A Review on Coordination Properties of Thiol-Containing Chelating Agents Towards Mercury, Cadmium, and Lead
Abstract
1. Introduction
2. Exposure and Effects
2.1. Mercury
2.2. Cadmium
2.3. Lead
3. Endogenous Protective SH-Compounds: Metallothioneins and Glutathione
3.1. Metallothioneins (MTs)
3.2. Glutathione (GSH)
- It is the principal endogenous antioxidant that the cells produce, it participates directly in the neutralization of ROS and free radicals, and it is a cofactor of the selenoenzyme glutathione peroxidase (GPx).
- GSH is an important substrate for conjugation reactions, catalyzed by the glutathione-S-transferase enzyme. Thus, in the case of the reactive metabolite formed by a paracetamol overdose, GSH acts as an antidote. GSH can also conjugate and detoxify organometallic compounds, such as MeHg [111].
- It has important roles in binding, transport, and storing of several metals, thus affecting the homeostasis of metals in biological systems [112].
4. SH-Containing Chelating Agents: Clinical Use and Environmental Remediation
4.1. BAL (2,3 dimercaptopropan-1-ol)
4.2. DMSA (meso-dimercaptosuccinic acid, Succimer)
4.3. DMPS (2,3-dimercaptopropane-1-sulfonic acid, Unitiol)
4.4. Penicillamine (D-2-amino-3-mercapto-3-methylbutanoic acid)
4.5. Lipoic and dihydrolipoic acids
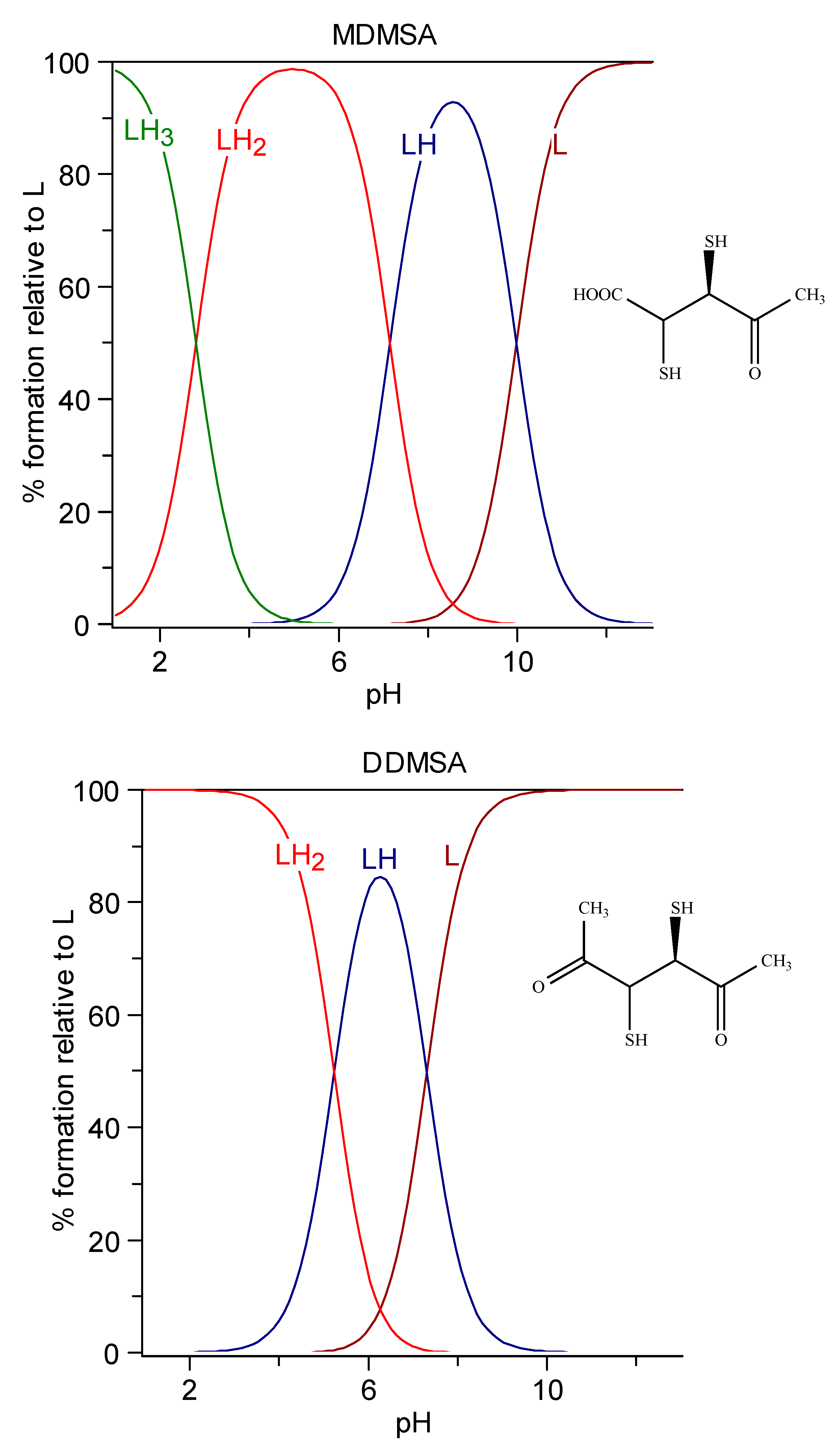
4.6. MiADMSA (monoisoamyl 2, 3-dimercaptosuccinic acid)
4.7. Thiocarbamates (Diethyldithiocarbamate and Derivatives)
5. Combination of Chelating Agents
6. Chemical Features of BAL, DMSA, DMPS, Penicillamine, Lipoic Acid, Dihydrolipoic Acid, and their Metal Chelates
6.1. Protonation of the Thiolate Anions
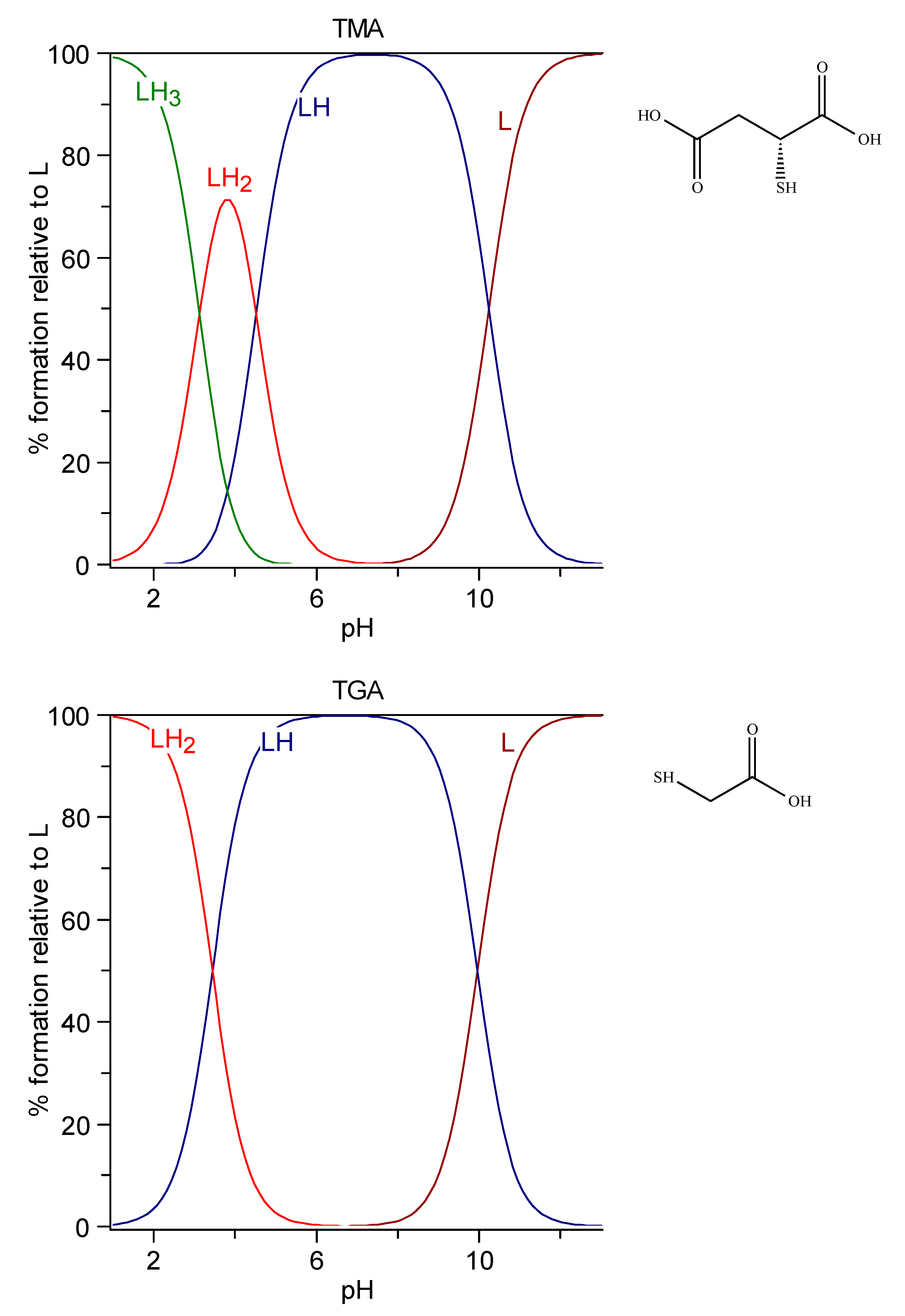
6.1.1. TGA and TMA
6.1.2. BAL
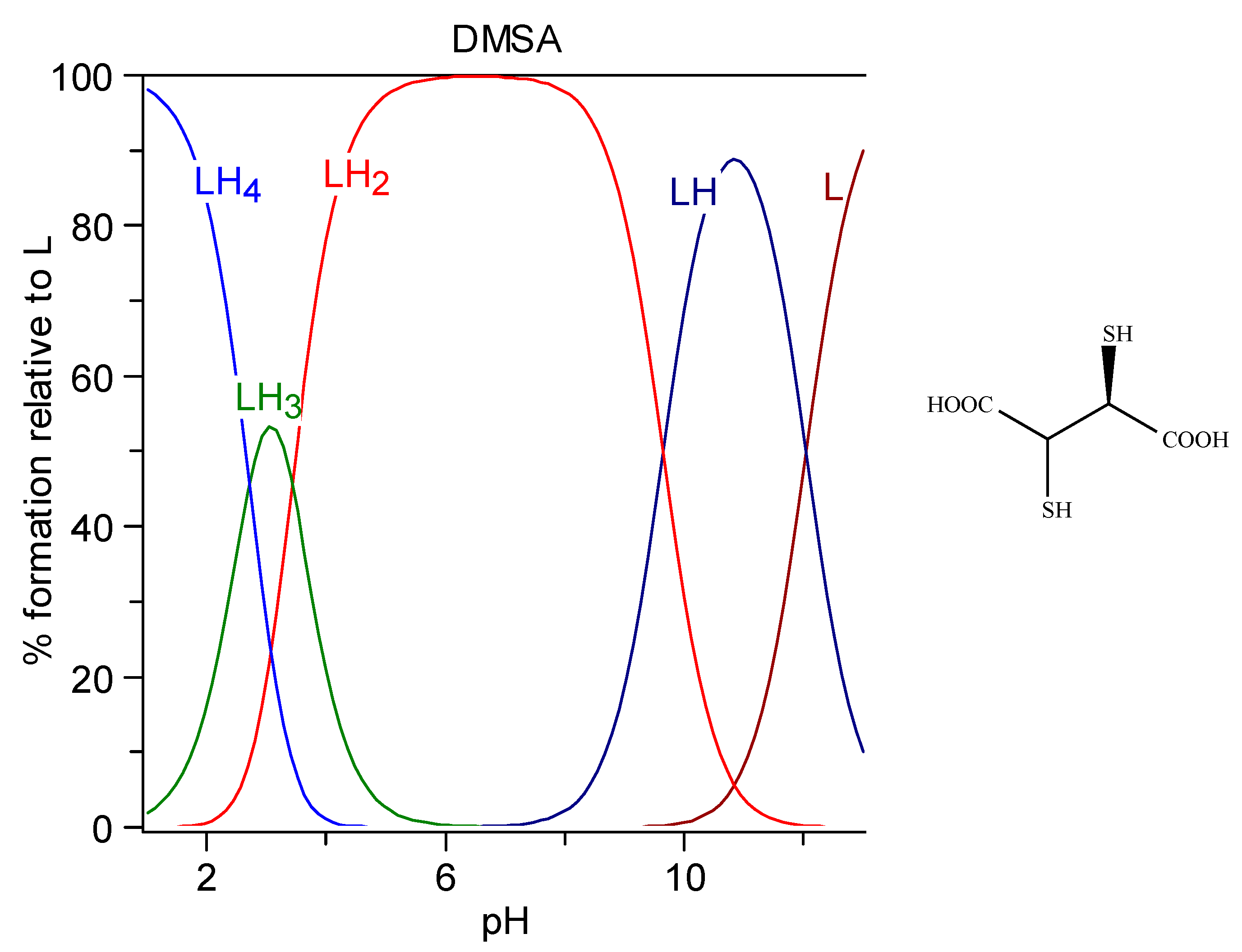
6.1.3. DMSA
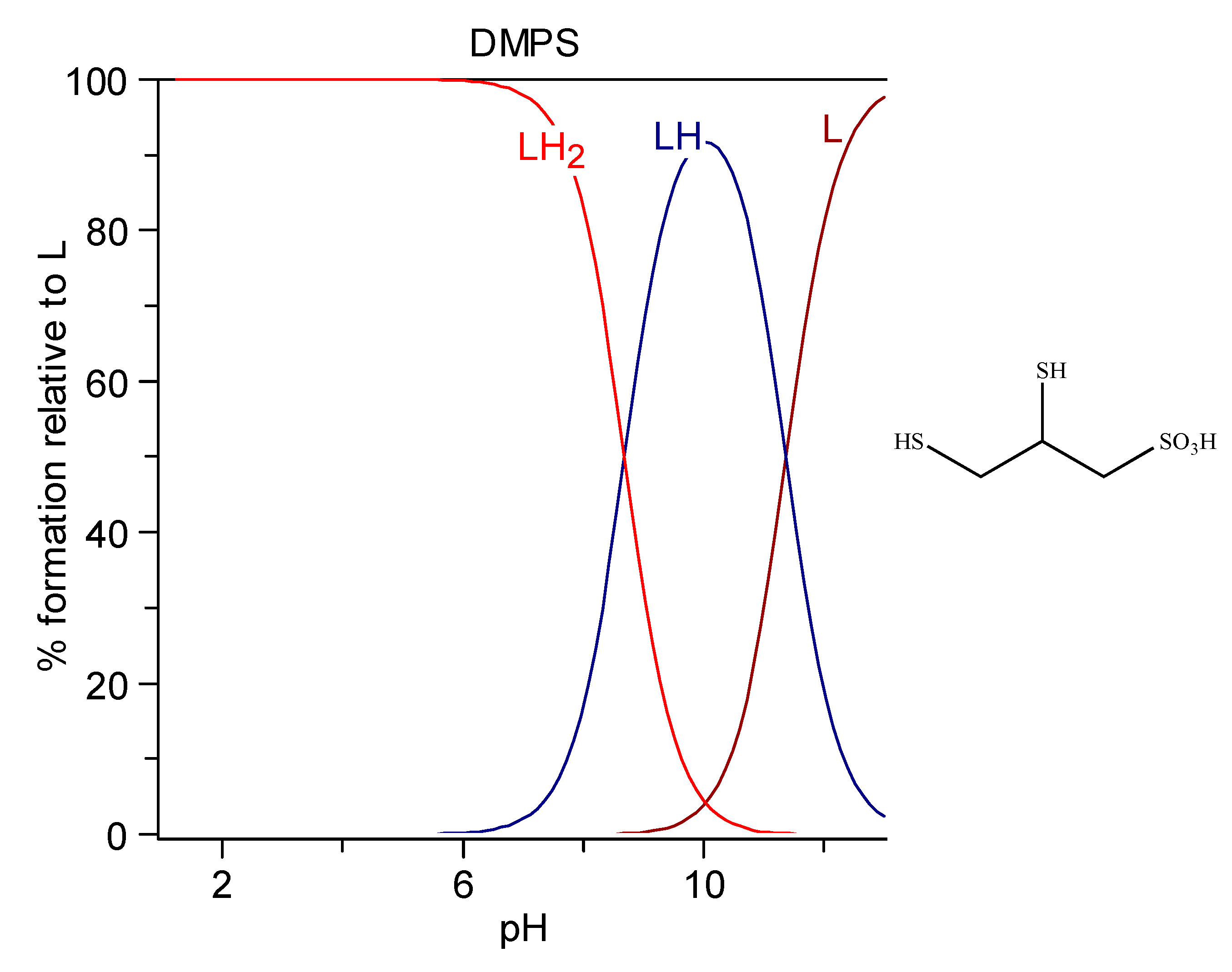
6.1.4. DMPS
6.1.5. d-penicillamine
6.1.6. Lipoic and dihydrolipoic acids
6.2. Complex Formation Reactions between Hg2+, Cd2+, and Pb2+ and Thiol Chelating Agents
7. Conclusions
Supplementary Materials
Funding
Acknowledgments
Conflicts of Interest
References
- ATSDR. ATSDR Substance Priority List; ATSDR: Atlanta, GA, USA, 2017. [Google Scholar]
- World Health Organisation. Ten Chemicals of Major Public Health Concern; WHO: Geneva, Switzerland, 2010; pp. 1–4. [Google Scholar]
- Pearson, R.G. Hard and Soft Acids and Bases. J. Am. Chem. Soc. 1963, 85, 3533–3539. [Google Scholar] [CrossRef]
- Aaseth, J.; Skaug, M.A.; Cao, Y.; Andersen, O. Chelation in metal intoxication-Principles and paradigms. J. Trace Elem. Med. Biol. 2015, 31, 260–266. [Google Scholar] [CrossRef]
- Clarkson, T.W. The toxicology of mercury. Crit. Rev. Clin. Lab. Sci. 1997, 34, 369–403. [Google Scholar] [CrossRef] [PubMed]
- Bjørklund, G.; Dadar, M.; Mutter, J.; Aaseth, J. The toxicology of mercury: Current research and emerging trends. Environ. Res. 2017, 159, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Maqbool, F.; Niaz, K.; Hassan, F.I.; Khan, F.; Abdollahi, M. Immunotoxicity of mercury: Pathological and toxicological effects. J. Environ. Sci. Health Part C 2017, 35, 29–46. [Google Scholar] [CrossRef] [PubMed]
- Branco, V.; Caito, S.; Farina, M.; Teixeira da Rocha, J.; Aschner, M.; Carvalho, C. Biomarkers of mercury toxicity: Past, present, and future trends. J. Toxicol. Environ. Health Part B 2017, 20, 119–154. [Google Scholar] [CrossRef]
- Scott, R.A. (Ed.) Encyclopedia of Inorganic and Bioinorganic Chemistry; John Wiley and Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Nordberg, G.F.; Fowler, B.A.; Nordberg, M. Handbook on the Toxicology of Metals: Fourth Edition; Academic Press: Cambridge, MA, USA, 2014; ISBN 9780123973399. [Google Scholar]
- Bose-O’Reilly, S.; McCarty, K.M.; Steckling, N.; Lettmeier, B. Mercury exposure and children’s health. Curr. Probl. Pediatric Adolesc. Health Care 2010, 40, 186–215. [Google Scholar] [CrossRef]
- Selin, N.E. Global Biogeochemical Cycling of Mercury: A Review Noelle Eckley Selin. Annu. Rev. Environ. Resour. 2009, 34, 43–63. [Google Scholar] [CrossRef]
- UNEP. Global Mercury Assessment 2013: Sources, Emissions, Releases, and Environmental Transport; UNEP Chemicals Branch: Geneva, Switzerland, 2013. [Google Scholar]
- Bjørklund, G.; Lindh, U.; Aaseth, J.; Mutter, J.; Chirumbolo, S. Mercury in dental amalgams: A great concern for clinical toxicology in developing countries? J. Trace Elem. Med. Biol. 2019, 51, 9–11. [Google Scholar] [CrossRef]
- Rice, K.M.; Walker, E.M.; Wu, M.; Gillette, C.; Blough, E.R. Environmental mercury and its toxic effects. J. Prev. Med. Public Heal. 2014, 47, 74–83. [Google Scholar] [CrossRef]
- Bjørklund, G. The history of dental amalgam. Tidsskr. Nor. Laegeforen. 1989, 109, 3582–3585. (In Norwegian) [Google Scholar]
- Risher, J. Toxicological Profile for Mercury. In ATSDR’s Toxicological Profiles; ATSDR: Atlanta, GA, USA, 2010. [Google Scholar]
- Rooney, J.P.K. The retention time of inorganic mercury in the brain—A systematic review of the evidence. Toxicol. Appl. Pharmacol. 2014, 274, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Pendergrass, J.C.; Haley, B.E.; Vimy, M.J.; Winfield, S.A.; Lorscheider, F.L. Mercury vapor inhalation inhibits binding of GTP to tubulin in rat brain: Similarity to a molecular lesion in Alzheimer diseased brain. Neurotoxicology 1997, 18, 315–324. [Google Scholar] [PubMed]
- Bjørklund, G.; Tinkov, A.A.; Dadar, M.; Rahman, M.M.; Chirumbolo, S.; Skalny, A.V.; Skalnaya, M.G.; Haley, B.E.; Ajsuvakova, O.P.; Aaseth, J. Insights into the Potential Role of Mercury in Alzheimer’s Disease. J. Mol. Neurosci. 2019, 67, 511–533. [Google Scholar] [CrossRef] [PubMed]
- Ask, K.; Åkesson, A.; Berglund, M.; Vahter, M. Inorganic mercury and methylmercury in placentas of Swedish women. Environ. Health Perspect. 2002, 110, 523–526. [Google Scholar] [CrossRef]
- Björnberg, K.A.; Vahter, M.; Petersson-Grawé, K.; Glynn, A.; Cnattingius, S.; Darnerud, P.O.; Atuma, S.; Aune, M.; Becker, W.; Berglund, M. Methyl mercury and inorganic mercury in Swedish pregnant women and in cord blood: Influence of fish consumption. Environ. Health Perspect. 2003, 111, 637–641. [Google Scholar]
- Aaseth, J.; Hilt, B.; Bjørklund, G. Mercury exposure and health impacts in dental personnel. Environ. Res. 2018, 164, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Bjørklund, G.; Hilt, B.; Dadar, M.; Lindh, U.; Aaseth, J. Neurotoxic effects of mercury exposure in dental personnel. Basic Clin. Pharmacol. Toxicol. 2019, 124, 568–574. [Google Scholar] [CrossRef]
- Mathiesen, T.; Ellingsen, D.G.; Kjuus, H. Neuropsychological effects associated with exposure to mercury vapor among former chloralkali workers. Scand. J. Work Environ. Health 1999, 25, 342–350. [Google Scholar] [CrossRef]
- Albers, J.W.; Kallenbach, L.R.; Fine, L.J.; Langolf, G.D.; Wolfe, R.A.; Donofrio, P.D.; Alessi, A.G.; Stolp-Smith, K.A.; Bromberg, M.B. Neurological abnormalities associated with remote occupational elemental mercury exposure. Ann. Neurol. 1988, 24, 651–659. [Google Scholar] [CrossRef]
- Letz, R.; Gerr, F.; Cragle, D.; Green, R.; Watkins, J.; Fidler, A. Residual neurologic deficits 30 years after occupational exposure to elemental mercury. Neurotoxicology 2000, 21, 459–474. [Google Scholar] [PubMed]
- Clarkson, T.W.; Magos, L. The toxicology of mercury and its chemical compounds. Crit. Rev. Toxicol. 2006, 36, 609–662. [Google Scholar] [CrossRef] [PubMed]
- International Programme on Chemical Safety (IPCS). Inorganic Mercury, Environmental Health Criteria 118; World Health Organization: Geneva, Switzerland, 1991. [Google Scholar]
- Brooks, W.E. Industrial Use of Mercury in the Ancient World. In Mercury in the Environment; USGS: Reston, VA, USA, 2012. [Google Scholar]
- Buckell, M.; Hunter, D.; Milton, R.; Perry, K.M. Chronic mercury poisoning. Br. J. Ind. Med. 1993, 50, 97–106. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Park, J.D.; Zheng, W. Human exposure and health effects of inorganic and elemental mercury. J. Prev. Med. Public Health 2012, 45, 344–352. [Google Scholar] [CrossRef] [PubMed]
- WHO. Exposure to Mercury: A Major Public Health Concern; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Likens, G.E. Biogeochemistry of Inland Waters; Academic Press: Cambridge, MA, USA, 2010; ISBN 9780123819963. [Google Scholar]
- Grandjean, P.; Satoh, H.; Murata, K.; Eto, K. Adverse effects of methylmercury: Environmental health research implications. Environ. Health Perspect. 2010, 118, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Harada, M. Minamata Disease: Methylmercury Poisoning in Japan Caused by Environmental Pollution. Crit. Rev. Toxicol. 1995, 25, 1–24. [Google Scholar] [CrossRef]
- Hinton, J.; Veiga, M. Mercury contaminated sites: A review of remedial solutions. In Proceedings of the National Institute for Minamata Disease, Kumamoto, Japan, 1 March 2001. [Google Scholar]
- Amin Zaki, L.; Elhassani, S.; Majeed, M.A.; Clarkson, T.W.; Doherty, R.A.; Greenwood, M.R.; Giovanoli-Jakubczak, T. Perinatal Methylmercury Poisoning in Iraq. Am. J. Dis. Child. 1976, 130, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, T.W.; Vyas, J.B.; Ballatori, N. Mechanisms of mercury disposition in the body. Am. J. Ind. Med. 2007, 50, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Fernandes Azevedo, B.; Barros Furieri, L.; Peçanha, F.M.; Wiggers, G.A.; Frizera Vassallo, P.; Ronacher Simões, M.; Fiorim, J.; Rossi de Batista, P.; Fioresi, M.; Rossoni, L.; et al. Toxic Effects of Mercury on the Cardiovascular and Central Nervous Systems. J. Biomed. Biotechnol. 2012. [Google Scholar] [CrossRef]
- Cariccio, V.L.; Samà, A.; Bramanti, P.; Mazzon, E. Mercury Involvement in Neuronal Damage and in Neurodegenerative Diseases. Biol. Trace Elem. Res. 2019, 187, 341–356. [Google Scholar] [CrossRef] [PubMed]
- Vejrup, K.; Brandlistuen, R.E.; Brantsæter, A.L.; Knutsen, H.K.; Caspersen, I.H.; Alexander, J.; Lundh, T.; Meltzer, H.M.; Magnus, P.; Haugen, M. Prenatal mercury exposure, maternal seafood consumption and associations with child language at five years. Environ. Int. 2018, 110, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Bjørklund, G.; Skalny, A.V.; Rahman, M.M.; Dadar, M.; Yassa, H.A.; Aaseth, J.; Chirumbolo, S.; Skalnaya, M.G.; Tinkov, A.A. Toxic metal (loid)-based pollutants and their possible role in autism spectrum disorder. Environ. Res. 2018, 166, 234–250. [Google Scholar] [CrossRef] [PubMed]
- IARC. IARC Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Man; IARC: Lyon, France, 1993. [Google Scholar]
- Remelli, M.; Nurchi, V.M.; Lachowicz, J.I.; Medici, S.; Zoroddu, M.A.; Peana, M. Competition between Cd (II) and other divalent transition metal ions during complex formation with amino acids, peptides, and chelating agents. Coord. Chem. Rev. 2016, 327–328, 55–69. [Google Scholar] [CrossRef]
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Painting, Firefighting, and Shiftwork; IARC: Lyon, France, 2010; Volume 100. [Google Scholar]
- EFSA. Cadmium in food-Scientific opinion of the Panel on Contaminants in the Food Chain: Cadmium in food-Scientific opinion of the Panel on Contaminants in the Food Chain. EFSA J. 2009, 7, 980. [Google Scholar]
- WHO. Exposure to Cadmium: A Major Public Health Concern; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- Crea, F.; Foti, C.; Milea, D.; Sammartano, S. Speciation of Cadmium in the Environment. In Cadmium: From Toxicity to Essentiality; Sigel, A., Sigel, H., Sigel, R.K.O., Eds.; Springer Science & Business Media: Dordrecht, The Netherlands, 2013; Chapter 3; Volume 11, pp. 63–83. [Google Scholar]
- Järup, L.; Åkesson, A. Current status of cadmium as an environmental health problem. Toxicol. Appl. Pharmacol. 2009, 238, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S. Dietary Cadmium Intake and Its Effects on Kidneys. Toxics 2018, 6, 15. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Current health risk assessment practice for dietary cadmium: Data from different countries. Food Chem. Toxicol. 2017, 106, 430–445. [Google Scholar] [CrossRef]
- Olsson, I.M.; Bensryd, I.; Lundh, T.; Ottosson, H.; Skerfving, S.; Oskarsson, A. Cadmium in blood and urine —Impact of sex, age, dietary intake, iron status, and former smoking—Association of renal effects. Environ. Health Perspect. 2002, 110, 1185–1190. [Google Scholar] [CrossRef]
- FAO/WHO; Food and Agriculture Organization of the United Nations; World Health Organization. Seventy-third Meeting, Geneva, 8–17 June 2010. Summary and Conclusions. JECFA/73/SC. 2010. Available online: http://www.who.int/foodsafety/publications/chem/summary73.pdf (accessed on 28 July 2019).
- Mortensen, M.E.; Wong, L.Y.; Osterloh, J.D. Smoking status and urine cadmium above levels associated with subclinical renal effects in U.S. adults without chronic kidney disease. Int. J. Hyg. Environ. Health 2011, 214, 305–310. [Google Scholar] [CrossRef]
- Zhang, H.; Reynolds, M. Cadmium exposure in living organisms: A short review. Sci. Total Environ. 2019, 678, 761–767. [Google Scholar] [CrossRef]
- Aoshima, K. Itai-itai disease: Renal tubular osteomalacia induced by environmental exposure to cadmium—Historical review and perspectives. Soil Sci. Plant Nutr. 2016, 62, 319–326. [Google Scholar] [CrossRef]
- Suwazono, Y.; Kido, T.; Nakagawa, H.; Nishijo, M.; Honda, R.; Kobayashi, E.; Dochi, M.; Nogawa, K. Biological half-life of cadmium in the urine of inhabitants after cessation of cadmium exposure. Biomarkers 2009, 14, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Fransson, M.N.; Barregard, L.; Sallsten, G.; Akerstrom, M.; Johanson, G. Physiologically-based toxicokinetic model for cadmium using markov-chain monte carlo analysis of concentrations in blood, urine, and kidney cortex from living kidney donors. Toxicol. Sci. 2014, 141, 365–376. [Google Scholar] [CrossRef] [PubMed]
- Mezynska, M.; Brzóska, M.M. Environmental exposure to cadmium—A risk for health of the general population in industrialized countries and preventive strategies. Environ. Sci. Pollut. Res. 2018, 25, 3211–3232. [Google Scholar] [CrossRef] [PubMed]
- Hyder, O.; Chung, M.; Cosgrove, D.; Herman, J.M.; Li, Z.; Firoozmand, A.; Gurakar, A.; Koteish, A.; Pawlik, T.M. Cadmium Exposure and Liver Disease among US Adults. J. Gastrointest. Surg. 2013, 17, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- James, K.A.; Meliker, J.R. Environmental cadmium exposure and osteoporosis: A review. Int. J. Public Health 2013, 58, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Deering, K.E.; Callan, A.C.; Prince, R.L.; Lim, W.H.; Thompson, P.L.; Lewis, J.R.; Hinwood, A.L.; Devine, A. Low-level cadmium exposure and cardiovascular outcomes in elderly Australian women: A cohort study. Int. J. Hyg. Environ. Health 2018, 221, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.X.; Sun, Y.; Feng, W.; Wang, P.; Yang, P.; Li, J.; Huang, Z.; Chen, Y.J.; Liu, C.; Sun, L.; et al. Association of urinary metal levels with human semen quality: A cross-sectional study in China. Environ. Int. 2016, 91, 51–59. [Google Scholar] [CrossRef]
- Silva, N.; Peiris-John, R.; Wickremasinghe, R.; Senanayake, H.; Sathiakumar, N. Cadmium a metalloestrogen: Are we convinced? J. Appl. Toxicol. 2012, 32, 318–332. [Google Scholar] [CrossRef]
- Chen, X.; Zhu, G.; Jin, T. Effects of Cadmium Exposure on Age of Menarche and Menopause. Toxics 2017, 6, 6. [Google Scholar] [CrossRef]
- Buha, A.; Matovic, V.; Antonijevic, B.; Bulat, Z.; Curcic, M.; Renieri, E.A.; Tsatsakis, A.M.; Schweitzer, A.; Wallace, D. Overview of cadmium thyroid disrupting effects and mechanisms. Int. J. Mol. Sci. 2018, 19, 1501. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Xia, W.; Zhang, B.; Zhou, A.; Huang, Z.; Zhang, H.; Liu, H.; Jiang, Y.; Hu, C.; Chen, X.; et al. Relation between cadmium exposure and gestational diabetes mellitus. Environ. Int. 2018, 113, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Jacquet, A.; Ounnas, F.; Lénon, M.; Arnaud, J.; Demeilliers, C.; Moulis, J.-M. Chronic Exposure to Low-Level Cadmium in Diabetes: Role of Oxidative Stress and Comparison with Polychlorinated Biphenyls. Curr. Drug Targets 2016, 17, 1385–1413. [Google Scholar] [CrossRef] [PubMed]
- Renieri, E.A.; Sfakianakis, D.G.; Alegakis, A.A.; Safenkova, I.V.; Buha, A.; Matović, V.; Tzardi, M.; Dzantiev, B.B.; Divanach, P.; Kentouri, M.; et al. Nonlinear responses to waterborne cadmium exposure in zebrafish. An in vivo study. Environ. Res. 2017, 157, 173–181. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). IARC Cadmium and Cadmium Compounds; 100C ed.; WHO Press: Lyon, France, 2012; Volume 100C. [Google Scholar]
- Buha, A.; Wallace, D.; Matovic, V.; Schweitzer, A.; Oluic, B.; Micic, D.; Djordjevic, V. Cadmium Exposure as a Putative Risk Factor for the Development of Pancreatic Cancer: Three Different Lines of Evidence. BioMed Res. Int. 2017, 2017, 1981837. [Google Scholar] [CrossRef] [PubMed]
- Djordjevic, V.R.; Wallace, D.R.; Schweitzer, A.; Boricic, N.; Knezevic, D.; Matic, S.; Grubor, N.; Kerkez, M.; Radenkovic, D.; Bulat, Z.; et al. Environmental cadmium exposure and pancreatic cancer: Evidence from case control, animal and in vitro studies. Environ. Int. 2019, 128, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Feki-Tounsi, M.; Hamza-Chaffai, A. Cadmium as a possible cause of bladder cancer: A review of accumulated evidence. Environ. Sci. Pollut. Res. 2014, 21, 10561–10573. [Google Scholar] [CrossRef]
- Vinceti, M.; Venturelli, M.; Sighinolfi, C.; Trerotoli, P.; Bonvicini, F.; Ferrari, A.; Bianchi, G.; Serio, G.; Bergomi, M.; Vivoli, G. Case-control study of toenail cadmium and prostate cancer risk in Italy. Sci. Total Environ. 2007, 373, 77–81. [Google Scholar] [CrossRef]
- Van Maele-Fabry, G.; Lombaert, N.; Lison, D. Dietary exposure to cadmium and risk of breast cancer in postmenopausal women: A systematic review and meta-analysis. Environ. Int. 2016, 86, 1–13. [Google Scholar] [CrossRef]
- Matović, V.; Buha, A.; Dukić-Ćosić, D.; Bulat, Z. Insight into the oxidative stress induced by lead and/or cadmium in blood, liver and kidneys. Food Chem. Toxicol. 2015, 78, 130–140. [Google Scholar] [CrossRef]
- Matović, V.; Buha, A.; Bulat, Z.; Đukić-Ćosić, D. Cadmium Toxicity Revisited: Focus on Oxidative Stress Induction and Interactions with Zinc and Magnesium. Arch. Ind. Hyg. Toxicol. 2011, 62, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Bulat, Z.; Dukić-Ćosić, D.; Antonijević, B.; Buha, A.; Bulat, P.; Pavlović, Z.; Matović, V. Can zinc supplementation ameliorate cadmium-induced alterations in the bioelement content in rabbits? Arh. Hig. Rada Toksikol. 2017, 68, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Bulat, Z.; Đukić-Ćosić, D.; Antonijević, B.; Bulat, P.; Vujanović, D.; Buha, A.; Matović, V. Effect of Magnesium Supplementation on the Distribution Patterns of Zinc, Copper, and Magnesium in Rabbits Exposed to Prolonged Cadmium Intoxication. Sci. World J. 2012, 2012, 572514. [Google Scholar] [CrossRef] [PubMed]
- Buha, A.; Bulat, Z.; Dukic-Cosic, D.; Matovic, V. Effects of oral and intraperitoneal magnesium treatment against cadmium-induced oxidative stress in plasma of rats. Arch. Ind. Hyg. Toxicol. 2012, 63, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Buha, A.; Jugdaohsingh, R.; Matovic, V.; Bulat, Z.; Antonijevic, B.; Kerns, J.G.; Goodship, A.; Hart, A.; Powell, J.J. Bone mineral health is sensitively related to environmental cadmium exposure-experimental and human data. Environ. Res. 2019, 176, 108539. [Google Scholar] [CrossRef] [PubMed]
- Wallace, D.; Spandidos, D.; Tsatsakis, A.; Schweitzer, A.; Djordjevic, V.; Djordjevic, A. Potential interaction of cadmium chloride with pancreatic mitochondria: Implications for pancreatic cancer. Int. J. Mol. Med. 2019, 44, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Fay, M.; Alt, L.; Ryba, D.; Salamah, R.; Peach, R.; Papaeliou, A.; Zawadzka, S.; Weiss, A.; Patel, N.; Rahman, A.; et al. Cadmium Nephrotoxicity Is Associated with Altered MicroRNA Expression in the Rat Renal Cortex. Toxics 2018, 6, 16. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, N.C.; Hatzidaki, E.G.; Belivanis, S.; Tzanakakis, G.N.; Tsatsakis, A.M. Lead toxicity update. A brief review. Med. Sci. Monit. 2005, 11, 329–336. [Google Scholar]
- Patrick, L. Lead toxicity, a review of the literature. Part I: Exposure, evaluation, and treatment. Altern. Med. Rev. 2006, 11, 2–22. [Google Scholar]
- Oudijk, G. The rise and fall of organometallic additives in automotive gasoline. Environ. Forensics 2010, 11, 17–49. [Google Scholar] [CrossRef]
- Maas, R.P.; Patch, S.C.; Morgan, D.M.; Pandolfo, T.J. Reducing lead exposure from drinking water: Recent history and current status. Public Health Rep. 2005, 120, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Casas, S.J.; Sordo, J. Lead Chemistry, Analytical Aspects, Environmental Impact and Health Effects; Elsevier: Amsterdam, The Netherlands, 2006; ISBN 9780444529459. [Google Scholar]
- EFSA (European Food Safety Authority). Scientific Opinion on Lead in Food. EFSA J. 2010, 8, 1570. [Google Scholar] [CrossRef]
- Laidlaw, M.A.S.; Filippelli, G.M.; Sadler, R.C.; Gonzales, C.R.; Ball, A.S.; Mielke, H.W. Children’s blood lead seasonality in flint, Michigan (USA), and soil-sourced lead hazard risks. Int. J. Environ. Res. Public Health 2016, 13, 358. [Google Scholar] [CrossRef]
- Safruk, A.M.; McGregor, E.; Whitfield Aslund, M.L.; Cheung, P.H.; Pinsent, C.; Jackson, B.J.; Hair, A.T.; Lee, M.; Sigal, E.A. The influence of lead content in drinking water, household dust, soil, and paint on blood lead levels of children in Flin Flon, Manitoba and Creighton, Saskatchewan. Sci. Total Environ. 2017, 593–594, 202–210. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, D.; Hou, D.; Ye, J.; Zhang, Y.; Ok, Y.S.; Song, Y.; Coulon, F.; Peng, T.; Tian, L. Lead-based paint remains a major public health concern: A critical review of global production, trade, use, exposure, health risk, and implications. Environ. Int. 2018, 121, 85–101. [Google Scholar] [CrossRef] [PubMed]
- Philip, A.T.; Gerson, B. Lead poisoning—Part I. Incidence, etiology, and toxicokinetics. Clin. Lab. Med. 1994, 14, 423–444. [Google Scholar] [CrossRef]
- Markowitz, M. Lead poisoning: A disease for the next millennium. Curr. Probl. Pediatr. 2000, 30, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Gulson, B.L.; Mizon, K.J.; Korsch, M.J.; Palmer, J.M.; Donnelly, J.B. Mobilization of lead from human bone tissue during pregnancy and lactation—A summary of long-term research. Sci. Total Environ. 2003, 303, 79–104. [Google Scholar] [CrossRef]
- Abadin, H.; Ashizawa, A.; Llados, F.; Stevens, Y.W. Toxicological Profile for Lead. In ATSDR’s Toxicological Profiles; ATSDR: Atlanta, GA, USA, 2010. [Google Scholar]
- Bjørklund, G.; Dadar, M.; Chirumbolo, S.; Aaseth, J. High content of lead is associated with the softness of drinking water and raised cardiovascular morbidity: A review. Biol. Trace Elem. Res. 2018, 186, 384–394. [Google Scholar] [CrossRef] [PubMed]
- American Academy of Pediatrics Committee on Environmental Health. Lead exposure in children: Prevention, detection, and management; CCHMC guideline. Pediatrics 2005, 116, 1036. [Google Scholar] [CrossRef]
- Grandjean, P. Even low-dose lead exposure is hazardous. Lancet 2010, 376, 855–856. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Inorganic and Organic Lead Compounds. IARC Monogr. Eval. Carcinog. Risks Hum. 2006, 87, 1. [Google Scholar]
- Andjelkovic, M.; Djordjevic, A.B.; Antonijevic, E.; Antonijevic, B.; Stanic, M.; Kotur-Stevuljevic, J.; Spasojevic-Kalimanovska, V.; Jovanovic, M.; Boricic, N.; Wallace, D.; et al. Toxic effect of acute cadmium and lead exposure in rat blood, liver, and kidney. Int. J. Environ. Res. Public Health 2019, 16, 274. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, Y.F.; Eldebaky, H.A.A.; Mahmoud, K.G.M.; Nawito, M. Effects of lead exposure on DNA damage and apoptosis in reproductive and vital organs in female rabbits. Glob. Vet. 2012, 9, 401–408. [Google Scholar]
- Margoshes, M.; Valiee, B.L. A Cadmium Protein from Equine Kidney Cortex. J. Am. Chem. Soc. 1957, 79, 4813–4814. [Google Scholar] [CrossRef]
- Felizola, S.J.A.; Nakamura, Y.; Arata, Y.; Ise, K.; Satoh, F.; Rainey, W.E.; Midorikawa, S.; Suzuki, S.; Sasano, H. Metallothionein-3 (MT-3) in the Human Adrenal Cortex and its Disorders. Endocr. Pathol. 2013, 25, 229–235. [Google Scholar] [CrossRef]
- Coyle, P.; Philcox, J.C.; Carey, L.C.; Rofe, A.M. Metallothionein: The multipurpose protein. Cell. Mol. Life Sci. 2002, 59, 627–647. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Koh, J.Y. Roles of zinc and metallothionein-3 in oxidative stress-induced lysosomal dysfunction, cell death, and autophagy in neurons and astrocytes. Mol. Brain 2010, 3, 30. [Google Scholar] [CrossRef]
- Kumari, M.V.R.; Hiramatsu, M.; Ebadi, M. Free radical scavenging actions of metallothionein isoforms I and II. Free Radic. Res. 1998, 29, 93–101. [Google Scholar] [CrossRef]
- Bolognin, S.; Cozzi, B.; Zambenedetti, P.; Zatta, P. Metallothioneins and the central nervous system: From a deregulation in neurodegenerative diseases to the development of new therapeutic approaches. J. Alzheimer’s Dis. 2014, 41, 29–42. [Google Scholar] [CrossRef]
- Couto, N.; Malys, N.; Gaskell, S.J.; Barber, J. Partition and turnover of glutathione reductase from saccharomyces cerevisiae: A proteomic approach. J. Proteome Res. 2013, 12, 2885–2894. [Google Scholar] [CrossRef] [PubMed]
- Ballatori, N.; Clarkson, T.W. Sulfobromophthalein inhibition of glutathione and methylmercury secretion into bile. Am. J. Physiol. Liver Physiol. 2017, 248, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Aaseth, J. Biliary excretion of copper and zinc in the rat as influenced by diethylmaleate, selenite and diethyldithiocarbamate. Biochem. Pharmacol. 1980, 29, 2129–2133. [Google Scholar] [CrossRef]
- Cigala, R.M.; Crea, F.; De Stefano, C.; Lando, G.; Milea, D.; Sammartano, S. Modeling the acid-base properties of glutathione in different ionic media, with particular reference to natural waters and biological fluids. Amino Acids 2013, 43, 629–648. [Google Scholar] [CrossRef] [PubMed]
- Morgan, G.T.; Drew, H.D.K. CLXII.—Researches on residual affinity and co-ordination. Part II. Acetylacetones of selenium and tellurium. J. Chem. Soc. Trans. 1920, 117, 1456–1465. [Google Scholar] [CrossRef]
- Aaseth, J. Recent Advance in the Therapy of Metal Poisonings with Chelating Agents. Hum. Exp. Toxicol. 1983, 2, 257–272. [Google Scholar] [CrossRef]
- Aaseth, J.; Crisponi, G.; Anderson, O. Chelation Therapy in the Treatment of Metal Intoxication; Academic Press: Cambridge, MA, USA, 2016; ISBN 9780128030721. [Google Scholar]
- Crisponi, G.; M Nurchi, V.; Crespo-Alonso, M.; Toso, L. Chelating Agents for Metal Intoxication. Curr. Med. Chem. 2012, 7, 2745–2788. [Google Scholar]
- Aaseth, J.; Ajsuvakova, O.P.; Skalny, A.V.; Skalnaya, M.G.; Tinkov, A.A. Chelator combination as therapeutic strategy in mercury and lead poisonings. Coord. Chem. Rev. 2018, 358, 1–12. [Google Scholar] [CrossRef]
- Voegtlin, C.; Smith, H.W.; Crane, M.M.; Wright, K.D.; Connell, M.A. Quantitative Studies in Chemotherapy: I. The Trypanocidal Action of Arsenic and Antimony Compounds. J. Pharmacol. Exp. Ther. 1920, 15, 453–473. [Google Scholar]
- Kety, S.S.; Letonoff, T.V. Treatment of lead poisoning by sodium citrate. Amer. J.Med. Sci. 1943, 205, 406–414. [Google Scholar] [CrossRef]
- Peters, R.A. Development and theortical significance of british anti-lewisite (bal). Br. Med. Bull. 1948, 5, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.D. Lead poisoning in children and its therapy with EDTA. Ind. Med. Surg. 1959, 28, 148–151. [Google Scholar] [PubMed]
- Ding, G.-S.; Liang, Y.-Y. Antidotal effects of dimercaptosuccinic acid. J. Appl. Toxicol. 1991, 11, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Oginski, M. Use of unitiol for speeding up renal excretion of Chlormerodrin203Hg. Int. Urol. Nephrol. 1971, 3, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Aposhian, H.V.; Maiorino, R.M.; Gonzalez-Ramirez, D.; Zuniga-Charles, M.; Xu, Z.; Hurlbut, K.M.; Junco-Munoz, P.; Dart, R.C.; Aposhian, M.M. Mobilization of heavy metals by newer, therapeutically useful chelating agents. Toxicology 1995, 97, 23–38. [Google Scholar] [CrossRef]
- Luetscher, J.A.; Eagle, H.; Long-cope, W.T. Clinical uses of 2,3-dimercaptopropanol (BAL); the effect of BAL on the excretion of arsenic in arsenical intoxication. J. Clin. Investig. 1946, 25, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Longcope, W.T.; Luetscher, J.A. Clinical uses of 2,3-dimercaptopropanol (BAL); the treatment of acute mercury poisoning by BAL. J. Clin. Investig. 1946, 25, 557–567. [Google Scholar] [CrossRef] [PubMed]
- Berlin, M.; Ullrebg, S. Increased uptake of mercury in mouse brain caused by 2,3-dimercaptopropanol. Nature 1963, 197, 84–85. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Skaug, M.A.; Andersen, O.; Aaseth, J. Chelation therapy in intoxications with mercury, lead and copper. J. Trace Elem. Med. Biol. 2015, 31, 188–192. [Google Scholar] [CrossRef]
- Andersen, O. Principles and recent developments in chelation treatment of metal intoxication. Chem. Rev. 1999, 99, 2683–2710. [Google Scholar] [CrossRef]
- Chisolm, J.J. The use of chelating agents in the treatment of acute and chronic lead intoxication in childhood. J. Pediatr. 1968, 73, 1–38. [Google Scholar] [CrossRef]
- Andersen, O.; Aaseth, J. Molecular mechanisms of in vivo metal chelation: Implications for clinical treatment of metal intoxications. Environ. Health Perspect. 2002, 110, 887–890. [Google Scholar] [CrossRef] [PubMed]
- Bjørklund, G.; Mutter, J.; Aaseth, J. Metal chelators and neurotoxicity: Lead, mercury, and arsenic. Arch. Toxicol. 2017, 91, 3787–3797. [Google Scholar] [CrossRef] [PubMed]
- Maiorino, R.M.; Akins, J.M.; Blaha, K.; Carter, D.E.; Aposhian, H.V. Determination and metabolism of dithiol chelating agents: X. In humans, meso-2,3-dimercaptosuccinic acid is bound to plasma proteins via mixed disulfide formation. J. Pharmacol. Exp. Ther. 1990, 254, 570–577. [Google Scholar] [PubMed]
- Dart, R.C.; Hurlbut, K.M.; Maiorino, R.M.; Mayersohn, M.; Aposhian, H.V.; Hassen, L.V.B. Pharmacokinetics of meso-2,3-dimercaptosuccinic acid in patients with lead poisoning and in healthy adults. J. Pediatr. 1994, 125, 309–316. [Google Scholar] [CrossRef]
- Sears, M.E. Chelation: Harnessing and Enhancing Heavy Metal Detoxification—A Review. Sci. World J. 2013, 2013, 219840. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.L. Dimercaptosuccinic acid (DMSA), a non-toxic, water-soluble treatment for heavy metal toxicity. Altern. Med. Rev. 1998, 3, 199–207. [Google Scholar]
- Aposhian, H.V.; Maiorino, R.M.; Dart, R.C.; Perry, D.F. Urinary excretion of meso-2,3-dimercaptosuccinic acid in human subjects. Clin. Pharmacol. Ther. 1989, 45, 520–526. [Google Scholar] [CrossRef]
- Asledu, P.; Moulton, T.; Blum, C.B.; Roldan, E.; Lolacono, N.J.; Graziano, J.H. Metabolism of meso-2,3-dimercaptosuccinic acid in lead-poisoned children and normal adults. Environ. Health Perspect. 1995, 103, 734–739. [Google Scholar]
- Bradberry, S.; Vale, A. Dimercaptosuccinic acid (succimer; DMSA) in inorganic lead poisoning. Clin. Toxicol. 2009, 47, 617–631. [Google Scholar] [CrossRef]
- Aaseth, J.; Jacobsen, D.; Andersen, O.; Wickstrøm, E. Treatment of mercury and lead poisonings with dimercaptosuccinic acid and sodium dimercaptopropanesulfonate: A review. Analyst 1995, 120, 853–854. [Google Scholar] [CrossRef] [PubMed]
- Aaseth, J.; Friedheim, E.A.H. Treatment of Methyl Mercury Poisoning in Mice with 2,3-dimercaptosuccinic Acid and other Complexing Thiols. Acta Pharmacol. Toxicol. 1978, 42, 248–252. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Guldager, B.; Larsen, I.B.; Jørgensen, P.J.; Holmstrup, P. Placebo response in environmental disease: Chelation therapy of patients with symptoms attributed to amalgam fillings. J. Occup. Environ. Med. 1997, 39, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Aposhian, H.V. DMSA and DMPS-Water Soluble Antidotes for Heavy Metal Poisoning. Annu. Rev. Pharmacol. Toxicol. 2003, 23, 193–215. [Google Scholar] [CrossRef] [PubMed]
- George, G.N.; Prince, R.C.; Gailer, J.; Buttigieg, G.A.; Denton, M.B.; Harris, H.H.; Pickering, I.J. Mercury binding to the chelation therapy agents DMSA and DMPS and the rational design of custom chelators for mercury. Chem. Res. Toxicol. 2004, 17, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Graziano, J.H. Role of 2,3-Dimercaptosuccinic Acid in the Treatment of Heavy Metal Poisoning. Med. Toxicol. 1986, 1, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, D.T.; Casteel, S.W.; Faggella, A.M.; Chastain, C.B.; Nunn, J.W.; Schaeffer, D.J. Use of orally administered succimer (meso-2,3-dimercaptosuccinic acid) for treatment of lead poisoning in dogs. J. Am. Vet. Med. Assoc. 1996, 208, 371–375. [Google Scholar]
- Roels, H.A.; Boeckx, M.; Ceulemans, E.; Lauwerys, R.R. Urinary excretion of mercury after occupational exposure to mercury vapour and influence of the chelating agent meso-2,3-dimercaptosuccinic acid (DMSA). Occup. Environ. Med. 2008, 48, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Gustavsson, P.; Gerhardsson, L. Intoxication from an accidentally ingested lead shot retained in the gastrointestinal tract. Environ. Health Perspect. 2005, 113, 491–493. [Google Scholar] [CrossRef]
- Flora, S.J.S.; Pachauri, V. Chelation in metal intoxication. Int. J. Environ. Res. Public Health 2010, 7, 2745–2788. [Google Scholar] [CrossRef]
- Gersl, V.; Hrdina, R.; Vavrova, J.; Holeckova, M.; Palicka, V.; Vogkova, J.; Mazurova, Y.; Bajgar, J. Effects of repeated administration of dithiol chelating agent-sodium 2,3-dimercapto 1-propanesulphonate (DMPS)-on biochemical and hematological parameters in rabbits. Acta. Medica. 1997, 40, 3–8. [Google Scholar] [PubMed]
- Hurlbut, K.M.; Maiorino, R.M.; Mayersohn, M.; Dart, R.C.; Bruce, D.C.; Aposhian, H.V. Determination and metabolism of dithiol chelating agents. XVI: Pharmacokinetics of 2,3-dimercapto-1-propanesulfonate after intravenous administration to human volunteers. J. Pharmacol. Exp. Ther. 1994, 268, 662–668. [Google Scholar]
- Gabard, B. Distribution and excretion of the mercury chelating agent sodium 2,3-dimercaptopropane-1-sulfonate in the rat. Arch. Toxicol. 1978, 39, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Torres-Alanís, O.; Garza-Ocañas, L.; Bernal, M.A.; Piñeyro-López, A. Urinary excretion of trace elements in humans after sodium 2,3-dimercaptopropane-1-sulfonate challenge test. J. Toxicol. Clin. Toxicol. 2000, 38, 697–700. [Google Scholar] [CrossRef]
- Walshe, J.M. Penicillamine, a new oral therapy for Wilson’s disease. Am. J. Med. 1956, 21, 487–495. [Google Scholar] [CrossRef]
- Rodriguez-Castro, K.I.; Hevia-Urrutia, F.J.; Sturniolo, G.C. Wilson’s disease: A review of what we have learned. World J. Hepatol. 2015, 7, 2859–2870. [Google Scholar] [CrossRef] [PubMed]
- Horn, N.; Møller, L.B.; Nurchi, V.M.; Aaseth, J. Chelating principles in Menkes and Wilson diseases: Choosing the right compounds in the right combinations at the right time. J. Inorg. Biochem. 2019, 190, 98–112. [Google Scholar] [CrossRef] [PubMed]
- Patrick, L. Mercury toxicity and antioxidants: Part I: Role of glutathione and alpha-lipoic acid in the treatment of mercury toxicity. Altern. Med. Rev. 2002, 7, 456–471. [Google Scholar] [PubMed]
- Bjørklund, G.; Aaseth, J.; Crisponi, G.; Rahman, M.M.; Chirumbolo, S. Insights on alpha lipoic and dihydrolipoic acids as promising scavengers of oxidative stress and possible chelators in mercury toxicology. J. Inorg. Biochem. 2019, 195, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Bakka, A.; Aaseth, J.; Rugstad, H.E. Influence of Certain Chelating Agents on Egress of Cadmium from Cultured Epithelial Cells Containing High Amounts of Metallothionein: A Screening of Cd-Releasing and Toxic Effects. Acta Pharmacol. Toxicol. 1981, 49, 432–437. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.M.; Singh, P.K.; Gale, G.R.; Smith, A.B.; Atkins, L.M. Cadmium Mobilization in Vivo by Intraperitoneal or Oral Administration of Monoalkyl Esters of meso–2, 3–Dmercaptosuccinic Acid in the Mouse. Pharmacol. Toxicol. 1992, 70, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Ram Kumar, M.; Flora, S.J.S.; Reddy, G.R. Monoisoamyl 2,3-dimercaptosuccinic acid attenuates arsenic induced toxicity: Behavioral and neurochemical approach. Environ. Toxicol. Pharmacol. 2013, 36, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Flora, S.J.S.; Bhadauria, S.; Pachauri, V.; Yadav, A. Monoisoamyl 2, 3-Dimercaptosuccinic Acid (MiADMSA) Demonstrates Higher Efficacy by Oral Route in Reversing Arsenic Toxicity: A Pharmacokinetic Approach. Basic Clin. Pharmacol. Toxicol. 2012, 110, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Kannan, G.M.; Flora, S.J.S. Combined administration of N-acetylcysteine and monoisoamyl DMSA on tissue oxidative stress during arsenic chelation therapy. Biol. Trace Elem. Res. 2006, 110, 43–59. [Google Scholar] [CrossRef]
- Gale, G.R.; Smith, A.B.; Walker, E.M. Diethyldithiocarbamate in treatment of acute cadmium poisoning. Ann. Clin. Lab. Sci. 1981, 11, 476–483. [Google Scholar] [PubMed]
- Aaseth, J.; Alexander, J.; Raknerud, N. Treatment of mercuric chloride poisoning with dimercaptosuccinic acid and diuretics: Preliminary studies. Clin. Toxicol. 1982, 19, 173–186. [Google Scholar] [CrossRef]
- Oskarsson, A.; Lind, B. Increased Lead Levels in Brain after Long-term Treatment with Lead and Dithiocarbamate or Thiuram Derivatives in Rats. Acta Pharmacol. Toxicol. 1985, 56, 309–315. [Google Scholar] [CrossRef]
- Kargačin, B.; Kostial, K.; Arežina, R.; Singh, P.K.; Jones, M.M.; Cikrt, M. Influence of age and time of administration of dithiocarbamate analogues on cadmium retention in rats. J. Appl. Toxicol. 1991, 11, 273–277. [Google Scholar] [CrossRef]
- Hamidinia, S.A.; Erdahl, W.L.; Chapman, C.J.; Steinbaugh, G.E.; Taylor, R.W.; Pfeiffer, D.R. Monensin improves the effectiveness of meso-dimercaptosuccinate when used to treat lead intoxication in rats. Environ. Health Perspect. 2006, 114, 484–493. [Google Scholar] [CrossRef]
- Chisolm, J.J. BAL, EDTA, DMSA and DMPS in the treatment of lead poisoning in children. Clin. Toxicol. 1992, 30, 493–504. [Google Scholar] [CrossRef]
- Lee, B.K.; Schwartz, B.S.; Stewart, W.; Ahn, K.D. Provocative chelation with DMSA and EDTA: Evidence for differential access to lead storage sites. Occup. Environ. Med. 1995, 52, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Besunder, J.B.; Super, D.M.; Anderson, R.L. Comparison of dimercaptosuccinic acid and calcium disodium ethylenediaminetetraacetic acid versus dimercaptopropanol and ethylenediaminetetraacetic acid in children with lead poisoning. J. Pediatr. 1997, 130, 966–971. [Google Scholar] [CrossRef]
- Dargan, P.I.; Giles, L.J.; Wallace, C.I.; House, I.M.; Thomson, A.H.; Beale, R.J.; Jones, A.L. Case report: Severe mercuric sulphate poisoning treated with 2,3-dimercaptopropane-1-sulphonate and haemodiafiltration. Crit. Care 2003, 7, R1–R6. [Google Scholar] [CrossRef]
- Aragoni, M.C.; Arca, M.; Crisponi, G.; Cristiani, F.; Isaia, F.; Nurchi, V.M. Characterization of the ionization and spectral properties of mercapto-carboxylic acids. Correlation with substituents and structural features. Talanta 1996, 43, 1357–1366. [Google Scholar]
- Arnold, A.P.; Canty, A.J.; Reid, R.S.; Rabenstein, D.L. Nuclear magnetic resonance and potentiometric studies of the complexation of methylmercury (II) by dithiols. Can. J. Chem. 2006, 63, 2430–2436. [Google Scholar] [CrossRef]
- Sallam, S.A.; Bahgat, K.M.; El-Tanany, A.Z.; Mahmoud, M.A. Lanthanide complexes of D-penicillamine: Formation constants, spectral and thermal properties. J. Coord. Chem. 2006, 59, 2055–2073. [Google Scholar] [CrossRef]
- Sigel, H.; Prijs, B.; McCormick, D.B.; Shih, J.C.H. Stability and structure of binary and ternary complexes of α-lipoate and lipoate derivatives with Mn2+, Cu2+, and Zn2+ in solution. Arch. Biochem. Biophys. 1978, 187, 208–214. [Google Scholar] [CrossRef]
- Bonomi, F.; Pagani, S.; Cariati, F.; Pozzi, A.; Crisponi, G.; Cristiani, F.; Diaz, A.; Zanoni, R. Synthesis and characterization of metal derivatives of dihydrolipoic acid and dihydrolipoamide. Inorg. Chim. Acta 1992, 192, 237–242. [Google Scholar] [CrossRef]
- The Merck Index, 12th ed.; Merck: New York, NY, USA, 1996.
- Williams, M. The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals, 14th ed.; Merck Inc.: Kenilworth, NJ, USA, 2006; p. 2564. [Google Scholar]
- Pettit, L.D.; Powell, K.J. The IUPAC Stability Constants Database (SC-Database); Timble: Otley, Yorks, UK, 2008. [Google Scholar]
- Ruprecht, J. Dimaval®: Scientific Product Monograph, 7th ed.; Heyl Chem.-Pharm Fabrik: Berlin, Germany, 2008. [Google Scholar]
- Weigert, W.M.; Offermanns, H.; Degussa, P.S. D-Penicillamine—Production and Properties. Angew. Chem. Int. Ed. Engl. 1975, 14, 330–336. [Google Scholar] [CrossRef]
- Nurchi, V.M.; Crisponi, G.; Lachowicz, J.I.; Jaraquemada-Pelaez, M.D.G.; Bretti, C.; Peana, M.; Medici, S.; Zoroddu, M.A. Equilibrium studies of new bis-hydroxypyrone derivatives with Fe3+, Al3+, Cu2+ and Zn2+. J. Inorg. Biochem. 2018, 189, 103–114. [Google Scholar] [CrossRef]
- Nurchi, V.M.; Crespo-Alonso, M.; Toso, L.; Lachowicz, J.I.; Crisponi, G. Chelation Therapy for Metal Intoxication: Comments from a Thermodynamic Viewpoint. Mini-Rev. Med. Chem 2013, 13, 1541–1549. [Google Scholar] [CrossRef] [PubMed]
- Basinger, M.A.; Casas, J.S.; Jones, M.M.; Weaver, A.D.; Weinstein, N.H. Structural requirements for Hg (II) antidotes. J. Inorg. Nucl. Chem. 1981, 43, 1419–1425. [Google Scholar] [CrossRef]
- Matsui, H.; Ohtaki, H. A potentiometric study on complex formation of cadmium (II) ion with 2-mercaptoacetic and 2-mercaptopropionic acids. Polyhedron 1983, 2, 631–633. [Google Scholar] [CrossRef]
- Li, N.C.; Manning, R.A. Some Metal Complexes of Sulfur-containing Amino Acids. J. Am. Chem. Soc. 1955, 77, 5225–5228. [Google Scholar] [CrossRef]
- Lenz, G.R.; Martell, A.E. Metal Chelates of Some Sulfur-Containing Amino Acids. Biochemistry 1964, 3, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Urbańska, J.; Kozlowski, H.; Kurzak, B. Polarographic and potentiometric study of ternary complexes of cadmium with penicillamine and histidine. J. Coord. Chem. 1992, 25, 149–154. [Google Scholar] [CrossRef]
- Harris, W.R.; Chen, Y.; Stenback, J.; Shah, B. Stability constants for dimercaptosuccinic acid with bismuth (III), zinc (II), and lead (II). J. Coord. Chem. 1991, 23, 173–186. [Google Scholar] [CrossRef]
- Casas, J.S.; Jones, M.M. Mercury (II) complexes with sulfhydryl containing chelating agents: Stability constant inconsistencies and their resolution. J. Inorg. Nucl. Chem. 1980, 42, 99–102. [Google Scholar] [CrossRef]
- Avdeef, A.; Chemotti, A.R. Cadmium binding by biological ligands. Part 4. Polynuclear complexes of cadmium with 2,3-dimercaptopropane-1-sulphonic acid. J. Chem. Soc. Dalt. Trans. 1991, 5, 1189–1194. [Google Scholar] [CrossRef]
- Strand, R.; Lund, W.; Aaseth, J. Complex formation of zinc, cadmium, and mercury with penicillamine. J. Inorg. Biochem. 1983, 19, 301–309. [Google Scholar] [CrossRef]
- Rivera, M.; Zheng, W.; Aposhian, H.V.; Fernando, Q. Determination and metabolism of dithiol chelating agents. VIII. Metal complexes of meso-dimercaptosuccinic acid. Toxicol. Appl. Pharmacol. 1989, 100, 96–106. [Google Scholar] [CrossRef]
- Chekmeneva, E.; Díaz-Cruz, J.M.; Ariño, C.; Esteban, M. Study of the Hg2+ binding with chelation therapy agents by differential pulse voltammetry on rotating Au-disk electrode and electrospray ionization mass-spectrometry. Anal. Chim. Acta 2009, 653, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Kõszegi-Szalai, H.; Paál, T.L. Equilibrium studies of mercury (II) complexes with penicillamine. Talanta 1999, 48, 393–402. [Google Scholar] [CrossRef]
- Leung, B.O.; Jalilehvand, F.; Mah, V. Mercury (II) penicillamine complex formation in alkaline aqueous solution. Dalt. Trans. 2007, 41, 4666–4674. [Google Scholar] [CrossRef] [PubMed]
- Strasdeit, H.; von Döllen, A.; Saak, W.; Wilhelm, M. Intracellular Degradation of Diorganomercury Compounds by Biological Thiols—Insights from Model Reactions. Angew. Chem. Int. Ed. 2000, 39, 784–786. [Google Scholar] [CrossRef]
- Chekmeneva, E.; Díaz-Cruz, J.M.; Ariño, C.; Esteban, M. Complexation of Hg2+ with α-Lipoic and dihydrolipoic acids: Study by differential pulse voltammetry on rotating Au-Disk electrode and ESI-MS. Electroanalysis 2010, 22, 177–184. [Google Scholar] [CrossRef]
- Zeini Jahromi, E.; Gailer, J.; Pickering, I.J.; George, G.N. Structural characterization of Cd2+ complexes in solution with DMSA and DMPS. J. Inorg. Biochem. 2014. [Google Scholar] [CrossRef] [PubMed]
- De La Gala Morales, M.; Ariño, C.; Díaz-Cruz, J.M.; Esteban, M. Study of the complexation of Pb (II) with meso-2,3-dimercaptosuccinic acid (DMSA) and 2,3-dimercapto-1-propanesulfonic acid (DMPS) using a bismuth-bulk rotating disk electrode. Electroanalysis 2014, 26, 1912–1919. [Google Scholar] [CrossRef]



| Metal Ions | Coordinating Groups | ||||
|---|---|---|---|---|---|
| Hard | Borderline | Soft | Hard | Borderline | Soft |
| Li+, Na+, K+, Be2+ Mg2+, Ca2+, Sr2+, Mn2+, Al3+, Ga3+, Cr3+, Fe3+, Sn4+, (CH3)2Sn2+, UO22+, VO2+ | Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Pb2+, Sn2+, Sb3+, Bi3+ | Cu+, Ag+, Au+, Hg+, Pd2+, Cd2+, Pt2+, Hg2+, CH3Hg+, Pt4+ | H2O, OH−, F−, RCOO−, Cl−, RO−, NH3, RNH2 | C6H5NH2 | R2S, RSH, RS− |
| Important Sources of Occupational Exposure | Routes of Exposure | Important Sources of Environmental Exposure | Routes of Exposure | Target Organs of Toxicity | |
|---|---|---|---|---|---|
| Elemental mercury | Coal-burning, waste incineration, gold extraction, dental amalgam handling, fluorescent lamp manufacturing | Inhalation | Dental amalgam in teeth | Inhalation | Central and peripheral nervous system |
| Inorganic mercury salts | - | - | Use of skin lightening products and medicinal use of mercury salts | Gastrointestinal ingestion, transdermal | Kidneys |
| Methyl mercury | - | - | Food (fish, seafood) | Gastrointestinal ingestion | Central nervous system |
| Cadmium | Production of nickel-Cd batteries, Cd plating, Cd-containing paint production | Inhalation | Food (rice, potato, and wheat, offal, seafood) Tobacco smoke | Gastrointestinal ingestion Inhalation | Kidneys Skeleton |
| Lead | Mining, smelting, battery manufacturing, traditional printing technology | Inhalation | Food, drinking water, dust and soil (in children) | Gastrointestinal ingestion | Central nervous system, hematopoietic system, kidneys |
| Structure | Name | Acronym | Formula | MW | log K1 | log K2 | log K3 | log K4 |
|---|---|---|---|---|---|---|---|---|
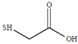 | Thioglycolic acid | TGA | C2H4O2S | 92.11 | [174] 9.96 | 3.44 | ||
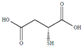 | Thiomalic acid | TMA | C4H6O4S | 150.15 | [174] 10.24 | 4.52 | 3.12 | |
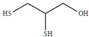 | 2,3 Dimercapto propan-1-ol | BAL | C3H8OS2 | 124.23 | 10.8 | 8.7 | ||
 | meso-Dimercapto succinic acid | DMSA | C4H6O4S2 | 182.22 | [174] 12.05 | 9.65 | 3.43 | 2.71 |
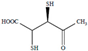 | Methyl-DMSA | MDMSA | C5H8O4S2 | 196.22 | [174] 9.98 | 7.15 | 2.8 | |
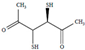 | Dimethyl-DMSA | DDMSA | C6H10O4S2 | 210.22 | [174] 7.31 | 5.23 | ||
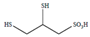 | Unitiol | DMPS | C3H8O3S3 | 188.289 | [175] 11.38 | 8.69 | ||
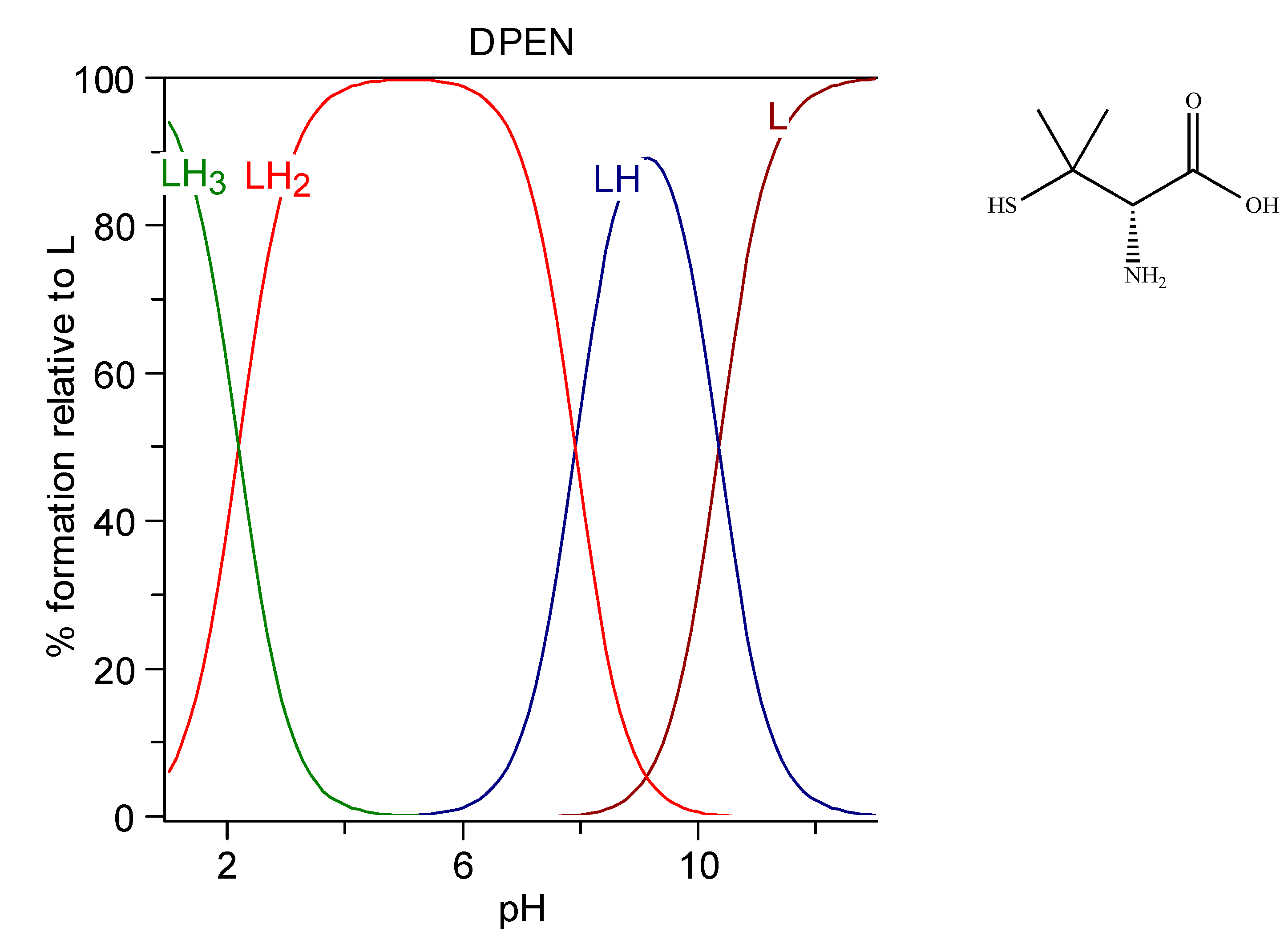
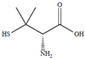 | d-Penicillamine | DPEN | C5H11NO2S | 149.212 | [176] 10.35 | 7.91 | 2.19 | |
 | Lipoic acid | LA | C8H14O2S2 | 206.343 | [177] 6.37 * $ 4.704(1) | |||
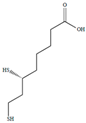 | Dihydrolipoic acid | DHLA | C8H16O2S2 | 208.343 | [178] 11.02 | 9.86 | 4.73 |
| Hg2+ | Cd2+ | Pb2+ | ||||||
|---|---|---|---|---|---|---|---|---|
| Ligand | Species | Logβ | Species | Logβ | Species | Logβ | Temp./Ion. Str. | Method |
| TGA | [186] HgL | 34.5 | [187] CdLH | 11.08 | [188] PbL | 8.5 | [186] 25, 0.1 M NaClO4 | EMF |
| HgL2 | 40.5 | CdL | 4.34 | [187] 25, 3 M LiClO4 | gl | |||
| CdL2 | 6.49 | [188] 25, 0.15 M | gl | |||||
| pM | 32.9 | 6.00 | 6.95 | |||||
| TMA | [189] HgL | 9.94 | [190] CdL | 10.05 | [181] PbL | 10.80 | [189] 25, 0.1 M KNO3 | gl |
| HgL2 | 18.07 | CdL2 | 13.51 | [190] 25, 0.2 M KNO3 | gl | |||
| Cd3L4 | 41.59 | [181] 30, 0.007 ClO4− | gl | |||||
| pM | 8.24 | 7.78 | 8.90 | |||||
| DMSA | § | ---- | [181] CdLH3 | 28.73 | [191] PbL | 17.4 | [181] 25, 0.1 M KCl | gl |
| CdLH | 23.50 | [191] 25, 0.1 M | Spect. | |||||
| CdL | 17.11 | |||||||
| pM | 11.48 | 11.45 | ||||||
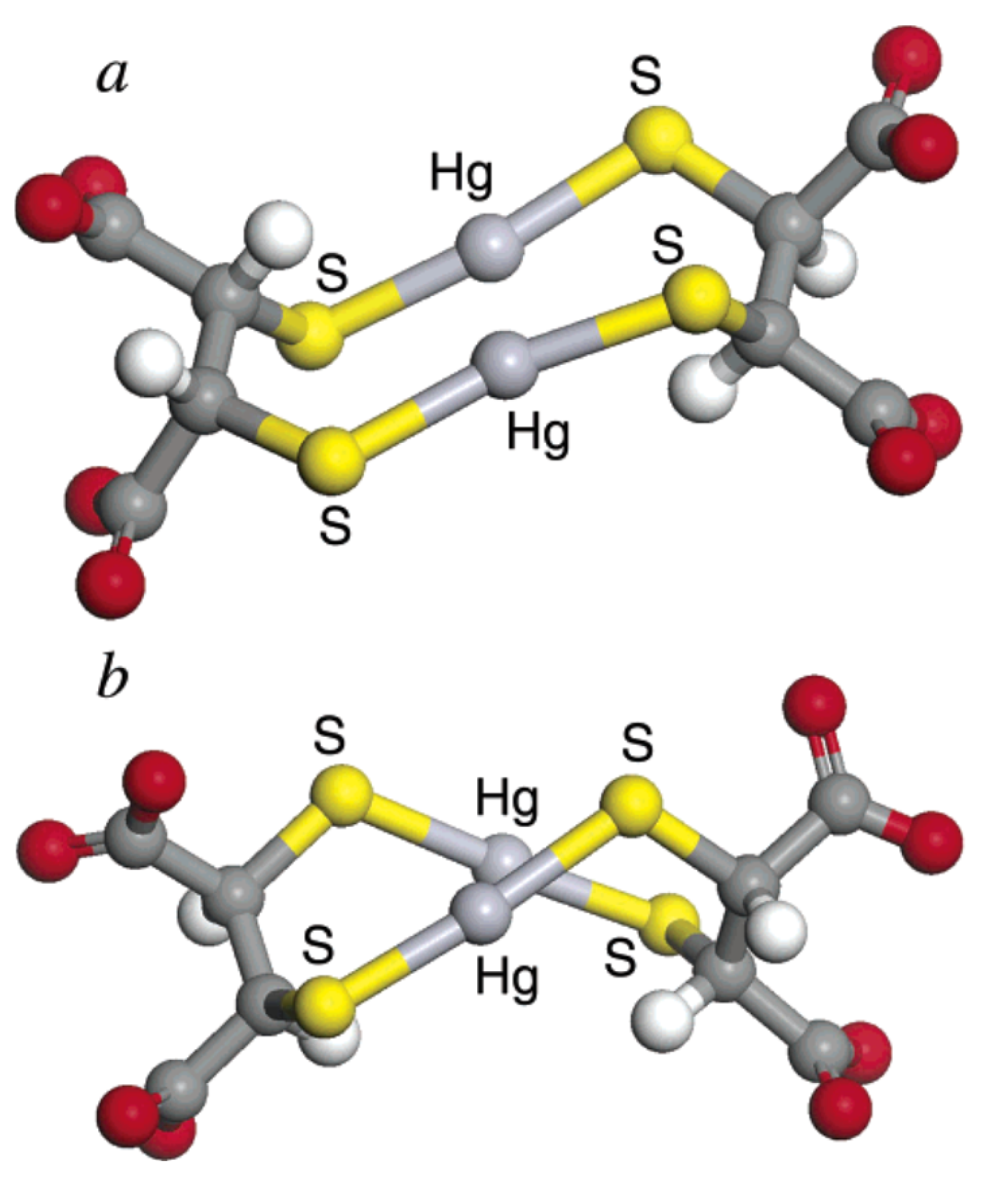
| DMPS | [192] HgL | 42.2 | [193] CdL2 | 28.27 | [181] PbL | 16.38 | [192] 25, 0.1 M NaClO4 | ISE |
| HgL2 | 53.1 | Cd3L3 | 59.9 | PbL2 | 22.21 | [193] 25, 0.2 M KNO3 | gl | |
| Cd3L4 | 71.9 | [181] 20, 0.1 KNO3 | EMF | |||||
| Cd5L6 | 114.3 | |||||||
| Cd7L8 | 156.7 | |||||||
| pM | 37.60 | 13.24 | 12.00 | |||||
| [181] HgL | 39.71 | [181] CdL | 17.32 | [181] 20, 0.1 KNO3 | EMF | |||
| CdL2H | 35.19 | [181] 37, 0.15 NaCl | gl | |||||
| CdL2 | 28.22 | |||||||
| Cd2L2 | 37.72 | |||||||
| Cd3L3H | 61.91 | |||||||
| pM | 34.80 | 12.14 | ||||||
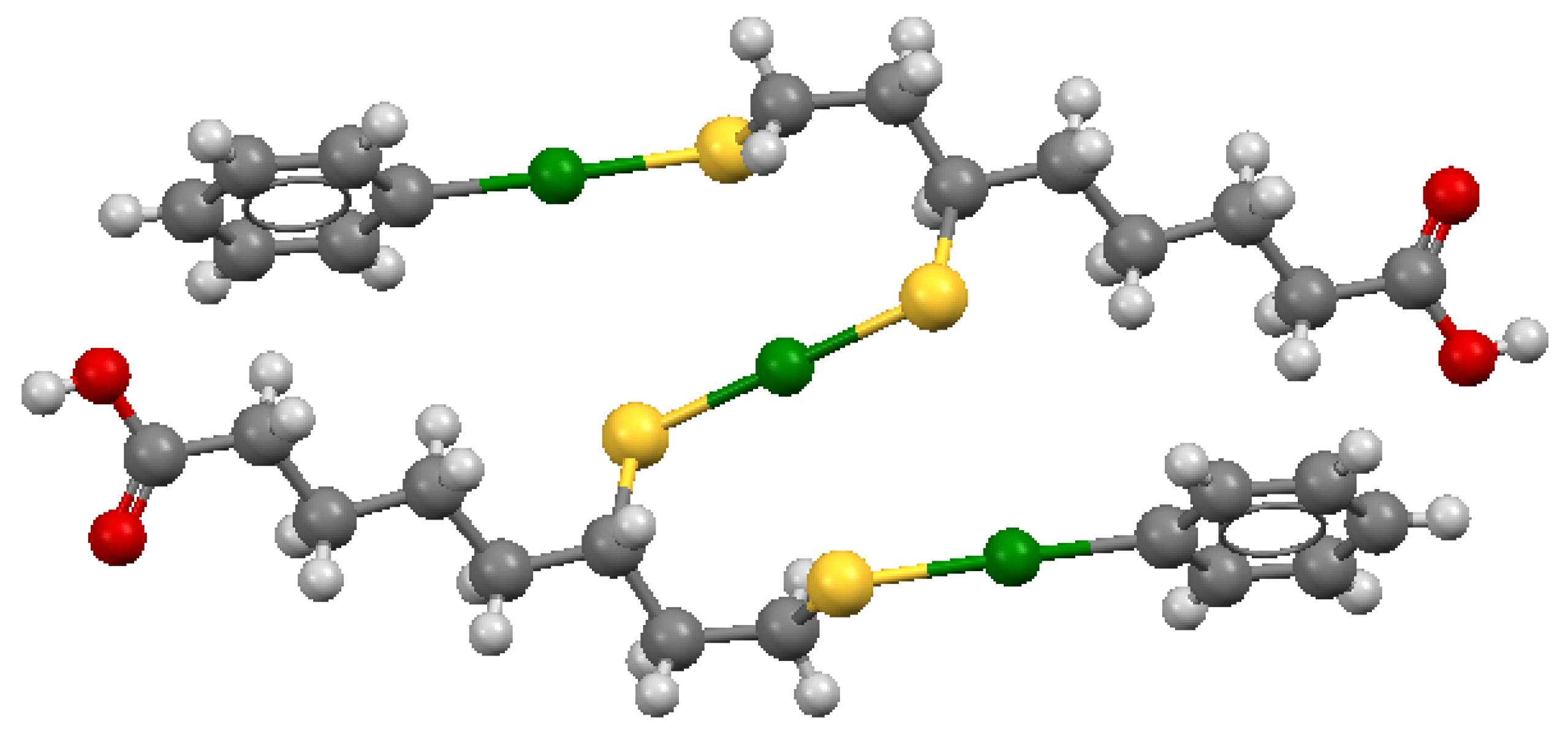
| DPEN | [194] HgL | 37.6 | [190] CdL | 11.53 | [181] PbLH | 15.87 | [194] 25, 0.1 M KNO3 | |
| HgL2H | 52.31 | CdL2 | 19.64 | PbL | 13.12 | [190] 25, 0.2 M KNO3 | gl | |
| HgL2 | 43.69 | Cd3L4 | 50.22 | PbL2H | 26.19 | [181] 25, 0.1 M KCl | gl | |
| PbL2 | 17.7 | |||||||
| pM | 34.50 | 8.61 | 10.50 | |||||
| [186] HgL | 37.8 | [194] CdL | 11.51 | [186] 25, 0.1 M NaClO4 | gl | |||
| HgL2H | 53.6 | CdL2H | 15.94 | [194] 25, 0.1 M KNO3 | ||||
| HgL2 | 44.50 | CdL2 | 19.52 | |||||
| CdL3 | 22.35 | |||||||
| pM | 35.30 | 8.46 | ||||||
| Toxic Agent | Recommended Chelation Treatment |
|---|---|
| Inorganic mercuric salts | DMPS |
| Methyl mercury | DMSA |
| Elemental mercury vapor | DMPS (initially combined with BAL) |
| Cadmium | DMSA (combined with MiADMSA) |
| Lead | DMSA (combined with Monensin) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bjørklund, G.; Crisponi, G.; Nurchi, V.M.; Cappai, R.; Buha Djordjevic, A.; Aaseth, J. A Review on Coordination Properties of Thiol-Containing Chelating Agents Towards Mercury, Cadmium, and Lead. Molecules 2019, 24, 3247. https://doi.org/10.3390/molecules24183247
Bjørklund G, Crisponi G, Nurchi VM, Cappai R, Buha Djordjevic A, Aaseth J. A Review on Coordination Properties of Thiol-Containing Chelating Agents Towards Mercury, Cadmium, and Lead. Molecules. 2019; 24(18):3247. https://doi.org/10.3390/molecules24183247
Chicago/Turabian StyleBjørklund, Geir, Guido Crisponi, Valeria Marina Nurchi, Rosita Cappai, Aleksandra Buha Djordjevic, and Jan Aaseth. 2019. "A Review on Coordination Properties of Thiol-Containing Chelating Agents Towards Mercury, Cadmium, and Lead" Molecules 24, no. 18: 3247. https://doi.org/10.3390/molecules24183247
APA StyleBjørklund, G., Crisponi, G., Nurchi, V. M., Cappai, R., Buha Djordjevic, A., & Aaseth, J. (2019). A Review on Coordination Properties of Thiol-Containing Chelating Agents Towards Mercury, Cadmium, and Lead. Molecules, 24(18), 3247. https://doi.org/10.3390/molecules24183247











