Preparation of Chitosan–Hexaconazole Nanoparticles as Fungicide Nanodelivery System for Combating Ganoderma Disease in Oil Palm
Abstract
1. Introduction
2. Results and Discussions
2.1. Nanoparticle Characterizations
2.1.1. Reaction Yield, Hexaconazole Loading Content, and Encapsulation Efficiency
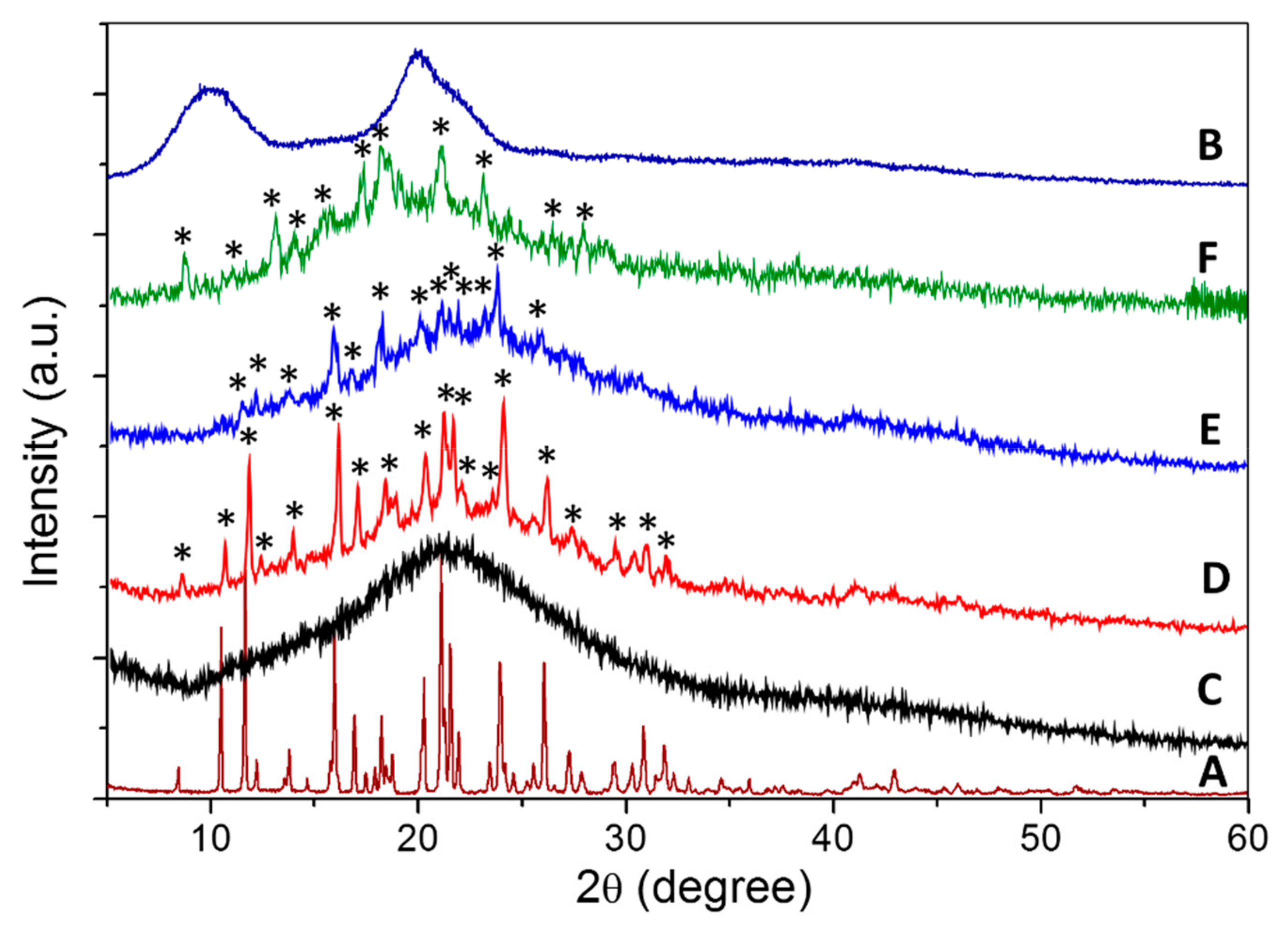
2.1.2. Powder X-Ray Diffraction
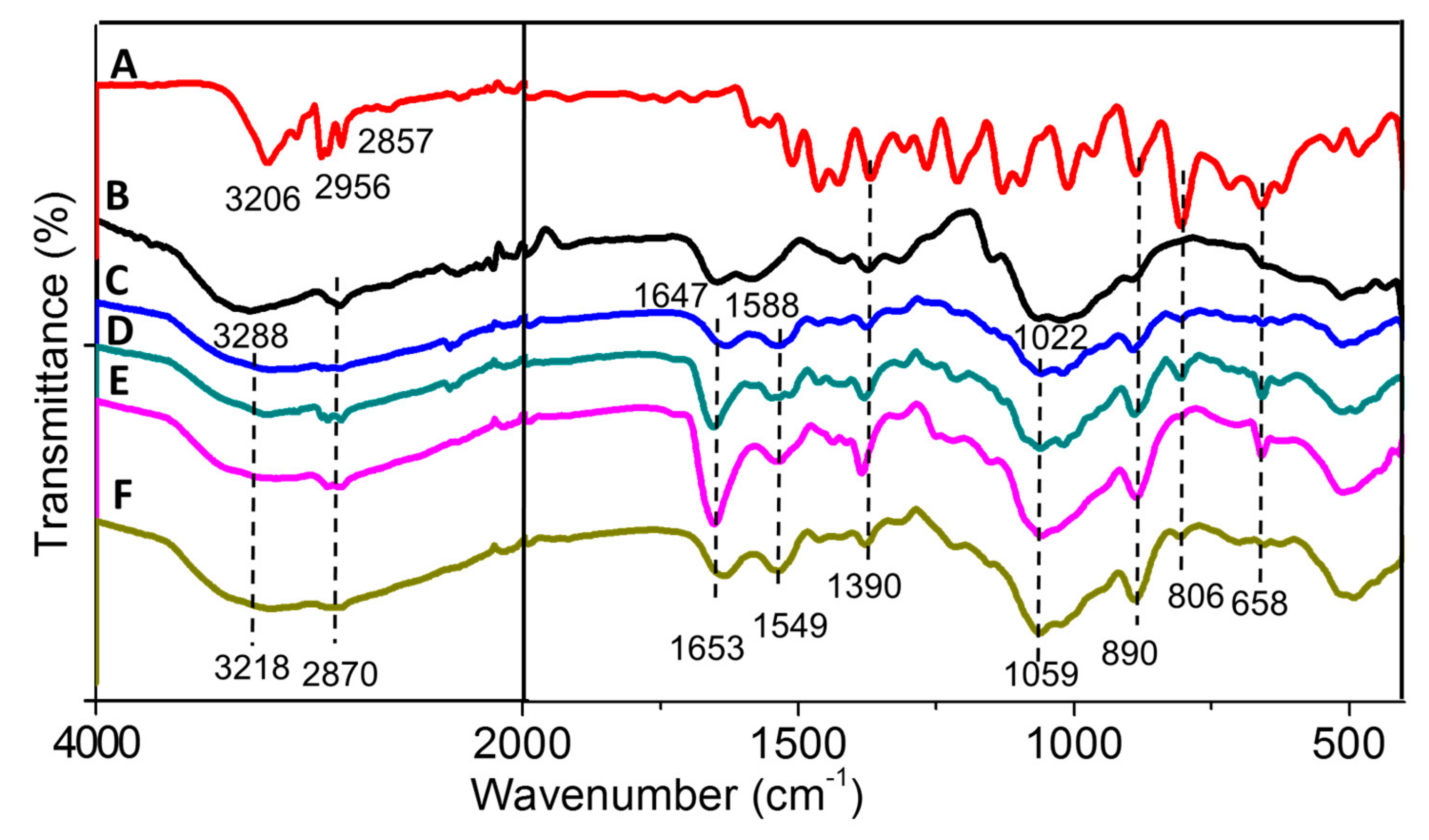
2.1.3. FTIR Spectroscopy
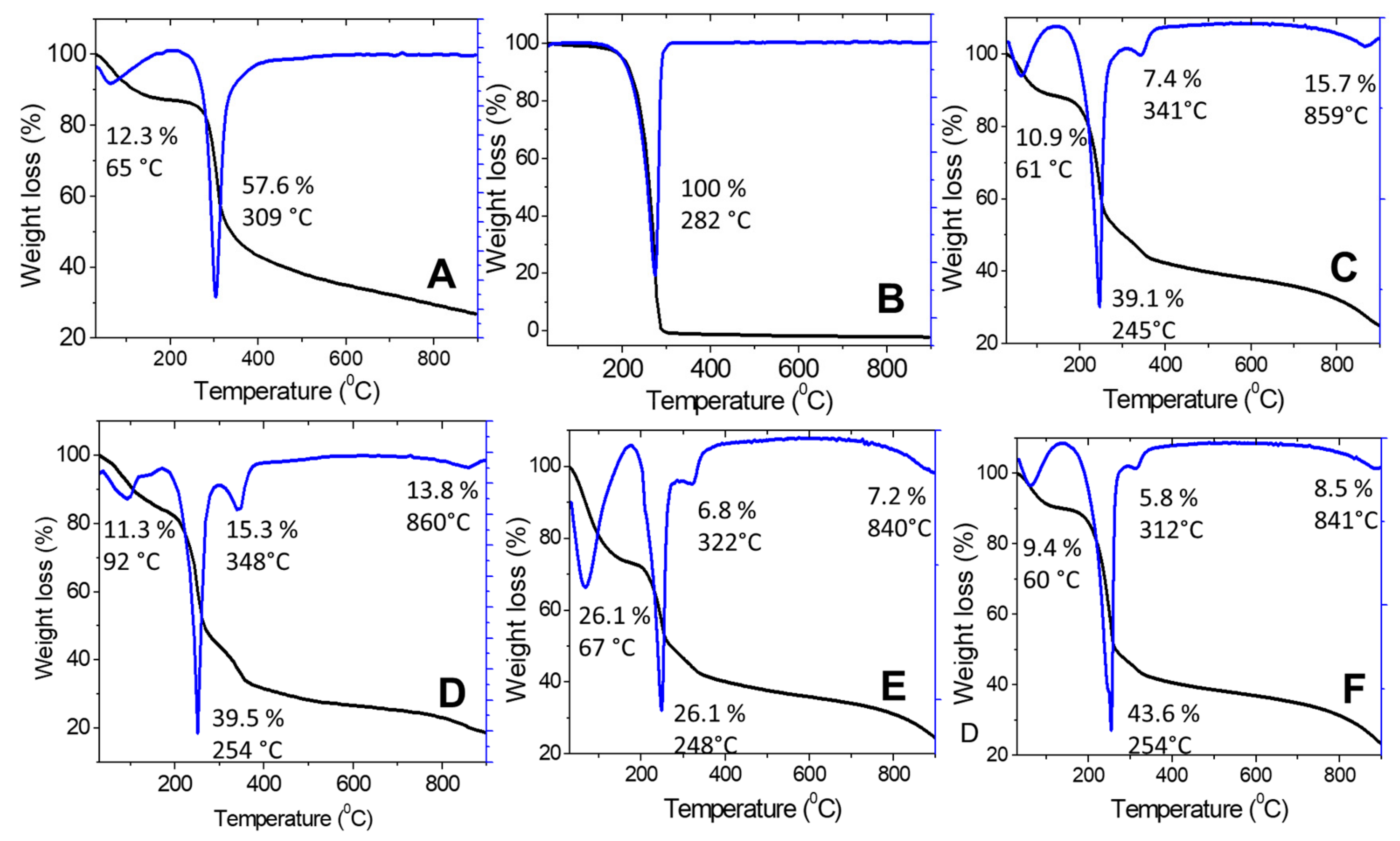
2.1.4. Thermal Analysis
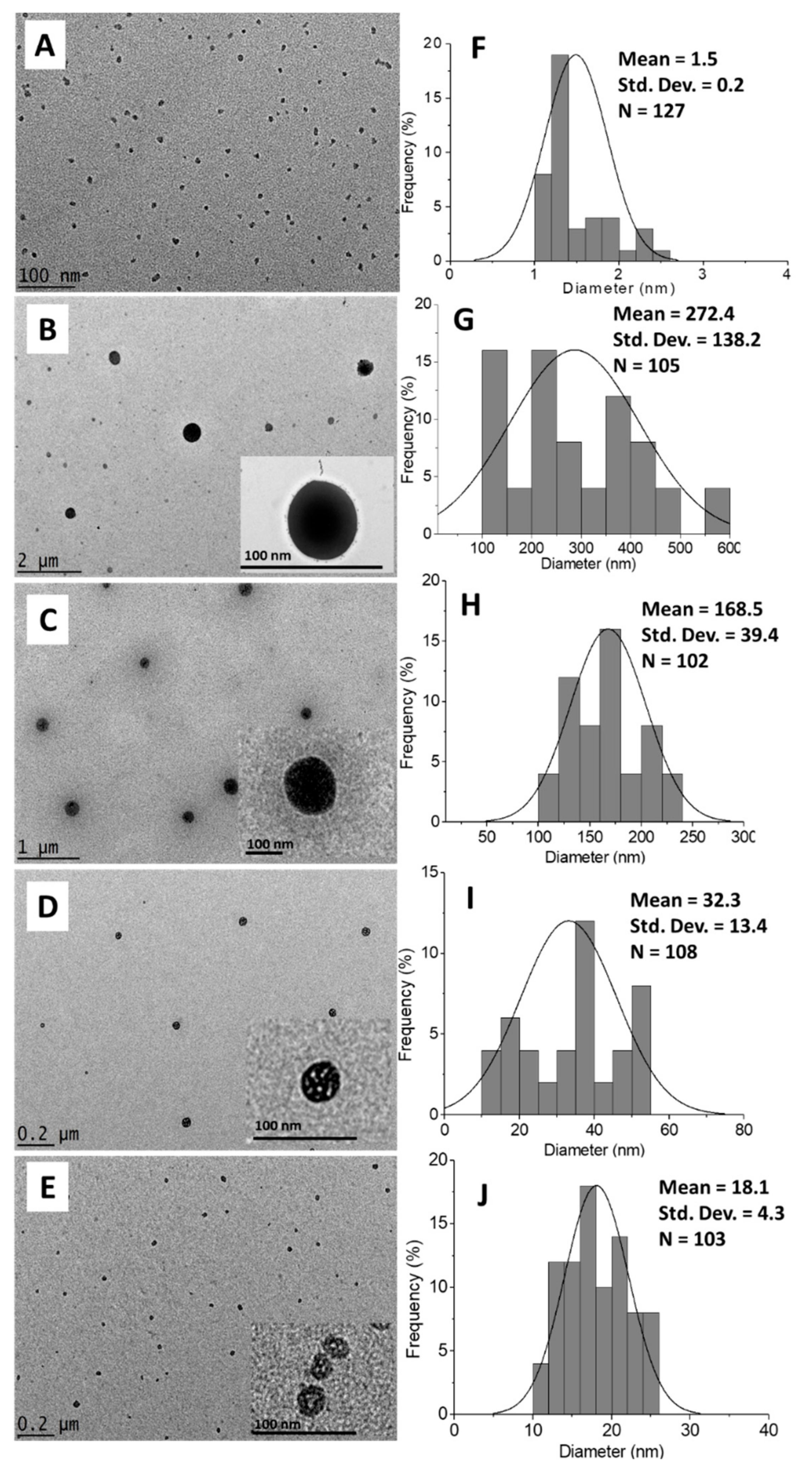
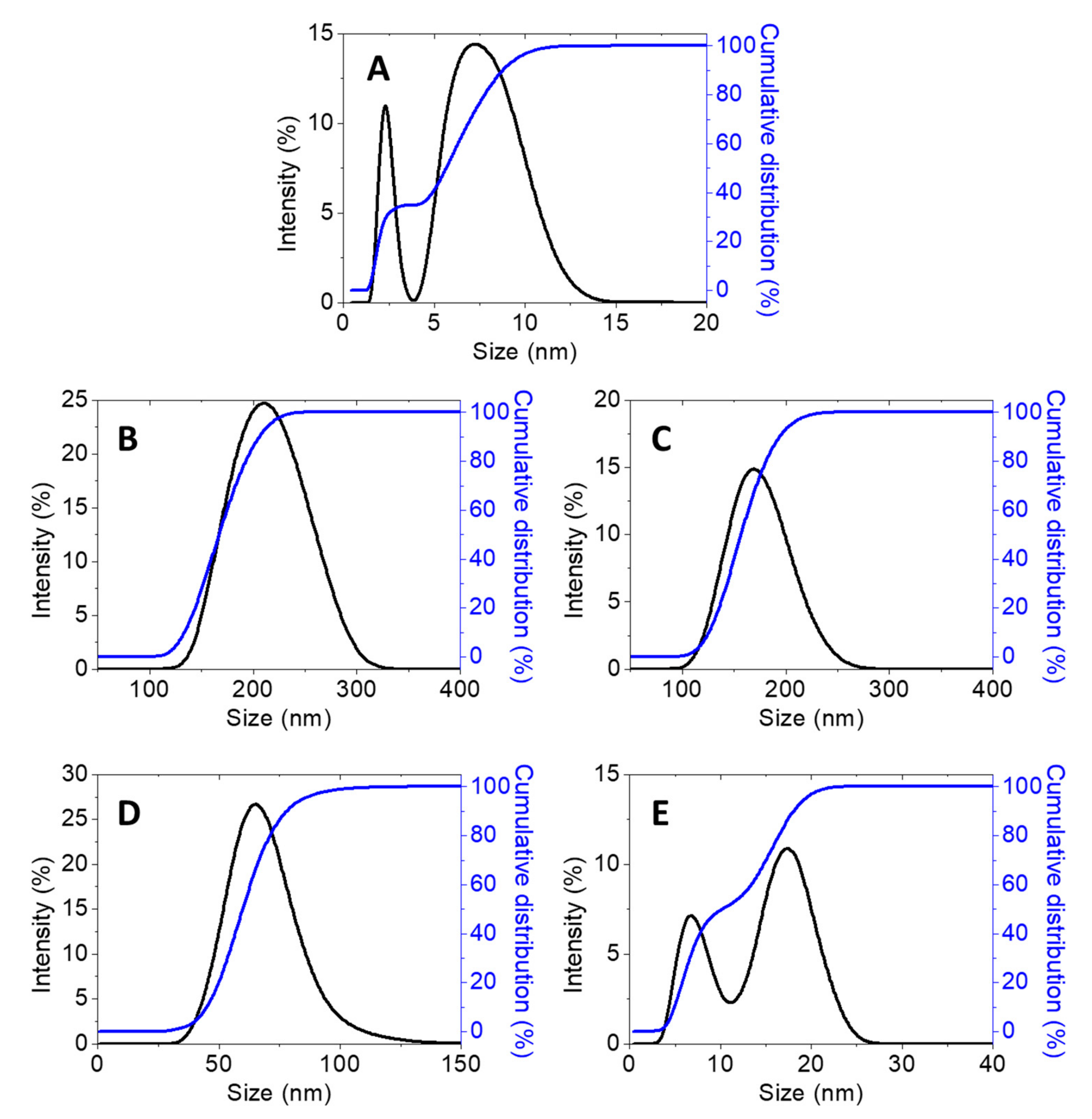
2.1.5. Morphology and Particle Size Distribution
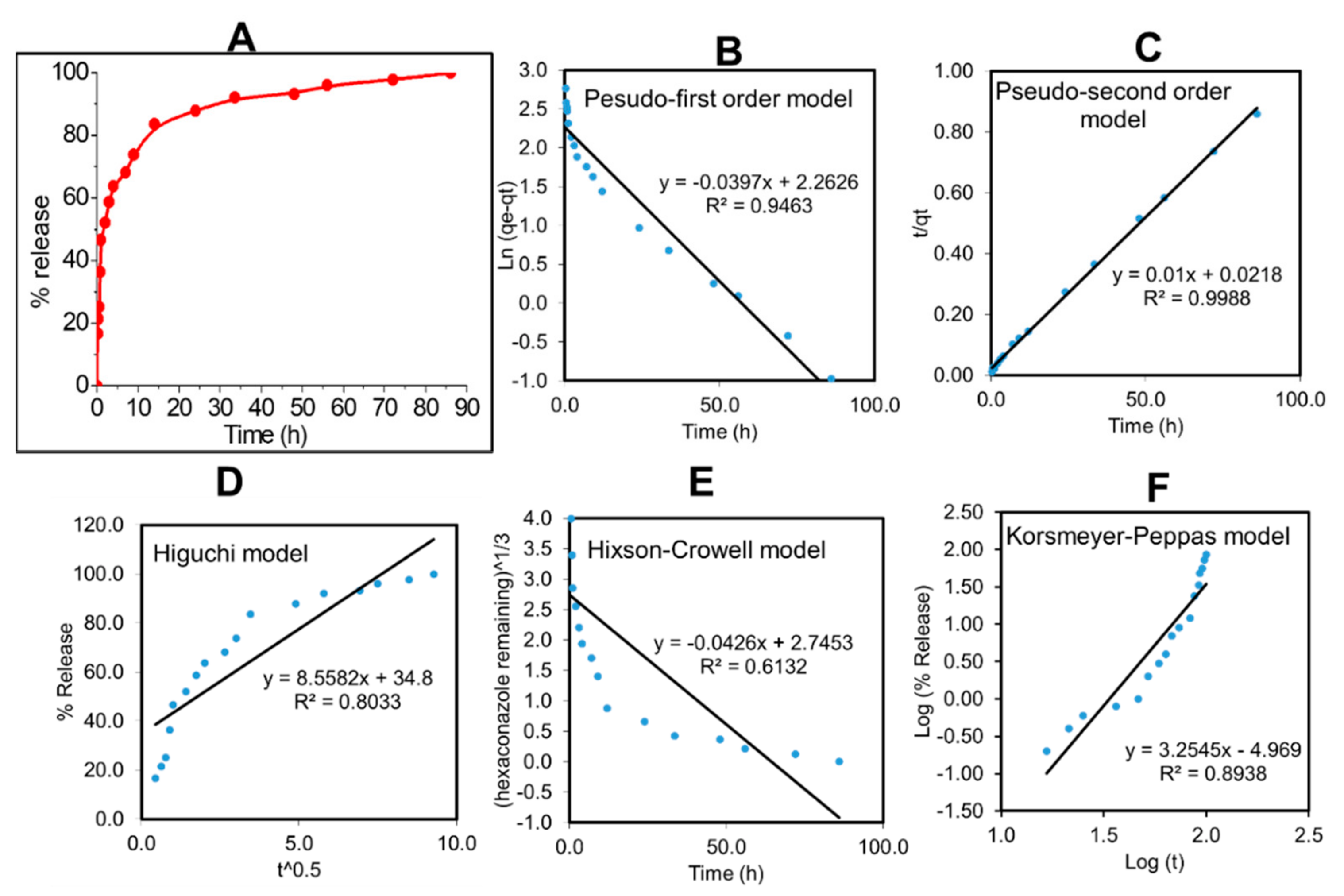
2.2. In Vitro Hexaconazole Release
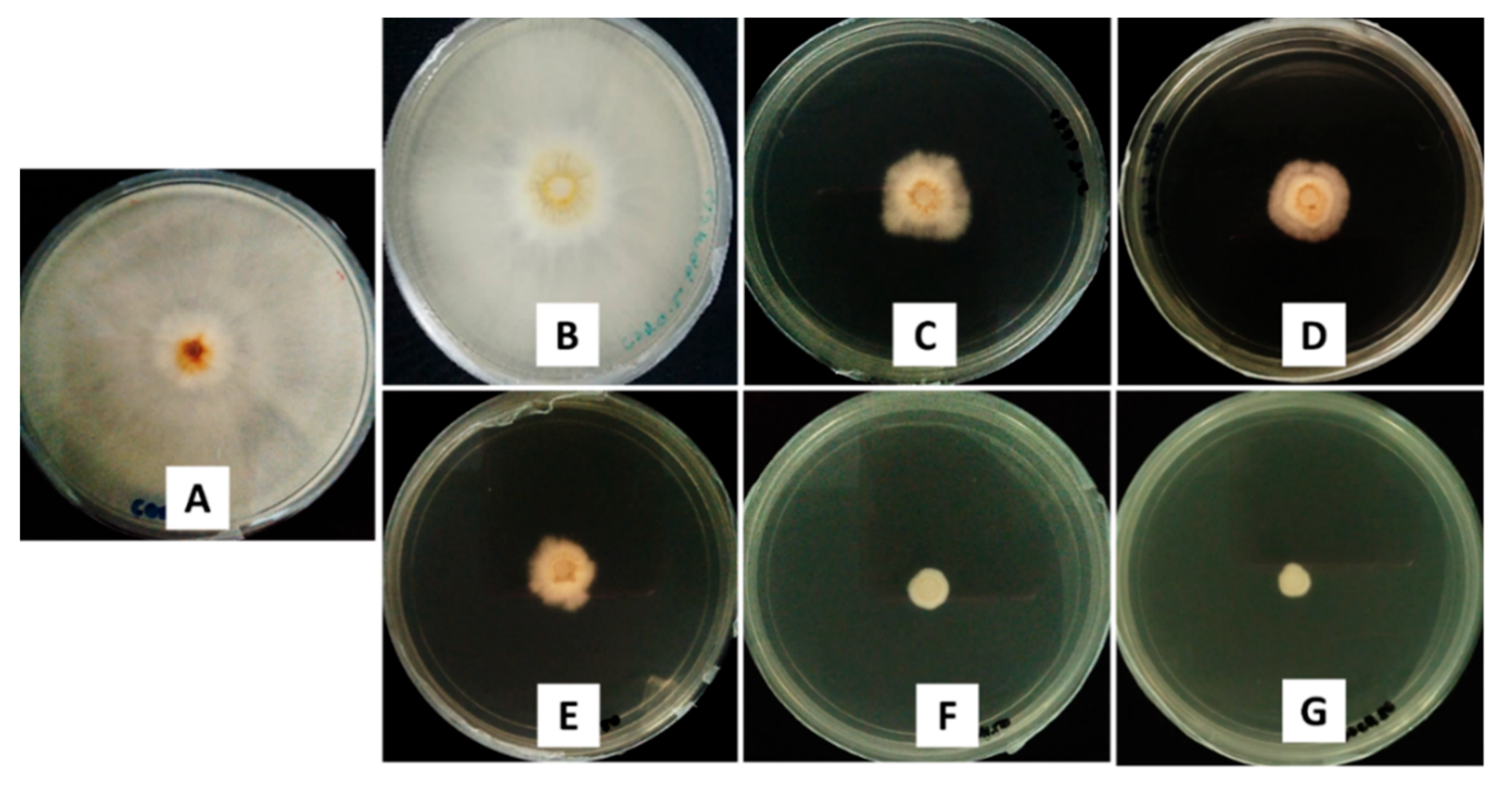
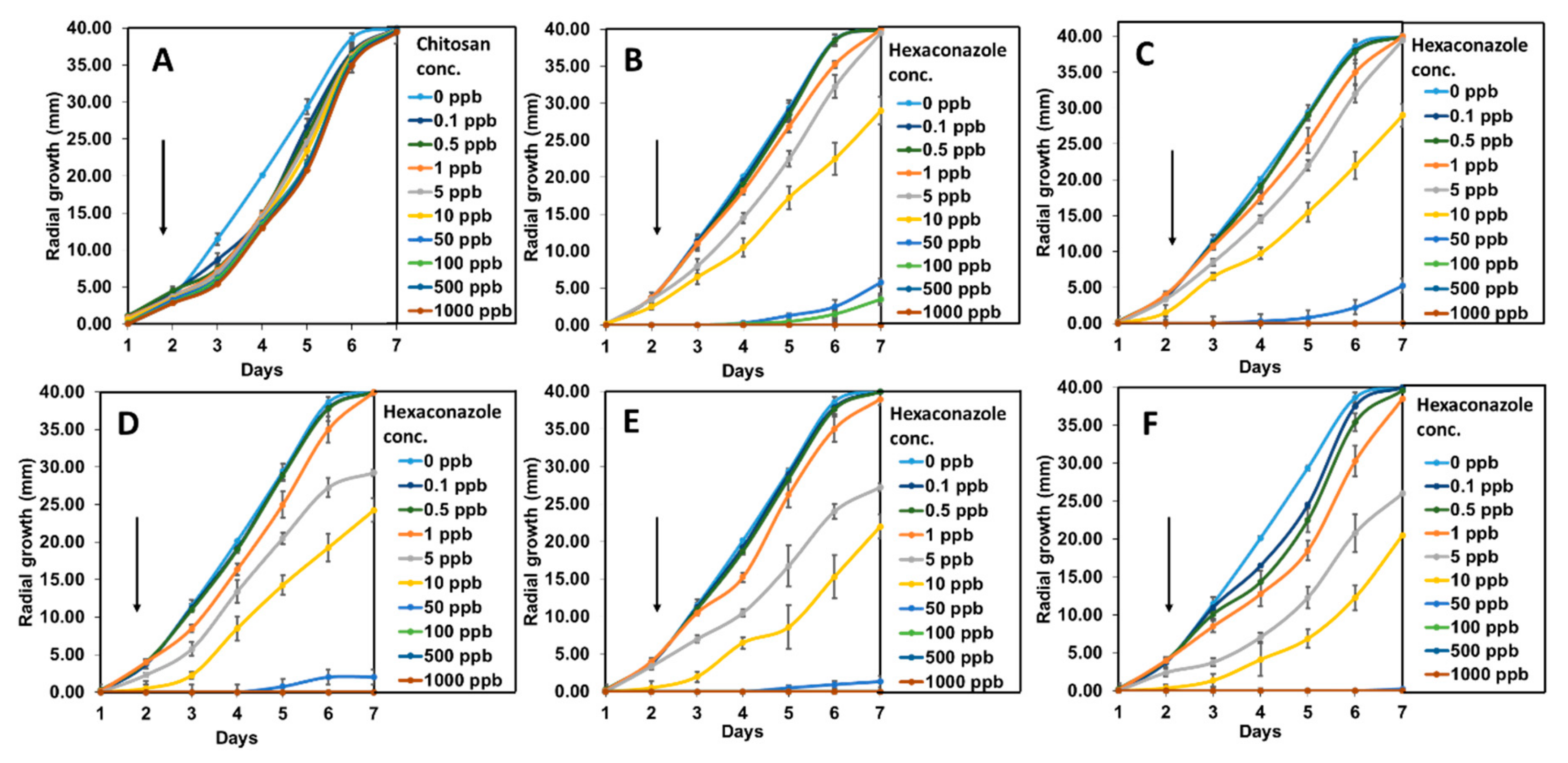
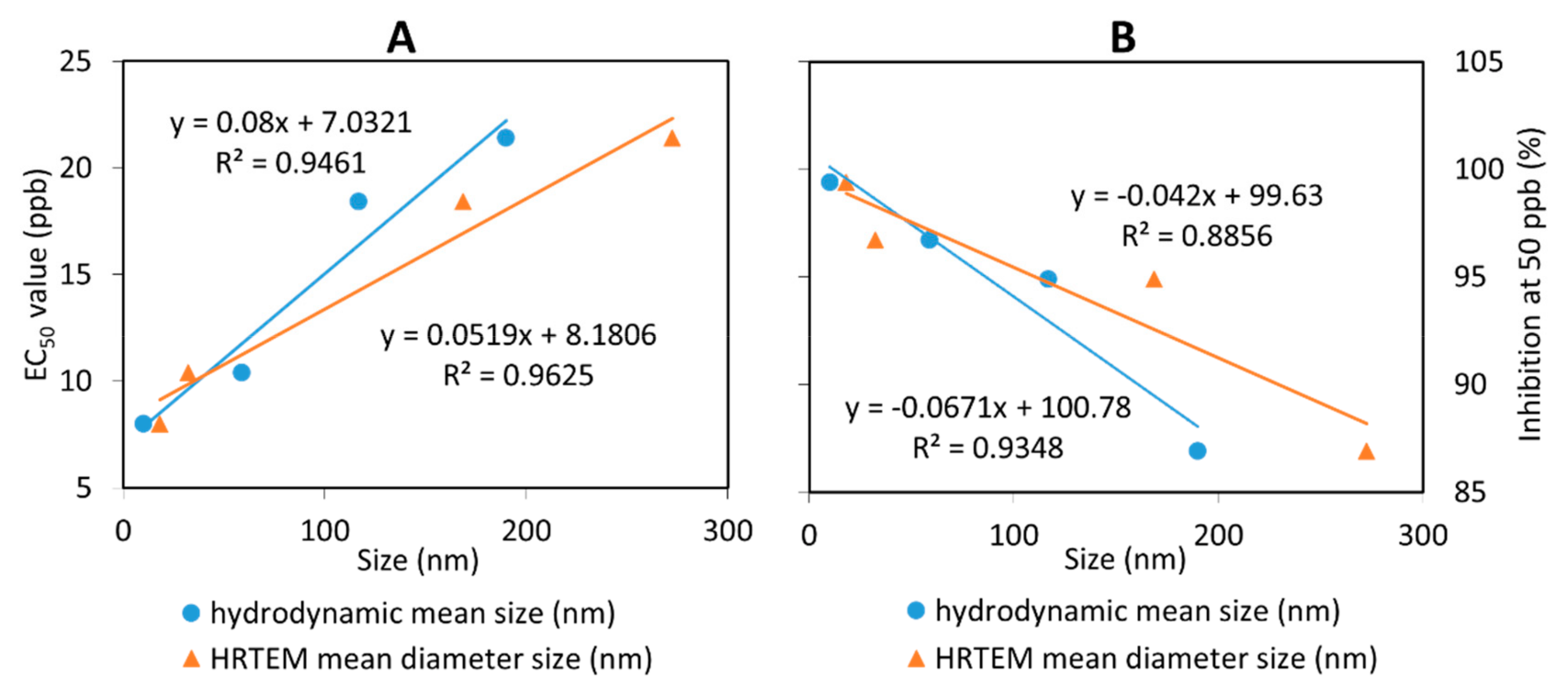
2.3. In Vitro Antifungal Activity Assay on G. boninense
3. Materials and Methods
3.1. Materials
3.2. Preparation of Chitosan and Its Encapsulation of Hexaconazole Nanoparticles
3.3. Reaction Yield, Hexaconazole Loading Content, and Encapsulation Efficiency
3.4. Characterizations
3.5. Hexaconazole Release Profile Study
3.6. In Vitro Antifungal Assay
3.7. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nair, R.; Varghese, S.H.; Nair, B.G.; Maekawa, T.; Yoshida, Y.; Kumar, D.S. Nanoparticulate material delivery to plants. Plant Sci. 2010, 179, 154–163. [Google Scholar] [CrossRef]
- Sabir, S.; Arshad, M.; Chaudhari, S.K. Zinc oxide nanoparticles for revolutionizing agriculture: Synthesis and applications. Sci. World J. 2014, 2014, 925494. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.S. Nanotechnology in agriculture: Prospects and constraints. Nanotechnol. Sci. Appl. 2014, 7, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Campos, E.V.R.; de Oliveira, J.L.; Fraceto, L.F. Applications of controlled release systems for fungicides, herbicides, acaricides, nutrients, and plant growth hormones: A review. Adv. Sci. Eng. Med. 2014, 6, 373–387. [Google Scholar] [CrossRef]
- Prasad, R.; Kumar, V.; Prasad, K.S. Nanotechnology in sustainable agriculture: Present concerns and future aspects. Afr. J. Biotechnol. 2014, 13, 705–713. [Google Scholar]
- Mousavi, S.R.; Rezaei, M. Nanotechnology in agriculture and food production. J. Appl. Environ. Biol. Sci. 2011, 1, 414–419. [Google Scholar]
- Ghormade, V.; Deshpande, M.V.; Paknikar, K.M. Perspectives for nano-biotechnology enabled protection and nutrition of plants. Biotechnol. Adv. 2011, 29, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Rai, M.; Ingle, A. Role of nanotechnology in agriculture with special reference to management of insect pests. Appl. Microbiol. Biotechnol. 2012, 94, 287–293. [Google Scholar] [CrossRef]
- Jha, Z.; Behar, N.; Sharma, S.N.; Chandel, G.; Sharma, D.; Pandey, M. Nanotechnology: Prospects of agricultural advancement. Nano Vision 2011, 1, 88–100. [Google Scholar]
- Manjunatha, S.; Biradar, D.; Aladakatti, Y. Nanotechnology and its applications in agriculture: A review. J. Farm Sci. 2016, 29, 1–13. [Google Scholar]
- Nanoscience in Food and Agriculture; Ranjan, S., Dasgupta, N., Lichtfouse, E., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 20. [Google Scholar]
- Yamamoto, C.F.; Pereira, E.I.; Mattoso, L.H.; Matsunaka, T.; Ribeiro, C. Slow release fertilizers based on urea/urea–formaldehyde polymer nanocomposites. Chem. Eng. J. 2016, 287, 390–397. [Google Scholar] [CrossRef]
- Pereira, E.I.; Da Cruz, C.C.; Solomon, A.; Le, A.; Cavigelli, M.A.; Ribeiro, C. Novel slow-release nanocomposite nitrogen fertilizers: The impact of polymers on nanocomposite properties and function. Ind. Eng. Chem. Res. 2015, 54, 3717–3725. [Google Scholar] [CrossRef]
- De Britto, D.; de Moura, M.R.; Aouada, F.A.; Pinola, F.G.; Lundstedt, L.M.; Assis, O.B.; Mattoso, L.H. Entrapment characteristics of hydrosoluble vitamins loaded into chitosan and N,N,N-trimethyl chitosan nanoparticles. Macromol. Res. 2014, 22, 1261–1267. [Google Scholar] [CrossRef]
- Giroto, A.S.; Guimarães, G.G.; Foschini, M.; Ribeiro, C. Role of slow-release nanocomposite fertilizers on nitrogen and phosphate availability in soil. Sci. Rep. 2017, 7, 46032. [Google Scholar] [CrossRef] [PubMed]
- Ditta, A. How helpful is nanotechnology in agriculture? Adv. Nat. Sci. Nanosci. Nanotech. 2012, 3, 033002. [Google Scholar] [CrossRef]
- Wanyika, H.; Gatebe, E.; Kioni, P.; Tang, Z.; Gao, Y. Mesoporous silica nanoparticles carrier for urea: Potential applications in agrochemical delivery systems. Journal Nanosci. Nanotechnol. 2012, 12, 2221–2228. [Google Scholar] [CrossRef]
- Chen, H.; Yada, R. Nanotechnologies in agriculture: New tools for sustainable development. Trends Food Sci. Technol. 2011, 22, 585–594. [Google Scholar] [CrossRef]
- Benjamin, M.; Chee, K. Basal stem rot of oil palm-a serious problem on inland soils. MAPPS Newsl. 1995, 19. [Google Scholar]
- Ariffin, D.; Idris, A.; Singh, G. Status of Ganoderma in oil palm. Ganoderma Dis. Perenn. Crops 2000, 1, 49–68. [Google Scholar]
- Sahebi, M.; Hanafi, M.M.; Wong, M.-Y.; Idris, A.; Azizi, P.; Jahromi, M.F.; Shokryazdan, P.; Abiri, R.; Mohidin, H. Towards immunity of oil palm against Ganoderma fungus infection. Acta Physiol. Plant. 2015, 37, 195. [Google Scholar] [CrossRef]
- Naher, L.; Yusuf, U.K.; Ismail, A.; Tan, S.G.; Mondal, M. Ecological status of Ganoderma and basal stem rot disease of oil palms (Elaeis guineensisJacq). Aust. J. Crop Sci. 2013, 7, 1723–1727. [Google Scholar]
- Durand-Gasselin, T.; Asmady, H.; Flori, A.; Jacquemard, J.-C.; Hayun, Z.; Breton, F.; De Franqueville, H. Possible sources of genetic resistance in oil palm (Elaeis guineensis Jacq.) to basal stem rot caused by Ganoderma boninense–prospects for future breeding. Mycopathologia 2005, 159, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Sheil, D.; Casson, A.; Meijaard, E.; Van Noordwjik, M.; Gaskell, J.; Sunderland-Groves, J.; Wertz, K.; Kanninen, M. The Impacts and Opportunities of Oil Palm in Southeast Asia: What Do We Know and What Do We Need to Know? Center for International Forestry Research (CIFOR): Bogor, Indonesia, 2009. [Google Scholar]
- Goy, R.C.; Britto, D.d.; Assis, O.B. A review of the antimicrobial activity of chitosan. Polímeros 2009, 19, 241–247. [Google Scholar] [CrossRef]
- Plascencia-Jatomea, M.; Viniegra, G.; Olayo, R.; Castillo-Ortega, M.M.; Shirai, K. Effect of chitosan and temperature on spore germination of Aspergillus niger. Macromol. Biosci. 2003, 3, 582–586. [Google Scholar] [CrossRef]
- Rabea, E.I.; Badawy, M.E.; Steurbaut, W.; Stevens, C.V. In vitro assessment of N-(benzyl) chitosan derivatives against some plant pathogenic bacteria and fungi. Eur. Polym. J. 2009, 45, 237–245. [Google Scholar] [CrossRef]
- Qiu, M.; Wu, C.; Ren, G.; Liang, X.; Wang, X.; Huang, J. Effect of chitosan and its derivatives as antifungal and preservative agents on postharvest green asparagus. Food Chem. 2014, 155, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Badawy, M.E. Structure and antimicrobial activity relationship of quaternary N-alkyl chitosan derivatives against some plant pathogens. J. Appl. Polym. Sci. 2010, 117, 960–969. [Google Scholar] [CrossRef]
- Fan, W.; Yan, W.; Xu, Z.; Ni, H. Formation mechanism of monodisperse, low molecular weight chitosan nanoparticles by ionic gelation technique. Coll. Surf. B Biointerf. 2012, 90, 21–27. [Google Scholar] [CrossRef]
- Hu, B.; Pan, C.; Sun, Y.; Hou, Z.; Ye, H.; Hu, B.; Zeng, X. Optimization of fabrication parameters to produce chitosan− tripolyphosphate nanoparticles for delivery of tea catechins. J. Agri. Food Chem. 2008, 56, 7451–7458. [Google Scholar] [CrossRef]
- Shi, Y.; Wan, A.; Shi, Y.; Zhang, Y.; Chen, Y. Experimental and mathematical studies on the drug release properties of aspirin loaded chitosan nanoparticles. BioMed. Res. Int. 2014, 2014. [Google Scholar] [CrossRef]
- Agarwal, M.; Nagar, D.; Srivastava, N.; Agarwal, M. Chitosan nanoparticles-based drug delivery: An update. Int. J. Adv. Multidiscip. Res. 2015, 2, 1–13. [Google Scholar]
- Idris, A.; Arifurrahman, R.; dan Kushairi, A. Hexaconazole as a preventive treatment for managing Ganoderma in oil palm. MPOB TS Infor. Ser. 2010, 75, 533–534. [Google Scholar]
- Idris, A.; Arifurrahman, R.; Kushairi, D. An Evaluation of Hexaconazole for Controlling Ganoderma Basal Stem Rot of Oil Palm in the Field as a Preventive Treatment. PIPOC (Agric. Biotechnol. Sustain.) Malaysia 2009. [Google Scholar]
- Russell, P. A Century of fungicide evolution. J. Agric. Sci. 2005, 143, 11–25. [Google Scholar] [CrossRef]
- Khalfallah, S.; Menkissoglu-Spiroudi, U.; Constantinidou, H.A. Dissipation study of the fungicide tetraconazole in greenhouse-grown cucumbers. J. Agric. Food Chem. 1998, 46, 1614–1617. [Google Scholar] [CrossRef]
- Hussein-Al-Ali, S.H.; Kura, A.; Hussein, M.Z.; Fakurazi, S. Preparation of chitosan nanoparticles as a drug delivery system for perindopril erbumine. Polym. Compos. 2018, 39, 544–552. [Google Scholar] [CrossRef]
- Mustafa, I.F.; Hussein, M.Z.; Saifullah, B.; Idris, A.S.; Hilmi, N.H.Z.; Fakurazi, S. Synthesis of (hexaconazole-zinc/aluminum-layered double hydroxide nanocomposite) fungicide nanodelivery system for controlling Ganoderma disease in oil palm. J. Agric. Food Chem. 2018, 66, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, S.; Goycoolea, F.M.; Moerschbacher, B.M.; Rivera-Rodriguez, G.R. Parameters influencing the size of chitosan-TPP nano-and microparticles. Sci. Rep. 2018, 8, 4695. [Google Scholar] [CrossRef]
- Morris, G.A.; Castile, J.; Smith, A.; Adams, G.G.; Harding, S.E. The effect of prolonged storage at different temperatures on the particle size distribution of tripolyphosphate (TPP)–chitosan nanoparticles. Carbohydr. Polym. 2011, 84, 1430–1434. [Google Scholar] [CrossRef]
- Kuen, C.; Fakurazi, S.; Othman, S.; Masarudin, M. Increased loading, efficacy and sustained release of silibinin, a poorly soluble drug using hydrophobically-modified chitosan nanoparticles for enhanced delivery of anticancer drug delivery systems. Nanomaterials 2017, 7, 379. [Google Scholar] [CrossRef]
- Hanafy, A.S.; Farid, R.M.; Elgamal, S.S. Complexation as an approach to entrap cationic drugs into cationic nanoparticles administered intranasally for Alzheimer’s disease management: preparation and detection in rat brain. Drug Dev. Ind. Pharm. 2015, 41, 2055–2068. [Google Scholar] [CrossRef]
- Subbiah, R.; Ramalingam, P.; Ramasundaram, S.; Park, K.; Ramasamy, M.K.; Choi, K.J. N, N, N-Trimethyl chitosan nanoparticles for controlled intranasal delivery of HBV surface antigen. Carbohydr. Polym. 2012, 89, 1289–1297. [Google Scholar] [CrossRef]
- Sharma, N.; Madan, P.; Lin, S. Effect of process and formulation variables on the preparation of parenteral paclitaxel-loaded biodegradable polymeric nanoparticles: A co-surfactant study. Asian J. Pharm. 2016, 11, 404–416. [Google Scholar] [CrossRef]
- Chauhan, N.; Dilbaghi, N.; Gopal, M.; Kumar, R.; Kim, K.-H.; Kumar, S. Development of chitosan nanocapsules for the controlled release of hexaconazole. Int. J. Biol. Macromol. 2017, 97, 616–624. [Google Scholar] [CrossRef]
- Ho, Y.; McKay, G. A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process Saf. Environ. 1998, 76, 332–340. [Google Scholar] [CrossRef]
- Tan, J.M.; Karthivashan, G.; Gani, S.A.; Fakurazi, S.; Hussein, M.Z. Biocompatible polymers coated on carboxylated nanotubes functionalized with betulinic acid for effective drug delivery. J. Mater. Sci. Mater. Med. 2016, 27, 26. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, F.; Scherer, G.G.; Kötz, R.; Wokaun, A. Nanoparticles in energy technology: Examples from electrochemistry and catalysis. Angew. Chem. Int. Ed. 2005, 44, 2190–2209. [Google Scholar] [CrossRef] [PubMed]
- Maluin, F.N.; Hussein, M.Z.; Yusof, N.A.; Fakurazi, S.; Idris, A.S.; Hilmi, N.H.Z.; Jeffery Daim, L.D. A potent antifungal agent for basal stem rot disease treatment in oil palms based on chitosan-dazomet nanoparticles. Int. J. Mol. Sci. 2019, 20, 2247. [Google Scholar] [CrossRef]
- Khaira, R.; Sharma, J.; Saini, V. Development and characterization of nanoparticles for the delivery of gemcitabine hydrochloride. Sci. World J. 2014, 2014. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds CHEN2.5, CHEN5, CHEN10, and CHEN20 are available from the authors. |

| Synthesized Nanoparticles | Reaction Yield * (%) | Loading Content * (%) | Encapsulation Efficiency * (%) |
|---|---|---|---|
| CHEN2.5 | 65.5 ± 3.5 a | 10.7 ± 2.2 a | 55.7 ± 4.3 a |
| CHEN5 | 75.0 ± 4.0 b | 16.7 ± 3.5 b | 66.7 ± 1.5 b |
| CHEN10 | 74.5 ± 2.5 b | 15.4 ± 2.5 b | 65.4 ± 2.0 b |
| CHEN20 | 76.0 ± 2.5 b | 15.2 ± 3.0 b | 65.3 ± 3.5 b |
| Sample | Saturation Release (%) | Pseudo-First-Order | Higuchi Model | Pseudo-Second-Order | ||||
|---|---|---|---|---|---|---|---|---|
| CHEN5 | 99.91 | R2 | K1 (ln mg h−1) | R2 | KH (mg √h−1) | R2 | K2 (mg h−1) | t1/2 (h) |
| 0.9463 | −0.0397 | 0.8033 | 8.5582 | 0.9988 | 0.0100 | 41.97 | ||
| Hixson–Crowell Model | Korsmeyer–Peppas Model | |||||||
| R2 | KHC (h−1) | R2 | K (h−1) | |||||
| 0.6132 | −0.0426 | 0.8938 | 3.2545 | |||||
| Parameter | Type of Fungicides | |||||
|---|---|---|---|---|---|---|
| CEN | Hexaconazole | CHEN2.5 | CHEN5 | CHEN10 | CHEN20 | |
| EC50 (ppb) | 1534.5 | 21.4 | 18.4 | 10.8 | 9.1 | 8.0 |
| Fiducial limit (ppb) (lower-upper) | 494.0–13280.4 | 16.7–27.3 | 13.0–32.8 | 8.1–16.3 | 6.8–12.9 | 6.0–10.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maluin, F.N.; Hussein, M.Z.; Yusof, N.A.; Fakurazi, S.; Idris, A.S.; Zainol Hilmi, N.H.; Jeffery Daim, L.D. Preparation of Chitosan–Hexaconazole Nanoparticles as Fungicide Nanodelivery System for Combating Ganoderma Disease in Oil Palm. Molecules 2019, 24, 2498. https://doi.org/10.3390/molecules24132498
Maluin FN, Hussein MZ, Yusof NA, Fakurazi S, Idris AS, Zainol Hilmi NH, Jeffery Daim LD. Preparation of Chitosan–Hexaconazole Nanoparticles as Fungicide Nanodelivery System for Combating Ganoderma Disease in Oil Palm. Molecules. 2019; 24(13):2498. https://doi.org/10.3390/molecules24132498
Chicago/Turabian StyleMaluin, Farhatun Najat, Mohd Zobir Hussein, Nor Azah Yusof, Sharida Fakurazi, Abu Seman Idris, Nur Hailini Zainol Hilmi, and Leona Daniela Jeffery Daim. 2019. "Preparation of Chitosan–Hexaconazole Nanoparticles as Fungicide Nanodelivery System for Combating Ganoderma Disease in Oil Palm" Molecules 24, no. 13: 2498. https://doi.org/10.3390/molecules24132498
APA StyleMaluin, F. N., Hussein, M. Z., Yusof, N. A., Fakurazi, S., Idris, A. S., Zainol Hilmi, N. H., & Jeffery Daim, L. D. (2019). Preparation of Chitosan–Hexaconazole Nanoparticles as Fungicide Nanodelivery System for Combating Ganoderma Disease in Oil Palm. Molecules, 24(13), 2498. https://doi.org/10.3390/molecules24132498






