Hepatoprotective Effect of Baicalein Against Acetaminophen-Induced Acute Liver Injury in Mice
Abstract
1. Introduction
2. Result
2.1. Effects of BAI on Body Weight and Organ Index in Mice
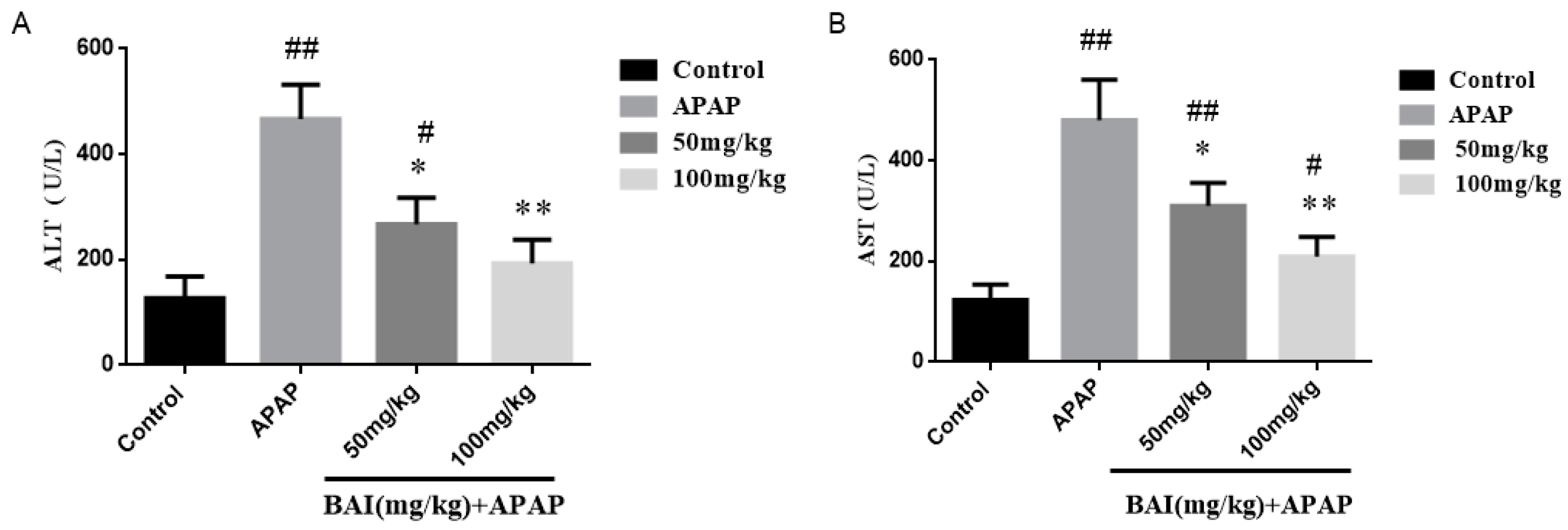
2.2. Effects of Baicalein on APAP-Induced Liver Injury
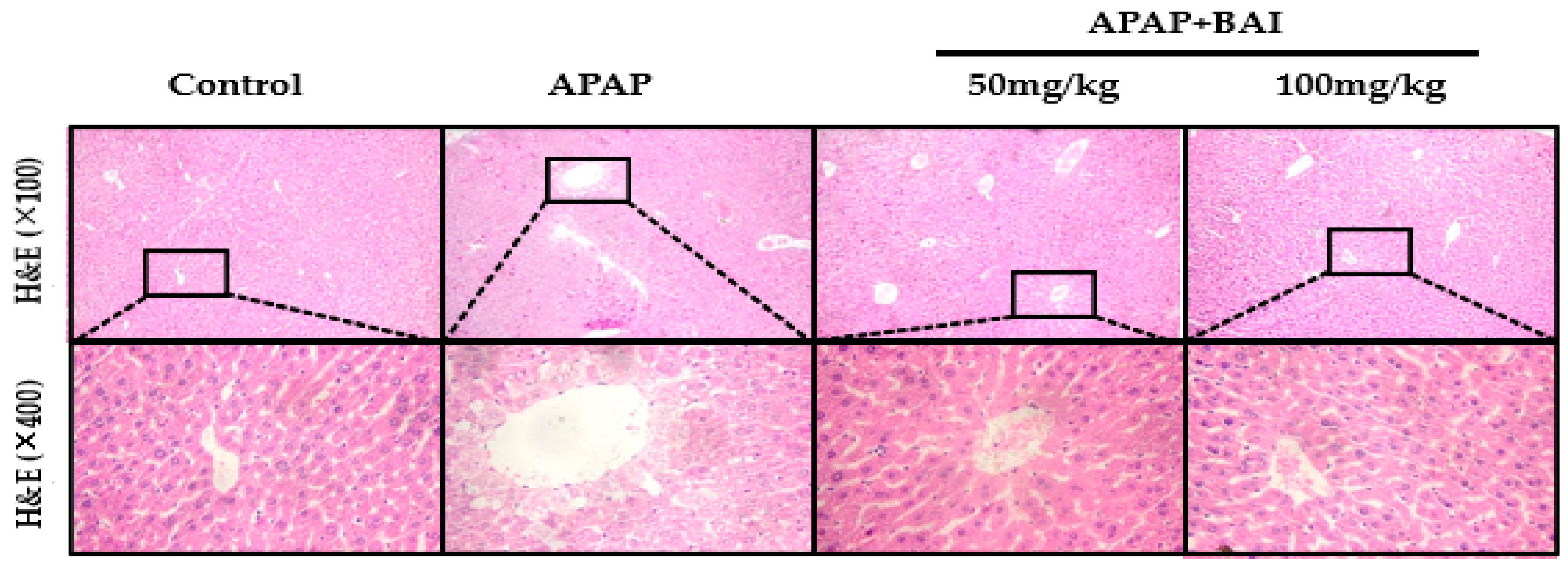
2.3. Effect of Baicalein on Histopathologic Changes
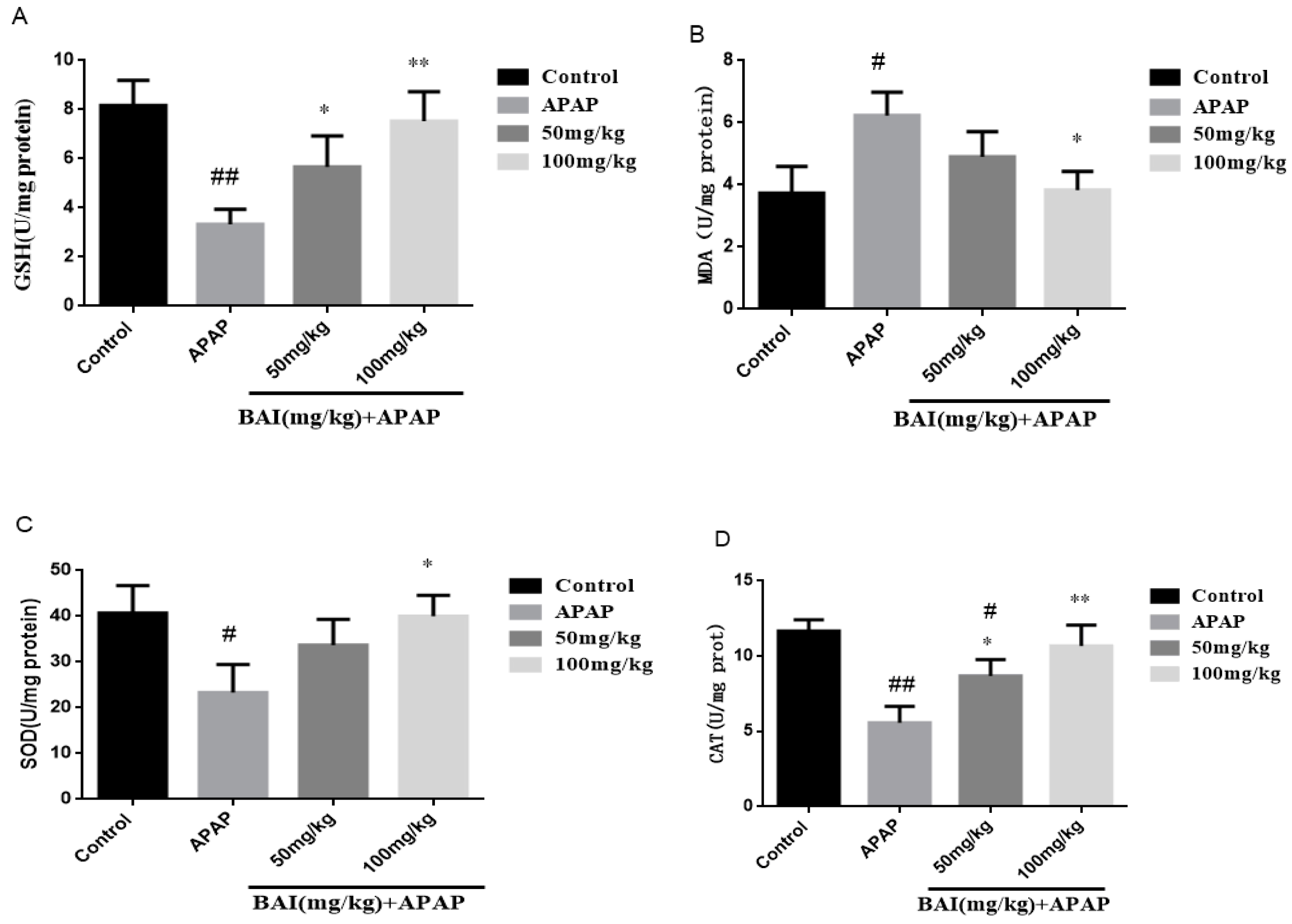
2.4. Effects of Baicalein on APAP-Induced Liver Oxidative Stress
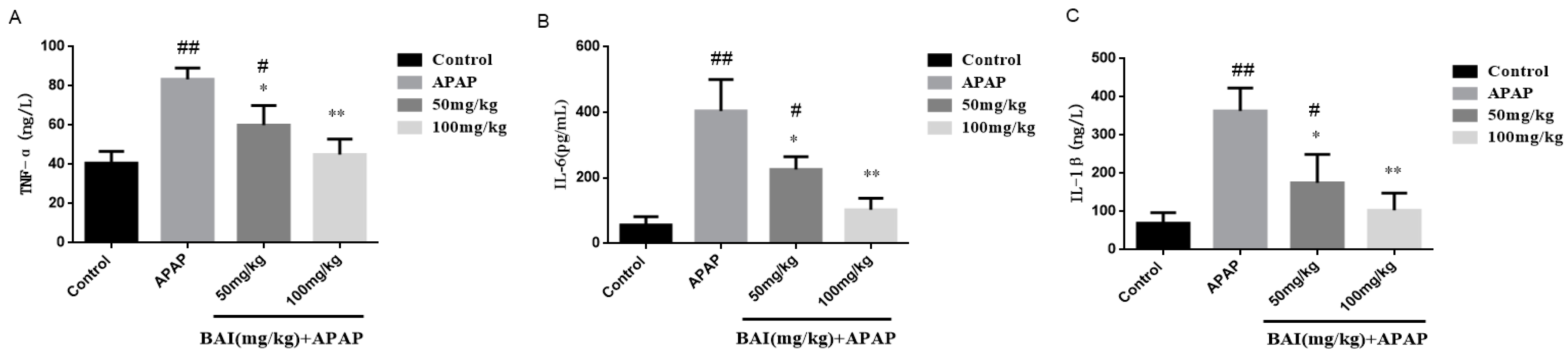
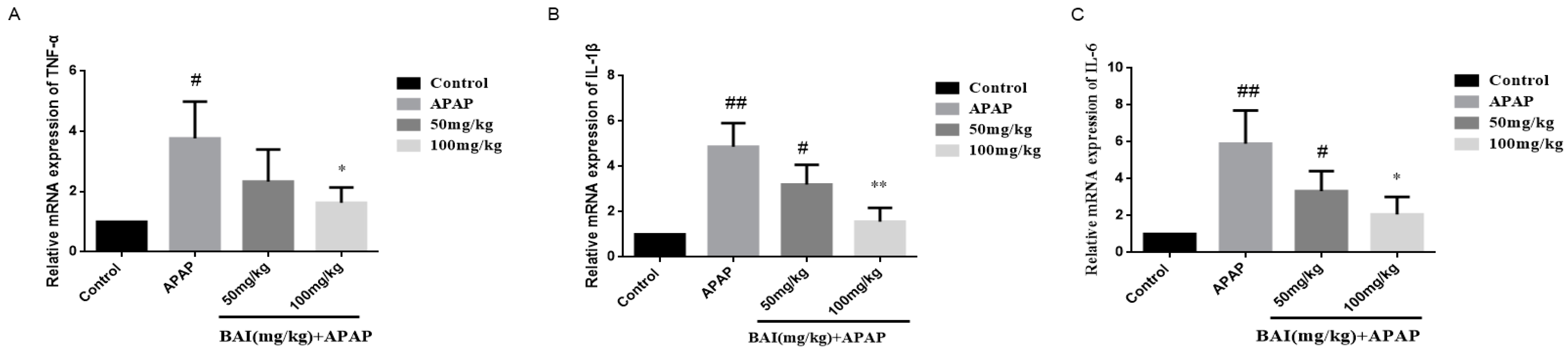
2.5. Effects of Baicalein on Inflammatory Cytokines Levels
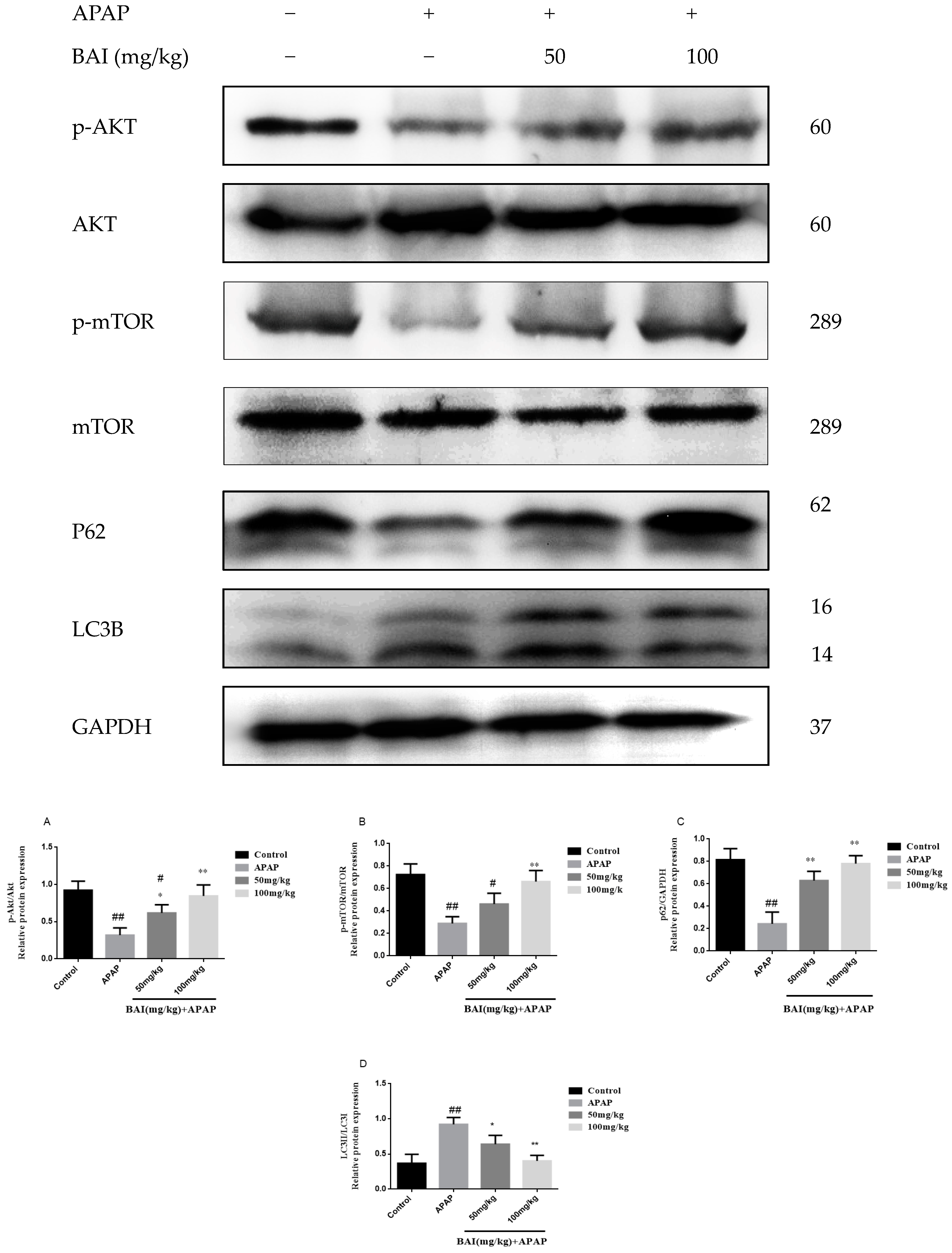
2.6. Baicalein Regulates Autophagy in Response to APAP Liver Injury
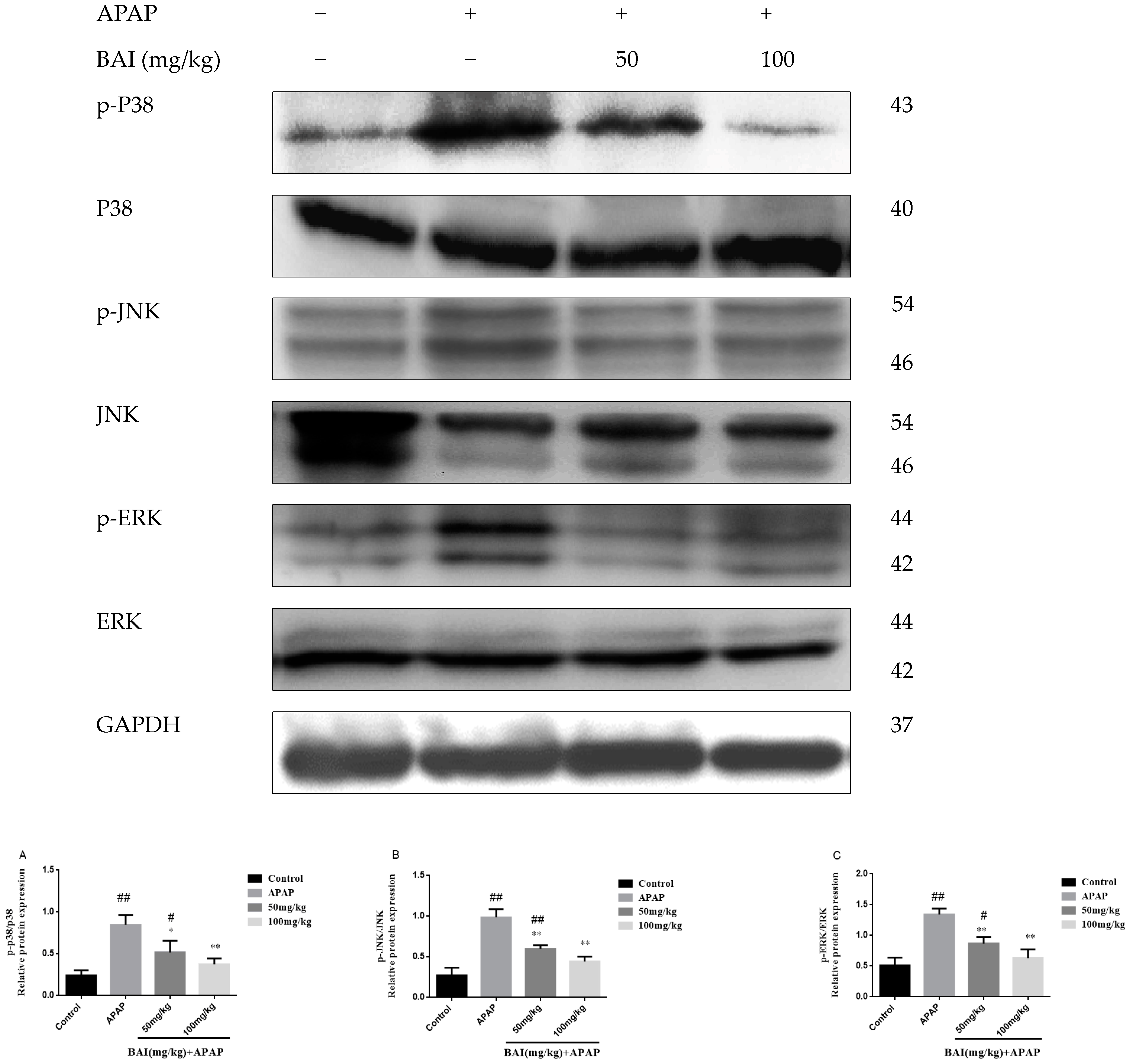
2.7. Baicalein Prevented the MAPK Pathway Activation
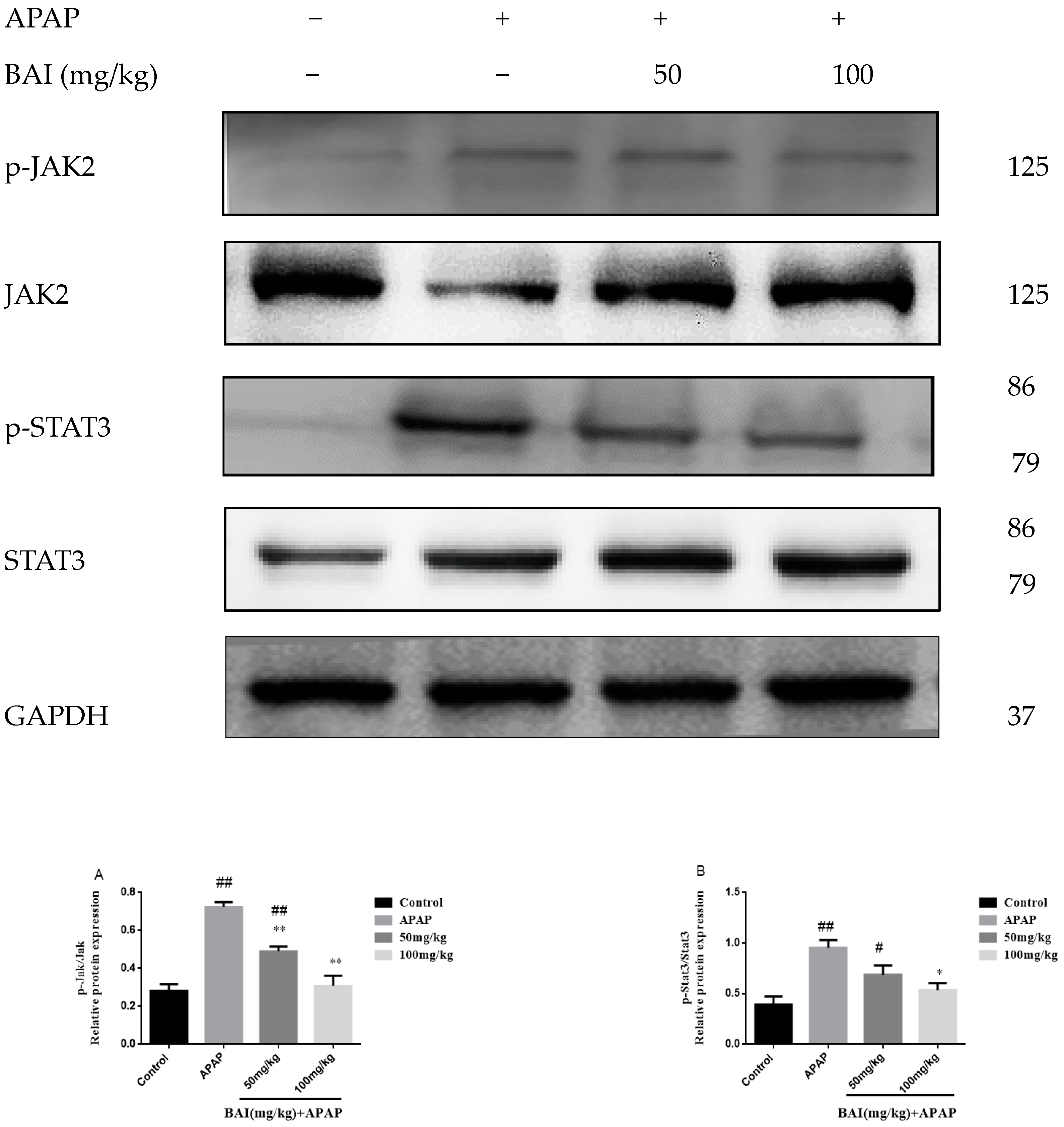
2.8. BAI Suppressed the Expression of p-JAK2 and p-STAT3 Proteins in APAP Liver Injury
3. Discussion
4. Materials and Methods
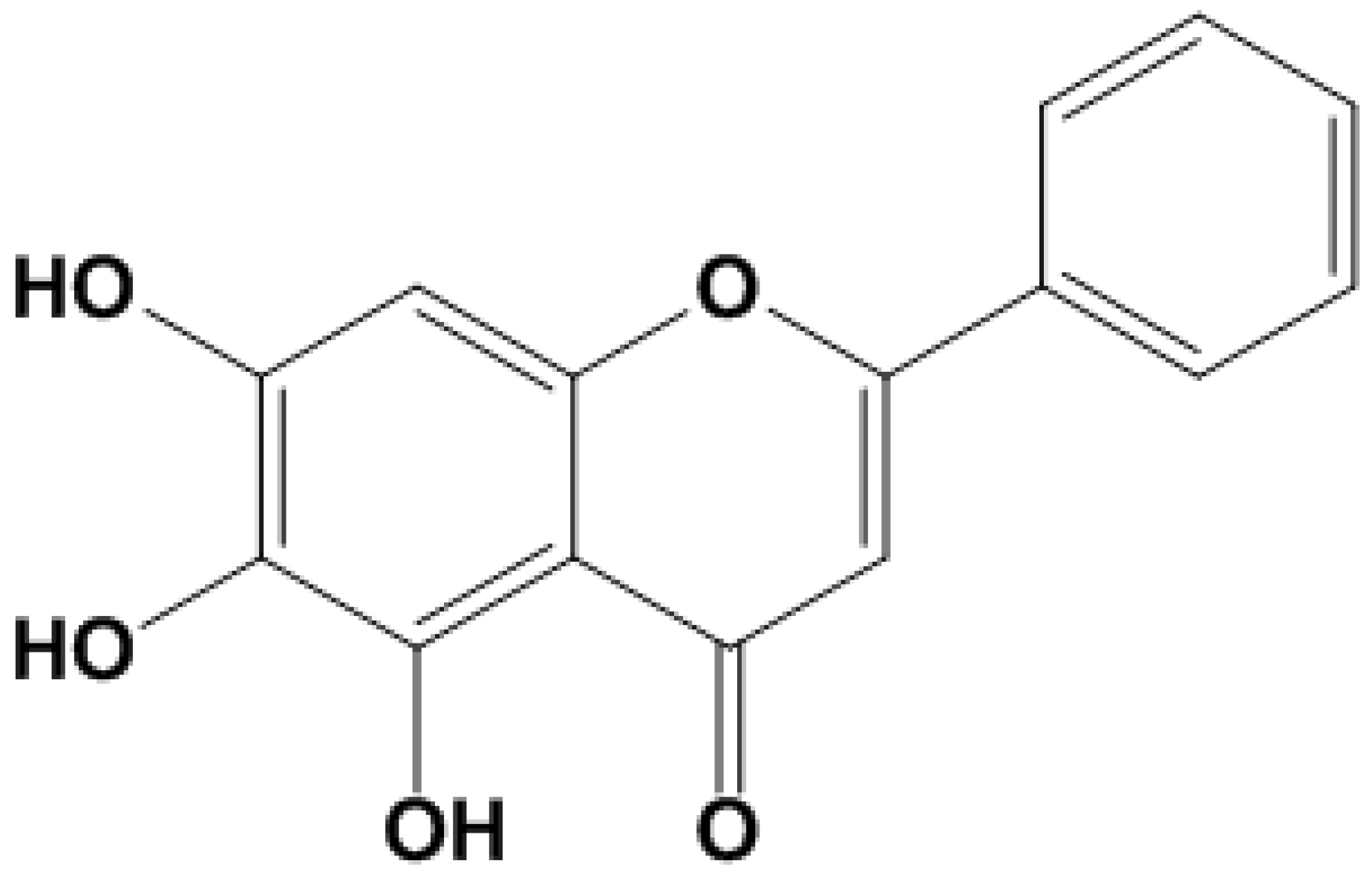
4.1. Chemicals and Reagents
4.2. Experimental Setting
4.3. Serum ALT and AST Assays
4.4. Histopathological Analysis
4.5. Measurement of Biochemical Index of GSH, SOD, CAT and MDA
4.6. TNF-α IL-6and IL-1β Analysis by ELISA
4.7. Quantitative Real-Time PCR
4.8. Western Blotting Analysis
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Formica, D.; Sultana, J.; Cutroneo, P.M.; Lucchesi, S.; Angelica, R.; Crisafulli, S.; Trifirò, G. The economic burden of preventable adverse drug reactions: A systematic review of observational studies. Expert Opin. Drug Saf. 2018, 17, 681–695. [Google Scholar] [CrossRef] [PubMed]
- Björnsson, E.S.; Hoofnagle, J.H. Categorization of drugs implicated in causing liver injury: Critical assessment based on published case reports. Hepatology 2015, 63, 590–603. [Google Scholar] [CrossRef]
- Getachew, Y.; James, L.; Lee, W.M.; Thiele, D.L.; Miller, B.C. Susceptibility to acetaminophen (apap) toxicity unexpectedly is decreased during acute viral hepatitis in mice. Biochem. Pharmacol. 2010, 79, 1363–1371. [Google Scholar] [CrossRef] [PubMed]
- Yan, M.; Huo, Y.; Yin, S.; Hu, H. Mechanisms of acetaminophen-induced liver injury and its implications for therapeutic interventions. Redox Biol. 2018, 17, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Herndon, C.M.; Dankenbring, D.M. Patient perception and knowledge of acetaminophen in a large family medicine service. J. Pain Palliat. Care Pharmacother. 2014, 28, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Rubin, J.B.; Hameed, B.; Gottfried, M.; Lee, W.M.; Sarkar, M. Acetaminophen-induced Acute Liver Failure Is More Common and More Severe in Women. Clin. Gastroenterol. Hepatol. 2018, 16, 936–946. [Google Scholar] [CrossRef] [PubMed]
- Watkins, P.B.; Seeff, L.B. Drug-induced liver injury: Summary of a single topic clinical research conference. Hepatology 2006, 43, 618–631. [Google Scholar] [CrossRef]
- McGreal, S.R.; Bhushan, B.; Walesky, C.; McGill, M.R.; Lebofsky, M.; Kandel, S.E.; Tan, E.P. Modulation of O-GlcNAc Levels in the Liver Impacts Acetaminophen-Induced Liver Injury by Affecting Protein Adduct Formation and Glutathione Synthesis. Toxicol. Sci. 2018, 162, 599–610. [Google Scholar] [CrossRef]
- Huang, M.; Williams, J.; Kong, B.; Zhu, Y.; Li, G.; Zhu, Z.; Guo, G.L. Fibroblast growth factor 15 deficiency increases susceptibility but does not improve repair to acetaminophen-induced liver injury in mice. Dig. Liver Dis. 2018, 50, 175–180. [Google Scholar] [CrossRef]
- Lee, B.W.; Jeon, B.S.; Yoon, B.I. Exogenous recombinant human thioredoxin-1 prevents acetaminophen-induced liver injury by scavenging oxidative stressors, restoring the thioredoxin-1 system and inhibiting receptor interacting protein-3 overexpression. J. Appl. Toxicol. 2018, 38, 1008–1017. [Google Scholar] [CrossRef]
- Letelier, M.E.; López-Valladares, M.; Peredo-Silva, L.; Rojas-Sepúlveda, D.; Aracena, P. Microsomal oxidative damage promoted by acetaminophen metabolism. Toxicol. Vitro 2011, 25, 1310–1313. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; Ramachandran, A.; Jaeschke, H. Oxidative stress during acetaminophen hepatotoxicity: Sources, pathophysiological role and therapeutic potential. Redox Biol. 2016, 10, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Jaeschke, H.; McGill, M.R.; Ramachandran, A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: Lessons learned from acetaminophen hepatotoxicity. Drug Metab. Rev. 2012, 44, 88–106. [Google Scholar] [CrossRef] [PubMed]
- McNulty, R.; Lim, J.M.E.; Chandru, P.; Gunja, N. Fewer adverse effects with a modified two-bag acetylcysteine protocol in paracetamol overdose. Clin. Toxicol. 2018, 56, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Sandilands, E.A.; Bateman, D.N. Adverse reactions associated with acetylcysteine. Clin. Toxicol. 2009, 47, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Bochorakova, H.; Paulová, H.; Slanina, J.; Musil, P.; Táborská, E. Main flavonoids in the root of Scutellaria baicalensis cultivated in Europe and their comparative antiradical properties. Phytother. Res. 2003, 17, 640–644. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Chen, X.Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Sci. Bull. 2016, 61, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Cui, M.Y.; Levsh, O.; Yang, D.; Liu, J.; Li, J.; Chen, X.Y. Two CYP82D Enzymes Function as Flavone Hydroxylases in the Biosynthesis of Root-Specific 4′-Deoxyflavones in Scutellaria baicalensis. Mol. Plant 2018, 11, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Huang, K.; Xu, H. Protective effects of flavonoids in the roots of scutellaria baicalensis georgi against hydrogen peroxide-induced oxidative stress in hs-sy5y cells. Pharmacol. Res. 2001, 43, 173–178. [Google Scholar] [CrossRef]
- Gong, G.; Wang, H.; Kong, X.; Duan, R.; Dong, T.T.; Tsim, K.W. Flavonoids are identified from the extract of Scutellariae Radix to suppress inflammatory-induced angiogenic responses in cultured RAW 264.7 macrophages. Sci. Rep. 2018, 8, 17412–17413. [Google Scholar] [CrossRef]
- Morgillo, F.; Sasso, F.C.; Della Corte, C.M.; Vitagliano, D.; D’Aiuto, E.; Troiani, T.; Martinelli, E.; De Vita, F.; Orditura, M.; De Palma, R.; et al. Synergistic effects of metformin treatment in combination with gefitinib, a selective egfr tyrosine kinase inhibitor, in lkb1 wild-type nsclc cell lines. Clin. Cancer Res. 2013, 19, 3508–3519. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Huang, L.; Ouyang, T.; Chen, L. Baicalein improves liver inflammation in diabetic db/db mice by regulating HMGB1/TLR4/NF-kappaB signaling pathway. Int. Immunopharmacol. 2018, 55, 55–62. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Yu, X.; Wang, X.; Tang, L.; Cao, Y.; Xia, J.; Cheng, J. Baicalein and Ly294002 induces liver cancer cells apoptosis via regulating phosphatidyl inositol 3-kinase/Akt signaling pathway. J. Cancer Res. Ther. 2018, 14, S519–S525. [Google Scholar]
- Rahimian, R.; Fakhfouri, G.; Ejtemaei Mehr, S.; Ghia, J.E.; Genazzani, A.A. Tropisetron attenuates amyloid-beta-induced inflammatory and apoptotic responses in rats. Eur. J. Clin. Investig. 2013, 43, 1039–1051. [Google Scholar] [CrossRef]
- Woolbright, B.L.; Jaeschke, H. Role of the inflammasome in acetaminophen-induced liver injury and acute liver failure. J. Hepatol. 2016, 66, 836–848. [Google Scholar] [CrossRef] [PubMed]
- McGill, M.R.; Williams, C.D.; Xie, Y.; Ramachandran, A.; Jaeschke, H. Acetaminophen-induced liver injury in rats and mice: Comparison of protein adducts, mitochondrial dysfunction, and oxidative stress in the mechanism of toxicity. Toxicol. Appl. Pharmacol. 2012, 264, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.D.; Antoine, D.J.; Shaw, P.J.; Benson, C.; Farhood, A.; Williams, D.P.; Jaeschke, H. Role of the Nalp3 inflammasome in acetaminophen-induced sterile inflammation and liver injury. Toxicol. Appl. Pharmacol. 2011, 252, 2892–2897. [Google Scholar] [CrossRef]
- Chao, X.; Wang, H.; Jaeschke, H.; Ding, W.X. Role and mechanisms of autophagy in acetaminophen-induced liver injury. Liver Int. 2018, 38, 1363–1374. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Chen, C. Inhibition of acetaminophen-induced hepatotoxicity in mice by exogenous thymosinbeta4 treatment. Int. Immunopharmacol. 2018, 61, 20–28. [Google Scholar] [CrossRef]
- Xu, X.Y.; Hu, J.N.; Liu, Z.; Zhang, R.; He, Y.F.; Hou, W.; Li, W. Saponins (Ginsenosides) from the Leaves of Panax quinquefolius Ameliorated Acetaminophen-Induced Hepatotoxicity in Mice. J. Agric. Food. Chem. 2017, 65, 3684–3692. [Google Scholar] [CrossRef]
- Uchida, N.S.; Silva-Filho, S.E.; Aguiar, R.P.; Wiirzler, L.A.M.; Cardia, G.F.E.; Cavalcante, H.A.O.; Cuman, R.K.N. Protective Effect of Cymbopogon citratus Essential Oil in Experimental Model of Acetaminophen-Induced Liver Injury. Am. J. Chin. Med. 2017, 45, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; MacMillan-Crow, L.A.; Rafferty, T.M.; Saba, H.; Roberts, D.W.; Fifer, E.K.; James, L.P.; Hinson, J.A. Acetaminophen-induced hepatotoxicity in mice occurs with inhibition of activity and nitration of mitochondrial manganese superoxide dismutase. J. Pharmacol. Exper. Ther. 2010, 337, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Feng, Y.; Cui, R.; Qiu, M.; Zhang, J.; Liu, C. Simvastatin protects against acetaminophen-induced liver injury in mice. Biomed. Pharmacother. 2018, 98, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Sun, R.; Wei, H.; Tian, Z. High-mobility group box 1 (hmgb1)-toll-like receptor (tlr)4-interleukin (il)-23-il-17a axis in drug-induced damage-associated lethal hepatitis: Interaction of γδ t cells with macrophages. Hepatology 2013, 57, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, X.; Wang, B.; Xu, X.; Wu, X.; Guo, M.; Wang, F. Effect of JAK2/STAT3 signaling pathway on liver injury associated with severe acute pancreatitis in rats. Exp. Ther. Med. 2018, 16, 2013–2021. [Google Scholar] [CrossRef] [PubMed]
- Silver, D.L.; Naora, H.; Liu, J.; Cheng, W.; Montell, D.J. Activated signal transducer and activator of transcription (STAT) 3: Localization in focal adhesions and function in ovarian cancer cell motility. Cancer Res. 2004, 64, 3550–3558. [Google Scholar] [CrossRef] [PubMed]
- Muhl, H. STAT3, a Key Parameter of Cytokine-Driven Tissue Protection during Sterile Inflammation—The Case of Experimental Acetaminophen (Paracetamol)-Induced Liver Damage. Front. Immunol. 2016, 7, 163. [Google Scholar] [CrossRef]
- Qi, Z.; Yin, F.; Lu, L.; Shen, L.; Qi, S.; Lan, L.; Luo, L.; Yin, Z. Baicalein reduces lipopolysaccharide-induced inflammation via suppressing jak/stats activation and ros production. Inflamm. Res. 2013, 62, 845–855. [Google Scholar] [CrossRef]
- Xu, S.; Niu, P.; Chen, K.; Xia, Y.; Yu, Q.; Liu, N.; Wang, W. The liver protection of propylene glycol alginate sodium sulfate preconditioning against ischemia reperfusion injury: Focusing MAPK pathway activity. Sci. Rep. 2017, 7, 15175–15176. [Google Scholar] [CrossRef]
- Wang, J.X.; Zhang, C.; Fu, L.; Zhang, D.G.; Wang, B.W.; Zhang, Z.H.; Xu, D.X. Protective effect of rosiglitazone against acetaminophen-induced acute liver injury is associated with down-regulation of hepatic NADPH oxidases. Toxicol. Lett. 2017, 265, 38–46. [Google Scholar] [CrossRef]
- Yang, C.; Li, L.; Ma, Z.; Zhong, Y.; Pang, W.; Xiong, M.; Li, Y. Hepatoprotective effect of methyl ferulic acid against carbon tetrachloride-induced acute liver injury in rats. Exp. Ther. Med. 2018, 15, 2228–2238. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.U.; Stamper, B.D. Polyphenols reported to shift APAP-induced changes in MAPK signaling and toxicity outcomes. Chem. Biol. Interact. 2017, 277, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Z.; Zhi-Na, M.; Yin-Shi, S.U.N.; Shen, R.E.N.; JIANG, S.; Zhang, W.Z.; Wei, L.I. Protective effects of extracts of Schisandra chinensis stems against acetaminophen-induced hepatotoxicity via regulation of MAPK and caspase-3 signaling pathways. Chin. J. Nat. Med. 2018, 16, 700–713. [Google Scholar] [CrossRef]
- Yin, Y.; Zhang, Y.; Li, H.; Zhao, Y.; Cai, E.; Zhu, H.; Liu, J. Triterpenoids from fruits of Sorbus pohuashanensis inhibit acetaminophen-induced acute liver injury in mice. Biomed. Pharmacothr. 2019, 109, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.L.; Liu, Y.; Leng, J.; Zhang, J.; He, Y.F.; Chen, C.; Li, W. Platycodin D protects acetaminophen-induced hepatotoxicity by inhibiting hepatocyte MAPK pathway and apoptosis in C57BL/6J mice. Biomed. Pharmacother. 2018, 107, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Whelan, K.A.; Merves, J.F.; Giroux, V.; Tanaka, K.; Guo, A.; Chandramouleeswaran, P.M.; Fernando, S.D. Autophagy mediates epithelial cytoprotection in eosinophilic oesophagitis. Gut 2017, 66, 1197–1207. [Google Scholar] [CrossRef]
- Jiang, P.; Mizushima, N. LC3- and p62-based biochemical methods for the analysis of autophagy progression in mammalian cells. Methods 2015, 75, 13–18. [Google Scholar] [CrossRef]
- Moscat, J.; Karin, M.; Diaz-Meco, M.T. p62 in Cancer: Signaling Adaptor Beyond Autophagy. Cell 2016, 167, 606–609. [Google Scholar] [CrossRef]
- Reina-Campos, M.; Shelton, P.M.; Diaz-Meco, M.T.; Moscat, J. Metabolic reprogramming of the tumor microenvironment by p62 and its partners. Biochem. Biophys. Acta Rev. Cancer 2018, 1870, 88–95. [Google Scholar] [CrossRef]
- Ni, H.M.; McGill, M.R.; Chao, X.; Du, K.; Williams, J.A.; Xie, Y.; Ding, W.X. Removal of acetaminophen protein adducts by autophagy protects against acetaminophen-induced liver injury in mice. J. Hepatol. 2016, 65, 354–362. [Google Scholar] [CrossRef]
- Lin, Z.; Wu, F.; Lin, S.; Pan, X.; Jin, L.; Lu, T.; Li, X. Adiponectin protects against acetaminophen-induced mitochondrial dysfunction and acute liver injury by promoting autophagy in mice. J. Hepatol. 2014, 61, 825–831. [Google Scholar] [CrossRef] [PubMed]
- Mo, R.; Lai, R.; Lu, J.; Zhuang, Y.; Zhou, T.; Jiang, S.; Chen, L. Enhanced autophagy contributes to protective effects of IL-22 against acetaminophen-induced liver injury. Theranostics 2018, 8, 4170–4180. [Google Scholar] [CrossRef] [PubMed]
- Duan, P.; Hu, C.; Quan, C.; Yu, T.; Zhou, W.; Yuan, M.; Yang, K. 4-Nonylphenol induces apoptosis, autophagy and necrosis in Sertoli cells: Involvement of ROS-mediated AMPK/AKT-mTOR and JNK pathways. Toxicology 2016, 341–343, 28–40. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Wang, L.; Huang, J.; Lv, H.; Deng, X.; Ci, X. Pterostilbene Reduces Acetaminophen-Induced Liver Injury by Activating the Nrf2 Antioxidative Defense System via the AMPK/Akt/GSK3beta Pathway. Cell Physiol. Biochem. 2018, 49, 1943–1958. [Google Scholar] [CrossRef] [PubMed]
- Saberi, B.; Ybanez, M.D.; Johnson, H.S.; Gaarde, W.A.; Han, D.; Kaplowitz, N. Protein kinase C (PKC) participates in acetaminophen hepatotoxicity through c-jun-N-terminal kinase (JNK)-dependent and -independent signaling pathways. Hepatology 2014, 59, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Liu, C.; Chen, S.; Ye, Y.; Guo, M.; Ren, Q.; Huang, S. Activation of AMPK and inactivation of Akt result in suppression of mTOR-mediated S6K1 and 4E-BP1 pathways leading to neuronal cell death in in vitro models of Parkinson’s disease. Cell. Signal. 2014, 26, 1680–1689. [Google Scholar] [CrossRef] [PubMed]
- Yim, N.H.; Hwang, Y.H.; Liang, C.; Ma, J.Y. A platycoside-rich fraction from the root of Platycodon grandiflorum enhances cell death in A549 human lung carcinoma cells via mainly AMPK/mTOR/AKT signal-mediated autophagy induction. J. Ethnopharmacol. 2016, 194, 1060–1068. [Google Scholar] [CrossRef]
Sample Availability: Samples of Baicalein are not available from the authors. Baicalein was purchased from Chengdu Pufei De Biotech Co., Ltd. (Chengdu, China). |
| Groups | Dosage (mg/kg) | Weight (g) | Organ Index (mg/g × 100) | ||
|---|---|---|---|---|---|
| Initial | Final | liver | Kidney | ||
| Control | - | 29.87 ± 1.03 | 29.35 ± 1.22 | 1.32 ± 0.03 | 0.39 ± 0.04 |
| APAP+BAI | 50 | 29.45 ± 1.12 | 29.52 ± 1.21 | 1.33 ± 0.34 | 0.39 ± 0.05 |
| APAP+BAI | 100 | 29.66 ± 1.09 | 29.41 ± 1.13 | 1.36 ± 0.32 | 0.40 ± 0.03 |
| APAP | - | 29.58 ± 1.16 | 27.64 ± 1.01 | 1.47 ± 0.21 | 0.41 ± 0.02 |
| Primer | Sequence | Length (bp) | |
|---|---|---|---|
| IL-1β | F | CCCAACTGGTACATCAGCACCTC | 23 |
| R | GACACGGATTCCATGGTGAAGTC | 23 | |
| IL-6 | F | CAAAGCCAGAGTCCTTCAGAG | 21 |
| R | GCCACTCCTTCTGTGACTCC | 20 | |
| TNF-α | F | TGGCCTCCCTCTCATCAG | 18 |
| R | ACTTGGTGGTTTGCTACGAC | 20 | |
| GAPDH | F | GTGCTATGTTGCTCTAGACTTCG | 23 |
| R | ATGCCACAGGATTCCATACC | 20 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, H.-C.; Wang, H.; Shi, K.; Li, J.-M.; Zong, Y.; Du, R. Hepatoprotective Effect of Baicalein Against Acetaminophen-Induced Acute Liver Injury in Mice. Molecules 2019, 24, 131. https://doi.org/10.3390/molecules24010131
Zhou H-C, Wang H, Shi K, Li J-M, Zong Y, Du R. Hepatoprotective Effect of Baicalein Against Acetaminophen-Induced Acute Liver Injury in Mice. Molecules. 2019; 24(1):131. https://doi.org/10.3390/molecules24010131
Chicago/Turabian StyleZhou, Hong-Chao, Hui Wang, Kun Shi, Jian-Ming Li, Ying Zong, and Rui Du. 2019. "Hepatoprotective Effect of Baicalein Against Acetaminophen-Induced Acute Liver Injury in Mice" Molecules 24, no. 1: 131. https://doi.org/10.3390/molecules24010131
APA StyleZhou, H.-C., Wang, H., Shi, K., Li, J.-M., Zong, Y., & Du, R. (2019). Hepatoprotective Effect of Baicalein Against Acetaminophen-Induced Acute Liver Injury in Mice. Molecules, 24(1), 131. https://doi.org/10.3390/molecules24010131




