Putative Roles of Plant-Derived Tannins in Neurodegenerative and Neuropsychiatry Disorders: An Updated Review
Abstract
1. Introduction
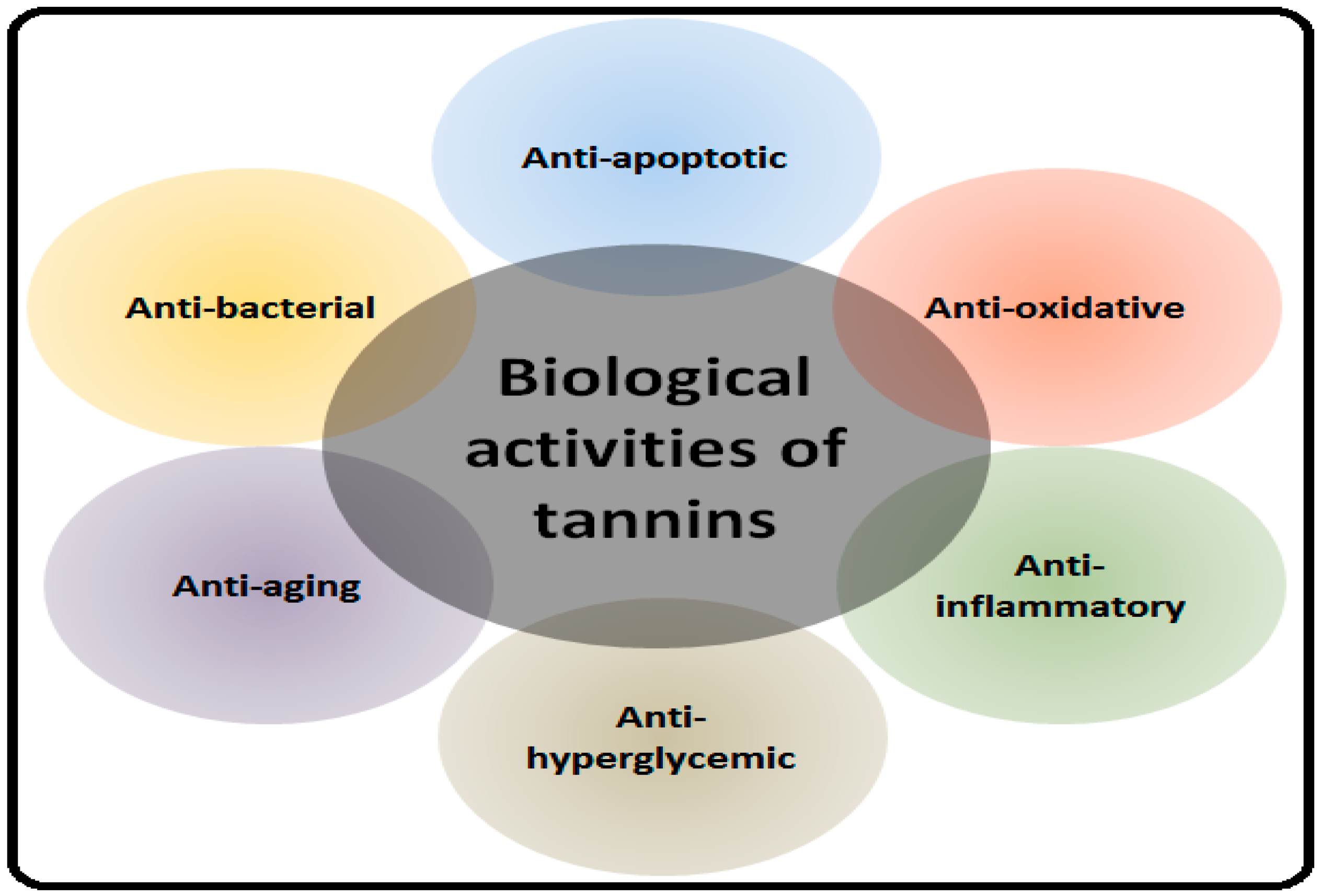
2. Health Benefits of Tannins
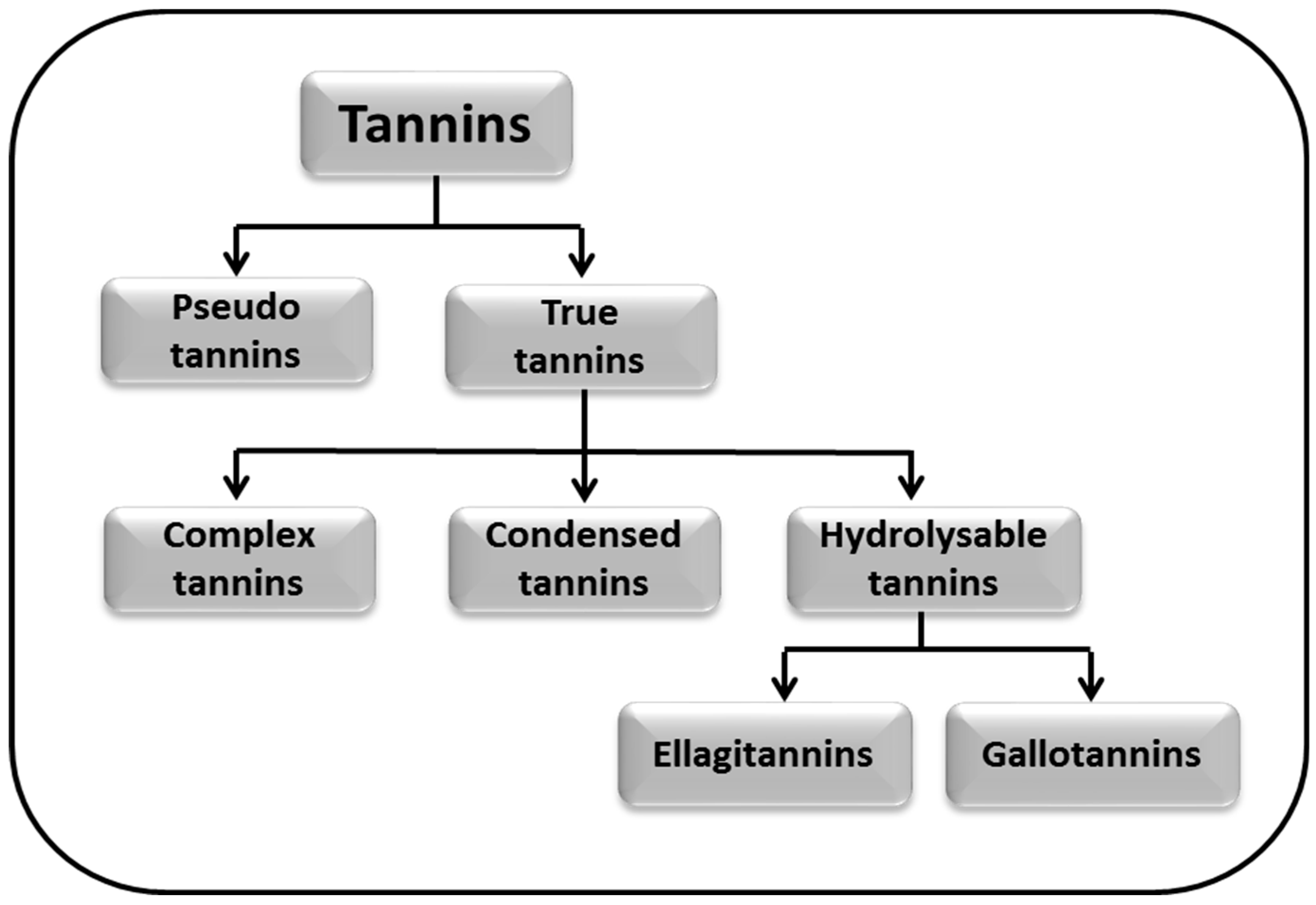
3. Classification of Tannins
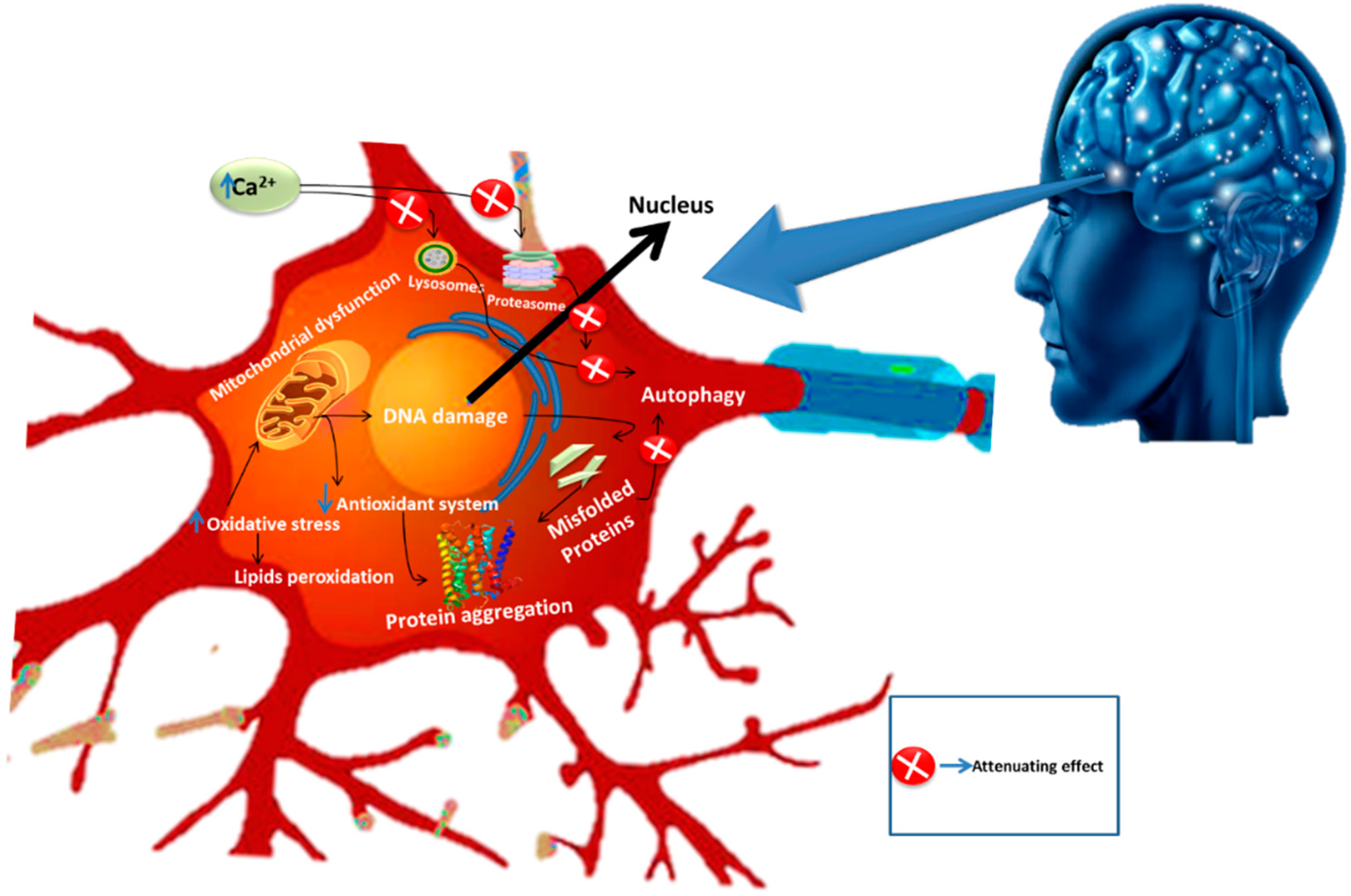
4. Neurodegenerative Diseases
4.1. Alzheimer’s Disease and Its Pathogenesis
4.2. Parkinson’s Disease and Its Pathogenesis
4.3. Depression and Its Pathogenesis
5. Tannins and Their Neuroprotective Functions
5.1. Pseudo Tannins
Pseudo Tannins in Alzheimer’s Disease
5.2. True Tannins
5.2.1. Complex Tannins
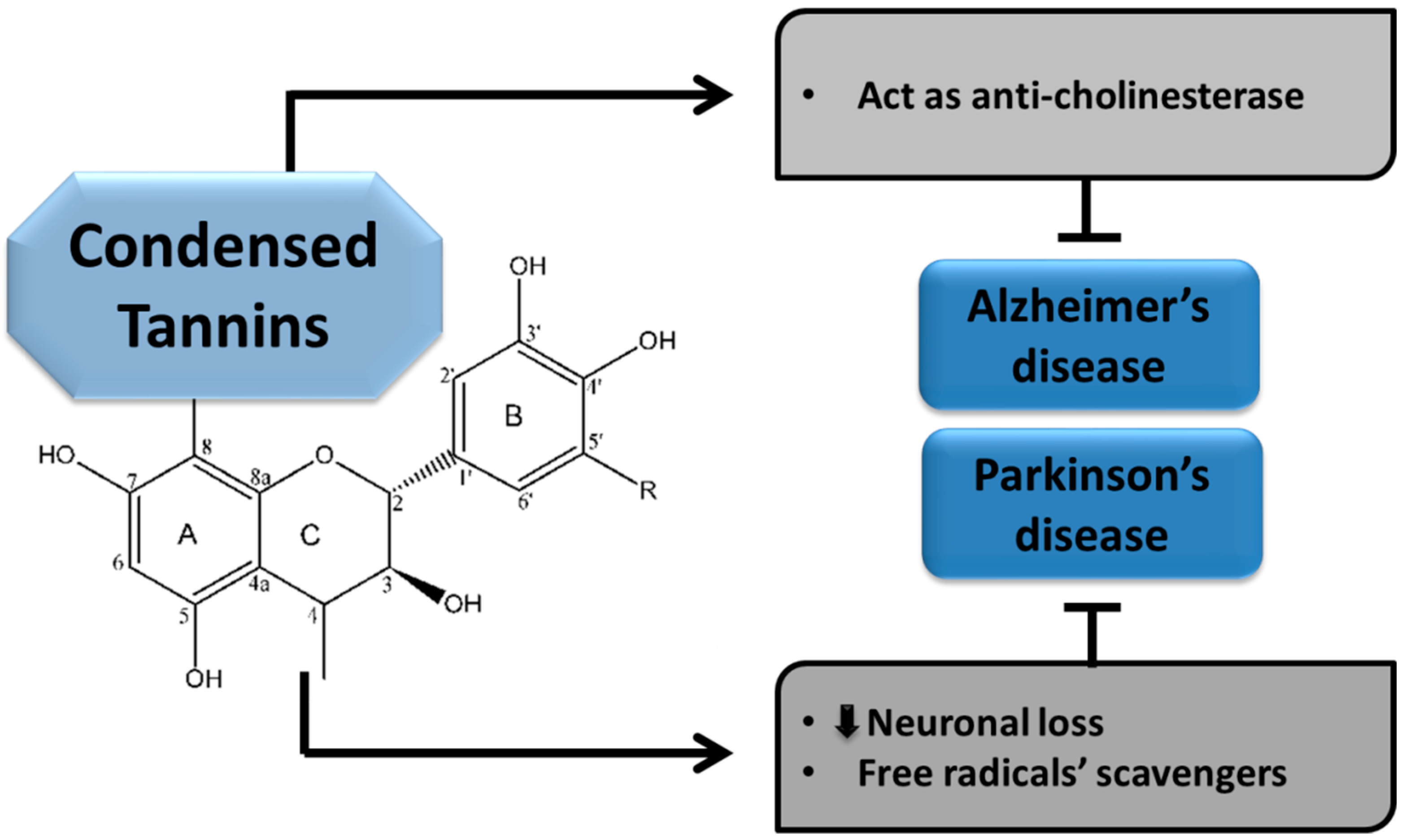
5.2.2. Condensed Tannins
Condensed Tannins in Alzheimer’s disease
Condensed Tannins in Parkinson’s disease
5.2.3. Hydrolysable Tannins
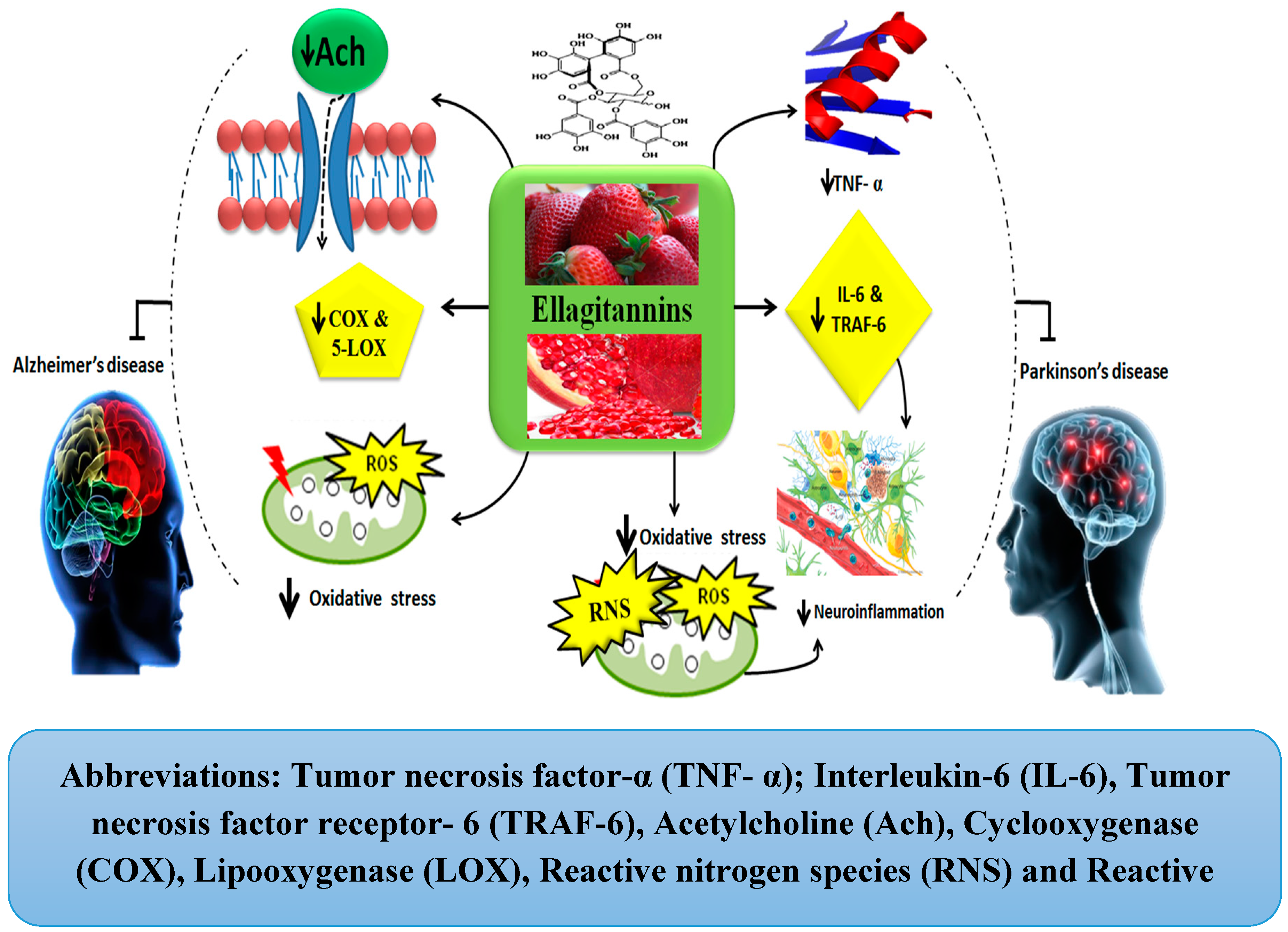
Ellagitannins
Gallotannins
Gallotannins in Alzheimer’s disease
5.3. Tannic Acid in Depression
6. Negative Effects of Tannins on Health
7. Conclusions and Future Perspectives
Funding
Conflicts of Interest
References
- Hussain, G.; Rasul, A.; Anwar, H.; Aziz, N.; Razzaq, A.; Wei, W.; Ali, M.; Li, J.; Li, X. Role of plant derived alkaloids and their mechanism in neurodegenerative disorders. Int. J. Biol. Sci. 2018, 14, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Hussain, G.; Zhang, L.; Rasul, A.; Anwar, H.; Sohail, M.; Razzaq, A.; Aziz, N.; Shabbir, A.; Ali, M.; Sun, T. Role of plant-derived flavonoids and their mechanism in attenuation of Alzheimer’s and Parkinson’s diseases: An update of recent data. Molecules 2018, 23, 814. [Google Scholar] [CrossRef] [PubMed]
- Aziz, N.; Rasul, A.; Malik, S.A.; Anwar, H.; Imran, A.; Razzaq, A.; Shaukat, A.; Kashif, S.; Kamran, S.; De Aguilar, J.G.; et al. Supplementation of Cannabis sativa L. leaf powder accelerates functional recovery and ameliorates haemoglobin level following an induced injury to sciatic nerve in mouse model. Pak. J. Pharm. Sci. 2019, 32, 785–792. [Google Scholar] [PubMed]
- Kennedy, D.O.; Wightman, E.L. Herbal extracts and phytochemicals: Plant secondary metabolites and the enhancement of human brain function. Adv. Nutr. 2011, 2, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Uriarte Pueyo, I.; Calvo, M.I. Phytochemical study and evaluation of antioxidant, neuroprotective and acetylcholinesterase inhibitor activities of Galeopsis ladanum L. extracts. Pharmacogn. Mag. 2009, 5, 287–290. [Google Scholar] [CrossRef]
- Ky, I.; Le Floch, A.; Zeng, L.; Pechamat, L.; Jourdes, M.; Teissedre, P.L. Tannins. In Encyclopedia of Food and Health; Elsevier: New York, NY, USA, 2015; pp. 247–255. ISBN 9780123849533. [Google Scholar]
- Kumari, M. Screening of Potential Sources of Tannin and Its Therapeutic Application. Int. J. Nutr. Food Sci. 2017, 4, 26. [Google Scholar] [CrossRef]
- Makkar, H.; Siddhuraju, P.; Becker, K. Plant Secondary Metabolites: Tannins. Methods Mol. Biol. 2007, 393, 67–81. [Google Scholar] [CrossRef]
- Delimont, N.M.; Haub, M.D.; Lindshield, B.L. The Impact of Tannin Consumption on Iron Bioavailability and Status: A Narrative Review. Curr. Dev. Nutr. 2017, 1, e000042. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.L.R.M.G.; Filho, J.M.B. Anti-inflammatory actions of tannins isolated from the bark of Anacardwm occidentale L. J. Ethnopharmacol. 1985, 13, 289–300. [Google Scholar] [CrossRef]
- Wafa, N.; Sofiane, G.; Mouhamed, K. The antioxidant and antimicrobial activities of flavonoids and tannins extracted from Phlomis bovei De Noé. Pelagia Res. Libr. Eur. J. Exp. Biol. 2016, 6, 55–61. [Google Scholar] [CrossRef]
- Yazdi, A.; Sardari, S.; Sayyah, M.; Ezzati, M.H. Evaluation of the anticonvulsant activity of the leaves of glycyrrhiza glabra var. glandulifera grown in Iran, as a possible renewable source for anticonvulsant compounds. Iran. J. Pharm. Res. 2011, 10, 75–82. [Google Scholar] [PubMed]
- Al-Ayyoubi, S.; Gali-Muhtasib, H. Anti-tumor signaling pathways modulated by plant polyphenols. New Cell Apoptosis Res. 2007, 85–105. [Google Scholar]
- Basli, A.; Soulet, S.; Chaher, N.; Mérillon, J.-M.; Chibane, M.; Monti, J.-P.; Richard, T. Wine Polyphenols: Potential Agents in Neuroprotection. Oxidative Med. Cell. Longev. 2012, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bora, K.S.; Arora, S.; Shri, R. Role of Ocimum basilicum L. in prevention of ischemia and reperfusion-induced cerebral damage, and motor dysfunctions in mice brain. J. Ethnopharmacol. 2011, 137, 1360–1365. [Google Scholar] [CrossRef] [PubMed]
- Kashiwada, Y.; Huang, L.; Kilkuskie, R.E.; Bodner, A.J.; Lee, K.-H. New hexahydroxydiphenoyl derivatives as potent inhibitors of HIV replication in H9 lymphocytes. Bioorganic Med. Chem. Lett. 1992, 2, 235–238. [Google Scholar] [CrossRef]
- Winterbourn, C.C. Reconciling the chemistry and biology of reactive oxygen species. Nat. Chem. Biol. 2008, 4, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Shen, T.; Lou, H. Dietary polyphenols and their biological significance. Int. J. Mol. Sci. 2007, 8, 950–988. [Google Scholar] [CrossRef]
- Weiss, E.I.; Houri-Haddad, Y.; Greenbaum, E.; Hochman, N.; Ofek, I.; Zakay-Rones, Z. Cranberry juice constituents affect influenza virus adhesion and infectivity. Antivir. Res. 2005, 66, 9–12. [Google Scholar] [CrossRef]
- Chung, K.-T.; Wong, T.Y.; Wei, C.-I.; Huang, Y.-W.; Lin, Y. Tannins and Human Health: A Review. Crit. Rev. Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef]
- Ovaskainen, M.-L.; Törrönen, R.; Koponen, J.M.; Sinkko, H.; Hellström, J.; Reinivuo, H.; Mattila, P. Dietary intake and major food sources of polyphenols in Finnish adults. J. Nutr. 2008, 138, 562–566. [Google Scholar] [CrossRef]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Møller, C.; Hansen, S.H.; Cornett, C. Characterisation of tannin-containing herbal drugs by HPLC. Phytochem. Anal. 2009, 20, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Rasul, A.; Di, J.; Millimouno, F.; Malhi, M.; Tsuji, I.; Ali, M.; Li, J.; Li, X. Reactive Oxygen Species Mediate Isoalantolactone-Induced Apoptosis in Human Prostate Cancer Cells. Molecules 2013, 18, 9382–9396. [Google Scholar] [CrossRef] [PubMed]
- Sarfraz, I.; Rasul, A.; Jabeen, F.; Younis, T.; Zahoor, M.K.; Arshad, M.; Ali, M. Fraxinus: A Plant with Versatile Pharmacological and Biological Activities. Evid.-Based Complementary Altern. Med. 2017, 2, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Khanbabaee, K.; van Ree, T. Tannins: Classification and definition. Nat. Prod. Rep. 2001, 18, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, H.; Singh, O.J.; Chauhan, A.; Sahu, M.K.; Dv, S.P. A review on tannins. Biosci. J. 2018, 6, 16–17. [Google Scholar]
- Maji Amal, K.; Pratim, B. Strychnos nux-vomica: A poisonous plant with various aspects of therapeutic significance. J. Basic Clin. Pharm. 2017, 8, S087–S103. [Google Scholar]
- Duval, A.; Averous, L. Characterization and physico-chemical properties of condensed tannins from Acacia catechu Characterization and physico-chemical properties of condensed tannins from Acacia catechu. J. Agric. Food Chem. 2016, 64, 1751–1760. [Google Scholar] [CrossRef]
- Zengin, G.; Locatelli, M.; Carradori, S.; Mocan, A.M.; Aktumsek, A. Total phenolics, flavonoids, condensed tannins content of eight centaurea species and their broad inhibitory activities against cholinesterase, tyrosinase, α-amylase and α-glucosidase. Not. Bot. Horti Agrobot. Cluj-Napoca 2016, 44, 195–200. [Google Scholar] [CrossRef]
- Calou, I.; Bandeira, M.A.; Aguiar-Galvão, W.; Cerqueira, G.; Siqueira, R.; Neves, K.R.; Brito, G.A.; Viana, G. Neuroprotective Properties of a Standardized Extract from Myracrodruon urundeuva Fr. All. (Aroeira-Do-Sertão), as Evaluated by a Parkinson’s Disease Model in Rats. Parkinson’s Dis. 2014, 2014, 519615. [Google Scholar] [CrossRef]
- Nile, S.H.; Park, S.W. Edible berries: Bioactive components and their effect on human health. Nutrition 2014, 30, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Afshari, A.R.; Sadeghnia, H.R.; Mollazadeh, H. A Review on Potential Mechanisms of Terminalia chebula in Alzheimer’s Disease. Adv. Pharmacol. Sci. 2016, 2016, 8964849. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Brijeshlata, D.S.; Dixit, S. Screening of traditional indian spices for inhibitory activity of acetylcholinesterase and butyrylcholinesterase enzymes. Int. J. Pharma Bio Sci. 2012, 3, P59–P65. [Google Scholar]
- Dhivya, P.S.; Sobiya, M.; Selvamani, P.; Latha, S. An approach to alzheimer’s disease treatment with cholinesterase inhibitory activity from various plant species. Int. J. Pharmtech Res. 2014, 6, 1450–1468. [Google Scholar]
- Olajide, O.A.; Kumar, A.; Velagapudi, R.; Okorji, U.P.; Fiebich, B.L. Punicalagin inhibits neuroinflammation in LPS-activated rat primary microglia. Mol. Nutr. Food Res. 2014, 58, 1843–1851. [Google Scholar] [CrossRef] [PubMed]
- Tapias, V. Pomegranate juice exacerbates oxidative stress and nigrostriatal degeneration in Parkinson’s disease. Neurobiol. Aging 2014, 35, 1162–1176. [Google Scholar] [CrossRef] [PubMed]
- Kujawski, R. Perspectives for gallotannins neuroprotective potential-current experimental evidences Comparison of extracts from root of rhodiola rosea inhibitory action on EtOH tolerance development in rats View project Małgorzata Kujawska. J. Med Sci. 2016, 85, 313–318. [Google Scholar] [CrossRef]
- Thenmozhi, A.J.; Manivasagam, T.; Essa, M.M. Role of plant polyphenols in Alzheimer’s disease. Adv. Neurobiol. 2016, 12, 153–171. [Google Scholar] [PubMed]
- Braidy, N.; Jugder, B.-E.; Poljak, A.; Jayasena, T.; Nabavi, S.M.; Sachdev, P.; Grant, R. Molecular targets of tannic acid in Alzheimer’s disease. Curr. Alzheimer Res. 2017, 14, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.W.; Chen, Y.C.; Hsieh, W.L.; Chiou, S.H.; Kao, C.L. Ageing and neurodegenerative diseases. Ageing Res. Rev. 2010, 9, S36–S46. [Google Scholar] [CrossRef]
- Hussain, G.; Rasul, A.; Anwar, H.; Sohail, M.U.; Kamran, S.K.S.; Baig, S.M.; Shabbir, A.; Iqbal, J. Epidemiological Data of Neurological Disorders in Pakistan and Neighboring Countries: A Review. Pak. J. Neurol. Sci. 2017, 12, 52–70. [Google Scholar]
- Kovacs, G.G. Current Concepts of Neurodegenerative Diseases. EMJ Neurol 2014, 1, 78–86. [Google Scholar]
- Association, A. Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2009, 5, 234–270. [Google Scholar] [CrossRef]
- Cai, Z.; Wang, C.; Yang, W. Role of berberine in Alzheimer’s disease. Neuropsychiatr. Dis. Treat. 2016, 12, 2509–2520. [Google Scholar] [CrossRef] [PubMed]
- Prasanthi, J.R.P.; Dasari, B.; Marwarha, G.; Larson, T.; Chen, X.; Geiger, J.D.; Ghribi, O. Caffeine protects against oxidative stress and Alzheimer’s disease-like pathology in rabbit hippocampus induced by cholesterol-enriched diet. Free Radic. Biol. Med. 2010, 49, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.; Wu, F.; Ma, Y.; Liu, G.; Li, Z.; Sun, Y.; Pei, Z. Decrease in the production of beta-amyloid by berberine inhibition of the expression of beta-secretase in HEK293 cells. BMC Neurosci. 2011, 12. [Google Scholar] [CrossRef] [PubMed]
- Castellani, R.J.; Zhu, X.; Lee, H.; Moreira, P.I.; Perry, G.; Smith, M.A. Neuropathology and treatment of Alzheimer disease: Did we lose the forest for the trees? Expert Rev. Neurother. 2007, 7, 473–485. [Google Scholar] [CrossRef] [PubMed]
- Heron, M. Deaths: Leading causes for 2008. Natl. Vital Stat. Rep. 2012, 60, 1–94. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, A. Pharmacological Reports Review article A review on Alzheimer’s disease pathophysiology and its management: An update. Pharm. Rep 2015, 67, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Duffy, F.H.; Albert, M.S.; McAnulty, G. Brain electrical activity in patients with presenile and senile dementia of the Alzheimer type. Ann. Neurol. 1984, 16, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Rupasinghe, H.P.V. Polyphenols: Multipotent Therapeutic Agents in Neurodegenerative Diseases. Oxidative Med. Cell. Longev. 2013, 2013, 891748. [Google Scholar] [CrossRef] [PubMed]
- Van Bulck, M.; Sierra-Magro, A.; Alarcon-Gil, J.; Perez-Castillo, A.; Morales-Garcia, J.A. Novel approaches for the treatment of Alzheimer’s and Parkinson’s disease. Int. J. Mol. Sci. 2019, 20, 719. [Google Scholar] [CrossRef] [PubMed]
- Sarrafchi, A.; Bahmani, M.; Shirzad, H.; Rafieian-Kopaei, M. Oxidative Stress and Parkinson’s Disease: New Hopes in Treatment with Herbal Antioxidants. Curr. Pharm. Des. 2016, 22, 238–246. [Google Scholar] [CrossRef] [PubMed]
- Oguru, M.; Tachibana, H.; Toda, K.; Okuda, B.; Oka, N. Apathy and depression in parkinson disease. J. Geriatr. Psychiatry Neurol. 2010, 23, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Neurol, J.J. Parkinson’s disease: Clinical features and diagnosis. Psychiatry Interpers. Biol. Process. 2008, 72, 368–376. [Google Scholar] [CrossRef]
- Gaki, G.S.; Papavassiliou, A.G. Oxidative stress-induced signaling pathways implicated in the pathogenesis of Parkinson’s disease. Neuromol. Med. 2014, 16, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Magrinelli, F.; Picelli, A.; Tocco, P.; Federico, A.; Roncari, L.; Smania, N.; Zanette, G.; Tamburin, S. Pathophysiology of Motor Dysfunction in Parkinson’s Disease as the Rationale for Drug Treatment and Rehabilitation. Parkinson’s Dis. 2016, 2016, 9832839. [Google Scholar] [CrossRef] [PubMed]
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Imran, A.; Qasim, M.; Zafar, S.; Kamran, S.K.S.; Razzaq, A.; Aziz, N.; et al. Role of cholesterol and sphingolipids in brain development and neurological diseases. Lipids Health Dis. 2019, 18, 26. [Google Scholar] [CrossRef] [PubMed]
- Wideman, T.H.; Zautra, A.J.; Edwards, R.R. Rethinking the fear avoidance model: Toward a multidimensional framework of pain-related disability. NIH Public Access. 2014, 154, 2262–2265. [Google Scholar] [CrossRef] [PubMed]
- Hurley, L.L.; Tizabi, Y. Neuroinflammation, neurodegeneration, and depression. Neurotox. Res. 2013, 23, 131–144. [Google Scholar] [CrossRef]
- Bajpai, A.; Verma, A.K.; Srivastava, M.; Srivastava, R. Oxidative stress and major depression. J. Clin. Diagn. Res. 2014, 8, CC04–CC07. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.A.; Drinnan, C.T.; Pleshko, N.; Fisher, O.Z. Pseudotannins self-assembled into antioxidant complexes. Soft Matter 2015, 11, 7783–7791. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Gu, L.; Prior, R.L.; McKay, S.; Nakajima, J.; Tanaka, I.; Seo, S.; Yamazaki, M.; Saito, K. Characterization of anthocyanins and proanthocyanidins in some cultivars of Ribes, Aronia, and Sambucus and their antioxidant capacity. J. Agric. Food Chem. 2004, 5, 7846–7856. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, D.; Bagchi, M.; Stohs, S.J.; Das, D.K.; Ray, S.D.; Kuszynski, C.A.; Joshi, S.S.; Pruess, H.G. Free radicals and grape seed proanthocyanidin extract: Importance in human health and disease prevention. Toxicology 2000, 148, 187–197. [Google Scholar] [CrossRef]
- Liu, X.; Li, X.N.; Bao, L.X.; Ling, B.Y. Effect of grape seed extract proanthocyanidin on loaded swimming time in mice. Chin. J. Clin. Rehabil. 2005, 9, 245–247. [Google Scholar]
- Amarowicz, R.; Janiak, M. Hydrolysable Tannins. In Encyclopedia of Food Chemistry; Elsevier: New York, NY, USA, 2018; pp. 337–343. ISBN 9780081005965. [Google Scholar]
- Zhao, J.; Khan, I.A.; Fronczek, F.R. Gallic acid. Acta Crystallogr. Sect. E Struct. Rep. Online 2011, 67, 1. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M.; Di Lorenzo, A.; Nabavi, S.F.; Talas, Z.S.; Nabavi, S.M. Polyphenols: Well beyond the Antioxidant Capacity: Gallic Acid and Related Compounds as Neuroprotective Agents: You are What You Eat! Curr. Pharm. Biotechnol. 2014, 15, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.A.; Collins, P.B. The anti-oxidative and anti-inflammatory roles of gallic acid on transcriptional regulation. In Handbook on Gallic Acid; Nova Science Publishers: New York, NY, USA, 2013; pp. 135–155. ISBN 9781626189218. [Google Scholar]
- Dumitriu, A.; Moser, C.; Hadzi, T.C.; Williamson, S.L.; Pacheco, C.D.; Hendricks, A.E.; Latourelle, J.C.; Wilk, J.B.; Destefano, A.L.; Myers, R.H. Postmortem Interval Influences α-Synuclein Expression in Parkinson Disease Brain. Parkinsons Dis. 2012, 2012, 614212. [Google Scholar] [CrossRef]
- Sameri, M.J.; Sarkaki, A.; Farbood, Y.; Mansouri, S.M.T. Motor disorders and impaired electrical power of pallidal EEG improved by gallic acid in animal model of Parkinson’s disease. Pak. J. Biol. Sci. 2011, 14, 1109–1116. [Google Scholar] [CrossRef]
- Stoner, G.D.; Seeram, N.P. Berries and Cancer Prevention; Springer: New York, NY, USA, 2011; ISBN 9781441975539. [Google Scholar]
- Jourdes, M. Hydrolyzable tannins: Gallotannins and ellagitannins. In Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes; Springer: Heidelberg, Germany, 2013; pp. 1975–2010. ISBN 9783642221446. [Google Scholar]
- Reddy, D.B. Reddy Chebulagic acid, a COX-LOX dual inhibitor isolated from the fruits of Terminalia chebula Retz., induces apoptosis in COLO-205 cell line. J. Ethnopharmacol. 2009, 124, 506–512. [Google Scholar] [CrossRef]
- Park, J.H.; Joo, H.S.; Yoo, K.Y.; Shin, B.N.; Kim, I.H.; Lee, C.H.; Choi, J.H.; Byun, K.; Lee, B.; Lim, S.S.; et al. Extract from Terminalia chebula seeds protect against experimental ischemic neuronal damage via maintaining SODs and BDNF levels. Neurochem. Res. 2011, 36, 2043–2050. [Google Scholar] [CrossRef] [PubMed]
- Subash, S.; Essa, M.M.; Al-Adawi, S.; Memon, M.A.; Manivasagam, T.; Akbar, M. Neuroprotective effects of berry fruits on neurodegenerative diseases. Neural Regen. Res. 2014, 9, 1557–1566. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Momin, R.A.; Nair, M.G.; Bourquin, L.D. Cyclooxygenase inhibitory and antioxidant cyanidin glycosides in cherries and berries. Phytomedicine 2001, 8, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Ferencik, M.; Novak, M.; Rovensky, J.; Rybar, I. Alzheimer’s disease, inflammation and non-steroidal anti-inflammatory drugs. Bratisl. Lek. Listy 2001, 102, 123–132. [Google Scholar] [PubMed]
- Sylla, T.; Pouységu, L.; Dacosta, G.; Deffieux, D.; Monti, J.P.; Quideau, S. Gallotannins and Tannic Acid: First Chemical Syntheses and in Vitro Inhibitory Activity on Alzheimer’s Amyloid β-Peptide Aggregation. Angew. Chem. Int. Ed. 2015, 54, 8217–8221. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, A.J.; Begg, D.; Mathai, M.; Weisinger, R.S. Omega 3 fatty acids and the brain: Review of studies in depression. Asia Pac. J. Clin. Nutr. 2007, 16, 391–397. [Google Scholar] [PubMed]
- Gold, P.W.; Machado-Vieira, R.; Pavlatou, M.G. Clinical and Biochemical Manifestations of Depression: Relation to the Neurobiology of Stress. Neural Plast. 2015, 2015, 581976. [Google Scholar] [CrossRef] [PubMed]
- Chandra Shekar, D.; Manohar, V.R.; Rao, S.N. Antidepressant activity of aqueous extract of fruits of terminalia chebula in rats. Int. J. Pharm. Pharm. Sci. 2012, 4, 449–451. [Google Scholar]
- Chandrasekhar, Y.; Ramya, E.M.; Navya, K.; Phani Kumar, G.; Anilakumar, K.R. Antidepressant like effects of hydrolysable tannins of Terminalia catappa leaf extract via modulation of hippocampal plasticity and regulation of monoamine neurotransmitters subjected to chronic mild stress (CMS). Biomed. Pharmacother. 2017, 86, 414–425. [Google Scholar] [CrossRef]
- Pariante, C.M.; Benkert, O.; Szegedi, A.; Müller, M.J. Antidepressant Drugs. In International Encyclopedia of the Social & Behavioral Sciences, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 778–783. ISBN 9780080970875. [Google Scholar]
- Cai, L.; Li, R.; Tang, W.J.; Meng, G.; Hu, X.Y.; Wu, T.N. Antidepressant-like effect of geniposide on chronic unpredictable mild stress-induced depressive rats by regulating the hypothalamus-pituitary-adrenal axis. Eur. Neuropsychopharmacol. 2015, 25, 1332–1341. [Google Scholar] [CrossRef]
- Lin, P.; Campbell, D.G.; Chaney, E.F.; Liu, C.F.; Heagerty, P.; Felker, B.L.; Hedrick, S.C. The influence of patient preference on depression treatment in primary care. Ann. Behav. Med. 2005, 30, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Robles, H. Tannic Acid. In Encyclopedia of Toxicology, 3rd ed.; Academic Press: Cambridge, MA, USA, 2014; pp. 474–475. ISBN 9780123864543. [Google Scholar]
- Serrano, J.; Puupponen-Pimiä, R.; Dauer, A.; Aura, A.M.; Saura-Calixto, F. Tannins: Current knowledge of food sources, intake, bioavailability and biological effects. Mol. Nutr. Food Res. 2009, 53, 310–329. [Google Scholar] [CrossRef] [PubMed]
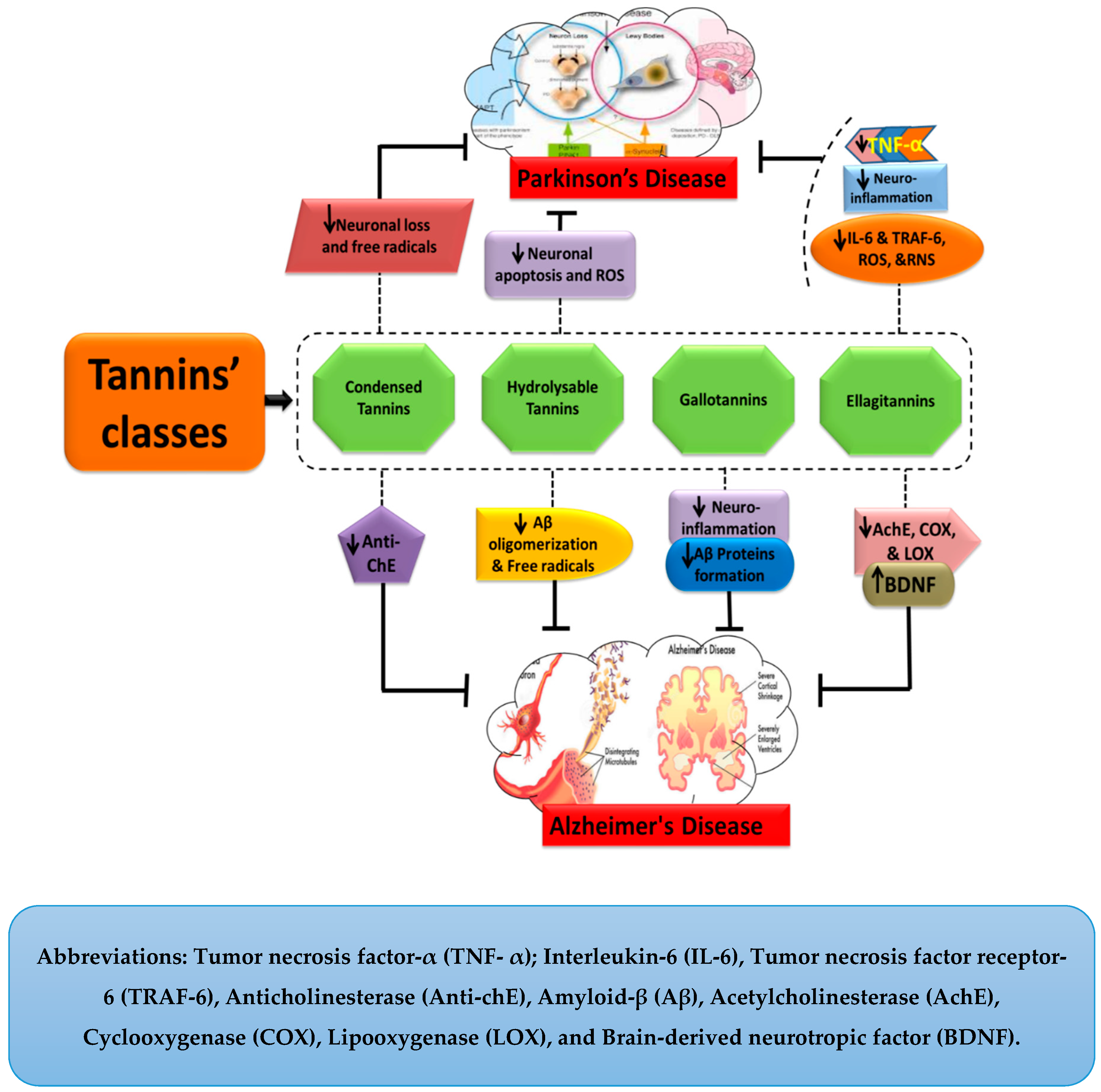
| Tannins | Classes | Sub-Classes | Mechanism of Action in NDDs | ||
| Alzheimer’s Disease | Parkinson’s Disease | ||||
| Pseudo tannins | — | Stimulate neurotransmission and upregulate Ach binding with receptors [28]. | — | ||
| True tannins | Complex tannins | — | — | ||
| Condensed tannins | Exhibit anti-chE activity [29,30]. | Anti-oxidants (free radicals scavengers) and attenuate neuronal loss [31,32]. | |||
| Hydrolysable tannins | Ellagitannins | Inhibit AChE, COX, and LOX activity, free radicals’ scavengers, maintain BDNF and SOD levels, attenuate neuronal damage [33,34,35]. | Attenuate neuro-inflammation, inhibit IL-6, TNF-α, TRAF-6, ROS, and RNS [36]. Upregulate neuroplasticity [37]. | ||
| Gallotannins | Attenuate oxidative stress, Aβ formation and deposition, and neuro-inflammation [38,39,40]. | — | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, G.; Huang, J.; Rasul, A.; Anwar, H.; Imran, A.; Maqbool, J.; Razzaq, A.; Aziz, N.; Makhdoom, E.u.H.; Konuk, M.; et al. Putative Roles of Plant-Derived Tannins in Neurodegenerative and Neuropsychiatry Disorders: An Updated Review. Molecules 2019, 24, 2213. https://doi.org/10.3390/molecules24122213
Hussain G, Huang J, Rasul A, Anwar H, Imran A, Maqbool J, Razzaq A, Aziz N, Makhdoom EuH, Konuk M, et al. Putative Roles of Plant-Derived Tannins in Neurodegenerative and Neuropsychiatry Disorders: An Updated Review. Molecules. 2019; 24(12):2213. https://doi.org/10.3390/molecules24122213
Chicago/Turabian StyleHussain, Ghulam, Jia Huang, Azhar Rasul, Haseeb Anwar, Ali Imran, Javeria Maqbool, Aroona Razzaq, Nimra Aziz, Ehtisham ul Haq Makhdoom, Muhsin Konuk, and et al. 2019. "Putative Roles of Plant-Derived Tannins in Neurodegenerative and Neuropsychiatry Disorders: An Updated Review" Molecules 24, no. 12: 2213. https://doi.org/10.3390/molecules24122213
APA StyleHussain, G., Huang, J., Rasul, A., Anwar, H., Imran, A., Maqbool, J., Razzaq, A., Aziz, N., Makhdoom, E. u. H., Konuk, M., & Sun, T. (2019). Putative Roles of Plant-Derived Tannins in Neurodegenerative and Neuropsychiatry Disorders: An Updated Review. Molecules, 24(12), 2213. https://doi.org/10.3390/molecules24122213







