Antischistosomal Properties of Hederacolchiside A1 Isolated from Pulsatilla chinensis
Abstract
1. Introduction
2. Results
2.1. HSA Has Antischistosomal Activity against Juvenile and Adult S. japonicum
2.2. HSA Was Superior in Inhibiting S. japonicum Less than 11 Days Old
2.3. The Efficacy Advantage of HSA against 1-Day-Old and 7-Day-Old Juvenile S. mansoni
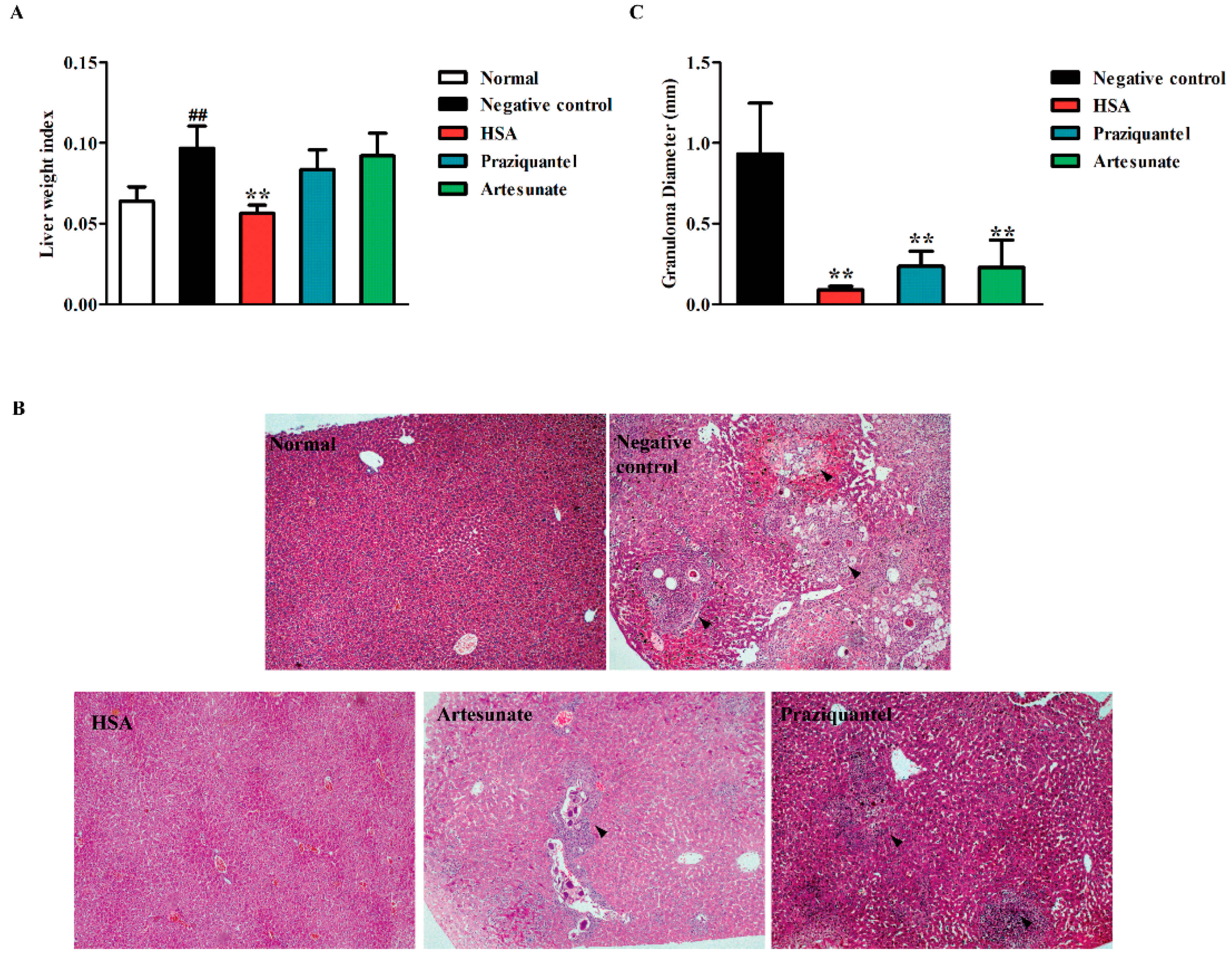
2.4. HSA Inhibited Liver Damage in S. japonicum-Infected Mice
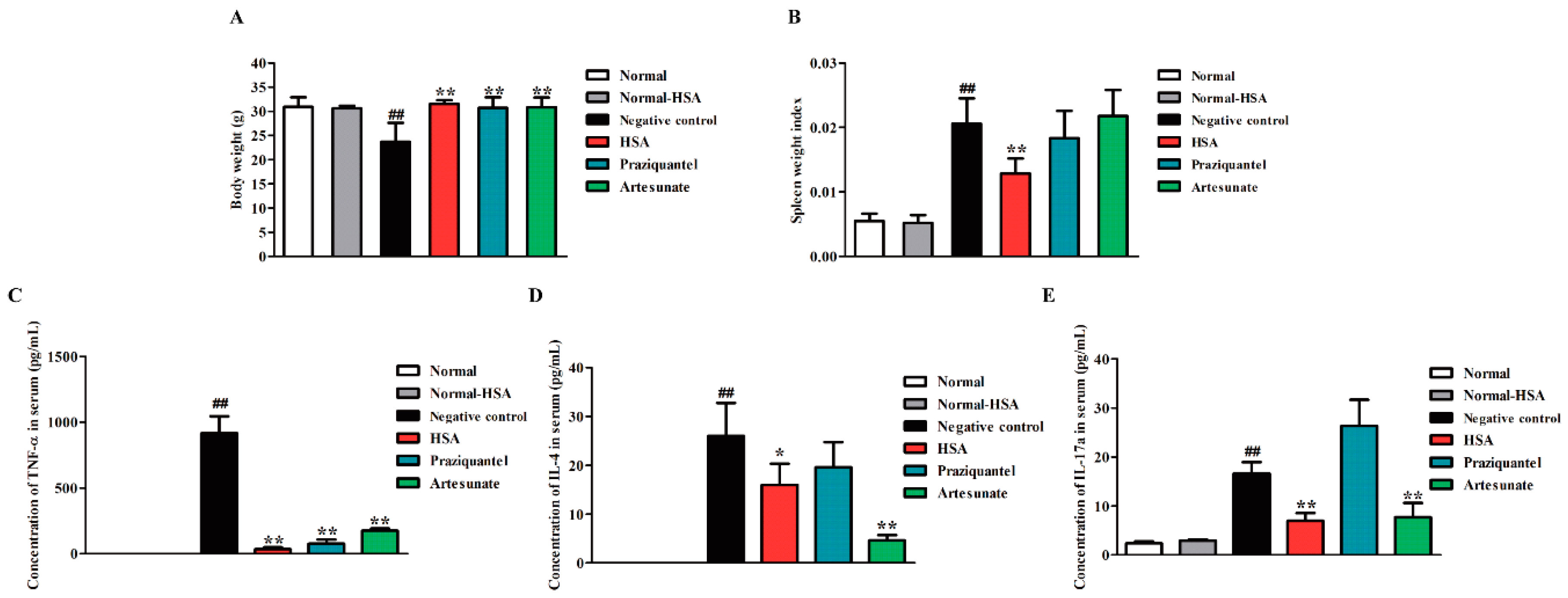
2.5. HSA Altered Cytokine Profile in S. japonicum Infected Mice
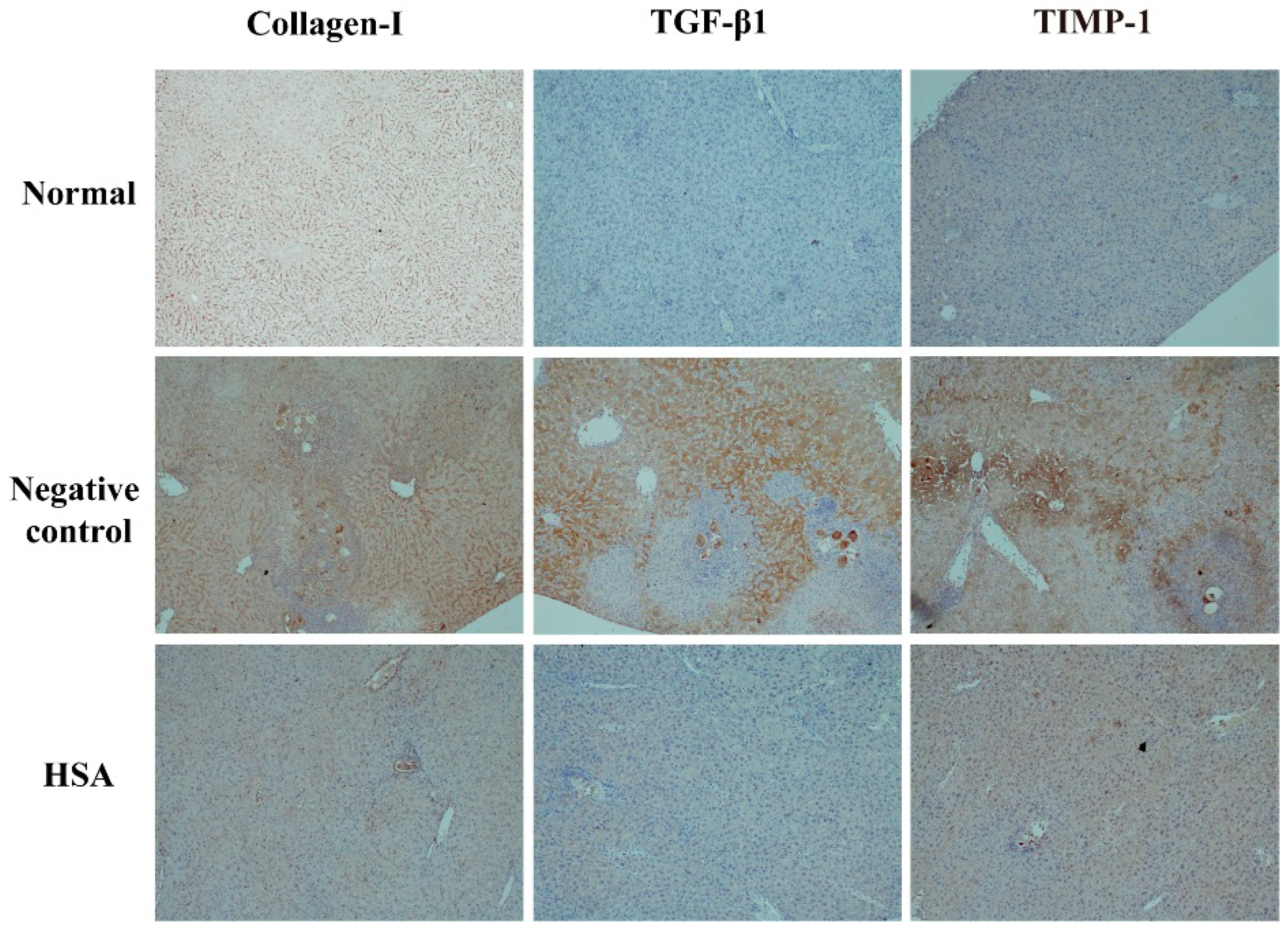
2.6. HSA Protects the Liver with Anti-Fibrotic Effects
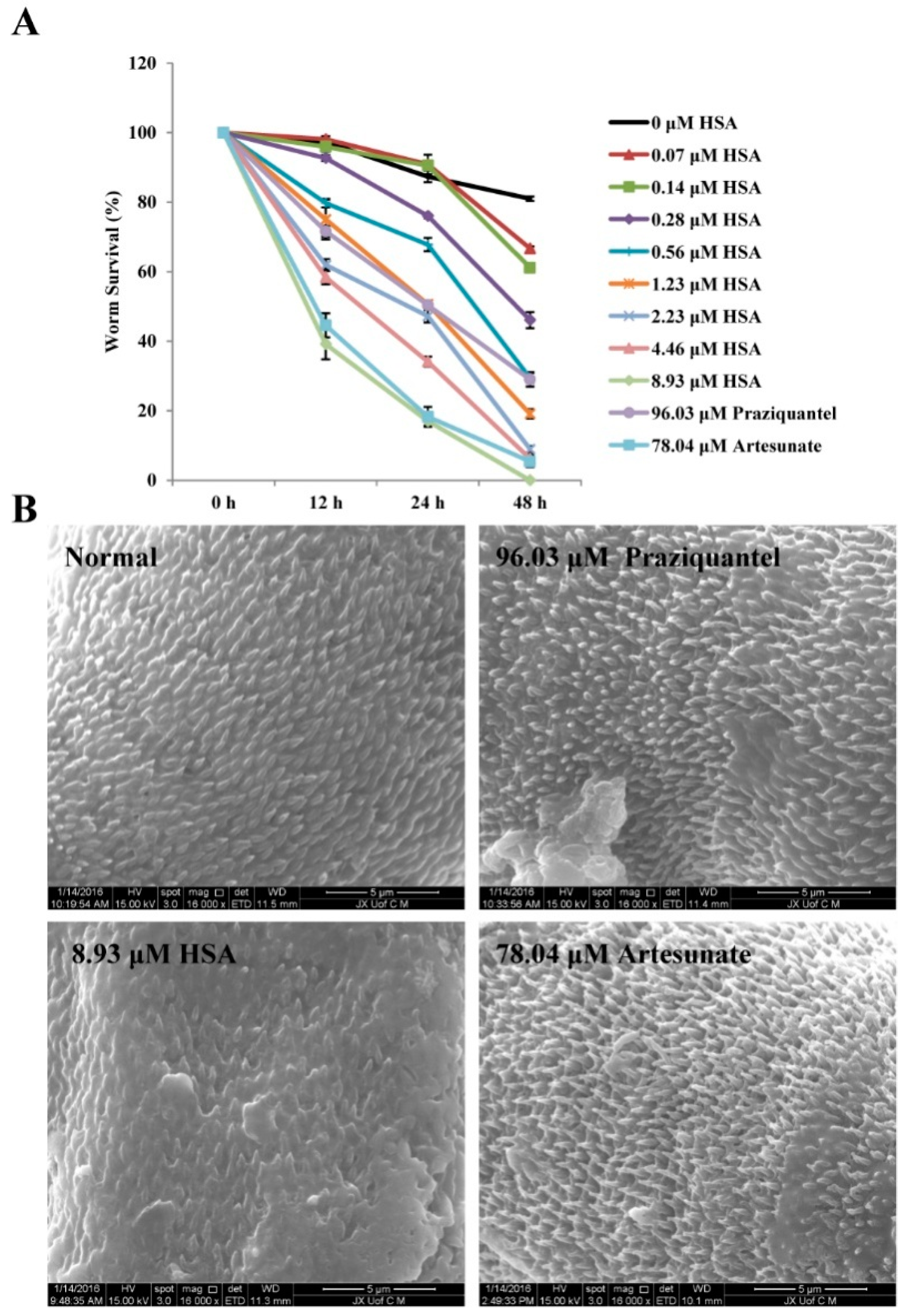
2.7. The Inhibition of S. japonicum NTS by HSA Is Partially Due to the Tegumental Disruption
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Compounds
4.3. In Vivo Studies with S. japonicum
4.4. In Vivo Studies with S. mansoni
4.5. Histology Analysis
4.6. Immunological Analysis
4.7. Immunohistochemistry Analysis
4.8. Cultivation of Newly Transformed Schistosomula (NTS-the Larval Stage) S. japonicum and Scanning Electron Microscopy (SEM)
4.8.1. Collection of NTS S. japonicum
4.8.2. Schistosome Incubation In Vitro with Treatment
4.8.3. SEM
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- King, C.H. Lifting the burden of schistosomiasis—Defining elements of infection-associated disease and the benefits of antiparasite treatment. J. Infect. Dis. 2007, 196, 653–655. [Google Scholar] [CrossRef] [PubMed]
- King, C.H.; Dangerfield-Cha, M. The unacknowledged impact of chronic schistosomiasis. Chronic Illn. 2008, 4, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Gray, D.J.; McManus, D.P.; Li, Y.; Williams, G.M.; Bergquist, R.; Ross, A.G. Schistosomiasis elimination: Lessons from the past guide the future. Lancet Infect. Dis. 2010, 10, 733–736. [Google Scholar] [CrossRef]
- King, C.H.; Dickman, K.; Tisch, D.J. Reassessment of the cost of chronic helmintic infection: A meta-analysis of disability-related outcomes in endemic schistosomiasis. Lancet 2005, 365, 1561–1569. [Google Scholar] [CrossRef]
- Bergquist, R.; Utzinger, J.; McManus, D.P. Trick or treat: The role of vaccines in integrated schistosomiasis control. PLoS Negl. Trop. Dis. 2008, 2, e244. [Google Scholar] [CrossRef] [PubMed]
- Hotez, P.J.; Molyneux, D.H.; Fenwick, A.; Ottesen, E.; Ehrlich Sachs, S.; Sachs, J.D. Incorporating a rapid-impact package for neglected tropical diseases with programs for HIV/AIDS, tuberculosis, and malaria. PLoS Med. 2006, 3, e102. [Google Scholar] [CrossRef] [PubMed]
- Steinmann, P.; Keiser, J.; Bos, R.; Tanner, M.; Utzinger, J. Schistosomiasis and water resources development: Systematic review, meta-analysis, and estimates of people at risk. Lancet Infect. Dis. 2006, 6, 411–425. [Google Scholar] [CrossRef]
- Gryseels, B.; Polman, K.; Clerinx, J.; Kestens, L. Human schistosomiasis. Lancet 2006, 368, 1106–1118. [Google Scholar] [CrossRef]
- Cioli, D.; Pica-Mattoccia, L.; Basso, A.; Guidi, A. Schistosomiasis control: Praziquantel forever? Mol. Biochem. Parasitol. 2014, 195, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Fallon, P.G.; Doenhoff, M.J. Drug-resistant schistosomiasis: Resistance to praziquantel and oxamniquine induced in Schistosoma mansoni in mice is drug specific. Am. J. Trop. Med. Hyg. 1994, 51, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Metwally, A.; Farghaly, A.; Bruce, J.; Tao, L.F.; Bennett, J.L. Characterization of isolates of Schistosoma mansoni from egyptian villagers that tolerate high doses of praziquantel. Am. J. Trop. Med. Hyg. 1996, 55, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Ghani, R.A.; Loutfy, N.; Hassan, A. Experimentally promising antischistosomal drugs: A review of some drug candidates not reaching the clinical use. Parasitol. Res. 2009, 105, 899–906. [Google Scholar] [CrossRef] [PubMed]
- Colley, D.G. Morbidity control of Schistosomiasis by mass drug administration: How can we do it best and what will it take to move on to elimination? Trop. Med. Health 2014, 42, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Kayser, O.; Kiderlen, A.F.; Croft, S.L. Natural products as antiparasitic drugs. Parasitol. Res. 2003, 90 (Suppl. 2), S55–S62. [Google Scholar] [CrossRef] [PubMed]
- Tagboto, S.; Townson, S. Antiparasitic properties of medicinal plants and other naturally occurring products. Adv. Parasitol. 2001, 50, 199–295. [Google Scholar] [PubMed]
- Chan-Bacab, M.J.; Pena-Rodriguez, L.M. Plant natural products with leishmanicidal activity. Nat. Prod. Rep. 2001, 18, 674–688. [Google Scholar] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Utzinger, J.; Xiao, S.H.; Tanner, M.; Keiser, J. Artemisinins for schistosomiasis and beyond. Curr. Opin. Investig. Drugs 2007, 8, 105–116. [Google Scholar] [PubMed]
- Mimaki, Y.; Kuroda, M.; Asano, T.; Sashida, Y. Triterpene saponins and lignans from the roots of Pulsatilla chinensis and their cytotoxic activity against hl-60 cells. J. Nat. Prod. 1999, 62, 1279–1283. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Ye, L.; Guan, X.; Liang, X.; Li, C.; Sun, Q.; Liu, Y.; Chen, S.; Bang, F.; Liu, B. Immunopontentiating and antitumor activities of a polysaccharide from Pulsatilla chinensis (bunge) regel. Int. J. Biol. Macromol. 2013, 54, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Zhang, M.; Zhang, P.; Song, Z.; Ma, Z.; Qu, H. Silver complexation and tandem mass spectrometry for differentiation of triterpenoid saponins from the roots of Pulsatilla chinensis (bunge) regel. Rapid Commun. Mass Spectrom. 2008, 22, 3783–3790. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.M.; Shu, Z.; He, W.J.; Chen, L.Y.; Yang, S.L.; Yang, G.; Liu, Y.L.; Li, X.R. Antitumor activity of Pulsatilla chinensis (bunge) regel saponins in human liver tumor 7402 cells in vitro and in vivo. Phytomedicine 2012, 19, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Li, L.D.; Li, W.C.; Liu, C.W.; Shi, W.J.; Gong, P.T.; Li, J.H.; Zhang, G.C.; Yang, J.; Li, H.; Zhang, X.C. Giardia intestinalis: Effects of Pulsatilla chinensis extracts on trophozoites. Parasitol. Res. 2012, 111, 1929–1935. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Beon, M.S.; Kim, M.K. Selective growth inhibitor toward human intestinal bacteria derived from Pulsatilla cernua root. J. Agric. Food Chem. 2001, 49, 4656–4661. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Wang, R.; He, M.; Huang, H.; Wang, Q.; Yang, Z.; Li, Y.; Yang, S.; Jin, Y. Nitric oxide-donating derivatives of hederacolchiside A1: Synthesis and biological evaluation in vitro and in vivo as potential anticancer agents. Bioorg. Med. Chem. Lett. 2017, 27, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.S.; Mentink-Kane, M.M.; Pesce, J.T.; Ramalingam, T.R.; Thompson, R.; Wynn, T.A. Immunopathology of schistosomiasis. Immunol. Cell Biol. 2007, 85, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Mantawy, M.M.; Ali, H.F.; Rizk, M.Z. Therapeutic effects of allium sativum and allium cepa in Schistosoma mansoni experimental infection. Rev. Inst. Med. Trop. Sao Paulo 2011, 53, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Mantawy, M.M.; Aly, H.F.; Zayed, N.; Fahmy, Z.H. Antioxidant and schistosomicidal effect of allium sativum and allium cepa against Schistosoma mansoni different stages. Eur. Rev. Med. Pharmacol. Sci. 2012, 16 (Suppl. 3), 69–80. [Google Scholar] [PubMed]
- Seifel-Din, S.H.; El-Lakkany, N.M.; Mohamed, M.A.; Hamed, M.M.; Sterner, O.; Botros, S.S. Potential effect of the medicinal plants Calotropis procera, Ficus elastica and Zingiber officinale against Schistosoma mansoni in mice. Pharm. Biol. 2014, 52, 144–150. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mostafa, O.M.; Eid, R.A.; Adly, M.A. Antischistosomal activity of ginger (Zingiber officinale) against Schistosoma mansoni harbored in c57 mice. Parasitol. Res. 2011, 109, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.M.; Menezes, R.M.; de Oliveira, S.A.; Andrade, Z.A. Chemotherapeutic effects on larval stages of Schistosoma mansoni during infection and re-infection of mice. Rev. Soc. Bras. Med. Trop. 2003, 36, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Awney, H.A.; Ghzlan, H.E.; Sheweita, S.A.; Mostafa, M.H. Different levels of Schistosoma mansoni infection increased the mutagenicity of benzo(a)pyrene, the activity of aryl hydrocarbon hydroxylase and the formation of hepatic microsomal hydrogen peroxide. Toxicology 2001, 163, 213–218. [Google Scholar] [CrossRef]
- Bergquist, N.R. Schistosomiasis: From risk assessment to control. Trends Parasitol. 2002, 18, 309–314. [Google Scholar] [CrossRef]
- Burke, M.L.; Jones, M.K.; Gobert, G.N.; Li, Y.S.; Ellis, M.K.; McManus, D.P. Immunopathogenesis of human schistosomiasis. Parasite Immunol. 2009, 31, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Thompson, R.W.; Cheever, A.W.; Mentink-Kane, M.M. Immunopathogenesis of schistosomiasis. Immunol. Rev. 2004, 201, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E.J.; MacDonald, A.S. The immunobiology of schistosomiasis. Nat. Rev. Immunol. 2002, 2, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Cheever, A.W.; Hoffmann, K.F.; Wynn, T.A. Immunopathology of Schistosomiasis mansoni in mice and men. Immunol. Today 2000, 21, 465–466. [Google Scholar] [CrossRef]
- Shainheit, M.G.; Lasocki, K.W.; Finger, E.; Larkin, B.M.; Smith, P.M.; Sharpe, A.H.; Dinarello, C.A.; Rutitzky, L.I.; Stadecker, M.J. The pathogenic th17 cell response to major schistosome egg antigen is sequentially dependent on il-23 and il-1beta. J. Immunol. 2011, 187, 5328–5335. [Google Scholar] [CrossRef] [PubMed]
- Friedman, S.L. Mechanisms of hepatic fibrogenesis. Gastroenterology 2008, 134, 1655–1669. [Google Scholar] [CrossRef] [PubMed]
- Bartley, P.B.; Ramm, G.A.; Jones, M.K.; Ruddell, R.G.; Li, Y.; McManus, D.P. A contributory role for activated hepatic stellate cells in the dynamics of Schistosoma japonicum egg-induced fibrosis. Int. J. Parasitol. 2006, 36, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Gomez, D.E.; De Lorenzo, M.S.; Alonso, D.F.; Andrade, Z.A. Expression of metalloproteinases (MMP-1, MMP-2, and MMP-9) and their inhibitors (TIMP-1 and TIMP-2) in schistosomal portal fibrosis. Am. J. Trop. Med. Hyg. 1999, 61, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Hemmann, S.; Graf, J.; Roderfeld, M.; Roeb, E. Expression of mmps and timps in liver fibrosis—A systematic review with special emphasis on anti-fibrotic strategies. J. Hepatol. 2007, 46, 955–975. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Matsuzaki, K. Differential regulation of TGF-beta/Smad signaling in hepatic stellate cells between acute and chronic liver injuries. Front. Physiol. 2012, 3, 53. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, J. Natural products with antischistosomal activity. Future Med. Chem. 2015, 7, 801–820. [Google Scholar] [CrossRef] [PubMed]
- Staudt, U.; Schmahl, G.; Blaschke, G.; Mehlhorn, H. Light and scanning electron microscopy studies on the effects of the enantiomers of praziquantel and its main metabolite on Schistosoma mansoni in vitro. Parasitol. Res. 1992, 78, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Jiraungkoorskul, W.; Sahaphong, S.; Sobhon, P.; Riengrojpitak, S.; Kangwanrangsan, N. Effects of praziquantel and artesunate on the tegument of adult Schistosoma mekongi harboured in mice. Parasitol. Int. 2005, 54, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Jiraungkoorskul, W.; Sahaphong, S.; Sobhon, P.; Riengrojpitak, S.; Kangwanrangsan, N. Schistosoma mekongi: The in vitro effect of praziquantel and artesunate on the adult fluke. Exp. Parasitol. 2006, 113, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Manneck, T.; Haggenmuller, Y.; Keiser, J. Morphological effects and tegumental alterations induced by mefloquine on schistosomula and adult flukes of Schistosoma mansoni. Parasitology 2010, 137, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Eissa, M.M.; El-Azzouni, M.Z.; Amer, E.I.; Baddour, N.M. Miltefosine, a promising novel agent for schistosomiasis mansoni. Int. J. Parasitol. 2011, 41, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Veras, L.M.; Guimaraes, M.A.; Campelo, Y.D.; Vieira, M.M.; Nascimento, C.; Lima, D.F.; Vasconcelos, L.; Nakano, E.; Kuckelhaus, S.S.; Batista, M.C.; et al. Activity of epiisopiloturine against Schistosoma mansoni. Curr. Med. Chem. 2012, 19, 2051–2058. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, J.; Nascimento, C.; Yamaguchi, L.F.; Kato, M.J.; Nakano, E. Schistosoma mansoni: In vitro schistosomicidal activity and tegumental alterations induced by piplartine on schistosomula. Exp. Parasitol. 2012, 132, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Ekabo, O.A.; Farnsworth, N.R.; Henderson, T.O.; Mao, G.; Mukherjee, R. Antifungal and molluscicidal saponins from Serjania salzmanniana. J. Nat. Prod. 1996, 59, 431–435. [Google Scholar] [CrossRef] [PubMed]
- Yolles, T.K.; Moore, D.V.; DeGiusti, D.L.; Ripsom, C.A.; Meleney, H.E. A technique for the perfusion of laboratory animals for the recovery of schistosomes. J. Parasitol. 1947, 33, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Smithers, S.R.; Terry, R.J. The infection of laboratory hosts with cercariae of Schistosoma mansoni and the recovery of the adult worms. Parasitology 1965, 55, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.H.; Keiser, J.; Chollet, J.; Utzinger, J.; Dong, Y.; Endriss, Y.; Vennerstrom, J.L.; Tanner, M. In vitro and in vivo activities of synthetic trioxolanes against major human schistosome species. Antimicrob. Agents Chemother. 2007, 51, 1440–1445. [Google Scholar] [CrossRef] [PubMed]
- Tendler, M.; Pinto, R.M.; Oliveira Lima, A.; Gebara, G.; Katz, N. Schistosoma mansoni: Vaccination with adult worm antigens. Int. J. Parasitol. 1986, 16, 347–352. [Google Scholar] [CrossRef]
- Cheever, A.W.; Anderson, L.A. Rate of destruction of Schistosoma mansoni eggs in the tissues of mice. Am. J. Trop. Med. Hyg. 1971, 20, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J. In vitro and in vivo trematode models for chemotherapeutic studies. Parasitology 2010, 137, 589–603. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds Hederacolchiside A1 are available from the authors. |
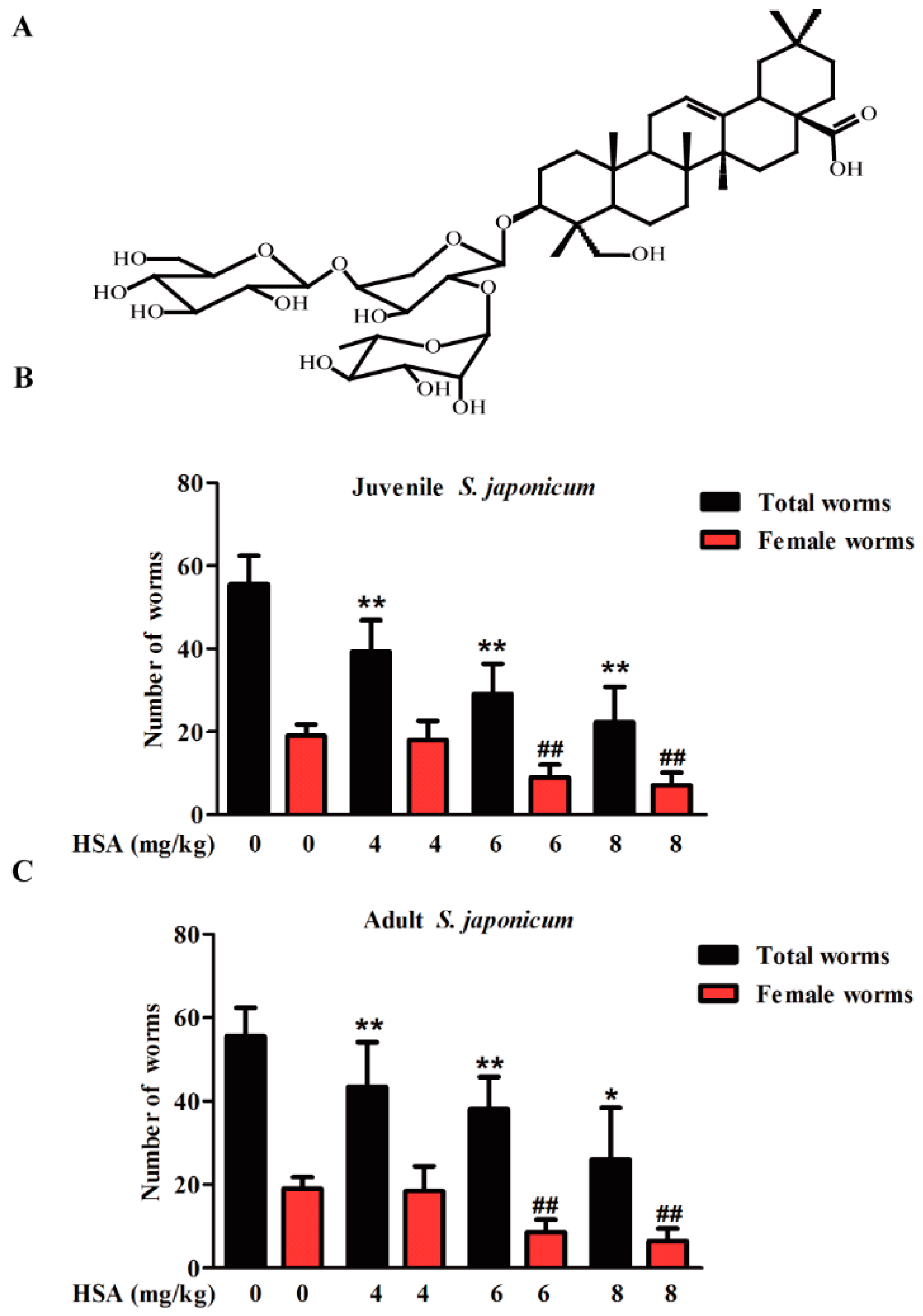
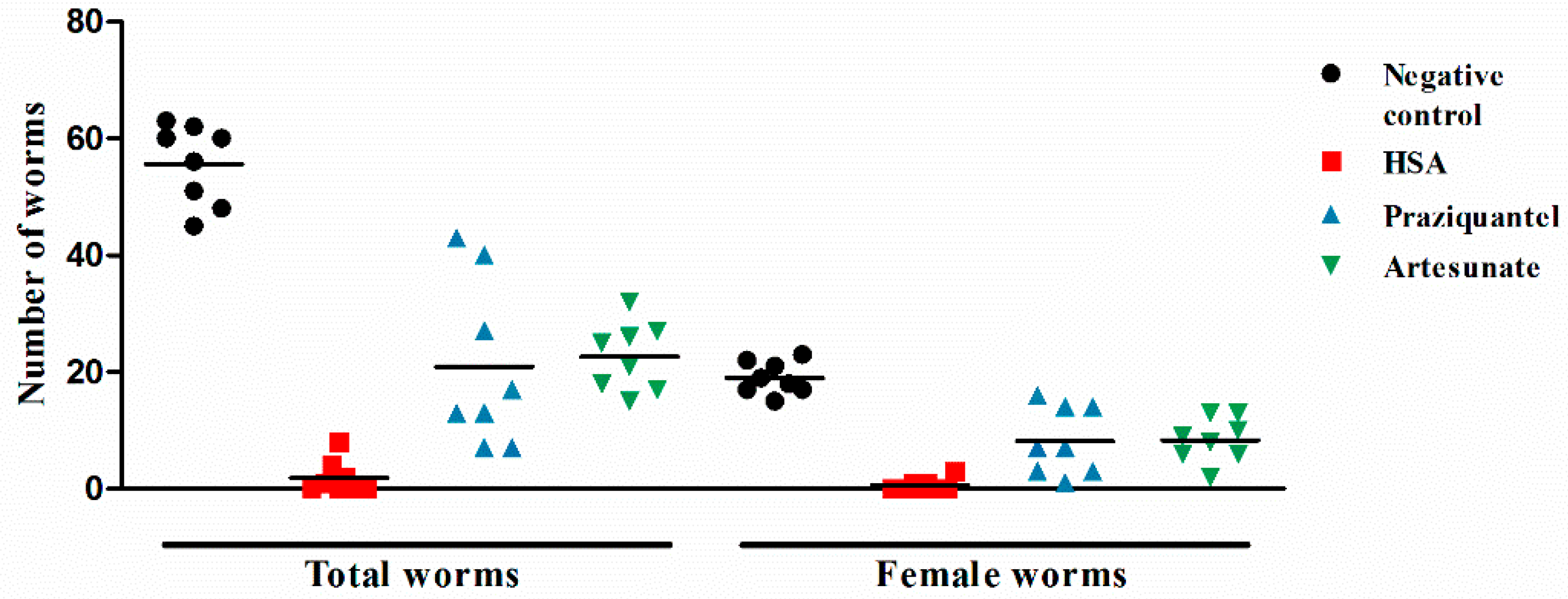
| Treatment (Dose mg·kg−1) | Worm Reduction Rate (%) | |||
|---|---|---|---|---|
| Total | Female | Total | Female | |
| 14–18 days | 35–39 days | |||
| Control | __ | __ | __ | __ |
| HSA (8) | 59.9 | 62.4 | 53.2 | 65.8 |
| HSA (6) | 47.6 | 52.6 | 32.4 | 52.6 |
| HSA (4) | 29.3 | 5.3 | 21.8 | 2.9 |
| praziquantel (300) | 52.0 | 45.6 | 85.2 | 83.2 |
| artesunate (300) | 85.1 | 85.0 | 92.4 | 94.7 |
| Treatment Stage of Post-Infection | Treatment (Dose mg/kg) | Mean Number of Worms (SD) | Worm Burden Reduction (%) | ||
|---|---|---|---|---|---|
| Total | Female | Total | Female | ||
| 1–5 days | - | 55.6 (6.8) | 19.0 (2.8) | — | — |
| HSA (8) | 1.9 (2.7) *** | 0.6 (1.0) *** | 97.2 | 94.7 | |
| praziquantel (300) | 20.9 (13.3) *** | 8.1 (5.4) *** | 62.4 | 57.2 | |
| artesunate (300) | 22.6 (5.5) *** | 8.4 (3.5) *** | 59.3 | 55.9 | |
| 7–11 days | HSA (8) | 5.5(3.3) *** | 1.6(3.3) *** | 90.0 | 89.5 |
| praziquantel (300) | 22.4(6.8) *** | 7.6(2.9) *** | 55.6 | 59.9 | |
| artesunate (300) | 26.4(11.9) *** | 10.0(5.0) ** | 52.5 | 47.4 | |
| 14–19 days | HSA (8) | 22.3 (8.6) *** | 7.2 (3.1) *** | 60.7 | 63.2 |
| praziquantel (300) | 26.7 (9.5) *** | 10.3 (3.2) *** | 52.0 | 45.6 | |
| artesunate (300) | 8.3 (5.9) *** | 2.9 (2.2) *** | 85.1 | 85.0 | |
| 21–25 days | HSA (8) | 24.6 (9.6) *** | 6.4 (4.1) *** | 55.4 | 68.4 |
| praziquantel (300) | 7.9 (5.8) *** | 5.0 (3.5) *** | 85.9 | 73.7 | |
| artesunate (300) | 8.7 (3.8) *** | 5.4 (2.8) *** | 84.3 | 71.4 | |
| 28–32 days | HSA (8) | 32.6 (7.1) *** | 6.1 (1.8) *** | 41.1 | 68.4 |
| praziquantel (300) | 4.3 (3.4) *** | 3.5 (2.5) *** | 92.2 | 81.6 | |
| artesunate (300) | 1.5 (1.9) *** | 1.3 (1.6) *** | 97.3 | 93.0 | |
| 35–39 days | HSA (8) | 26.0 (12.4) ** | 6.5 (3.1) *** | 53.2 | 65.8 |
| praziquantel (300) | 8.2 (14.9) ** | 3.2 (5.9) ** | 85.2 | 83.2 | |
| artesunate (300) | 4.3 (4.0) *** | 1 (1.2) *** | 92.4 | 94.7 | |
| Treatment Stage of Post-Infection | Treatment (Dose mg/kg) | Mean Number of Worms (SD) | Worm Burden Reduction (%) | ||
|---|---|---|---|---|---|
| Total | Females | Total | Females | ||
| 1–5 days | - | 38.3 (16.6) | 14.1 (7.8) | — | — |
| HSA (8) | 4.4 (2.7) *** | 1.0 (1.5) ** | 88.6 | 92.9 | |
| praziquantel (300) | 5.3 (5.4) *** | 2.3 (2.6) ** | 86.2 | 83.8 | |
| artesunate (300) | 13.6 (4.7) ** | 5.0 (1.8) * | 64.4 | 64.6 | |
| 7–11 days | HSA (8) | 7.4 (3.1) ** | 2.1 (1.5) ** | 80.7 | 85.0 |
| praziquantel (300) | 13.2 (6.3) ** | 2.5 (1.5) ** | 65.6 | 82.3 | |
| artesunate (300) | 23.8 (11.2) | 6.5 (3.6) * | 37.7 | 54.0 | |
| 21–25 days | HSA (8) | 12.1 (7.0) ** | 4.3 (3.3) * | 68.3 | 70.0 |
| praziquantel (300) | 16.7 (10.3) * | 5.0 (3.7) * | 56.4 | 64.6 | |
| artesunate (300) | 1.5 (1.3) *** | 0.2 (0.4) ** | 96.1 | 98.8 | |
| 42–46 days | HSA (8) | 9.5 (4.4) ** | 2.3 (1.4) ** | 75.2 | 84.1 |
| praziquantel (300) | 13.2 (8.5) * | 4.5 (3.5) ** | 65.6 | 68.1 | |
| artesunate (300) | 2.5 (2.5) *** | 0.7 (0.7) ** | 93.5 | 95.3 | |
| 49–53 days | HSA (8) | 11.3 (4.1) ** | 2.5 (1.4) ** | 70.6 | 82.3 |
| praziquantel (300) | 1.3 (0.7) ** | 0.3 (0.5) *** | 96.5 | 97.6 | |
| artesunate (300) | 13.7(7.0) ** | 3.8 (2.0) ** | 64.3 | 72.9 | |
| Treatment Stage of Post-Infection | Treatment (Dose mg/kg) | Hepatic Ova Reduction Rate (%) |
|---|---|---|
| 1–5 days | - | — |
| HSA (8) | 99.3 | |
| praziquantel (300) | 64.7 | |
| artesunate (300) | 61.3 | |
| 7–11 days | HSA (8) | 98.7 |
| praziquantel (300) | 68.1 | |
| artesunate (300) | 54.6 | |
| 14–19 days | HSA (8) | 72.6 |
| praziquantel (300) | 59.5 | |
| artesunate (300) | 97.6 | |
| 21–25 days | HSA (8) | 79.7 |
| praziquantel (300) | 89.9 | |
| artesunate (300) | 89.6 | |
| 28–32 days | HSA (8) | 75.7 |
| praziquantel (300) | 98.2 | |
| artesunate (300) | 99.8 | |
| 35–39 days | HSA (8) | 75.4 |
| praziquantel (300) | 97.8 | |
| artesunate (300) | 99.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, N.; Shen, W.; Gao, H.; Feng, Y.; Zhu, W.; Yang, S.; Liu, Y.; Xu, Q.; Yu, D. Antischistosomal Properties of Hederacolchiside A1 Isolated from Pulsatilla chinensis. Molecules 2018, 23, 1431. https://doi.org/10.3390/molecules23061431
Kang N, Shen W, Gao H, Feng Y, Zhu W, Yang S, Liu Y, Xu Q, Yu D. Antischistosomal Properties of Hederacolchiside A1 Isolated from Pulsatilla chinensis. Molecules. 2018; 23(6):1431. https://doi.org/10.3390/molecules23061431
Chicago/Turabian StyleKang, Naixin, Wenhua Shen, Hongwei Gao, Yulin Feng, Weifeng Zhu, Shilin Yang, Yanli Liu, Qiongming Xu, and Di Yu. 2018. "Antischistosomal Properties of Hederacolchiside A1 Isolated from Pulsatilla chinensis" Molecules 23, no. 6: 1431. https://doi.org/10.3390/molecules23061431
APA StyleKang, N., Shen, W., Gao, H., Feng, Y., Zhu, W., Yang, S., Liu, Y., Xu, Q., & Yu, D. (2018). Antischistosomal Properties of Hederacolchiside A1 Isolated from Pulsatilla chinensis. Molecules, 23(6), 1431. https://doi.org/10.3390/molecules23061431





