Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling
Abstract
1. Introduction
2. Results
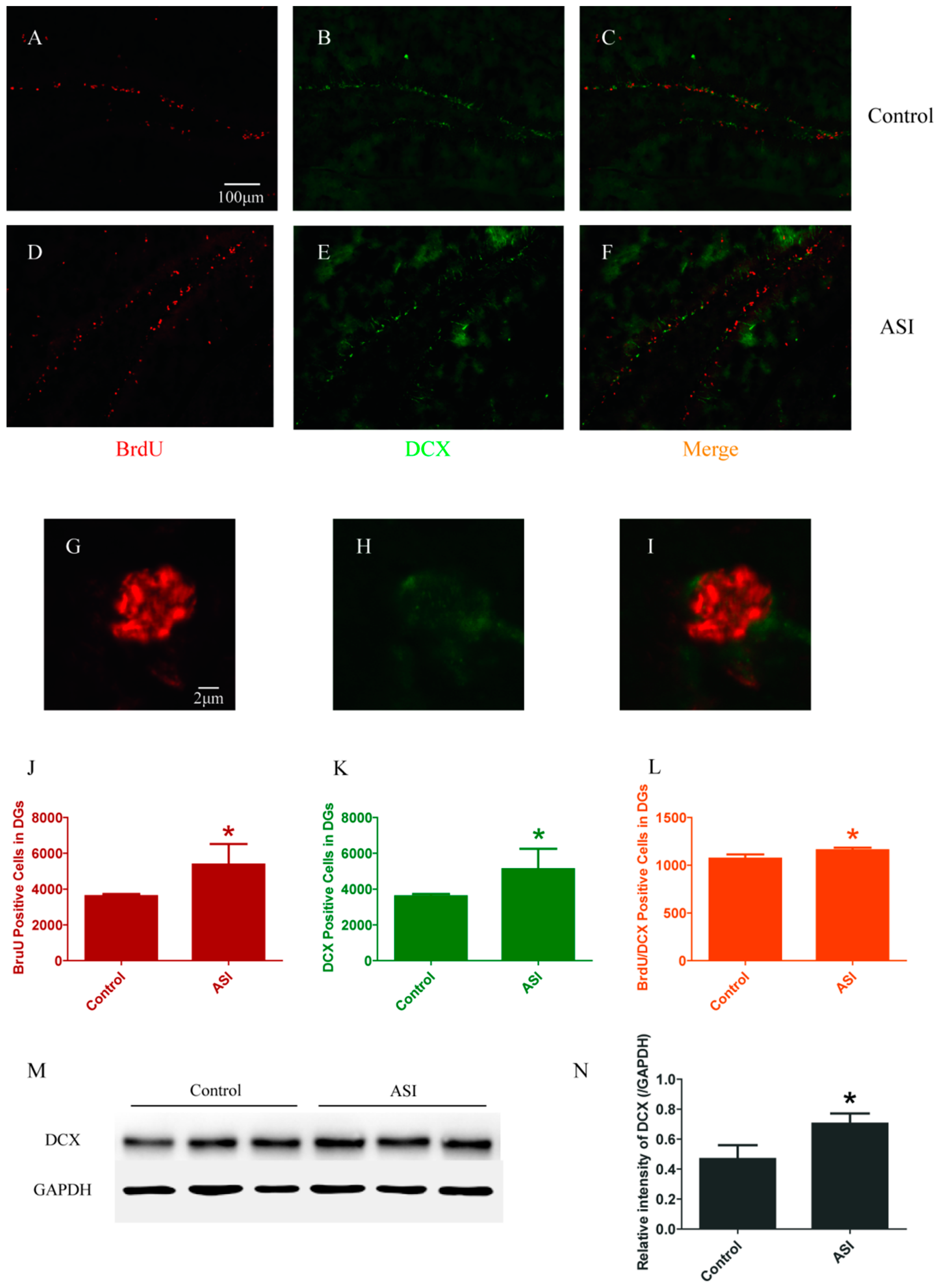
2.1. ASI Increased the Total Number of Proliferative Cells (BrdU+), Pre-Mature Neurons (DCX+) and Early Proliferative Cells (BrdU+/DCX+)
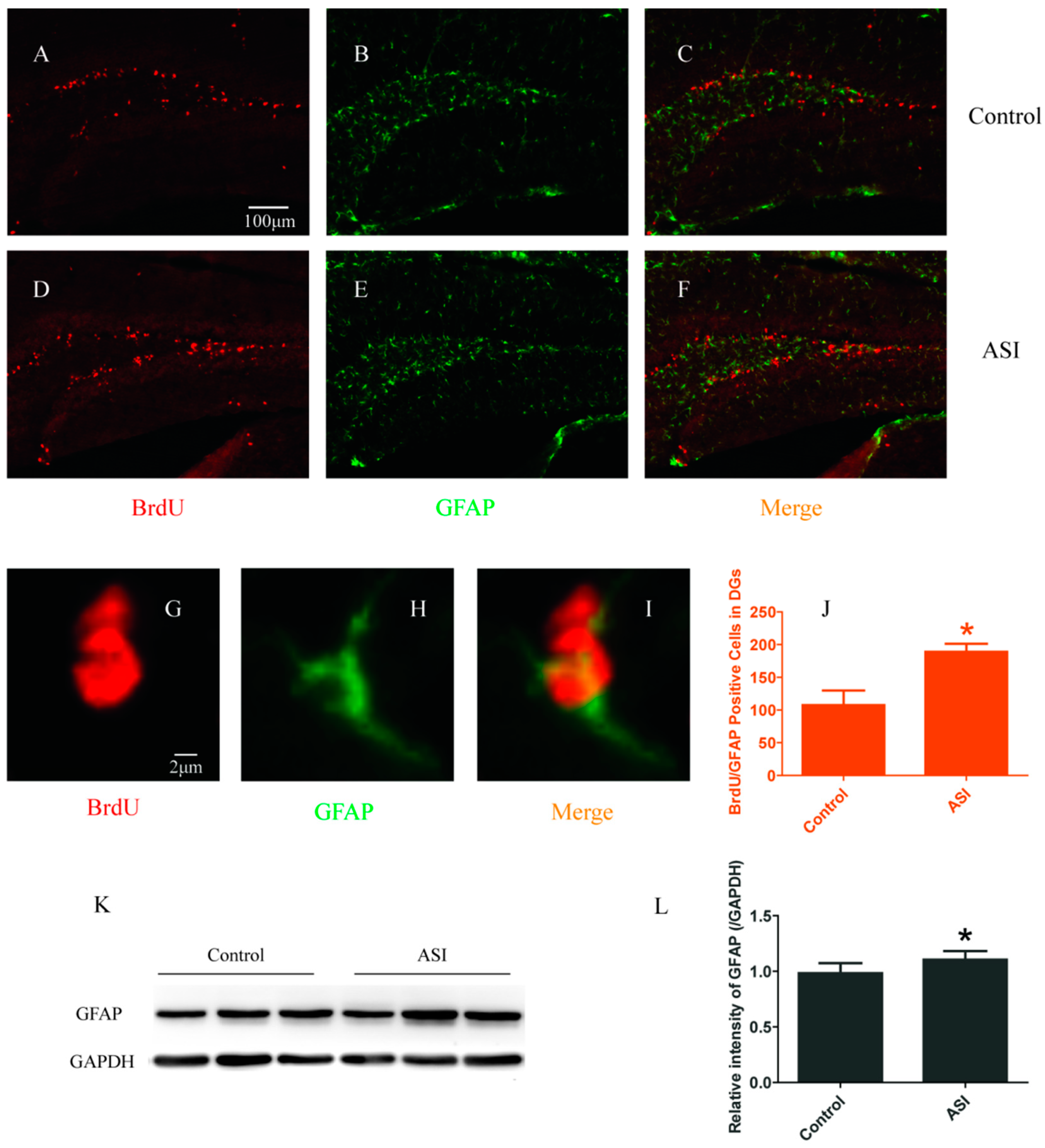
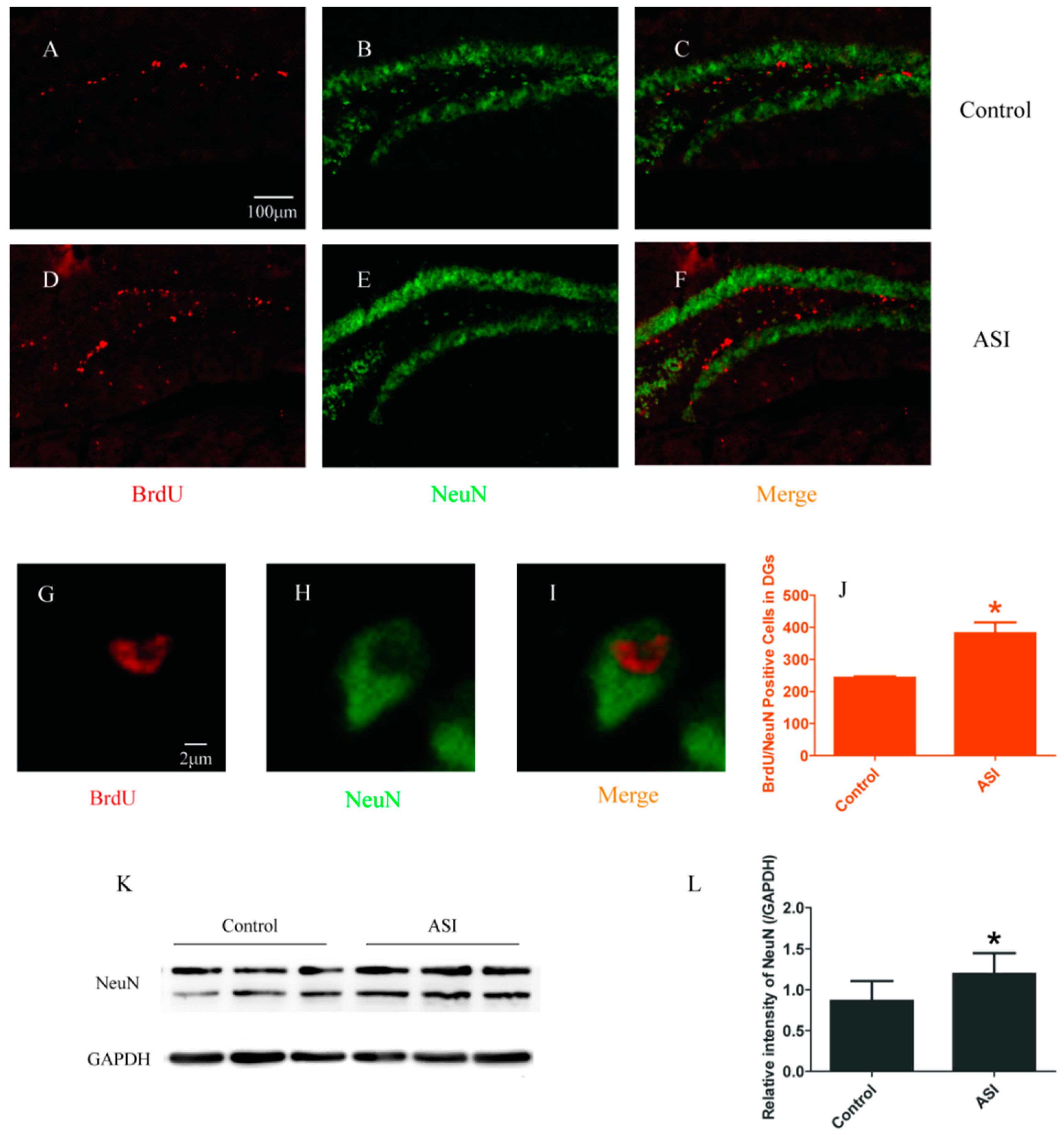
2.2. ASI Increased the Total Number of Proliferative Radial Glia-Like Cells (BrdU+/GFAP+) and Newly Generated Neurons (BrdU+/NeuN+)
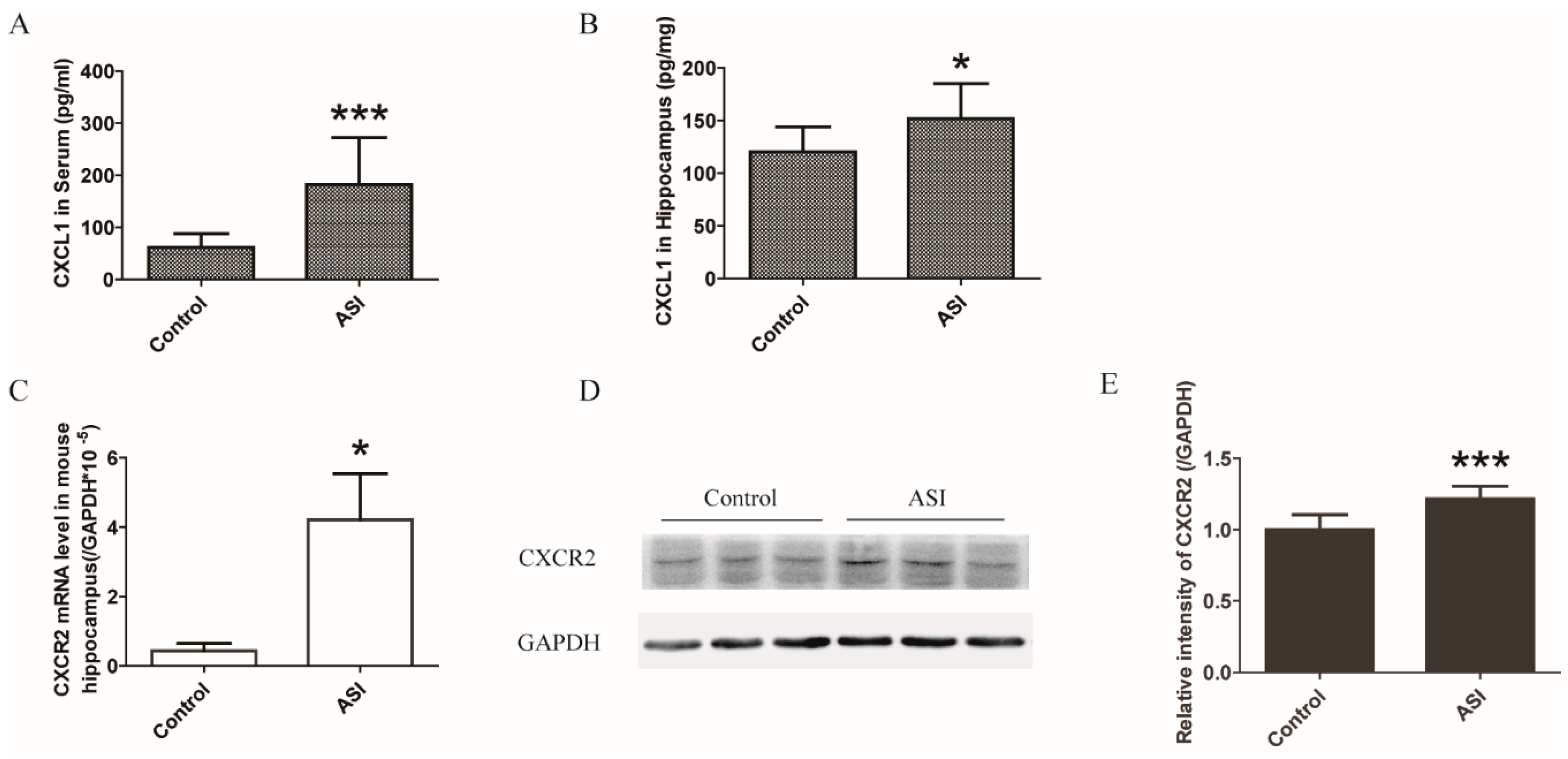
2.3. ASI Enhanced CXCL1/CXCR2 Signaling Pathway
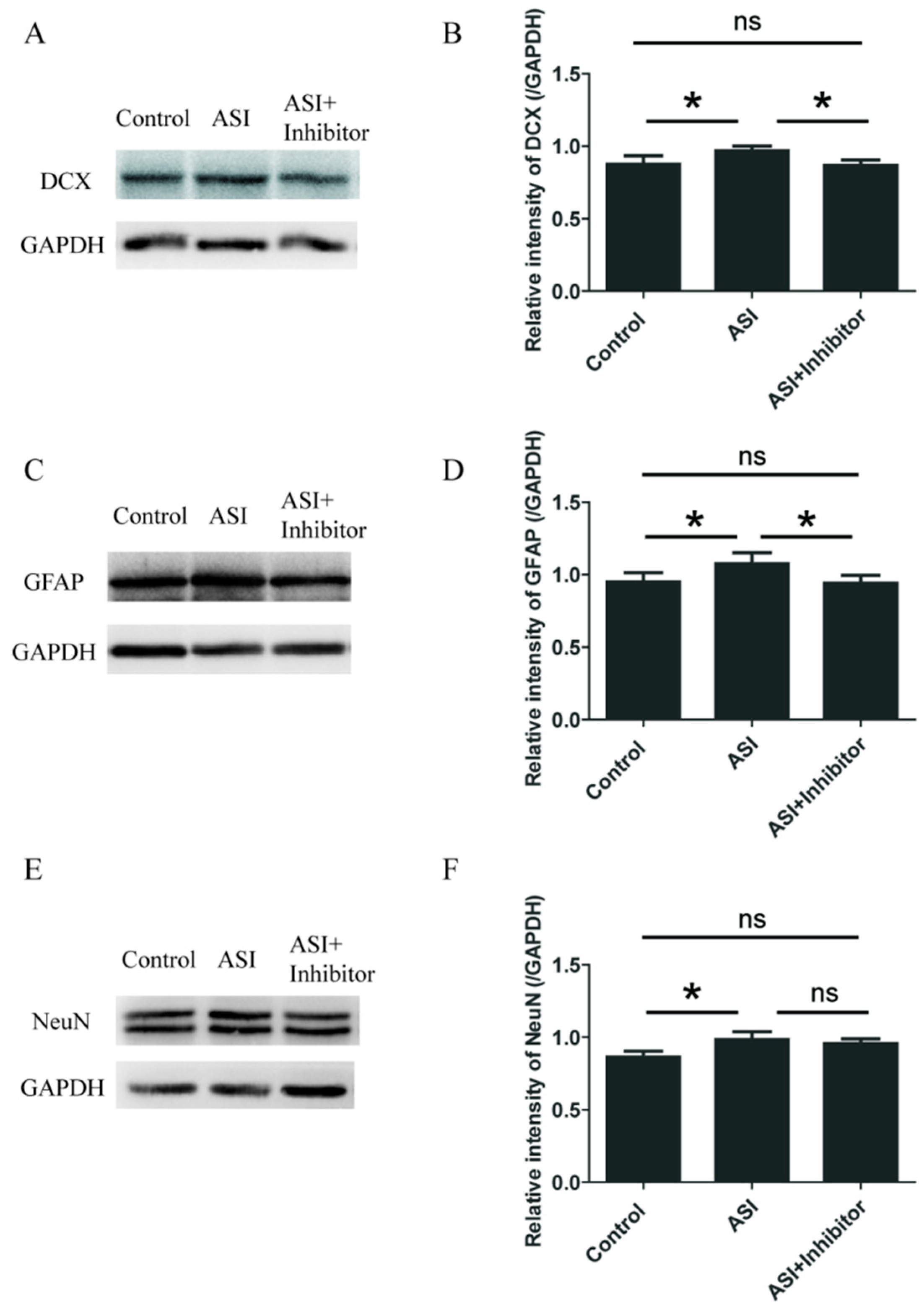
2.4. ASI Promoted Neurogenesis through CXCL1/CXCR2 Signaling Pathway
3. Discussion
4. Materials and Methods
4.1. Animal and Drug Administration
4.2. Immunohistochemistry
4.3. RT2 Profiler PCR Array Analysis
4.4. Real-Time PCR
4.5. Western Blotting Analysis
4.6. ELISA Analysis
4.7. Statistical Analysis
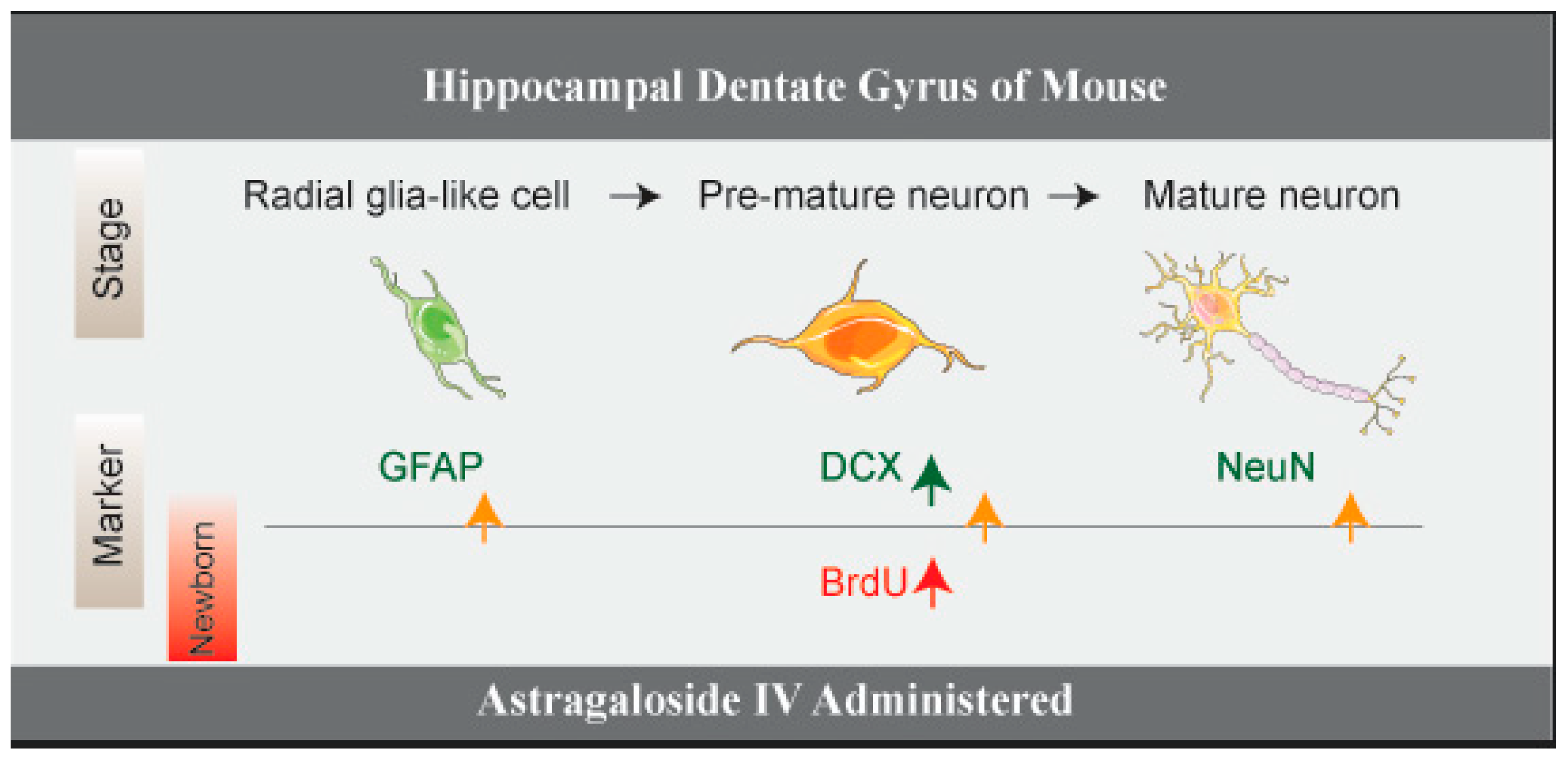
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kempermann, G.; Song, H.; Gage, F.H. Neurogenesis in the adult hippocampus. CSH Perspect. Med. 2015, 5, a018812. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, H.G.; Dickinson-Anson, H.; Gage, F.H. Neurogenesis in the dentate gyrus of the adult rat: Age-related decrease of neuronal progenitor proliferation. J. Neurosci. Off. Soc. Neurosci. 1996, 16, 2027–2033. [Google Scholar] [CrossRef]
- Ramirez-Amaya, V.; Marrone, D.F.; Gage, F.H.; Worley, P.F.; Barnes, C.A. Integration of new neurons into functional neural networks. J. Neurosci. Off. Soc. Neurosci. 2006, 26, 12237–12241. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.H.; Kim, Y.N.; Kim, I.Y.; Choi, D.H.; Yi, S.S.; Seong, J.K. Increased cell proliferations and neurogenesis in the hippocampal dentate gyrus of ahnak deficient mice. Neurochem. Res. 2015, 40, 1457–1462. [Google Scholar] [CrossRef] [PubMed]
- Dupret, D.; Revest, J.M.; Koehl, M.; Ichas, F.; De Giorgi, F.; Costet, P.; Abrous, D.N.; Piazza, P.V. Spatial relational memory requires hippocampal adult neurogenesis. PLoS ONE 2008, 3, e1959. [Google Scholar] [CrossRef] [PubMed]
- Imayoshi, I.; Sakamoto, M.; Ohtsuka, T.; Takao, K.; Miyakawa, T.; Yamaguchi, M.; Mori, K.; Ikeda, T.; Itohara, S.; Kageyama, R. Roles of continuous neurogenesis in the structural and functional integrity of the adult forebrain. Nat. Neurosci. 2008, 11, 1153–1161. [Google Scholar] [CrossRef] [PubMed]
- Saxe, M.D.; Battaglia, F.; Wang, J.W.; Malleret, G.; David, D.J.; Monckton, J.E.; Garcia, A.D.; Sofroniew, M.V.; Kandel, E.R.; Santarelli, L.; et al. Ablation of hippocampal neurogenesis impairs contextual fear conditioning and synaptic plasticity in the dentate gyrus. Proc. Natl. Acad. Sci. USA 2006, 103, 17501–17506. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, T.; Matsushima, K.; Tanaka, S.; Robinson, E.A.; Appella, E.; Oppenheim, J.J.; Leonard, E.J. Purification of a human monocyte-derived neutrophil chemotactic factor that has peptide sequence similarity to other host defense cytokines. Proc. Natl. Acad. Sci. USA 1987, 84, 9233–9237. [Google Scholar] [CrossRef] [PubMed]
- Bonavia, R.; Bajetto, A.; Barbero, S.; Pirani, P.; Florio, T.; Schettini, G. Chemokines and their receptors in the cns: Expression of cxcl12/sdf-1 and cxcr4 and their role in astrocyte proliferation. Toxicol. Lett. 2003, 139, 181–189. [Google Scholar] [CrossRef]
- Araujo, D.M.; Cotman, C.W. Trophic effects of interleukin-4, -7 and -8 on hippocampal neuronal cultures: Potential involvement of glial-derived factors. Brain Res. 1993, 600, 49–55. [Google Scholar] [CrossRef]
- Hesselgesser, J.; Horuk, R. Chemokine and chemokine receptor expression in the central nervous system. J. Neurovirol. 1999, 5, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.H.; Frost, E.; To, V.; Robinson, S.; Ffrench-Constant, C.; Geertman, R.; Ransohoff, R.M.; Miller, R.H. The chemokine receptor cxcr2 controls positioning of oligodendrocyte precursors in developing spinal cord by arresting their migration. Cell 2002, 110, 373–383. [Google Scholar] [CrossRef]
- Fu, J.; Wang, Z.; Huang, L.; Zheng, S.; Wang, D.; Chen, S.; Zhang, H.; Yang, S. Review of the botanical characteristics, phytochemistry, and pharmacology of Astragalus membranaceus (HUANGQI). Phytother. Res. PTR 2014, 28, 1275–1283. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.G.; Wu, L.; Wang, J.L.; Yang, J.D.; Zhang, J.; Zhang, J.; Li, L.H.; Xia, Y.; Yao, L.B.; Qin, H.Z.; et al. Astragaloside iv prevents mpp(+)-induced sh-sy5y cell death via the inhibition of bax-mediated pathways and ros production. Mol. Cell. Biochem. 2012, 364, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, X.; Han, C.; Wang, X.; Xing, G.; Zhou, L.; Li, G.; Niu, Y. Astragaloside iv suppresses collagen production of activated hepatic stellate cells via oxidative stress-mediated p38 mapk pathway. Free Radic. Biol. Med. 2013, 60, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.Y.; Han, J.; Chu, Z.G.; Song, H.P.; Zhang, D.X.; Zhang, Q.; Huang, Y.S. Astragaloside iv attenuates hypoxia-induced cardiomyocyte damage in rats by upregulating superoxide dismutase-1 levels. Clin. Exp. Pharmacol. Physiol. 2009, 36, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Qin, Z.; Hong, Z.; Zhang, X.; Ding, D.; Fu, J.H.; Zhang, W.D.; Chen, J. Astragaloside iv protects against ischemic brain injury in a murine model of transient focal ischemia. Neurosci. Lett. 2004, 363, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Qu, Y.Z.; Zhao, Z.W.; Wu, S.X.; Liu, Y.Y.; Wei, X.Y.; Gao, L.; Gao, G.D. Astragaloside iv protects against focal cerebral ischemia/reperfusion injury correlating to suppression of neutrophils adhesion-related molecules. Neurochem. Int. 2012, 60, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Song, J.; Guo, Y.; Wang, T.; Zhou, Z. Astragalus injection protects cerebral ischemic injury by inhibiting neuronal apoptosis and the expression of jnk3 after cerebral ischemia reperfusion in rats. BBF 2013, 9, 36. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.Z.; Li, M.; Zhao, Y.L.; Zhao, Z.W.; Wei, X.Y.; Liu, J.P.; Gao, L.; Gao, G.D. Astragaloside iv attenuates cerebral ischemia-reperfusion-induced increase in permeability of the blood-brain barrier in rats. Eur. J. Pharmacol. 2009, 606, 137–141. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Du, M.; Gao, Y.; Liu, H.; Wang, H.; Wu, X.; Wang, Z. Astragaloside iv attenuates experimental autoimmune encephalomyelitis of mice by counteracting oxidative stress at multiple levels. PLoS ONE 2013, 8, e76495. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kang, I.H.; Nam, J.B.; Cho, Y.; Chung, D.Y.; Kim, S.H.; Kim, J.S.; Cho, Y.D.; Hong, E.K.; Sohn, N.W.; et al. Ameliorating the effect of astragaloside iv on learning and memory deficit after chronic cerebral hypoperfusion in rats. Molecules 2015, 20, 1904–1921. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, H.; Fang, F.; Deng, X.; Ma, S. Astragaloside IV attenuates cognitive impairments induced by transient cerebral ischemia and reperfusion in mice via anti-inflammatory mechanisms. Neurosci. Lett. 2017, 639, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Yang, R.; Jin, G.; Zhang, X.; Xia, H.; Xu, Y. Effect of astragaloside IV on neural stem cell transplantation in alzheimer’s disease rat models. Evid.-Based Complement. Altern. Med. 2016, 2016, 3106980. [Google Scholar]
- Huang, P.; Lu, X.; Yuan, B.; Liu, T.; Dai, L.; Liu, Y.; Yin, H. Astragaloside iv alleviates e. Coli-caused peritonitis via upregulation of neutrophil influx to the site of infection. Int. Immunopharmacol. 2016, 39, 377–382. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Aimone, J.B.; Gage, F.H. New neurons and new memories: How does adult hippocampal neurogenesis affect learning and memory? Nat. Rev. Neurosci. 2010, 11, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.; Tani, M.; Strieter, R.M.; Ransohoff, R.M.; Miller, R.H. The chemokine growth-regulated oncogene-alpha promotes spinal cord oligodendrocyte precursor proliferation. J. Neurosci. Off. J. Soc. Neurosci. 1998, 18, 10457–10463. [Google Scholar] [CrossRef]
- Wu, Q.; Miller, R.H.; Ransohoff, R.M.; Robinson, S.; Bu, J.; Nishiyama, A. Elevated levels of the chemokine gro-1 correlate with elevated oligodendrocyte progenitor proliferation in the jimpy mutant. J. Neurosci. Off. J. Soc. Neurosci. 2000, 20, 2609–2617. [Google Scholar] [CrossRef]
- Edman, L.C.; Mira, H.; Erices, A.; Malmersjo, S.; Andersson, E.; Uhlen, P.; Arenas, E. Alpha-chemokines regulate proliferation, neurogenesis, and dopaminergic differentiation of ventral midbrain precursors and neurospheres. Stem Cells 2008, 26, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Valles, A.; Grijpink-Ongering, L.; de Bree, F.M.; Tuinstra, T.; Ronken, E. Differential regulation of the cxcr2 chemokine network in rat brain trauma: Implications for neuroimmune interactions and neuronal survival. Neurobiol. Dis. 2006, 22, 312–322. [Google Scholar] [CrossRef] [PubMed]
- Watson, K.; Fan, G.H. Macrophage inflammatory protein 2 inhibits beta-amyloid peptide (1-42)-mediated hippocampal neuronal apoptosis through activation of mitogen-activated protein kinase and phosphatidylinositol 3-kinase signaling pathways. Mol. Pharmacol. 2005, 67, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.Y.; Tu, Y.F.; Lin, Y.C.; Huang, C.C. Cxcl5 signaling is a shared pathway of neuroinflammation and blood-brain barrier injury contributing to white matter injury in the immature brain. J. Neuroinflam. 2016, 13, 6. [Google Scholar] [CrossRef] [PubMed]
- Filipkowski, R.K.; Kaczmarek, L. Severely impaired adult brain neurogenesis in cyclin d2 knock-out mice produces very limited phenotypic changes. Prog. Neuro-Psychopharm. Biol. Psychiatry 2018, 80, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Franklin, K.B.J.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates, 2nd ed.; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
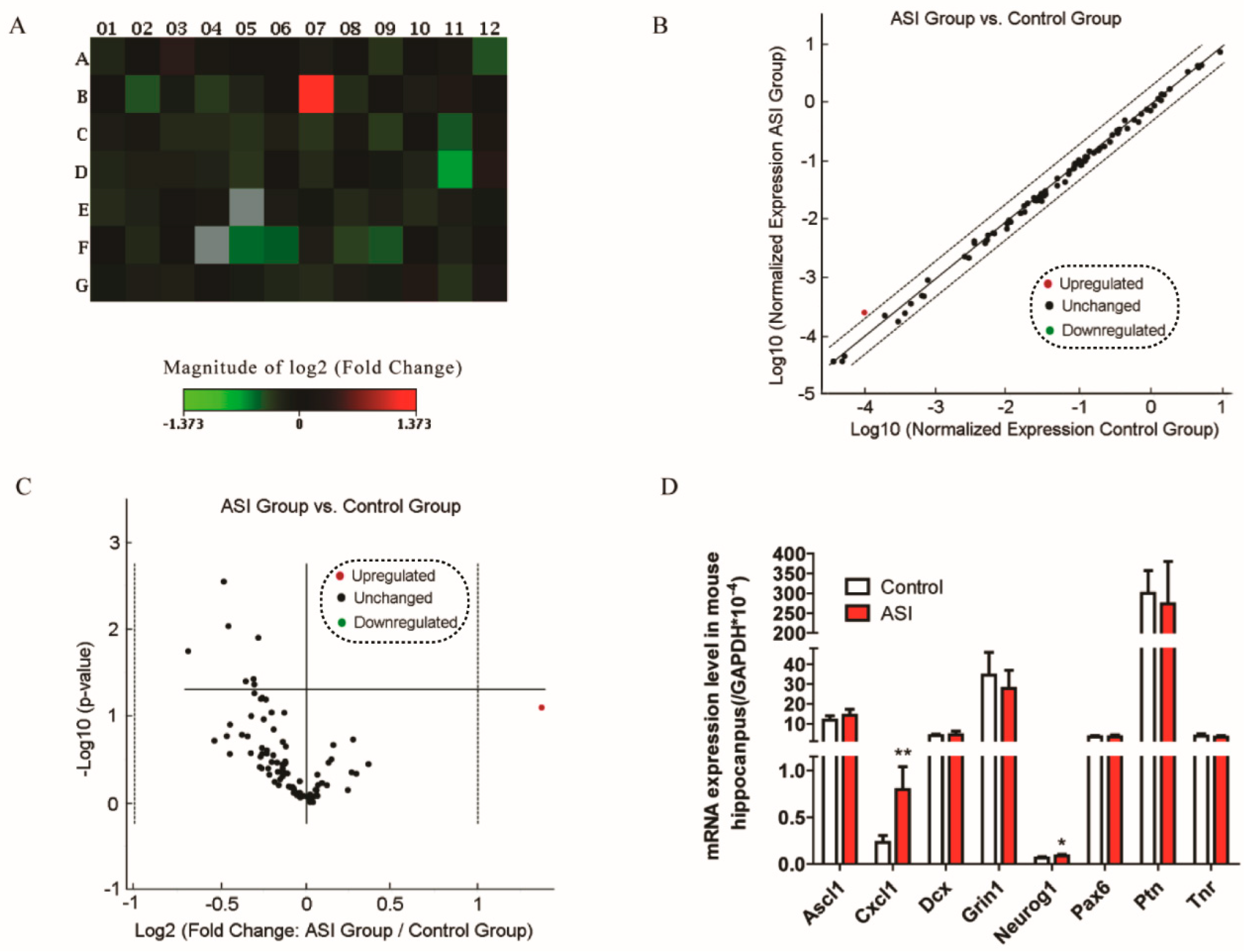
| Description | Symbol | Fold Change | p-Value |
|---|---|---|---|
| Acetylcholinesterase | Ache | 0.857 | 0.408505 |
| Adenosine A1 receptor | Adora1 | 1.016 | 0.958737 |
| Adenosine A2a receptor | Adora2a | 1.2858 | 0.364259 |
| Anaplastic lymphoma kinase | Alk | 0.9632 | 0.831172 |
| Amyloid beta (A4) precursor protein-binding, family B, member 1 | Apbb1 | 1.0156 | 0.930706 |
| Apolipoprotein E | Apoe | 1.0062 | 0.874886 |
| Amyloid beta (A4) precursor protein | App | 0.9095 | 0.20132 |
| Artemin | Artn | 1.0458 | 0.845847 |
| Achaete-scute complex homolog 1 (Drosophila) | Ascl1 | 0.808 | 0.038118 |
| B-cell leukemia/lymphoma 2 | Bcl2 | 1.0486 | 0.632784 |
| Brain derived neurotrophic factor | Bdnf | 0.9419 | 0.678017 |
| Bone morphogenetic protein 2 | Bmp2 | 0.734 | 0.277842 |
| Bone morphogenetic protein 4 | Bmp4 | 1.0426 | 0.725701 |
| Bone morphogenetic protein 8b | Bmp8b | 0.7345 | 0.127788 |
| Cyclin-dependent kinase 5, regulatory subunit 1 (p35) | Cdk5r1 | 0.9182 | 0.33772 |
| CDK5 regulatory subunit associated protein 2 | Cdk5rap2 | 0.7886 | 0.173978 |
| Cholinergic receptor, muscarinic 2, cardiac | Chrm2 | 0.8612 | 0.481537 |
| CAMP responsive element binding protein 1 | Creb1 | 1.0151 | 0.998111 |
| Chemokine (C-X-C motif) ligand 1 | Cxcl1 | 2.5899 | 0.081082 |
| Doublecortin | Dcx | 0.8235 | 0.012748 |
| Discs, large homolog 4 (Drosophila) | Dlg4 | 0.9898 | 0.847781 |
| Delta-like 1 (Drosophila) | Dll1 | 0.9084 | 0.501594 |
| Dopamine receptor D2 | Drd2 | 1.1828 | 0.723812 |
| Dishevelled 3, dsh homolog (Drosophila) | Dvl3 | 1.0458 | 0.480882 |
| Ephrin B1 | Efnb1 | 0.9166 | 0.35916 |
| Epidermal growth factor | Egf | 0.9504 | 0.69603 |
| E1A binding protein p300 | Ep300 | 0.8368 | 0.062456 |
| V-erb-b2 erythroblastic leukemia viral oncogene homolog 2, neuro/glioblastoma derived oncogene homolog (avian) | Erbb2 | 0.8359 | 0.236367 |
| Fibroblast growth factor 2 | Fgf2 | 0.8105 | 0.055528 |
| Filamin, alpha | Flna | 0.8888 | 0.349493 |
| Glial cell line derived neurotrophic factor | Gdnf | 0.7991 | 0.271871 |
| Glucose phosphate isomerase 1 | Gpi1 | 0.946 | 0.668135 |
| Glutamate receptor, ionotropic, NMDA1 (zeta 1) | Grin1 | 0.7829 | 0.040383 |
| Histone deacetylase 4 | Hdac4 | 1.0155 | 0.813732 |
| Hairy and enhancer of split 1 (Drosophila) | Hes1 | 0.725 | 0.173203 |
| Hairy/enhancer-of-split related with YRPW motif 1 | Hey1 | 1.1153 | 0.21879 |
| Hairy/enhancer-of-split related with YRPW motif 2 | Hey2 | 0.8526 | 0.251846 |
| Hairy/enhancer-of-split related with YRPW motif-like | Heyl | 0.8945 | 0.635531 |
| Interleukin 3 | Il3 | 0.88 | 0.582914 |
| Midkine | Mdk | 0.8764 | 0.145008 |
| Myocyte enhancer factor 2C | Mef2c | 0.8003 | 0.101506 |
| Myeloid/lymphoid or mixed-lineage leukemia 1 | Kmt2a | 1.0275 | 0.997062 |
| Microtubule-associated protein 2 | Map2 | 0.8362 | 0.276201 |
| Necdin | Ndn | 1.0382 | 0.718334 |
| Norrie disease (pseudoglioma) (human) | Ndp | 0.9318 | 0.657985 |
| Neurogenic differentiation 1 | Neurod1 | 0.8694 | 0.344025 |
| Neurogenin 1 | Neurog1 | 0.62 | 0.018182 |
| Neurogenin 2 | Neurog2 | 1.2238 | 0.468483 |
| Neurofibromatosis 1 | Nf1 | 0.8321 | 0.06433 |
| Noggin | Nog | 0.875 | 0.288031 |
| Notch gene homolog 1 (Drosophila) | Notch1 | 1.0654 | 0.599656 |
| Notch gene homolog 2 (Drosophila) | Notch2 | 0.9508 | 0.77945 |
| Nuclear receptor subfamily 2, group E, member 3 | Nr2e3 | 1.0185 | 0.936243 |
| Neuron-glia-CAM-related cell adhesion molecule | Nrcam | 0.9158 | 0.460346 |
| Neuregulin 1 | Nrg1 | 0.9564 | 0.765114 |
| Neuropilin 1 | Nrp1 | 0.835 | 0.406853 |
| Neuropilin 2 | Nrp2 | 0.9089 | 0.406554 |
| Neurotrophin 3 | Ntf3 | 0.831 | 0.299471 |
| Netrin 1 | Ntn1 | 0.9076 | 0.44445 |
| Odd Oz/ten-m homolog 1 (Drosophila) | Tenm1 | 0.9365 | 0.665725 |
| Oligodendrocyte transcription factor 2 | Olig2 | 1.0327 | 0.853541 |
| Platelet-activating factor acetylhydrolase, isoform 1b, subunit 1 | Pafah1b1 | 0.8419 | 0.110867 |
| Par-3 (partitioning defective 3) homolog (C. elegans) | Pard3 | 0.976 | 0.878636 |
| Paired box gene 3 | Pax3 | 1.0185 | 0.936243 |
| Paired box gene 5 | Pax5 | 0.6895 | 0.195764 |
| Paired box gene 6 | Pax6 | 0.7163 | 0.002868 |
| POU domain, class 3, transcription factor 3 | Pou3f3 | 0.8996 | 0.542927 |
| POU domain, class 4, transcription factor 1 | Pou4f1 | 0.7709 | 0.167186 |
| Pleiotrophin | Ptn | 0.7296 | 0.009374 |
| RAS-related C3 botulinum substrate 1 | Rac1 | 0.9743 | 0.778785 |
| Roundabout homolog 1 (Drosophila) | Robo1 | 0.8688 | 0.092542 |
| Reticulon 4 | Rtn4 | 1.0437 | 0.826015 |
| S100 calcium binding protein A6 (calcyclin) | S100a6 | 0.9265 | 0.466286 |
| S100 protein, beta polypeptide, neural | S100b | 0.9084 | 0.453236 |
| Sonic hedgehog | Shh | 0.8914 | 0.445715 |
| Slit homolog 2 (Drosophila) | Slit2 | 1.0868 | 0.636415 |
| Superoxide dismutase 1, soluble | Sod1 | 0.9206 | 0.228334 |
| SRY-box containing gene 2 | Sox2 | 0.8523 | 0.274519 |
| SRY-box containing gene 3 | Sox3 | 0.8288 | 0.392865 |
| Signal transducer and activator of transcription 3 | Stat3 | 0.9089 | 0.366775 |
| Transforming growth factor, beta 1 | Tgfb1 | 0.9192 | 0.351884 |
| Tyrosine hydroxylase | Th | 1.2009 | 0.452233 |
| Tenascin R | Tnr | 0.8102 | 0.04399 |
| Vascular endothelial growth factor A | Vegfa | 1.107 | 0.320206 |
| Gene | Forward Primer | Reverse Primer |
|---|---|---|
| Ascl1 | GTCACAAGTCAGCGGCCAAGCA | TTCTTGTTGGCCGCGCCGTT |
| CXCL1 | GCCAATGAGCTGCGCTGTCAGT | AAGGCAAGCCTCGCGACCATTC |
| CXCR2 | ATGCTGTCCCATGCCACTCAGAGA | CCATTTACTTTAGATGCAGCCCAGACA |
| Dcx | CATCTAGAAATATGAGAGGGTCACGGATG | TCTTCCAGTTCATCCATGCTTCCAAT |
| Neurog1 | CCTCTCCGGGGCATCGAATGTT | TGAGCTTGGTGTCGTCGGGGAA |
| GAPDH | ATGTGTCCGTCGTGGATCTGA | ATGCCTGCTTCACCACCTTCT |
| Grin1 | CAAGCCCAACGCCATACAGATGG | AGCAACGTCTCCAGGCGCTTCT |
| Pax6 | CCAGGGCAATCGGAGGGAGTAA | CGCCCATCTGTTGCTTTTCGCTA |
| Ptn | GCAACGTAGAAAATTTGCAGCTGCCTTC | TCTCTGAGTCTTCATGGTCTGTTTGCAC |
| Tnr | AGGTGACTACAGAAAGGGCTCAGAGACA | GCTCAGCAGTTCCTGCAGTACCTGG |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, F.; Lan, Y.; Qin, L.; Dong, H.; Shi, H.; Wu, H.; Zou, Q.; Hu, Z.; Wu, X. Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling. Molecules 2018, 23, 2178. https://doi.org/10.3390/molecules23092178
Huang F, Lan Y, Qin L, Dong H, Shi H, Wu H, Zou Q, Hu Z, Wu X. Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling. Molecules. 2018; 23(9):2178. https://doi.org/10.3390/molecules23092178
Chicago/Turabian StyleHuang, Fei, Yunyi Lan, Liyue Qin, Huaihuai Dong, Hailian Shi, Hui Wu, Qinrui Zou, Zhibi Hu, and Xiaojun Wu. 2018. "Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling" Molecules 23, no. 9: 2178. https://doi.org/10.3390/molecules23092178
APA StyleHuang, F., Lan, Y., Qin, L., Dong, H., Shi, H., Wu, H., Zou, Q., Hu, Z., & Wu, X. (2018). Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling. Molecules, 23(9), 2178. https://doi.org/10.3390/molecules23092178




