Osteopathic Manipulation Treatment Improves Cerebro–splanchnic Oximetry in Late Preterm Infants
Abstract
1. Introduction
2. Results
2.1. Main Perinatal Outcomes
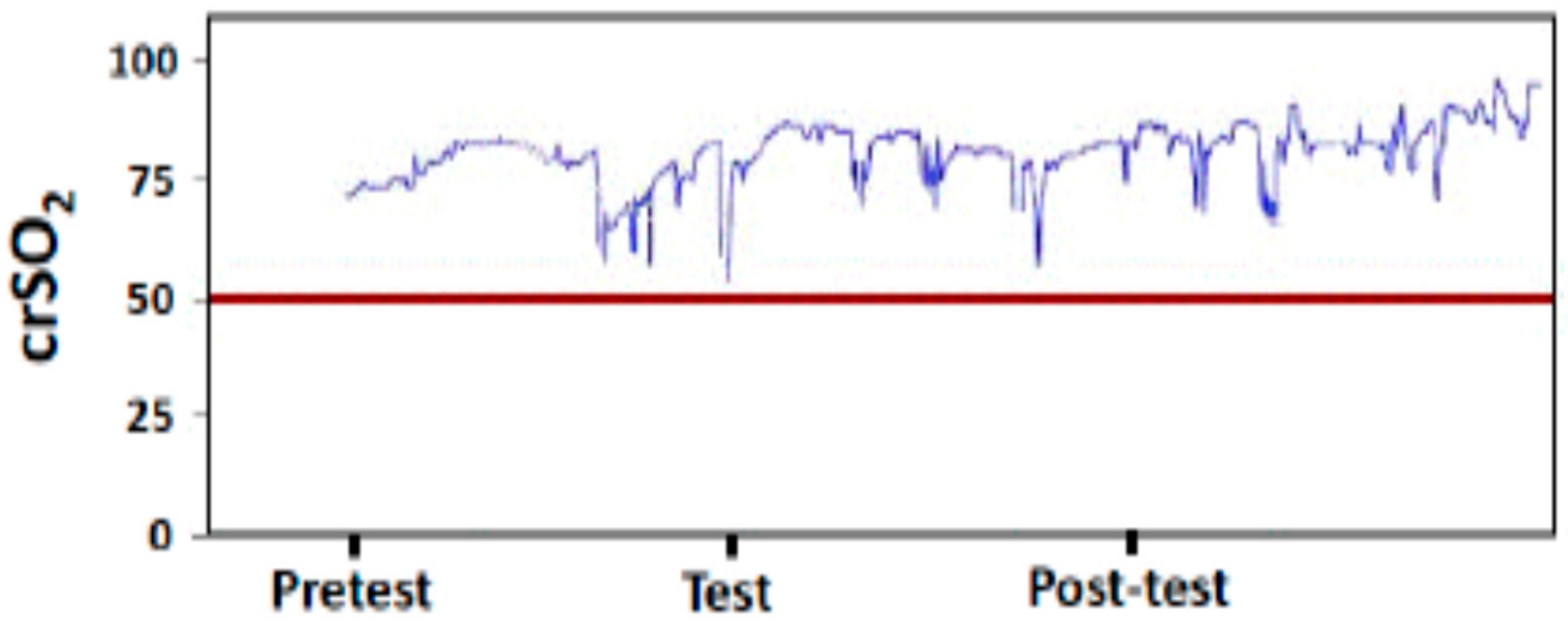
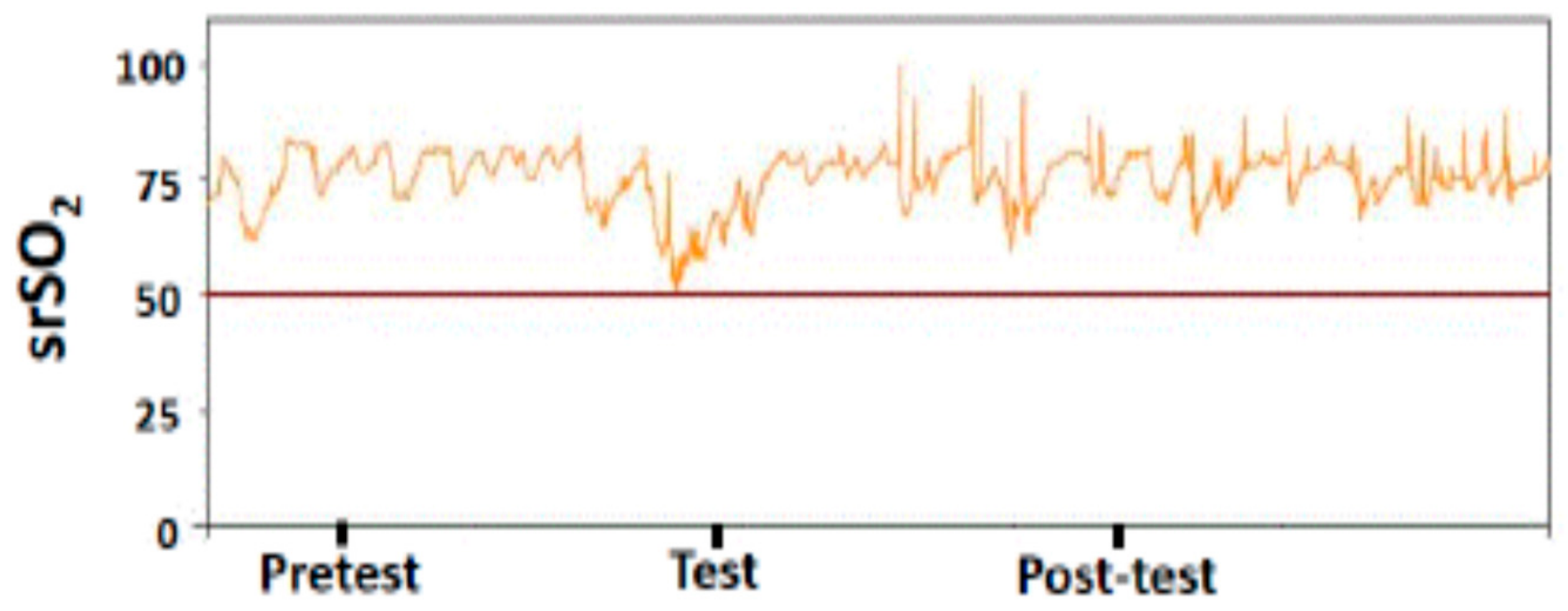
2.2. Monitoring Parameters
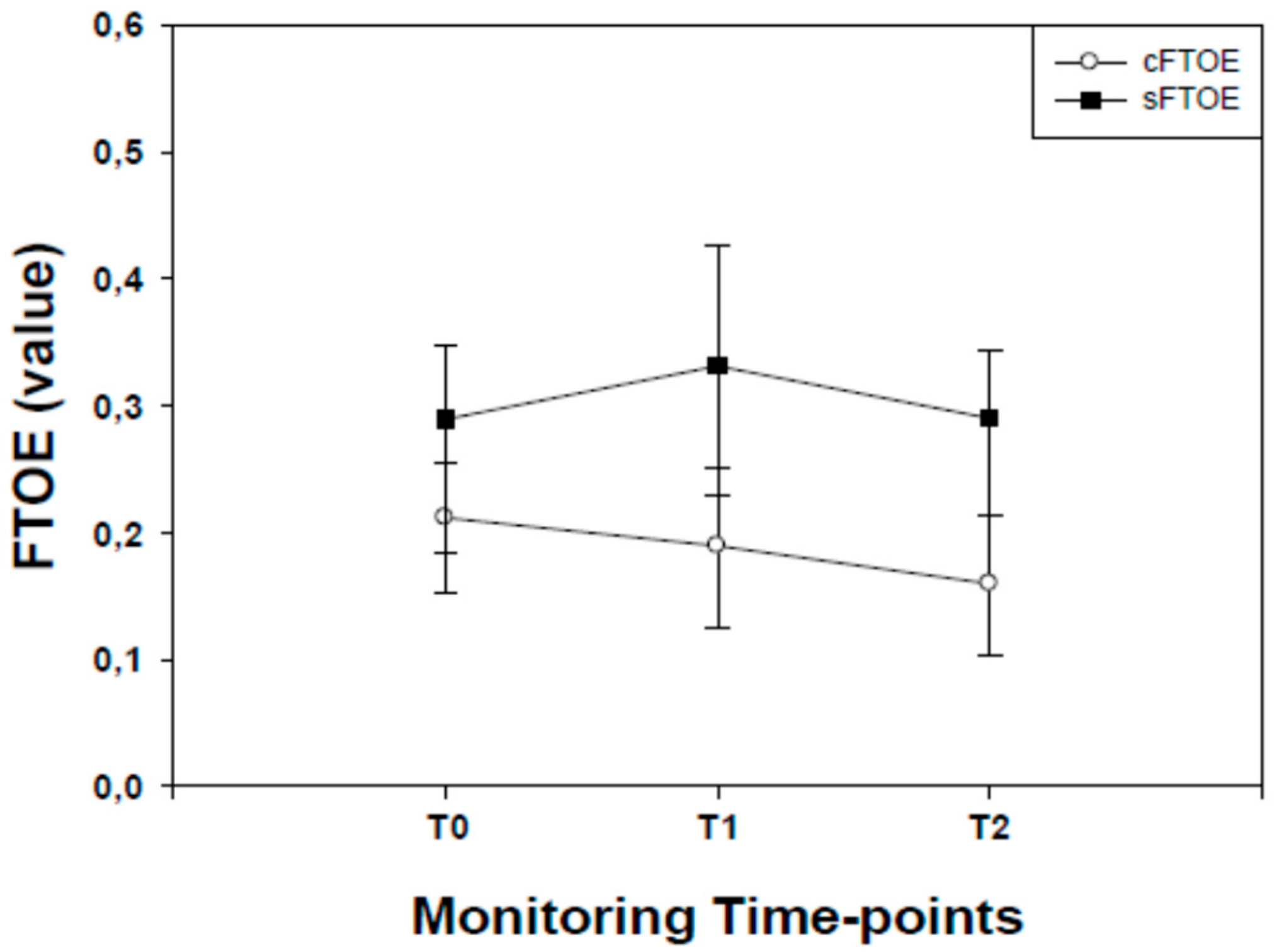
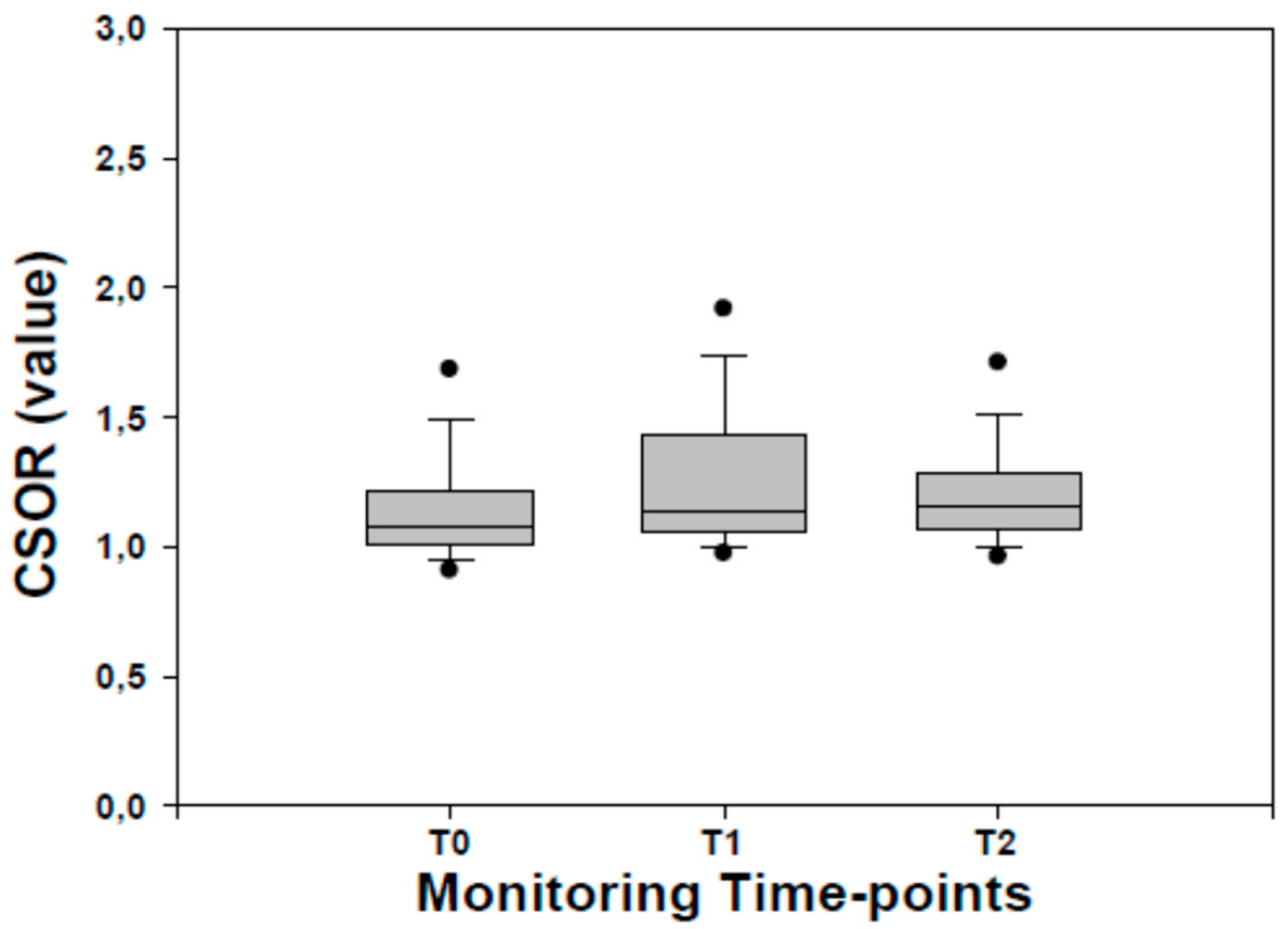
2.3. NIRS Parameters
3. Discussion
4. Materials and Methods
4.1. NIRS Monitoring
4.2. Standard Monitoring Parameters and Main Outcomes
4.3. Cranial Assessment
4.4. Neurological Examination
4.5. Osteopathic Procedure
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. March of Dimes, PMNCH, Save the Children. In Born Too Soon: The Global Action Report on Preterm Birth; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar] [CrossRef]
- Beck, S.; Wojdyla, D.; Say, L.; Betran, A.P.; Merialdi, M.; Requejo, J.H.; Rubens, C.; Menon, R.; Van Look, P.F.A. The worldwide incidence of preterm birth: A systematic review of maternal mortality and morbidity. Bull. World Health Organ. 2010, 88, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Italian Ministry of Health. Annual Report on Hospitals Activity; Ministry of Health: Rome, Italy, 2012. [Google Scholar]
- National Academy Press. Preterm Birth: Causes, Consequences, and Prevention; National Academy Press: Cambridge, MA, USA, 2007. [Google Scholar]
- Boyle, E.M.; Poulsen, G.; Field, D.J.; Kurinczuk, J.J.; Wolke, D.; Alfirevic, Z.; Quigley, M.A. Effects of gestational age at birth on health outcomes at 3 and 5 years of age: Population based cohort study. BMJ 2012, 344, e896. [Google Scholar] [CrossRef] [PubMed]
- Pineda, R.; Guth, R.; Herring, A.; Reynolds, L.; Oberle, S.; Smith, J. Enhancing sensory experiences for very preterm infants in the NICU: An integrative review. J. Perinatol. 2017, 37, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Cerritelli, F.; Pizzolorusso, G.; Renzetti, C.; Cozzolino, V.; D’Orazio, M.; Lupacchini, M.; Marinelli, B.; Accorsi, A.; Lucci, C.; Lancellotti, J.; et al. A multicenter, randomized, controlled trial of osteopathic manipulative treatment on preterms. PLoS ONE 2015, 10, e0127370. [Google Scholar] [CrossRef] [PubMed]
- Pizzolorusso, G.; Turi, P.; Barlafante, G.; Cerritelli, F.; Renzetti, C.; Cozzolino, V.; D’Orazio, M.; Fusilli, P.; Carinci, F.; D’Incecco, C. Effect of osteopathic manipulative treatment on gastrointestinal function and length of stay of preterm infants: An exploratory study. Chiropr. Man. Ther. 2011, 19, 15. [Google Scholar] [CrossRef]
- Henley, C.; Ivins, D.; Mills, M.; Wen, F.; Benjamin, B. Osteopathic manipulative treatment and its relationship to autonomic nervous system activity as demonstrated by heart rate variability: A repeated measures study. Osteopath Med. Prim. Care 2008, 2, 7. [Google Scholar] [CrossRef]
- Giles, P.; Hensel, K.; Pacchia, C.; Smith, M. Suboccipital decompression enhances heart rare variability indices of cardiac control in healthy subjects. J. Altern. Complement. Med. 2013, 19, 92–96. [Google Scholar] [CrossRef]
- Ruffini, N.; D’Alessandro, G.; Mariani, N.; Pollastrelli, A.; Cardinali, L.; Cerritelli, F. Variations of high frequency parameter of heart rate variability following osteopathic manipulative treatment in healthy subjects compared to control group and sham therapy: Randomized controlled trial. Front. Neurosci. 2015, 9, 272. [Google Scholar] [CrossRef]
- McGlone, F.; Cerritelli, F.; Walker, S.; Esteves, J. The role of gentle touch in perinatal osteopathic manual therapy. Neurosci. Biobehav. Rev. 2017, 72, 1–9. [Google Scholar] [CrossRef]
- Volpe, J.J. Brain injury in premature infants: A complex amalgam of destructive and developmental disturbances. Lancet Neurol. 2009, 8, 110–124. [Google Scholar] [CrossRef]
- Arpino, C.; Argenzio, L.; Ticconi, C.; Di Paolo, A.; Stellin, V.; Lopez, L.; Curatolo, P. Brain damage in preterm infants: Etiological pathways. Ann. Ist. Super. Sanità 2005, 41, 229–237. [Google Scholar]
- Prechtl, H.F.R. Assessment methods for the newborn infant: A critical evaluation. In Psychobiology of Human Newborn; Stratton, P., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 1982; pp. 21–52. [Google Scholar]
- Cerritelli, F.; Martelli, M.; Renzetti, C.; Pizzolorusso, G.; Cozzolino, V.; Barlafante, G. Introducing an osteopathic approach into neonatology ward: The NE-O model. Chiropr. Man Ther. 2014, 22, 18. [Google Scholar] [CrossRef]
- Naulaers, G.; Meyns, B.; Miserez, M.; Leunens, V.; Van Huffel, S.; Casaer, P.; Weindling, M.; Devlieger, H. Use of tissue oxygenation index and fractional tissue oxygen extraction as non-invasive parameters for cerebral oxygenation: A validation study in piglets. Neonatology 2007, 92, 120–126. [Google Scholar] [CrossRef]
- Wardle, S.P.; Yoxall, C.W.; Weindling, A.M. Determinants of cerebral fractional oxygen extraction using near infrared spectroscopy in preterm neonates. J. Cereb. Blood Flow Metab. 2000, 20, 272–279. [Google Scholar] [CrossRef]
- Dani, C.; Pratesi, S.; Fontanelli, G.; Barp, J.; Bertini, G. Blood transfusions increase, cerebral, splanchnic and renal oxygenation in anemic preterm infants. Transfusion 2010, 50, 1220–1226. [Google Scholar] [CrossRef]
- Bozzetti, V.; Paterlini, G.; Van Bel, F.; Visser, G.H.A.; Tosetti, L.; Gazzolo, D.; Tagliabue, P.E. Cerebral and somatic NIRS-determined oxygenation in IUGR and non-IUGR preterm infants during transition. J. Matern. Fetal Neonatal Med. 2015, 25, 1–4. [Google Scholar]
- Kinney, H.C. The near-term (late preterm) human brain and risk for periventricular leukomalacia: A review. Semin. Perinatol. 2006, 30, 81–88. [Google Scholar] [CrossRef]
- Haynes, R.L.; Borenstein, N.S.; Desilva, T.M.; Folkerth, R.D.; Liu, L.G.; Volpe, J.J.; Kinney, H.C. Axonal development in the cerebral white matter of the human fetus and infant. J. Comp. Neurol. 2005, 484, 156–167. [Google Scholar] [CrossRef]
- Back, S.A.; Luo, N.L.; Borenstein, N.S.; Levine, J.M.; Volpe, J.J.; Kinney, H.C. Late oligodendrocyte progenitors coincide with the developmental window of vulnerability for human perinatal white matter injury. J. Neurosci. 2001, 21, 1302–1312. [Google Scholar] [CrossRef]
- Huppi, P.S.; Warfield, S.; Kikinis, R.; Barnes, P.D.; Zientara, G.P.; Jolesz, F.A.; Tsuji, M.K.; Volpe, J.J. Quantitative magnetic resonance imaging of brain development in premature and mature newborns. Ann. Neurol. 1998, 43, 224–235. [Google Scholar] [CrossRef]
- Sannia, A.; Zimmermann, L.J.; Gavilanes, A.W.; Vles, H.J.; Serpero, L.; Frulio, R.; Michetti, F.; Gazzolo, D. S100B protein maternal and fetal bloodstreams gradient in healthy and small for gestational age pregnancies. Clin. Chim. Acta 2014, 12, 1337–1340. [Google Scholar] [CrossRef]
- Sannia, A.; Risso, F.M.; Zimmermann, L.J.; Gavilanes, A.W.; Vles, H.J.; Gazzolo, D. S100B urine concentrations in late preterm infants are gestational age and gender dependent. Clin. Chim. Acta 2013, 417, 31–34. [Google Scholar] [CrossRef]
- Tina, L.G.; Frigiola, A.; Abella, R.; Artale, B.; Puleo, G.; D’Angelo, S.; Musmarra, C.; Tagliabue, P.; Li Volti, G.; Florio, P.; et al. Near infrared spectroscopy in healthy preterm and term newborns: Correlation with gestational age and standard monitoring parameters. Curr. Neurovascular Res. 2009, 6, 148–154. [Google Scholar] [CrossRef]
- Gazzolo, D.; Pluchinotta, F.; Lapergola, G.; Franchini, S. The Ca2+-Binding S100B Protein: An Important Diagnostic and Prognostic Neurobiomarker in Pediatric Laboratory Medicine. Methods Mol. Biol. 2019, 1929, 701–728. [Google Scholar] [CrossRef]
- Kleiser, S.; Ostojic, D.; Andresen, B.; Nasseri, N.; Isler, H.; Scholkmann, F.; Karen, T.; Greisen, G.; Wolf, M. Comparison of tissue oximeters on a liquid phantom with adjustable optical properties: An extension. Biomed. Opt. Express 2018, 9, 86–101. [Google Scholar] [CrossRef]
- Isler, H.; Shenk, D.; Bernhard, J.; Kleiser, S.; Scholkmann, F.; Ostojic, D.; Kalyanov, A.; Ahnen, L.; Wolf, M.; Karen, T. Absorption spectra of early stool from preterm infants need to be considered in abdominal NIRS oximetry. Biomed. Opt. Express 2019, 10, 2784–2791. [Google Scholar] [CrossRef]
- Naulaers, G.; Morren, G.; Van Huffel, S.; Casaer, P.; Devlieger, H. Cerebral tissue oxygenation index in very premature infants. Arch. Dis. Child. Fetal Neonatal Ed. 2002, 87, F189–F192. [Google Scholar] [CrossRef]
- Toet, M.C.; Lemmers, P.M.; van Schelven, L.J.; Van Bel, F. Cerebral oxygenation and electrical activity after birth asphyxia: Their relation to outcome. Pediatrics 2006, 117, 333–339. [Google Scholar] [CrossRef]
- Grometto, A.; Pizzo, B.; Strozzi, M.C.; Gazzolo, F.; Gazzolo, D. Near-infrared spectroscopy is a promising noninvasive technique for monitoring the effects of feeding regimens on the cerebral and splanchnic regions. Acta Paediatr. 2018, 107, 234–239. [Google Scholar] [CrossRef]
- Campbell, S.; Thoms, A. Ultrasound measurement of the fetal head to abdomen circumference ratio in the assessment of growth retardation. Br. J. Obstet. Gynaecol. 1977, 84, 165–174. [Google Scholar] [CrossRef]
- Villar, J.; Cheikh Ismail, L.; Victora, C.G.; Ohuma, E.O.; Bertino, E.; Altman, D.G.; Lambert, A.; Papageorghiou, A.T.; Carvalho, M.; Jaffer, Y.A.; et al. International standards for newborn weight, length, and head cir- cumference by gestational age and sex: The newborn Cross-Sectional Study of the INTERGROWTH-21st Project. Lancet 2014, 384, 857–868. [Google Scholar] [CrossRef]
Sample Availability: Not Available. |


| Late Preterm (N = 18) | |
|---|---|
| Maternal Characteristics | |
| Maternal age (y) | 32 ± 2 |
| Chorioamnionitis (n°/tot) | 0/18 |
| Glucocorticoids (n°/tot) | 7/18 |
| PROM (n°/tot) | 3/18 |
| EPH (n°/tot) | 0/10 |
| Neonatal Characteristics | |
| GA (wks) | 35 ± 1 |
| BW (g) | 1762 ± 111 |
| CS (n°/tot) | 4/18 |
| Gender (M/F) | 11/7 |
| Outborn/Inborn (n°/tot) | 0/18 |
| Apgar 1’ | 8 ± 1 |
| Apgar 5’ | 8 ± 1 |
| Main Outcomes | |
| RDS (n°/tot) | 5/18 |
| Surfactant administration (n°/tot) | 5/18 |
| MV (n°/tot) | 5/18 |
| PDA (n°/tot) | 0/18 |
| IVH (n°/tot) | 0/18 |
| EOS (n°/tot) | 2/18 |
| NEC (n°/tot) | 0/18 |
| ROP (n°/tot) | 0/18 |
| LOS (n°/tot) | 2/18 |
| BPD (n°/tot) | 0/10 |
| Late Preterm (N = 18) | |
|---|---|
| Monitoring Parameters | |
| GA (wks) | 36 ± 1 |
| BW (g) | 1846 ± 265 |
| pH | 7.35 ± 0.02 |
| pCO2 (mmHg) | 43.9 ± 4.7 |
| pO2 (mmHg) | 40.1 ± 2.3 |
| Base excess | 0.9 ± 1.1 |
| Bilirubinemia (mg/dL) | 4.3 ± 1.5 |
| Hb (g/dL) | 13.9 ± 1.3 |
| Hematocrit rate (%) | 40.1 ± 2.1 |
| HR (bpm) | 146 ± 12 |
| RR (breath pm) | 56 ± 9 |
| SaO2 at T0 | 98 ± 1 |
| SaO2 at T1 | 98 ± 1 |
| SaO2 at T2 | 99 ± 1 |
| Neurological Examination | |
| Normal/suspect/abnormal | 18/0/0 |
| Cerebral Ultrasound | |
| Normal/abnormal | 18/0/0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marinelli, B.; Pluchinotta, F.; Cozzolino, V.; Barlafante, G.; Strozzi, M.C.; Marinelli, E.; Franchini, S.; Gazzolo, D. Osteopathic Manipulation Treatment Improves Cerebro–splanchnic Oximetry in Late Preterm Infants. Molecules 2019, 24, 3221. https://doi.org/10.3390/molecules24183221
Marinelli B, Pluchinotta F, Cozzolino V, Barlafante G, Strozzi MC, Marinelli E, Franchini S, Gazzolo D. Osteopathic Manipulation Treatment Improves Cerebro–splanchnic Oximetry in Late Preterm Infants. Molecules. 2019; 24(18):3221. https://doi.org/10.3390/molecules24183221
Chicago/Turabian StyleMarinelli, Benedetta, Francesca Pluchinotta, Vincenzo Cozzolino, Gina Barlafante, Maria Chiara Strozzi, Eleonora Marinelli, Simone Franchini, and Diego Gazzolo. 2019. "Osteopathic Manipulation Treatment Improves Cerebro–splanchnic Oximetry in Late Preterm Infants" Molecules 24, no. 18: 3221. https://doi.org/10.3390/molecules24183221
APA StyleMarinelli, B., Pluchinotta, F., Cozzolino, V., Barlafante, G., Strozzi, M. C., Marinelli, E., Franchini, S., & Gazzolo, D. (2019). Osteopathic Manipulation Treatment Improves Cerebro–splanchnic Oximetry in Late Preterm Infants. Molecules, 24(18), 3221. https://doi.org/10.3390/molecules24183221







