Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Proximate Composition
2.3. Polyphenols Extraction
2.4. Total Polyphenols
2.5. DPPH Radical Scavenging Activity
2.6. Preparation of FAME
2.7. GC-FID Analysis of Fatty Acids
2.8. GC-FID Analysis of Unsaponificable
2.9. Mineralization of Samples
2.10. ICP-OES Analysis
2.11. ATR-FTIR Analysis
2.12. Statistical Analysis
3. Results and Discussion
3.1. Proximate Composition
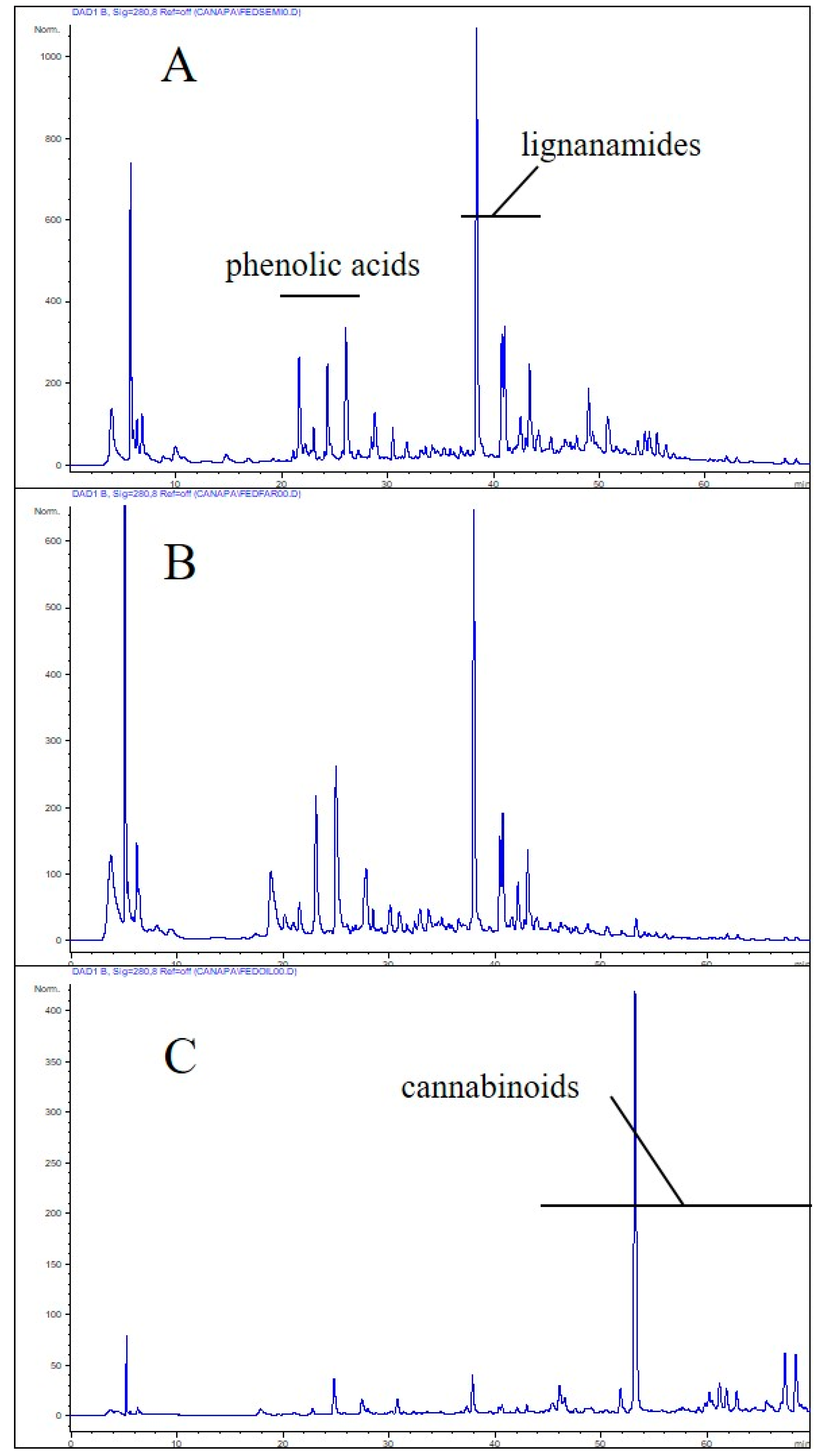
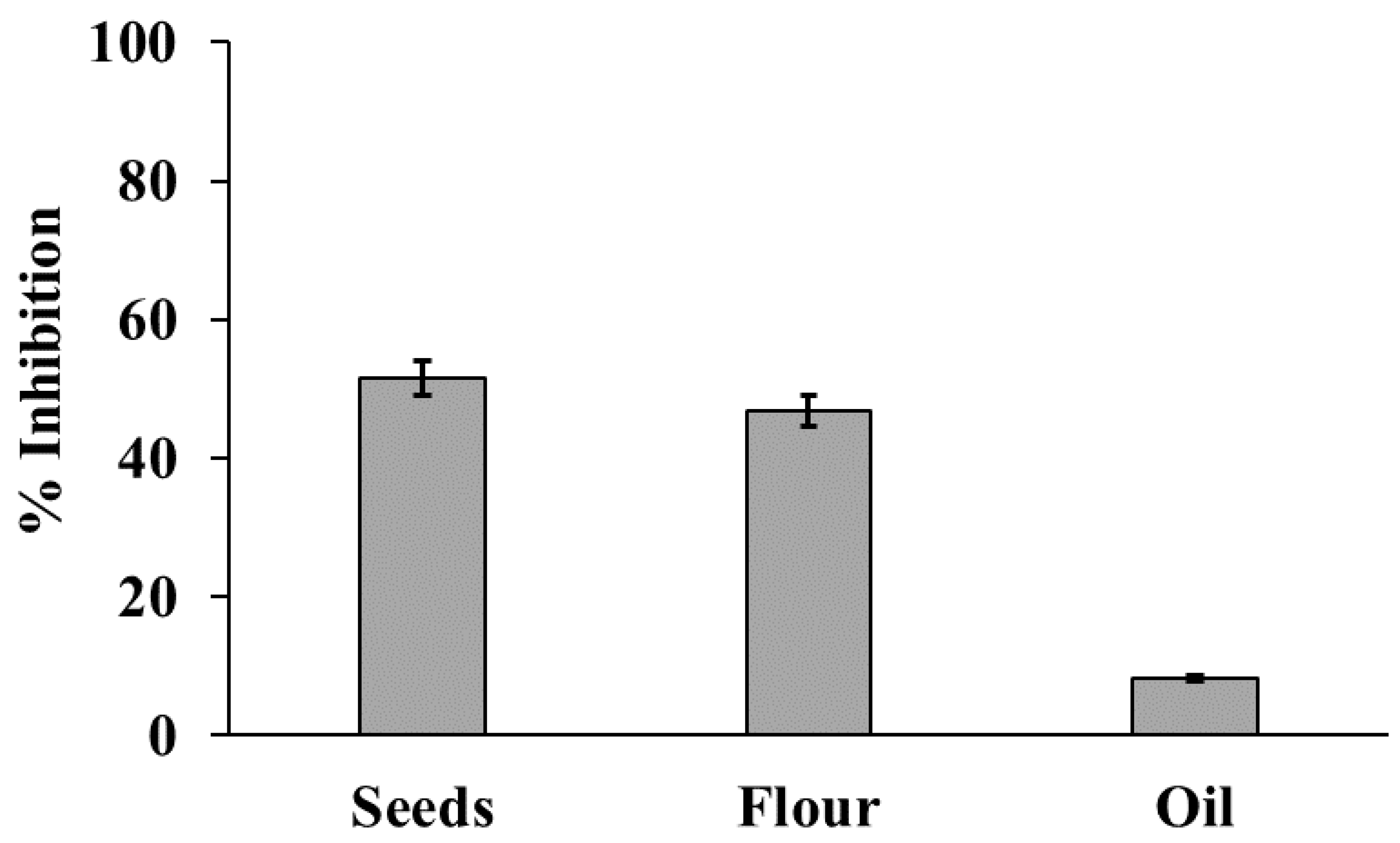
3.2. Total Polyphenolic Contents and Antioxidant Activity of Hemp Products
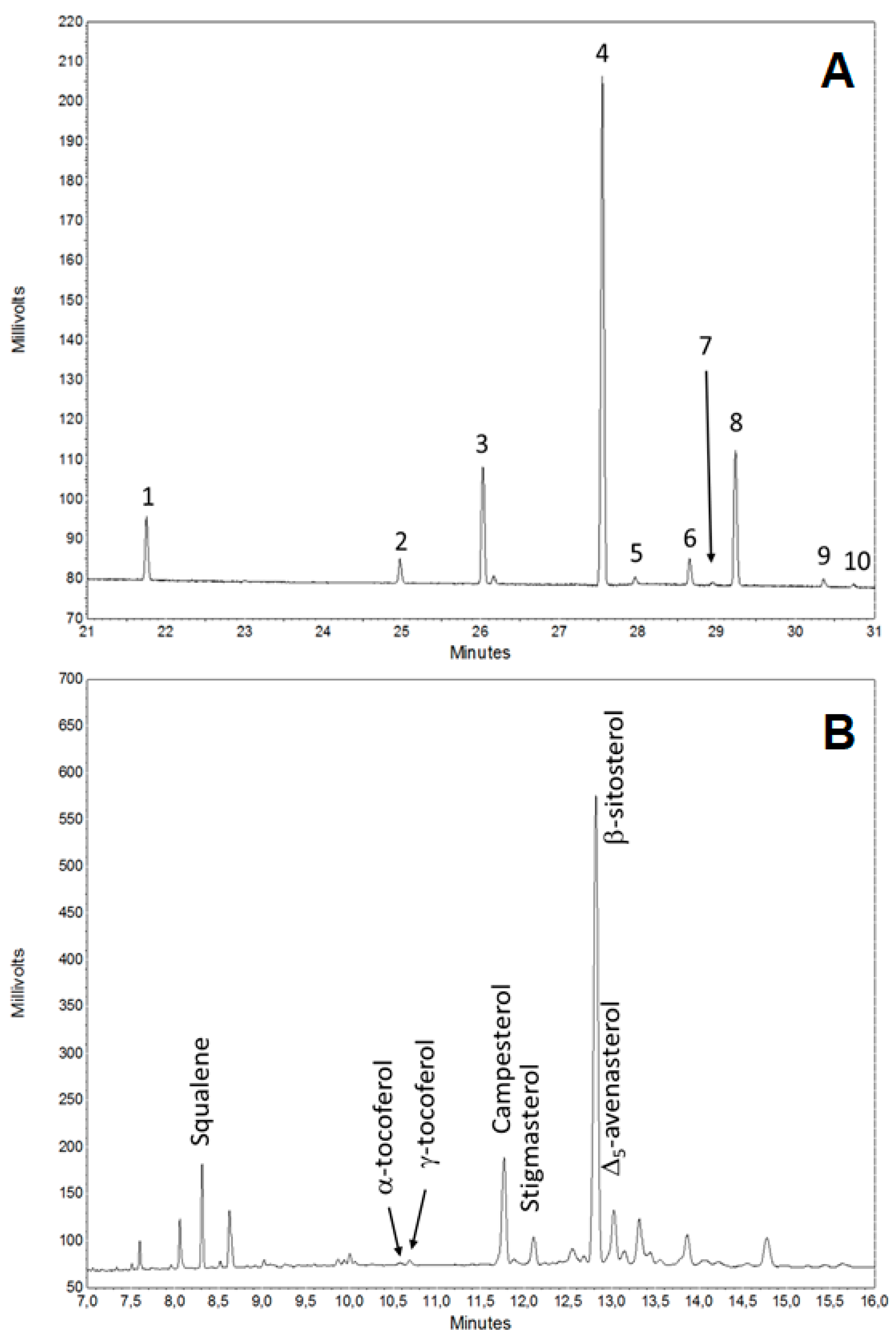
3.3. Saponificable and Unsaponificable Analysis
3.4. Minerals Content
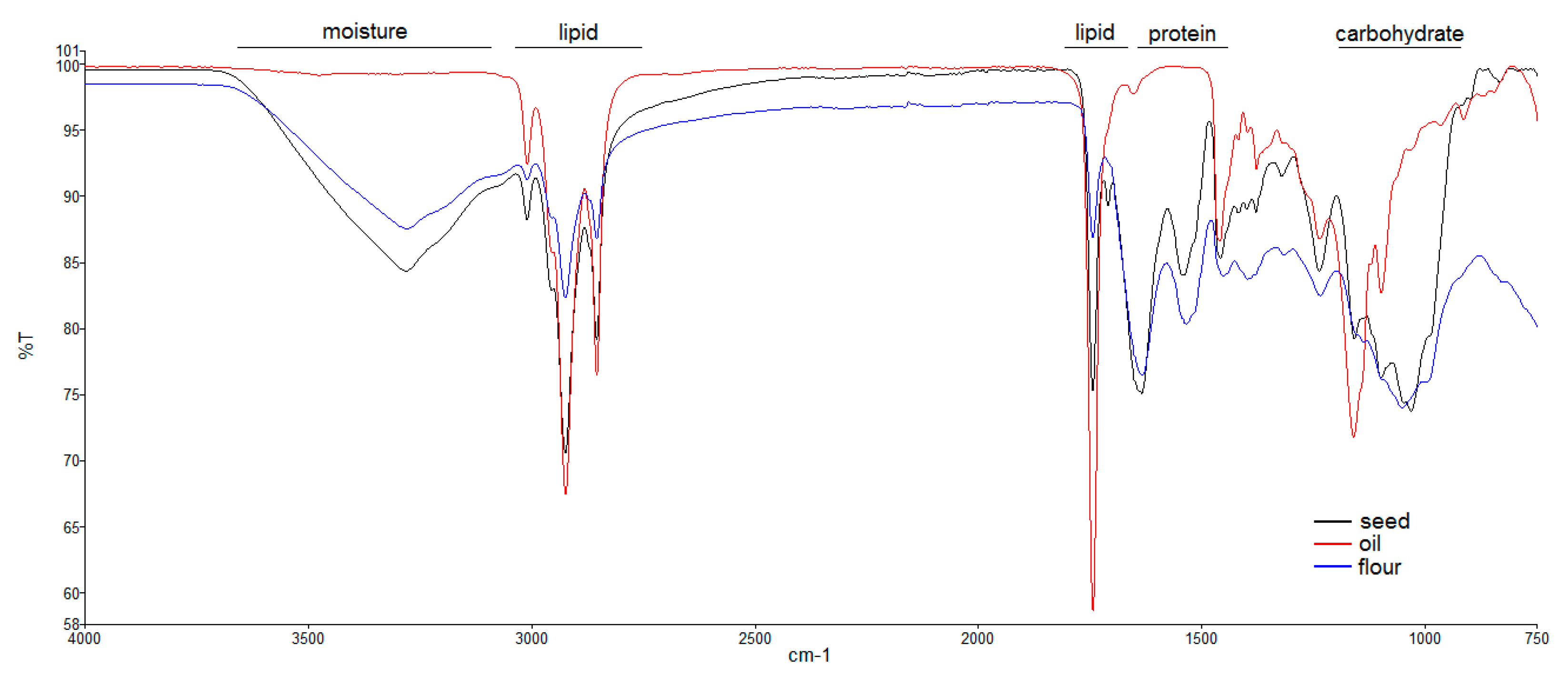
3.5. ATR-FTIR Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Callaway, J.C. Hempseed as a nutritional resource: An overview. Euphytica 2004, 140, 65–72. [Google Scholar] [CrossRef]
- Kriese, U.; Schumann, E.; Weber, W.E.; Beyer, M.; Bruhl, L.; Matthaus, B. Oil content, tocopherol composition and fatty acid patterns of the seeds of 51 Cannabis sativa L. genotypes. Euphytica 2004, 137, 339–351. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Evolutionary aspects of diet and essential fatty acids. In Fatty Acids and Lipids-New Findings; Hamazaki, T., Okuyama, H., Eds.; Karger: Basel, Switzerland, 2001; Volume 88, pp. 18–27. [Google Scholar]
- Kang, J.X. The importance of omega-6/omega-3 fatty acid ratio in cell function. The gene transfer of omega-3 fatty acid desaturase. In Omega-6/Omega-3 Essential Fatty Acid Ratio: The Scientific Evidence; Simopoulos, A.P., Cleland, L.G., Eds.; Karger: Basel, Switzerland, 2003; Volume 92, pp. 23–36. [Google Scholar]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The Plant of the Thousand and One Molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Bilotto, S.; Spagnuolo, C.; Russo, M.; Tedesco, I.; Laratta, B.; Russo, G.L. Dietary phytochemicals in chemoprevention of cancer: an update. Immunol. Endocr. Metab. Agents Med. Chem. 2013, 13, 2–24. [Google Scholar] [CrossRef]
- Smeriglio, A.; Galati, E.M.; Monforte, M.T.; Lanuzza, F.; D’Angelo, V.; Circosta, C. Polyphenolic Compounds and Antioxidant Activity of Cold-Pressed Seed Oil from Finola Cultivar of Cannabis sativa L. Phytother. Res. 2016, 30, 1298–1307. [Google Scholar] [CrossRef] [PubMed]
- Andre, C.M.; Larondelle, Y.; Evers, D. Dietary antioxidants and oxidative stress from a human and plant perspective: A review. Curr. Nutr. Food Sci. 2010, 6, 2–12. [Google Scholar] [CrossRef]
- Wang, C.; Kurzer, M.S. Effects of phytoestrogens on DNA synthesis in MCF-7 cells in the presence of estradiol or growth factors. Nutr. Cancer. 1998, 31, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Murthy, K.N.C.; Kim, J.; Vikram, A.; Patil, B.S. Differential inhibition of human colon cancer cells by structurally similar flavonoids of citrus. Food Chem. 2012, 132, 27–34. [Google Scholar] [CrossRef]
- Yan, X.; Tang, J.; dos Santos Passos, C.; Nurisso, A.; Simões-Pires, C.A.; Ji, M.; Lou, H.; Fan, P. Characterization of lignanamides from hemp (Cannabis sativa L.) seed and their antioxidant and acetylcholinesterase inhibitory activities. J. Agr. Food Chem. 2015, 63, 10611–10619. [Google Scholar] [CrossRef]
- Struik, P.C.; Amaducci, S.; Bullard, M.J.; Stutterheim, N.C.; Venturi, G.; Cromack, H.T.H. Agronomy of fibre hemp (Cannabis sativa L.) in Europe. Ind. Crop Prod. 2000, 11, 107–118. [Google Scholar] [CrossRef]
- Ranalli, P.; Venturi, G. Hemp as a raw material for industrial applications. Euphytica 2004, 140, 1–6. [Google Scholar] [CrossRef]
- Regulation (EC) N.1251/99 and Subsequent Amendments. Available online: https://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX%3A31999R1251 (accessed on 23 December 2018).
- AOAC INTERNATIONAL. Official Methods of Analysis, 20th ed.; Association of Official Agricultural Chemists: Rockville, MD, USA, 2016. [Google Scholar]
- Vonapartis, E.; Aubin, M.; Seguin, P.; Mustafa, A.F.; Charron, J.-B. Seed composition of ten industrial hemp cultivars approved fornproduction in Canada. J. Food Compost. Anal. 2015, 39, 8–12. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colourimetry of total phenolics with phosphomolybdic-phospholungistic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- IOC. Determination of Biophenols in Olive Oils by HPLC; COI/T.20/Doc.N. 29/1-8; IOC: Madrid, Spain, 2009. [Google Scholar]
- Ricciutelli, M.; Marconi, S.; Boarelli, M.C.; Caprioli, G.; Sagratini, G.; Ballini, R.; Fiorini, D. Olive oil polyphenols: A quantitative method by high-performanceliquid-chromatography-diode-array detection for their determination and theassessment of the related health claim. J. Chromatogr. A. 2017, 1481, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Chung, H.; Chang, P.-S.; Lee, J.H. Development of a method predicting the oxidative stability of edible oils using 2,2-diphenyl-1-picrylhydrazyl (DPPH). Food Chem. 2007, 103, 662–669. [Google Scholar] [CrossRef]
- Siano, F.; Addeo, F.; Volpe, M.G.; Paolucci, M.; Picariello, G. Oxidative Stability of Pomegranate (Punica granatum L.) Seed Oil to Simulated Gastric Conditions and Thermal Stress. J. Agric. Food Chem. 2016, 64, 8369–8378. [Google Scholar] [CrossRef]
- Caligiani, A.; Bonzanini, F.; Palla, G.; Cirlini, M.; Bruni, R. Characterization of a potential nutraceutical ingredient: Pomegranate (Punicagranatum L.) seed oil unsaponifiable fraction. Plant Foods Hum. Nutr. 2010, 65, 277–283. [Google Scholar] [CrossRef]
- Volpe, M.G.; Nazzaro, M.; Di Stasio, M.; Siano, F.; Coppola, R.; De Marco, A. Content of micronutrients, mineral and trace elements in some Mediterranean spontaneous edible herbs. Chem. Cent. J. 2015, 9, 57. [Google Scholar] [CrossRef]
- Folegatti, L.; Rovellini, P.; Baglio, D.; De Cesarei, S.; Fusari, P.; Venturini, S.; Cavalieri, A. Caratterizzazione chimica della farina ottenuta dopo la spremitura a freddo dei semi di Cannabis sativa L. Riv. Ital. Sostanze Gr. 2014, 91, 3–16. [Google Scholar]
- Choe, E.; Min, D.B. Mechanisms and Factors for Edible Oil Oxidation. Compr. Rev. Food Sci. Food Saf. 2006, 5, 169–186. [Google Scholar] [CrossRef]
- Pojić, M.; Mišan, A.; Sakač, M.; Hadnađev, T.D.; Šarić, B.; Milovanović, I.; Hadnađev, M. Characterization of Byproducts Originating from Hemp Oil Processing. J. Agric. Food Chem. 2014, 62, 12436–12442. [Google Scholar] [CrossRef] [PubMed]
- Aizpurua-Olaizola, O.; Omar, J.; Navarro, P.; Olivares, M.; Etxebarria, N.; Usobiaga, A. Identification and quantification of cannabinoids in Cannabis sativa L. plants by high performance liquid chromatography-mass spectrometry. Anal. Bioanal. Chem. 2014, 406, 7549–7560. [Google Scholar] [CrossRef] [PubMed]
- Da Porto, C.; Decorti, D.; Natolino, A. Potential Oil Yield, Fatty Acid Composition, and Oxidation Stability of the Hempseed Oil from Four Cannabis sativa L. Cultivars. J. Diet Suppl. 2015, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.G.; Song, Z.X.; Yin, H.; Wang, Y.Y.; Shu, G.F.; Lu, H.X.; Wang, S.K.; Sun, G.J. Low n-6/n-3 PUFA Ratio Improves Lipid Metabolism, Inflammation, Oxidative Stress and Endothelial Function in Rats Using Plant Oils as n-3 Fatty Acid Source. Lipids 2016, 51, 49–59. [Google Scholar] [CrossRef]
- Saeidnia, S.; Manayi, A.; Gohari, A.R.; Abdollahi, M. The Story of Beta-sitosterol- A Review. European J. Med. Plants. 2014, 4, 590–609. [Google Scholar] [CrossRef]
- Lees, A.M.; Mok, H.Y.I.; Lees, R.S.; McCluskey, M.A.; Grundy, S.M. Plant sterols as cholesterol-lowering agents: Clinical trials in patients with hypercholesterolemia and studies of sterol balance. Atherosclerosis 1977, 28, 325–338. [Google Scholar] [CrossRef]
- Rao, C.V.; Newmark, H.L.; Reddy, B.S. Chemopreventive effect of squalene on colon cancer. Carcinogenesis 1998, 19, 287–290. [Google Scholar] [CrossRef]
- Mihoc, M.; Pop, G.; Alexa, E.; Radulov, I. Nutritive quality of romanian hemp varieties (Cannabis sativa L.) with special focus on oil and metal contents of seeds. Chem. Cent. J. 2012, 6, 122–134. [Google Scholar] [CrossRef]
- Zerihun, A.; Chandravanshi, B.S.; Debebe, A.; Mehari, B. Levels of selected metals in leaves of Cannabis sativa L. cultivated in Ethiopia. SpringerPlus 2015, 4, 359. [Google Scholar] [CrossRef]
- Poisa, L.; Adamovics, A. Evaluate of hemp (Cannabis sativa L.) quality parameters for bioenergy production. Eng. Rural Dev. 2011, 26, 358–362. [Google Scholar]
- Korkmaz, K.; Kara, S.M.; Ozkutlu, F.; Gu, V. Monitoring of heavy metals and selected micronutrients in hempseeds from North-western Turkey. Afr. J. Agric. Res. 2010, 5, 463–467. [Google Scholar]
- Eboh, L.O.; Thomas, B.E. Analysis of Heavy Metal Content in Cannabis Leaf and seed Cultivated in Southern Part of Nigeria. Pak. J. Nutr. 2005, 4, 349–351. [Google Scholar]
- Official Journal of the European Union. Setting maximum levels for certain contaminants in foodstuffs. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=LEGISSUM%3Al21290 (accessed on 27 July 2018).
- Siano, F.; Sorrentino, G.; Riccardi, M.; De Cunzo, F.; Orefice, G.; Volpe, M.G. Chemical, nutritional, and spectroscopic characterization of typical ecotypes of Mediterranean area beans. Eur. Food Res. Technol. 2018, 244, 795–804. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
| Fedora cv | Moisture (%) | Protein (%) | Lipids (%) | Carbohydrates (%) | Ash (%) |
|---|---|---|---|---|---|
| Flour | 7.9 ± 0.9 | 30.7 ± 1.2 | 13.6 ± 1.9 | 41.6 ± 2.5 | 6.2 ± 0.5 |
| Seeds | 7.3 ± 0.8 | 24.8 ± 1.1 | 24.5 ± 2.0 | 38.1 ± 2.5 | 5.3 ± 0.6 |
| Flour Lipids | Cold Pressed Oil | Seed Lipids | ||
|---|---|---|---|---|
| N. | Component (Area %) | Fatty Acids | ||
| 1 | Palmitic, C16:0 | 7.35 ± 0.35 | 7.15 ± 0.42 | 7.03 ± 0.31 |
| 2 | Stearic, C18:0 | 2.62 ± 0.25 | 2.73 ± 0.21 | 2.78 ± 0.38 |
| 3 | Oleic, C18:1 ω-9c | 12.79 ± 1.14 | 12.75 ± 1.10 | 12.74 ± 1.25 |
| 4 | Linoleic, C18:2 ω-6c | 56.42 ± 3.98 | 56.08 ± 3.05 | 56.16 ± 3.45 |
| 5 | Arachidic, C20:0 | 0.74 ± 0.07 | 0.89 ± 0.05 | 0.81 ± 0.02 |
| 6 | γ-Linolenic, C18:3 ω-6 | 3.00 ± 0.40 | 3.03 ± 0.42 | 2.94 ± 0.37 |
| 7 | cis-11-Eicosenoic, C20:1 | 0.45 ± 0.05 | 0.26 ± 0.02 | 0.37 ± 0.07 |
| 8 | α-Linolenic, C18:3 ω-3 | 14.55 ± 1.47 | 14.89 ± 1.18 | 15.02 ± 1.12 |
| 9 | cis-11,14-Eicosadienoic, C20:2 | 0.82 ± 0.27 | 1.03 ± 0.32 | 0.99 ± 0.21 |
| 10 | Behenic, C22:0 | 0.29 ± 0.05 | 0.20 ± 0.06 | 0.27 ± 0.03 |
| Σ-SFA | 11.00 ± 0.42 | 10.97 ± 0.47 | 10.89 ± 0.49 | |
| Σ-MUFA | 13.24 ± 1.14 | 13.01 ± 1.10 | 13.11 ± 1.25 | |
| Σ-PUFA | 74.79 ± 4.27 | 75.03 ± 3.31 | 75.11 ± 3.65 | |
| Σ-PUFA/Σ-SFA | 6.80 | 6.84 | 6.90 | |
| Component (mg kg−1) | Unsaponificable Fraction | |||
| Squalene | 40.1 ± 6.1 | 43.5 ± 5.2 | 49.3 ± 5.6 | |
| α-tocoferol | 3.2 ± 0.7 | 2.7 ± 0.5 | 3.5 ± 0.9 | |
| γ-tocoferol | 5.8 ± 0.6 | 5.0 ± 0.8 | 5.5 ± 1.3 | |
| Brassicasterol | - | - | - | |
| Campesterol | 125.1 ± 8.2 | 117.4 ± 9.3 | 115.4 ± 10.3 | |
| Stigmasterol | 27.4 ± 2.8 | 28.2 ± 2.1 | 24.7 ± 2.5 | |
| β-sitosterol | 528.4 ± 28.8 | 530.4 ± 25.4 | 536.1 ± 31.5 | |
| Δ5-avenasterol | 69.6 ± 8.6 | 72.6 ± 6.6 | 76.2 ± 8.4 | |
| (mg kg−1) | Seed | Flour | Oil | (µg kg−1) | Seed | Flour | Oil |
|---|---|---|---|---|---|---|---|
| Na | 67.54 | 90.55 | 88.79 | Fe | 97.97 | 152.47 | 1.71 |
| Ca | 944.41 | 1907.20 | 53.86 | Cu | 5.02 | 11.94 | 1.48 |
| Mg | 2682.13 | 2310.54 | 199.07 | Zn | 48.43 | 54.68 | 0.91 |
| K | 2517.35 | 5064.45 | 20.72 | Mn | 44.42 | 94.71 | 0.82 |
| Ba | 2.04 | 4.08 | 0.43 | ||||
| Mo | 0.50 | 0.19 | 0.57 | ||||
| Co | 0.03 | 0.03 | 0.06 | ||||
| Al | 2.12 | 3.48 | 1.83 | ||||
| Ni | 0.79 | 0.85 | 0.03 | ||||
| Pb | 0.04 | 0.03 | 0.09 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siano, F.; Moccia, S.; Picariello, G.; Russo, G.L.; Sorrentino, G.; Di Stasio, M.; La Cara, F.; Volpe, M.G. Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.). Molecules 2019, 24, 83. https://doi.org/10.3390/molecules24010083
Siano F, Moccia S, Picariello G, Russo GL, Sorrentino G, Di Stasio M, La Cara F, Volpe MG. Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.). Molecules. 2019; 24(1):83. https://doi.org/10.3390/molecules24010083
Chicago/Turabian StyleSiano, Francesco, Stefania Moccia, Gianluca Picariello, Gian Luigi Russo, Giuseppe Sorrentino, Michele Di Stasio, Francesco La Cara, and Maria Grazia Volpe. 2019. "Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.)" Molecules 24, no. 1: 83. https://doi.org/10.3390/molecules24010083
APA StyleSiano, F., Moccia, S., Picariello, G., Russo, G. L., Sorrentino, G., Di Stasio, M., La Cara, F., & Volpe, M. G. (2019). Comparative Study of Chemical, Biochemical Characteristic and ATR-FTIR Analysis of Seeds, Oil and Flour of the Edible Fedora Cultivar Hemp (Cannabis sativa L.). Molecules, 24(1), 83. https://doi.org/10.3390/molecules24010083










