Applications of Photonics in Agriculture Sector: A Review
Abstract
1. Introduction
2. Classification of Photonics Systems in Agriculture
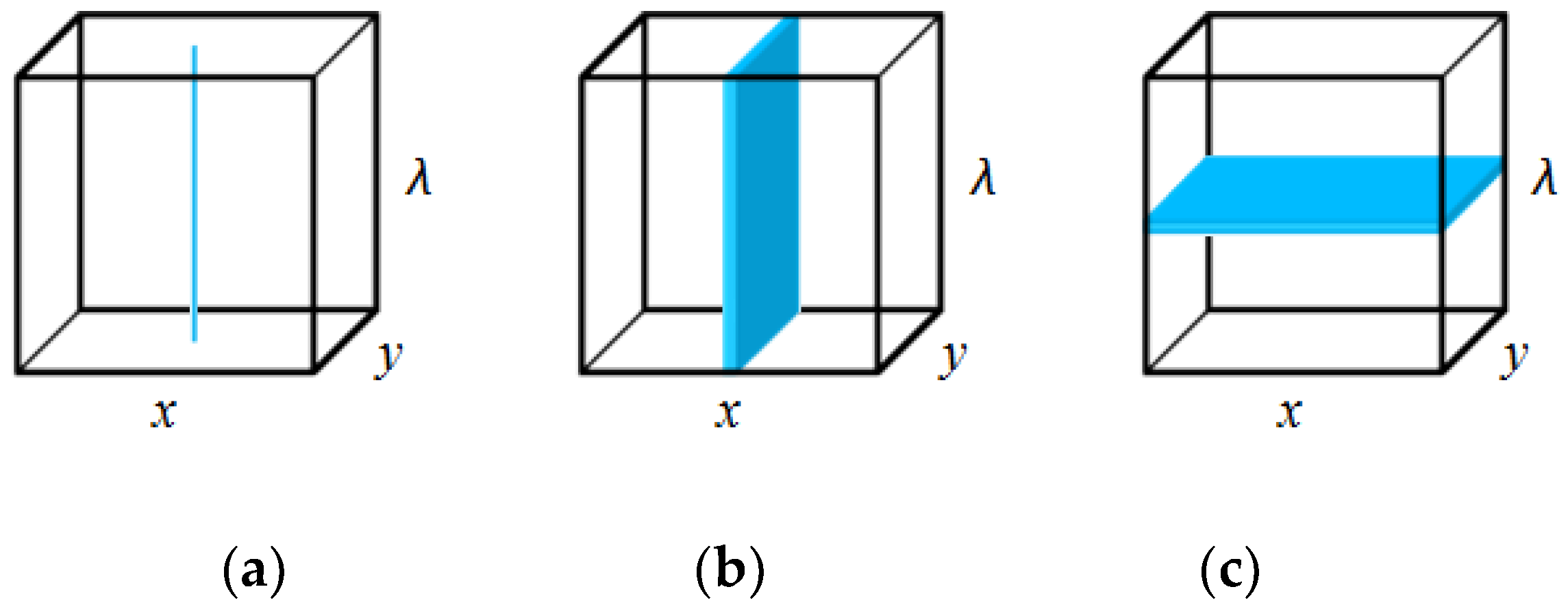
2.1. Imaging Technique
2.2. Spectroscopy Technique
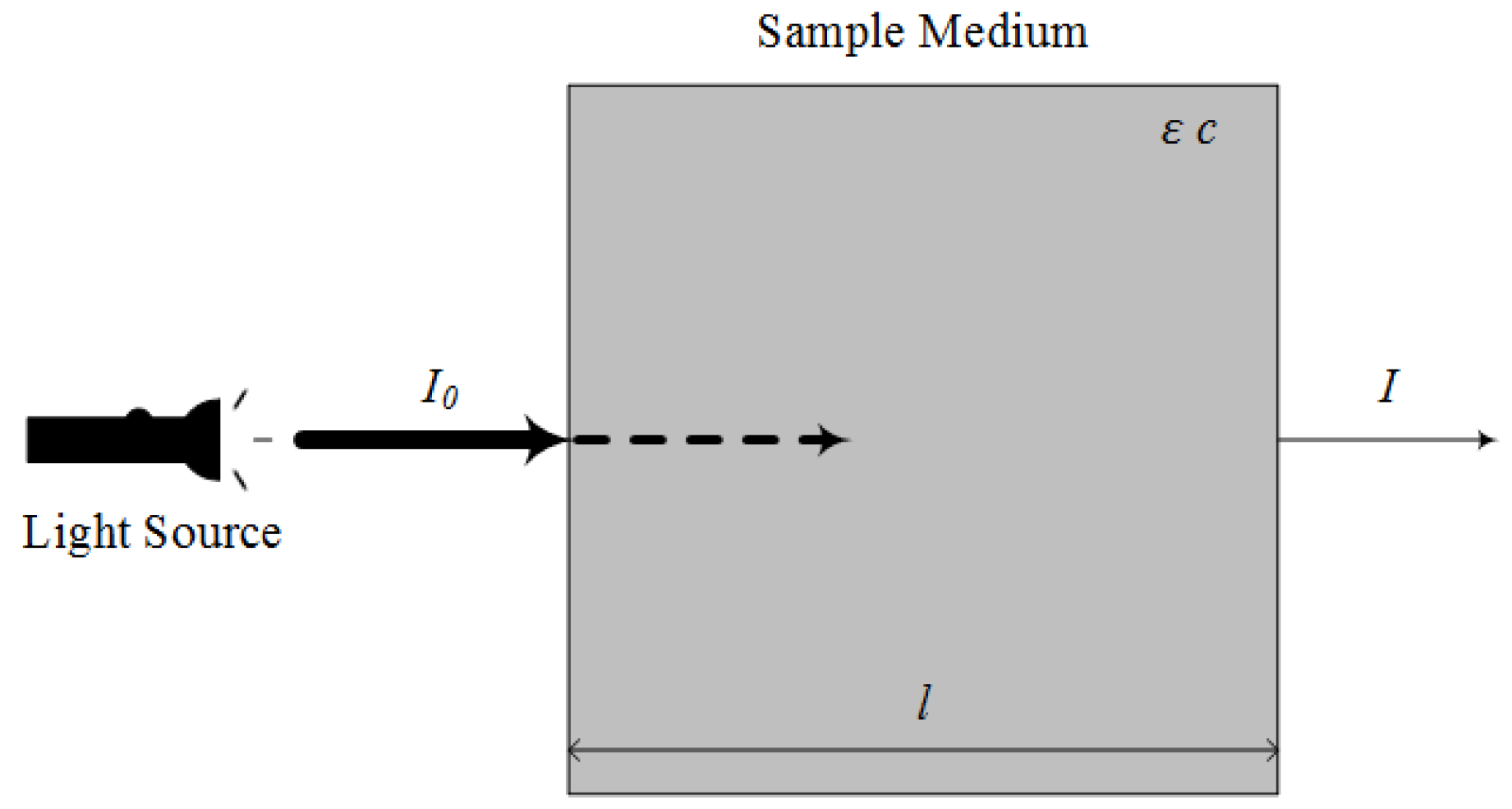
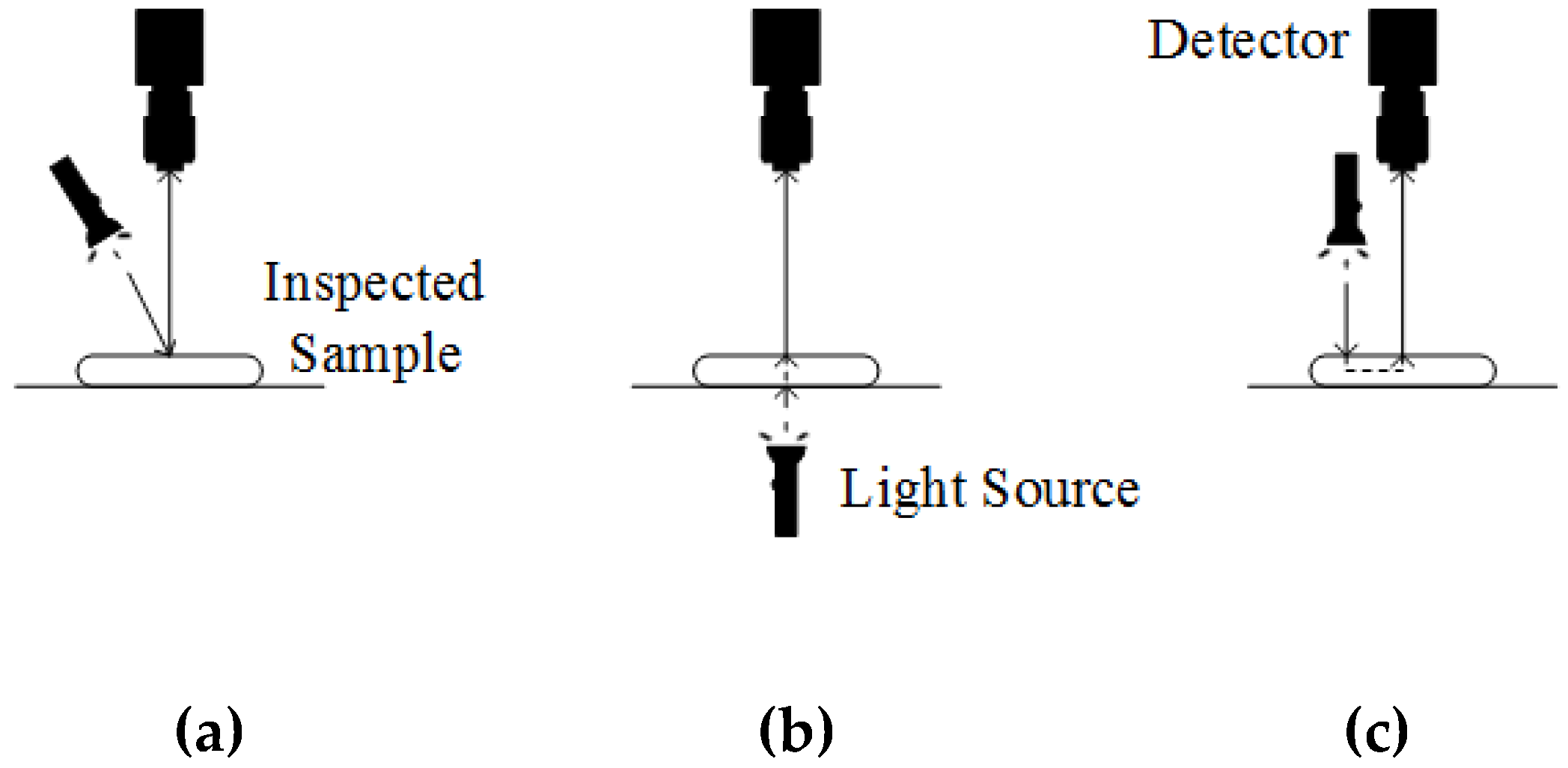
2.2.1. Ultraviolet-Visible (UV-VIS) Spectroscopy
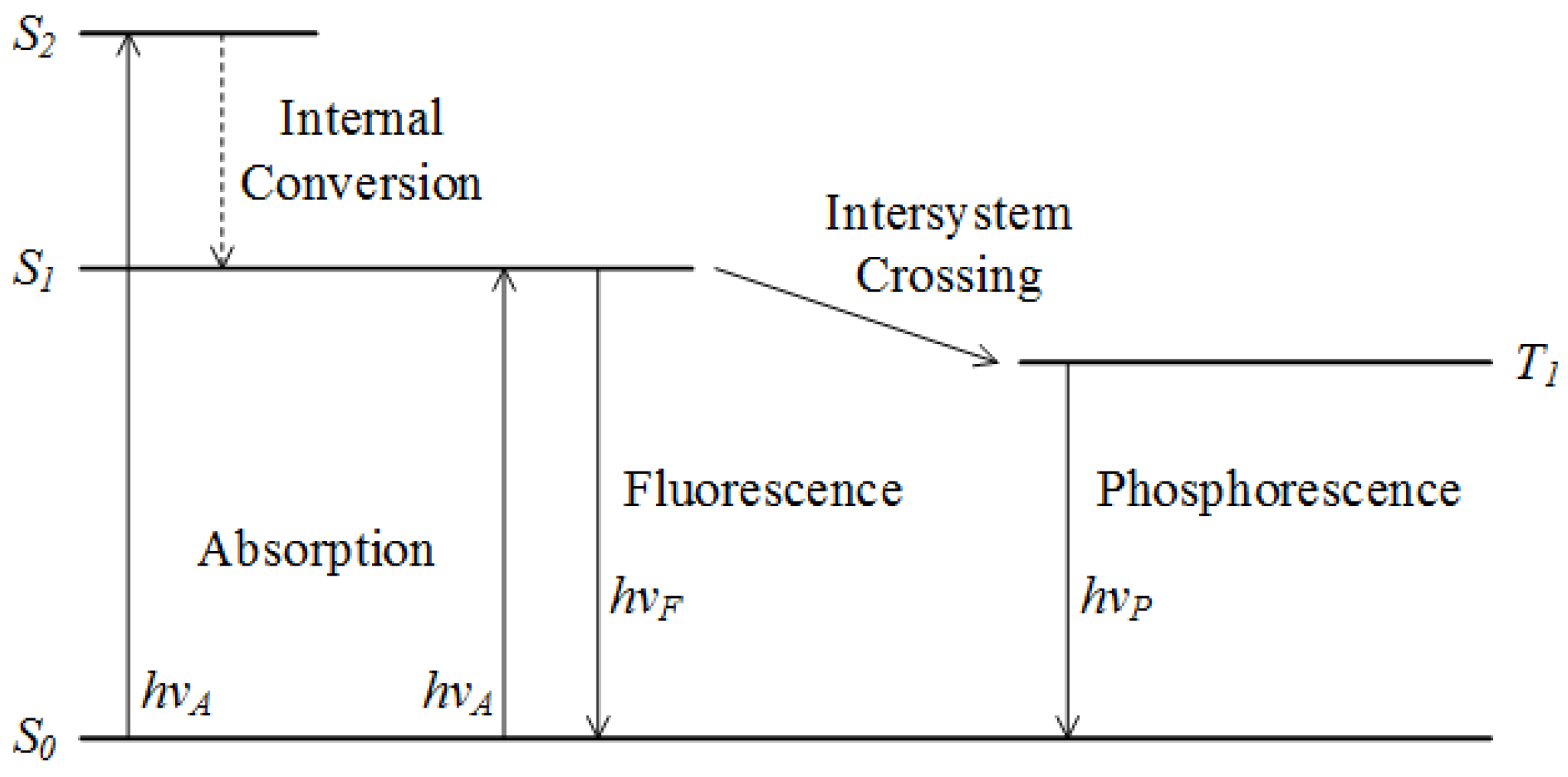
2.2.2. Fluorescence Spectroscopy
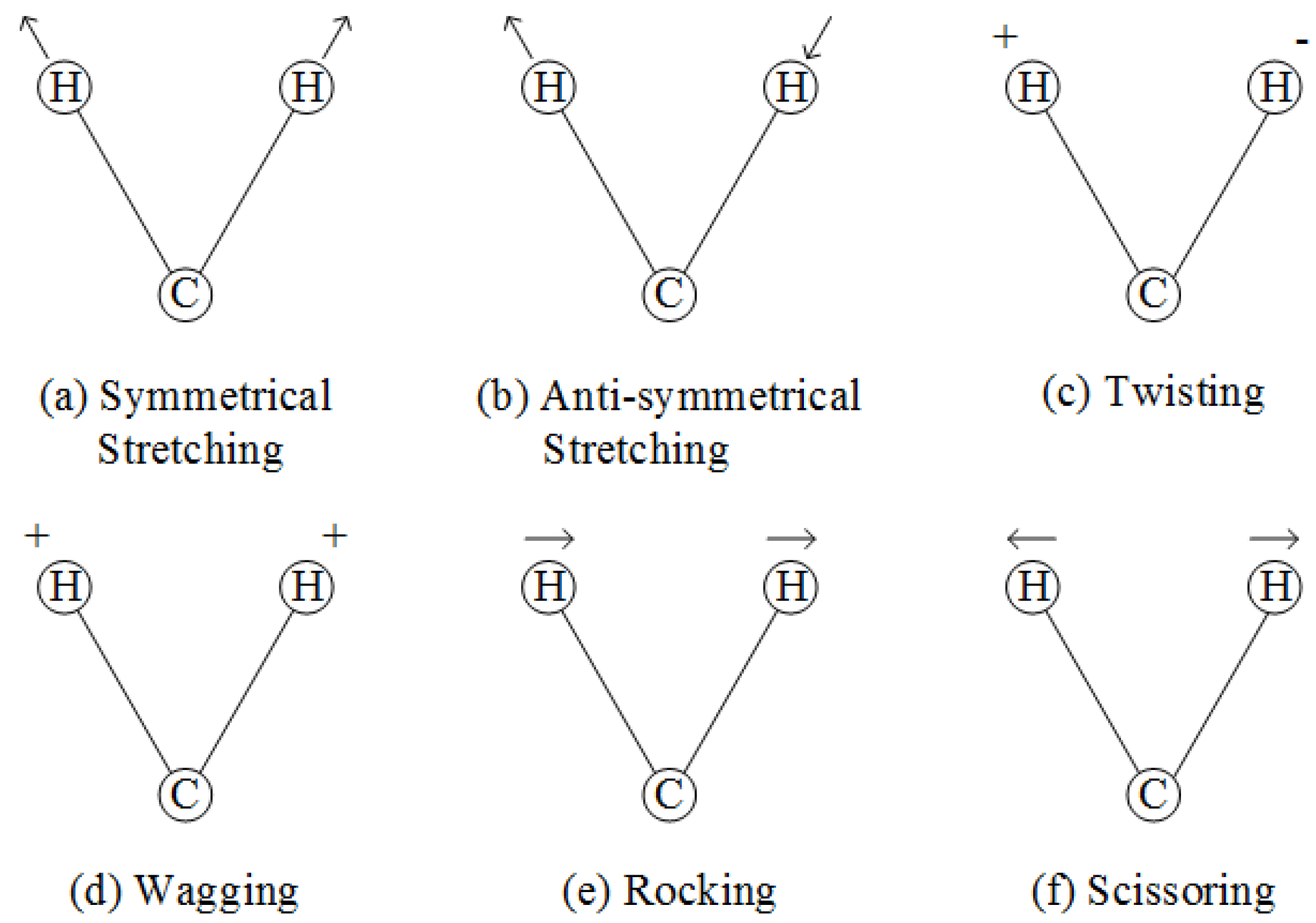
2.2.3. Infrared (IR) Spectroscopy
2.2.4. Near-Infrared (NIR) Spectroscopy
2.2.5. Mid-Infrared (MIR) Spectroscopy
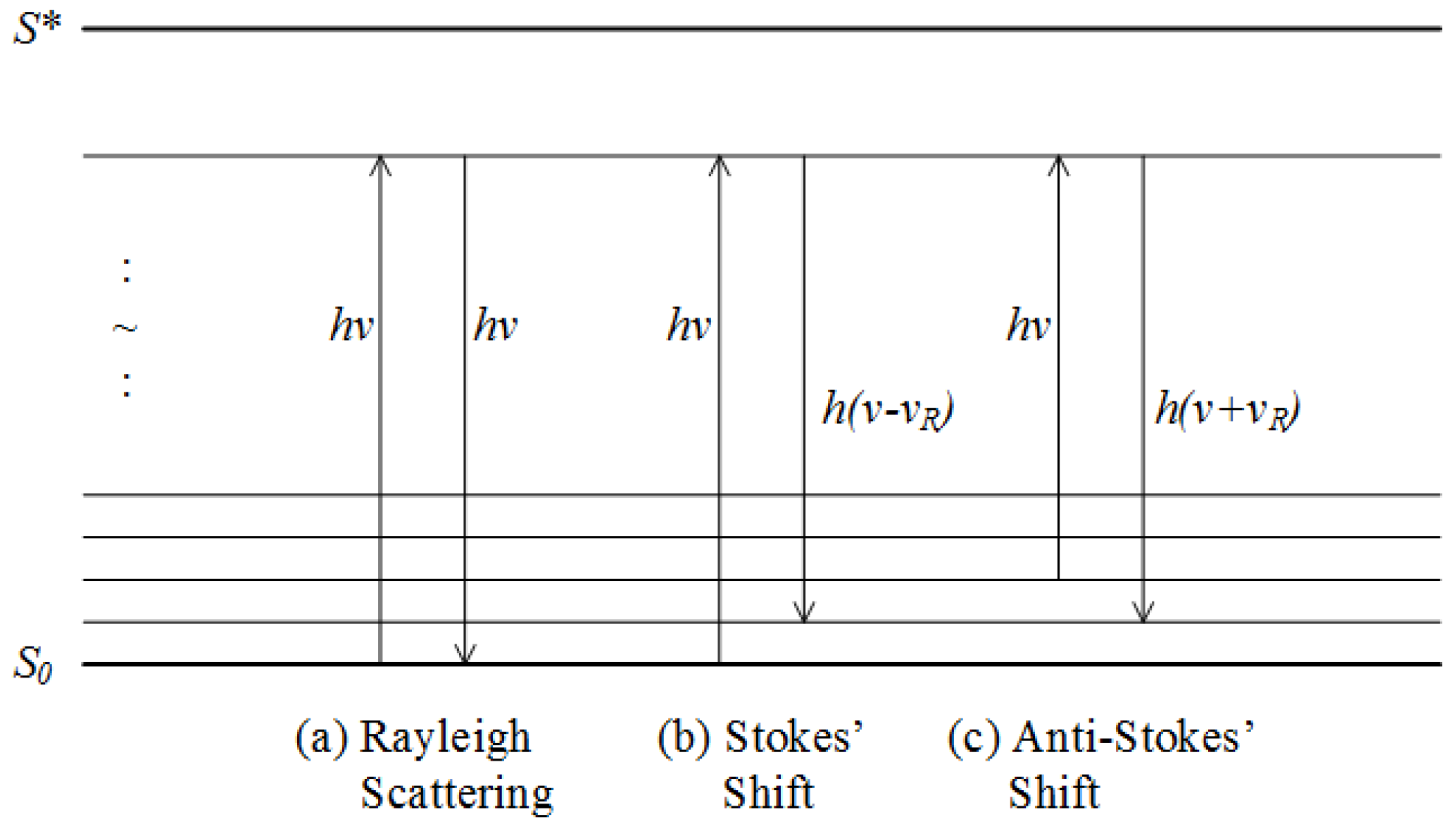
2.2.6. Raman Spectroscopy
2.2.7. Additional Spectroscopy Techniques
2.2.8. Spectroscopy Processing and Analysis
2.3. Spectral Imaging Technique
2.3.1. Classes of Spectral Imaging
2.3.2. Spectral Image Acquisition Methods
2.3.3. Spectral Imaging Sensing Modes
2.3.4. Spectral Imaging System Construction
2.3.5. Spectral Imaging Processing and Analysis
2.3.6. Pros and Cons of Spectral Imaging
2.4. Technique Comparison
3. Optics and Photonics Applications in Agriculture
3.1. Applications of Imaging Technique
3.2. Applications of Spectroscopy Technique
3.3. Applications of Spectral Imaging Technique
4. Photonics Techniques Implementation in Food Safety Inspection and Quality Control
5. Photonics Techniques Implementation in Tropical Countries Agriculture
5.1. Implementation in Palm Oil-Related Activities
5.2. Implementation in Natural Rubber Related Activities
5.3. Implementation in Agro-Food Crops Related Activities
5.4. Possible Challenges
6. Conclusions
- The incorporation of optical sensors into photonics detection techniques that serve as an early warning for drinking water pollution.
- The characterization of canned food or bottled beverages in the NIR (>1100 nm) and MIR wavebands for their optical “fingerprint” that correlates to the quality and food safety level of the product, such as preservatives concentration.
- The characterization on hazardous residual materials in food using optical spectroscopy, Raman spectroscopy and fluorescence.
- The implementation of an agricultural robot to perform better palm oil plantation management, scheduled collection of field latex and weed removal.
- The spectral imaging provides early detection of disease-causing G. boninense in the oil palm.
- Spectroscopy provides moisture content inspection, protein and lipid content detection, as well as improving the rubber vulcanizing process.
- The imaging technique detects external damage or bruises on organic fruits and vegetables.
Author Contributions
Funding
Conflicts of Interest
References
- National Research Council. Light: Wave-Particle Duality; The National Academic Press: Washington, DC, USA, 2013. [Google Scholar]
- An Overview of Optics & Photonics, Essential Technologies for Our Nation. Available online: https://www.scribd.com/document/113237529/ HLII-Brochure (accessed on 22 November 2018).
- Brown, L.M.; Pais, A.; Pippard, A.B. A History of Optical and Optoelectronic Physics in the Twentieth Century; American Institute of Physics Press: New York, NY, USA, 1995. [Google Scholar]
- Sternberg, E. Photonic Technology and Industrial Policy: U.S. Responses to Technological Change; State University of New York Press: New York, NY, USA, 1992. [Google Scholar]
- Sumriddetchkajorn, S. How Optics and Photonics is Simply Applied in Agriculture? In Proceedings of the International Conference on Photonics Solutions (ICPS), Pattaya City, Thailand, 7 June 2013; p. 888311. [Google Scholar] [CrossRef]
- Machine Vision in Agricultural Robotics—A Short Overview. Available online: https://pdfs.semanticscholar.org/ef13/5ac11c38022029da4d607343b33abb033758.pdf (accessed on 22 November 2018).
- Ji, B.; Zhu, W.; Liu, B.; Ma, C.; Li, X. Review of Recent Machine-Vision Technologies in Agriculture. In Proceedings of the Knowledge Acquisition and Modeling, 2009. KAM’09. Second International Symposium, Wuhan, China, 30 November–1 December 2009; pp. 330–334. [Google Scholar]
- Ishimwe, R.; Abutaleb, K.; Ahmed, F. Applications of Thermal Imaging in Agriculture—A Review. Adv. Remote Sens. 2014, 3, 128. [Google Scholar] [CrossRef]
- Szeliski, R. Computer Vision: Algorithms and Applications; Springer: London, UK, 2010. [Google Scholar]
- Gunasekaran, S.; Ding, K. Using computer vision for food quality evaluation. Food Technol. 1994, 6, 151–154. [Google Scholar]
- Bhargava, A.; Bansal, A. Fruits and vegetables quality evaluation using computer vision: A review. J. King Saud Univ. Comput. Inf. Sci. 2018. [Google Scholar] [CrossRef]
- Raj, M.P.; Swaminarayan, P.R.; Istar, A. Applications of image processing for grading agriculture products. Int. J. Recent Innov. Trends Comput. Commun. 2015, 3, 1194–1201. [Google Scholar]
- Mahendran, R.; Jayashree, G.C.; Alagusundaram, K. Application of computer vision technique on sorting and grading of fruits and vegetables. J. Food Process. Technol. 2012, 10, 2157–7110. [Google Scholar]
- Nezhad, M.A.K.B.; Massh, J.; Komleh, H.E. Tomato Picking Machine Vision Using with the Open CV’s library. In Proceedings of the 7th Iranian Conference on Machine Vision and Image Processing, Tehran, Iran, 16–17 November 2011; pp. 1–5. [Google Scholar]
- Bora, G.C.; Pathak, R.; Ahmadi, M.; Mistry, P. Image processing analysis to track colour changes on apple and correlate to moisture content in drying stage. Food Qual. Saf. 2018, 2, 105–110. [Google Scholar] [CrossRef]
- Digital Agriculture. Available online: https://sites.tufts.edu/eeseniordesignhandbook/2015/digital-agriculture/ (accessed on 29 April 2019).
- Nawrocka, A.; Lamorska, L. Advances in Agrophysical Research; IntechOpen: Rijeka, Croatia, 2013. [Google Scholar]
- Swinehart, D.F. The Beer-Lambert Law. J. Chem. Educ. 1962, 39, 333. [Google Scholar] [CrossRef]
- Animal and Vegetable Fats and Oils—Determination of Anisidine Value. Available online: https://www.iso.org/standard/69593.html (accessed on 22 November 2018).
- Gray, J.I. Measurement of Lipid Oxidation: A Review. J. Am. Oil Chem. Soc. 1978, 55, 539–546. [Google Scholar] [CrossRef]
- Animal and Vegetable Fats and Oils: Determination of Iodine Value. Available online: https://www.researchgate.net/publication/38999704_Animal_and_Vegetable_Fats_and_Oils_Determination_of_Iodine_Value (accessed on 23 November 2018).
- Mińkowski, K.; Grześkiewicz, S.; Jerzewska, M.; Ropelewska, M. Characteristic of chemical composition of vegetable oil about high contents of linoleic acids (in Polish). ŻYWNOŚĆ Nauka Technologia Jakość 2010, 73, 146–157. [Google Scholar]
- Psomiadou, E.; Tsimidou, M. Pigments in Greek virgin olive oils: Occurrence and levels. J. Sci. Food Agric. 2001, 81, 640–647. [Google Scholar] [CrossRef]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy; Springer: New York, NY, USA, 2006. [Google Scholar]
- Albani, J.R. Fluorescence Spectroscopy in Food Analysis. Encycl. Anal. Chem. 2006. [Google Scholar] [CrossRef]
- Karoui, R.; Blecker, C. Fluorescence spectroscopy measurement for quality assessment of food systems—A review. Food Bioprocess Technol. 2011, 4, 364–386. [Google Scholar] [CrossRef]
- Stuart, B.H. Infrared Spectroscopy: Fundamentals and Applications; John Wiley and Sons Ltd.: Chichester, UK, 2004. [Google Scholar]
- Shurvell, H. Spectra-Structure Correlations in the Mid- and Far-Infrared; John Wiley and Sons Ltd.: Chichester, UK, 2006. [Google Scholar]
- Li-Chan, E.C.Y.; Ismail, A.A.; Sedman, J.; Voort, F.R. Vibrational Spectroscopy of Food and Food Products; John Wiley and Sons Ltd.: Chichester, UK, 2006. [Google Scholar]
- Niaura, G. Raman Spectroscopy in Analysis of Biomolecules; John Wiley and Sons: New York, NY, USA, 2006. [Google Scholar]
- Nelson, S.O. Dielectric spectroscopy in agriculture. J. Non-Cryst. Solids 2005, 351, 2940–2944. [Google Scholar] [CrossRef]
- Mlynárik, V. Introduction to nuclear magnetic resonance. Anal. Biochem. 2016, 529, 4–9. [Google Scholar] [CrossRef]
- Guo, Z.; Huang, W.; Peng, Y.; Chen, Q.; Ouyang, Q.; Zhao, J. Color compensation and comparison of shortwave near infrared and long wave near infrared spectroscopy for determination of soluble solids content of ‘Fuji’ apple. Postharvest Biol. Technol. 2016, 115, 81–90. [Google Scholar] [CrossRef]
- Guo, Z.; Chen, Q.; Chen, L.; Huang, W.; Zhang, C.; Zhao, C. Optimization of Informative Spectral Variables for the Quantification of EGCG in Green Tea Using Fourier Transform Near-Infrared (FT-NIR) Spectroscopy and Multivariate Calibration. Appl. Spectrosc. 2011, 65, 1062–1067. [Google Scholar] [CrossRef]
- Qin, J.; Chao, K.; Kim, M.S.; Lu, R.; Burks, T.F. Hyperspectral and multispectral imaging for evaluating food safety and quality. J. Food Eng. 2013, 118, 157–171. [Google Scholar] [CrossRef]
- Wu, D.; Sun, D. Advanced applications of hyperspectral imaging technology for food quality and safety analysis and assessment: A review—Part I: Fundamentals. Innov. Food Sci. Emerg. Technol. 2013, 19, 1–14. [Google Scholar] [CrossRef]
- Kim, M.S.; Chao, K.; Chan, D.E.; Jun, W.; Lefcourt, A.M.; Delwiche, S.R.; Kang, S.; Lee, K. Line-scan hyperspectral imaging platform for agro-food safety and quality evaluation: System enhancement and characterization. Trans. ASABE 2011, 54, 703–711. [Google Scholar] [CrossRef]
- Morris, H.R.; Hoyt, C.C.; Treado, P.J. Imaging spectrometers for fluorescence and Raman microscopy–acousto-optic and liquid-crystal tunable filters. Appl. Spectrosc. 1994, 48, 857–866. [Google Scholar] [CrossRef]
- Kise, M.; Park, B.; Heitschmidt, G.W.; Lawrence, K.C.; Windham, W.R. Multispectral imaging system with interchangeable filter design. Comput. Electron. Agric. 2010, 72, 61–68. [Google Scholar] [CrossRef]
- Kim, M.S.; Chen, Y.R.; Mehl, P.M. Hyperspectral reflectance and fluorescence imaging system for quality and safety. Trans. ASAE 2001, 44, 721–729. [Google Scholar]
- Lu, R. Detection of bruises on apples using near-infrared hyperspectral imaging. Trans. ASAE 2003, 46, 523–530. [Google Scholar]
- Manley, M.; Williams, P.; Nilsson, D.; Geladi, P. Near infrared hyperspectral imaging for the evaluation of endosperm texture in whole yellow maize (Zea maize L.) kernels. J. Agric. Food Chem. 2009, 57, 8761–8769. [Google Scholar] [CrossRef]
- Litwiller, D. CMOS vs. CCD: Maturing technologies, maturing markets. Photonics Spectra 2005, 39, 54–61. [Google Scholar]
- ElMasry, G.; Wang, N.; Vigneault, C. Detecting chilling injury in Red Delicious apple using hyperspectral imaging and neural networks. Postharvest Biol. Technol. 2009, 52, 1–8. [Google Scholar] [CrossRef]
- Ngadi, M.O.; Liu, L. Hyperspectral Image Processing Techniques; Academic Press/Elsevier: Cambridge, MA, USA, 2010; pp. 99–127. [Google Scholar]
- ElMasry, G.; Wang, N.; ElSayed, A.; Ngadi, M. Hyperspectral imaging for non-destructive determination of some quality attributes for strawberry. J. Food Eng. 2007, 81, 98–107. [Google Scholar] [CrossRef]
- Qiao, J.; Wang, N.; Ngadi, M.O.; Gunenc, A.; Monroy, M.; Gariepy, C. Prediction of drip-loss, pH, and color for pork using a hyperspectral imaging technique. Meat Sci. 2007, 76, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.W.; Burks, T.F.; Ritenour, M.A.; Bonn, W.G. Detection of citrus canker using hyperspectral reflectance imaging with spectral information divergence. J. Food Eng. 2009, 93, 183–191. [Google Scholar] [CrossRef]
- Daugman, J.G. Uncertainty relation for resolution in space, spatial-frequency, and orientation optimized by two-dimensional visual cortical filters. J. Opt. Soc. Am. A Opt. Image Sci. Vis. 1985, 2, 1160–1169. [Google Scholar] [CrossRef]
- Lee, K.; Kang, S.; Delwiche, S.R.; Kim, M.S.; Noh, S. Correlation analysis of hyperspectral imagery for multispectral wavelength selection for detection of defects on apples. Sens. Instrum. Food Qual. Saf. 2008, 2, 90–96. [Google Scholar] [CrossRef]
- Kim, M.S.; Lefcourt, A.M.; Chao, K.; Chen, Y.R.; Kim, I.; Chan, D.E. Multispectral detection of fecal contamination on apples based on hyperspectral imagery: Part I–Application of visible and near-infrared reflectance imaging. Trans. ASAE 2002, 45, 2027. [Google Scholar]
- Park, B.; Lawrence, K.C.; Windham, W.R.; Buhr, R.J. Hyperspectral imaging for detecting fecal and ingesta contaminants on poultry carcasses. In 2001 ASAE Annual Meeting; American Society of Agricultural and Biological Engineers: St. Joseph, MI, USA, 1998. [Google Scholar] [CrossRef]
- Bajwa, S.G.; Bajcsy, P.; Groves, P.; Tian, L. Hyperspectral image data mining for band selection in agricultural applications. Trans. ASAE 2004, 47, 895. [Google Scholar] [CrossRef]
- Nakariyakul, S.; Casasent, D.P. Hyperspectral waveband selection for contaminant detection on poultry carcasses. Opt. Eng. 2008, 47, 087202. [Google Scholar]
- Xing, J.; Guver, D.; Ariana, D.; Lu, R. Determining optimal wavebands using genetic algorithm for detection of internal insect infestation in tart cherry. Sens. Instrum. Food Qual. Saf. 2008, 2, 161–167. [Google Scholar] [CrossRef]
- Leiva-Valenzuela, G.A.; Aguilera, J.M. Automatic detection of orientation and diseases in blueberries using image analysis to improve their postharvest storage quality. Food Control 2013, 33, 166–173. [Google Scholar] [CrossRef]
- Arakeri, M.P.; Lakshmana. Computer Vision Based Fruit Grading System for Quality Evaluation of Tomato in Agriculture industry. Procedia Comput. Sci. 2016, 79, 426–433. [Google Scholar] [CrossRef]
- Sumriddetchkajorn, S.; Somboonkaew, A.; Chanhorm, S. Mobile Device-Based Digital Microscopy for Education, Healthcare, and Agriculture. In Proceedings of the 9th International Conference on Electrical Engineering/Electronics, Computer, Telecommunications and Information Technology (ECTI-CON), Phetchaburi, Thailand, 16–18 May 2012; IEEE: Piscataway, NJ, USA, 2012; pp. 1–4. [Google Scholar] [CrossRef]
- Kaur, S.; Pandey, S.; Goel, S. Semi-automatic leaf disease detection and classification system for soybean culture. IET Image Process. 2018, 12, 1038–1048. [Google Scholar] [CrossRef]
- Billingsley, J. The Counting of Macadamia Nuts; Research Studies Press Ltd.: Baldock, UK, 2002. [Google Scholar]
- Chen, S.W.; Shivakumar, S.S.; Dcunha, S.; Das, J.; Okon, E.; Qu, C.; Taylor, C.J.; Kumar, V. Counting Apples and Oranges with Deep Learning: A Data-Driven Approach. IEEE Robot. Autom. Lett. 2017, 2, 781–788. [Google Scholar] [CrossRef]
- Intaravanne, Y.; Sumriddetchkajorn, S.; Nukeaw, J. Cell phone-based two-dimensional spectral analysis for banana ripeness estimation. Sens. Actuators B-Chem. 2012, 168, 390–394. [Google Scholar] [CrossRef]
- Marimuthu, S.; Roomi, S.M.M. Particle Swarm Optimized Fuzzy Model for the Classification of Banana Ripeness. IEEE Sens. J. 2017, 17, 4903–4915. [Google Scholar] [CrossRef]
- Intaravanne, Y.; Sumriddetchkajorn, S. Android-based rice leaf color analyzer for estimating the needed amount of nitrogen fertilizer. Comput. Electron. Agric. 2015, 116, 228–233. [Google Scholar] [CrossRef]
- Sulistyo, S.B.; Woo, W.L.; Dlav, S.S. Regularized Neural Networks Fusion and Genetic Algorithm Based On-Field Nitrogen Status Estimation of Wheat Plants. IEEE Trans. Ind. Inform. 2017, 13, 103–114. [Google Scholar] [CrossRef]
- Varith, J.; Hyde, G.; Baritelle, A.; Fellman, J.; Sattabongkot, T. Non-Contact Bruise Detection in Apples by Thermal Imaging. Innov. Food Sci. Emerg. Technol. 2003, 4, 211–218. [Google Scholar] [CrossRef]
- Danno, A.; Miyazato, M.; Ishiguro, E. Quality Evaluation of Agricultural Products by Infrared Imaging Method: Grading of Fruits for Bruise and Other Surface Defects. Mem. Fac. Agric. Kagoshima Univ. 1978, 14, 123–138. [Google Scholar]
- Oerke, E.; Fröhling, P.; Steiner, U. Thermographic Assessment of Scab Disease on Apple Leaves. Precis. Agric. 2011, 12, 699–715. [Google Scholar] [CrossRef]
- Oerke, E.; Steiner, U.; Dehne, H.; Lindenthal, M. Thermal Imaging of Cucumber Leaves Affected by Downy Mildew and Environmental Conditions. J. Exp. Bot. 2006, 57, 2121–2132. [Google Scholar] [CrossRef] [PubMed]
- Hellebrand, H.J.; Linke, M.; Beuche, H.; Herold, B.; Geyer, M. Horticultural Products Evaluated by Thermography; The Leibniz Institute for Agricultural Engineering Potsdam-Bornim: Potsdam, Germany, 2000. [Google Scholar]
- Danno, A.; Miyazato, M.; Ishiguro, E. Quality Evaluation of Agricultural Products by Infrared Imaging Method: Maturity Evaluation of Fruits and Vegetables. Mem. Fac. Agric. Kagoshima Univ. 1980, 16, 157–164. [Google Scholar]
- Wu, G.; Tan, Y.; Zheng, Y.; Wang, S. Walking Goal Line Detection Based on Machine Vision on Harvesting Robot. In Proceedings of the 2011 Third Pacific-Asia Conference on Circuits, Communications and System (PACCS), Wuhan, China, 17–18 July 2011; IEEE: Piscataway, NJ, USA, 2011; pp. 1–4. [Google Scholar]
- Tillett, N.D.; Hague, T.; Miles, S.J. A field assessment of a potential method for weed and crop mapping on the basis of crop planting geometry. Comput. Electron. Agric. 2001, 32, 229–246. [Google Scholar] [CrossRef]
- Irie, N.; Taguchi, N.; Horie, T.; Ishimatsu, T. Asparagus harvesting robot coordinated with 3-D vision sensor. In Proceedings of the IEEE International Conference on Industrial Technology (ICIT 2009), Gippsland, VIC, Australia, 10–13 February 2009; IEEE: Piscataway, NJ, USA, 2009; pp. 1–6. [Google Scholar]
- Astrand, B.; Baerveldt, A.J. A vision based row-following system for agricultural field machinery. Mechatronics 2005, 15, 251–269. [Google Scholar] [CrossRef]
- Songa, X.Y.; Lerova, T.; Vrankena, E.; Maertens, W.; Sonck, B.; Berckmans, D. Automatic detection of lameness in dairy cattle Vision-based trackway analysis in cow’s locomotion. Comput. Electron. Agric. 2008, 64, 39–44. [Google Scholar] [CrossRef]
- Kane, A.S.; Salierno, J.D.; Gipson, G.T.; Molteno, T.C.A.; Hunter, C. A video-based movement analysis system to quantify behavioural stress responses of fish. Water Res. 2004, 38, 3993–4001. [Google Scholar] [CrossRef]
- Baranowski, P.; Mazurek, W.; Wozniak, J.; Majewska, U. Detection of early bruises in apples using hyperspectral data and thermal imaging. J. Food Eng. 2012, 110, 345–355. [Google Scholar] [CrossRef]
- Stajnko, D.; Lakota, M.; Hocevar, M. Estimation of Number and Diameter of Apple Fruits in an Orchard during the Growing Season by Thermal Imaging. Comput. Electron. Agric. 2004, 42, 31–42. [Google Scholar] [CrossRef]
- Lv, J.; Shen, G.; Ma, Z. Acquisition of Fruit Region in Green Apple Image Based on the Combination of Segmented Regions. In Proceedings of the 2nd International Conference on Image, Vision and Computing, Chengdu, China, 2–4 June 2017. [Google Scholar]
- Dunn, M.; Billingsley, J. A Machine Vision System for Surface Texture Measurements of Citrus. In Proceedings of the 11th IEEE conference on Mechatronics and Machine Vision in Practice, Macau, China, 30 November–2 December 2004; pp. 73–76. [Google Scholar]
- Ballester, C.; Castel, J.; Jiménez-Bello, M.; Castel, J.; Intrigliolo, D. Thermographic Measurement of Canopy Temperature Is a Useful Tool for Predicting Water Deficit Effects on Fruit Weight in Citrus Trees. Agric. Water Manag. 2013, 122, 1–6. [Google Scholar] [CrossRef]
- Bonilla, J.; Prieto, F.; Pérez, C. Mass and Volume Estimation of Passion Fruit using Digital Images. IEEE. Lat. Am. Trans. 2017, 15, 275–282. [Google Scholar] [CrossRef]
- Stoll, M.; Schultz, H.R.; Loehnertz, B.B. Exploring the Sensitivity of Thermal Imaging for Plasmopara viticola Pathogen Detection in Grapevines under Different Water Status. Funct. Plant Biol. 2008, 35, 281–288. [Google Scholar] [CrossRef]
- Xiao-Lian, L.; Xiao-Rong, L.; Bing-Fu, L. Identification and Location of Picking Tomatoes Based on Machine Vision. In Proceedings of the 2011 International Conference on Intelligent Computation Technology and Automation (ICICTA), Shengzhen, Guangdong, China, 28–29 March 2011; pp. 101–107. [Google Scholar]
- Yang, L.; Dickinson, J.; Wu, Q.M.J.; Lang, S. A fruit recognition method for automatic harvesting. In Proceedings of the 14th International Conference on Mechatronics and Machine Vision in Practice (M2VIP2007), Xiamen, China, 3–5 December 2007; pp. 152–157. [Google Scholar]
- Vanlinden, V.; Vereycken, R.; Ramon, H.; Baerdemaeker, J.D. Detection technique for tomato bruise damage by thermal imaging. Acta Hortic. 2003, 599, 389–394. [Google Scholar] [CrossRef]
- Quan, Q.; Lanlan, T.; Xiaojun, Q.; Kai, J.; Qingchun, F. Selecting Candidate Regions of Clustered Tomato Fruits under Complex Greenhouse Scenes Using RGB-D Data. In Proceedings of the 3rd International Conference on Control, Automation and Robotics, Nagoya, Japan, 22–24 April 2017; pp. 389–393. [Google Scholar]
- Sa, I.; Lehnert, C.; McCool, C.; Dayoub, F.; Upcroft, B.; Perez, T. Peduncle Detection of Sweet Pepper for Autonomous Crop Harvesting-Combined Color and 3-D Information. IEEE Robot. Autom. Lett. 2017, 2, 765–772. [Google Scholar] [CrossRef]
- Speir, R.A.; Heidekker, M.A. Onion postharvest quality assessment with X-ray computed tomography–A pilot study. IEEE Instrum. Meas. Mag. 2017, 20, 15–19. [Google Scholar] [CrossRef]
- Shi-Gang, C.; Heng, L.; Xing-Li, W.; Yong-Li, Z.; Lin, H. Study on segmentation of lettuce image based on morphological reorganization and watershed algorithm. In Proceedings of the IEEE Chinese Control and Decision Conference (CCDC), IEEE, Shenyang, China, 9–11 June 2018; pp. 6595–6597. [Google Scholar]
- Lindenthal, M.; Steiner, U.; Dehne, H.; Oerke, E. Effect of Downy Mildew Development on Transpiration of Cucumber Leaves Visualized by Digital Infrared Thermography. Phytopathology 2005, 95, 233–240. [Google Scholar] [CrossRef]
- Oerke, E.; Lindenthal, M.; Fröhling, P.; Steiner, U. Digital Infrared Thermography for the Assessment of Leaf Pathogens. In Proceedings of the 5th European Conference on Precision Agriculture, Uppsala, Sweden, 9–11 June 2005; pp. 91–98. [Google Scholar]
- Smith, R.; Barrs, H.; Steiner, J.; Stapper, M. Relationship between Wheat Yield and Foliage Temperature: Theory and Its Application to Infrared Measurements. Agric. For. Meteorol. 1985, 36, 129–143. [Google Scholar] [CrossRef]
- Du, W.Y.; Zhang, L.D.; Hu, Z.F.; Shamaila, Z.; Zeng, A.J.; Song, J.L.; Liu, Y.J.; Wolfram, S.; Joachim, M.; He, X.K. Utilization of Thermal Infrared Image for Inversion of Winter Wheat Yield and Biomass. Spectrosc. Spectr. Anal. 2011, 31, 1476–1480. [Google Scholar]
- Hu, Z.; Zhang, L.; Wang, Y.; Shamaila, Z.; Zeng, A.; Song, J.; Liu, Y.; Wolfram, S.; Joachim, M.; He, X. Application of BP Neural Network in Predicting Winter Wheat Yield Based on Thermography Technology. Spectrosc. Spectr. Anal. 2013, 33, 1587–1592. [Google Scholar]
- Wanjura, D.; Upchurch, D.R. Water Status Response of Corn and Cotton to Altered Irrigation. Irrig. Sci. 2002, 21, 45–55. [Google Scholar] [CrossRef]
- Tetila, E.C.; Machado, B.B.; de Souza Belete, N.A.; Guimarães, D.A.; Pistori, H. Identification of Soybean Foliar Diseases Using Unmanned Aerial Vehicle Images. IEEE Geosci. Remote Sens. Lett. 2017, 14, 2190–2194. [Google Scholar] [CrossRef]
- Zhou, C.; Yang, G.; Liang, D.; Yang, X.; Xu, B. An Integrated Skeleton Extraction and Pruning Method for Spatial Recognition of Maize Seedlings in MGV and UAV Remote Images. IEEE Geosci. Remote Sens. Lett. 2018, 56, 4618–4632. [Google Scholar] [CrossRef]
- Zhang, X.; Qiao, Y.; Meng, F.; Fan, C.; Zhang, M. Identification of Maize Leaf Diseases Using Improved Deep Convolutional Neural Networks. IEEE Access 2018, 6, 30370–30377. [Google Scholar] [CrossRef]
- Lu, H.; Cao, Z.; Xiao, Y.; Fang, Z.; Zhu, Y. Toward good practices for fine-grained maize cultivar identification with filter-specific convolutional activations. IEEE Trans. Autom. Sci. Eng. 2018, 15, 430–442. [Google Scholar] [CrossRef]
- Padhi, J.; Misra, R.; Payero, J. Estimation of Soil Water Deficit in an Irrigated Cotton Field with Infrared Thermography. Field Crops Res. 2012, 126, 45–55. [Google Scholar] [CrossRef]
- Kamtongdee, C.; Sumriddetchkajorn, S.; Chanhorm, S.; Kaewhom, W. Noise reduction and accuracy improvement in optical-penetration-based silkworm gender identification. Appl. Opt. 2015, 54, 1844–1851. [Google Scholar] [CrossRef]
- Kranner, I.; Kastbergerb, G.; Hartbauerb, M.; Pritcharda, H.W. Noninvasive Diagnosis of Seed Viability Using Infrared Thermography. Proc. Natl. Acad. Sci. USA 2010, 107, 3912–3917. [Google Scholar] [CrossRef]
- Henry, D.; Aubert, H.; Véronèse, T.; Serrano, É. Remote estimation of intra-parcel grape quantity from three-dimensional imagery technique using ground-based microwave FMCW radar. IEEE Instrum. Meas. Mag. 2017, 20, 20–24. [Google Scholar] [CrossRef]
- Tosi, M.V.; Ferrante, V.; Mattiello, S.; Canali, E.; Verga, M. Comparison of video and direct observation methods for measuring oral behaviourin veal calves. Ital. J. Anim. Sci. 2006, 5, 19–27. [Google Scholar] [CrossRef][Green Version]
- Dunn, M.; Billingsley, J.; Finch, N. Machine Vision Classification of Animals; Research Studies Press Ltd.: Baldock, UK, 2003. [Google Scholar]
- Stien, L.H.; Brafland, S.; Austevollb, I.; Oppedal, F.; Kristiansen, T.S. A video analysis procedure for assessing vertical fish distribution in aquaculture tanks. Aquac. Eng. 2007, 37, 115–124. [Google Scholar] [CrossRef]
- Sumriddetchkajorna, S.; Chaitavonb, K.; Intaravanne, Y. Mobile-platform based colorimeter for monitoring chlorine concentration in water. Sens. Actuators B-Chem. 2014, 191, 561–566. [Google Scholar] [CrossRef]
- Iqbal, Z.; Bjorklund, R.B. Colorimetric analysis of water and sand samples performed on a mobile phone. Talanta 2011, 84, 1118–1123. [Google Scholar] [CrossRef] [PubMed]
- Nagy, A.; Riczu, P.; Tamás, J. Spectral evaluation of apple fruit ripening and pigment contentalteration. Sci. Hortic. 2016, 201, 256–264. [Google Scholar] [CrossRef]
- Roy, S.; Anantheswaran, R.C.; Shenk, J.S.; Beelman, R. Determination of moisture content of mushrooms by Vis-NIR spectroscopy. J. Sci. Food Agric. 1993, 63, 355–360. [Google Scholar] [CrossRef]
- Hartmann, R.; Büning-Pfaue, H. NIR determination of potato constituents. Potato Res. 1998, 41, 327–334. [Google Scholar] [CrossRef]
- Lee, M.; Hwang, Y.; Lee, J.; Choung, M. The characterization of caffeine and nine individual catechins in the leaves of green tea (Camellia sinensis L.) by near-infrared reflectance spectroscopy. Food Chem. 2014, 158, 351–357. [Google Scholar] [CrossRef]
- Sun, X.; Liu, Y.; Li, Y.; Wu, M.; Zhu, D. Simultaneous measurement of brown core and soluble solids content in pear by on-line visible and near infrared spectroscopy. Postharvest Biol. Technol. 2016, 116, 80–87. [Google Scholar] [CrossRef]
- Viegas, T.R.; Mata, A.L.M.L.; Duarte, M.M.L.; Lima, K.M.G. Determination of quality attributes in wax jambu fruit using NIRS and PLS. Food Chem. 2016, 190, 1–4. [Google Scholar] [CrossRef]
- Gente, R.; Busch, S.F.; Stübling, E.; Schneider, L.M.; Hirschmann, C.B.; Balzer, J.C.; Koch, M. Quality control of sugar beet seeds with THz time-domain spectroscopy. IEEE Trans. Terahertz Sci. Technol. 2016, 6, 754–756. [Google Scholar] [CrossRef]
- Nunes, K.M.; Andrade, M.V.O.; Filho, A.M.P.S.; Lasmar, M.C.; Sena, M.M. Detection and characterisation of frauds in bovine meat in natura by non-meat ingredient additions using data fusion of chemical parameters and ATR-FTIR spectroscopy. Food Chem. 2016, 205, 14–22. [Google Scholar] [CrossRef] [PubMed]
- ElMasry, G.; Nagai, H.; Moria, K.; Nakazawa, N.; Tsuta, M.; Sugiyama, J.; Okazaki, E.; Nakauchi, S. Freshness estimation of intact frozen fish using fluorescence spectroscopy and chemometrics of excitation-emission matrix. Talanta 2015, 143, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Huang, Y.; Lai, K.; Rasco, B.A.; Fan, Y. Surface-enhanced Raman spectroscopy coupled with gold nanoparticles for rapid detection of phosmet and thiabendazole residues in apples. Food Control 2016, 68, 229–235. [Google Scholar] [CrossRef]
- Cozzolino, D. Near infrared spectroscopy as a tool to monitor contaminants in soil, sediments and water–State of the art, advantages and pitfalls. Trends Environ. Anal. Chem. 2016, 9, 1–7. [Google Scholar] [CrossRef]
- Nie, P.; Dong, T.; He, Y.; Xiao, S. Research on the effects of drying temperature on nitrogen detection of different soil types by near infrared sensors. Sensors 2018, 18, 391. [Google Scholar] [CrossRef]
- Xiao, S.; He, Y.; Dong, T.; Nie, P. Spectral Analysis and Sensitive Waveband Determination Based on Nitrogen Detection of Different Soil Types Using Near Infrared Sensors. Sensors 2018, 18, 523. [Google Scholar] [CrossRef]
- Hernández, M.; Valentín, J.L.; López-Manchado, M.A.; Ezquerra, T.A. Influence of the vulcanization system on the dynamics and structure of natural rubber: Comparative study by means of broadband dielectric spectroscopy and solid-state NMR spectroscopy. Eur. Polym. J. 2015, 68, 90–103. [Google Scholar] [CrossRef]
- Mahani, R.; Atia, F.; Neklawy, M.M.A.; Fahem, A. Dielectric spectroscopic studies on the water hyacinth plant collected from agriculture drainage. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2016, 162, 81–85. [Google Scholar] [CrossRef]
- Jha, S.N.; Narsaiah, K.; Jaiswal, P.; Bhardwaj, R.; Gupta, M.; Kumar, R.; Sharma, R. Nondestructive prediction of maturity of mango using near infrared spectroscopy. J. Food Eng. 2014, 124, 152–157. [Google Scholar] [CrossRef]
- Guo, W.; Gu, J.; Liu, D.; Shang, L. Peach variety identification using near-infrared diffuse reflectance spectroscopy. Comput. Electron. Agric. 2016, 123, 297–303. [Google Scholar] [CrossRef]
- González-Fernández, A.B.; Rodríguez-Pérez, J.R.; Marabel, M.; Álvarez-Taboada, F. Spectroscopic estimation of leaf water content in commercial vineyards using continuum removal and partial least squares regression. Sci. Hortic. 2015, 188, 15–22. [Google Scholar] [CrossRef]
- Schulz, H.; Drews, H.; Quilitzsch, R.; Krüger, H. Application of near infrared spectroscopy for the quantification of quality parameters in selected vegetables and essential oil plants. J. Near Infrared Spectrosc. 1998, 6, A125–A130. [Google Scholar] [CrossRef]
- Evans, S.D.; Muir, A.Y. Reflectance Spectrophotometry of Bruising in Potatoes. I. Ultraviolet to Near Infrared. Int. Agrophys. 1999, 13, 203–210. [Google Scholar]
- Birth, G.S.; Dull, G.G.; Renfroe, W.T.; Kays, S.J. Nondestructive Spectrophotometric Determination of Dry Matter in Onions. J. Am. Soc. Hortic. Sci. 1985, 110, 297–303. [Google Scholar]
- Zhang, C.; Kong, W.; Liu, F.; He, Y. Measurement of aspartic acid in oilseed rape leaves under herbicide stress using near infrared spectroscopy and chemometrics. Heliyon 2016, 2, e00064. [Google Scholar] [CrossRef]
- Ambrose, A.; Lohumi, S.; Lee, W.; Cho, B.K. Comparative nondestructive measurement of corn seed viability using Fourier transform near-infrared (FT-NIR) and Raman spectroscopy. Sens. Actuators B-Chem. 2016, 224, 500–506. [Google Scholar] [CrossRef]
- Pearson, T.C. Spectral Properties and Effect of Drying Temperature on Almonds with Concealed Damage. LWT-Food Sci. Technol. 1999, 32, 67–72. [Google Scholar] [CrossRef]
- Pearson, T.C. Use of Near Infrared Transmittance to Automatically Detect Almonds with Concealed Damage. LWT-Food Sci. Technol. 1999, 32, 73–78. [Google Scholar] [CrossRef]
- Lian, F.; Xu, D.; Fu, M.; Ge, H.; Jiang, Y.; Zhang, Y. Identification of Transgenic Ingredients in Maize Using Terahertz Spectra. IET Nanobiotechnol. 2017, 7, 378–384. [Google Scholar] [CrossRef]
- Prasad, T.N.; Adam, S.; Rao, P.V.; Reddy, B.R.; Krishna, T.G. Size dependent effects of antifungal phytogenic silver nanoparticles on germination, growth and biochemical parameters of rice (Oryza sativa L), maize (Zea mays L) and peanut (Arachis hypogaea L). IET Nanobiotechnol. 2016, 11, 277–285. [Google Scholar] [CrossRef]
- Sahar, A.; Rahman, U.; Kondjoyan, A.; Portanguen, S.; Dufour, E. Monitoring of thermal changes in meat by synchronous fluorescence spectroscopy. J. Food Eng. 2016, 168, 160–165. [Google Scholar] [CrossRef]
- Liu, M.; Yao, L.; Wang, T.; Li, J.; Yu, C. Rapid determination of egg yolk contamination in egg white by VIS spectroscopy. J. Food Eng. 2014, 124, 117–121. [Google Scholar] [CrossRef]
- Núñez-Sánchez, N.; Martínez-Marín, A.L.; Polvillo, O.; Fernández-Cabanás, V.M.; Carrizosa, J.; Urrutia, B.; Serradilla, J.M. Near Infrared Spectroscopy (NIRS) for the determination of the milk fat fatty acid profile of goats. Food Chem. 2016, 190, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Almoselhy, R.I.M.; Allam, M.H.; El-Kalyoubi, M.H.; El-Sharkawy, A.A. 1H NMR spectral analysis as a new aspect to evaluate the stability of some edible oils. Ann. Agric. Sci. 2014, 59, 201–206. [Google Scholar] [CrossRef]
- Mabood, F.; Boqué, R.; Folcarelli, R.; Busto, O.; Jabeen, F.; Al-Harrasi, A.; Hussain, J. The effect of thermal treatment on the enhancement of detection of adulteration in extra virgin olive oils by synchronous fluorescence spectroscopy and chemometric analysis. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2016, 161, 83–87. [Google Scholar] [CrossRef]
- Hzounda, J.B.F.; Jazet, P.M.D.; Lazar, G.; Răducanu, D.; Caraman, I.; Bassene, E.; Boyom, F.F.; Lazarca, I.M. Spectral and chemometric analyses reveal antioxidant properties ofessential oils from four Cameroonian Ocimum. Ind. Crops Prod. 2016, 80, 101–108. [Google Scholar] [CrossRef]
- Dutta, D.; Das, P.K.; Bhunia, U.K.; Singh, U.; Singh, S.; Sharma, J.R.; Dadhwal, V.K. Retrieval of tea polyphenol at leaf level using spectral transformation and multi-variate statistical approach. Int. J. Appl. Earth Obs. Geoinf. 2015, 36, 22–29. [Google Scholar] [CrossRef]
- Marquetti, I.; Link, J.V.; Lemes, A.L.G.; dos Santos Scholz, M.B.; Valderrama, P.; Bona, E. Partial least square with discriminant analysis and near infrared spectroscopy for evaluation of geographic and genotypic origin of arabica coffee. Comput. Electron. Agric. 2016, 121, 313–319. [Google Scholar] [CrossRef]
- Bertone, E.; Venturello, A.; Giraudo, A.; Pellegrino, G.; Geobaldo, F. Simultaneous determination by NIR spectroscopy of the roasting degree and Arabica/Robusta ratio in roasted and ground coffee. Food Control 2016, 59, 683–689. [Google Scholar] [CrossRef]
- Ayvaz, H.; Sierra-Cadavid, A.; Aykas, D.P.; Mulqueeney, B.; Sullivan, S.; Rodriguez-Saona, L.E. Monitoring multicomponent quality traits in tomato juice using portable mid-infrared (MIR) spectroscopy and multivariate analysis. Food Control 2016, 66, 79–86. [Google Scholar] [CrossRef]
- Ye, M.; Gao, Z.; Li, Z.; Yuan, Y.; Yue, T. Rapid detection of volatile compounds in apple wines using FT-NIR spectroscopy. Food Chem. 2016, 190, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Cho, B.; Lee, S.H.; Kwon, K.; Park, E.S.; Lee, W. Application of Fourier transform-mid infrared reflectance spectroscopy for monitoring Korean traditional rice wine ‘Makgeolli’ fermentation. Sens. Actuators B-Chem. 2016, 230, 753–760. [Google Scholar] [CrossRef]
- Fortier, C.; Rodgers, J. Preliminary Examinations for the Identification of U.S. Domestic and International Cotton Fibers by Near-Infrared Spectroscopy. Fibers 2014, 2, 264–274. [Google Scholar] [CrossRef]
- Liu, Y.; Delhom, C.; Campbell, B.T.; Martin, V. Application of near infrared spectroscopy in cotton fiber micronaire measurement. Inf. Process. Agric. 2016, 3, 30–35. [Google Scholar] [CrossRef]
- Rolere, S.; Liengprayoon, S.; Vaysse, L.; Sainte-Beuve, J.; Bonfils, F. Investigating natural rubber composition with Fourier Transform Infrared (FT-IR) spectroscopy: A rapid and non-destructive method to determine both protein and lipid contents simultaneously. Polym. Test. 2015, 43, 83–93. [Google Scholar] [CrossRef]
- Musto, P.; Larobina, D.; Cotugno, S.; Straffi, P.; Florio, G.D.; Mensitieri, G. Confocal Raman imaging, FTIR spectroscopy and kinetic modelling of the zinc oxide/stearic acid reaction in a vulcanizing rubber. Polymer 2013, 54, 685–693. [Google Scholar] [CrossRef]
- Yrieix, M.; Cruz-Boisson, F.D.; Majesté, J. Rubber/silane reaction sand grafting rates investigated by liquid-state NMR spectroscopy. Polymer 2016, 87, 90–97. [Google Scholar] [CrossRef]
- Suchat, S.; Theanjumol, P.; Karrila, S. Rapid moisture determination for cup lump natural rubber by near infrared spectroscopy. Ind. Crops Prod. 2015, 76, 772–780. [Google Scholar] [CrossRef]
- Avinash, B.; Venu, R.; Prasad, T.N.; Rao, K.S.; Srilatha, C. Synthesis and characterisation of neem leaf extract, 2, 3-dehydrosalanol and quercetin dihydrate mediated silver nano particles for therapeutic applications. IET Nanobiotechnol. 2016, 11, 383–389. [Google Scholar] [CrossRef]
- Cécillon, L.; Barthès, B.; Gomez, C.; Ertlen, D.; Génot, V.; Hedde, M.; Stevens, A.; Brun, J. Assessment and monitoring of soil quality using near infrared reflectance spectroscopy (NIRS). Eur. J. Oral Sci. 2009, 60, 770–784. [Google Scholar]
- Zhang, Y.; Li, M.; Zheng, L.; Zhao, Y.; Pei, X. Soil nitrogen content forecasting based on real-time NIR spectroscopy. Comput. Electron. Agric. 2016, 124, 29–36. [Google Scholar] [CrossRef]
- Ludwig, B.; Linsler, D.; Höper, H.; Schimdt, H.; Piepho, H.; Vohland, M. Pitfalls in the use of middle-infrared spectroscopy: Representativeness and ranking criteria for the estimation of soil properties. Geoderma 2016, 268, 165–175. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, F.; Gupta, B.B.; Rho, S.; Liu, Q.; Hou, H.; Jing, D.; Shen, W. Variable Selection and Optimization in Rapid Detection of Soybean Straw Biomass Based on CARS. Cellulose 2018, 144, 28–51. [Google Scholar] [CrossRef]
- Symonds, P.; Paap, A.; Alameh, K.; Rowe, J.; Miller, C. A real-time plant discrimination system utilising discrete reflectance spectroscopy. Comput. Electron. Agric. 2015, 117, 57–69. [Google Scholar] [CrossRef]
- Bennedsen, B.S.; Peterson, D.L.; Tabb, A. Identifying defects in images of rotating apples. Comput. Electron. Agric. 2005, 48, 92–102. [Google Scholar] [CrossRef]
- Gowen, A.A.; O’Donnell, C.P.; Taghizadeh, M.; Cullen, P.J.; Frias, J.M.; Downey, G. Hyperspectral imaging combined with principal component analysis for bruise damage detection on white mushrooms (Agaricus bisporus). J. Chemom. 2008, 22, 259–267. [Google Scholar] [CrossRef]
- Lleo, L.; Barreiro, P.; Ruiz-Altisent, M.; Herrero, A. Multispectral images of peach related to firmness and maturity at harvest. J. Food Eng. 2009, 93, 229–235. [Google Scholar] [CrossRef]
- Zhang, C.; Guo, C.; Liu, F.; Kong, W.; He, Y.; Lou, B. Hyperspectral imaging analysis for ripeness evaluation of strawberry with support vector machine. J. Food Eng. 2016, 179, 11–18. [Google Scholar] [CrossRef]
- Polder, G.; van der Heijden, G.W.; Young, I.T. Spectral image analysis for measuring ripeness of tomatoes. Trans. ASAE 2002, 45, 1155–1161. [Google Scholar] [CrossRef]
- Hahn, F. Multi-spectral prediction of unripe tomatoes. Biosyst. Eng. 2002, 81, 147–155. [Google Scholar] [CrossRef]
- Chen, J.; Cai, F.; He, R.; He, S. Experimental Demonstration of Remote and Compact Imaging Spectrometer Based on Mobile Devices. Sensors 2018, 18, 1989. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Peng, Y. Development of a multispectral imaging prototype for real-time detection of apple fruit firmness. Opt. Eng. 2007, 46, 123201. [Google Scholar]
- Baiano, A.; Terracone, C.; Peri, G.; Romaniello, R. Application of hyperspectral imaging for prediction of physico-chemical and sensory characteristics of table grapes. Comput. Electron. Agric. 2012, 87, 142–151. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, Q.; Cai, J.; Ouyang, Q. Automated tea quality classification by hyperspectral imaging. Appl. Opt. 2009, 48, 3557–3564. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Burks, T.F.; Zhao, X.; Niphadkar, N.; Ritenour, M.A. Development of a two-band spectral imaging system for real-time citrus canker detection. J. Food Eng. 2012, 108, 87–93. [Google Scholar] [CrossRef]
- Wang, W.; Li, C.; Tollner, E.W.; Gitaitis, R.D.; Rains, G.C. Shortwave infrared hyperspectral imaging for detecting sour skin (Burkholderia cepacia)-infected onions. J. Food Eng. 2012, 109, 38–48. [Google Scholar] [CrossRef]
- Chao, K.; Mehl, P.M.; Chen, Y.R. Use of hyper- and multi-spectral imaging for detection of chicken skin tumors. Appl. Eng. Agric. 2002, 18, 113. [Google Scholar] [CrossRef]
- Kumar, A.; Manjunath, K.R.; Meenakshi; Bala, R.; Sud, R.K.; Singh, R.D.; Panigrahy, S. Field hyperspectral data analysis for discriminating spectral behavior of tea plantations under various management practices. Int. J. Appl. Earth Obs. Geoinf. 2013, 23, 352–359. [Google Scholar] [CrossRef]
- Martins, G.D.; Galo, M.D.L.B.T.; Vieira, B.S. Detecting and Mapping Root-Knot Nematode Infection in Coffee Crop Using Remote Sensing Measurements. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2017, 10, 5395–5403. [Google Scholar] [CrossRef]
- Leiva-Valenzuela, G.A.; Lu, R.; Aguilera, J.M. Prediction of firmness and soluble solids content of blueberries using hyperspectral reflectance imaging. J. Food Eng. 2013, 115, 91–98. [Google Scholar] [CrossRef]
- Qin, J.; Lu, R. Measurement of the absorption and scattering properties of turbid liquid foods using hyperspectral imaging. Appl. Spectrosc. 2007, 61, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Gila, D.M.M.; Marchal, P.C.; García, J.G.; Ortega, J.G. On-line system based on hyperspectral information to estimate acidity, moisture and peroxides in olive oil samples. Comput. Electron. Agric. 2015, 116, 1–7. [Google Scholar] [CrossRef]
- Keresztes, J.C.; Goodarzi, M.; Saeys, W. Real-time pixel based early apple bruise detection using short wave infrared hyperspectral imaging in combination with calibration and glare correction techniques. Food Control 2016, 66, 215–226. [Google Scholar] [CrossRef]
- Baranowski, P.; Mazurek, W.; Pastuszka-Woźniak, J. Supervised classification of bruised apples with respect to the time after bruising on the basis of hyperspectral imaging data. Postharvest Biol. Technol. 2013, 86, 249–258. [Google Scholar] [CrossRef]
- Kim, M.S.; Lee, K.; Chao, K.; Lefcourt, A.M.; Jun, W.; Chan, D.E. Multispectral line-scan imaging system for simultaneous fluorescence and reflectance measurements of apples: Multitask apple inspection system. Sens. Instrum. Food Qual. Saf. 2008, 2, 123–129. [Google Scholar] [CrossRef]
- Lu, R.; Peng, Y. Hyperspectral scattering for assessing peach fruit firmness. Biosyst. Eng. 2006, 93, 161–171. [Google Scholar] [CrossRef]
- Rajkumar, P.; Wang, N.; EImasry, G.; Raghavan, G.S.V.; Gariepy, Y. Studies on banana fruit quality and maturity stages using hyperspectral imaging. J. Food Eng. 2012, 108, 194–200. [Google Scholar] [CrossRef]
- Leiva-Valenzuela, G.A.; Lu, R.; Aguilera, J.M. Assessment of internal quality of blueberries using hyperspectral transmittance and reflectance images with whole spectra or selected wavelengths. Innov. Food Sci. Emerg. Technol. 2014, 24, 2–13. [Google Scholar] [CrossRef]
- Qin, J.; Lu, R. Detection of pits in tart cherries by hyperspectral transmission imaging. Trans. ASAE 2005, 48, 1963–1970. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Y.R.; Wang, C.; Chan, D.E.; Kim, M.S. Development of a simple algorithm for the detection of chilling injury in cucumbers from visible/near-infrared hyperspectral imaging. Appl. Spectrosc. 2005, 59, 78–85. [Google Scholar] [CrossRef]
- Hernández-Hierro, J.M.; Esquerre, C.; Valverde, J.; Villacreces, S.; Reilly, K.; Gaffne, M.; González-Miret, M.L.; Heredia, F.J.; O’Donnell, C.P.; Downey, G. Preliminary study on the use of near infrared hyperspectral imaging for quantitation and localisation of total glucosinolates in freeze-dried broccoli. J. Food Eng. 2014, 126, 107–112. [Google Scholar] [CrossRef]
- Trong, D.; Nyugen, N.; Tsuta, M.; Nicolaï, B.M.; Baerdemaeker, J.D.; Saeys, W. Prediction of optimal cooking time for boiled potatoes by hyperspectral imaging. J. Food Eng. 2011, 105, 617–624. [Google Scholar] [CrossRef]
- Onoyama, H.; Ryu, C.; Suguri, M.; Iida, M. Estimation of Nitrogen Contents in Rice Plant at the Panicle Initiation Stage Using Ground-Based Hyperspectral Remote Sensing. IFAC Proc. Vol. 2010, 43, 166–171. [Google Scholar] [CrossRef]
- Onoyama, H.; Ryu, C.; Suguri, M.; Iida, M. Potential of Hyperspectral Imaging for Constructing a Year-invariant Model to Estimate the Nitrogen Content of Rice Plants at the Panicle Initiation Stage. IFAC Proc. Vol. 2013, 46, 219–224. [Google Scholar] [CrossRef]
- Suwansukho, K.; Sumriddetchkajorn, S.; Buranasiri, P. Demonstration of a single-wavelength spectral-imaging-based Thai jasmine rice identification. Appl. Opt. 2011, 50, 4024–4030. [Google Scholar] [CrossRef]
- Zhang, H.; Paliwal, J.; Jayas, D.S.; White, N.D.G. Classification of fungal infected wheat kernels using near-infrared reflectance hyperspectral imaging and support vector machine. Trans. ASABE 2007, 50, 1779–1785. [Google Scholar] [CrossRef]
- Xing, J.; Huang, P.; Symons, S.; Shahin, M.; Hatcher, D. Using a short wavelength infrared (SWIR) hyperspectral imaging system to predict alpha amylase activity in individual Canadian western wheat kernels. Sens. Instrum. Food Qual. Saf. 2009, 3, 211. [Google Scholar] [CrossRef]
- Patrick, A.; Pelham, S.; Culbreath, A.; Holbrook, C.C.; De Godoy, I.J.; Li, C. High throughput phenotyping of tomato spot wilt disease in peanuts using unmanned aerial systems and multispectral imaging. IEEE Instrum. Meas. Mag. 2017, 20, 4–12. [Google Scholar] [CrossRef]
- Weinstock, B.A.; Janni, J.; Hagen, L.; Wright, S. Prediction of oil and oleic acid concentrations in individual corn (Zea mays L.) kernels using near-infrared reflectance hyperspectral imaging and multivariate analysis. Appl. Spectrosc. 2006, 60, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Hruska, Z.; Kincaid, R.; Brown, R.; Cleveland, T.; Bhatnagar, D. Correlation and classification of single kernel fluorescence hyperspectral data with aflatoxin concentration in corn kernels inoculated with Aspergillus flavus spores. Food Addict. Contam. Part A Chem. 2010, 27, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Chao, K.; Chen, Y.R.; Hruschka, W.R.; Park, B. Chicken heart disease characterization by multi-spectral imaging. Appl. Eng. Agric. 2001, 17, 99. [Google Scholar] [CrossRef]
- Kise, M.; Park, B.; Lawrence, K.C.; Windham, W.R. Design and calibration of a dual-band imaging system. Sens. Instrum. Food Qual. Saf. 2007, 1, 113–121. [Google Scholar] [CrossRef]
- Chao, K.; Yang, C.C.; Kim, M.S.; Chan, D.E. High throughput spectral imaging system for wholesomeness inspection of chicken. Appl. Eng. Agric. 2008, 24, 475–485. [Google Scholar] [CrossRef]
- Naganathan, G.K.; Grimes, L.M.; Subbiah, J.; Calkins, C.R.; Samal, A.; Meyer, G.E. Visible/near-infrared hyperspectral imaging for beef tenderness prediction. Comput. Electron. Agric. 2008, 64, 225–233. [Google Scholar] [CrossRef]
- Peng, Y.; Zhang, J.; Wang, W.; Li, Y.; Wu, J.; Huang, H.; Gao, X.; Jiang, W. Potential prediction of the microbial spoilage of beef using spatially resolved hyperspectral scattering profiles. J. Food Eng. 2011, 102, 163–169. [Google Scholar] [CrossRef]
- Kamruzzaman, M.; ElMasry, G.; Sun, D.; Allen, P. Application of NIR hyperspectral imaging for discrimination of lamb muscles. J. Food Eng. 2011, 104, 332–340. [Google Scholar] [CrossRef]
- Tao, F.; Peng, Y. A method for nondestructive prediction of pork meat quality and safety attributes by hyperspectral imaging technique. J. Food Eng. 2014, 126, 98–106. [Google Scholar] [CrossRef]
- Barbin, D.; Elmasry, G.; Sun, D.; Allen, P. Near-infrared hyperspectral imaging for grading and classification of pork. Meat Sci. 2012, 90, 259–268. [Google Scholar] [CrossRef] [PubMed]
- ElMasry, G.; Wold, J.P. High-speed assessment of fat and water content distribution in fish fillets using online imaging spectroscopy. J. Agric. Food Chem. 2008, 56, 7672–7677. [Google Scholar] [CrossRef] [PubMed]
- Sivertsen, A.H.; Chu, C.K.; Wang, L.C.; Godtliebsen, F.; Heia, K.; Nilsen, H. Ridge detection with application to automatic fish fillet inspection. J. Food Eng. 2009, 90, 317–324. [Google Scholar] [CrossRef]
- Wu, D.; Sun, D. Potential of time series-hyperspectral imaging (TS-HSI) for non-invasive determination of microbial spoilage of salmon flesh. Talanta 2013, 111, 39–46. [Google Scholar] [CrossRef]
- Wu, D.; Shi, H.; Wang, S.; Hea, Y.; Bao, Y.; Liu, K. Rapid prediction of moisture content of dehydrated prawns using online hyperspectral imaging system. Anal. Chim. Acta 2012, 726, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Shi, H.; He, Y.; Yu, X.; Bao, Y. Potential of hyperspectral imaging and multivariate analysis for rapid and non-invasive detection of gelatin adulteration in prawn. J. Food Eng. 2013, 119, 680–686. [Google Scholar] [CrossRef]
- Lim, J.; Kim, G.; Mo, C.; Kim, M.S.; Chao, K.; Qin, J.; Fu, X.; Baek, I.; Cho, B. Detection of melamine in milk powders using near-infrared hyperspectral imaging combined with regression coefficient of partial least square regression model. Talanta 2016, 151, 183–191. [Google Scholar] [CrossRef]
- Qin, J.; Chao, K.; Kim, M.S. Raman chemical imaging system for food safety and quality inspection. Trans. ASABE 2010, 53, 1873–1882. [Google Scholar] [CrossRef]
- Deng, S.; Xu, Y.; Li, X.; He, Y. Moisture content prediction in tealeaf with near infrared hyperspectral imaging. Comput. Electron. Agric. 2015, 118, 38–46. [Google Scholar] [CrossRef]
- Wu, D.; Yang, H.; Chen, X.; He, Y.; Li, X. Application of image texture for the sorting of tea categories using multi-spectral imaging technique and support vector machine. J. Food Eng. 2008, 88, 474–483. [Google Scholar] [CrossRef]
- Chemura, A.; Mutanga, O.; Odindi, J. Empirical Modeling of Leaf Chlorophyll Content in Coffee (Coffea Arabica) Plantations with Sentinel-2 MSI Data: Effects of Spectral Settings, Spatial Resolution, and Crop Canopy Cover. IEEE J. Sel. Top. Appl. Earth Obs. Remote Sens. 2017, 10, 5541–5550. [Google Scholar] [CrossRef]
- Heibati, M.; Stedmon, C.A.; Stenroth, K.; Rauch, S.; Toljander, J.; Säve-Söderbergh, M.; Murphy, K.R. Assessment of drinking water quality at the tap using fluorescence spectroscopy. Water Res. 2017, 125, 1–10. [Google Scholar] [CrossRef]
- Sorensen, J.P.R.; Vivanco, A.; Ascott, M.J.; Gooddy, D.C.; Lapworth, D.J.; Read, D.S.; Rushworth, C.M.; Bucknall, J.; Herbert, K.; Karapanos, I.; et al. Online fluorescence spectroscopy for the real-time evaluation of the microbial quality of drinking water. Water Res. 2018, 137, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Højris, B.; Christensen, S.C.B.; Albrechtsen, H.J.; Smith, C.; Dahlqvist, M. A novel, optical, on-line bacteria sensor for monitoring drinking water quality. Sci. Rep. 2016, 6, 23935. [Google Scholar] [CrossRef] [PubMed]
- Mignani, A.G.; Ciaccheri, L.; Ottevaere, H.; Thienpont, H.; Conte, L.; Marega, M.; Cichelli, A.; Attilio, C.; Cimato, A. Visible and near-infrared absorption spectroscopy by an integrating sphere and optical fibers for quantifying and discriminating the adulteration of extra virgin olive oil from Tuscany. Anal. Bioanal. Chem. 2011, 399, 1315–1324. [Google Scholar] [CrossRef]
- Woodcock, T.; Downey, G.; O’Donnel, C. Near infrared spectral fingerprinting for confirmation of claimed PDO provenance of honey. Food Chem. 2009, 114, 742–746. [Google Scholar] [CrossRef]
- Li, X.; He, Y.; Wu, C.; Sun, D.W. Non desctructive measurement and fingerprint analysis of soluble content of tea soft drink based on Vis/NIR spectroscopy. J. Food Eng. 2007, 82, 316–323. [Google Scholar] [CrossRef]
- Fagan, C.C.; Castillo, M.; O’Donnel, C.P.; Callaghan, D.J.; Payne, F.A. Online prediction of cheese making indices using backscatter of near infrared light. Int. Dairy J. 2008, 18, 120–128. [Google Scholar] [CrossRef]
- Egidio, V.D.; Oliveri, P.; Woodcock, T.; Downey, G. Confirmation of brand identity in foods by near infrared transflectance spectroscopy using classification and class-modelling chemometric techniques—The example of a Belgian beer. Food Res. Int. 2011, 44, 544–549. [Google Scholar] [CrossRef]
- Leong, Y.S.; Ker, P.J.; Jamaludin, M.Z.; Nomanbhay, S.M.; Ismail, A.; Abdullah, F.; Looe, H.M.; Shukri, C.N.S. New near-infrared absorbance peak for inhibitor content detection in transformer insulating oil. Sens. Actuators B Chem. 2018, 266, 577–582. [Google Scholar] [CrossRef]
- Li, T.L.; Chung-Wang, Y.J.; Shih, Y.C. Determination and confirmation of chloramphenicol residues in swine muscle and liver. J. Food Sci. 2002, 67, 21–28. [Google Scholar] [CrossRef]
- Overview: Malaysian Agricultural Biotechnology. BiotechCorp, 2009. Available online: http://www.bioeconomycorporation.my/wp-content/uploads/2011/11/publications/White_Paper_Agricultural.pdf (accessed on 25 November 2018).
- Ahmad, T.T.M.A.; Suntharalingam, C. Transformation and Economic Growth of the Malaysian Agricultural Sector. Econ. Technol. Manag. Rev. 2009, 4, 1–10. [Google Scholar]
- Matahir, H. The Empirical Investigation of the Nexus between Agricultural and Industrial Sectors in Malaysia. Int. J. Bus. Manag. Soc. Res. 2012, 3, 225–231. [Google Scholar]
- Onn, F.C. Small and Medium Industries in Malaysia: Economic Efficiency and Entrepreneurship. Dev. Econ. 1990, 28, 152–179. [Google Scholar]
- Rahman, A.A.Z. Economic Reforms and Agricultural Development in Malaysia. ASEAN Econ. Bull. 1998, 15, 59–76. [Google Scholar] [CrossRef]
- Shaffril, M.H.A.; Asmuni, A.; Ismail, A. The Ninth Malaysian Plan and Agriculture Extension Officer Competency: A Combination for Intensification of Paddy Industry in Malaysian. J. Int. Soc. Res. 2010, 3, 450–457. [Google Scholar]
- Eleventh Malaysia Plan 2016–2020 Anchoring Growth on People; Percetakan Nasional Malaysia: Kuala Lumpur, Malaysia, 2015; Available online: https://www.mkma.org/Notice%20Board/2015/MP11Book.pdf (accessed on 25 November 2018).
- Murad, M.W.; Mustapha, N.H.; Siwar, C. Review of Agricultural Policies with Regards to Sustainability. Am. J. Environ. Sci. 2008, 4, 608–614. [Google Scholar] [CrossRef]
- Tenth Malaysia Plan 2011–2015; Percetakan Nasional Malaysia: Kuala Lumpur, Malaysia, 2010. Available online: http://www.pmo.gov.my/dokumenattached/RMK/RMK10_E.pdf (accessed on 25 November 2018).
- Tagoe, S.M.A.; Dickinson, M.J.; Apetorgbor, M.M. Factors influencing quality of palm oil produced at the cottage industry level in Ghana. Int. Food Res. J. 2012, 19, 271–278. [Google Scholar]
- Hushiarian, R.; Yusof, N.A.; Dutse, S.W. Detection and control of Ganoderma boninense: Strategies and perspectives. SpringerPlus 2013, 2, 555. [Google Scholar] [CrossRef] [PubMed]
- Lelong, C.C.; Roger, J.; Brégand, S.; Dubertret, F.; Lanore, M.; Sitorus, N.; Raharjo, D.; Caliman, J. Evaluation of Oil-palm fungal disease infestation with canopy hyperspectral reflectance data. Sensors 2010, 10, 734–747. [Google Scholar] [CrossRef] [PubMed]
| Wavelength (nm) | Wavenumber (cm−1) | Assignment |
|---|---|---|
| Water | ||
| 1454 | 6878 | 1st overtone O–H stretching |
| 1932 | 5176 | O–H combination |
| Proteins | ||
| 1208 | 8278 | 2nd overtone C–H stretching |
| 1465 | 6826 | 1st overtone N–H and O–H stretching |
| 1734 | 5767 | 1st overtone C–H stretching |
| 1932 2058 2180 | 5176 4859 4587 | N–H combination and O–H stretching |
| 2302 2342 | 4344 4270 | C–H stretching combination |
| Oil | ||
| 1210 | 8264 | 2nd overtone C–H stretching |
| 1406 | 7112 | 1st overtone N–H and O–H stretching |
| 1718 1760 | 5821 5682 | 1st overtone C–H stretching |
| 2114 | 4730 | N–H combination and O–H stretching |
| 2308 2346 | 4333 4263 | C–H stretching combination |
| Starch | ||
| 1204 | 8306 | 2nd overtone C–H stretching |
| 1464 | 6831 | 1st overtone N–H and O–H stretching |
| 1932 2100 | 5176 4762 | N–H combination and O–H stretching |
| 2290 2324 | 4367 4303 | C–H stretching combination |
| Wavelength (nm) | Wavenumber (cm−1) | Assignment |
|---|---|---|
| Water | ||
| 2.778–3.125 | 3200–3600 | O–H stretching |
| 6.061 | 1650 | H–OH stretching |
| Proteins | ||
| 5.917–6.250 | 1600–1690 | Amide I (C=O stretching) |
| 6.349–6.757 | 1480–1575 | Amide II (C–N stretching and N–H bending) |
| 7.692–8.130 | 1230–1300 | Amide III (C–N stretching and N–H bending) |
| Fats | ||
| 3.333–3.571 | 2800–3000 | C–H stretching |
| 5.731–5.797 | 1725–1745 | C=O stretching |
| 10.309 | 970 | C=C–H bending |
| Carbohydrates | ||
| 3.333–3.571 | 2800–3000 | C–H stretching |
| 7.143–12.500 | 800–1400 | Skeletal stretching and bending |
| Wavelength (nm) | Wavenumber (cm−1) | Assignment |
|---|---|---|
| Water | ||
| 2.778–3.125 | 3200–3600 | O–H stretching |
| Proteins | ||
| 19.608 19.048 18.349 | 510 525 545 | S–S stretching |
| 14.925–15.873 13.423–14.286 | 630–670 700–745 | C–S stretching |
| 5.882–6.250 | 1600–1700 | Amide I (C=O stretching and N–H bending) |
| 8.032–8.097 | 1235–1245 | Amide III (C–N stretching and N–H bending) |
| 3.876–3.922 | 2550–2580 | S–H stretching |
| 3.333–3.571 | 2800–3000 | C–H stretching |
| Fats | ||
| 6.940 | 1441 | CH2 bending |
| 6.863 | 1457 | CH3–CH2 bending |
| 6.039 | 1656 | C=C stretching |
| 3.378–3.503 | 2855–2960 | C–H stretching |
| Carbohydrates | ||
| 11.962 | 836 | C–C stretching |
| 9.398 | 1064 | C–O stretching |
| 3.434 3.397 | 2912 2944 | C–H stretching |
| 2.898 | 3451 | O–H stretching |
| Characteristics | Imaging | Spectroscopy | Spectral Imaging |
|---|---|---|---|
| Spectral information | × | ✓ | ✓ |
| Spatial information | ✓ | × | ✓ |
| Multi-constituent information | × | ✓ | ✓ |
| Sensitivity to small-sized objects | ✓ | × | ✓ |
| Flexibility of spectral extraction | × | × | ✓ |
| Generation of quality-attribute distribution | × | × | ✓ |
| Class | Product | Application | Ref. |
|---|---|---|---|
| Fruit | Apple | Bruise detection (thermal) | [66,67,70,78] |
| Apple | Maturity evaluation (thermal) | [70] | |
| Apple | Yield estimation (thermal) | [79] | |
| Apple | Scab disease detection (thermal) | [68] | |
| Green apple | Acquisition of segmented fruit region | [80] | |
| Green apple and orange | Yield estimation | [61] | |
| Orange | Texture analysis | [81] | |
| Orange | Bruise detection (thermal) | [67] | |
| Citrus | Water stress evaluation (thermal) | [82] | |
| Pear | Maturity evaluation (thermal) | [71] | |
| Banana | Maturity evaluation | [62] | |
| Banana | Maturity evaluation | [63] | |
| Persimmon | Maturity evaluation (thermal) | [71] | |
| Passion fruit | Mass and volume estimation | [83] | |
| Blueberry | Bruise detection | [56] | |
| Grapevine | Pathogen detection (thermal) | [84] | |
| Tomato | Fruit detection | [85,86] | |
| Tomato | Bruise detection and maturity evaluation | [57] | |
| Tomato | Bruise detection (thermal) | [87] | |
| Tomato | Maturity evaluation (thermal) | [71] | |
| Tomato | Clustered fruit detection | [88] | |
| Sweet peppers | Peduncle detection | [89] | |
| Onion | Post-harvest quality assessment (thermal) | [90] | |
| Lettuce | Segmentation of vegetable | [91] | |
| Cucumber | Downy mildew disease detection (thermal) | [69,92,93] | |
| Grain | Rice leaf | Nitrogen content detection | [64] |
| Wheat | Yield estimation (thermal) | [94,95,96] | |
| Corn | Water stress evaluation (thermal) | [97] | |
| Macadamia nuts | Yield estimation | [60] | |
| Soybean | Identification of foliar disease | [98] | |
| Soybean | Identification of leaf disease | [59] | |
| Maize | Yield estimation (thermal) | [99] | |
| Maize | Identification of leaf disease | [100] | |
| Maize | Cultivar identification | [101] | |
| Commercial | Cotton | Water stress evaluation (thermal) | [97,102] |
| Silkworm | Gender identification | [103] | |
| Farm and Plantation | Seed | Viability evaluation (thermal) | [104] |
| Wheat field | Estimation of nutrient content | [65] | |
| Cauliflower plantation | Weed detection | [73] | |
| Asparagus plantation | Crop harvest robot vision | [74] | |
| Sugar beet and rape plantation | Agriculture robot vision | [75] | |
| Grapevines | Estimation of intra-parcel grape quantities | [105] | |
| Cow farm | Behavioural studies | [76,106] | |
| Goat and sheep farm | Animal species identification | [107] | |
| Fish aquarium | Behavioural studies | [77,108] | |
| Baby shrimp farm | Chlorine level detection | [109] | |
| Orchid farm | Disease and pest detection | [58] | |
| Surface and ground water | Chemical content detection | [110] |
| Class | Product | Application | Method | Wavelength (nm) | Ref. |
|---|---|---|---|---|---|
| Fruit | Apple | Pigment content change during ripening | UV-VIS-NIR | 400–1000 | [111] |
| Apple | Soluble solid content detection | VIS-NIR | 500–1100, 1000–2500 | [33] | |
| Apple | Pesticide residue detection | Raman | 5–18 µm | [120] | |
| Pear | Brown core and soluble solid content detection | UV-VIS-NIR | 200–1100 | [115] | |
| Mango | Maturity evaluation | NIR | 1200–2200 | [126] | |
| Peach | Peach variety identification | NIR | 833–2500 | [127] | |
| Wax jambu | Quality inspection | NIR | 1000–2400 | [116] | |
| Grape leaf | Water content estimation | UV-VIS-NIR | 350–2500 | [128] | |
| Vegetable | Carrot | Carotenoid, fructose, glucose, sucrose and sugar content detection | NIR | 1108–2490 | [129] |
| Potato | Bruise detection | UV-VIS-NIR | 250–1750 | [130] | |
| Potato | Protein, fructose, glucose, starch and sucrose content detection | NIR | 1100–2500 | [113] | |
| Onion | Soluble solid content detection | VIS-NIR | 500–1200 | [131] | |
| Oilseed rape leaf | Aspartic acid content detection | NIR | 1100–2500 | [132] | |
| Sugar beet seeds | Quality control | Time-domain spectroscopy | 250–350 GHz | [117] | |
| Mushroom | Moisture content detection | VIS-NIR | 600–2200 | [112] | |
| Grain | Corn seed | Viability evaluation | NIR Raman | 1000–2500 3.125–59 µm | [133] |
| Almond | Internal defect detection | VIS-NIR | 700–1400 | [134,135] | |
| Maize | Identification of transgenic ingredients | THz spectral | 0–4.5 THz | [136] | |
| Rice, maize and peanut | Germination and growth of crop | UV-VIS FTIR | 380.85–796.62 nm 562.72–3865.11 cm−1 | [137] | |
| Meat | Beef | Thermal change inspection | Fluorescence | 250–550 | [138] |
| Beef | Adulteration detection | NIR-MIR | 2.5–19 µm | [118] | |
| Frozen fish | Freshness evaluation | Fluorescence | 250–800 | [119] | |
| Dairy | Egg | Contamination detection | UV-VIS-NIR | 200–860 | [139] |
| Goat milk | Fatty acid content detection | VIS-NIR | 400–2498 | [140] | |
| Oil | Edible oil | Stability analysis | NMR | 300 MHz (1H) | [141] |
| Olive oil | Adulteration detection | Fluorescence | 250–720 | [142] | |
| Ocimum essential oil | Antioxidant property identification | NIR-MIR | 2.5–18 µm | [143] | |
| Beverage | Tea leaf | Tea polyphenol level detection | UV-VIS-NIR | 347–2506 | [144] |
| Green tea leaf | Caffeine and catechins content detection | VIS-NIR | 400–2500 | [114] | |
| Coffee | Geographic and genotypic origin identification | NIR | 1100–2498 | [145] | |
| Coffee | Roasting degree and blend composition detection | NIR | 800–2857 | [146] | |
| Tomato juice | Quality inspection | NIR-MIR | 2.5–14 µm | [147] | |
| Apple wine | Volatile compound detection | NIR | 833–2500 | [148] | |
| Rice wine | Fermentation monitoring | NIR-MIR | 2.5–25 µm | [149] | |
| Commercial | Cotton fibre | Cotton type identification | NIR | 800–2500 | [150] |
| Cotton fibre | Cotton fibre micronaire measurement | VIS-NIR | 400–2500 | [151] | |
| Natural rubber | Protein and lipid content detection | NIR-MIR | 2.5–25 µm | [152] | |
| Natural rubber | Chemical interaction during vulcanizing process | NIR-MIR Raman | 2.5–25 µm 3.125–100 µm, 6.25–50 µm | [153] | |
| Natural rubber | Rubber silane reaction | NMR | 400 MHz (1H), 100.6 MHz (13C) | [154] | |
| Natural rubber | Moisture content detection | VIS-NIR | 400–1100 | [155] | |
| Natural rubber | Vulcanization system effect | Dielectric NMR | 10-1 < Hz < 107 20 MHz (1H) | [124] | |
| Neem leaf | Pest control | UV-VIS FTIR XRD | 200–800 nm 250–4000 cm−1 10–80° | [156] | |
| Farm and Plantation | Soil | Quality inspection | NIR | 780–5000 | [157] |
| Soil | Nitrogen content detection | NIR | 800–2564 | [158] | |
| Soil | Chemical and physical property estimation | NIR-MIR | 1430–2500, 2.5–27 µm | [159] | |
| Soil | Nitrogen detection | NIR | 900–1700 | [122] | |
| Soil | Nitrogen detection | NIR | 900–1700 | [123] | |
| Soil and water | Contaminant detection | VIS-NIR | 400–2500 | [121] | |
| Water hyacinth Soybean straw | Pollutant concentration detection Detection of biomass | Dielectric Fluorescence Near infrared spectroscopy | 10-1 < Hz < 106 N/A 4000–12,000 cm−1 | [125] [160] | |
| Flower | Plant type identification | VIS | 635, 685, 785 | [161] |
| Class | Product | Application | Method | Wavelength (nm) | Ref. |
|---|---|---|---|---|---|
| Fruit | Apple | Bruise detection | Hyper. line scan | 400–2500, 1000–2500 | [78,180] |
| Apple | Bruise detection timing | Hyper. line scan | 400–2500 | [181] | |
| Apple | Bruise detection | Multi. area scan | 740, 950 | [162] | |
| Apple | Bruise and faeces detection | Multi. line scan | 530, 665, 750, 800 | [182] | |
| Apple | Firmness evaluation | Multi. area scan | 680, 880, 905, 940 | [169] | |
| Citrus | Canker detection | Multi. area scan | 730, 830 | [172] | |
| Peach | Firmness evaluation | Hyper. line scan | 500–1000 | [183] | |
| Peach | Maturity evaluation | Multi. area scan | 450, 675, 800 | [164] | |
| Cantaloupe | Faeces detection | Hyper. line scan | 425–774 | [184] | |
| Blueberry | Firmness evaluation, soluble solid content detection | Hyper. line scan | 400–1000 | [177,185] | |
| Strawberry | Maturity evaluation | Hyper. line scan | 380–1030 874–1734 | [165] | |
| Cherry | Pit detection | Hyper. line scan | 450–1000 | [186] | |
| Grape | Quality evaluation | Hyper. line scan | 400–1000 | [170] | |
| Banana | Maturity evaluation | Hyper. area scan | 500–700 | [168] | |
| Tomato | Maturity evaluation | Hyper. line scan | 396–736 | [166] | |
| Tomato | Maturity evaluation | Multi. area scan | 530, 595, 630, 850 | [167] | |
| Cucumber | Chilling injury detection | Hyper. line scan | 447–951 | [187] | |
| Vegetable | Freeze-dried broccoli | Glucosinolate detection | Hyper. line scan | 400–1700 | [188] |
| Potato | Cooking time prediction | Hyper. line scan | 400–1000 | [189] | |
| Onion | Sour skin disease detection | Hyper. area scan | 950–1650 | [173] | |
| Mushroom | Bruise detection | Hyper. line scan | 400–1000 | [163] | |
| Grain | Rice plant | Nitrogen content detection | Hyper. line scan | 400–1000 | [190,191] |
| Thai jasmine rice | Rice variety identification | Multi. area scan | 545, 575 | [192] | |
| Wheat | Fungus detection | Hyper. area scan | 1000-1600 | [193] | |
| Wheat | Damage detection | Hyper. line scan | 1000–2500 | [194] | |
| Peanut | Tomato spot wilt disease detection | Multi. Area scan | 475, 560, 668, 717, 840 | [195] | |
| Corn | Oil and oleic acid content detection | Hyper. area scan | 950-1700 | [196] | |
| Corn | Aflatoxin detection | Hyper. line scan | 400–600 | [197] | |
| Meat | Chicken | Skin tumour detection | Hyper. line scan | 420–850 | [174] |
| Chicken | Heart disease detection | Multi. area scan | 495, 535, 585, 605 | [198] | |
| Chicken | Faeces detection | Multi. area scan | 520, 560 | [199] | |
| Chicken | Wholesomeness inspection | Multi. line scan | 580, 620 | [200] | |
| Beef | Tenderness evaluation | Hyper. line scan | 400–1000 | [201] | |
| Beef | Microbial spoilage detection | Hyper. line scan | 400–1100 | [202] | |
| Lamb | Lamb variety identification | Hyper. line scan | 900–1700 | [203] | |
| Pork meat | E. coli detection | Hyper. line scan | 470–960 | [204] | |
| Pork meat | Quality inspection | Hyper. line scan | 900–1700 | [205] | |
| Fish | Moisture and fat content detection | Hyper. line scan | 460–1040 | [206] | |
| Fish | Ridge detection | Hyper. line scan | 400–1000 | [207] | |
| Salmon | Microbial spoilage detection | Hyper. line scan | 400–1000 880–1720 | [208] | |
| Dehydrated prawn | Moisture content detection | Hyper. line scan | 380–1100 | [209] | |
| Prawn | Adulteration detection | Hyper. line scan | 380–1030 900–1700 | [210] | |
| Dairy | Milk powder | Melamine detection | Hyper. line scan | 990–1700 | [211] |
| Milk | Fat content detection | Hyper. line scan | 530–900 | [178] | |
| Milk | Melamine detection | Hyper. point scan | 4–98 µm | [212] | |
| Oil | Olive oil | Free acidity, peroxide and moisture content detection | Hyper. line scan | 900–1700 | [179] |
| Beverage | Tea | Quality inspection | Hyper. line scan | 408–1117 | [171] |
| Tea | Moisture content detection | Hyper. line scan | 874–1734 | [213] | |
| Tea | Tea variety identification | Multi. area scan | 580, 680, 800 | [214] | |
| Farm and Plantation | Tea bush | Tea variety, growth status and disease identification | Hyper. area scan | 325–1075 | [175] |
| Coffee crop | Detection of disease/infection | Hyper. area scan | 440–850 | [176] | |
| Coffee plantation | Monitoring chlorophyll content | Multi. area scan | 490–2190 | [215] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, J.Y.; Ker, P.J.; Lau, K.Y.; Hannan, M.A.; Tang, S.G.H. Applications of Photonics in Agriculture Sector: A Review. Molecules 2019, 24, 2025. https://doi.org/10.3390/molecules24102025
Tan JY, Ker PJ, Lau KY, Hannan MA, Tang SGH. Applications of Photonics in Agriculture Sector: A Review. Molecules. 2019; 24(10):2025. https://doi.org/10.3390/molecules24102025
Chicago/Turabian StyleTan, Jin Yeong, Pin Jern Ker, K. Y. Lau, M. A. Hannan, and Shirley Gee Hoon Tang. 2019. "Applications of Photonics in Agriculture Sector: A Review" Molecules 24, no. 10: 2025. https://doi.org/10.3390/molecules24102025
APA StyleTan, J. Y., Ker, P. J., Lau, K. Y., Hannan, M. A., & Tang, S. G. H. (2019). Applications of Photonics in Agriculture Sector: A Review. Molecules, 24(10), 2025. https://doi.org/10.3390/molecules24102025






