Simultaneous Photodiagnosis and Photodynamic Treatment of Metastatic Melanoma
Abstract
1. Introduction
2. Photodynamic Diagnosis and Treatment
2.1. Photodynamic Diagnosis (PDD) and Photodynamic Therapy (PDT)
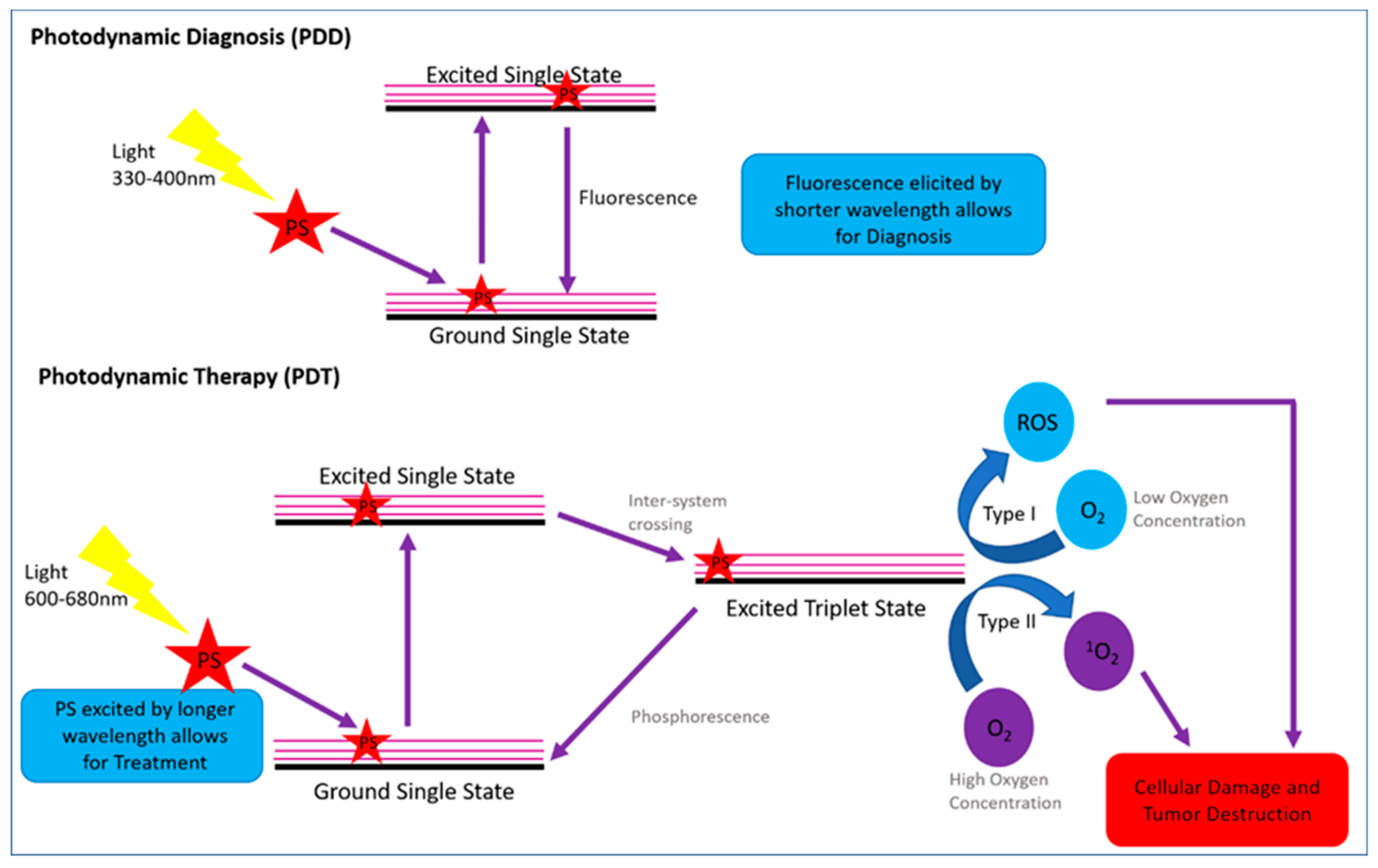
2.1.1. Photodynamic Modality Mechanism Action for PDD and PDT
2.1.2. Specific Challenges and Limitations of PDD and PDT in Relation to MM
2.2. Photosensitizers (PSs) for MM PDD and PDT Applications
2.2.1. First-Generation PSs for Simultaneous MM PDD and PDT Applications
2.2.2. Second-Generation PSs for Simultaneous MM PDD and PDT Applications
2.2.3. Third-Generation PSs for Simultaneous MM PDD and PDT Applications
2.3. Nanoparticles and Biomarkers for Enhanced PSs MM Uptake
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moody, J.A.; Botham, S.J.; Dahill, K.E.; Wallace, D.L.; Hardwicke, J.T. Complications following completion lymphadenectomy versus therapeutic lymphadenectomy for melanoma–A systematic review of the literature. Eur. J. Surg. Oncol. 2017, 43, 1760–1767. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Available online: https://www.who.int/uv/faq/skincancer/en/index1.html (accessed on 27 June 2019).
- Del Marmol, V.; Stratigos, A.J. New Frontiers in Skin Cancer. Curr. Opin. Oncol. 2019, 31, 53. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Available online: https://www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2018.html (accessed on 28 June 2019).
- Skin Cancer Foundation. Available online: https://www.skincancer.org/skin-cancer-information/skin-cancer-facts#melanoma (accessed on 3 July 2019).
- Bulir, D.; Liang, S.; O’Malley, M.; McWhirter, E. Update on Immunotherapeutics in the Management of Metastatic Melanoma. Skin Therapy Lett. 2019, 24, 8–11. [Google Scholar] [PubMed]
- Tas, F. Metastatic Behavior in Melanoma: Timing, Pattern, Survival, and Influencing Factors. J. Oncol. 2012, 1, 9. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, C.; Kruger, C.A.; Abrahamse, H. Photodynamic therapy for metastatic melanoma treatment: A review. Technol. Cancer Res. Treat. 2018, 17. [Google Scholar] [CrossRef] [PubMed]
- Moura, N.; Veras, R.; Aires, K.; Machado, V.; Silva, R.; Araújo, F.; Claro, M. ABCD rule and pre-trained CNNs for melanoma diagnosis. Multimed. Tools Appl. 2019, 78, 6869–6888. [Google Scholar] [CrossRef]
- Danielsen, M.; Kjaer, A.; Wu, M.; Martineau, L.; Nosrati, M.; Leong, S.P.; Sagebiel, R.W.; Miller III, J.R.; Kashani-Sabet, M. Prediction of positron emission tomography/computed tomography (PET/CT) positivity in patients with high-risk primary melanoma. Am. J. Nucl. Med. Mol. Imaging 2016, 6, 277–285. [Google Scholar]
- Huang, S.K.; Hoon, D.S. Liquid biopsy utility for the surveillance of cutaneous malignant melanoma patients. Mol. Oncol. 2016, 10, 450–463. [Google Scholar] [CrossRef]
- Murchie, P.; Raja, E.A.; Brewster, D.H.; Iversen, L.; Lee, A.J. Is initial excision of cutaneous melanoma by General Practitioners (GPs) dangerous? Comparing patient outcomes following excision of melanoma by GPs or in hospital using national datasets and meta-analysis. Eur. J. Cancer 2017, 86, 373–384. [Google Scholar] [CrossRef][Green Version]
- Ng, P.C.; Barzilai, D.A.; Ismail, S.A.; Averitte Jr, R.L.; Gilliam, A.C. Evaluating invasive cutaneous melanoma: Is the initial biopsy representative of the final depth. J. Am. Acad. Dermatol. 2003, 48, 420–424. [Google Scholar] [CrossRef]
- Doubrovsky, A.; Scolyer, R.A.; Murali, R.; McKenzie, P.R.; Watson, G.F.; Lee, C.S.; McLeod, D.J.; McCarthy, W.H.; Uren, R.F.; Stretch, J.R.; et al. Diagnostic accuracy of fine needle biopsy for metastatic melanoma and its implications for patient management. Ann. Surg. Oncol. 2008, 15, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Kell, M.R.; Kerin, M.J. Sentinel lymph node biopsy. BMJ 2004, 328, 1330. [Google Scholar] [CrossRef] [PubMed]
- Gandini, S.; Ferrucci, P.F.; Botteri, E.; Tosti, G.; Barberis, M.; Pala, L.; Battaglia, A.; Clerici, A.; Spadola, G.; Cocorocchio, E.; et al. Prognostic significance of hematological profiles in melanoma patients. Int. J. Cancer 2016, 139, 1618–1625. [Google Scholar] [CrossRef] [PubMed]
- Timerman, D.; McEnery-Stonelake, M.; Joyce, C.J.; Nambudiri, V.E.; Stephen, F.H.; Claus, E.B.; Ibrahim, N.; Lin, J.Y. Vitamin D deficiency is associated with a worse prognosis in metastatic melanoma. Oncotarget 2017, 8, 6873. [Google Scholar] [CrossRef] [PubMed]
- Palmer, S.R.; Erickson, L.A.; Ichetovkin, I.; Knauer, D.J.; Markovic, S.N. Circulating serologic and molecular biomarkers in malignant melanoma. Mayo Clin. Proc. 2011, 86, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Alrabadi, N.; Gibson, N.; Curless, K.; Cheng, L.; Kuhar, M.; Chen, S.; Warren, S.J.; Alomari, A.K. Detection of driver mutations in BRAF can aid in diagnosis and early treatment of dedifferentiated metastatic melanoma. Mod. Pathol. 2018, 32, 330–337. [Google Scholar] [CrossRef]
- Ross, C.L.; Kaushik, S.; Valdes-Rodriguez, R.; Anvekar, R. MicroRNAs in cutaneous melanoma: Role as diagnostic and prognostic biomarkers. J. Cell. Physiol. 2018, 233, 5133–5141. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Pick, H.; Gasilova, N.; Li, X.; Lin, T.E.; Laeubli, H.P.; Zippelius, A.; Ho, P.C.; Girault, H.H. MALDI detection of exosomes: A potential tool for cancer studies. Chem 2019, 5, 1318–1336. [Google Scholar] [CrossRef]
- Hong, H.; Sun, J.; Cai, W. Anatomical and molecular imaging of skin cancer. Clin. Cosmet. Investig. Dermatol. 2008, 1, 1–17. [Google Scholar]
- Histed, S.N.; Lindenberg, M.L.; Mena, E.; Turkbey, B.; Choyke, P.L.; Kurdziel, K.A. Review of functional/anatomical imaging in oncology. Nucl. Med. Commun. 2012, 4, 349–361. [Google Scholar] [CrossRef]
- Huang, Y.; He, S.; Cao, W.; Cai, K.; Liang, X.J. Biomedical nanomaterials for imaging-guided cancer therapy. Nanoscale 2012, 4, 6135–6149. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Masamune, K. Flexible laser endoscope for minimally invasive photodynamic diagnosis (PDD) and therapy (PDT) toward efficient tumor removal. Opt. Express 2017, 25, 16795–16812. [Google Scholar] [CrossRef] [PubMed]
- Shanbhag, P.P.; Jog, S.V.; Chogale, M.M.; Gaikwad, S.S. Theranostics for cancer therapy. Curr. Drug Deliv. 2013, 10, 357–362. [Google Scholar] [CrossRef]
- He, J.; Yang, L.; Yi, W.; Fan, W.; Wen, Y.; Miao, X.; Xiong, L. Combination of fluorescence-guided surgery with photodynamic therapy for the treatment of cancer. Mol. Imaging 2017, 16, 1536012117722911. [Google Scholar] [CrossRef] [PubMed]
- Lipiński, M.I.; Różański, W.; Markowski, M.P. Photodynamic diagnosis - current tool in diagnosis of carcinoma in situ of the urinary bladder. Contemp. Oncol. 2015, 19, 341–342. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.Y.; Liao, Y.H.; Tan, G.S.; Wang, X.M.; Lu, G.H.; Yang, Y.H. Targeted photosensitizer nanoconjugates based on human serum albumin selectively kill tumor cells upon photo-irradiation. RSC Adv. 2015, 5, 50572–50579. [Google Scholar] [CrossRef]
- Zhu, T.C.; Finlay, J.C. The role of photodynamic therapy (PDT) physics. Med. Phys. 2008, 7, 3127–3136. [Google Scholar] [CrossRef]
- Mokwena, M.G.; Kruger, C.A.; Tygna, I.M.; Abrahamse, H. A review of nanoparticle photosensitizer drug delivery uptake systems for photodynamic treatment of lung cancer. Photodiagn. Photodyn. 2018, 22, 147–154. [Google Scholar] [CrossRef]
- Kruger, C.; Abrahamse, H. Utilisation of Targeted Nanoparticle Photosensitiser Drug Delivery Systems for the Enhancement of Photodynamic Therapy. Molecules 2018, 23, 2628. [Google Scholar] [CrossRef]
- Dobson, J.; de Queiroz, G.F.; Golding, J.P. Photodynamic therapy and diagnosis: Principles and comparative aspects. Vet. J. 2018, 233, 8–18. [Google Scholar] [CrossRef]
- Oniszczuk, A.; Wojtunik-Kulesza, K.A.; Oniszczuk, T.; Kasprzak, K. The potential of photodynamic therapy (PDT)—Experimental investigations and clinical use. Biomed. Pharmacother. 2016, 83, 912–929. [Google Scholar] [CrossRef] [PubMed]
- Abrahamse, H.; Kruger, C.A.; Kadanyo, S.; Mishra, A. Nanoparticles for advanced photodynamic therapy of cancer. Photomed. Laser Surg. 2017, 35, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.; Pera, P.; Joshi, P.; Dukh, M.; Tabaczynski, W.A.; Siters, K.E.; Kryman, M.; Cheruku, R.R.; Durrani, F.; Missert, J.R.; et al. Highly effective dual-function near-infrared (NIR) photosensitizer for fluorescence imaging and photodynamic therapy (PDT) of cancer. J. Med. Chem. 2016, 59, 9774–9787. [Google Scholar] [CrossRef] [PubMed]
- Lucky, S.S.; Muhammad Idris, N.; Li, Z.; Huang, K.; Soo, K.C.; Zhang, Y. Titania coated upconversion nanoparticles for near-infrared light triggered photodynamic therapy. ACS Nano 2015, 9, 191–205. [Google Scholar] [CrossRef] [PubMed]
- Slominski, R.M.; Zmijewski, M.A.; Slominski, A.T. The role of melanin pigment in melanoma. Exp. Dermatol. 2015, 24, 258–259. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Song, J.; Nie, L.; Chen, X. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 2016, 45, 6597–6626. [Google Scholar] [CrossRef] [PubMed]
- Benov, L. Photodynamic therapy: Current status and future directions. Med. Princ. Pract. 2015, 24, 14–28. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.O.; Ha, K.S. New insights into the mechanisms for photodynamic therapy-induced cancer cell death. Int. Rev. Cell Mol. Biol. 2012, 295, 139–174. [Google Scholar] [CrossRef]
- Allison, R.R.; Sibata, C.H. Oncologic photodynamic therapy photosensitizers: A clinical review. Photodiagn. Photodyn. 2010, 7, 61–75. [Google Scholar] [CrossRef]
- Abrahamse, H.; Hamblin, M.R. New photosensitizers for photodynamic therapy. Biochem. J. 2016, 473, 347–364. [Google Scholar] [CrossRef]
- Inoue, K.; Fukuhara, H.; Shimamoto, T.; Kamada, M.; Iiyama, T.; Miyamura, M.; Kurabayashi, A.; Furihata, M.; Tanimura, M.; Watanabe, H.; et al. Comparison between intravesical and oral administration of 5-aminolevulinic acid in the clinical benefit of photodynamic diagnosis for nonmuscle invasive bladder cancer. Cancer 2012, 118, 1062–1074. [Google Scholar] [CrossRef] [PubMed]
- Ndhundhuma, I.M.; Abrahamse, H. A Review of the Photodynamic Application of 5-Aminolevulinic acid, Hypericin and Phthalocyanines in dermatology. Med. Technol. SA 2015, 29, 20–26. [Google Scholar]
- Fonda-Pascual, P.; Moreno-Arrones, O.M.; Alegre-Sanchez, A.; Saceda-Corralo, D.; Buendia-Castaño, D.; Pindado-Ortega, C.; Fernandez-Gonzalez, P.; Velazquez-Kennedy, K.; Calvo-Sánchez, M.I.; Harto-Castaño, A.; et al. In situ production of ROS in the skin by photodynamic therapy as a powerful tool in clinical dermatology. Methods 2016, 109, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Martinez De Pinillos Bayona, A.; Mroz, P.; Thunshelle, C.; Hamblin, M.R. Design features for optimization of tetrapyrrole macrocycles as antimicrobial and anticancer photosensitizers. Chem. Biol Drug Des. 2017, 89, 192–206. [Google Scholar] [CrossRef]
- Jocham, D.; Stepp, H.; Waidelich, R. Photodynamic diagnosis in urology: State-of-the-art. Eur. Urol. 2008, 53, 1138–1150. [Google Scholar] [CrossRef]
- Sharma, K.V.; Davids, L.M. Hypericin-PDT-induced rapid necrotic death in human squamous cell carcinoma cultures after multiple treatment. Cell Biol. Int. 2012, 36, 1261–1266. [Google Scholar] [CrossRef]
- Kleemann, B.; Loos, B.; Scriba, T.J.; Lang, D.; Davids, L.M. St John’s Wort (Hypericum perforatum L.) photomedicine: Hypericin-photodynamic therapy induces metastatic melanoma cell death. PLoS ONE 2014, 9, 103762. [Google Scholar] [CrossRef]
- Josefsen, L.B.; Boyle, R.W. Photodynamic therapy and the development of metal-based photosensitisers. Met. Based Drugs 2008. [Google Scholar] [CrossRef]
- Silva, C.O.; Martinho, N.; Aniceto, N.; Reis, C.P. Melanoma prevention: Challenges and progresses in nanotechnology for melanoma prevention and treatment. In CRC Concise Encyclopedia of Nanotechnology; Kharisov, B.I., Kharissova, O.V., Ortiz-Mendez, U., Eds.; 2016; pp. 453–465. [Google Scholar]
- Luo, D.; Carter, K.A.; Miranda, D.; Lovell, J.F. Chemophototherapy: An Emerging Treatment Option for Solid Tumors. Adv. Sci. (Weinh.) 2016, 4, 1600106. [Google Scholar] [CrossRef]
- Conde, J.; Doria, G.; Baptista, P. Noble metal nanoparticles applications in cancer. J. Drug Deliv. 2012. [Google Scholar] [CrossRef]
- Buríková, M.; Bilcik, B.; Macajova, M.; Vyboh, P.; Bizik, J.; Mateasik, A.; Miskovsky, P.; Cavarga, I. Hypericin fluorescence kinetics in the presence of low-density lipoproteins: Study on quail CAM assay for topical delivery. Gen. Physiol. Biophys. 2016, 33, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Naves, L.B.; Dhand, C.; Venugopal, J.R.; Rajamani, L.; Ramakrishna, S.; Almeida, L. Nanotechnology for the treatment of melanoma skin cancer. Progr. Biomater. 2017, 6, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Pelaz, B.; Alexiou, C.; Alvarez-Puebla, R.A.; Alves, F.; Andrews, A.M.; Ashraf, S.; Balogh, L.P.; Ballerini, L.; Bestetti, A.; Brendel, C.; et al. Diverse applications of nanomedicine. ACS Nano 2017, 11, 2313–2381. [Google Scholar] [CrossRef] [PubMed]
- Bosserhoff, A.K.; Buettner, R. Expression, function and clinical relevance of MIA (melanoma inhibitory activity). Histol. Histopathol. 2002, 17, 289–300. [Google Scholar] [PubMed]
- Tang, J.Q.; Hou, X.Y.; Yang, C.S.; Li, Y.X.; Xin, Y.; Guo, W.W.; Wei, Z.P.; Liu, Y.Q.; Jiang, G. Recent developments in nanomedicine for melanoma treatment. Int. J. Cancer 2017, 141, 646–653. [Google Scholar] [CrossRef]
- Li, D.; He, Q.; Li, J. Smart core/shell nanocomposites: Intelligent polymers modified gold nanoparticles. Adv. Colloid Interfac. 2009, 149, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Kaleta-Richter, M.; Kawczyk-Krupka, A.; Aebisher, D.; Bartusik-Aebisher, D.; Czuba, Z.; Cieślar, G. The capabilities and hope of the combination the new forms of personalized colon cancer treatment–immunotherapy and immune photodynamic therapy. Photodiagn. Photodyn. 2019, 25, 253–258. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naidoo, C.; Kruger, C.A.; Abrahamse, H. Simultaneous Photodiagnosis and Photodynamic Treatment of Metastatic Melanoma. Molecules 2019, 24, 3153. https://doi.org/10.3390/molecules24173153
Naidoo C, Kruger CA, Abrahamse H. Simultaneous Photodiagnosis and Photodynamic Treatment of Metastatic Melanoma. Molecules. 2019; 24(17):3153. https://doi.org/10.3390/molecules24173153
Chicago/Turabian StyleNaidoo, Channay, Cherie Ann Kruger, and Heidi Abrahamse. 2019. "Simultaneous Photodiagnosis and Photodynamic Treatment of Metastatic Melanoma" Molecules 24, no. 17: 3153. https://doi.org/10.3390/molecules24173153
APA StyleNaidoo, C., Kruger, C. A., & Abrahamse, H. (2019). Simultaneous Photodiagnosis and Photodynamic Treatment of Metastatic Melanoma. Molecules, 24(17), 3153. https://doi.org/10.3390/molecules24173153






