Essential Oils and Their Natural Active Compounds Presenting Antifungal Properties
Abstract
1. Introduction
1.1. Public Health Problem, Invasive Fungal Infections (IFIs)
1.2. Crop Disease and Fungal Infections
1.3. Current Antifungal Treatments and Therapeutic Failures
- Polyenes, such as amphotericin B, target the plasma membrane, and in particular, ergosterol, a major component of the fungal membrane that increases membrane fluidity and causes cell death.
- Echinocandins, such as caspofungin, target β (1–3) glucan in the fungal cell wall.
- Azole derivatives, such as fluconazole or voriconazole, act on the biosynthetic pathway of ergosterol, increasing membrane fluidity and accumulation of a toxic sterol.
- Allylamines, such as terbinafine, also target the plasma membrane, and in particular, the first steps of ergosterol synthesis.
- Pyrimidines, such as 5-fluorocytosine, act on the nucleus, and in particular, on DNA synthesis.
2. The Main Essential Oils with Antifungal Activity
2.1. Lamacieae Family
2.2. Myrtaceae Family
2.3. Geraniaceae, Lauraceae and Apiaceae Family
3. Natural Compounds and Mechanism of Action
3.1. Thymol
3.2. Carvacrol
3.3. Geraniol
3.4. Cinnamaldehyde
4. Synergetic Effects of Essential Oils
- the inhibition of different stages in the fungal intracellular pathways that are essential for cell survival,
- the action of another antifungal agent on the fungal cell membrane,
- the inhibition of carrier proteins,
- the simultaneous inhibition of different cell targets [77].
4.1. Thymol
4.2. Carvacrol
4.3. Geraniol
4.4. Cinnamaldehyde
5. Toxicity and Side Effects
6. Resistance
7. Example of Patents of Natural Compounds with Antifungal Properties
8. Conclusions
Funding
Conflicts of Interest
References
- Bitar, D.; Lortholary, O.; Le Strat, Y.; Nicolau, J.; Coignard, B.; Tattevin, P.; Che, D.; Dromer, F. Population-Based Analysis of Invasive Fungal Infections, France, 2001–2010. Emerg. Infect. Dis. 2014, 20, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.; Ferreira, S.; Duarte, A.; Mendonça, D.I.; Domingues, F.C. Antifungal activity of Coriandrum sativum essential oil, its mode of action against Candida species and potential synergism with amphotericin B. Phytomedicine 2011, 19, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.D.; Denning, D.W.; Gow, N.A.R.; Levitz, S.M.; Netea, M.G.; White, T.C. Hidden killers: Human fungal infections. Sci. Transl. Med. 2012, 4, 165rv13. [Google Scholar] [CrossRef] [PubMed]
- Blot, M.; Lanternier, F.; Lortholary, O. Epidemiology of Visceral Fungal Infection In France And In The World. Rev. Prat. 2015, 65, 1318–1321. [Google Scholar]
- Parkin, D.M.; Bray, F.; Ferlay, J.; Pisani, P. Global cancer statistics, 2002. CA Cancer J. Clin. 2005, 55, 74–108. [Google Scholar] [CrossRef]
- Rapp, R.P. Changing strategies for the management of invasive fungal infections. Pharmacotherapy 2004, 24, 4S–28S. [Google Scholar] [CrossRef]
- Enoch, D.A.; Yang, H.; Aliyu, S.H.; Micallef, C. The Changing Epidemiology of Invasive Fungal Infections. Methods Mol. Biol. 2017, 1508, 17–65. [Google Scholar]
- Savary, S.; Ficke, A.; Aubertot, J.-N.; Hollier, C. Crop losses due to diseases and their implications for global food production losses and food security. Food Sec. 2012, 4, 519–537. [Google Scholar] [CrossRef]
- Oerke, E.-C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Plesken, C.; Weber, R.W.S.; Rupp, S.; Leroch, M.; Hahn, M. Botrytis pseudocinerea Is a Significant Pathogen of Several Crop Plants but Susceptible to Displacement by Fungicide-Resistant B. cinerea Strains. Appl. Environ. Microbiol. 2015, 81, 7048–7056. [Google Scholar] [CrossRef]
- Nganje, W.E.; Bangsund, D.A.; Leistritz, F.L.; Wilson, W.W.; Tiapo, N.M. Estimating The Economic Impact Of A Crop Disease: The Case Of Fusarium Head Blight In U.S. Wheat Barley 2002, 275–281. [Google Scholar]
- Abuhammour, W.; Habte-Gaber, E. Newer antifungal agents. Indian J. Pediatr 2004, 71, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, C. Antifongiques. Available online: https://www.em-consulte.com/article/946740/antifongiques (accessed on 19 December 2018).
- De Pascale, G.; Tumbarello, M. Fungal infections in the ICU: Advances in treatment and diagnosis. Curr. Opin. Crit. Care 2015, 21, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Robbins, N.; Wright, G.D.; Cowen, L.E. Antifungal Drugs: The Current Armamentarium and Development of New Agents. Microbiol. Spectr. 2016, 4. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Divband, K.; Shokri, H.; Khosravi, A.R. Down-regulatory effect of Thymus vulgaris L. on growth and Tri4 gene expression in Fusarium oxysporum strains. Microb. Pathog. 2017, 104, 1–5. [Google Scholar] [CrossRef]
- De Groot, A.C.; Schmidt, E. Essential Oils, Part I: Introduction. Dermatitis 2016, 27, 39–42. [Google Scholar] [CrossRef]
- Rajkowska, K.; Otlewska, A.; Kunicka-Styczyńska, A.; Krajewska, A. Candida albicans Impairments Induced by Peppermint and Clove Oils at Sub-Inhibitory Concentrations. Int. J. Mol. Sci. 2017, 18, 1307. [Google Scholar] [CrossRef]
- Pinto, E.; Pina-Vaz, C.; Salgueiro, L.; Gonçalves, M.J.; Costa-de-Oliveira, S.; Cavaleiro, C.; Palmeira, A.; Rodrigues, A.; Martinez-de-Oliveira, J. Antifungal activity of the essential oil of Thymus pulegioides on Candida, Aspergillus and dermatophyte species. J. Med. Microbiol. 2006, 55, 1367–1373. [Google Scholar] [CrossRef]
- Daferera, D.J.; Ziogas, B.N.; Polissiou, M.G. The effectiveness of plant essential oils on the growth of Botrytis cinerea, Fusarium sp. and Clavibacter michiganensis subsp. michiganensis. Crop Prot. 2003, 22, 39–44. [Google Scholar] [CrossRef]
- Segvić Klarić, M.; Kosalec, I.; Mastelić, J.; Piecková, E.; Pepeljnak, S. Antifungal activity of thyme (Thymus vulgaris L.) essential oil and thymol against moulds from damp dwellings. Lett. Appl. Microbiol. 2007, 44, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Banani, H.; Olivieri, L.; Santoro, K.; Garibaldi, A.; Gullino, M.L.; Spadaro, D. Thyme and Savory Essential Oil Efficacy and Induction of Resistance against Botrytis cinerea through Priming of Defense Responses in Apple. Foods 2018, 7, 11. [Google Scholar] [CrossRef] [PubMed]
- Diánez, F.; Santos, M.; Parra, C.; Navarro, M.J.; Blanco, R.; Gea, F.J. Screening of antifungal activity of 12 essential oils against eight pathogenic fungi of vegetables and mushroom. Lett. Appl. Microbiol. 2018, 67, 400–410. [Google Scholar] [CrossRef] [PubMed]
- Gill, T.A.; Li, J.; Saenger, M.; Scofield, S.R. Thymol-based submicron emulsions exhibit antifungal activity against Fusarium graminearum and inhibit Fusarium head blight in wheat. J. Appl. Microbiol. 2016, 121, 1103–1116. [Google Scholar] [CrossRef] [PubMed]
- Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Vazquez-Olivo, G.; Heredia, J.B. Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules 2017, 22, 989. [Google Scholar] [CrossRef] [PubMed]
- Santoyo, S.; Cavero, S.; Jaime, L.; Ibañez, E.; Señoráns, F.J.; Reglero, G. Supercritical carbon dioxide extraction of compounds with antimicrobial activity from Origanum vulgare L.: Determination of optimal extraction parameters. J. Food Prot. 2006, 69, 369–375. [Google Scholar] [CrossRef]
- Khosravi, A.R.; Shokri, H.; Kermani, S.; Dakhili, M.; Madani, M.; Parsa, S. Antifungal properties of Artemisia sieberi and Origanum vulgare essential oils against Candida glabrata isolates obtained from patients with vulvovaginal candidiasis. J. Mycol. Médicale 2011, 21, 93–99. [Google Scholar] [CrossRef]
- Mahboubi, M.; Haghi, G. Antimicrobial activity and chemical composition of Mentha pulegium L. essential oil. J. Ethnopharmacol. 2008, 119, 325–327. [Google Scholar] [CrossRef]
- Stringaro, A.; Colone, M.; Angiolella, L. Antioxidant, Antifungal, Antibiofilm, and Cytotoxic Activities of Mentha spp. Essential Oils. Medicines 2018, 5, 112. [Google Scholar] [CrossRef]
- De Groot, A.; Schmidt, E. Essential Oils, Part V: Peppermint Oil, Lavender Oil, and Lemongrass Oil. Dermatitis 2016, 27, 325–332. [Google Scholar] [CrossRef]
- Tampieri, M.P.; Galuppi, R.; Macchioni, F.; Carelle, M.S.; Falcioni, L.; Cioni, P.L.; Morelli, I. The inhibition of Candida albicans by selected essential oils and their major components. Mycopathologia 2005, 159, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Behmanesh, F.; Pasha, H.; Sefidgar, A.A.; Taghizadeh, M.; Moghadamnia, A.A.; Adib Rad, H.; Shirkhani, L. Antifungal Effect of Lavender Essential Oil (Lavandula angustifolia) and Clotrimazole on Candida albicans: An In Vitro Study. Scientifica (Cairo) 2015, 2015, 261397. [Google Scholar] [PubMed]
- D’Auria, F.D.; Tecca, M.; Strippoli, V.; Salvatore, G.; Battinelli, L.; Mazzanti, G. Antifungal activity of Lavandula angustifolia essential oil against Candida albicans yeast and mycelial form. Med. Mycol. 2005, 43, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.; Yousef, M.; Tsiani, E. Anticancer Effects of Rosemary (Rosmarinus officinalis L.) Extract and Rosemary Extract Polyphenols. Nutrients 2016, 8, 731. [Google Scholar] [CrossRef] [PubMed]
- Ksouri, S.; Djebir, S.; Bentorki, A.A.; Gouri, A.; Hadef, Y.; Benakhla, A. Antifungal activity of essential oils extract from Origanum floribundum Munby, Rosmarinus officinalis L. and Thymus ciliatus Desf. against Candida albicans isolated from bovine clinical mastitis. J. Mycol. Med. 2017, 27, 245–249. [Google Scholar] [CrossRef]
- De Oliveira, J.R.; de Jesus, D.; Figueira, L.W.; de Oliveira, F.E.; Pacheco Soares, C.; Camargo, S.E.A.; Jorge, A.O.C.; de Oliveira, L.D. Biological activities of Rosmarinus officinalis L. (rosemary) extract as analyzed in microorganisms and cells. Exp. Biol. Med. (Maywood) 2017, 242, 625–634. [Google Scholar] [CrossRef]
- Golab, M.; Skwarlo-Sonta, K. Mechanisms involved in the anti-inflammatory action of inhaled tea tree oil in mice. Exp. Biol. Med. (Maywood) 2007, 232, 420–426. [Google Scholar]
- Carson, C.F.; Hammer, K.; Riley, T. Melaleuca alternifolia (Tea Tree) Oil: A Review of Antimicrobial and Other Medicinal Properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar] [CrossRef]
- De Campos Rasteiro, V.M.; da Costa, A.C.B.P.; Araújo, C.F.; de Barros, P.P.; Rossoni, R.D.; Anbinder, A.L.; Jorge, A.O.C.; Junqueira, J.C. Essential oil of Melaleuca alternifolia for the treatment of oral candidiasis induced in an immunosuppressed mouse model. BMC Complement. Altern. Med. 2014, 14, 489. [Google Scholar] [CrossRef]
- Dhakad, A.K.; Pandey, V.V.; Beg, S.; Rawat, J.M.; Singh, A. Biological, medicinal and toxicological significance of Eucalyptus leaf essential oil: a review. J. Sci. Food Agric. 2018, 98, 833–848. [Google Scholar] [CrossRef]
- Dutta, B.K.; Karmakar, S.; Naglot, A.; Aich, J.C.; Begam, M. Anticandidial activity of some essential oils of a mega biodiversity hotspot in India. Mycoses 2007, 50, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Elansary, H.O.; Salem, M.Z.M.; Ashmawy, N.A.; Yessoufou, K.; El-Settawy, A.A.A. In vitro antibacterial, antifungal and antioxidant activities of Eucalyptus spp. leaf extracts related to phenolic composition. Nat. Prod. Res. 2017, 31, 2927–2930. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Meng, X.; Li, Y.; Zhao, C.-N.; Tang, G.-Y.; Li, H.-B. Antibacterial and Antifungal Activities of Spices. Int. J. Mol. Sci. 2017, 18, 1283. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, E.; Jirovetz, L.; Wlcek, K.; Buchbauer, G.; Gochev, V.; Girova, T.; Stoyanova, A.; Geissler, M. Antifungal Activity of Eugenol and Various Eugenol-Containing Essential Oils against 38 Clinical Isolates of Candida albicans. J. Essent. Oil Bear. Plants 2007, 10, 421–429. [Google Scholar] [CrossRef]
- Essid, R.; Hammami, M.; Gharbi, D.; Karkouch, I.; Hamouda, T.B.; Elkahoui, S.; Limam, F.; Tabbene, O. Antifungal mechanism of the combination of Cinnamomum verum and Pelargonium graveolens essential oils with fluconazole against pathogenic Candida strains. Appl. Microbiol. Biotechnol. 2017, 101, 6993–7006. [Google Scholar] [CrossRef]
- Ennaifer, M.; Bouzaiene, T.; Chouaibi, M.; Hamdi, M. Pelargonium graveolens Aqueous Decoction: A New Water-Soluble Polysaccharide and Antioxidant-Rich Extract. Biomed Res. Int. 2018, 2018, 2691513. [Google Scholar] [CrossRef]
- Gucwa, K.; Milewski, S.; Dymerski, T.; Szweda, P. Investigation of the Antifungal Activity and Mode of Action of Thymus vulgaris, Citrus limonum, Pelargonium graveolens, Cinnamomum cassia, Ocimum basilicum, and Eugenia caryophyllus Essential Oils. Molecules 2018, 23, 1116. [Google Scholar] [CrossRef]
- Chinh, H.V.; Luong, N.X.; Thin, D.B.; Dai, D.N.; Hoi, T.M.; Ogunwande, I.A. Essential Oils Leaf of Cinnamomum glaucescens and Cinnamomum verum from Vietnam. Am. J. Plant Sci. 2017, 8, 2712. [Google Scholar] [CrossRef][Green Version]
- Homa, M.; Fekete, I.P.; Böszörményi, A.; Singh, Y.R.B.; Selvam, K.P.; Shobana, C.S.; Manikandan, P.; Kredics, L.; Vágvölgyi, C.; Galgóczy, L. Antifungal Effect of Essential Oils against Fusarium Keratitis Isolates. Planta Med. 2015, 81, 1277–1284. [Google Scholar] [CrossRef]
- Manso, S.; Nerín, C.; Gómez-Lus, R. Antifungal activity of the essential oil of cinnamon (cinnamomum zeylanicum), oregano (origanum vulgare) and lauramide argine ethyl ester (LAE) against the mold aspergillus flavus CECT 2949. Ital. J. Food Sci. 2011, 23, 151–156. [Google Scholar]
- Manso, S.; Pezo, D.; Gómez-Lus, R.; Nerín, C. Diminution of aflatoxin B1 production caused by an active packaging containing cinnamon essential oil. Food Control 2014, 45, 101–108. [Google Scholar] [CrossRef]
- Reza, Z.M.; Atefeh, J.Y.; Faezeh, F. Effect of γ-Irradiation on the Antibacterial Activities of Cuminum cyminum L. Essential Oils In Vitro and In Vivo Systems. J. Essent. Oil Bear. Plants 2015, 18, 582–591. [Google Scholar] [CrossRef]
- Kedia, A.; Prakash, B.; Mishra, P.K.; Dubey, N.K. Antifungal and antiaflatoxigenic properties of Cuminum cyminum (L.) seed essential oil and its efficacy as a preservative in stored commodities. Int. J. Food Microbiol. 2014, 168, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Minooeianhaghighi, M.H.; Sepehrian, L.; Shokri, H. Antifungal effects of Lavandula binaludensis and Cuminum cyminum essential oils against Candida albicans strains isolated from patients with recurrent vulvovaginal candidiasis. J. Mycol. Med. 2017, 27, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.S.; Joshi, N.; Padalia, R.C.; Singh, V.R.; Goswami, P.; Kumar, A.; Iqbal, H.; Verma, R.K.; Chanda, D.; Chauhan, A.; et al. Chemical Composition and Allelopathic, Antibacterial, Antifungal, and Antiacetylcholinesterase Activity of Fish-mint (Houttuynia cordataThunb.) from India. Chem. Biodivers. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines (Basel) 2016, 3, 25. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; Coppola, R.; Feo, V.D. Essential Oils and Antifungal Activity. Pharmaceuticals (Basel) 2017, 10, 86. [Google Scholar] [CrossRef]
- De Oliveira Lima, M.I.; Araújo de Medeiros, A.C.; Souza Silva, K.V.; Cardoso, G.N.; de Oliveira Lima, E.; de Oliveira Pereira, F. Investigation of the antifungal potential of linalool against clinical isolates of fluconazole resistant Trichophyton rubrum. J. Mycol. Med. 2017, 27, 195–202. [Google Scholar] [CrossRef]
- Shreaz, S.; Shiekh, R.A.; Raja, V.; Wani, W.A.; Behbehani, J.M. Impaired ergosterol biosynthesis mediated fungicidal activity of Co(II) complex with ligand derived from cinnamaldehyde. Chem. Biol. Interact. 2016, 247, 64–74. [Google Scholar] [CrossRef]
- Kurita, N.; Koike, S. Synergistic Antimicrobial Effect of Ethanol, Sodium Chloride, Acetic Acid and Essential Oil Components. Agric. Biol. Chem. 1983, 47, 67–75. [Google Scholar]
- Marchese, A.; Orhan, I.E.; Daglia, M.; Barbieri, R.; Di Lorenzo, A.; Nabavi, S.F.; Gortzi, O.; Izadi, M.; Nabavi, S.M. Antibacterial and antifungal activities of thymol: A brief review of the literature. Food Chem. 2016, 210, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Gao, T.; Zhou, H.; Zhou, W.; Hu, L.; Chen, J.; Shi, Z. The Fungicidal Activity of Thymol against Fusarium graminearum via Inducing Lipid Peroxidation and Disrupting Ergosterol Biosynthesis. Molecules 2016, 21, 770. [Google Scholar] [CrossRef] [PubMed]
- De Castro, R.D.; de Souza, T.M.P.A.; Bezerra, L.M.D.; Ferreira, G.L.S.; Costa, E.M.M.; Cavalcanti, A.L. Antifungal activity and mode of action of thymol and its synergism with nystatin against Candida species involved with infections in the oral cavity: An in vitro study. BMC Complement. Altern. Med. 2015, 15, 417. [Google Scholar] [CrossRef] [PubMed]
- Darvishi, E.; Omidi, M.; Bushehri, A.A.; Golshani, A.; Smith, M.L. Thymol antifungal mode of action involves telomerase inhibition. Med. Mycol. 2013, 51, 826–834. [Google Scholar] [CrossRef]
- Zhou, D.; Wang, Z.; Li, M.; Xing, M.; Xian, T.; Tu, K. Carvacrol and eugenol effectively inhibit Rhizopus stolonifer and control postharvest soft rot decay in peaches. J. Appl. Microbiol. 2018, 124, 166–178. [Google Scholar] [CrossRef]
- Ahmad, A.; van Vuuren, S.; Viljoen, A. Unravelling the complex antimicrobial interactions of essential oils--the case of Thymus vulgaris (thyme). Molecules 2014, 19, 2896–2910. [Google Scholar] [CrossRef]
- Chaillot, J.; Tebbji, F.; Remmal, A.; Boone, C.; Brown, G.W.; Bellaoui, M.; Sellam, A. The Monoterpene Carvacrol Generates Endoplasmic Reticulum Stress in the Pathogenic Fungus Candida albicans. Antimicrob. Agents Chemother. 2015, 59, 4584–4592. [Google Scholar] [CrossRef]
- Lv, Y.; Zhang, L.; Li, N.; Mai, N.; Zhang, Y.; Pan, S. Geraniol promotes functional recovery and attenuates neuropathic pain in rats with spinal cord injury. Can. J. Physiol. Pharmacol. 2017, 95, 1389–1395. [Google Scholar] [CrossRef]
- Leite, M.C.A.; de Brito Bezerra, A.P.; de Sousa, J.P.; de Oliveira Lima, E. Investigating the antifungal activity and mechanism(s) of geraniol against Candida albicans strains. Med. Mycol. 2015, 53, 275–284. [Google Scholar] [CrossRef]
- Singh, S.; Fatima, Z.; Hameed, S. Insights into the mode of action of anticandidal herbal monoterpenoid geraniol reveal disruption of multiple MDR mechanisms and virulence attributes in Candida albicans. Arch. Microbiol. 2016, 198, 459–472. [Google Scholar] [CrossRef]
- Khan, S.N.; Khan, S.; Iqbal, J.; Khan, R.; Khan, A.U. Enhanced Killing and Antibiofilm Activity of Encapsulated Cinnamaldehyde against Candida albicans. Front Microbiol 2017, 8, 1641. [Google Scholar] [CrossRef] [PubMed]
- Pootong, A.; Norrapong, B.; Cowawintaweewat, S. Antifungal Activity of Cinnamaldehyde Against Candida Albicans. Southeast Asian J. Trop. Med. Public Health 2017, 48, 150–158. [Google Scholar] [PubMed]
- Sun, Q.; Shang, B.; Wang, L.; Lu, Z.; Liu, Y. Cinnamaldehyde inhibits fungal growth and aflatoxin B1 biosynthesis by modulating the oxidative stress response of Aspergillus flavus. Appl. Microbiol. Biotechnol. 2016, 100, 1355–1364. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Xing, F.; Selvaraj, J.N.; Liu, X.; Wang, L.; Hua, H.; Zhou, L.; Zhao, Y.; Wang, Y.; Liu, Y. Inhibitory Effect of Cinnamaldehyde, Citral, and Eugenol on Aflatoxin Biosynthetic Gene Expression and Aflatoxin B1 Biosynthesis in Aspergillus flavus. J. Food Sci. 2015, 80, M2917–M2924. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Ohashi, Y. Combination effect of antibiotics against bacteria isolated from keratitis using fractional inhibitory concentration index. Cornea 2013, 32, e156–e160. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.D.; MacDougall, C.; Ostrosky-Zeichner, L.; Perfect, J.R.; Rex, J.H. Combination antifungal therapy. Antimicrob. Agents Chemother. 2004, 48, 693–715. [Google Scholar] [CrossRef]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Khan, M.S.A.; Ahmad, I. Antifungal activity of essential oils and their synergy with fluconazole against drug-resistant strains of Aspergillus fumigatus and Trichophyton rubrum. Appl. Microbiol. Biotechnol. 2011, 90, 1083–1094. [Google Scholar] [CrossRef]
- Sharifzadeh, A.; Khosravi, A.R.; Shokri, H.; Shirzadi, H. Potential effect of 2-isopropyl-5-methylphenol (thymol) alone and in combination with fluconazole against clinical isolates of Candida albicans, C. glabrata and C. krusei. J. Mycol. Med. 2018, 28, 294–299. [Google Scholar] [CrossRef]
- Scalas, D.; Mandras, N.; Roana, J.; Tardugno, R.; Cuffini, A.M.; Ghisetti, V.; Benvenuti, S.; Tullio, V. Use of Pinus sylvestris L. (Pinaceae), Origanum vulgare L. (Lamiaceae), and Thymus vulgaris L. (Lamiaceae) essential oils and their main components to enhance itraconazole activity against azole susceptible/not-susceptible Cryptococcus neoformans strains. BMC Complement. Altern. Med. 2018, 18, 143. [Google Scholar] [CrossRef]
- Ahmad, A.; Khan, A.; Manzoor, N. Reversal of efflux mediated antifungal resistance underlies synergistic activity of two monoterpenes with fluconazole. Eur. J. Pharm. Sci. 2013, 48, 80–86. [Google Scholar] [CrossRef]
- Jesus, F.P.K.; Ferreiro, L.; Bizzi, K.S.; Loreto, É.S.; Pilotto, M.B.; Ludwig, A.; Alves, S.H.; Zanette, R.A.; Santurio, J.M. In vitro activity of carvacrol and thymol combined with antifungals or antibacterials against Pythium insidiosum. J. Mycol. Med. 2015, 25, e89–e93. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Fatima, Z.; Ahmad, K.; Hameed, S. Fungicidal action of geraniol against Candida albicans is potentiated by abrogated CaCdr1p drug efflux and fluconazole synergism. PLoS ONE 2018, 13, e0203079. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, N.N.R.; Alviano, C.S.; Blank, A.F.; Romanos, M.T.V.; Fonseca, B.B.; Rozental, S.; Rodrigues, I.A.; Alviano, D.S. Synergism Effect of the Essential Oil from Ocimum basilicum var. Maria Bonita and Its Major Components with Fluconazole and Its Influence on Ergosterol Biosynthesis. Evid Based Complement. Altern. Med. 2016, 2016, 5647182. [Google Scholar] [CrossRef] [PubMed]
- Giordani, R.; Regli, P.; Kaloustian, J.; Portugal, H. Potentiation of antifungal activity of amphotericin B by essential oil from Cinnamomum cassia. Phytother Res. 2006, 20, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Naganuma, M.; Hirose, S.; Nakayama, Y.; Nakajima, K.; Someya, T. A study of the phototoxicity of lemon oil. Arch. Dermatol. Res. 1985, 278, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Piątkowska, E.; Rusiecka-Ziółkowska, J. Influence of Essential Oils on Infectious Agents. Adv. Clin. Exp. Med. 2016, 25, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Shreaz, S.; Wani, W.A.; Behbehani, J.M.; Raja, V.; Irshad, M.; Karched, M.; Ali, I.; Siddiqi, W.A.; Hun, L.T. Cinnamaldehyde and its derivatives, a novel class of antifungal agents. Fitoterapia 2016, 112, 116–131. [Google Scholar] [CrossRef]
- McKay, D.L.; Blumberg, J.B. A review of the bioactivity and potential health benefits of peppermint tea (Mentha piperita L.). Phytother. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef]
- Prashar, A.; Locke, I.C.; Evans, C.S. Cytotoxicity of clove (Syzygium aromaticum) oil and its major components to human skin cells. Cell Prolif. 2006, 39, 241–248. [Google Scholar] [CrossRef]
- André, W.P.P.; Cavalcante, G.S.; Ribeiro, W.L.C.; Santos, J.M.L.D.; Macedo, I.T.F.; de Paula, H.C.B.; de Morais, S.M.; de Melo, J.V.; Bevilaqua, C.M.L. Anthelmintic effect of thymol and thymol acetate on sheep gastrointestinal nematodes and their toxicity in mice. Rev. Bras. Parasitol. Vet. 2017, 26, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, C.J.; Carson, C.F.; Chang, B.J.; Riley, T.V. Role of the MexAB-OprM efflux pump of Pseudomonas aeruginosa in tolerance to tea tree (Melaleuca alternifolia) oil and its monoterpene components terpinen-4-ol, 1,8-cineole, and alpha-terpineol. Appl. Environ. Microbiol. 2008, 74, 1932–1935. [Google Scholar] [CrossRef] [PubMed]
- Langeveld, W.T.; Veldhuizen, E.J.A.; Burt, S.A. Synergy between essential oil components and antibiotics: A review. Crit. Rev. Microbiol. 2014, 40, 76–94. [Google Scholar] [CrossRef] [PubMed]
- Becerril, R.; Nerín, C.; Gómez-Lus, R. Evaluation of bacterial resistance to essential oils and antibiotics after exposure to oregano and cinnamon essential oils. Foodborne Pathog. Dis. 2012, 9, 699–705. [Google Scholar] [CrossRef] [PubMed]
| IFI | Localization | Cases/Year | Mortality Rate |
|---|---|---|---|
| Invasive Aspergillosis | Worldwide | >200,000 | 30%–95% |
| Invasive Candidiasis | Worldwide | >400,000 | 46%–75% |
| Cryptococcosis | Worldwide | >1,000,000 | 20%–70% |
| Mucormycosis | Worldwide | >10,000 | 30%–90% |
| Essential Oil | Major Compounds | Pathogens Tested | MIC/Concentration Used in the Studies | Converted Values (µg/mL) | Number of Strains Tested | % of the Major Compound (When Presided) | References |
|---|---|---|---|---|---|---|---|
| Thymus vulgaris | Thymol Carvacrol p-Cymene | Candida albicans | 62 µg/mL | 1 | Thymol 60.8% Carvacrol 2.88% p-Cymene 15.4% | [67] | |
| Candida tropicalis | |||||||
| Fusarium sp | ED50 71 µg/mL | 1 | Thymol 0.2% Carvacrol 81.5% | [21] | |||
| Aspergillus sp | 9.85 µg/mL | 44 | Thymol 33% Carvacrol 3.9% | [22] | |||
| Penicillium sp | 19.17 µg/mL | 18 | |||||
| Cladosporum sp | 15.20 µg/mL | 6 | |||||
| Botrytis cinerea | - | 1 | - | [23] | |||
| Alternaria brassicae | ED50 67.7% v/v | ED50 677 µg/mL | 1 | - | [24] | ||
| Fusarium oxysporum | ED50 36.3% v/v | ED50 363 µg/mL | 1 | ||||
| Thymus pulegioides | Fusarium graminearum | 105–108 µg/mL | 1 | - | [25] | ||
| Aspergillus sp | 0.16–0.64 µL/mL | 160-–40 µg/mL | 9 | Thymol 26% Carvacrol 21% | [20] | ||
| Dermatophytes | 5 | ||||||
| Candida sp | 11 | ||||||
| Maleleuca alternifolia | Terpinen-4-ol | Aspergillus sp | 0.016%–0.12% v/v | 1.6–200 µg/mL | Terpinen-4-ol 40.1% | [39] | |
| Candida sp | 0.03%–8% v/v | 3–800 µg/mL | |||||
| Candida albicans | 1.95 mg/mL | 1 | - | [40] | |||
| Origanum vulgare | Thymol Carvacrol Sabinene Linalool | Botrytis cinerea | ED50 50 µg/mL | 1 | Thymol 63.7% Carvacrol 8.6% | [21] | |
| Fusarium sp | 1 | ||||||
| C. albicans | 1.48–1.75 mg/mL | 1 | Carvacrol 39.08%–49.03% Sabinene 1 9.81%–25.11% | [27] | |||
| A. niger | 2.75–2.85 mg/mL | 1 | |||||
| C. glabrata | 0.5–1100 µg/mL | 16 | Thymol 25.1% Linalool 42% | [28] | |||
| A. flavus | 400 ppm | 3.6 ug/mL | 1 | [51] | |||
| Mentha piperita | Linalool Menthol Piperitone | Candida albicans | 1 µL/mL | 1 mg/mL | 1 | Piperitone 38% Piperitenone 33% | [29] |
| Aspergillus niger | 0.25 µL/m | 250 mg/mL | 1 | ||||
| Candida sp | 800 µg/mL | - | [30] | ||||
| Aspergillus sp | 222 µg/mL | ||||||
| Mentha pulegium | Pulegone | Candida albicans | 500 ppm | 44.5 µg/mL | 1 | Menthol 37.88% | [32] |
| Candida sp | 400–7000 µg/mL | - | [30] | ||||
| Dermatophyte | 800–3500 µg/mL | ||||||
| Aspergillus sp | 400–3500 µg/mL | ||||||
| Lavendula angustifolia | Linalool Linalyl acetate | b.cinerea | ED50 223 µg/mL | 1 | Linalool 25.5% Linalyl acetate 17.7% | [21] | |
| Fusarium sp | 520 µg/mL | 1 | |||||
| F.oxysporum | ED50 372 µL/mL | 37.2 mg/mL | 1 | - | [24] | ||
| C. albicans | 1/40 of pure solution of essential oil | 20 | - | [33] | |||
| C. albicans | 5000 ppm | 445 µg/mL | 50 | Linalool 24.7% Linalyl acetate 31.1% | [34] | ||
| Rosmarinus officinialis | 1,8-Cineole Camphor α-pinene | B.cinierea | ED50 600 µg/mL | 1 | Eucalyptol 31.5% | [21] | |
| Fusarium sp | 660 µg/mL | 1 | |||||
| C. albicans | MIC 80% 24–31 µg/mL | 11 | 1,8-Cineole 31.5% | [36] | |||
| C. albicans | 0.78 mg/mL | 1 | 1,8-Cineole 52.2% Camphor 15.2% α-pinene 12.4% | [37] | |||
| Pelargonium graveolens | (Z)-geraniol Citronellol | C. albicans | 0.16% v/v | 1.6 mg/mL | 47 | Citronellol 11.94% | [48] |
| C. glabrata | 20 | ||||||
| C. albicans | 500–1000 µg/mL | 5 | Citronellol 27.23% | [46] | |||
| C. tropicalis | 250 µg/mL | 1 | |||||
| C. parasilopsisi | 500 µg/mL | 1 | |||||
| C. glabrata | 500 µg/mL | 2 | |||||
| C. riferi | 500 µg/mL | 1 | |||||
| Eucalyptus citriodora | Citronellol Citronellal | C. albicans | 318 µg/mL | 1 | - | [42] | |
| Eucalyptus camaldulensis | p-cymene 1,8-Cineole | P. funicuarum | 0.15 mg/mL | - | [43] | ||
| A. niger | 0.47 mg/mL | ||||||
| A. flavus | 0.43 mg/mL | ||||||
| Cinnamomum verum | Cinnamaldehyde | Fusarium | 31.25–500 µg/mL | 18 | Cinnamaldehyde 93.1% | [50] | |
| A. flavus | 100 ppm | 8.9 µg/mL | 1 | - | [52] | ||
| C. albicans | 31.25–62.5 µg/mL | 5 | Cinnamaldehyde 82.09% | [46] | |||
| C. parasilopsis | 1 | ||||||
| C. riferii | 1 | ||||||
| C. tropicalis | 1 | ||||||
| C. glabrata | 2 | ||||||
| Cuminum cyminum | Cuminaldehyde Cymene γ-terpinene 1,8-cineole | Fusarium sp | 0.6 µL/mL | 600 µg/mL | 1230 | Cymene 47.8% Cuminaldehyde 14.92% γ-terpinene 19.36% | [54] |
| Aspergillus sp | |||||||
| C. albicans | 3.90–11.71 µg/mL | 20 | 1,8-cineole 21.07% | [55] | |||
| Sysygium aromaticum | 1,8-cineole Eugenol | C. albicans | MIC 50% 6.2–7.5 µL/mL | 6.2–7.5 mg/mL | 38 | Eugenol 76.84% | [45] |
| A. brassicae | ED50 54% v/v | 540 µg/mL | 1 | Eugenol 86.38% | [24] | ||
| F.oxysporum | ED50 44.7% v/v | 447 µg/mL | 1 | ||||
| C. albicans | 125–250 µg/mL | 5 | Eugenol 90.43% | [46] | |||
| C. parapsilopsis | 1 | ||||||
| C. riferii | 1 | ||||||
| C. tropicalis | 1 | ||||||
| C. glabrata | 2 |
| Essential Oil | Pathogens Tested | MIC/Concentration Used in the Studies (Converted) | Atf Tested | MIC | References |
|---|---|---|---|---|---|
| Thymus vulgaris | Candida albicans | 62 µg/mL | Amphotericin B | 0.001 mg/mL | [67] |
| Candida tropicalis | Amphotericin B | 0.001 mg/mL | |||
| Thymus pulegioides | Aspergillus sp | 160–640 µg/mL | Amphotericin B | 2–8 µg/mL | [20] |
| Dermatophytes | Fluconazole | 16–168 µg/mL | |||
| Candida sp | Fluconazole | 1–168 µg/mL | |||
| Origanum vulgare | C. albicans | 1.48–1.75 mg/mL | Amphotericin B | 100 µg/mL | [27] |
| A. niger | 2.75–2.85 mg/mL | Amphotericin B | 100 µg/mL | ||
| Mentha piperita | Candida albicans | 1 mg/mL | Amphotericin B | 1 µL/mL | [29] |
| Aspergillus niger | 250 mg/mL | Amphotericin B | 0.25 µL/mL | ||
| Pelargonium graveolens Cinnamomum verum Sysygium aromaticum | C. albicans | 500–1000 µg/mL | Amphotericin B Fluconazole | 0.5–2 (µg/mL) 62.5–1000 (µg/mL) | [46] |
| C. tropicalis | 250 µg/mL | Amphotericin B Fluconazole | 2 (µg/mL) 1000 (µg/mL) | ||
| C. parasilopsisi | 500 µg/mL | Amphotericin B Fluconazole | 2 (µg/mL) 7.81 (µg/mL) | ||
| C. glabrata | 500 µg/mL | Amphotericin B Fluconazole | 2 (µg/mL) 15.62–31.25 (µg/mL) | ||
| C. riferi | 500 µg/mL | Amphotericin B Fluconazole | 2 (ug/mL) 1000 (ug/mL) | ||
| Cinnamomum verum | Fusarium sp | 31.25–500 µg/mL | Natamycin | 2–256 ug/mL | [50] |
| Cuminum cyminum | C. albicans | 3.90–11.71 µg/mL | Fluconazole | 3.24–54 ug/mL | [55] |
| Essential Oil | Part of the Plant | Familly | References |
|---|---|---|---|
| Thyme | Leaves Aerial part | Lamiaceae | [20,21] |
| Tea Tree | Leaves | Myrtaceae | [21] |
| Origano | Leaves | Lamiaceae | [21,27] |
| Mentha | Flowering aerial part | Lamiaceae | [29] |
| Lavander | Aerial part | Lamiaceae | [33] |
| Rosmarin | Aerial part Leaves | Lamiaceae | [21,36,37] |
| Geranium | Aerial Part | Geraniaceae | [46] |
| Eucalyptus | Leaves | Myrtaceae | [42,43] |
| Cinnamon | Leaves Bark | Lauraceae | [46,51] |
| Cumin | Seeds Aerial part | Apiaceae | [54,55] |
| Clove | Leaves | Myrtaceae | [45,46] |
| Compounds | Molecular Formula | Essential Oil | Structure |
|---|---|---|---|
| Thymol 2-Isopropyl-5-methylphenol | C10H14O | Thyme | 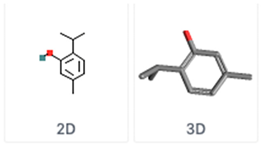 |
| Carvacrol 5-Isopropyl-2-methylphenol | C10H14O | Oregano Savory | 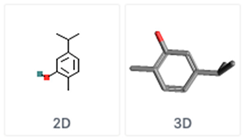 |
| Geraniol Geranyl alcohol | C10H18O | Lemongrass | 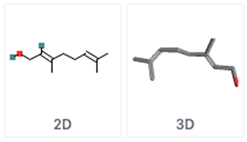 |
| Cinnamaldehyde trans-Cinnamaldehyde | C9H8O | Cinnamon | 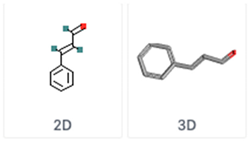 |
| Molecules | Mechanism of Action | Species Tested | Reference |
|---|---|---|---|
| Thymol | Ergosterol binding: plasma membrane permeability | Candida spp | [64] |
| Inhibition of hyphal growth, conidia production and germination Electrolytes leakage Lipid peroxidation | F. graminearum | [63] | |
| Telomerase activity inhibition: cell death, stop of the cellular cylce, apopotoe | S. cerevisae | [65] | |
| Carvacrol | Targeting plasma membrane protein and intracellular target: disruption and depolarization of the plasma membrane Endoplasmic reticulum disruption: unfold protein response | C. albicans/C. tropicalis C. albicans | [67] [68] |
| Geraniol | Inhibition of the calcineurin pathway: plasma membrane and cell wall damage, ROS production | C. albicans | [71] |
| Cinnamaldehyde | Decrease of the virulence factors Effect on spore production, fungal growth and aflatoxine | C. albicans A. flavus | [73] [74,75] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’agostino, M.; Tesse, N.; Frippiat, J.P.; Machouart, M.; Debourgogne, A. Essential Oils and Their Natural Active Compounds Presenting Antifungal Properties. Molecules 2019, 24, 3713. https://doi.org/10.3390/molecules24203713
D’agostino M, Tesse N, Frippiat JP, Machouart M, Debourgogne A. Essential Oils and Their Natural Active Compounds Presenting Antifungal Properties. Molecules. 2019; 24(20):3713. https://doi.org/10.3390/molecules24203713
Chicago/Turabian StyleD’agostino, Maurine, Nicolas Tesse, Jean Pol Frippiat, Marie Machouart, and Anne Debourgogne. 2019. "Essential Oils and Their Natural Active Compounds Presenting Antifungal Properties" Molecules 24, no. 20: 3713. https://doi.org/10.3390/molecules24203713
APA StyleD’agostino, M., Tesse, N., Frippiat, J. P., Machouart, M., & Debourgogne, A. (2019). Essential Oils and Their Natural Active Compounds Presenting Antifungal Properties. Molecules, 24(20), 3713. https://doi.org/10.3390/molecules24203713






