Anti-Haemophilus Activity of Selected Essential Oils Detected by TLC-Direct Bioautography and Biofilm Inhibition
Abstract
1. Introduction
2. Results
2.1. Chemical Composition of EOs
2.2. TLC-DB
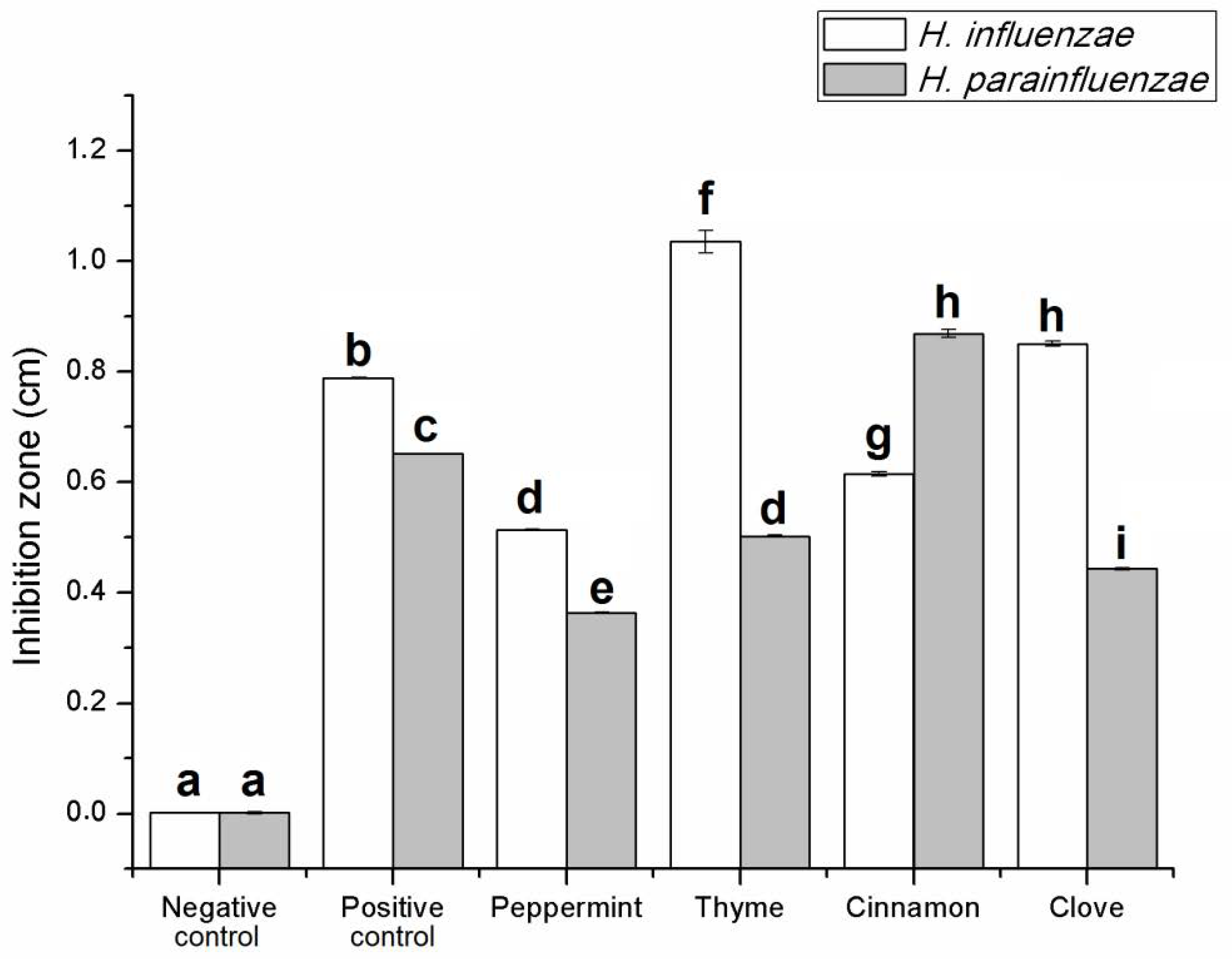
2.2.1. Antibacterial Activity of EOs
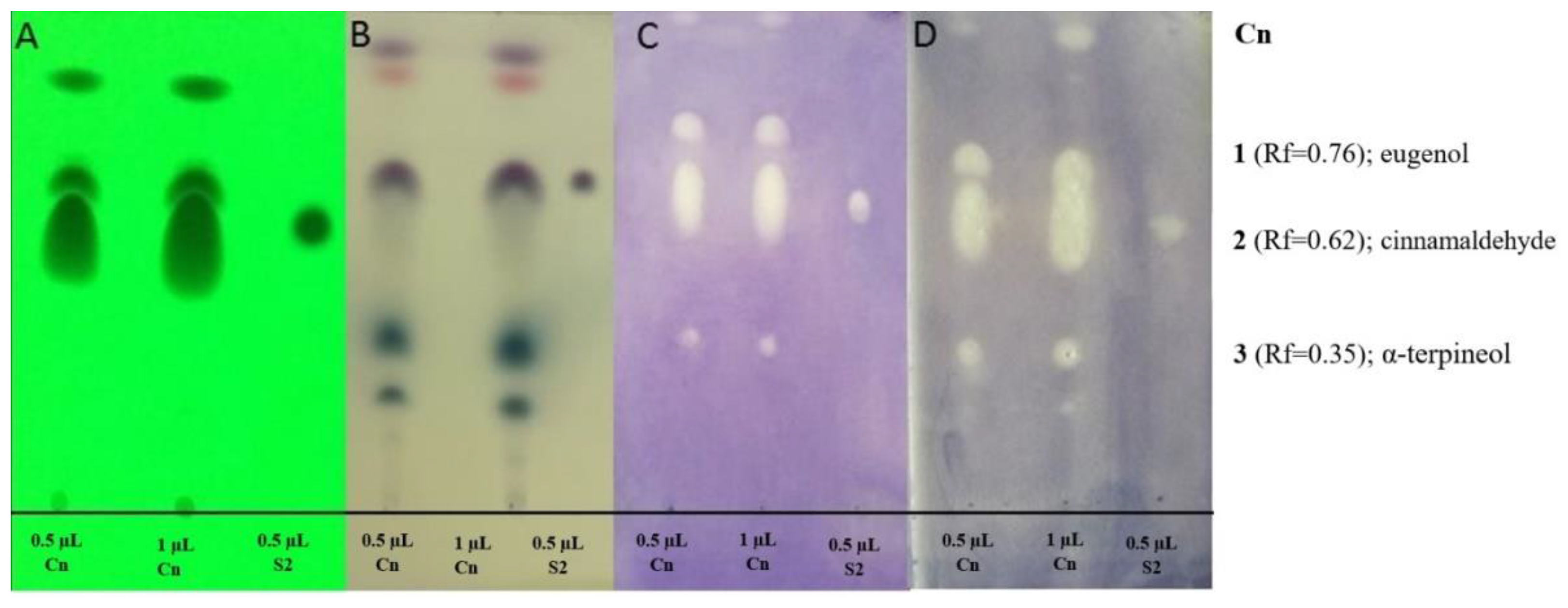
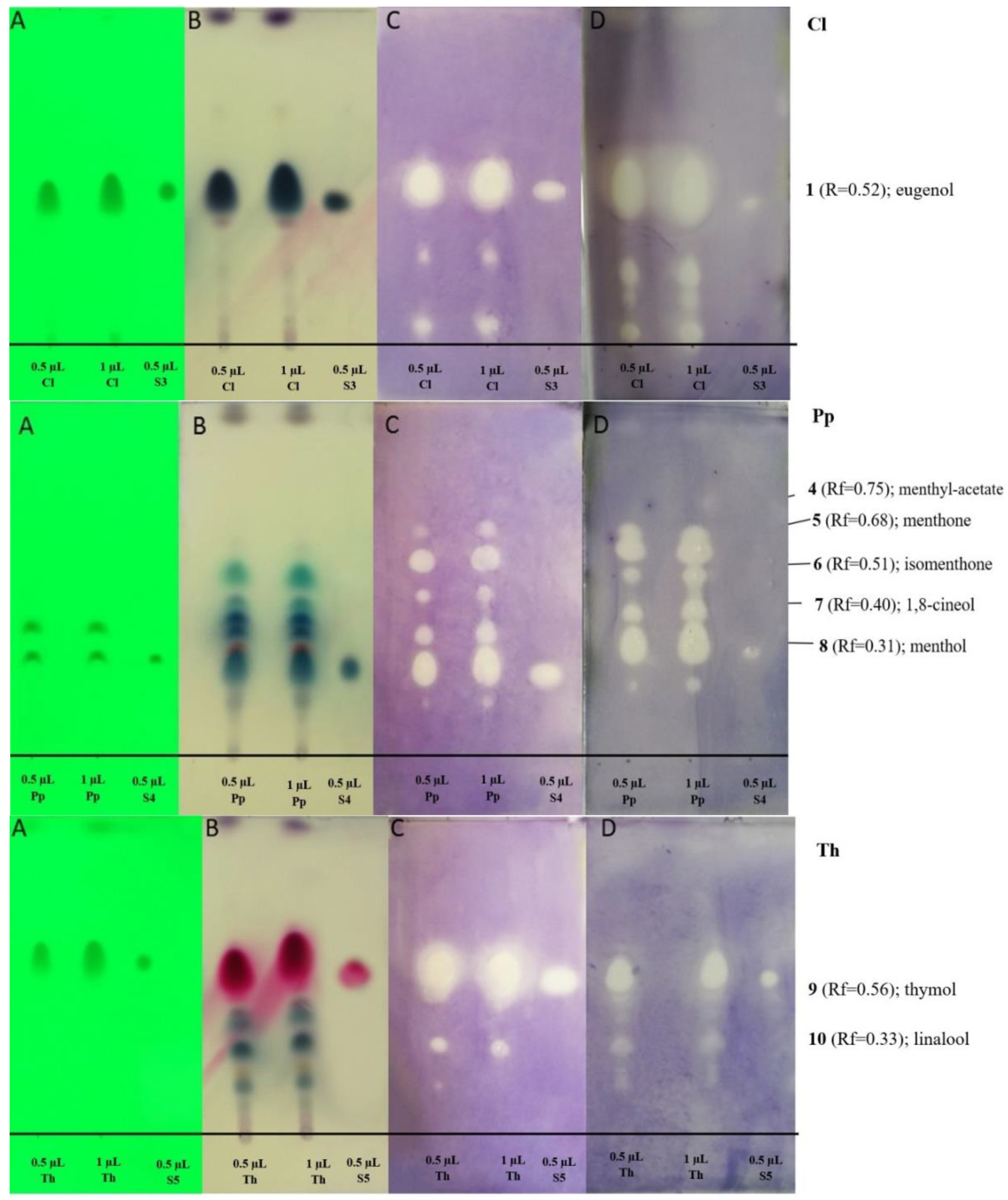
2.2.2. Antibacterial Activity of Main Components of EOs by TLC-DB Method
2.3. Preparation and Characterization of Pickering Nano-Emulsions of EOs
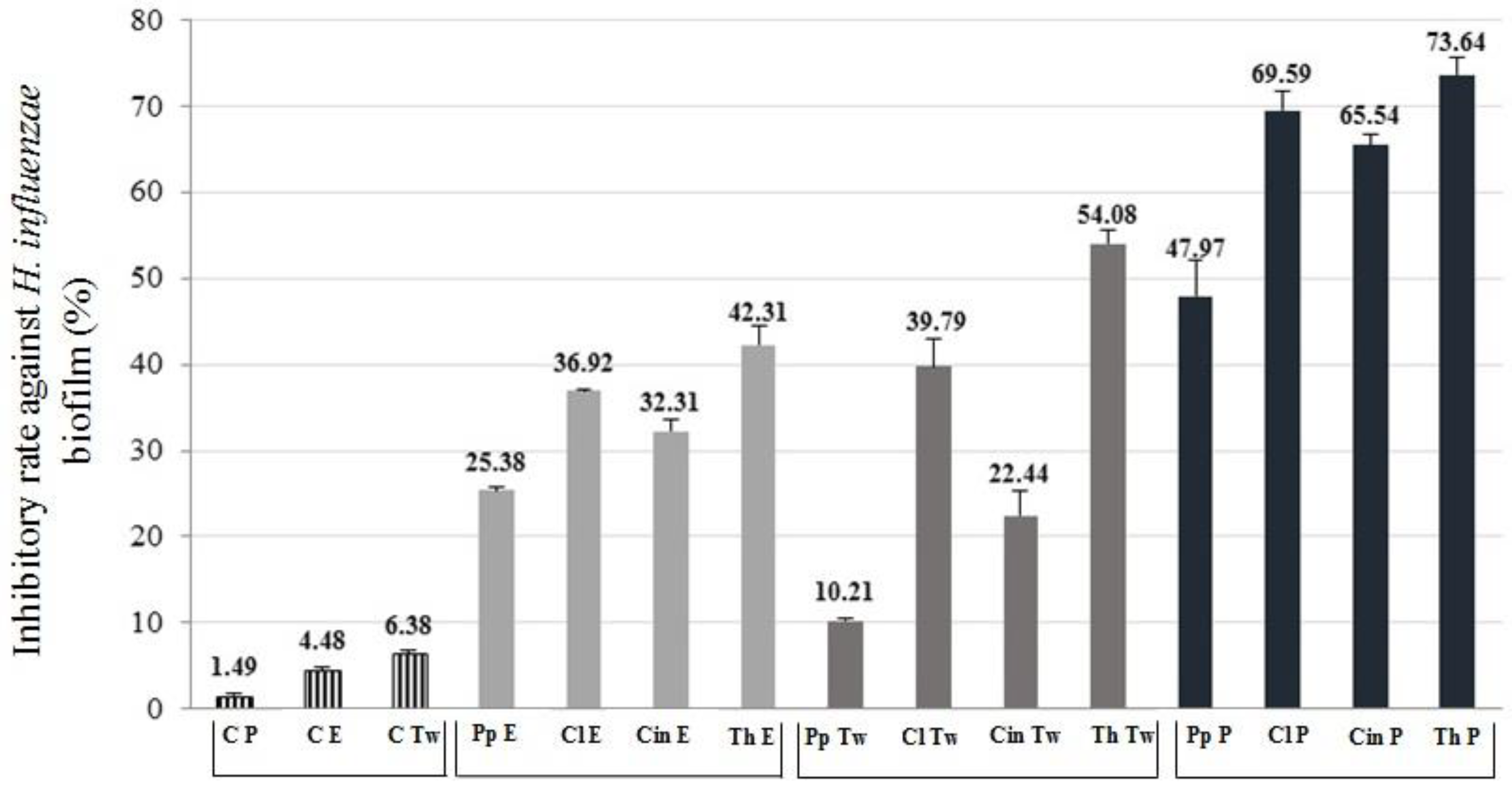
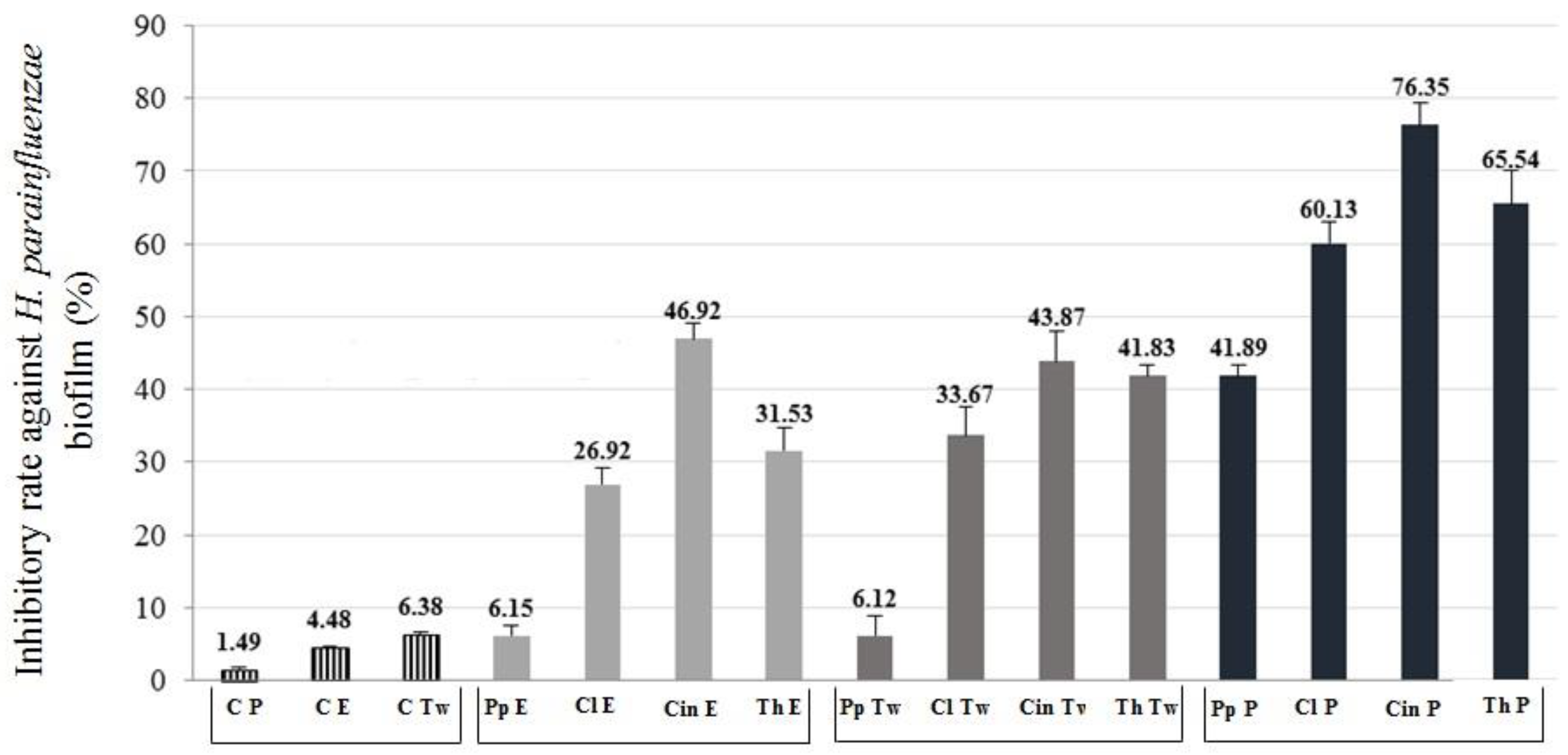
2.4. Anti-Biofilm Activity
3. Discussion
4. Materials and Methods
4.1. Essential Oils and Their Components
4.2. GC-FID and GC-MS
4.3. TLC-DB
4.3.1. Cultivation of Test Bacteria for Dipping
4.3.2. Layer Chromatography
4.3.3. Post-Chromatographic Detection
4.4. Statistical Analyses
4.5. Biofilm Inhibition Experiments
4.5.1. Broth Macrodilution Test (BDT)
4.5.2. Preparation of Pickering Emulsion of the EOs
Synthesis, Surface Modification and Characterization of Silica Nanoparticles
Preparation and Characterization of O/W (Oil/Water) Type EOs Emulsions
4.5.3. Anti-Biofilm Activity Test
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Van Welie, R.T.H. Alle Cosmetica Ingredienten En Hun Functies; Nederlandse Cosmetica Vereniging: Zeist, The Netherlands, 1997; Volume 126. [Google Scholar]
- Bauer, K.; Garbe, D. Common fragrance and flavor materials. In Preparation, Properties and Uses; VCH Verlagsgesellschaft: Weinheim, Germany, 1985; Volume 213. [Google Scholar]
- Lober, B. Update in infectious diseases. Ann. Intern. Med. 2006, 145, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Eloff, J.N. A proposal on expressing the antibacterial activity of plant extracts—A small first step in applying scientific knowledge to rural primary health care. S. Afr. J. Sci. 2000, 96, 116–118. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef] [PubMed]
- Jones, F.A. Herbs—Useful plants. Their role in history and today. Eur. J. Gastroenterol. Hepatol. 1996, 8, 1227–1231. [Google Scholar] [CrossRef] [PubMed]
- Botz, L.; Nagy, S.; Kocsis, B. Detection of microbiologically active compounds. In Planar Chromatography, A Retrospective View for the Third Millenium; Nyiredy, S., Ed.; Springer: Budapest, Hungary, 2001; pp. 489–516. [Google Scholar]
- Lund, B.M.; Lyon, G.D. Detection of inhibitors of Erwinia carotovora and E. herbicola on thin-layer chromatograms. J. Chromatogr. 1975, 110, 193–196. [Google Scholar] [CrossRef]
- Horváth, G.; Botz, L.; Kocsis, B.; Lemberkovics, É.; Szabó, L.G. Antimicrobial natural products and antibiotics detected by direct bioautography using plant pathogenic bacteria. Acta Bot. Hung. 2004, 46, 153–165. [Google Scholar] [CrossRef]
- Quiroga, E.N.; Sampietro, D.A.; Sgariglia, M.A.; Soberón, J.R.; Vattuone, M.A. Antimycotic activity of 5′-prenylisoflavanones of the plant Geoffroea decorticans, against Aspergillus species. Int. J. Food Microbiol. 2009, 132, 42–46. [Google Scholar] [CrossRef]
- Fabio, A.; Cermelli, C.; Fabio, G.; Nicoletti, P.; Quaglio, P. Screening of the antibacterial effects of a variety of essential oils on microorganisms responsible for respiratory infections. Phytother. Res. 2007, 21, 374–377. [Google Scholar] [CrossRef]
- Inouye, S.; Yamaguchi, H.; Takizawa, T. Screening of antibacterial effect of a variety of essential oils on respiratory tract pathogens, using a modified dilution assay method. J. Infect. Chemother. 2001, 7, 251–254. [Google Scholar] [CrossRef]
- World Health Organization: Ten Leading Causes of Death. Available online: http://apps.who.int/gho/data/view.wrapper.MGHEMORTCAUSE10-2012?lang=en (accessed on 3 September 2014).
- Ács, K.; Balázs, V.L.; Kocsis, B.; Bencsik, T.; Böszörményi, A.; Horváth, G. Antibacterial activity evaluation of selected essential oils in liquid and vapor phase on respiratory tract pathogenes. BMC Complement. Altern. Med. 2018, 18, 227. [Google Scholar] [CrossRef]
- Ács, K.; Bencsik, T.; Böszörményi, A.; Kocsis, B.; Horváth, G. Essential oils and their vapors as potential antibacterial agents against respiratory tract pathogens. Nat. Prod. Commun. 2016, 11, 1709–1712. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.C.; Neu, T.R.; Wozniak, D.J. The EPS matrix: The “house of biofilm cells”. J. Bacteriol. 2007, 189, 7945–7947. [Google Scholar] [CrossRef] [PubMed]
- Hall-Stoodley, L.; Hu, F.Z.; Gieseke, A.; Nistico, L.; Nguyen, D.; Hayes, J.; Forbes, M.; Greenberg, D.P.; Dice, B.; Burrows, A.; et al. Direct detection of bacterial biofilms on the middle-ear mucosa of children with chronic otitis media. JAMA 2006, 296, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Ciofu, O.; Lykkesfeldt, J. Antioxidant supplementation for lung disease in cystic fibrosis—Review. Cochrane Database Syst. Rev. 2014, 8, 1–84. [Google Scholar]
- Bryers, J.D. Medical biofilms. Biotechnol. Bioeng. 2008, 100, 1–18. [Google Scholar] [CrossRef] [PubMed]
- LeBel, G.; Haas, B.; Adam, A.A.; Veilleux, M.P.; Lagha, A.B.; Grenier, D. Effect of cinnamon (Cinnamomum verum) bark essential oil on the halitosis-associated bacterium Solobacterium moorei and in vitro cytotoxicity. Arch. Oral Biol. 2017, 83, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Vidács, A.; Kerekes, E.; Rajkó, R.; Petkovits, T.; Alharbi, N.S.; Khaled, J.M.; Vágvölgyi, C.; Krisch, J. Optimization of essential oil-based natural disinfectants against Listeria monocytogenes and Escherichia coli biofilms formed on polypropylene surfaces. J. Mol. Liq. 2018, 255, 257–262. [Google Scholar] [CrossRef]
- Jesionek, W.; Majer-Dziedzic, B.; Choma, M.I. TLC-Direct bioautography as a method for evaluation of antimicrobial properties of Thymus vulgaris L. and Salvia officinalis L. essential oils of different origin. J. Liq. Chromatogr. Relat. Technol. 2017, 40, 292–296. [Google Scholar] [CrossRef]
- Wagner, H.; Bladt, S. Plant Drug Analysis. In A Thin Layer Chromatography Atlas, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2001; pp. 150–161. [Google Scholar]
- Horváth, B.; Szilárd, P.; Széchenyi, A. Preparation and in vitro diffusion study of essential oil Pickering emulsions stabilized by silica nanoparticles. Flavour Fragr. J. 2018, 33, 1–12. [Google Scholar] [CrossRef]
- Kerekes, E.B.; Deák, É.; Takó, M.; Tserennadmid, R.; Petkovits, T.; Vágvölgyi, C.; Krisch, J. Anti-biofilm forming and anti-quorum sensing activity of selected essential oils and their main components on food-related microorganisms. J. Appl. Microbiol. 2013, 115, 933–942. [Google Scholar] [PubMed]
- Yanwei, S.; Sijia, C.; Chen, Z.; Yali, L.; Li, M.; Xiangyu, Z. Effect of sub-minimum inhibitory concentrations of lemon essential oil on the acid tolerance and biofilm formation of Streptococcus mutans. Arch. Oral Biol. 2018, 87, 235–241. [Google Scholar]
- Hammer, K.; Carson, C.; Riley, T. Antimicrobial activity of essential oils and other plant extracts. J. Appl. Microbiol. 1999, 86, 985–990. [Google Scholar] [CrossRef] [PubMed]
- Kavanaugh, N.L.; Ribbec, K. Selected antimicrobial essential oils eradicate Pseudomonas spp. and Staphylococcus aureus biofilms. Appl. Environ. Microbiol. 2012, 78, 4057–4061. [Google Scholar] [CrossRef] [PubMed]
- Saviuc, C.M.; Drumea, V.; Olariu, L.; Chifiriuc, M.C.; Bezirtzoglou, E.; Lazar, V. Essential oils with microbicidal and antibiofilm activity. Curr. Pharm. Biotechnol. 2015, 16, 137–151. [Google Scholar] [CrossRef] [PubMed]
- Skocibusic, M.; Bezic, N.; Dunkic, V.; Radonic, A. Antibacterial activity of Achillea clavennae essential oils on respiratory tract pathogens. Fitoterapia 2004, 75, 733–736. [Google Scholar] [CrossRef]
- Viljoen, A.M.; Subramoney, S.; Van Vuuren, S.F.; Baser, K.H.C.; Demirci, B. The composition, geographical variation and antimicrobial activity of Lippia javanica (Verbenaceae) leaf essential oils. J. Ethnopharmacol. 2005, 96, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Romero, J.; Gonza’lez-Rios, H.; Borges, A.; Simoes, M. Antibacterial effects and mode of action of selected essential oils components against Escherichia coli and Staphylococcus aureus. Evid. Based Complement. Altern. Med. 2015, 2015, 795435. [Google Scholar] [CrossRef]
- Houdkova, M.; Rondevaldova, J.; Doskocil, I.; Kokoska, L. Evaluation of antibacterial potential and toxicity of plant volatile compounds using new broth microdilution volatilization method and modified MTT assay. Fitoterapia 2017, 118, 56–62. [Google Scholar] [CrossRef]
- Derakhshan, S.; Sattari, M.; Bigdeli, M. Effect of cumin (Cuminum cyminum) seed essential oil on biofilm formation and plasmid integrity of Klebsiella pneumoniae. Pharmacogn. Mag. 2010, 6, 57–61. [Google Scholar]
- Lang, G.; Buchbauer, G. A review on recent research (2008–2010) on essential as antimicrobials and antifungals. A review. Flavour Fragr. J. 2011, 27, 13–39. [Google Scholar] [CrossRef]
- Khan, S.T.; Khan, M.; Ahmad, J.; Wahab, R.; Abd-Elkader, O.H.; Musarrat, J. Thymol and carvacrol induce autolysis, stress, growth inhibition and reduce the biofilm formation by Streptococcus mutans. AMB Express 2017, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- De Paula, S.B.; Bartelli, T.F.; Di Raimo, V.; Santos, J.P.; Morey, A.T.; Bosini, M.A. Effect of eugenol on cell surface hydrophobicity, adhesion, and biofilm of Candida tropicalis and Candida dubliniensis isolated from oral cavity of HIV-infected patients. Evid. Based Complement. Altern. Med. 2014, 2014, 505204. [Google Scholar] [CrossRef] [PubMed]
- Mathur, S.; Udgire, M.; Khambhapati, A.; Paul, D. Anti-biofilm activity and bioactive component analysis of eucalyptus oil against urinary tract pathogen. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 912–918. [Google Scholar]
- Kim, Y.G.; Lee, J.H.; Kim, S.I.; Baek, K.H.; Lee, J. Cinnamon bark oil and its components inhibit biofilm formation and toxin production. Int. J. Food Microbiol. 2015, 195, 30–39. [Google Scholar] [CrossRef]
- Almeida, L.F.; Paula, J.F.; Almeida, R.V.; Williams, D.W.; Hebling, J.; Cavalcanti, Y.W. Efficacy of citronella and cinnamon essential oils on Candida albicans biofilms. Acta Odontol. Scand. 2016, 74, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Gupta, C.; Garg, A.P.; Uniyal, R.C.; Kumari, A. Comparative analysis of the antimicrobial activity of cinnamon oil and cinnamon extract on some food-borne microbes. Afr. J. Microbiol. Res. 2008, 2, 247–251. [Google Scholar]
- Zhang, Y.; Liu, X.; Wang, Y.; Jiang, P.; Quek, S.Y. Antibacterial activity and mechanism of cinnamon essential oil against Escherichia coli and Staphylococcus aureus. Food Control 2016, 59, 282–289. [Google Scholar] [CrossRef]
- Trombetta, D.; Castelli, F.; Sarpietro, M.G.; Venuti, V.; Cristani, M.; Daniele, C. Mechanisms of antibacterial action of three monoterpenes. Antimicrob. Agents Chemother. 2005, 49, 2474–2478. [Google Scholar] [CrossRef]
- Hafedh, H.; Fethi, B.A.; Mejdi, S.; Emira, N.; Bakhrouf Amin, B. Effect of Mentha longifolia L. ssp. longifolia essential oil on the morphology of four pathogenic bacteria visualized by atomic force microscopy. Afr. J. Microbiol. Res. 2010, 4, 1122–1127. [Google Scholar]
- Latifah-Munirah, B.; Himratul-Aznita, W.H.; Zain, N.M. Eugenol, an essential oil of clove, causes disruption to the cell wall of Candida albicans (ATCC 14053). Front. Life Sci. 2015, 8, 231–240. [Google Scholar] [CrossRef]
- Yadav, M.K.; Chae, S.W.; Im, G.J.; Chung, J.W.; Song, J.J. Eugenol: A phytocompound effective against methicillin-resistant and methicillin-sensitive Staphylococcus aureus clinical strain biofilms. PLoS ONE 2015, 10, e0119564. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Meng, X.; Li, Y.; Zhao, C.N.; Tang, G.Y.; Li, H.B. Antibacterial and antifungal activities of spices. Int. J. Mol. Sci. 2017, 18, 1283. [Google Scholar] [CrossRef] [PubMed]
- Sambyal, S.S.; Sharma, P.; Shrivastava, D. Antibiofilm activity of selected plant essential oils against Pseudomonas aeruginosa and Staphylococcus aureus. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6, 444–450. [Google Scholar] [CrossRef]
- Rasooli, I.; Rezaei, M.B.; Allameli, A. Ultrastructural studies on antimicrobial efficacy of thyme essential oils on Listeria monocytogenes. Int. J. Infect. Dis. 2006, 10, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Huma, J.; Firoz, A.A.; Iqbal, A. CHAPTER 9 Prospects of Essential Oils in Controlling Pathogenic Biofilm. In New Look to Phytomedicine Advancements in Herbal Products as Novel Drug Leads; Academic Press: Cambridge, CA, USA, 2019; pp. 203–236. [Google Scholar]
- Baothong, S.; Sitthisak, S.; Kunthalert, D. In vitro interference of cefotaxime at subinhibitory concentrations on biofilm formation by nontypeable Haemophilus influenzae. Asian Pac. J. Trop. Med. 2016, 6, 745–750. [Google Scholar] [CrossRef]
- Kosikowska, U.; Andrzejczuk, S.; Plech, T.; Malm, A. Inhibitory effect of 1,2,4-triazole-ciprofloxacin hybrids on Haemophilus parainfluenzae and Haemophilus influenzae biofilm formation in vitro under stationary conditions. Res. Microbiol. 2016, 167, 647–654. [Google Scholar] [CrossRef] [PubMed]
- Takahata, M.; Sugiura, Y.; Shinmura, Y.; Fukuda, Y.; Nomura, N. Bactericidal activity of garenoxacin against in vitro biofilm formed by nontypeable Haemophilus influenzae. J. Infect. Chemother. 2013, 19, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Hindler, J.A.; Jorgensen, J.H. Susceptibility test methods: Fastidious bacteria. In Manual of Clinical Microbiology, 10th ed.; ASM: Washington, DC, USA, 2011; pp. 1180–1187. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Chambers, J.M. Linear models. In Chapter 4 of Statistical Models; Chambers, J.M., Hastie, T.J., Eds.; Wadsworth & Brooks/Cole: Pacific Grove, CA, USA, 1992. [Google Scholar]
- Crawley, M.J. Statistics: An Introduction Using R, 2nd ed.; John Wiley and Sons: Chichester, UK, 2014. [Google Scholar]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Stöber, W.; Fink, A.; Bohn, E. Controlled growth of monodispersed silica spheres in the micron size range. J. Colloid Interface Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Peeters, E.; Nelis, H.J.; Coenye, T. Comparison of multiple methods for quantification of microbial biofilms grown in microliter plates. J. Microbiol. Methods 2008, 72, 157–165. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds cinnamaldehyde, eugenol, menthone, thymol are available from the authors. |
| Component | RI | Percentage of Compounds (%) | |||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | ||
| α-Pinene | 939 | 1.1 | 1.0 | - | 5.1 |
| Camphene | 951 | - | 2.0 | - | - |
| β-Myrcene | 992 | - | 1.0 | - | - |
| α-Terpinene | 1017 | - | 3.2 | - | - |
| p-Cymene | 1026 | - | 19.2 | - | 1.9 |
| Limonene | 1044 | 1.4 | - | - | 1.8 |
| 1,8-Cineole | 1046 | 5.5 | 4.6 | - | 2.8 |
| γ-Terpinene | 1060 | - | 6.7 | - | - |
| Linalool | 1104 | - | 5.6 | - | 4.0 |
| Isopulegol | 1150 | 1.0 | - | - | - |
| Menthone | 1156 | 19.8 | - | - | - |
| Isomenthone | 1159 | 7.0 | - | - | - |
| Menthol | 1172 | 50.4 | - | - | - |
| Isomenthol | 1183 | 4.3 | - | - | - |
| α-Terpineol | 1190 | - | - | - | 2.2 |
| Pulegone | 1215 | 1.9 | - | - | - |
| trans-Cinnamaldehyde | 1266 | - | - | - | 63.7 |
| Bornyl acetate | 1289 | - | 1.0 | - | - |
| Thymol | 1297 | - | 39.8 | - | - |
| Isomenthyl acetate | 1305 | 5.5 | - | - | - |
| Eugenol | 1373 | - | - | 78.8 | 4.6 |
| β-Elemene | 1394 | - | - | - | - |
| β-Caryophyllene | 1417 | 1.3 | 4.2 | 13.5 | 4.2 |
| Cinnamyl acetate | 1446 | - | - | - | 9.4 |
| α-Humulene | 1452 | - | - | 4.6 | - |
| β-Cadinene | 1473 | - | - | 1.1 | - |
| Total: | - | 99.2 | 88.3 | 98.0 | 99.7 |
| Properties of Pickering and Conventional Emulsions of Essential Oils | ||||
|---|---|---|---|---|
| Essential Oil | Coil (mg/mL) | Stabilizing Agent | Droplet Size (nm) | Stability |
| Cinnamon bark EO | 0.03 | 20ET nanoparticles | 256.2 ± 12.3 | 2 months |
| 0.03 | Tween80 | 274.5 ± 28.5 | 1 month | |
| Clove EO | 0.125 | 20ET nanoparticles | 184.6 ± 8.8 | 2 weeks |
| 0.125 | Tween80 | 185.2 ± 10.7 | 2 weeks | |
| Peppermint EO | 0.105 | 20ET nanoparticles | 308.7 ± 15.5 | 5 months |
| 0.105 | Tween80 | 248.9 ± 4. | 1 months | |
| Thyme EO | 0.055 | 20ET nanoparticles | 180.5 ± 6.4 | 4 months |
| 0.055 | Tween80 | 163.2 ± 1.3 | 1 month | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balázs, V.L.; Horváth, B.; Kerekes, E.; Ács, K.; Kocsis, B.; Varga, A.; Böszörményi, A.; Nagy, D.U.; Krisch, J.; Széchenyi, A.; et al. Anti-Haemophilus Activity of Selected Essential Oils Detected by TLC-Direct Bioautography and Biofilm Inhibition. Molecules 2019, 24, 3301. https://doi.org/10.3390/molecules24183301
Balázs VL, Horváth B, Kerekes E, Ács K, Kocsis B, Varga A, Böszörményi A, Nagy DU, Krisch J, Széchenyi A, et al. Anti-Haemophilus Activity of Selected Essential Oils Detected by TLC-Direct Bioautography and Biofilm Inhibition. Molecules. 2019; 24(18):3301. https://doi.org/10.3390/molecules24183301
Chicago/Turabian StyleBalázs, Viktória Lilla, Barbara Horváth, Erika Kerekes, Kamilla Ács, Béla Kocsis, Adorján Varga, Andrea Böszörményi, Dávid U. Nagy, Judit Krisch, Aleksandar Széchenyi, and et al. 2019. "Anti-Haemophilus Activity of Selected Essential Oils Detected by TLC-Direct Bioautography and Biofilm Inhibition" Molecules 24, no. 18: 3301. https://doi.org/10.3390/molecules24183301
APA StyleBalázs, V. L., Horváth, B., Kerekes, E., Ács, K., Kocsis, B., Varga, A., Böszörményi, A., Nagy, D. U., Krisch, J., Széchenyi, A., & Horváth, G. (2019). Anti-Haemophilus Activity of Selected Essential Oils Detected by TLC-Direct Bioautography and Biofilm Inhibition. Molecules, 24(18), 3301. https://doi.org/10.3390/molecules24183301






