Sesquiterpene Lactones and Flavonoids from Psephellus pyrrhoblepharus with Antiproliferative Activity on Human Gynecological Cancer Cell Lines
Abstract
1. Introduction
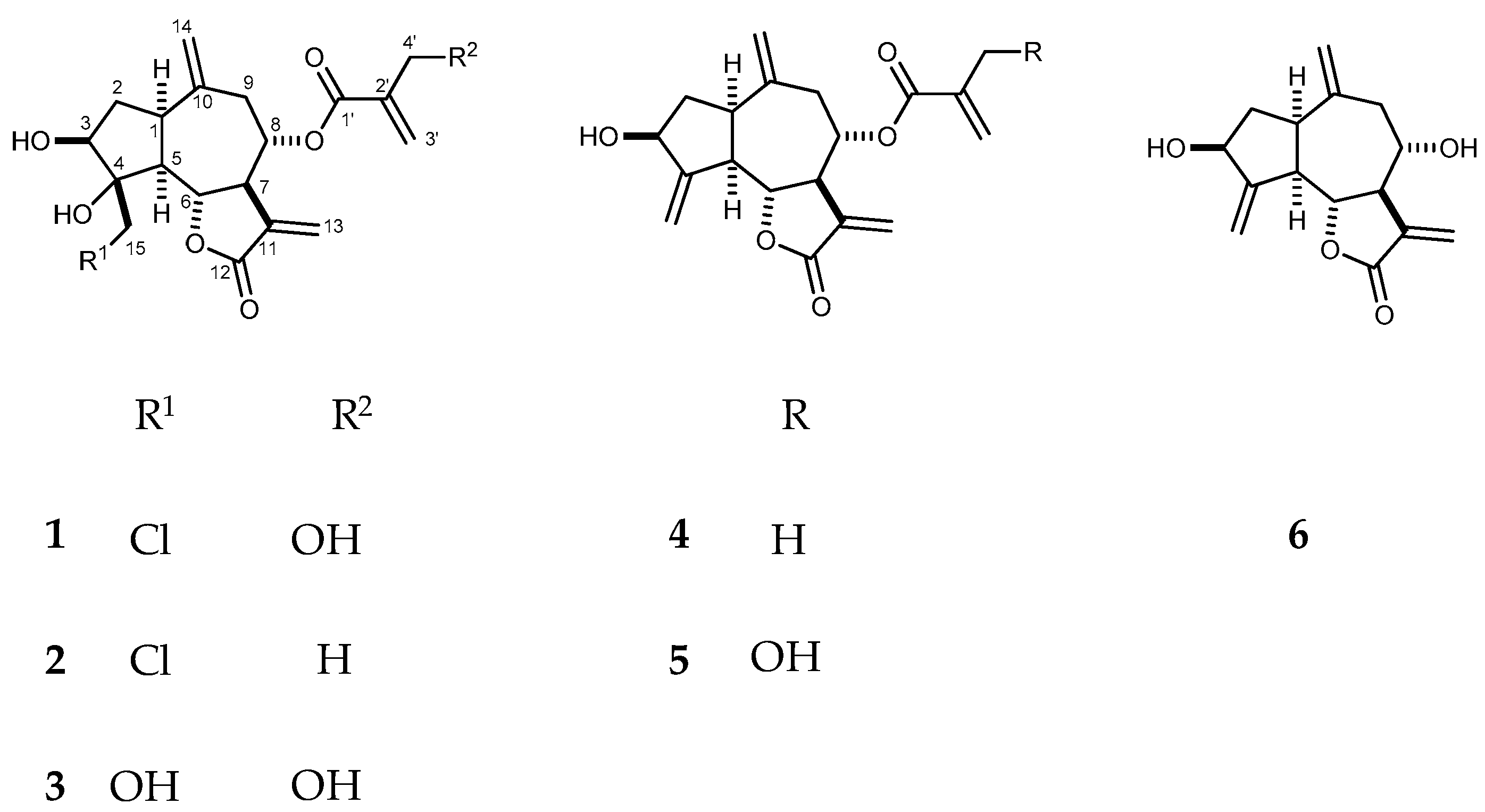
2. Results and Discussion
3. Materials and Methods
3.1. General Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Antiproliferative Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Greuter, W. The Euro+Med treatment of Cardueae (Compositae)–generic concepts and required new names. Willdenowia 2003, 33, 49–61. [Google Scholar] [CrossRef]
- Wagenitz, G.; Hellwig, F.H. The genus Psephellus Cass. (Compositae, Cardueae) revisited with a broadened concept. Willdenowia 2000, 30, 29–44. [Google Scholar] [CrossRef]
- Aydin, Ö.; Çoşkunçelebi, K.; Gültepe, M.; Güzel, M.E. A contribution to taxonomy of Centaurea including Psephellus (Asteraceae) based on anatomical and molecular data. Turk. J. Bot. 2013, 37, 419–427. [Google Scholar]
- Sağiroğlu, M.; Arslantürk, A.; Akdemir, Z.K.; Turna, M. An ethnobotanical survey from Hayrat (Trabzon) and Kalkandere (Rize/Turkey). Biol. Divers. Conserv. 2012, 5, 31–43. [Google Scholar]
- Dalar, A.; Konczak, I. Botanicals from Eastern Anatolia region of Turkey: Antioxidant capacity and phenolic constituents of endemic herbal medicines. J. Herb. Med. 2012, 2, 126–135. [Google Scholar] [CrossRef]
- Ekim, T. The Rare Endemics of Turkey; Türkiye İş Bankası Kültür Yayınları: İstanbul, Turkey, 2007; pp. 366–369. [Google Scholar]
- Mükemre, M.; Behçet, L.; Çakılcıoğlu, U. Ethnobotanical study on medicinal plants in villages of Çatak (Van-Turkey). J. Ethnopharmacol. 2015, 166, 361–374. [Google Scholar] [CrossRef] [PubMed]
- Hartwell, J. Plants used against cancer. J. Nat. Prod. 1968, 31, 71–170. [Google Scholar]
- Vallejo, J.R.; Peral, D.; Gemio, P.; Carrasco, M.C.; Heinrich, M.; Pardo-de-Santayana, M. Atractylis gummifera and Centaurea ornate in the Province of Badajoz (Extremadura, Spain)—Ethnopharmacological importance and toxicological risk. J. Ethnopharmacol. 2009, 126, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Moradi, M.; Mojab, F.; Bidgoliet, S.A. Toxicity assessment of Asteraceae Centaurea repens L extract in mice. Iran J. Pharm. Res. 2017, 16, 1071–1079. [Google Scholar] [PubMed]
- Csupor-Löffler, B.; Hajdú, Z.; Réthy, B.; Zupkó, I.; Máthé, I.; Rédei, T.; Falkay, G.; Hohmann, J. Antiproliferative activity of Hungarian Asteraceae species against human cancer cell lines. Part II. Phytother. Res. 2009, 23, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Taştan, P.; Armagan, G.; Dağcı, T.; Kıvçak, B. Potential cytotoxic activity of Psephellus pyrrhoblepharus extracts. Proceedings 2017, 1, 1047. [Google Scholar] [CrossRef]
- Nazaruk, J.; Karna, E.; Kalemba, D. The chemical composition of the essential oils of Cirsium palustre and C. rivulare and their antiproliferative effect. Nat. Prod. Commun. 2012, 7, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Machado, F.B.; Yamamoto, R.E.; Zanoli, K.; Nocchi, S.R.; Novello, C.R.; Schuquel, I.T.; Sakuragui, C.M.; Luftmann, H.; Ueda-Nakamura, T.; Nakamura, C.V.; et al. Evaluation of the antiproliferative activity of the leaves from Arctium lappa by a bioassay-guided fractionation. Molecules 2012, 17, 1852–1859. [Google Scholar] [CrossRef] [PubMed]
- Forgo, P.; Zupkó, I.; Molnár, J.; Vasas, A.; Dombi, G.; Hohmann, J. Bioactivity-guided isolation of antiproliferative compounds from Centaurea jacea L. Fitoterapia 2012, 83, 921–925. [Google Scholar] [CrossRef] [PubMed]
- Csapi, B.; Hajdú, Z.; Zupkó, I.; Berényi, A.; Forgo, P.; Szabó, P.; Hohmann, J. Bioactivity-guided isolation of antiproliferative compounds from Centaurea arenaria. Phytother. Res. 2010, 24, 1664–1669. [Google Scholar] [CrossRef] [PubMed]
- Nowak, G. A chemotaxonomic study of sesquiterpene lactones from subtribe Centaureinae of the Compositae. Phytochemistry 1992, 31, 2363–2368. [Google Scholar] [CrossRef]
- Cis, J.; Nowak, G.; Horoszkiewicz-Hassan, M.; Kisiel, W. Syringin in some species of the subtribe Centaureinae of the Asteraceae. Acta Soc. Bot. Pol. 2003, 72, 105–107. [Google Scholar] [CrossRef][Green Version]
- Hayta, S.; Bagci, E. Composition of the essential oil of endemic Psephellus pyrrhoblephara Boiss. (Asteraceae) from Turkey. J. Essent. Oil Bear. Plants 2015, 18, 627–632. [Google Scholar] [CrossRef]
- Ramos, P.A.; Guerra, Â.R.; Guerreiro, O.; Santos, S.A.; Oliveira, H.; Freire, C.S.; Silvestre, A.J.; Duarte, M.F. Antiproliferative effects of Cynara cardunculus L. var. altilis (DC) lipophilic extracts. Int. J. Mol. Sci. 2017, 18, 63. [Google Scholar] [CrossRef]
- Iranshahy, M.; Tayarani-Najaran, Z.; Kasaian, J.; Ghandadi, M.; Emami, S.A.; Asili, J.; Chandran, J.N.; Schneider, B.; Iranshahi, M. Highly oxygenated sesquiterpene lactones from Cousinia aitchisonii and their cytotoxic properties: Rhaserolide induces apoptosis in human T lymphocyte (Jurkat) cells via the activation of c-Jun n-terminal kinase phosphorylation. Phytother. Res. 2016, 30, 222–226. [Google Scholar] [CrossRef]
- Choi, S.Z.; Choi, S.U.; Lee, K.R. Cytotoxic sesquiterpene lactones from Saussurea calcicola. Arch. Pharm. Res. 2005, 28, 1142–1146. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Amini, E.; Asili, J.; Masullo, M.; Piacente, S.; Iranshahi, M. Screening of several biological activities induced by different sesquiterpene lactones isolated from Centaurea behen L. and Rhaponticum repens (L.) Hidalgo. Nat. Prod. Res. 2018, 32, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Ghantous, A.; Gali-Muhtasib, H.; Vuorela, H.; Saliba, N.A.; Darwiche, N. What made sesquiterpene lactones reach cancer clinical trials? Drug Discov. Today 2010, 15, 668–678. [Google Scholar] [CrossRef]
- Lone, S.H.; Bhat, K.A.; Khuroo, M.A. Arglabin: From isolation to antitumor evaluation. Chem. Biol. Interact. 2015, 240, 180–198. [Google Scholar] [CrossRef]
- Muhammad, I.; Takamatsu, S.; Mossa, J.S.; El-Feraly, F.S.; Walker, L.A.; Clark, A.M. Cytotoxic sesquiterpene lactones from Centaurothamnus maximus and Vicoa pentanema. C. Phytother. Res. 2003, 17, 168–173. [Google Scholar] [CrossRef]
- Cho, J.Y.; Kim, A.R.; Jung, J.H.; Chun, T.; Rhee, M.H.; Yoo, E.S. Cytotoxic and pro-apoptotic activities of cynaropicrin, a sesquiterpene lactone, on the viability of leukocyte cancer cell lines. Eur. J. Pharmacol. 2004, 492, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.Y.; Park, J.R.; Byun, D.S. 6-Methoxyluteolin from Chrysanthemum zawadskii var. latilobum suppresses histamine release and calcium influx via down-regulation of FcεRI α chain expression. J. Microbiol. Biotechnol. 2012, 22, 622–627. [Google Scholar] [CrossRef]
- Lee, K.H.; Choi, S.U.; Lee, K.R. Sesquiterpenes from Syneilesis palmata and their cytotoxicity against human cancer cell lines. Arch. Pharm. Res. 2005, 28, 280–284. [Google Scholar]
- Csupor-Löffler, B.; Hajdú, Z.; Zupkó, I.; Molnár, J.; Forgo, P.; Vasas, A.; Kele, Z.; Hohmann, J. Antiproliferative constituents of the roots of Conyza canadensis. Planta Med. 2011, 77, 1183–1188. [Google Scholar] [CrossRef]
- Ha, T.J.; Jang, D.S.; Lee, J.R.; Lee, K.D.; Lee, J.; Hwang, S.W.; Jung, H.J.; Nam, S.H.; Park, K.H.; Yang, M.S. Cytotoxic effects of sesquiterpene lactones from the flowers of Hemisteptia lyrata B. Arch. Pharm. Res. 2003, 26, 925–928. [Google Scholar] [CrossRef]
- Erenler, R.; Sen, O.; Yaglioglu, A.S.; Demirtas, I. Bioactivity-guided isolation of antiproliferative sesquiterpene lactones from Centaurea solstitialis L. ssp. solstitialis. Comb. Chem. High Throughput Screen 2016, 19, 66–72. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–6 are available from the authors. |
| 1 | 2 | 3 | 4 | 5 | 6 | |
|---|---|---|---|---|---|---|
| 1 | 3.56 ddd (9.6, 7.1, 7.1) | 3.60 m | 3.47 ddd (9.5, 7.8, 7.2) | 3.01 dd (9.7, 7.2, 7.2) | 3.01 ddd (9.4, 7.1, 7.1) | 2.96 ddd (9.3, 7.3, 7.3) |
| 2a | 2.45 ddd (14.7, 7.1, 5.3) | 2.49 ddd (14.6, 7.0, 5.5) | 2.40 m | 2.10 ddd (13.1, 7.2, 7.0) | 2.09 ddd (13.4, 7.1, 7.1) | 2.10 ddd (13.8, 7.3, 7.1) |
| 2b | 1.51 ddd (14.7, 10.0, 7.1) | 1.54 dd (14.6, 10.2, 7.0) | 1.60 ddd (14.0, 10.1, 7.8) | 1.75 ddd (13.1, 9.3, 7.2) | 1.74 ddd (13.4, 9.3, 7.1) | 1.71 ddd (13.8, 9.4, 7.3) |
| 3 | 4.07 m | 4.10 m | 4.07 m | 4.51 t (9.3, 7.0) | 4.51 dd (9.3, 7.1) | 4.48 dd (9.4, 7.1) |
| 5 | 2.26 t (9.6) | 2.28 dd (9.8, 9.2) | 2.28 dd (10.2, 9.7) | 2.90 t (10.1, 9.7) | 2.91 dd (10.2, 9.4) | 2.84 m |
| 6 | 4.86 dd (9.6, 9.2) | 4.91 dd (9.8, 9.2) | 4.80 m | 4.34 t (10.1) | 4.35 t (10.2) | 4.16 dd (10.3, 9.2) |
| 7 | 3.15 br t (9.2) | 3.17 m | 3.20 m | 3.26 m | 3.28 m | 2.87 m |
| 8 | 5.12 m | 5.12 m | 5.12 m | 5.12 m | 5.15 m | 3.87 ddd (9.3, 4.8, 4.3) |
| 9a | 2.67dd (15.0, 4.5) | 2.71 dd (15.0, 4.8) | 2.75 dd (14.5, 5.0) | 2.73 dd (14.5, 4.9) | 2.73 dd (14.5, 5.0) | 2.67 dd (13.8, 4.8) |
| 9b | 2.38 br d (15.0) | 2.42 br d (15.0) | 2.38 dd (14.5, 3.2) | 2.38 br d (14.5) | 2.40 dd (14.5, 2.9) | 2.25 dd (13.8, 4.3) |
| 13a | 6.07 d (2.8) | 6.10 d (3.3) | 6.11 d (3.0) | 6.18 d (2.5) | 6.13 d (3.1) | 6.16 d (3.2) |
| 13b | 5.59 d (2.8) | 5.57 d (3.3) | 5.45 d (3.0) | 5.59 d (2.5) | 5.65 d (3.1) | 6.14 d (3.2) |
| 14a | 5.08 br s | 5.10 br s | 5.12 br s | 5.15 br s | 5.17 br s | 5.09 br s |
| 14b | 4.75 br s | 4.77 br s | 4.85 br s | 4.91 br s | 4.92 br s | 4.97 br s |
| 15a | 4.14 d (11.7) | 4.19 d (11.7) | 4.02 d (11.8) | 5.44 br s | 5.44 br s | 5.40 br s |
| 15b | 3.80 d (11.7) | 3.83 d (11.7) | 3.81 d (11.8) | 5.34 br s | 5.34 br s | 5.31 br s |
| 3′a | 6.28 br s | 6.18 br s | 6.31 br s | 6.13 d (2.5) | 6.31 br s | - |
| 3′b | 5.94 br s | 5.73 br s | 5.97 br s | 5.73 d (2.5) | 5.97 br s | - |
| 4′ | 4.28 s (2H) | 1.98 s | 4.31 s (2H) | 1.97 s | 4.31s (2H) | - |
| Position | 1 | 2 | 3 | 5 | 6 |
|---|---|---|---|---|---|
| 1 | 48.73 | 48.95 | 46.90 | 46.18 | 45.93 |
| 2 | 39.80 | 40.02 | 39.15 | 40.01 | 40.01 |
| 3 | 76.94 | 76.99 | 77.81 | 74.14 | 74.11 |
| 4 | 85.77 | 85.84 | 85.82 | 154.04 | 154.26 |
| 5 | 59.39 | 59.64 | 57.53 | 52.02 | 51.81 |
| 6 | 78.45 | 78.52 | 78.84 | 80.30 | 80.87 |
| 7 | 47.43 | 47.60 | 48.17 | 48.45 | 51.66 |
| 8 | 75.47 | 75.57 | 75.63 | 75.63 | 73.07 |
| 9 | 35.74 | 35.80 | 37.48 | 37.66 | 42.84 |
| 10 | 144.71 | 145.05 | 144.68 | 144.04 | 144.63 |
| 11 | 139.11 | 139.52 | 139.23 | 139.71 | 140.53 |
| 12 | 170.97 | 170.84 | 171.11 | 171.21 | 172.0 |
| 13 | 122.53 | 122.01 | 122.45 | 122.39 | 122.95 |
| 14 | 117.71 | 117.48 | 117.27 | 118.15 | 117.03 |
| 15 | 50.04 | 50.08 | 64.35 | 112.72 | 112.15 |
| 1′ | 166.65 | 167.86 | 166.55 | 166.55 | - |
| 2′ | 141.61 | 137.76 | 141.91 | 141.92 | - |
| 3′ | 126.25 | 126.92 | 125.97 | 125.97 | - |
| 4′ | 61.53 | 18.31 | 61.62 | 61.63 | - |
| Inhibition of Cell Proliferation (%) ± SEM [Calculated IC50 (μM)] | ||||
|---|---|---|---|---|
| Compound | Conc. | SiHa | HeLa | MDA-MB-231 |
| Chlorojanerin (1) | 10 μM | 82.10 ± 0.97 | 51.83 ± 2.43 | 90.55 ± 1.27 |
| 30 μM | 92.93 ± 0.35 | 77.08 ± 0.80 | 92.00 ± 0.68 | |
| [6.71] | [11.37] | [2.21] | ||
| 19-Deoxychlorojanerin (2) | 10 μM | 90.93 ± 1.32 | 96.17 ± 0.38 | 96.14 ± 0.51 |
| 30 μM | 95.80 ± 0.15 | 96.14 ± 0.48 | 97.42 ± 0.36 | |
| [5.54] | [4.86] | [2.88] | ||
| 15-Hydroxyjanerin (3) | 10 μM | <10 | <10 | <10 |
| 30 μM | <10 | <10 | <10 | |
| Aguerin B (4) | 10 μM | 89.09 ± 1.54 | 28.87 ± 0.83 | 96.23 ± 0.45 |
| 30 μM | 97.92 ± 0.41 | 97.50 ± 0.31 | 96.64 ± 0.71 | |
| [5.70] | [12.64] | [4.26] | ||
| Cynaropicrin (5) | 10 μM | 63.03 ± 1.25 | 34.36 ± 2.37 | 92.16 ± 0.68 |
| 30 μM | 90.75 ± 0.55 | 68.26 ± 1.85 | 93.45 ± 0.27 | |
| [8.39] | [12.99] | [4.80] | ||
| Eleganin (6) | 10 μM | 64.97 ± 2.06 | 16.54 ± 1.48 | 89.44 ± 1.36 |
| 30 μM | 90.89 ± 0.43 | 73.99 ± 1.68 | 92.09 ± 0.9 | |
| [7.57] | [19.62] | [6.86] | ||
| Apigenin | 10 μM | <10* | 18.81 ± 0.80 | <10 |
| 30 μM | 29.92 ± 1.75 | 70.43 ± 1.09 | 28.42 ± 1.04 | |
| Jaceosidine | 10 μM | 18.88 ± 2.99 | <10 | <10 |
| 30 μM | 60.08 ± 0.74 | 75.18 ± 0.68 | 40.04 ± 1.68 | |
| 6-Methoxyluteolin | 10 μM | 14.16 ± 1.49 | 88.00 ± 0.63 | <10 |
| 30 μM | 35.20 ± 2.08 | 93.74 ± 0.65 | 26.55 ± 2.12 | |
| [4.50] | ||||
| Scopoletin | 10 μM | <10 | <10 | 12.09 ± 2.59 |
| 30 μM | 17.18 ± 2.18 | <10 | 15.85 ± 1.85 | |
| Cisplatin | 10 μM | 88.64 ± 0.50 | 42.61 ± 2.33 | 67.51 ± 1.01 |
| 30 μM | 90.18 ± 1.78 | 99.93 ± 0.26 | 87.75 ± 1.10 | |
| [7.84] | [12.43] | [3.74] | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tastan, P.; Hajdú, Z.; Kúsz, N.; Zupkó, I.; Sinka, I.; Kivcak, B.; Hohmann, J. Sesquiterpene Lactones and Flavonoids from Psephellus pyrrhoblepharus with Antiproliferative Activity on Human Gynecological Cancer Cell Lines. Molecules 2019, 24, 3165. https://doi.org/10.3390/molecules24173165
Tastan P, Hajdú Z, Kúsz N, Zupkó I, Sinka I, Kivcak B, Hohmann J. Sesquiterpene Lactones and Flavonoids from Psephellus pyrrhoblepharus with Antiproliferative Activity on Human Gynecological Cancer Cell Lines. Molecules. 2019; 24(17):3165. https://doi.org/10.3390/molecules24173165
Chicago/Turabian StyleTastan, Pelin, Zsuzsanna Hajdú, Norbert Kúsz, István Zupkó, Izabella Sinka, Bijen Kivcak, and Judit Hohmann. 2019. "Sesquiterpene Lactones and Flavonoids from Psephellus pyrrhoblepharus with Antiproliferative Activity on Human Gynecological Cancer Cell Lines" Molecules 24, no. 17: 3165. https://doi.org/10.3390/molecules24173165
APA StyleTastan, P., Hajdú, Z., Kúsz, N., Zupkó, I., Sinka, I., Kivcak, B., & Hohmann, J. (2019). Sesquiterpene Lactones and Flavonoids from Psephellus pyrrhoblepharus with Antiproliferative Activity on Human Gynecological Cancer Cell Lines. Molecules, 24(17), 3165. https://doi.org/10.3390/molecules24173165






