Layered Extraction and Adsorption Performance of Extracellular Polymeric Substances from Activated Sludge in the Enhanced Biological Phosphorus Removal Process
Abstract
1. Introduction
2. Results and Discussion
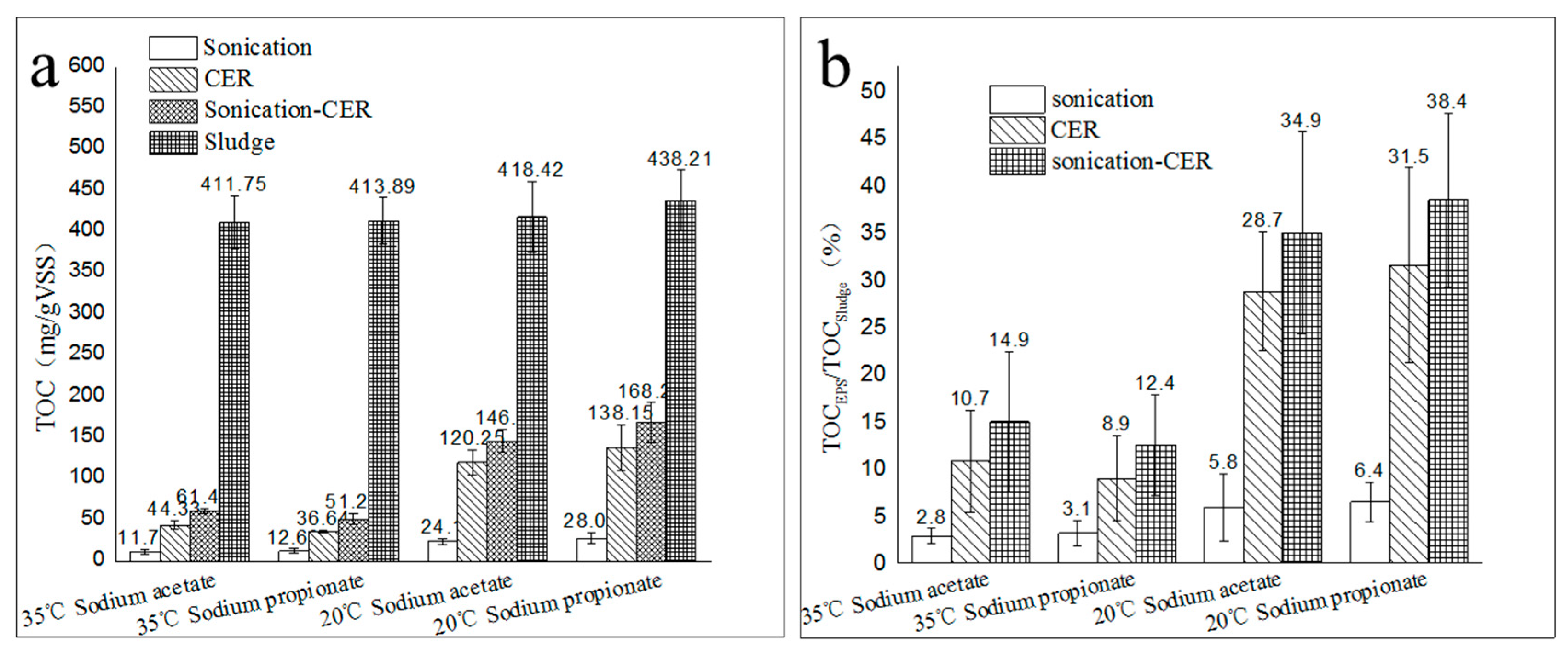
2.1. Extraction Efficacy of EPS
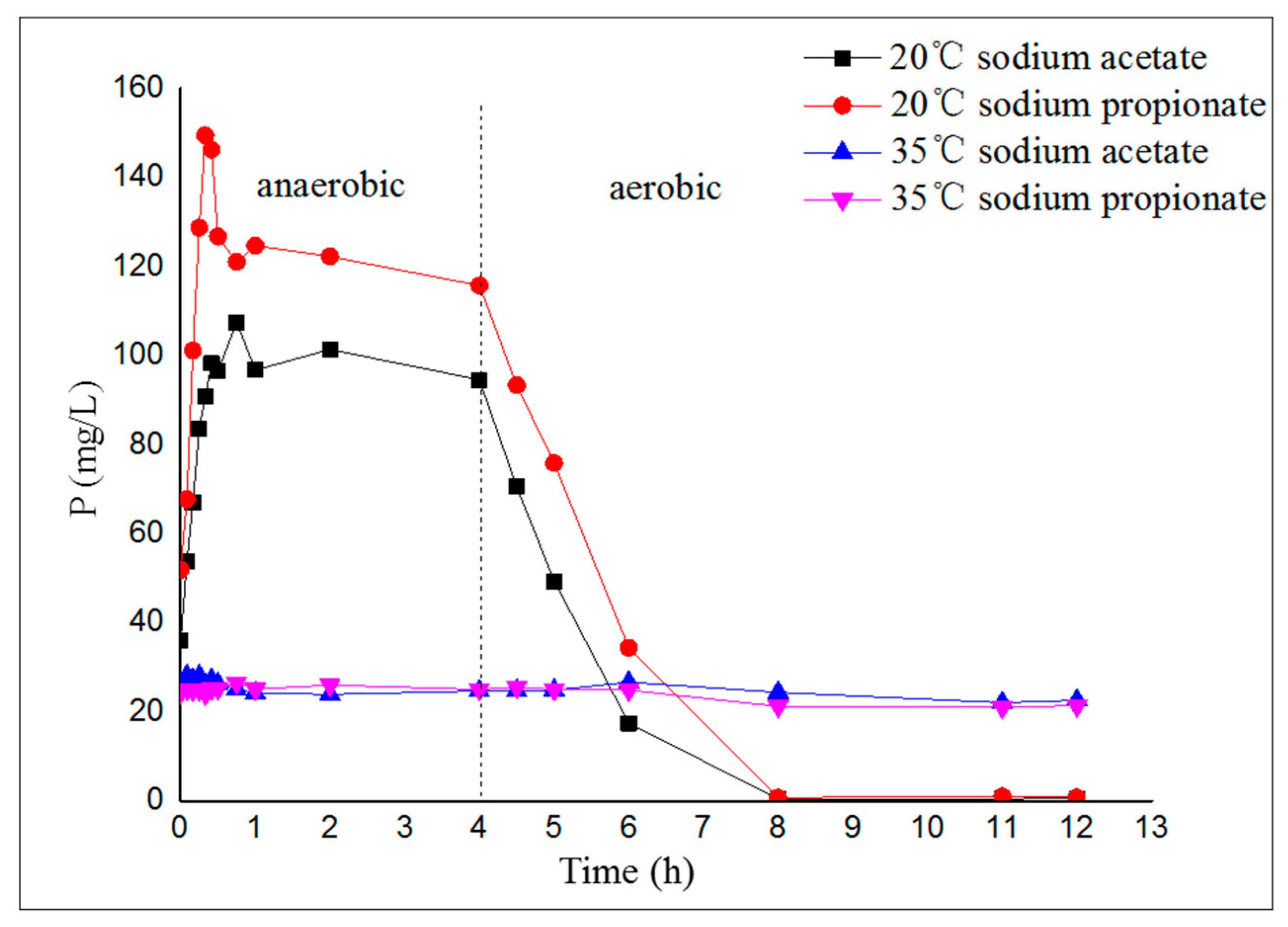
2.2. Phosphorus Removal Performance and Microbial Ecological Mechanism
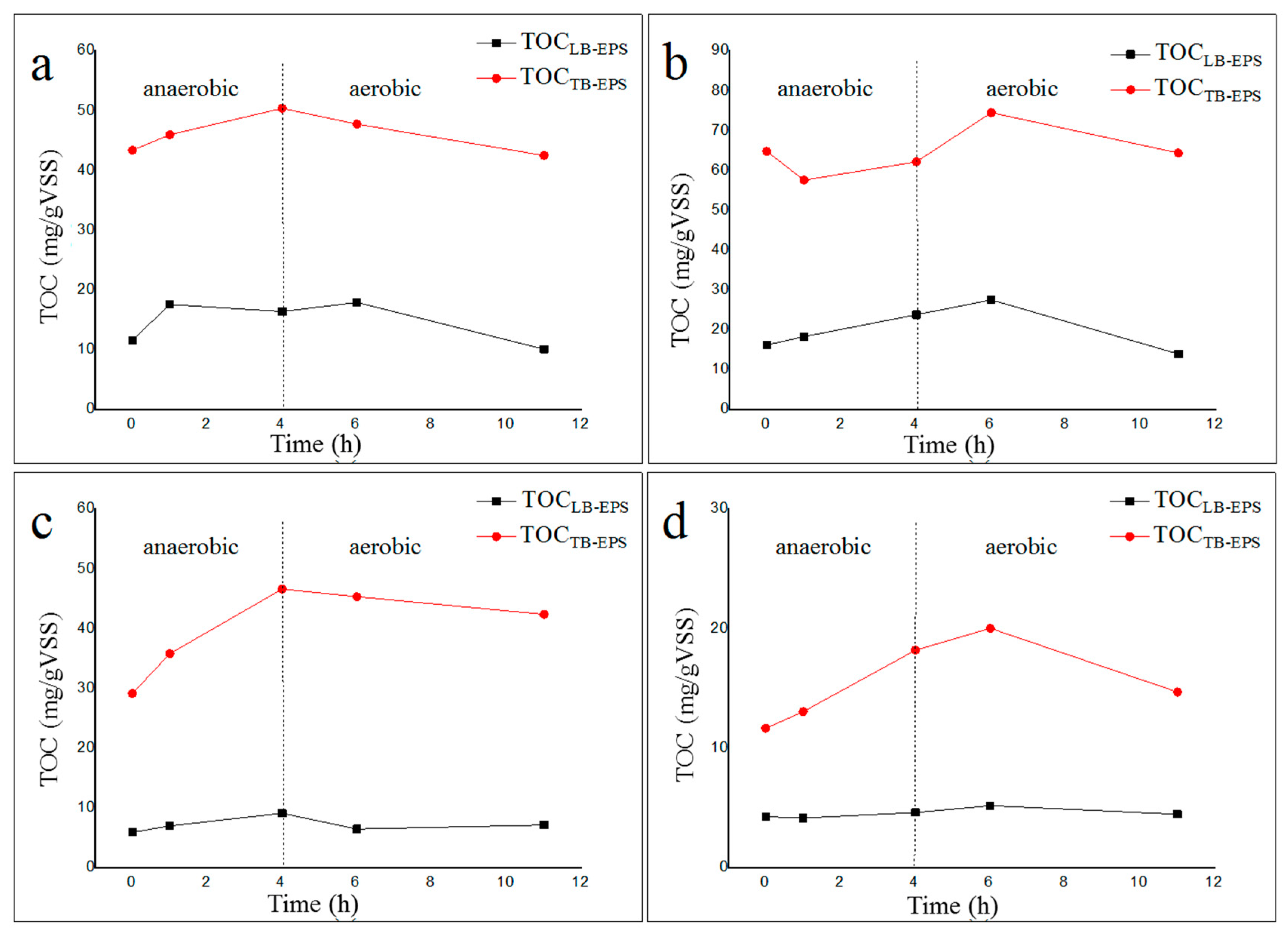
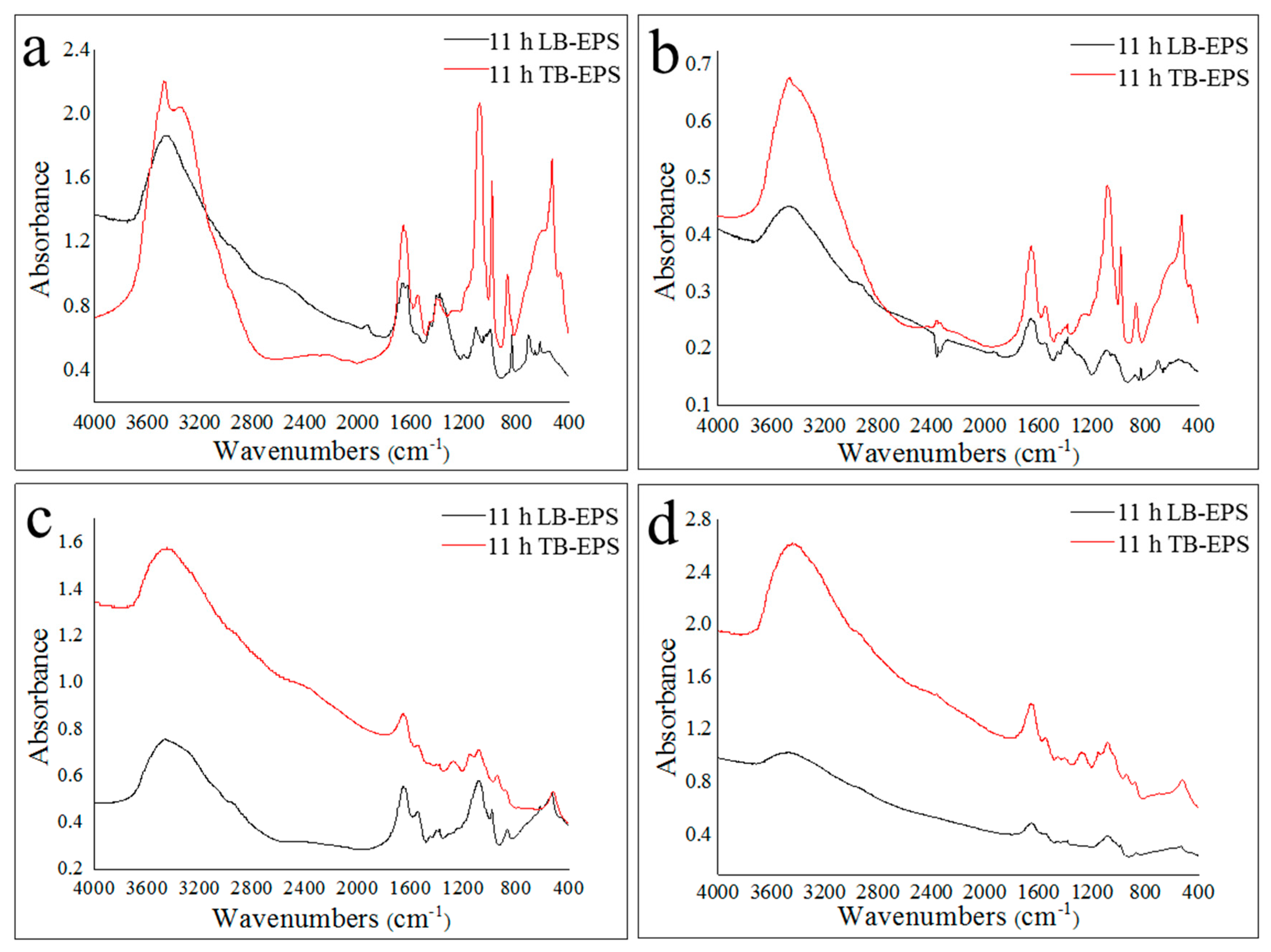
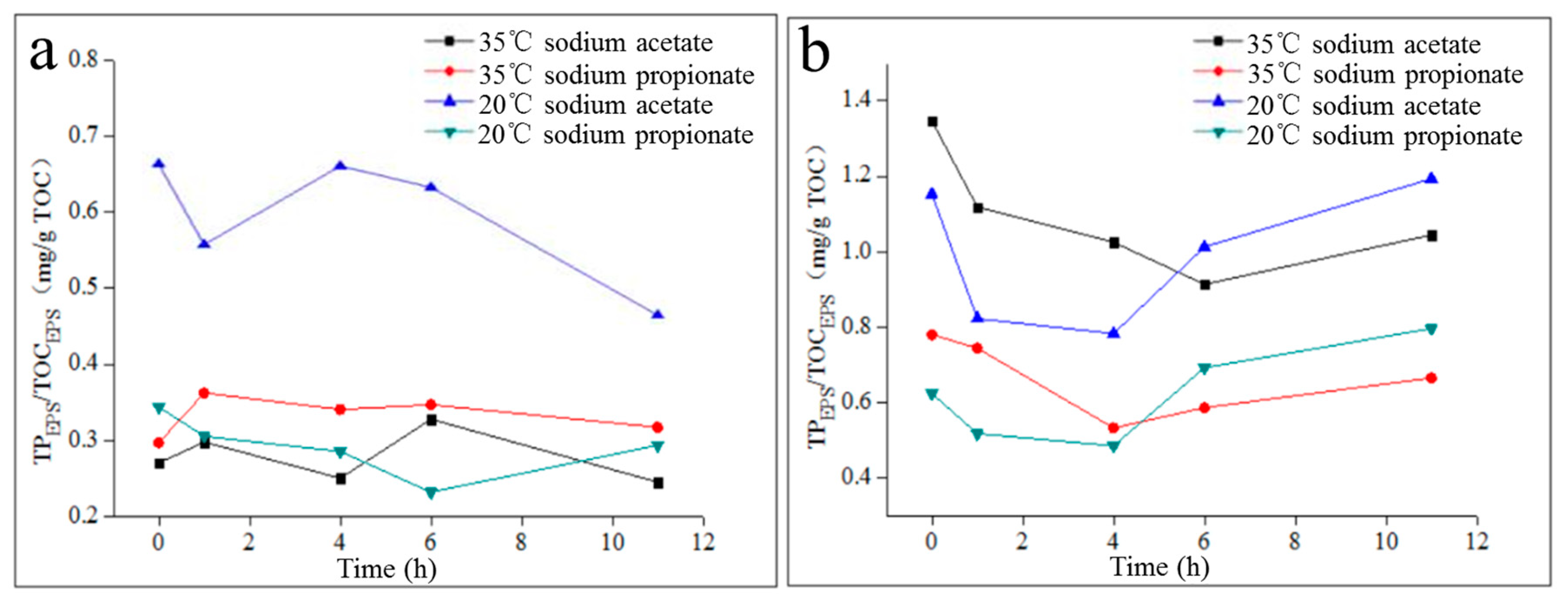
2.3. Properties of LB-EPS and TB-EPS Phosphorous Adsorption/Complexing
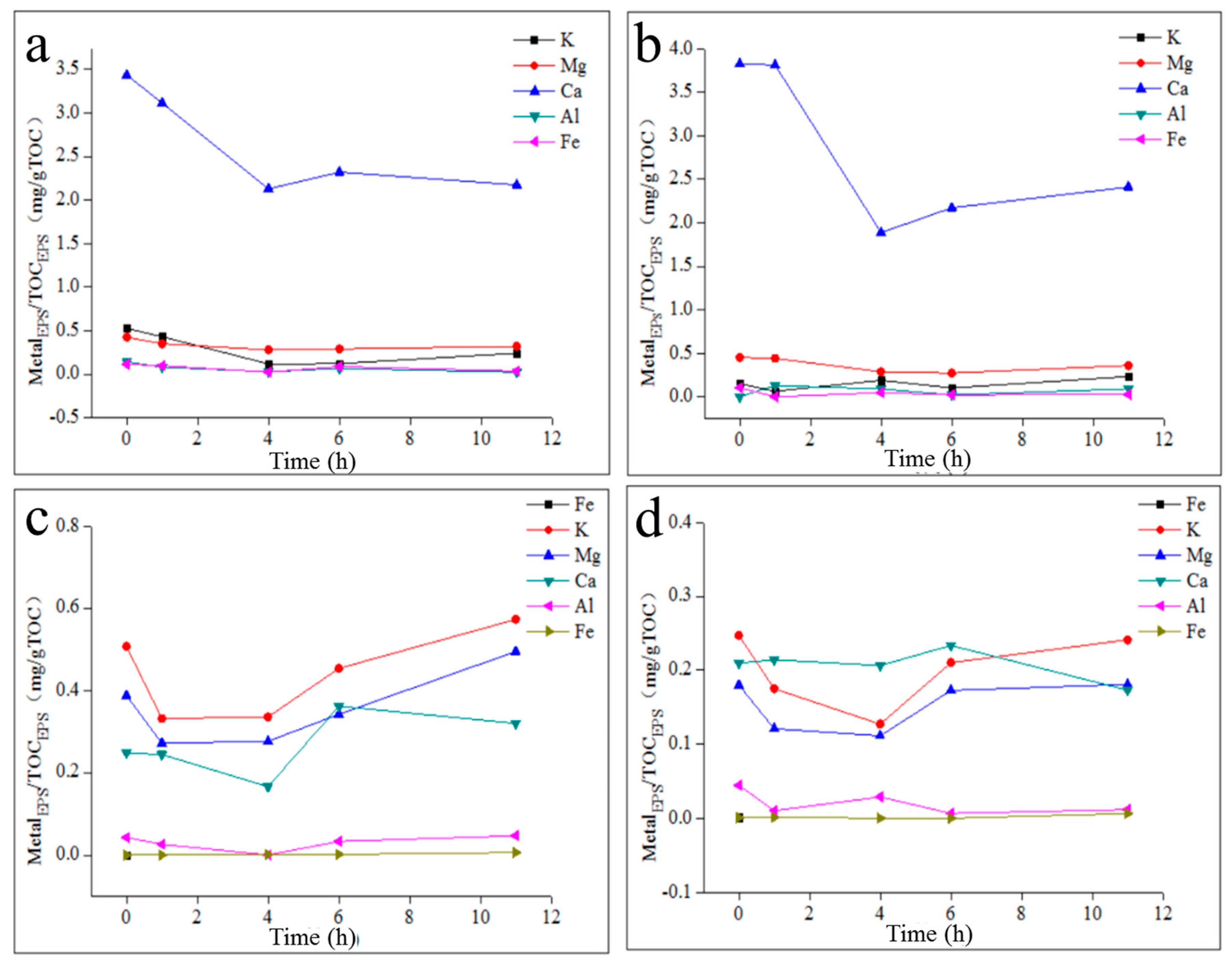
2.4. The Effect of Metal Elements on TB-EPS Phosphorous Adsorption/Complexing
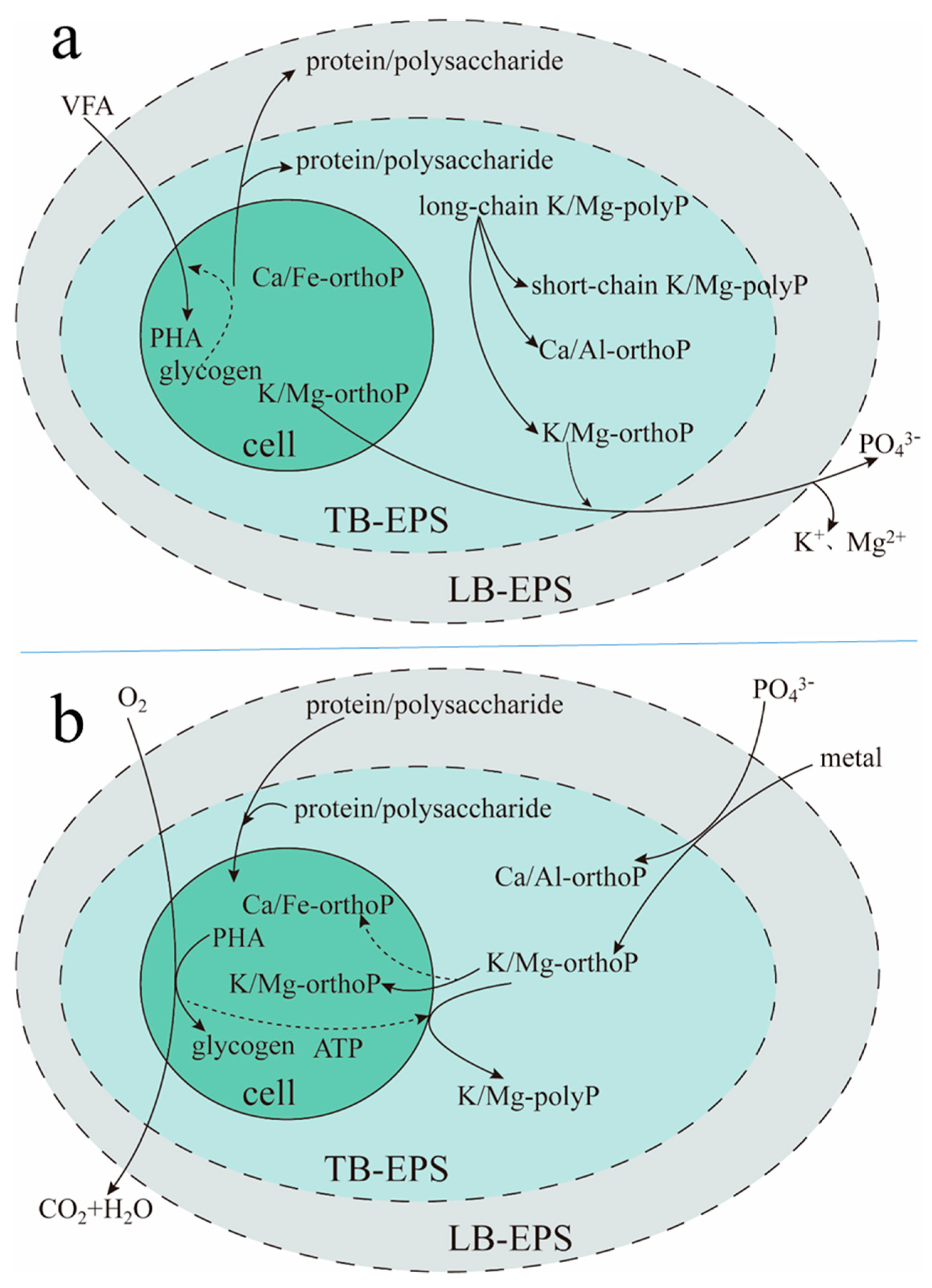
2.5. The Biochemical Reaction Process of TB-EPS Phosphate Adsorption/Complexing in EBPR-Activated Sludge
3. Materials and Methods
3.1. Culture of Activated Sludge
3.2. Method of Washing CER
3.3. Extraction of EPS
3.3.1. Extraction of LB-EPS
3.3.2. Extraction of TB-EPS
3.3.3. Extraction of EPS
3.4. FTIR Analysis
3.5. Other Analyses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yan, L.L.; Yu, L.B.; Liu, Q.P.; Zhang, X.L.; Liu, Y.; Zhang, M.Y.; Liu, S.; Ren, Y.; Chen, Z.L. Effects of phosphorus on loosely bound and tightly bound extracellular polymer substances in aerobic granular sludge. Chem. Biochem. Eng. Q. 2019, 33, 59–68. [Google Scholar] [CrossRef]
- Zhang, H.L.; Fang, W.; Wang, Y.P.; Sheng, G.P.; Zeng, R.J.; Li, W.W.; Yu, H.Q. Phosphorus removal in an enhanced biological phosphorus removal process: Roles of extracellular polymeric substances. Environ. Sci. Technol. 2013, 47, 11482–11489. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.L.; Fang, W.; Wang, Y.P.; Sheng, G.P.; Xia, C.W.; Zeng, R.J.; Yu, H.Q. Species of phosphorus in the extracellular polymeric substances of EBPR sludge. Bioresource Technol. 2013, 142, 714–718. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Wang, Z.; Zhu, C.; Wu, Z. Effect of ultrasonic power density on extracting loosely bound and tightly bound extracellular polymeric substances. Desalination 2013, 329, 35–40. [Google Scholar] [CrossRef]
- Basuvaraj, M.; Fein, J.; Liss, S.N. Protein and polysaccharide content of tightly and loosely bound extracellular polymeric substances and the development of a granular activated sludge floc. Water Res. 2015, 82, 104–117. [Google Scholar] [CrossRef] [PubMed]
- Iorhemen, O.T.; Hamza, R.A.; Zaghloul, M.S.; Tay, J.H. Aerobic granular sludge membrane bioreactor (AGMBR): Extracellular polymeric substances (EPS) analysis. Water Res. 2019, 156, 305–314. [Google Scholar] [CrossRef]
- Zhao, W.; Yang, S.; Huang, Q.; Cai, P. Bacterial cell surface properties: Role of loosely bound extracellular polymeric substances (LB-EPS). Colloid Surf. B Biointerfaces 2015, 128, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Xiao, B.Y.; Luo, M.; Yang, T.; Guo, X.S.; Yi, H. Evaluation of the secondary structures of protein in the extracellular polymeric substances extracted from activated sludge by different methods. J. Environ. Sci. 2018, 6, 1663–1673. [Google Scholar] [CrossRef]
- Sheng, G.P.; Yu, H.Q.; Li, X.Y. Extracellular polymeric substances (EPS) of microbial aggregates in biological wastewater treatment systems: A review. Biotechnol. Adv. 2010, 28, 882–894. [Google Scholar] [CrossRef]
- Wang, B.B.; Liu, X.T.; Chen, J.M.; Peng, D.C.; He, F. Composition and functional group characterization of extracellular polymeric substances (EPS) in activated sludge: The impacts of polymerization degree of proteinaceous substrates. Water Res. 2017, 129, 133–142. [Google Scholar] [CrossRef]
- Wang, H.; Deng, H.; Ma, L.; Ge, L. Influence of operating conditions on extracellular polymeric substances and surface properties of sludge flocs. Carbohyd. Polym. 2013, 92, 510–515. [Google Scholar] [CrossRef]
- Güler, G.; Vorob’Ev, M.M.; Vogel, V.; Mäntele, W. Proteolytically-induced changes of secondary structural protein conformation of bovine serum albumin monitored by Fourier transform infrared (FT-IR) and UV-circular dichroism spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2016, 161, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.X.; Lu, C.H.; Liu, P.; Chai, X.L.; Chen, X.; Min, X.B.; Tang, C.J.; Chai, L.Y. Insights into the role of extracellular polymeric substances in Zn2+ adsorption in different biological sludge systems. Environ. Sci. Pollut. Res. 2018, 25, 36680–36692. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dong, D.; Hua, X.; Guo, Z. Inhibitory effects of extracellular polymeric substances on ofloxacin sorption by natural biofilms. Sci. Total Environ. 2017, 625, 178. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.J.; Wang, J.J.; Liu, S.C.; Sun, X.D.; Yuan, X.Z.; Wang, S.G. Role of extracellular polymeric substances in the acute inhibition of activated sludge by polystyrene nanoparticles. Environ. Pollut. 2018, 238, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Guo, G.; Wu, D.; Ekama, G.A.; Hao, T.; Mackey, H.R.; Chen, G. Denitrifying sulfur conversion-associated EBPR: Effects of temperature and carbon source on anaerobic metabolism and performance. Water Res. 2018, 141, 36–43. [Google Scholar] [CrossRef]
- Oehmen, A.; Lemos, P.C.; Carvalho, G.; Yuan, Z.; Keller, J.; Blackall, L.L.; Reis, M.A.M. Advances in enhanced biological phosphorus removal: From micro to macro scale. Water Res. 2007, 41, 2271–2300. [Google Scholar] [CrossRef] [PubMed]
- Qiu, G.; Zuniga-Montanez, R.; Law, Y.; Thi, S.S.; Nguyen, T.Q.N.; Eganathan, K.; Liu, X.; Nielsen, P.H.; Williams, R.B.H.; Wuertz, S. Polyphosphate-accumulating organisms in full-scale tropical wastewater treatment plants use diverse carbon sources. Water Res. 2019, 149, 496–510. [Google Scholar] [CrossRef]
- Mielcarek, A.; Rodziewicz, J.; Janczukowicz, W.; Thornton, A. The feasibility of citric acid as external carbon source for biological phosphorus removal in a sequencing batch biofilm reactor (SBBR). Biochem. Eng. J. 2015, 93, 102–107. [Google Scholar] [CrossRef]
- Shen, N.; Chen, Y.; Zhou, Y. Multi-cycle operation of Enhanced Biological Phosphorus Removal (EBPR) with different carbon sources under high temperature. Water Res. 2017, 114, 308–315. [Google Scholar] [CrossRef]
- Kee, F.L.; Tadashi, S.; Ying, H.O.; Adeline, S.M.C.; Hak, K.Y.; Pei, Y. Kinetic and stoichiometric characterization for efficient enhanced biological phosphorus removal (EBPR) process at high temperatures. Bioproc. Biosyst. Eng. 2015, 38, 729–737. [Google Scholar]
- Sayi-Ucar, N.; Sarioglu, M.; Insel, G.; Cokgor, E.U.; Orhon, D.; Loosdrecht, M.C.M.V. Long-term study on the impact of temperature on enhanced biological phosphorus and nitrogen removal in membrane bioreactor. Water Res. 2015, 84, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Zeng, F.; Jin, W.; Zhao, Q. Temperature effect on extracellular polymeric substances (EPS) and phosphorus accumulating organisms (PAOs) for phosphorus release of anaerobic sludge. RSC Adv. 2019, 9, 2162–2171. [Google Scholar] [CrossRef]
- Frølund, B.; Palmgren, R.; Keiding, K.; Nielsen, P.H. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Res. 1996, 30, 1749–1758. [Google Scholar] [CrossRef]
- Wang, R.Y.; Li, Y.M.; Chen, W.L.; Zou, J.T.; Chen, Y.G. Phosphate release involving PAOs activity during anaerobic fermentation of EBPR sludge and the extension of ADM1. Chem. Eng. J. 2016, 287, 436–447. [Google Scholar] [CrossRef]
- Wang, R.D.; Peng, Y.Z.; Cheng, Z.L.; Ren, N.Q. Understanding the role of extracellular polymeric substances in an enhanced biological phosphorus removal granular sludge system. Bioresource Technol. 2014, 169, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Long, X.; Tang, R.; Fang, Z.; Xie, C.; Li, Y.; Xian, G. The roles of loosely-bound and tightly-bound extracellular polymer substances in enhanced biological phosphorus removal. Chemosphere. 2017, 189, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Clesceri, L.S.; Greenberg, A.E.; Eaton, A.D. Organic Contaminant. In Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 1999. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, D.; Xi, H. Layered Extraction and Adsorption Performance of Extracellular Polymeric Substances from Activated Sludge in the Enhanced Biological Phosphorus Removal Process. Molecules 2019, 24, 3358. https://doi.org/10.3390/molecules24183358
Li D, Xi H. Layered Extraction and Adsorption Performance of Extracellular Polymeric Substances from Activated Sludge in the Enhanced Biological Phosphorus Removal Process. Molecules. 2019; 24(18):3358. https://doi.org/10.3390/molecules24183358
Chicago/Turabian StyleLi, Daxue, and Hailing Xi. 2019. "Layered Extraction and Adsorption Performance of Extracellular Polymeric Substances from Activated Sludge in the Enhanced Biological Phosphorus Removal Process" Molecules 24, no. 18: 3358. https://doi.org/10.3390/molecules24183358
APA StyleLi, D., & Xi, H. (2019). Layered Extraction and Adsorption Performance of Extracellular Polymeric Substances from Activated Sludge in the Enhanced Biological Phosphorus Removal Process. Molecules, 24(18), 3358. https://doi.org/10.3390/molecules24183358





