2-Phenylacetamide Isolated from the Seeds of Lepidium apetalum and Its Estrogen-Like Effects In Vitro and In Vivo
Abstract
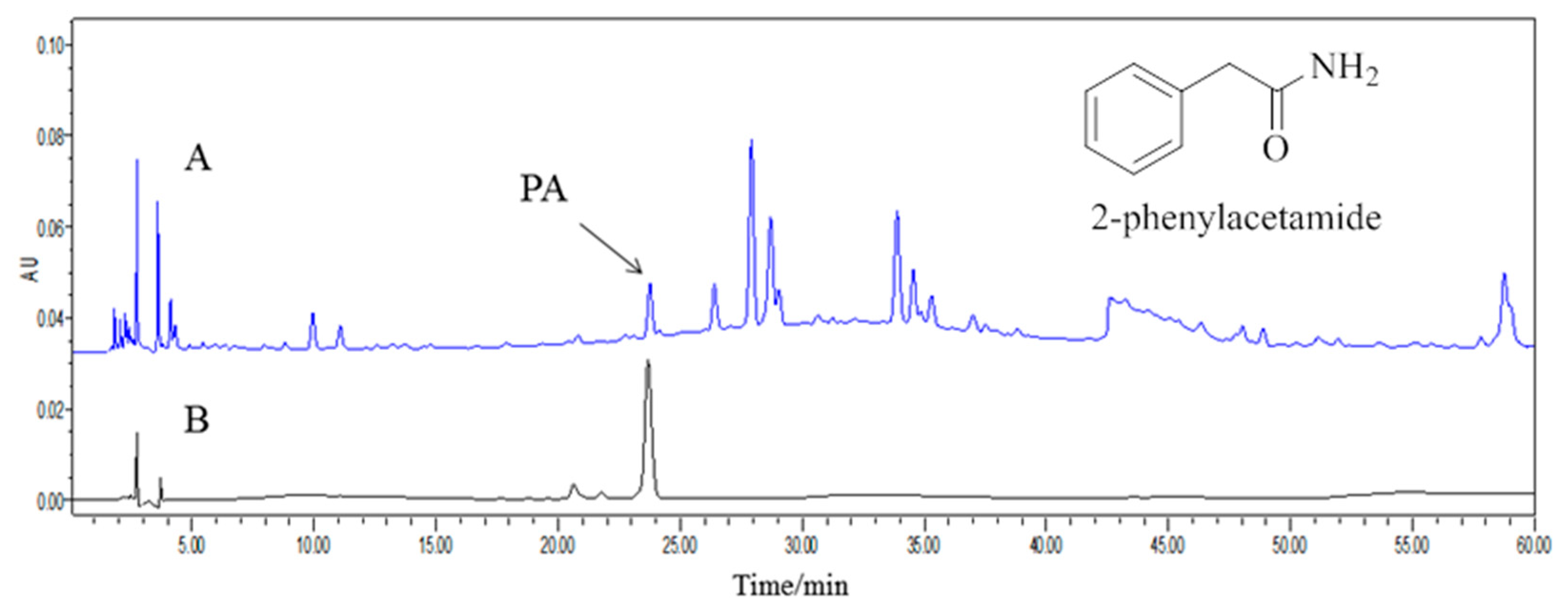
1. Introduction
2. Results
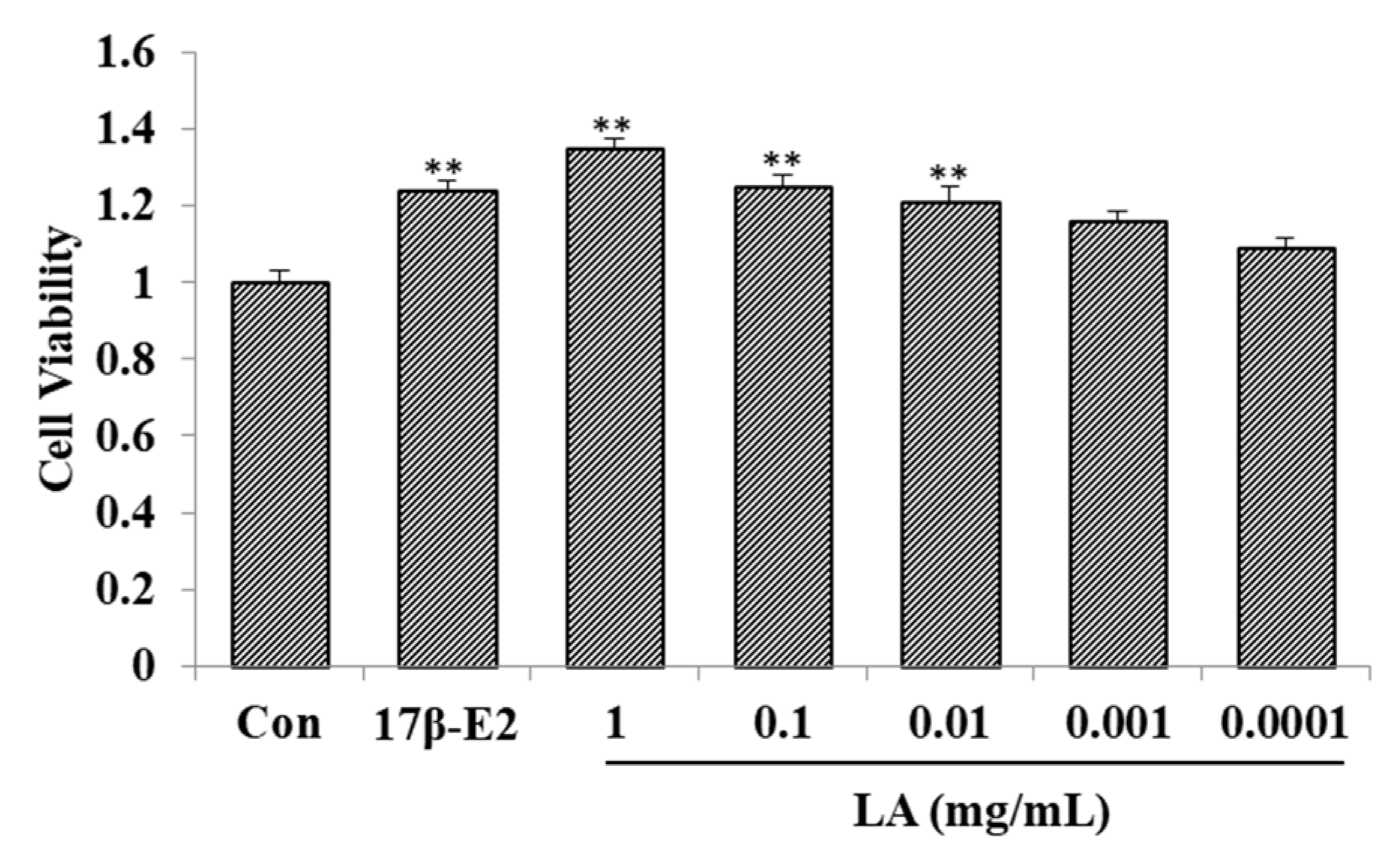
2.1. Effect of LA on MCF-7 Cell Proliferation
2.2. Effects of LA on Immature Female Swiss Mice
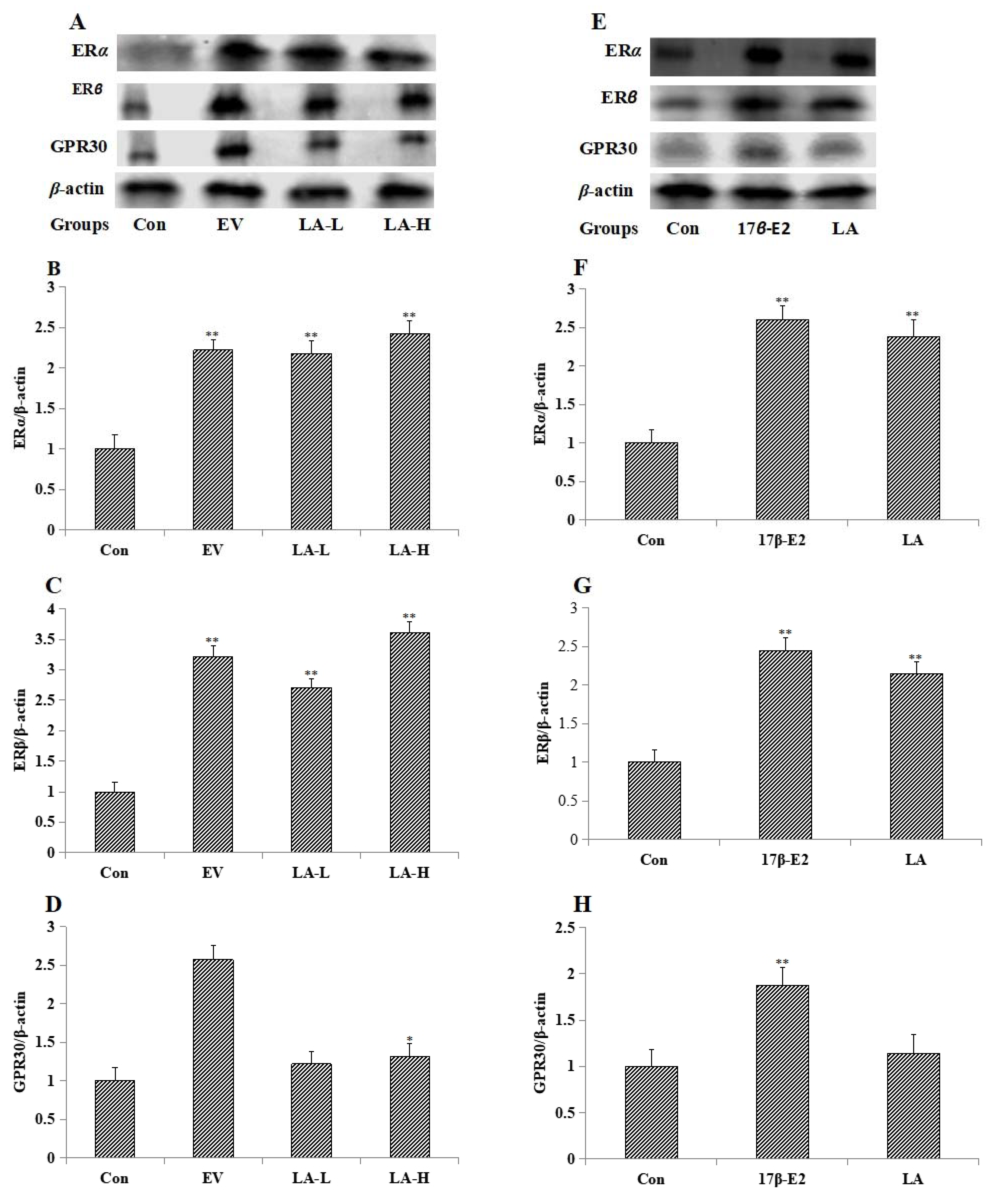
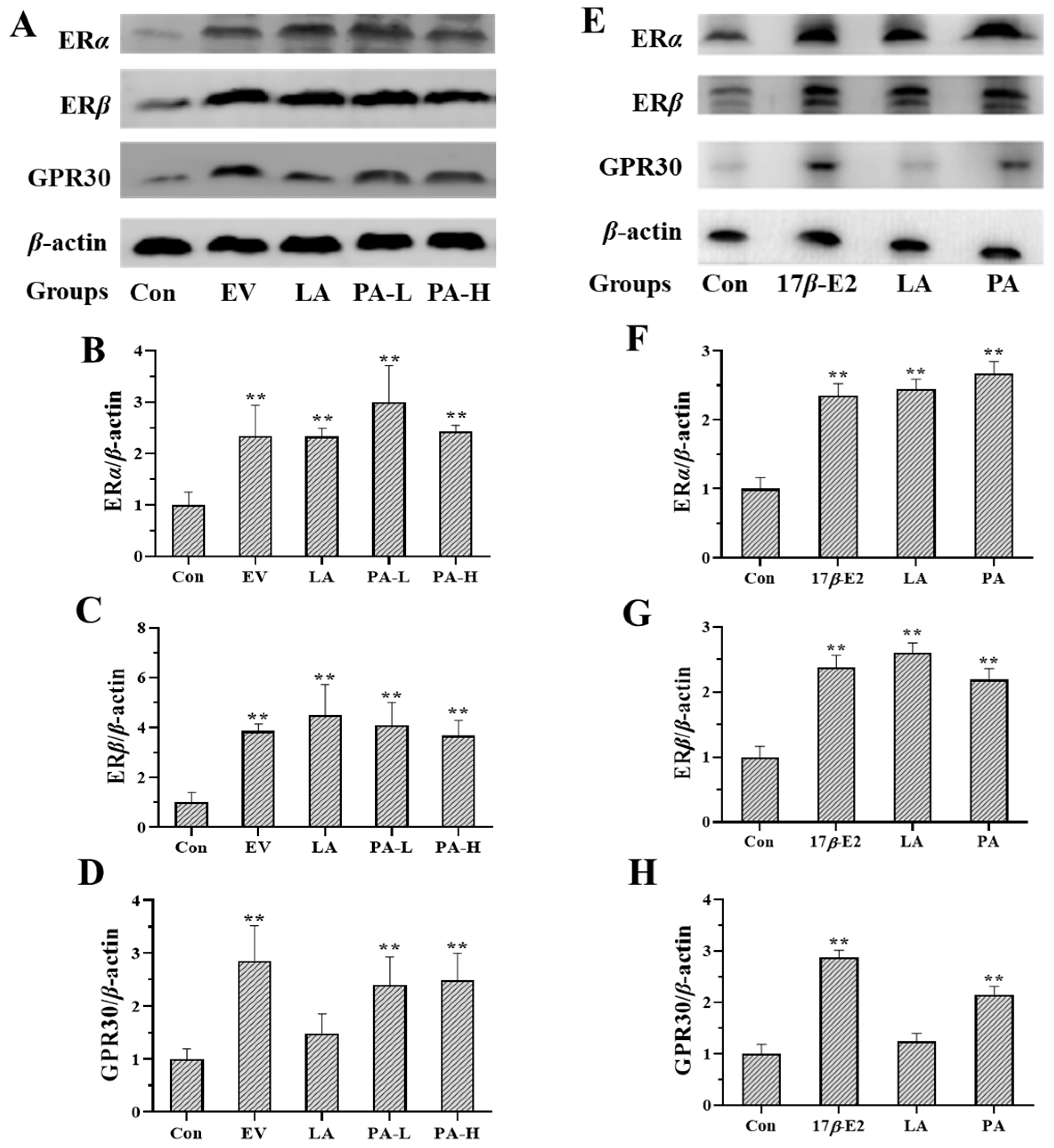
2.3. Effects of LA on the Expression of Estrogen Receptors
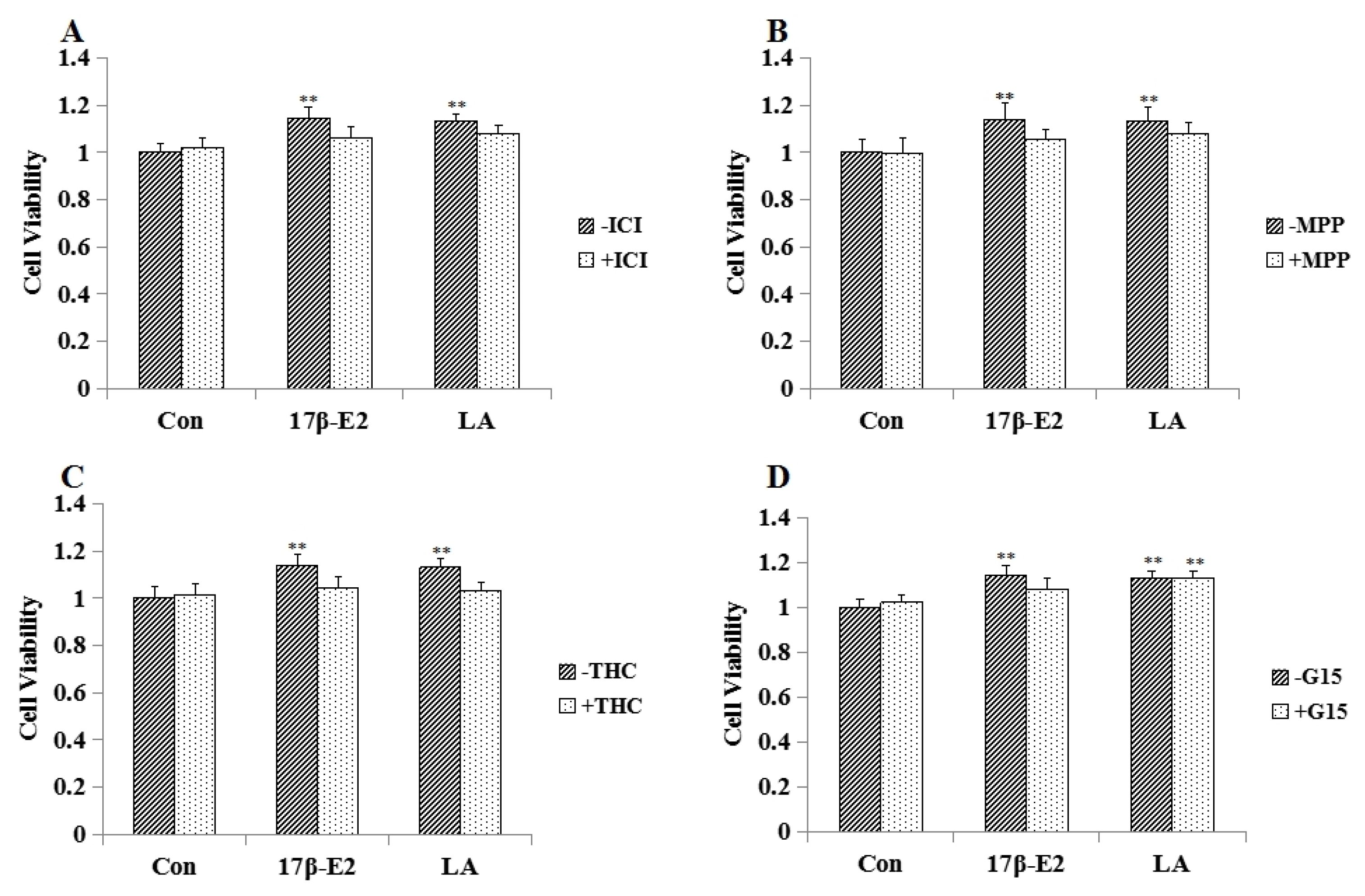
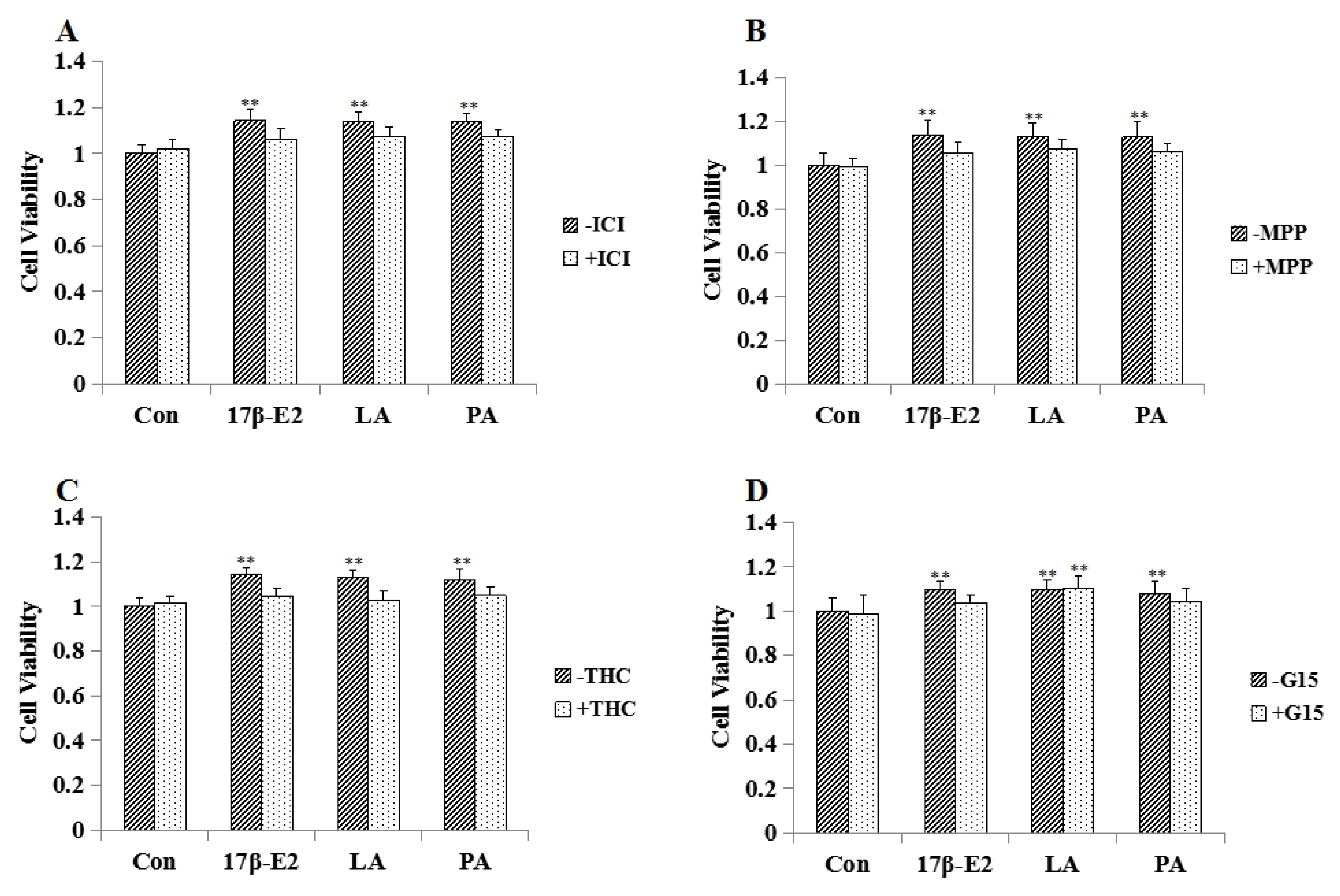
2.4. Effect of Estrogen Receptor Antagonists on LA Aroused MCF-7 Cell Proliferation
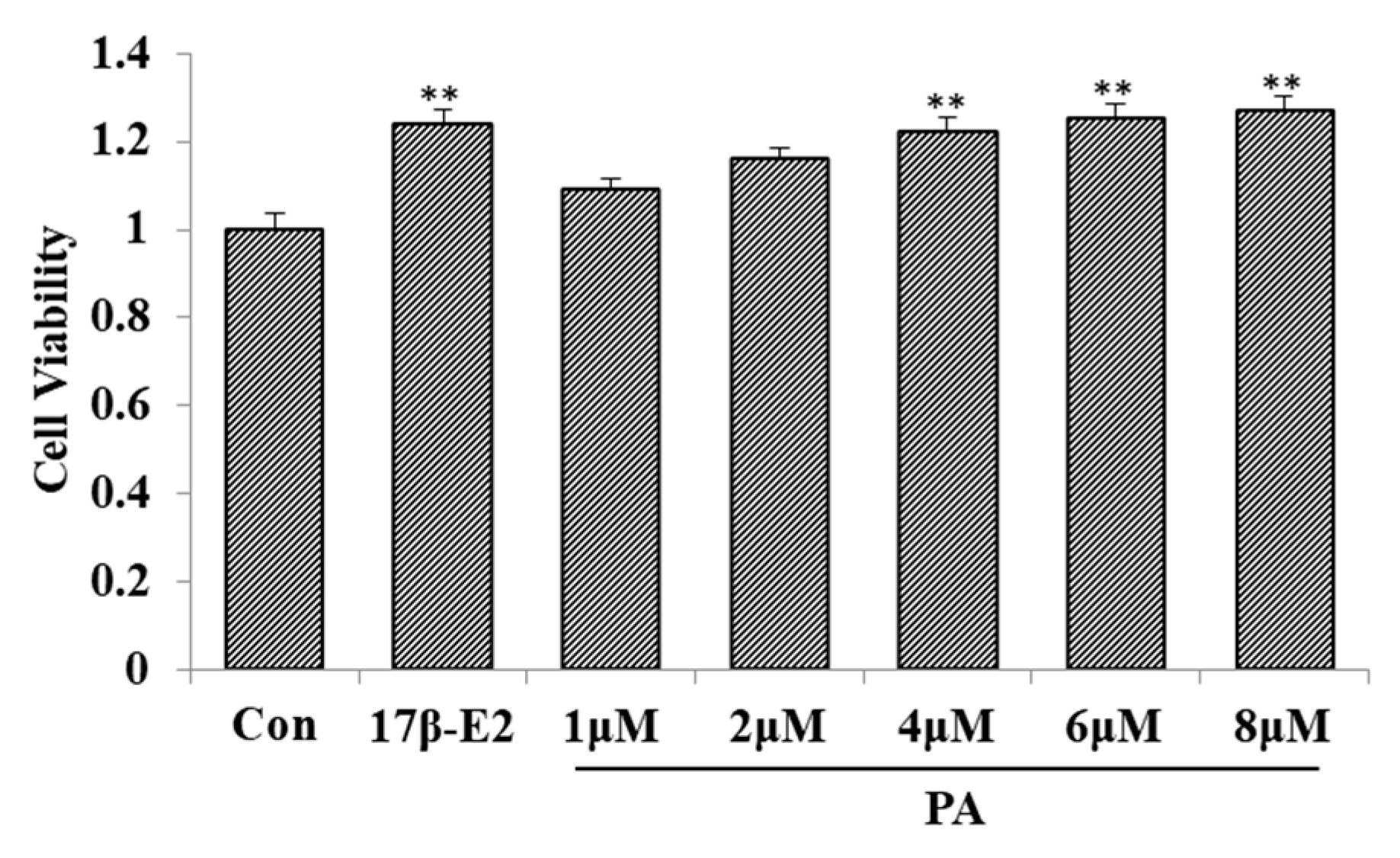
2.5. Effect of PA on MCF-7 Cell Proliferation
2.6. Effect of PA on MCF-7 Cell Proliferation
2.7. The Effect of PA on MCF-7 Cell Proliferation
2.8. Effect of Estrogen Receptor Antagonists on PA Aroused MCF-7 Cell Proliferation
3. Discussion
4. Materials and Methods
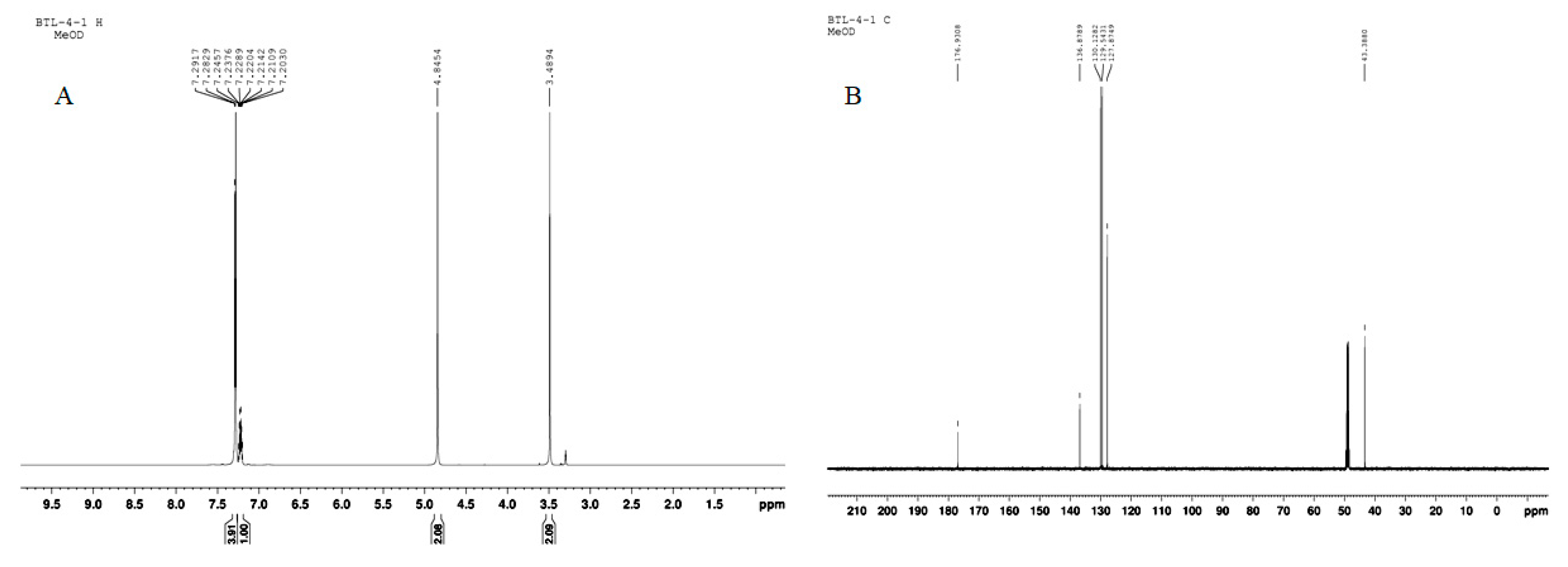
4.1. Plant Material
4.2. The Effects of LA and PA on MCF-7 Cell Proliferation
4.3. The Effects of ICI182780, MPP, THC and G15 on LA and PA Promoted MCF-7 Cell Proliferation
4.4. Animals
4.5. Western Blot
4.6. Enzyme-Linked Immuno Sorbent Assay
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PA | Phenylacetamide |
| LA | Lepidium apetalum Willd |
| ERT | Estrogen replacement therapy |
| TCM | Traditional Chinese medicine |
| PE | Phytoestrogen |
| MCF-7 cell | Breast adenocarcinoma cell line |
| LH | Luteinizing hormone |
| FSH | Follicle stimulating hormone |
| ICI182780 | Faslodex |
| MPP | Specific ERα antagonist methylpiperidino-pyrazole |
| THC | Specific ERβ antagonist Delta (9)-tetrahydrocannabinol |
| G15 | Specific GPR30 antagonist |
| EV | Estradiol valerate |
| TBST | Tris buffered saline with Tween-20 |
| MTT | Methyl thiazolyl tetrazolium |
| PBS | Phosphate buffer saline |
| DMSO | Dimethyl sulfoxide |
| 17β-E2 | 17-beta-estradiol |
| PVDF | Poly vinylidene fluoride |
| VEGF | Vascular endothelial growth factor |
References
- Kobayashi, N.; Fujino, T.; Shirogane, T.; Furuta, I.; Kobamatsu, Y.; Yaegashi, M.; Sakuragi, N.; Fujimoto, S. Estrogen receptor alpha polymorphism as a genetic marker for bone loss, vertebral fractures and susceptibility to estrogen. Maturitas 2002, 41, 193–201. [Google Scholar] [CrossRef]
- Dworatzek, E.; Mahmoodzadeh, S. Targeted basic research to highlight the role of estrogen and estrogen receptors in the cardiovascular system. Pharmacol. Res. 2017, 119, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Zhou, K.; Sun, P.; Zhang, Y.; You, X.; Li, P.; Wang, T. Estrogen stimulated migration and invasion of estrogen receptor-negative breast cancer cells involves an ezrin-dependent crosstalk between G protein-coupled receptor 30 and estrogen receptor beta signaling. Steroids 2016, 111, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.; Xu, W.; Li, H.; Chen, L.; Zhang, Y.; Tang, X. Extraction of Lepidium apetalum seed oil using supercritical carbon dioxide and anti-oxidant activity of the extracted oil. Molecules 2011, 16, 10029–10045. [Google Scholar] [PubMed]
- Shi, P.; Chao, L.; Wang, T.; Liu, E.; Han, L.; Zong, Q.; Li, X.; Zhang, Y.; Wang, T. New bioactive flavonoid glycosides isolated from the seeds of Lepidium apetalum Willd. Fitoterapia 2015, 103, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Hyun, J.W.; Shin, J.E.; Lim, K.H.; Sung, M.S.; Park, J.W.; Yu, J.H.; Kim, B.K.; Paik, W.H.; Kang, S.S.; Park, J.G. Evomonoside: The cytotoxic cardiac glycoside from Lepidium apetalum. Planta Med. 1995, 61, 294–295. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kim, H.Y.; Lee, Y.J.; Cui, H.Z.; Jang, J.Y.; Kang, D.G.; Lee, H.S. Ethanol extract of Lepidium apetalum seed elicits contractile response and attenuates atrial natriuretic peptide secretion in beating rabbit atria. Evid. Based Complement. Altern. Med. 2013, 2013, 404713. [Google Scholar] [CrossRef] [PubMed]
- Ren, C. Clinical observation on treatment of heart failure induced by chronicpneumocardial disease by Lepidium apetalum. Proc. Clin. Med. 2005, 14, 933. [Google Scholar]
- Zhang, W.; Zhang, Y.; Li, H. The application of Lepidium apetalum in treatment of chronic heart failure. World J. Integr. Tradit. West. Med. 2010, 2010, 349. [Google Scholar]
- Choi, H.; Ahn, S.; Lee, B.G.; Chang, I.; Hwang, J.S. Inhibition of skin pigmentation by an extract of Lepidium apetalum and its possible implication in IL-6 mediated signaling. Pigment Cell Res. 2005, 18, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Fantasia, H.C. A Nonhormonal Treatment for moderate to severe vasomotor symptoms of menopause. Nurs. Womens Health 2016, 20, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.S.; Zhang, Z.G.; Li, M.; Zhang, J.K.; Zhao, X.; Zheng, X.K.; Kuang, H.X. Chemical constituents from the seeds of Lepidium apetalum Willd. Acta Pharm. Sin. 2018, 53, 12–15. [Google Scholar]
- Webster, R.W. Aboriginal women and menopause. J. Obstet. Gynaecol. Can. 2002, 24, 938–940. [Google Scholar] [CrossRef]
- Sawka, A.M.; Huh, A.; Dolovich, L.; Papaioannou, A.; Eva, K.; Thabane, L.; Gafni, A.; Cullimore, A.; Macdougall, M.; Steiner, M. Attitudes of women who are currently using or recently stopped estrogen replacement therapy with or without progestins: Results of the AWARE survey. J. Obstet. Gynaecol. Can. 2004, 26, 967–973. [Google Scholar] [CrossRef]
- Seki, K.; Minami, Y.; Nishikawa, M.; Kawata, S.; Miyoshi, S.; Imai, Y.; Tarui, S. “Nonalcoholic steatohepatitis” induced by massive doses of synthetic estrogen. Gastroenterol. Jpn. 1983, 18, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Marks, K.J.; Hartman, T.J.; Taylor, E.V.; Rybak, M.E.; Northstone, K.; Marcus, M. Exposure to phytoestrogens in utero and age at menarche in a contemporary British cohort. Environ. Res. 2017, 155, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Ranich, T.; Bhathena, S.J.; Velasquez, M.T. Protective effects of dietary phytoestrogens in chronic renal disease. J. Ren. Nutr. 2001, 11, 183–193. [Google Scholar] [PubMed]
- Tabacova, S.; Kimmel, C.A.; Wall, K.; Hansen, D. Atenolol developmental toxicity: Animal-to-human comparisons. Birth Defects Res. A Clin. Mol. Teratol. 2003, 67, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Cheng, S.; Wei, D.; Ren, Y.; Zhang, D. Production of enantiomerically pure (S)-beta-phenylalanine and (R)-beta-phenylalanine by penicillin G acylase from Escherichia coli in aqueous medium. Biotechnol. Lett. 2007, 29, 1825–1830. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.T.; Lefkowitz, R.J. Regulation of rabbit myometrial alpha adrenergic receptors by estrogen and progesterone. J. Clin. Investig. 1977, 60, 815–818. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H. Coexistence of cytoplasmic and nuclear estrogen receptors. A histochemical study on human mammary cancer and rabbit uterus. Cancer 1989, 64, 1461–1466. [Google Scholar] [CrossRef]
- Carlson, R.A.; Gorski, J. Characterization of a unique population of unfilled estrogen-binding sites associated with the nuclear fraction of immature rat uteri. Endocrinology 1980, 106, 1776–1785. [Google Scholar] [CrossRef] [PubMed]
- Degani, H.; Victor, T.A.; Kaye, A.M. Effects of 17 beta-estradiol on high energy phosphate concentrations and the flux catalyzed by creatine kinase in immature rat uteri: 31P nuclear magnetic resonance studies. Endocrinology 1988, 122, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chung, M.H.; Xue, B. Estrogenic and antiestrogenic activities of phloridzin. Biol. Pharm. Bull. 2010, 33, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Dowsett, M.; Johnston, S.R.; Iveson, T.J.; Smith, I.E. Response to specific anti-oestrogen (ICI182780) in tamoxifen-resistant breast cancer. Lancet 1995, 345, 525–534. [Google Scholar] [CrossRef]
- Shennan, D.B.; Thomson, J. Estrogen regulation and ion dependence of taurine uptake by MCF-7 human breast cancer cells. Cell. Mol. Biol. Lett. 2007, 12, 396–406. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Motoya, E.; Matsuzawa, N.; Funahashi, T.; Kimura, T.; Matsunaga, T.; Arizono, K.; Yamamoto, I. Marijuana extracts possess the effects like the endocrine disrupting chemicals. Toxicology 2005, 206, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hong, D.Y.; Wang, J.; Ling-Hu, J.; Zhang, Y.Y.; Pan, D.; Xu, Y.N.; Tao, L.; Luo, H.; Shen, X.C. Baicalein, unlike 4-hydroxytamoxifen but similar to G15, suppresses 17β-estradiol-induced cell invasion, and matrix metalloproteinase-9 expression and activation in MCF-7 human breast cancer cells. Oncol. Lett. 2017, 14, 1823–1830. [Google Scholar] [CrossRef] [PubMed]
- Hastings, J.M.; Licence, D.R.; Burton, G.J.; Charnock-Jones, D.S.; Smith, S.K. Soluble vascular endothelial growth factor receptor 1 inhibits edema and epithelial proliferation induced by 17beta-estradiol in the mouse uterus. Endocrinology 2003, 144, 326–334. [Google Scholar] [PubMed]
Sample Availability: Not available. |
| Groups | Dose (mg/kg) | Uterus Coefficient (%) | LH (mIU/mL) | FSH (mIU/mL) | E2 (pmol/mL) |
|---|---|---|---|---|---|
| Con | — | 0.1019 ± 0.015 | 3.68 ± 0.69 | 40.11 ± 4.81 | 32.82 ± 2.53 |
| EV | 0.33 | 0.2795 ± 0.027 ** | 4.77 ± 0.74 ** | 46.91 ± 2.37 ** | 39.32 ± 2.10 ** |
| LA-L | 446 | 0.1262 ± 0.021 * | 5.22 ± 0.35 ** | 43.24 ± 4.36 | 45.06 ± 6.65 ** |
| LA-H | 892 | 0.1259 ± 0.022 * | 5.42 ± 0.44 ** | 43.04 ± 5.26 | 46.11 ± 5.78 ** |
| Groups | Dose (mg/kg) | Uterus Coefficient (%) | LH (mIU/mL) | FSH (mIU/mL) | E2 (pmol/mL) |
|---|---|---|---|---|---|
| Con | — | 0.1009 ± 0.014 | 4.01 ± 0.58 | 39.77 ± 5.12 | 33.01 ± 2.14 |
| EV | 0.33 | 0.2395 ± 0.027 ** | 5.11 ± 0.49 ** | 46.91 ± 3.27 ** | 38.41 ± 2.07 ** |
| LA | 446 | 0.1253 ± 0.021 * | 5.09 ± 0.61 ** | 42.37 ± 4.12 | 44.36 ± 4.37 ** |
| PA-L | 25 | 0.1219 ± 0.021 * | 4.98 ± 0.47 ** | 42.34 ± 5.26 | 41.19 ± 5.57 ** |
| PA-H | 50 | 0.1201 ± 0.019 * | 5.02 ± 0.79 ** | 41.14 ± 6.78 | 40.56 ± 6.01 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, M.; Li, M.; Li, M.; Zhang, B.; Li, B.; Zhang, L.; Feng, W.; Zheng, X. 2-Phenylacetamide Isolated from the Seeds of Lepidium apetalum and Its Estrogen-Like Effects In Vitro and In Vivo. Molecules 2018, 23, 2293. https://doi.org/10.3390/molecules23092293
Zeng M, Li M, Li M, Zhang B, Li B, Zhang L, Feng W, Zheng X. 2-Phenylacetamide Isolated from the Seeds of Lepidium apetalum and Its Estrogen-Like Effects In Vitro and In Vivo. Molecules. 2018; 23(9):2293. https://doi.org/10.3390/molecules23092293
Chicago/Turabian StyleZeng, Mengnan, Meng Li, Miao Li, Beibei Zhang, Benke Li, Li Zhang, Weisheng Feng, and Xiaoke Zheng. 2018. "2-Phenylacetamide Isolated from the Seeds of Lepidium apetalum and Its Estrogen-Like Effects In Vitro and In Vivo" Molecules 23, no. 9: 2293. https://doi.org/10.3390/molecules23092293
APA StyleZeng, M., Li, M., Li, M., Zhang, B., Li, B., Zhang, L., Feng, W., & Zheng, X. (2018). 2-Phenylacetamide Isolated from the Seeds of Lepidium apetalum and Its Estrogen-Like Effects In Vitro and In Vivo. Molecules, 23(9), 2293. https://doi.org/10.3390/molecules23092293