Synthesis of Trichodermin Derivatives and Their Antimicrobial and Cytotoxic Activities
Abstract
1. Introduction
2. Results and Discussion
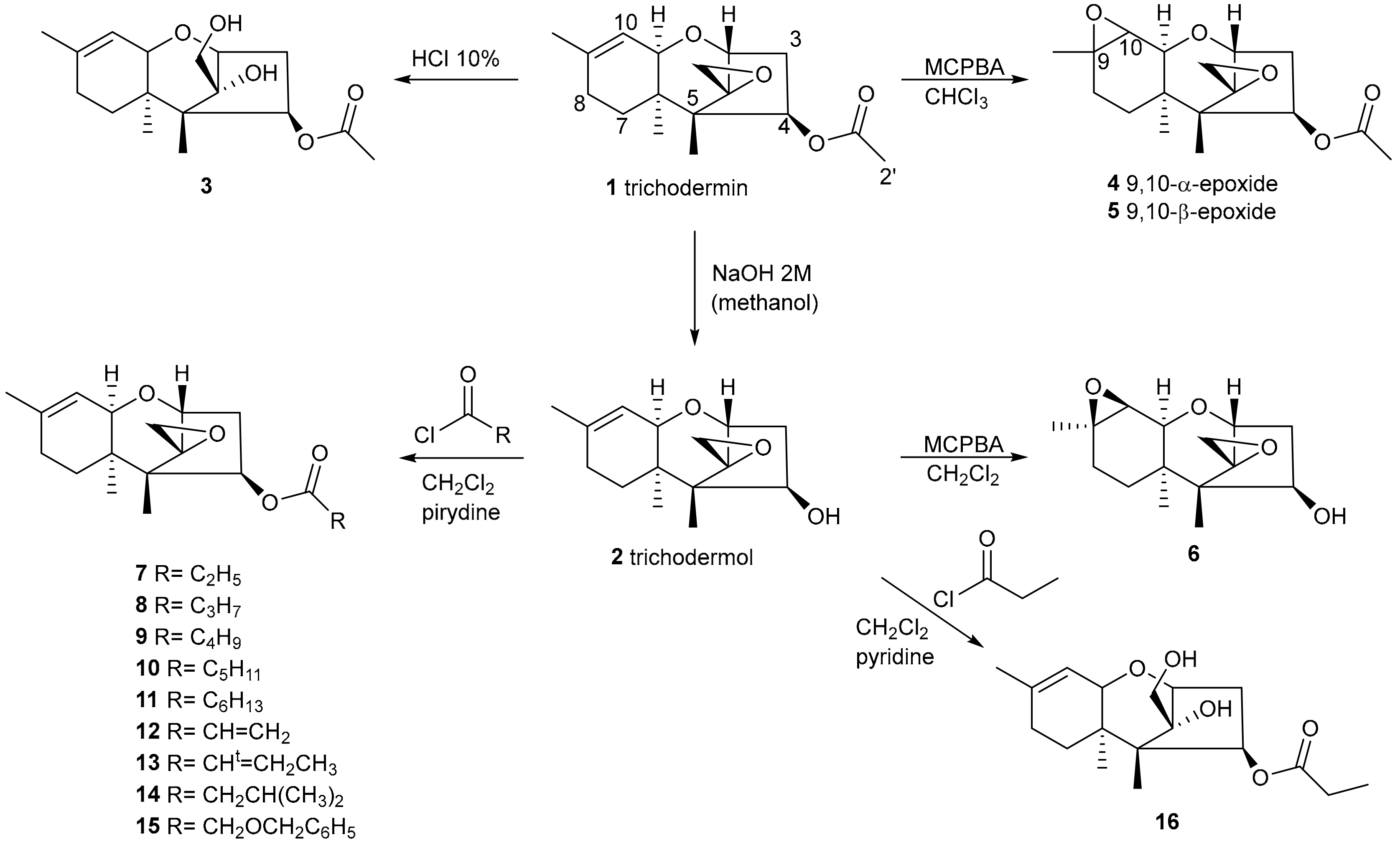
2.1. Synthesis of Trichodermin Derivatives
2.2. Evaluation of the Cytotoxic and Antimicrobial Activity of Trichodermin (1) Derivatives

3. Experimental Section
3.1. General
3.2. Fungal Strain and Culture Conditions
3.3. Microorganism Used in the Bioassays
3.4. Chemical Procedures
3.4.1. Trichodermin (1) Extraction
3.4.2. Preparation of Compound 3
3.4.3. General Procedure for the Preparation of Compounds 4–5 [21]
3.4.4. Preparation of Trichodermol (2)
3.4.5. Preparation of Compound 6
3.4.6. General Procedure for the Preparation of Trichodermol (2) C-4 derivatives (7–15)
3.4.7. Preparation of Compound 16
3.5. Evaluation of Antimicrobial Activity
3.5.1. Antifungal Assay
3.5.2. Antibacterial Test
3.5.3. MTT Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lachance, H.; Wetzel, S.; Kumar, K.; Waldmann, H. Charting, navigating, and populating natural product chemical space for drug discovery. J. Med. Chem. 2012, 55, 5989–6001. [Google Scholar] [CrossRef] [PubMed]
- Gerry, C.J.; Schreiber, S.L. Chemical probes and drug leads from advances in synthetic planning and methodology. Nat. Rev. Drug. Discov. 2018, 17, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Firn, R. Nature’s Chemicals. The Natural Products That Shaped Our World; Oxford University Press: Oxford, UK, 2010; Volume 106. [Google Scholar]
- Tijerino, A.; Cardoza, R.E.; Moraga, J.; Malmierca, M.G.; Vicente, F.; Aleu, J.; Collado, I.G.; Gutiérrez, S.; Monte, E.; Hermosa, R. Overexpression of the trichodiene synthase gene tri5 increases trichodermin production and antimicrobial activity in Trichoderma brevicompactum. Fungal Genet. Biol. 2011, 48, 285–296. [Google Scholar] [CrossRef]
- Dewick, P.M. Medicinal Natural Products: A Biosynthetic Approach; John Wiley & Sons, Ltd.: West Sussex, NJ, USA, 2009; Volume 84. [Google Scholar]
- Beremand, M.N.; Mc Cormick, S.P. Biosynthesis and regulation of trichothecene production by Fusarium species. In Handbook of Applied Mycology. Mycotoxins in Ecological Systems; Bhatnagar, D., Lilehoj, E.B., Arora, D.K., Eds.; Marcel Dekker, Inc.: New York, USA, 1992; Volume 5, pp. 360–389. [Google Scholar]
- Kimura, M.; Tokai, T.; Takahashi-Ando, N.; Ohsato, S.; Fujimura, M. Molecular and Genetic Studies of Fusarium Trichothecene Biosynthesis: Pathways, Genes, and Evolution. Biosci. Biotechnol. Biochem. 2007, 71, 2105–2123. [Google Scholar] [CrossRef]
- Reino, J.L.; Guerrero, R.F.; Hernández-Galán, R.; Collado, I.G. Secondary metabolites from species of the biocontrol agent Trichoderma. Phytochem. Rev. 2008, 7, 89–123. [Google Scholar] [CrossRef]
- Rocha, O.; Ansari, K.; Doohan, F.M. Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit. Contam. 2005, 22, 369–378. [Google Scholar] [CrossRef]
- Corley, D.G.; Miller-Wideman, M.; Durley, R.C. Isolation and structure of harzianum a: A new trichothecene from Trichoderma harzianum. J. Nat. Prod. 1994, 57, 422–425. [Google Scholar] [CrossRef]
- Pestka, J.J. Deoxynivalenol: Toxicity, mechanisms and animal health risks. Anim. Feed Sci. Technol. 2007, 137, 283–298. [Google Scholar] [CrossRef]
- Martin, L.J.; Doebler, J.A.; Anthony, A. Scanning cytophotometric analysis of brain neuronal nuclear chromatin changes in acute T-2 toxin-treated rats. Toxicol. Appl. Pharmacol. 1986, 85, 207–214. [Google Scholar] [CrossRef]
- Brown, D.W.; Dyer, R.B.; McCormick, S.P.; Kendra, D.F.; Plattner, R.D. Functional demarcation of the Fusarium core trichothecene gene cluster. Fungal. Genet. Biol. 2004, 41, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Cardoza, R.E.; Malmierca, M.G.; Hermosa, M.R.; Alexander, N.J.; McCormick, S.P.; Proctor, R.H.; Tijerino, A.M.; Rumbero, A.; Monte, E.; Gutiérrez, S. Identification of loci and functional characterization of trichothecene biosynthesis genes in filamentous fungi of the genus Trichoderma. Appl. Environ. Microbiol. 2011, 77, 4867–4877. [Google Scholar] [CrossRef] [PubMed]
- Proctor, R.H.; McCormick, S.P.; Kim, H.-S.; Cardoza, R.E.; Stanley, A.M.; Lindo, L.; Kelly, A.; Brown, D.W.; Lee, T.; Vaughan, M.M.; et al. Evolution of structural diversity of trichothecenes, a family of toxins produced by plant pathogenic and entomopathogenic fungi. PLoS Pathog. 2018, 14, e1006946. [Google Scholar]
- Nielsen, K.F.; Gräfenhan, T.; Zafari, D.; Thrane, U. Trichothecene production by Trichoderma brevicompactum. J. Agric. Food Chem. 2005, 53, 8190–8196. [Google Scholar] [CrossRef] [PubMed]
- Godtfredsen, W.O.; Vangedal, S. Trichodermin, a New Sesquiterpene Antibiotic. Acta Chem. Scand. 1965, 19, 1088–1102. [Google Scholar] [CrossRef]
- Westerberg, U.; Bolcsfoldi, G.; Eliasson, E.V.A. Control of tranfer RNA synthesis in the presence of inhibitors of protein synthesis. Biochim. Biophys. Acta 1976, 447, 203–213. [Google Scholar] [CrossRef]
- Tijerino, A.; Hermosa, R.; Cardoza, R.E.; Moraga, J.; Malmierca, M.G.; Aleu, J.; Collado, I.G.; Monte, E.; Gutiérrez, S. Overexpression of the Trichoderma brevicompactum tri5 gene: Effect on the expression of the trichodermin biosynthetic genes and on tomato seedlings. Toxins (Basel) 2011, 3, 1220. [Google Scholar] [CrossRef]
- Cheng, J.L.; Zhou, Y.; Zhao, J.H.; Zhang, C.; Lin, F.C. Synthesis and antifungal activity of trichodermin derivatives. Chinese Chem. Lett. 2010, 21, 1037–1040. [Google Scholar] [CrossRef]
- Xu, X.; Cheng, J.; Zhou, Y.; Zhang, C.; Ou, X.; Su, W.; Zhao, J.; Zhu, G. Synthesis and antifungal activities of trichodermin derivatives as fungicides on rice. Chem. Biodivers. 2013, 10, 600–611. [Google Scholar] [CrossRef]
- Cheng, J.L.; Zheng, M.; Yao, T.T.; Li, X.L.; Zhao, J.H.; Xia, M.; Zhu, G.N. Synthesis, antifungal activity, and QSAR study of novel trichodermin derivatives. J. Asian Nat.Prod. Res. 2015, 17, 47–55. [Google Scholar] [CrossRef]
- Colvin, E.W.; Malchenko, S.; Raphael, R.A.; Roberts, J.S. Total Synthesis of (±)-Trichodermin. J. Chem. Soc. Perkin Trans. 1973, 17, 1989–1997. [Google Scholar] [CrossRef]
- Still, W.C.; Tsai, M.-Y. Total synthesis of (±)-Trichodermol. J. Am. Chem. Soc. 1980, 102, 3654–3655. [Google Scholar] [CrossRef]
- O’Brien, M.K.; Pearson, A.J.; Pinkerton, A.A.; Schmidt, W.; Willman, K. A total synthesis of (±)-Trichodermol. J. Am. Chem. Soc. 1989, 111, 1499–1501. [Google Scholar] [CrossRef]
- Ayer, W.A.; Miao, S. Secondary metabolites of the aspen fungus Stachybotrys cylindrospora. Can. J. Chem. 1993, 71, 487–493. [Google Scholar] [CrossRef]
- Wei, C.M.; McLaughlin, C.S. Structure-function relationship in the 12, 13-epoxytrichothecenes: Novel inhibitors of protein synthesis. Biochem. Biophys. Res. Commun. 1974, 57, 838–844. [Google Scholar] [CrossRef]
- Carrasco, L.; Barbacid, M.; Vazquez, D. The trichodermin group of antibiotics, inhibitors of peptide bond formation by eukaryotic ribosomes. Biochim. Biophys. Acta 1973, 312, 368–376. [Google Scholar] [CrossRef]
- Plattner, R.D.; Al-Hetti, M.B.; Weisleder, D.; Sinclair, J.B. A new trichothecene from Trichothecium roseum. J. Chem. Res. 1988, 311, 2461–2473. [Google Scholar]
- Langley, P.; Shuttleworth, A.; Sidebottom, P.J.; Wrigley, S.K.; Fisher, P.J. A trichothecene from Spicellum roseum. Mycol. Res. 1990, 94, 705–720. [Google Scholar] [CrossRef]
- Tanaka, K.; Plattner, R.D.; Yamagishi, R.; Minamisawa, M.; Manabe, M.; Kawasugi, S.; Gareis, M.; Okada, G. 8-Deoxy-trichothecin production by Spicellum roseum isolated from a cultivated mushroom in Japan. Mycotoxins. 2001, 51, 71–77. [Google Scholar] [CrossRef]
- Kralj, A.; Gurgui, M.; König, G.M.; van Echten-Deckert, G. Trichothecenes induce accumulation of glucosylceramide in neural cells by interfering with lactosylceramide synthase activity. Toxicol. Appl. Pharmacol. 2007, 225, 113–122. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, L.; Li, G.-T.; Li, Z.H.; Dong, Z.J.; Li, Y.; Liu, J.K. Identification and Cytotoxic Activities of Two New Trichothecenes and a New Cuparane-Type Sesquiterpenoid from the Cultures of the Mushroom Engleromyces goetzii. Nat. Products Bioprospect. 2015, 5, 47–53. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 1 and 2 are available from the authors. |
| Entry | E. coli MB5746 | MRSA MB5393 | C. albicans MY1055 |
|---|---|---|---|
| 1 | >64 | >64 | <4 |
| 2 | >64 | >64 | 8–16 |
| 7 | >64 | >64 | 16–32 |
| 9 | 64 | >64 | 4–8 |
| 10 | 32 | >64 | 8 |
| 11 | 32 | >64 | >64 |
| 12 | >64 | >64 | 4 |
| 13 | >64 | >64 | 4 |
| 14 | >64 | >64 | 8–16 |
| 15 | >64 | >64 | 8 |
| Entry | HepG2 | MCF-7 | RCC4-VA | RCC4-VHL | Fa2N4 |
|---|---|---|---|---|---|
| 1 | >20 | <1.25 | 14.19 | >20 | 12.09 |
| 2 | >20 | 4.42 | >20 | >20 | >20 |
| 7 | >20 | 13.52 | >20 | >20 | >20 |
| 9 | >20 | 1.93 | >20 | >20 | >20 |
| 10 | 14.20 | 3.59 | >20 | >20 | >20 |
| 11 | >20 | 11.26 | >20 | >20 | >20 |
| 12 | >20 | 3.78 | 6.02 | 8.00 | >20 |
| 13 | 17.48 | 2.15 | >20 | >20 | >20 |
| 14 | >20 | 5.22 | >20 | >20 | >20 |
| 15 | >20 | 2.03 | 17.14 | >20 | >20 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barúa, J.E.; de la Cruz, M.; de Pedro, N.; Cautain, B.; Hermosa, R.; Cardoza, R.E.; Gutiérrez, S.; Monte, E.; Vicente, F.; Collado, I.G. Synthesis of Trichodermin Derivatives and Their Antimicrobial and Cytotoxic Activities. Molecules 2019, 24, 3811. https://doi.org/10.3390/molecules24203811
Barúa JE, de la Cruz M, de Pedro N, Cautain B, Hermosa R, Cardoza RE, Gutiérrez S, Monte E, Vicente F, Collado IG. Synthesis of Trichodermin Derivatives and Their Antimicrobial and Cytotoxic Activities. Molecules. 2019; 24(20):3811. https://doi.org/10.3390/molecules24203811
Chicago/Turabian StyleBarúa, Javier E., Mercedes de la Cruz, Nuria de Pedro, Bastien Cautain, Rosa Hermosa, Rosa E. Cardoza, Santiago Gutiérrez, Enrique Monte, Francisca Vicente, and Isidro G. Collado. 2019. "Synthesis of Trichodermin Derivatives and Their Antimicrobial and Cytotoxic Activities" Molecules 24, no. 20: 3811. https://doi.org/10.3390/molecules24203811
APA StyleBarúa, J. E., de la Cruz, M., de Pedro, N., Cautain, B., Hermosa, R., Cardoza, R. E., Gutiérrez, S., Monte, E., Vicente, F., & Collado, I. G. (2019). Synthesis of Trichodermin Derivatives and Their Antimicrobial and Cytotoxic Activities. Molecules, 24(20), 3811. https://doi.org/10.3390/molecules24203811










