A Novel Series of Acylhydrazones as Potential Anti-Candida Agents: Design, Synthesis, Biological Evaluation and In Silico Studies
Abstract
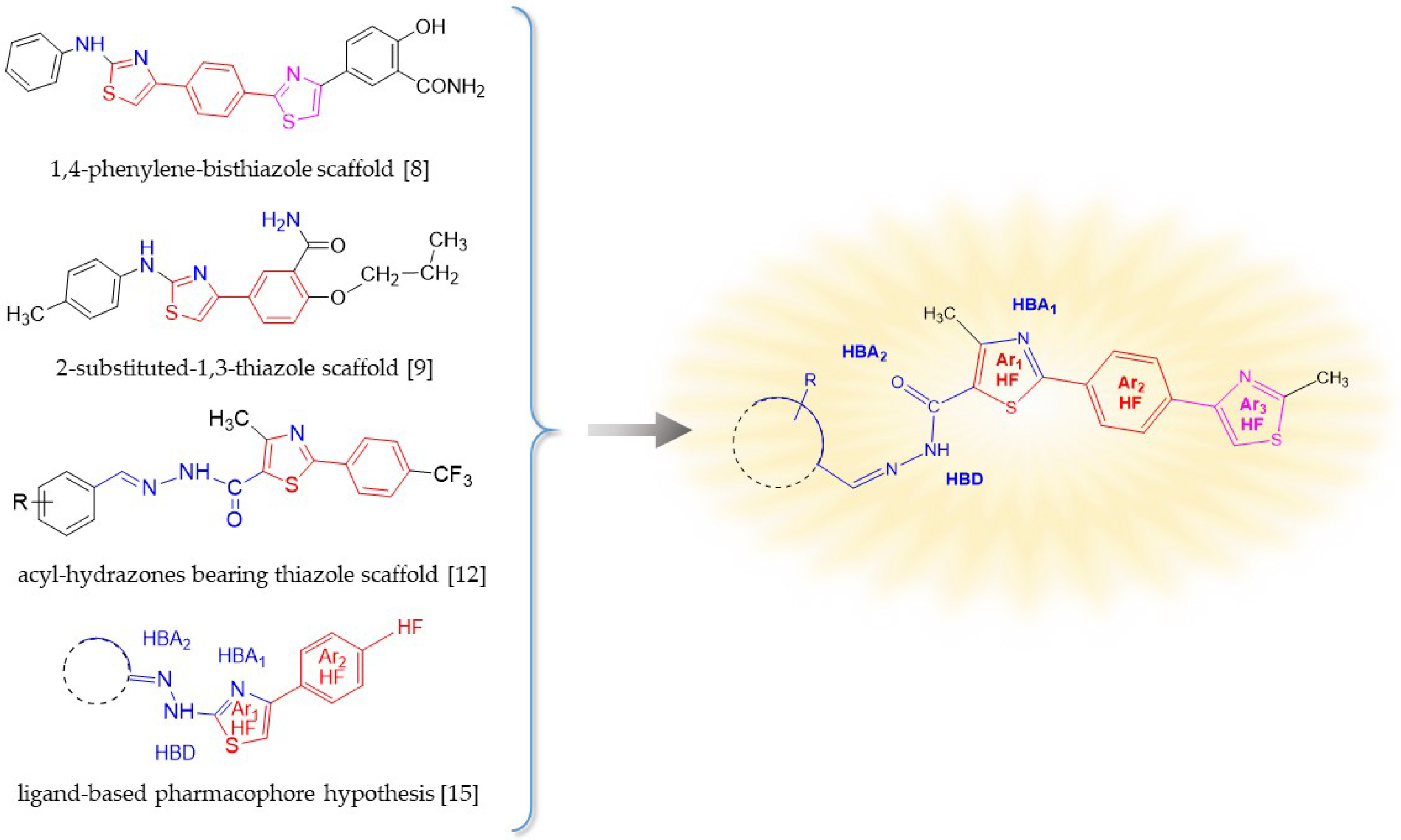
1. Introduction
2. Results and Discussion
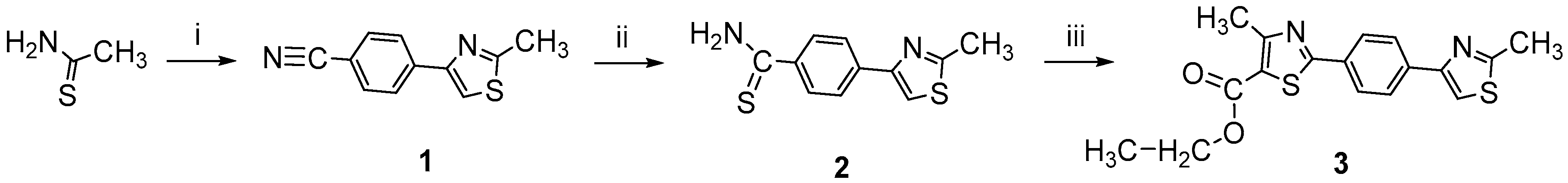
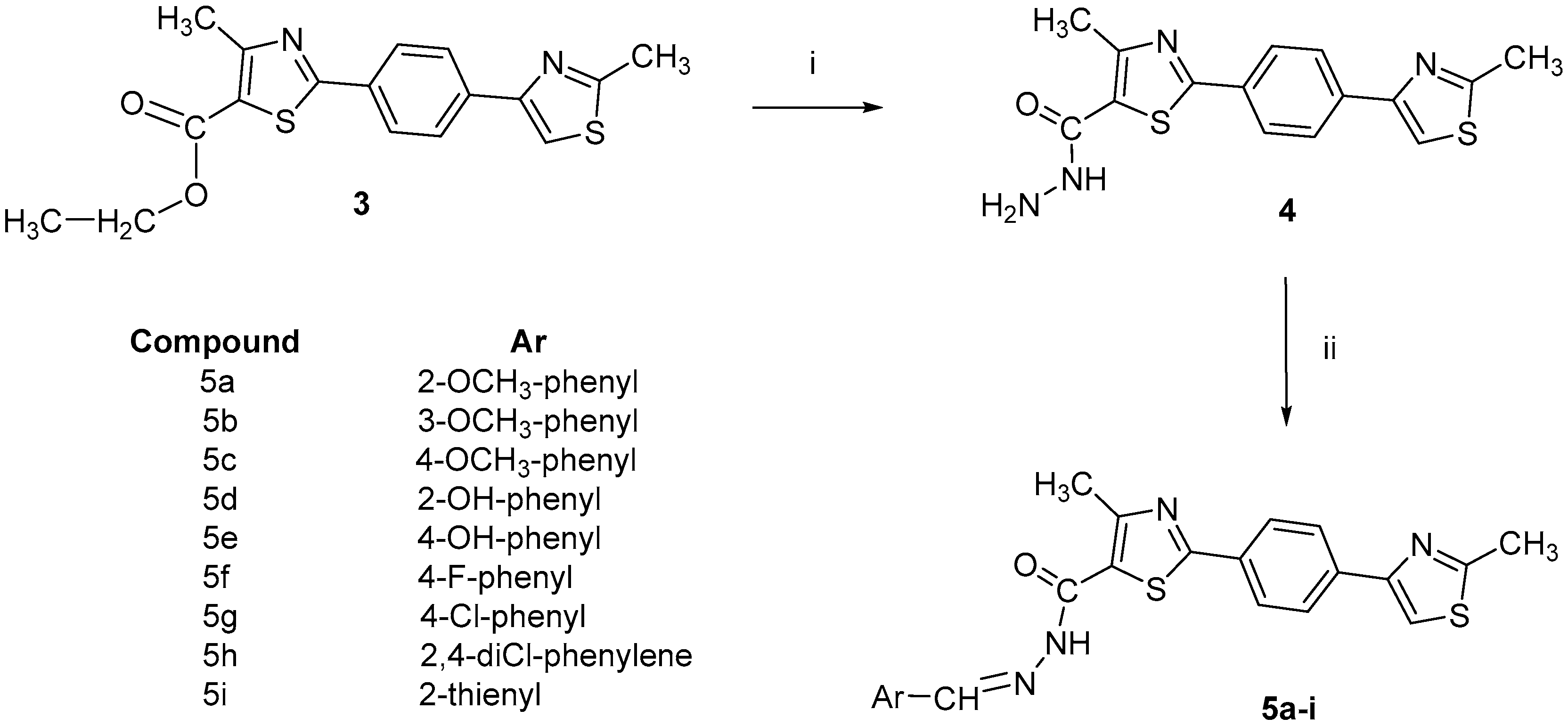
2.1. Chemistry
2.2. Anti-Candida Activity Assay
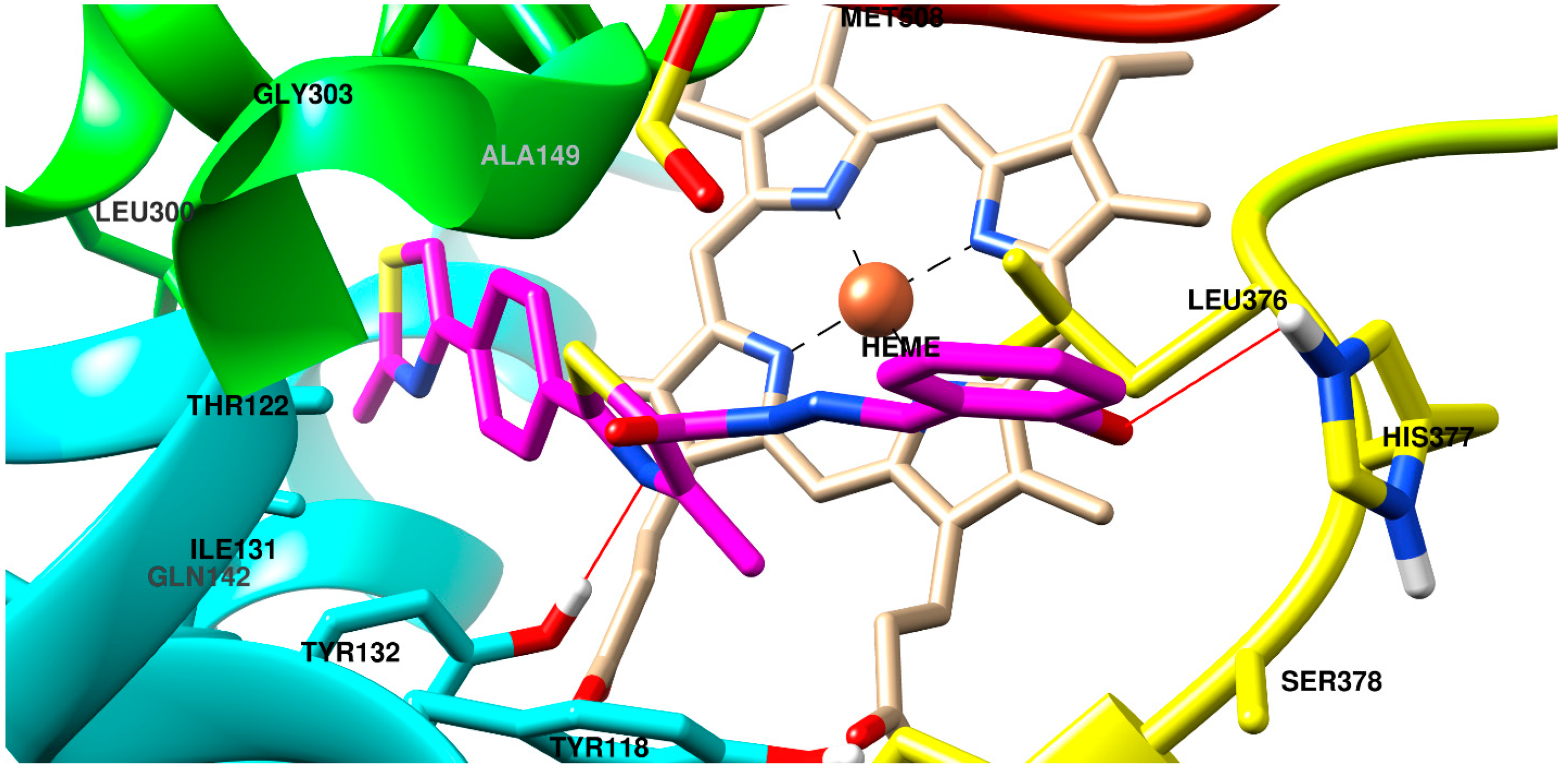
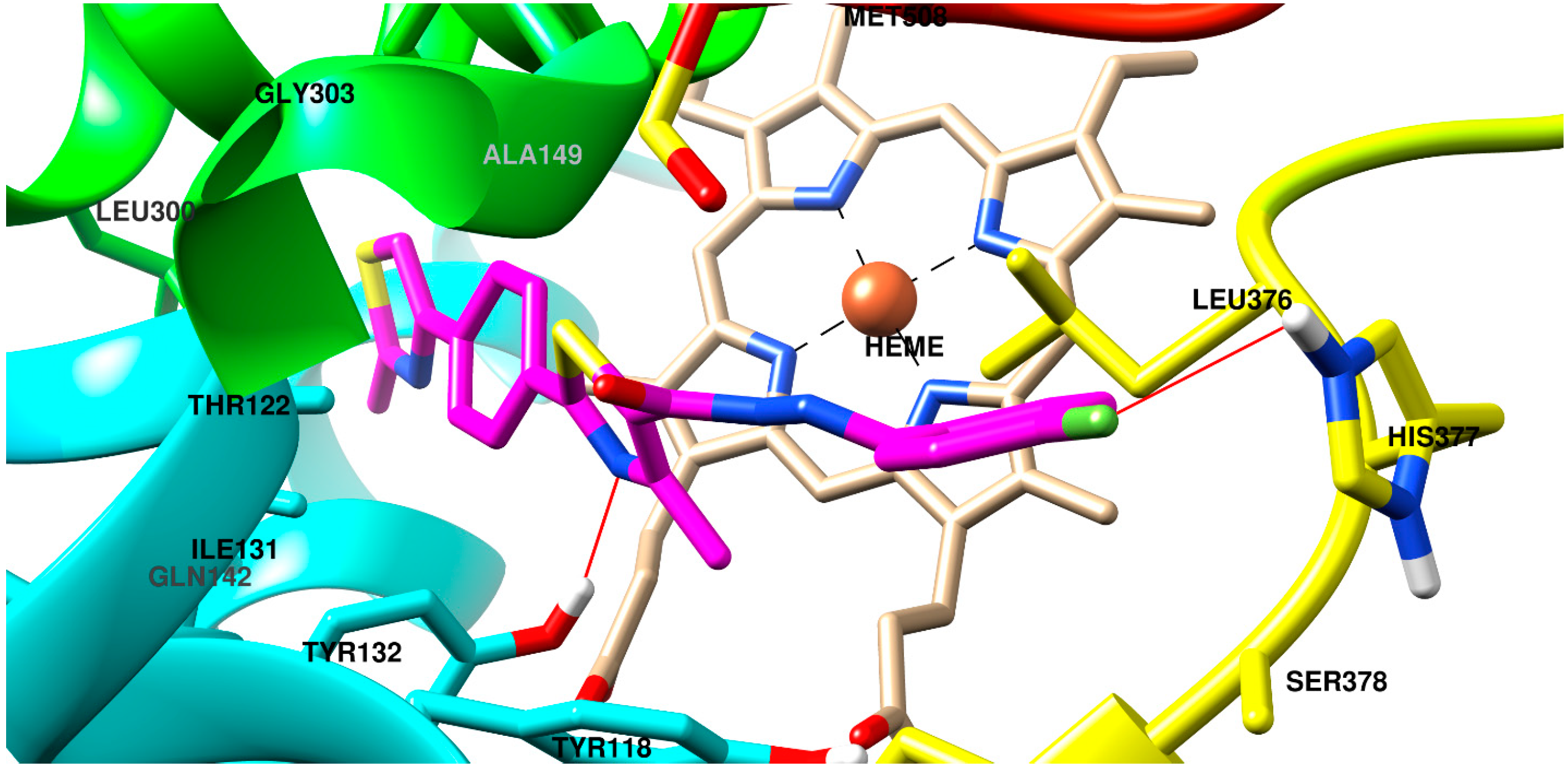
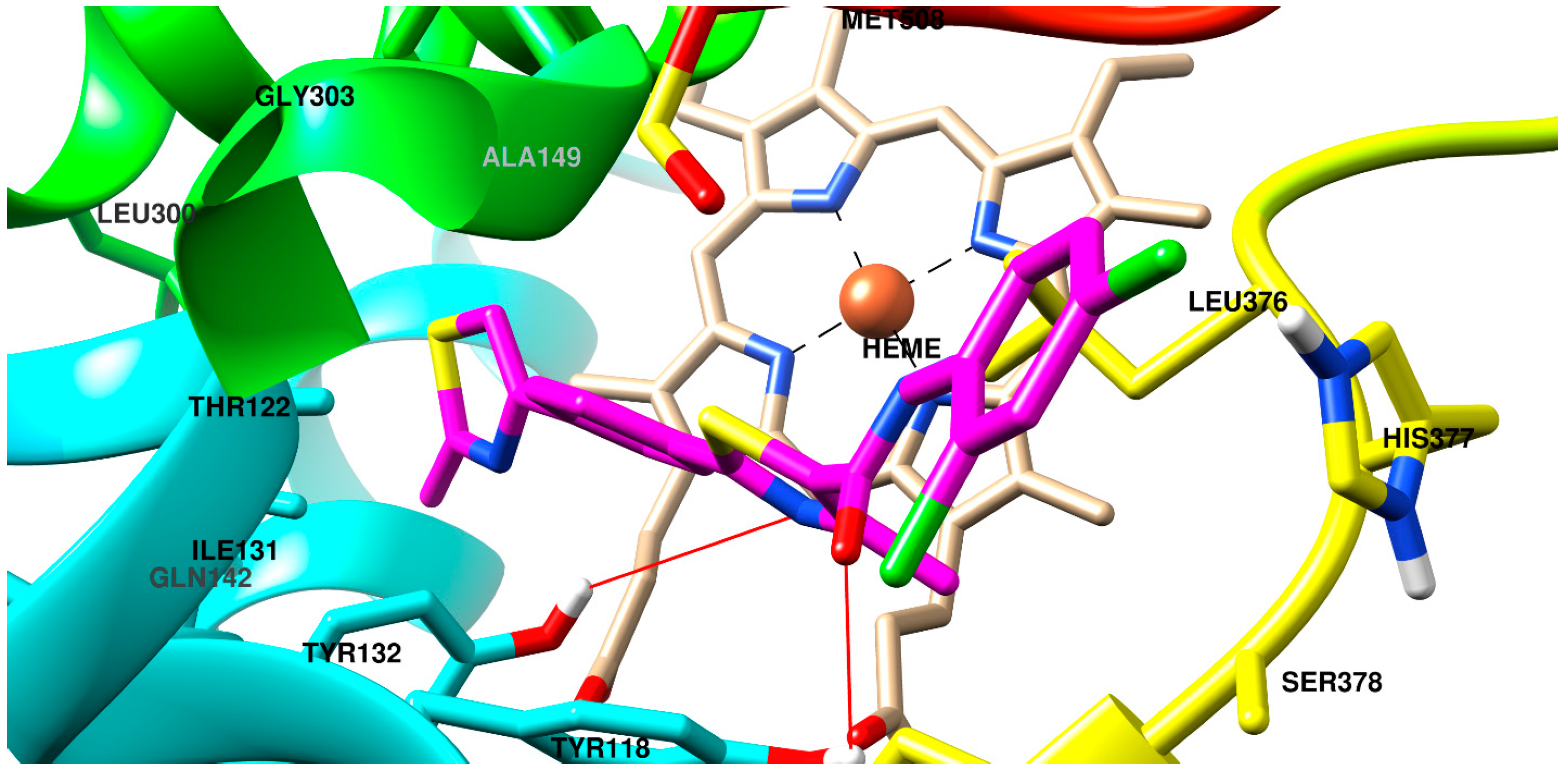
2.3. Molecular Docking
2.4. In Silico ADMET Evaluation
3. Materials and Methods
3.1. General Information
3.2. Chemistry
3.2.1. Synthesis of 4-methyl-2-(4-(2-methylthiazol-4-yl)phenyl)thiazole-5-carbohydrazide (4)
3.2.2. General Procedure for the Synthesis of 1,4-phenylene-bisthiazole acyl-hydrazones 5a–i
3.3. Anti-Candida Activity Assay
3.4. Molecular Docking
3.5. In Silico ADMET Evaluation
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lai, C.C.; Wang, C.Y.; Liu, W.L.; Huang, Y.T.; Hsueh, P.R. Time to positivity of blood cultures of different Candida species causing fungaemia. J. Med. Microbiol. 2012, 61, 701–704. [Google Scholar] [CrossRef] [PubMed]
- Sardi, J.C.O.; Scorzoni, L.; Bernardi, T.; Fusco-Almeida, A.M.; Mendes Giannini, M.J.S. Candida species: Current epidemiology, pathogenicity, biofilm formation, natural antifungal products and new therapeutic options. J. Med. Microbiol. 2013, 62, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Scorzoni, L.; de Lucas, M.P.; Mesa-Arango, A.C.; Fusco-Almeida, A.M.; Lozano, E.; Cuenca-Estrella, M.; Mendes-Giannini, M.J.; Zaragoza, O. Antifungal efficacy during Candida krusei infection in non-conventional models correlates with the yeast in vitro susceptibility profile. PLoS ONE 2013, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Trofa, D.; Gácser, A.; Nosanchuk, J.D. Candida parapsilosis, an emerging fungal pathogen. Clin. Microbiol. Rev. 2008, 21, 606–625. [Google Scholar] [CrossRef] [PubMed]
- Whaley, S.G.; Berkow, E.L.; Rybak, J.M.; Nishimoto, A.T.; Barker, K.S.; Rogers, P.D. Azole antifungal resistance in Candida albicans and emerging non-albicans Candida species. Front. Microbiol. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.E.; Warrilow, A.G.S.; Price, C.L.; Mullins, J.G.L.; Kelly, D.E.; Kelly, S.L. Resistance to antifungals that target CYP51. J. Chem. Biol. 2014, 7, 143–161. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, F.; Bizzarri, B.; Bolasco, A.; Secci, D.; Chimenti, P.; Granese, A.; Carradori, S.; D’Ascenzio, M.; Lilli, D.; Rivanera, D. Synthesis and biological evaluation of novel 2,4-disubstituted-1,3-thiazoles as anti-Candida spp. agents. Eur. J. Med. Chem. 2011, 46, 378–382. [Google Scholar] [CrossRef]
- Bikobo, D.S.N.; Vodnar, D.C.; Stana, A.; Tiperciuc, B.; Nastasă, C.; Douchet, M.; Oniga, O. Synthesis of 2-phenylamino-thiazole derivatives as antimicrobial agents. J. Saudi Chem. Soc. 2017, 21, 861–868. [Google Scholar] [CrossRef]
- Narayana, B.; Vijaya Raj, K.K.; Ashalatha, B.V.; Kumari, N.S.; Sarojini, B.K. Synthesis of some new 5-(2-substituted-1,3-thiazol-5-yl)-2-hydroxy benzamides and their 2-alkoxy derivatives as possible antifungal agents. Eur. J. Med. Chem. 2004, 39, 867–872. [Google Scholar] [CrossRef]
- Maillard, L.T.; Bertout, S.; Quinonéro, O.; Akalin, G.; Turan-Zitouni, G.; Fulcrand, P.; Demirci, F.; Martinez, J.; Masurier, N. Synthesis and anti-Candida activity of novel 2-hydrazino-1,3-thiazole derivatives. Bioorg. Med. Chem. Lett. 2013, 23, 1803–1807. [Google Scholar] [CrossRef]
- Kamal, R.; Kumar, V.; Bhardwaj, V.; Kumar, V.; Aneja, K.R. Synthesis, characterization and in vitro antimicrobial evaluation of some novel hydrazone derivatives bearing pyrimidinyl and pyrazolyl moieties as a promising heterocycles. Med. Chem. Res. 2015, 24, 2551–2560. [Google Scholar] [CrossRef]
- Nastasă, C.; Tiperciuc, B.; Duma, M.; Benedec, D.; Oniga, O. New hydrazones bearing thiazole scaffold: Synthesis, characterization, antimicrobial, and antioxidant investigation. Molecules 2015, 20, 17325–17338. [Google Scholar] [CrossRef] [PubMed]
- Kaplancikli, Z.A.; Levent, S.; Osmaniye, D.; Saǧlik, B.N.; Çevik, U.A.; Çavuşoǧlu, B.K.; Özkay, Y.; Ilgin, S. Synthesis and anticandidal activity evaluation of new benzimidazole-thiazole derivatives. Molecules 2017, 22, 2051. [Google Scholar] [CrossRef] [PubMed]
- Kauthale, S.; Tekale, S.; Damale, M.; Sangshetti, J.; Pawar, R. Synthesis, biological evaluation, molecular docking, and ADMET studies of some isoxazole-based amides. Med. Chem. Res. 2018, 27, 429–441. [Google Scholar] [CrossRef]
- Gidaro, M.C.; Alcaro, S.; Secci, D.; Rivanera, D.; Mollica, A.; Agamennone, M.; Giampietro, L.; Carradori, S. Identification of new anti-Candida compounds by ligand-based pharmacophore virtual screening. J. Enzym. Inhib. Med. Chem. 2016, 31, 1703–1706. [Google Scholar] [CrossRef] [PubMed]
- Geerts, T.; Vander Heyden, Y. In silico predictions of ADME-Tox properties: Drug absorption. Comb. Chem. High Throughput Screen. 2011, 14, 339–361. [Google Scholar] [CrossRef] [PubMed]
- Borcea, A.-M.; Marc, G.; Pîrnău, A.; Vlase, L.; Ionuț, I.; Tiperciuc, B.; Oniga, O. Synthesis and molecular docking study of some new 1,4-phenylene-bisthiazoles as fungal lanosterol 14α-demethylase inhibitors. Farmacia 2017, 65, 683–689. [Google Scholar]
- Secci, D.; Bizzarri, B.; Bolasco, A.; Carradori, S.; D’Ascenzio, M.; Rivanera, D.; Mari, E.; Polletta, L.; Zicari, A. Synthesis, anti-Candida activity, and cytotoxicity of new (4-(4-iodophenyl)thiazol-2-yl)hydrazine derivatives. Eur. J. Med. Chem. 2012, 53, 246–253. [Google Scholar] [CrossRef]
- Stana, A.; Enache, A.; Vodnar, D.C.; Nastasă, C.; Benedec, D.; Ionuț, I.; Login, C.; Marc, G.; Oniga, O.; Tiperciuc, B. New thiazolyl-triazole Schiff bases: Synthesis and evaluation of the anti-Candida potential. Molecules 2016, 21, 1595. [Google Scholar] [CrossRef]
- Borcea, A.-M.; Marc, G.; Vodnar, D.C.; Vlase, L.; Oniga, O. Synthesis of novel thiazolyl-phenyl-thiazole derivatives as promising anti-Candida agents. Pak. J. Pharm. Sci. 2018, 31, 2085–2090. [Google Scholar]
- Meletiadis, J.; Antachopoulos, C.; Stergiopoulou, T.; Pournaras, S.; Roilides, E.; Walsh, T.J. Differential fungicidal activities of amphotericin B and voriconazole against Aspergillus species determined by microbroth methodology. Antimicrob. Agents Chemother. 2007, 51, 3329–3337. [Google Scholar] [CrossRef] [PubMed]
- Marc, G.; Ionuț, I.; Pîrnǎu, A.; Vlase, L.; Vodnar, D.C.; Duma, M.; Tiperciuc, B.; Oniga, O. Microwave assisted synthesis of 3,5-disubstituted tiazolidine-2,4-diones with antifungal activity. Design, synthesis, virtual and in vitro antifungal screening. Farmacia 2017, 65, 414–422. [Google Scholar]
- Villaseñor-Granados, T.; García, S.; Vazquez, M.A.; Robles, J. Molecular docking-based screening of newly designed coumarin derivatives with potential antifungal activity against lanosterol 14α-demethylase. Theor. Chem. Acc. 2016, 135, 1–16. [Google Scholar] [CrossRef]
- Almi, Z.; Belaidi, S.; Lanez, T.; Tchouar, N. Structure activity relationships, QSAR modeling and drug-like calculations of TP inhibition of 1,3,4-oxadiazoline-2-thione derivatives. Int. Lett. Chem. Phys. Astron. 2013, 37, 113–124. [Google Scholar] [CrossRef]
- Wang, J.; Urban, L. The impact of early ADME profiling on drug discovery and development strategy. Drug Discov. World 2004, 73–86. [Google Scholar]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
- Ertl, P.; Rohde, B.; Selzer, P. Fast calculation of molecular polar surface area as a sum of fragment-based contributions and its application to the prediction of drug transport properties. J. Med. Chem. 2000, 43, 3714–3717. [Google Scholar] [CrossRef]
- Zhou, S.-F. Structure, function and regulation of P-glycoprotein and its clinical relevance in drug disposition. Xenobiotica 2008, 38, 802–832. [Google Scholar] [CrossRef]
- Ghabbour, H.A.; Qabeel, M.M.; Eldehna, W.M.; Al-Dhfyan, A.; Abdel-Aziz, H.A. Design, synthesis, and molecular docking of 1-(1-(4-chlorophenyl)-2-(phenylsulfonyl)ethylidene)-2-phenylhydrazine as potent nonazole anticandidal agent. J. Chem. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Alexander, B.D.; Procop, G.W.; Dufresne, P.; Fuller, J.; Ghannoum, M.A.; Hanson, K.E.; Holliday, D.; Holliday, N.M.; Kovanda, L.; Lockhart, S.R.; et al. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, 4th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017. [Google Scholar]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Belew, R.K.; Goodsell, D.S.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef]
- Marc, G.; Stana, A.; Pîrnău, A.; Vlase, L.; Vodnar, D.C.; Duma, M.; Tiperciuc, B.; Oniga, O. 3,5-Disubstituted thiazolidine-2,4-diones: Design, microwave-assisted synthesis, antifungal activity, and ADMET screening. SLAS Discov. 2018, 23, 807–814. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Daina, A.; Zoete, V. A BOILED-Egg to predict gastrointestinal absorption and brain penetration of small molecules. ChemMedChem 2016, 11, 1117–1121. [Google Scholar] [CrossRef]
- Zhao, Y.H.; Abraham, M.H.; Le, J.; Hersey, A.; Luscombe, C.N.; Beck, G.; Sherborne, B.; Cooper, I. Rate-limited steps of human oral absorption and QSAR studies. Pharm. Res. 2002, 19, 1446–1457. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 5a–i are available from the authors. |
| Compound | C. albicans ATCC 10231 | C. albicans ATCC 18804 | C. krusei ATCC 6258 | C. parapsilosis ATCC 22019 |
|---|---|---|---|---|
| 5a | 31.25 | 31.25 | 62.5 | 62.5 |
| 5b | 31.25 | 31.25 | 62.5 | 62.5 |
| 5c | 31.25 | 31.25 | 31.25 | 31.25 |
| 5d | 15.62 | 15.62 | 15.62 | 15.62 |
| 5e | 31.25 | 31.25 | 31.25 | 31.25 |
| 5f | 15.62 | 15.62 | 15.62 | 15.62 |
| 5g | 31.25 | 31.25 | 15.62 | 7.81 |
| 5h | 31.25 | 31.25 | 15.62 | 15.62 |
| 5i | 31.25 | 31.25 | 31.25 | 31.25 |
| Fluconazole | 15.62 | 15.62 | 15.62 | 7.81 |
| Drug-free | Growth in all concentrations | |||
| Broth control | No growth | |||
| Compound | C. albicans ATCC 10231 | C. albicans ATCC 18804 | C. krusei ATCC 6258 | C. parapsilosis ATCC 22019 |
|---|---|---|---|---|
| 5a | 62.5 | 62.5 | 125 | 125 |
| 5b | 62.5 | 62.5 | 125 | 125 |
| 5c | 62.5 | 62.5 | 62.5 | 62.5 |
| 5d | 31.25 | 31.25 | 31.25 | 31.25 |
| 5e | 62.5 | 62.5 | 62.5 | 62.5 |
| 5f | 31.25 | 31.25 | 31.25 | 31.25 |
| 5g | 62.5 | 62.5 | 31.25 | 15.62 |
| 5h | 62.5 | 62.5 | 31.25 | 31.25 |
| 5i | 62.5 | 62.5 | 62.5 | 62.5 |
| Fluconazole | 31.25 | 31.25 | 31.25 | 15.62 |
| Drug-free | Growth in all concentrations | |||
| Broth control | No growth | |||
| Compound | Best Binding Conformation | 2Å Cluster of the Best Binding Conformation | Number of Other Clusters | ||||
|---|---|---|---|---|---|---|---|
| ΔG (kcal/mol) | Ki (nM) | NoC 1 | Average ΔG (kcal/mol) | Standard Deviation | |||
| ΔG (kcal/mol) | RMSD 2 of Cartesian Coordinates | ||||||
| 5a | −11.86 | 2.03 | 74 | −11.68 | 0.39 | 0.44 | 8 |
| 5b | −11.92 | 1.83 | 66 | −11.82 | 0.38 | 0.43 | 5 |
| 5c | −11.87 | 1.99 | 65 | −11.66 | 0.24 | 0.56 | 6 |
| 5d | −12.16 | 1.22 | 81 | −11.91 | 0.30 | 0.39 | 4 |
| 5e | −11.97 | 1.68 | 70 | −11.89 | 0.30 | 0.48 | 7 |
| 5f | −11.46 | 3.98 | 77 | −11.29 | 0.14 | 0.25 | 3 |
| 5g | −11.98 | 1.65 | 68 | −11.82 | 0.32 | 0.48 | 3 |
| 5h | −12.36 | 0.87 | 76 | −12.17 | 0.26 | 0.31 | 6 |
| 5i | −11.85 | 2.06 | 65 | −11.52 | 0.36 | 0.45 | 8 |
| Compound | MW 1 (Da) | Rob 2 | HBA 3 | HBD 4 | MR 5 | TPSA 6 | mLogP 7 | Lipinski Violations |
|---|---|---|---|---|---|---|---|---|
| 5a | 448.56 | 7 | 5 | 1 | 126.25 | 132.95 | 2.25 | 0 |
| 5b | 448.56 | 7 | 5 | 1 | 126.25 | 132.95 | 2.25 | 0 |
| 5c | 448.56 | 7 | 5 | 1 | 126.25 | 132.95 | 2.25 | 0 |
| 5d | 434.53 | 6 | 5 | 2 | 121.78 | 143.95 | 2.04 | 0 |
| 5e | 434.53 | 6 | 5 | 2 | 121.78 | 143.95 | 2.04 | 0 |
| 5f | 436.52 | 6 | 5 | 1 | 119.72 | 123.72 | 2.96 | 0 |
| 5g | 452.98 | 6 | 4 | 1 | 124.77 | 123.72 | 3.06 | 0 |
| 5h | 487.42 | 6 | 4 | 1 | 129.78 | 123.72 | 3.54 | 0 |
| 5i | 424.56 | 6 | 4 | 1 | 117.64 | 151.96 | 2.17 | 0 |
| Compound | logS 1 | %Abs 2 | BBBP 3 | Pgp 4 Substrate | CYP2D6 Inhibitor |
|---|---|---|---|---|---|
| 5a | −7.86 | 63.13 | No | No | No |
| 5b | −7.86 | 63.13 | No | No | No |
| 5c | −7.86 | 63.13 | No | No | No |
| 5d | −7.76 | 59.33 | No | No | No |
| 5e | −7.76 | 59.33 | No | No | No |
| 5f | −7.8 | 66.31 | No | No | No |
| 5g | −8.35 | 66.31 | No | No | No |
| 5h | −9.01 | 66.31 | No | No | No |
| 5i | −8.31 | 56.57 | No | No | No |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borcea, A.-M.; Marc, G.; Ionuț, I.; Vodnar, D.C.; Vlase, L.; Gligor, F.; Pricopie, A.; Pîrnău, A.; Tiperciuc, B.; Oniga, O. A Novel Series of Acylhydrazones as Potential Anti-Candida Agents: Design, Synthesis, Biological Evaluation and In Silico Studies. Molecules 2019, 24, 184. https://doi.org/10.3390/molecules24010184
Borcea A-M, Marc G, Ionuț I, Vodnar DC, Vlase L, Gligor F, Pricopie A, Pîrnău A, Tiperciuc B, Oniga O. A Novel Series of Acylhydrazones as Potential Anti-Candida Agents: Design, Synthesis, Biological Evaluation and In Silico Studies. Molecules. 2019; 24(1):184. https://doi.org/10.3390/molecules24010184
Chicago/Turabian StyleBorcea, Anca-Maria, Gabriel Marc, Ioana Ionuț, Dan C. Vodnar, Laurian Vlase, Felicia Gligor, Andreea Pricopie, Adrian Pîrnău, Brîndușa Tiperciuc, and Ovidiu Oniga. 2019. "A Novel Series of Acylhydrazones as Potential Anti-Candida Agents: Design, Synthesis, Biological Evaluation and In Silico Studies" Molecules 24, no. 1: 184. https://doi.org/10.3390/molecules24010184
APA StyleBorcea, A.-M., Marc, G., Ionuț, I., Vodnar, D. C., Vlase, L., Gligor, F., Pricopie, A., Pîrnău, A., Tiperciuc, B., & Oniga, O. (2019). A Novel Series of Acylhydrazones as Potential Anti-Candida Agents: Design, Synthesis, Biological Evaluation and In Silico Studies. Molecules, 24(1), 184. https://doi.org/10.3390/molecules24010184










