Modulation of Skin Inflammatory Response by Active Components of Silymarin
Abstract
1. Introduction
2. Results
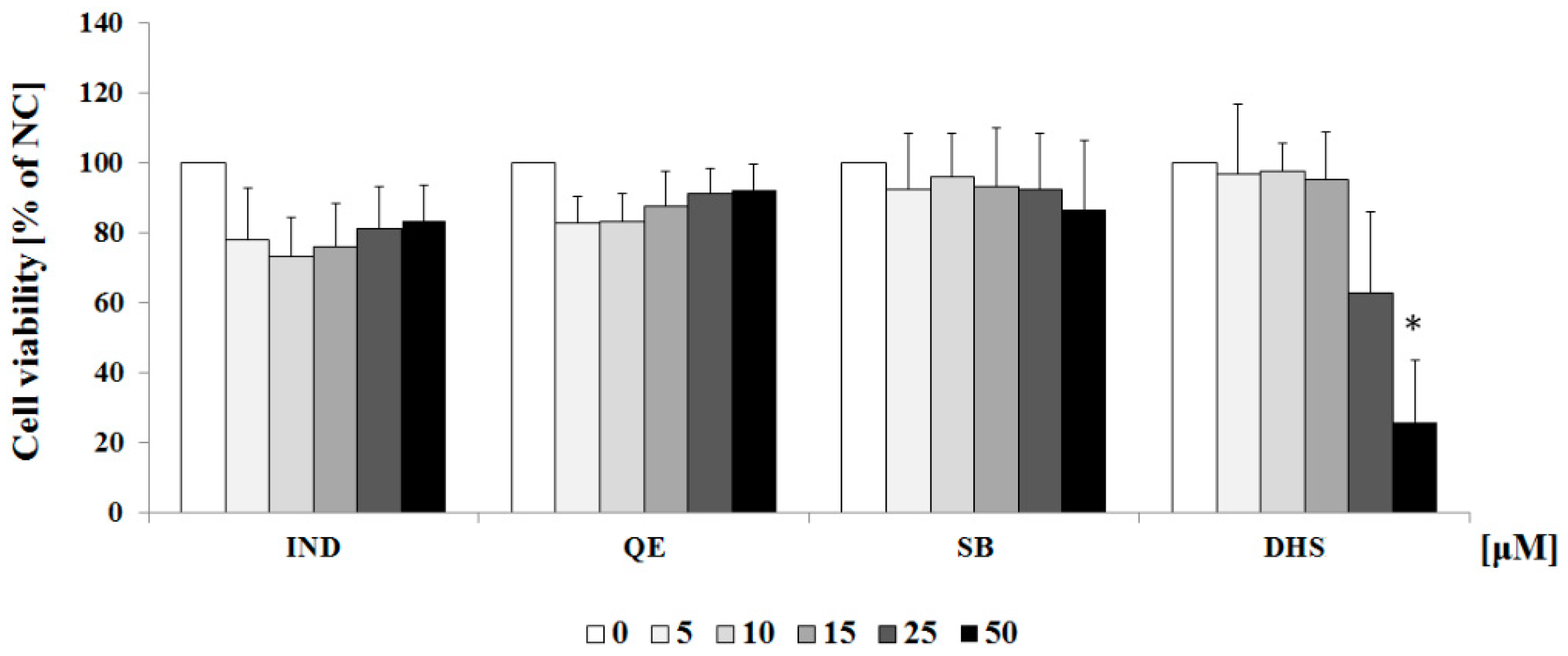
2.1. Cytotoxicity Profiles of the Studied Component of Silymarin and Indomethacin
2.2. Effect of Subtoxic Concentrations of Selected Components of Silymarin on Cytokine and Chemokine Secretion
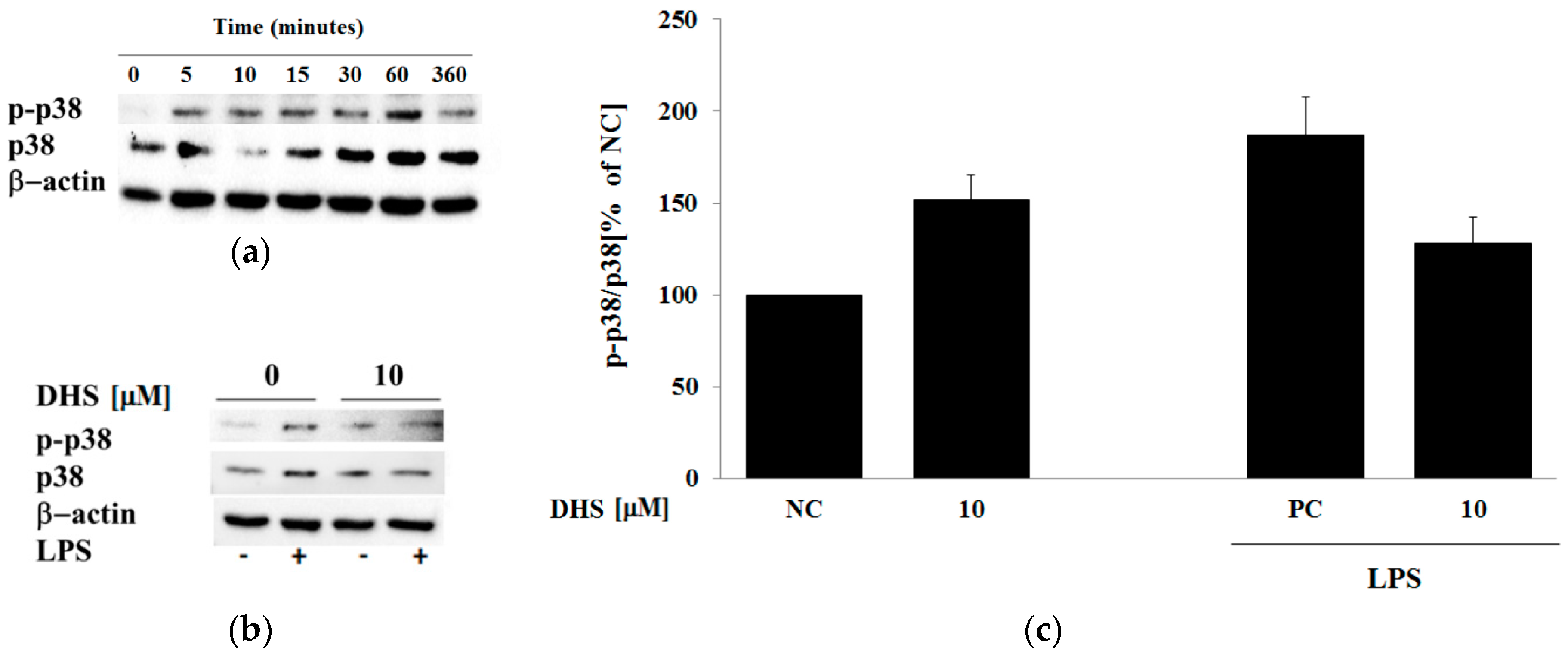
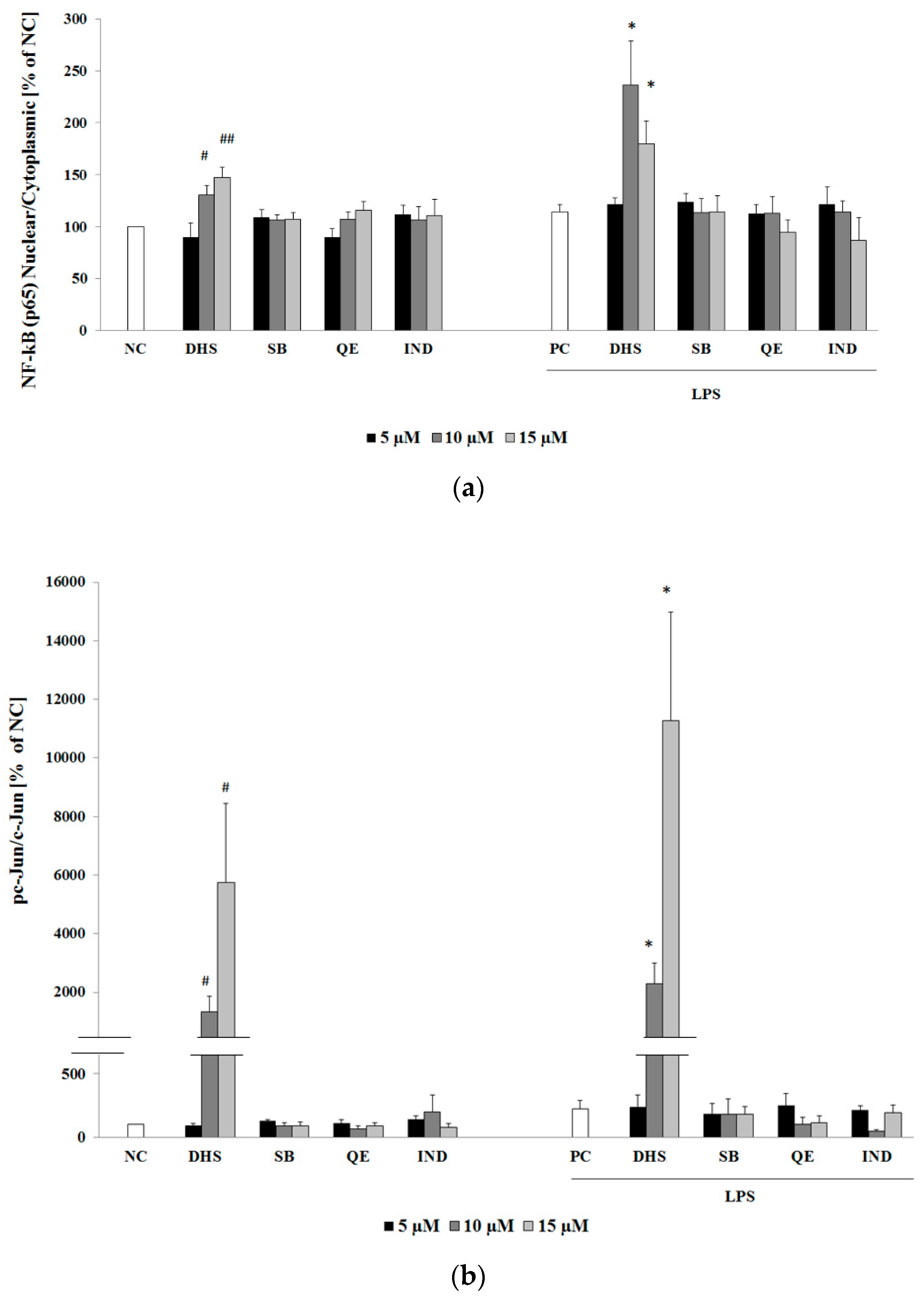
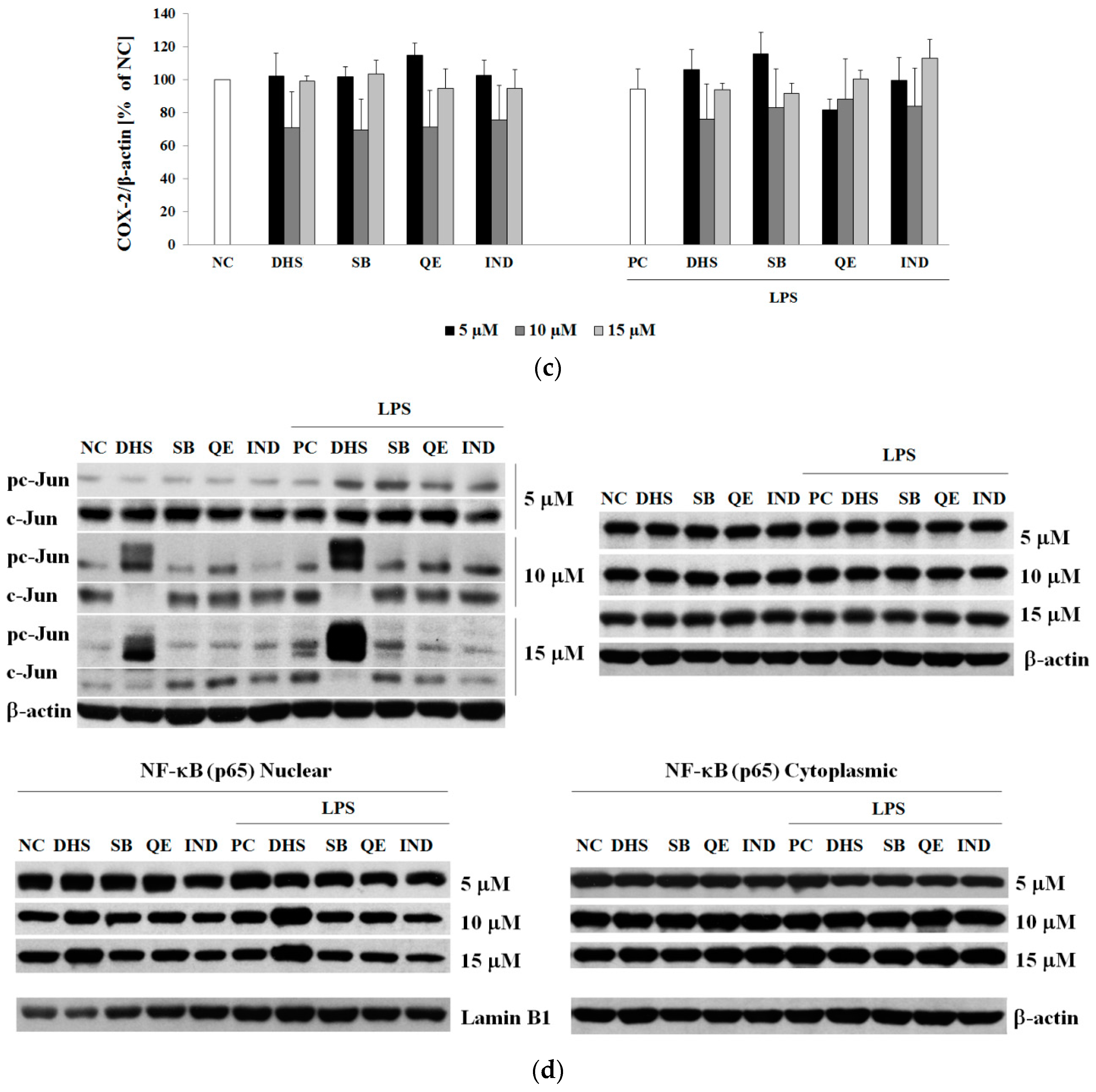
2.3. Western Blotting Analysis of Molecular Targets of the Potential Anti-Inflammatory Action of Subtoxic Concentrations of Selected Components of Silymarin
2.4. Silymarin Component Effects on Cytokines IL-6 and IL-8 and Upstream Transcription Factors NF-κB (p65) or AP-1 (c-Jun) mRNA Levels
2.5. Western Blotting Analysis of Cotreatment of Nontoxic Concentrations of Dehydrosilybin and Lipopolysaccharide
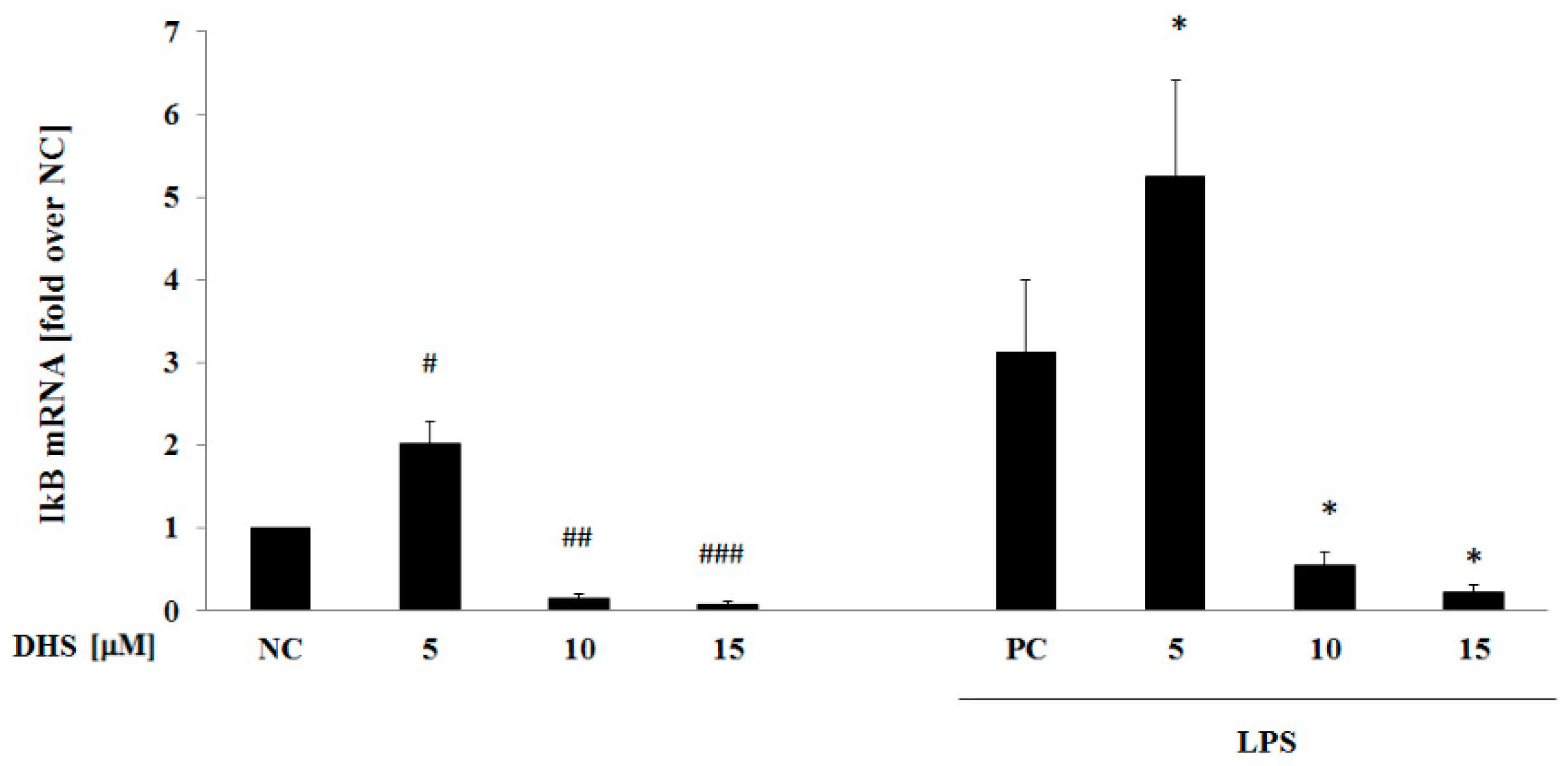
2.6. DHS Effect on IκB mRNA Level with or without LPS Pretreatment
2.7. DHS Effect on NF-κB DNA-Binding Activity with or without Cotreatment with Lipopolysaccharide
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Tested Compounds
4.3. Cell Culture
4.4. Cell Viability Assay
4.5. Treatment Protocols
4.6. Enzyme-Linked Immunosorbent Assay (ELISA)
4.7. Western Blot Analysis
4.8. RNA Isolation and Quantitative Real-Time RT-PCR
4.9. Electrophoretic Mobility Shift Assay (EMSA)
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Baum, C.L.; Arpey, C.J. Normal cutaneous wound healing: Clinical correlation with cellular and molecular events. Dermatol. Surg. 2005, 31, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Juranova, J.; Frankova, J.; Ulrichova, J. The role of keratinocytes in inflammation. J. Appl. Biomed. 2017, 15, 169–179. [Google Scholar] [CrossRef]
- Landen, N.X.; Li, D.; Stahle, M. Transition from inflammation to proliferation: A critical step during wound healing. Cell. Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed]
- Wullaert, A.; Bonnet, M.C.; Pasparakis, M. NF-kappaB in the regulation of epithelial homeostasis and inflammation. Cell Res. 2011, 21, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.R.; Dragoo, J.L. The effect of nonsteroidal anti-inflammatory drugs on tissue healing. Knee Surg. Sports Traumatol. Arthrosc. 2013, 21, 540–549. [Google Scholar] [CrossRef] [PubMed]
- Okamura, M.; Takano, Y.; Hiramatsu, N.; Hayakawa, K.; Yao, J.; Paton, A.W.; Paton, J.C.; Kitamura, M. Suppression of cytokine responses by indomethacin in podocytes: A mechanism through induction of unfolded protein response. Am. J. Physiol. Renal. Physiol. 2008, 295, F1495–F1503. [Google Scholar] [CrossRef] [PubMed]
- Tegeder, I.; Pfeilschifter, J.; Geisslinger, G. Cyclooxygenase-independent actions of cyclooxygenase inhibitors. FASEB J. 2001, 15, 2057–2072. [Google Scholar] [CrossRef] [PubMed]
- Dobrev, H. Evaluation of the inhibitory activity of topical indomethacin, betamethasone valerate and emollients on UVL-induced inflammation by means of non-invasive measurements of the skin elasticity. Photodermatol. Photoimmunol. Photomed. 2001, 17, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.; Rapuano, C.J.; Laibson, P.R.; Eagle, R.C.; Cohen, E.J. Corneal melting associated with use of topical nonsteroidal anti-inflammatory drugs after ocular surgery. Arch. Ophthalmol. 2000, 118, 1129–1132. [Google Scholar] [PubMed]
- Liu, W.; Li, Y.; Zheng, X.; Zhang, K.; Du, Z. Potent inhibitory effect of silibinin from milk thistle on skin inflammation stimuli by 12-O-tetradecanoylphorbol-13-acetate. Food Funct. 2015, 6, 3712–3719. [Google Scholar] [CrossRef] [PubMed]
- van Wenum, E.; Jurczakowski, R.; Litwinienko, G. Media effects on the mechanism of antioxidant action of silybin and 2,3-dehydrosilybin: Role of the enol group. J. Org. Chem. 2013, 78, 9102–9112. [Google Scholar] [CrossRef] [PubMed]
- Pastore, S.; Lulli, D.; Fidanza, P.; Potapovich, A.I.; Kostyuk, V.A.; De Luca, C.; Mikhal’chik, E.; Korkina, L.G. Plant polyphenols regulate chemokine expression and tissue repair in human keratinocytes through interaction with cytoplasmic and nuclear components of epidermal growth factor receptor system. Antioxid. Redox Signal. 2012, 16, 314–328. [Google Scholar] [CrossRef] [PubMed]
- Kren, V.; Walterova, D. Silybin and silymarin--new effects and applications. Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech. Repub. 2005, 149, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, R.; Bernuzzi, S.; Ciani, D.; Ascari, E. Silymarine during maintenance therapy of acute promyelocytic leukemia. Haematologica 1993, 78, 340–341. [Google Scholar] [PubMed]
- Molavi, O.; Narimani, F.; Asiaee, F.; Sharifi, S.; Tarhriz, V.; Shayanfar, A.; Hejazi, M.; Lai, R. Silibinin sensitizes chemo-resistant breast cancer cells to chemotherapy. Pharm. Biol. 2017, 55, 729–739. [Google Scholar] [CrossRef] [PubMed]
- El-Far, Y.M.; Zakaria, M.M.; Gabr, M.M.; El Gayar, A.M.; El-Sherbiny, I.M.; Eissa, L.A. A newly developed silymarin nanoformulation as a potential antidiabetic agent in experimental diabetes. Nanomedicine (Lond.) 2016, 11, 2581–2602. [Google Scholar] [CrossRef] [PubMed]
- Meng, R.; Mahadevan, J.; Oseid, E.; Vallerie, S.; Robertson, R.P. Silymarin activates c-AMP phosphodiesterase and stimulates insulin secretion in a glucose-dependent manner in HIT-T15 cells. Antioxidants 2016, 5, 47. [Google Scholar] [CrossRef]
- Stolf, A.M.; Cardoso, C.C.; Acco, A. Effects of silymarin on diabetes mellitus complications: A review. Phytother. Res. 2017, 31, 366–374. [Google Scholar] [CrossRef]
- Rao, P.R.; Viswanath, R.K. Cardioprotective activity of silymarin in ischemia-reperfusion-induced myocardial infarction in albino rats. Exp. Clin. Cardiol. 2007, 12, 179–187. [Google Scholar]
- Gabrielova, E.; Kren, V.; Jaburek, M.; Modriansky, M. Silymarin component 2,3-dehydrosilybin attenuates cardiomyocyte damage following hypoxia/reoxygenation by limiting oxidative stress. Physiol. Res. 2015, 64, 79–91. [Google Scholar]
- Oh, Y.S. Bioactive compounds and their neuroprotective effects in diabetic complications. Nutrients 2016, 8, 472. [Google Scholar] [CrossRef] [PubMed]
- Fehér, P.; Vecsernyés, M.; Fenyvesi, F.; Váradi, J.; Kiss, T.; Újhelyi, Z.; Nagy, K.; Bacskay, I. Topical application of Silybum marianum extract. Arad. Med. J. 2011, 14, 5–8. [Google Scholar]
- Han, M.H.; Yoon, W.K.; Lee, H.; Han, S.B.; Lee, K.; Park, S.K.; Yang, K.H.; Kim, H.M.; Kang, J.S. Topical application of silymarin reduces chemical-induced irritant contact dermatitis in BALB/c mice. Int. Immunopharmacol. 2007, 7, 1651–1658. [Google Scholar] [CrossRef]
- Sharifi, R.; Rastegar, H.; Kamalinejad, M.; Dehpour, A.R.; Tavangar, S.M.; Paknejad, M.; Mehrabani Natanzi, M.; Ghannadian, N.; Akbari, M.; Pasalar, P. Effect of topical application of silymarin (Silybum marianum) on excision wound healing in albino rats. Acta Med. Iran. 2012, 50, 583–588. [Google Scholar] [PubMed]
- Gomathi, K.; Gopinath, D.; Rafiuddin, A.M.; Jayakumar, R. Quercetin incorporated collagen matrices for dermal wound healing processes in rat. Biomaterials 2003, 24, 2767–2772. [Google Scholar] [CrossRef]
- Caddeo, C.; Diez-Sales, O.; Pons, R.; Fernandez-Busquets, X.; Fadda, A.M.; Manconi, M. Topical anti-inflammatory potential of quercetin in lipid-based nanosystems: In vivo and in vitro evaluation. Pharm. Res. 2014, 31, 959–968. [Google Scholar] [CrossRef] [PubMed]
- Shubina, V.S.; Shatalin, Y.V. Skin regeneration after chemical burn under the effect of taxifolin-based preparations. Bull. Exp. Biol. Med. 2012, 154, 152–157. [Google Scholar] [CrossRef]
- Perez-Victoria, J.M.; Perez-Victoria, F.J.; Conseil, G.; Maitrejean, M.; Comte, G.; Barron, D.; Di Pietro, A.; Castanys, S.; Gamarro, F. High-affinity binding of silybin derivatives to the nucleotide-binding domain of a Leishmania tropica p-glycoprotein-like transporter and chemosensitization of a multidrug-resistant parasite to daunomycin. Antimicrob. Agents Chemother. 2001, 45, 439–446. [Google Scholar] [CrossRef]
- Gažák, R.; Trouillas, P.; Biedermann, D.; Fuksová, K.; Marhol, P.; Kuzma, M.; Křen, V. Base-catalyzed oxidation of silybin and isosilybin into 2,3-dehydro derivatives. Tetrahedron. Lett. 2013, 54, 315–317. [Google Scholar] [CrossRef]
- Theodosiou, E.; Purchartová, K.; Stamatis, H.; Kolisis, F.; Křen, V. Bioavailability of silymarin flavonolignans: Drug formulations and biotransformation. Phytochem. Rev. 2014, 13, 1–18. [Google Scholar] [CrossRef]
- Biedermann, D.; Vavrikova, E.; Cvak, L.; Kren, V. Chemistry of silybin. Nat. Prod. Rep. 2014, 31, 1138–1157. [Google Scholar] [CrossRef] [PubMed]
- Svobodova, A.; Walterova, D.; Psotova, J. Influence of silymarin and its flavonolignans on H2O2-induced oxidative stress in human keratinocytes and mouse fibroblasts. Burns 2006, 32, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, R.; Pasalar, P.; Kamalinejad, M.; Dehpour, A.R.; Tavangar, S.M.; Paknejad, M.; Mehrabani Natanzi, M.; Nourbakhsh, M.; Ahmadi Ashtiani, H.R.; Akbari, M.; et al. The effect of silymarin (Silybum marianum) on human skin fibroblasts in an in vitro wound healing model. Pharm. Biol. 2013, 51, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Svobodova, A.R.; Zalesak, B.; Biedermann, D.; Ulrichova, J.; Vostalova, J. Phototoxic potential of silymarin and its bioactive components. J. Photochem. Photobiol. B-Biol. 2016, 156, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Maitrejean, M.; Comte, G.; Barron, D.; El Kirat, K.; Conseil, G.L.; Di Pietro, A. The flavanolignan silybin and its hemisynthetic derivatives, a novel series of potential modulators of P-glycoprotein. Bioorg. Med. Chem. Lett. 2000, 10, 157–160. [Google Scholar] [CrossRef]
- Rincon, M. Interleukin-6: From an inflammatory marker to a target for inflammatory diseases. Trends Immunol. 2012, 33, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, A.; Knapp, M.; Lang, I.; Kohler, G. Interleukin 8 (IL-8)—A universal biomarker? Int. Arch. Med. 2010, 3, 11. [Google Scholar] [CrossRef]
- Kim, B.R.; Seo, H.S.; Ku, J.M.; Kim, G.J.; Jeon, C.Y.; Park, J.H.; Jang, B.H.; Park, S.J.; Shin, Y.C.; Ko, S.G. Silibinin inhibits the production of pro-inflammatory cytokines through inhibition of NF-kappaB signaling pathway in HMC-1 human mast cells. Inflamm. Res. 2013, 62, 941–950. [Google Scholar] [CrossRef]
- Samanta, R.; Pattnaik, A.K.; Pradhan, K.K.; Mehta, B.K.; Pattanayak, S.P.; Banerjee, S. Wound Healing Activity of Silibinin in Mice. Pharmacogn. Res. 2016, 8, 298–302. [Google Scholar] [CrossRef]
- Ratz-Lyko, A.; Arct, J.; Majewski, S.; Pytkowska, K. Influence of polyphenols on the physiological processes in the skin. Phytother. Res. 2015, 29, 509–517. [Google Scholar] [CrossRef]
- Renner, F.; Schmitz, M.L. Autoregulatory feedback loops terminating the NF-kappaB response. Trends Biochem. Sci. 2009, 34, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Gabrielova, E.; Jaburek, M.; Gazak, R.; Vostalova, J.; Jezek, J.; Kren, V.; Modriansky, M. Dehydrosilybin attenuates the production of ROS in rat cardiomyocyte mitochondria with an uncoupler-like mechanism. J. Bioenerg. Biomembr. 2010, 42, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Huber, A.; Thongphasuk, P.; Erben, G.; Lehmann, W.D.; Tuma, S.; Stremmel, W.; Chamulitrat, W. Significantly greater antioxidant anticancer activities of 2,3-dehydrosilybin than silybin. Biochim. Biophys. Acta 2008, 1780, 837–847. [Google Scholar] [CrossRef] [PubMed]
- Pyszkova, M.; Biler, M.; Biedermann, D.; Valentova, K.; Kuzma, M.; Vrba, J.; Ulrichova, J.; Sokolova, R.; Mojovic, M.; Popovic-Bijelic, A.; et al. Flavonolignan 2,3-dehydroderivatives: Preparation, antiradical and cytoprotective activity. Free Radic. Biol. Med. 2016, 90, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Abe, M.; Yokoyama, Y.; Syuto, T.; Ishibuchi, H.; Ishikawa, O. Interleukin-6 counteracts effects of cyclosporin A on extracellular matrix metabolism by human dermal fibroblasts. Cell Tissue Res. 2008, 333, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Gallucci, R.M.; Sloan, D.K.; Heck, J.M.; Murray, A.R.; O’Dell, S.J. Interleukin 6 indirectly induces keratinocyte migration. J. Investig. Dermatol. 2004, 122, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Liechty, K.W.; Crombleholme, T.M.; Cass, D.L.; Martin, B.; Adzick, N.S. Diminished interleukin-8 (IL-8) production in the fetal wound healing response. J. Surg. Res. 1998, 77, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.; Debnath, B.; Neamati, N. Role of the CXCL8-CXCR1/2 axis in cancer and inflammatory diseases. Theranostics 2017, 7, 1543–1588. [Google Scholar] [CrossRef]
- Perfetto, B.; Donnarumma, G.; Criscuolo, D.; Paoletti, I.; Grimaldi, E.; Tufano, M.A.; Baroni, A. Bacterial components induce cytokine and intercellular adhesion molecules-1 and activate transcription factors in dermal fibroblasts. Res. Microbiol. 2003, 154, 337–344. [Google Scholar] [CrossRef]
- Hoesel, B.; Schmid, J.A. The complexity of NF-kappaB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef]
- Pasparakis, M. Regulation of tissue homeostasis by NF-kappaB signalling: Implications for inflammatory diseases. Nat. Rev. Immunol. 2009, 9, 778–788. [Google Scholar] [CrossRef] [PubMed]
- Tilley, C.; Deep, G.; Agarwal, C.; Wempe, M.F.; Biedermann, D.; Valentova, K.; Kren, V.; Agarwal, R. Silibinin and its 2,3-dehydro-derivative inhibit basal cell carcinoma growth via suppression of mitogenic signaling and transcription factors activation. Mol. Carcinog. 2016, 55, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Karin, M.; Lin, A. NF-kappaB at the crossroads of life and death. Nat. Immunol. 2002, 3, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Sollberger, G.; Strittmatter, G.E.; Garstkiewicz, M.; Sand, J.; Beer, H.D. Caspase-1: The inflammasome and beyond. Innate Immun. 2014, 20, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Kutuk, O.; Poli, G.; Basaga, H. Resveratrol protects against 4-hydroxynonenal-induced apoptosis by blocking JNK and c-JUN/AP-1 signaling. Toxicol. Sci. 2006, 90, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Gažák, R.; Svobodová, A.; Psotová, J.; Sedmera, P.; Přikrylová, V.; Walterová, D.; Křen, V. Oxidised derivatives of silybin and their antiradical and antioxidant activity. Bioorg. Med. Chem. 2004, 12, 5677–5687. [Google Scholar] [CrossRef] [PubMed]
- Křenek, K.; Marhol, P.; Peikerová, Ž.; Křen, V.; Biedermann, D. Preparatory separation of the silymarin flavonolignans by Sephadex LH-20 gel. Food Res. Int. 2014, 65, 115–120. [Google Scholar] [CrossRef]
- Pivodová, V.; Franková, J.; Galandáková, A.; Ulrichová, J. In vitro AuNPs’ cytotoxicity and their effect on wound healing. Nanobiomedicine 2015, 2, 7. [Google Scholar] [CrossRef]
- Zdarilova, A.; Svobodova, A.; Simanek, V.; Ulrichova, J. Prunella vulgaris extract and rosmarinic acid suppress lipopolysaccharide-induced alteration in human gingival fibroblasts. Toxicol. In Vitro 2009, 23, 386–392. [Google Scholar] [CrossRef]
- Bauge, C.; Legendre, F.; Leclercq, S.; Elissalde, J.M.; Pujol, J.P.; Galera, P.; Boumediene, K. Interleukin-1beta impairment of transforming growth factor beta1 signaling by down-regulation of transforming growth factor beta receptor type II and up-regulation of Smad7 in human articular chondrocytes. Arthritis Rheum. 2007, 56, 3020–3032. [Google Scholar] [CrossRef]
- Bauge, C.; Beauchef, G.; Leclercq, S.; Kim, S.J.; Pujol, J.P.; Galera, P.; Boumediene, K. NFkappaB mediates IL-1beta-induced down-regulation of TbetaRII through the modulation of Sp3 expression. J. Cell. Mol. Med. 2008, 12, 1754–1766. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds DHS, QE and SB are available from the authors. |




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juráňová, J.; Aury-Landas, J.; Boumediene, K.; Baugé, C.; Biedermann, D.; Ulrichová, J.; Franková, J. Modulation of Skin Inflammatory Response by Active Components of Silymarin. Molecules 2019, 24, 123. https://doi.org/10.3390/molecules24010123
Juráňová J, Aury-Landas J, Boumediene K, Baugé C, Biedermann D, Ulrichová J, Franková J. Modulation of Skin Inflammatory Response by Active Components of Silymarin. Molecules. 2019; 24(1):123. https://doi.org/10.3390/molecules24010123
Chicago/Turabian StyleJuráňová, Jana, Juliette Aury-Landas, Karim Boumediene, Catherine Baugé, David Biedermann, Jitka Ulrichová, and Jana Franková. 2019. "Modulation of Skin Inflammatory Response by Active Components of Silymarin" Molecules 24, no. 1: 123. https://doi.org/10.3390/molecules24010123
APA StyleJuráňová, J., Aury-Landas, J., Boumediene, K., Baugé, C., Biedermann, D., Ulrichová, J., & Franková, J. (2019). Modulation of Skin Inflammatory Response by Active Components of Silymarin. Molecules, 24(1), 123. https://doi.org/10.3390/molecules24010123





