Epitopes of MUC1 Tandem Repeats in Cancer as Revealed by Antibody Crystallography: Toward Glycopeptide Signature-Guided Therapy
Abstract
1. Introduction
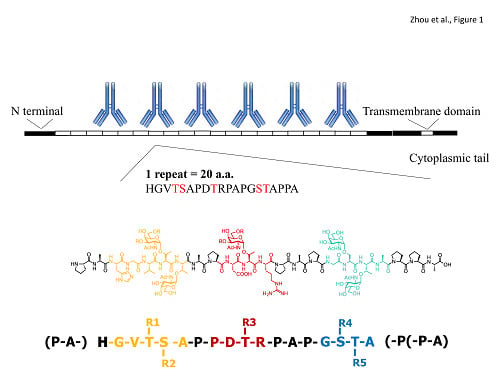
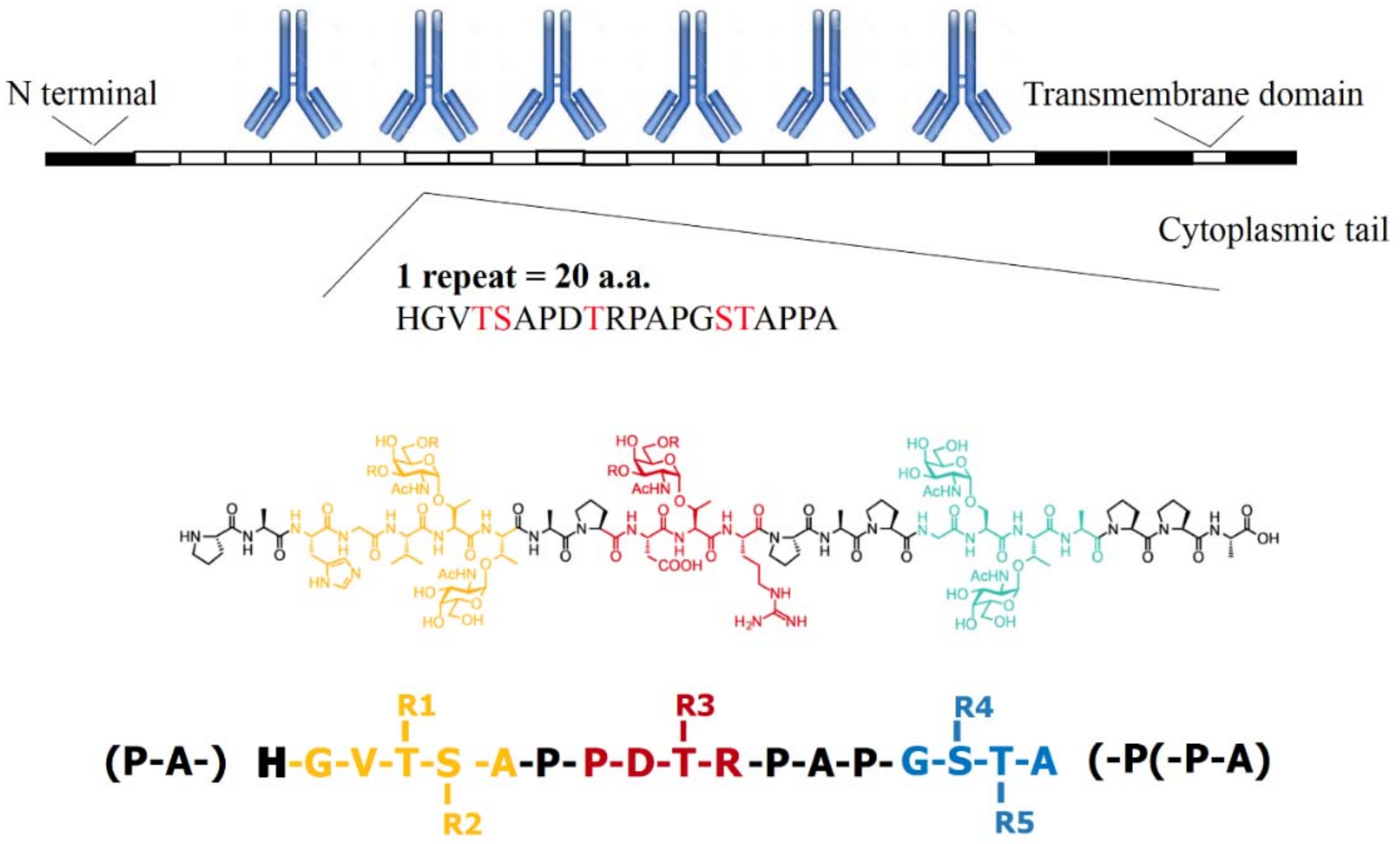
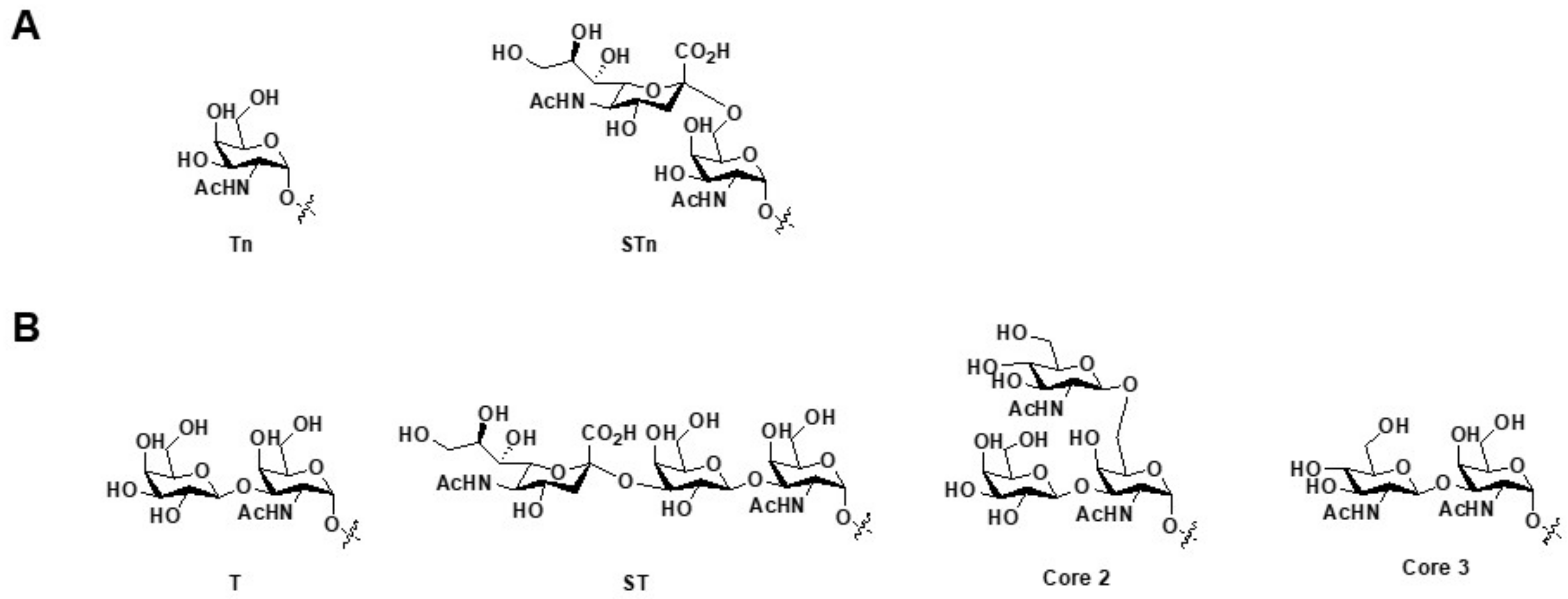
1.1. The Sugar and Peptide Portions of MUC1 in Clinical Medicine
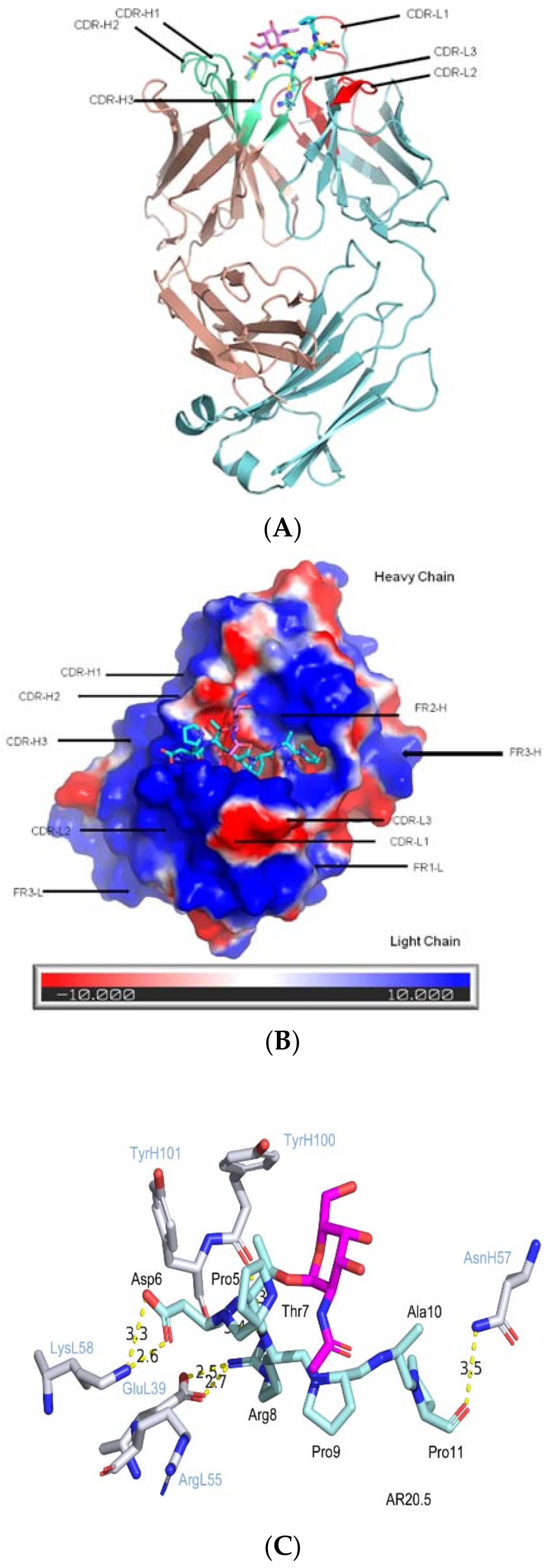
2. Published Co-Crystal Structures: 3 Hypothesized Models of CDR (Complementarity Determining Region) Binding to Glycopeptide Signature
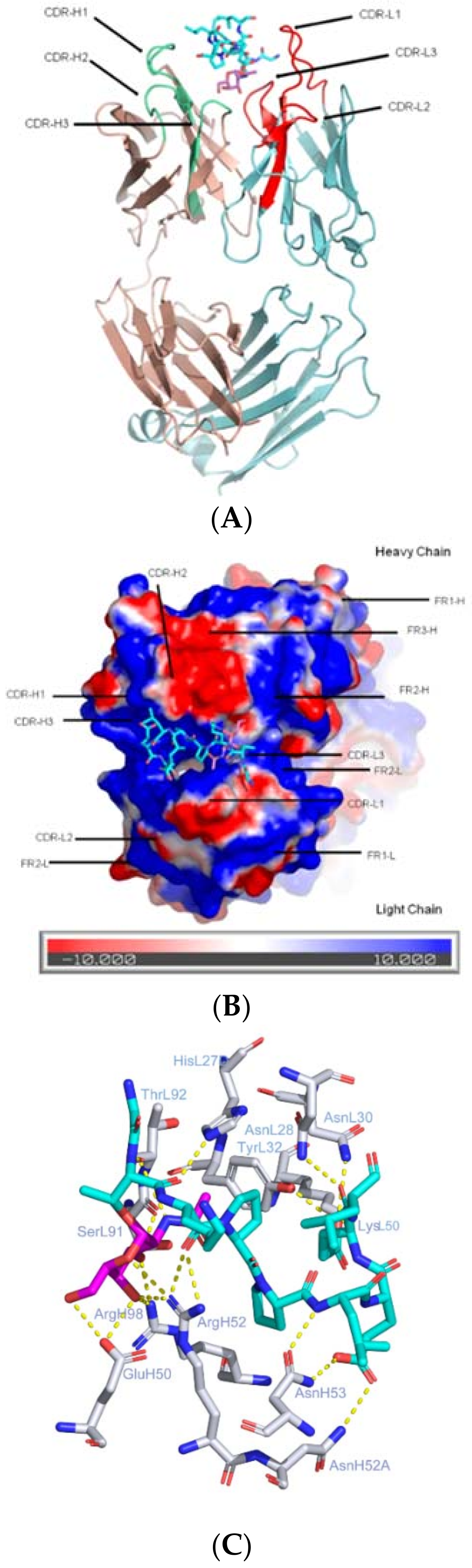
2.1. CDRs Bind to Sugar and Hypermutated FRs Bind to Peptide
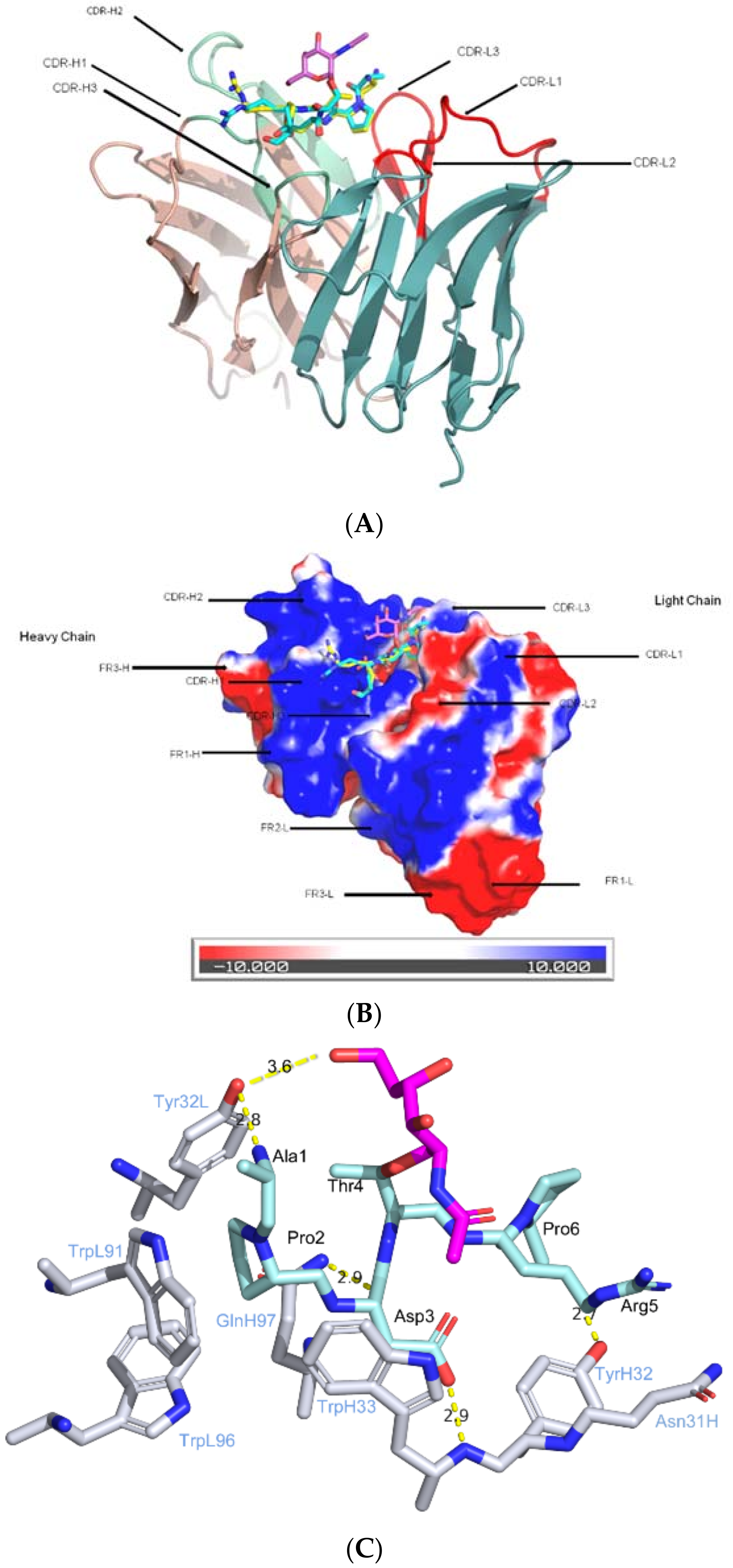
2.2. CDRs Bind to Both Sugar and Peptide Portions
2.3. CDRs Bind to Peptide Portion, while the Sugar Portion May Alter Peptide Confirmation
3. Hypothesized Functional Roles of Peptide and Sugar Portions: Four Types of Glycopeptide Signature Scanned by B Cells
3.1. Direct Stimulation by Sugar (Glycans)
3.2. Direct Stimulation of BCR by a Peptide Epitope
3.3. Glycan-Shield as Immune-Escape Mechanism to Suppress B Cell Stimulation by A Peptide
3.4. Glycan Modification to Improve the B Cell Stimulation by A Peptide
4. Glycopeptide Signature-Induced Monoclonal Antibodies: 3 Groups by Specificity
4.1. Antibodies Which Only Bind to Peptide Portion
4.2. Antibodies Which Only Bind to Sugar Portion
4.3. Antibodies Which Bind to Both Peptide and Sugar Portions
5. Glycopeptide Signature-Guided Design of MUC1 Vaccine
5.1. The Use of Peptide Motif as Priming Immunogens
5.2. Clustered Tandem Repeat Sequences for BCR Binding
5.3. The Choice of Peptide Backbone for Sugars
5.4. Modification of Glycans on MUC1 Glycopeptides in Vaccine Design
5.5. Double-Edged Sword: The Cross Reactivity of MUC1-Glycopeptide Binding Monoclonal Antibodies
6. Glycopeptide Signature-Guided Selection of Antibody Therapeutics
6.1. Antibody-Drug Conjugate
6.2. Bi-Specific Antibodies
6.3. Chimeric Antigen Receptors
7. New Tools for Decoding Glycopeptide Signatures in High Throughput
- (1)
- Glycopeptide array: the specificity of mAbs is always a most difficult issue when a mAb is used as a diagnostic tool. Several groups have published glycopeptide arrays to address this issue. In a 106 glycopeptide chip array designed by Westerlind group [65], MUC1 tandem repeat sequence containing abnormal and normal O-glycans at five sites of glycosylation were printed in a chip format, and used to test the polyclonal antibodies induced by synthetic MUC1 glycopeptide vaccines. It is anticipated that such glycopeptide chip arrays may be produced at a custom service level, with the cost of $50,000 for 106 glycopeptides each at microgram level, for 100 slides.
- (2)
- Next generation sequencing and single B cell sequencing. The development of next generation sequencing and single cell sequencing technologies [137,138] has made it possible to generate a complete picture of B cell repertoire after the individuals are immunized by MUC1 glycopeptide vaccine. By focusing on syndecan+ memory B cells and plasma cells, those mAb clones which expand after being stimulated by glycopeptide vaccines can be selected, and expressed as Fab at high throughput, allowing the functional studies of sequenced BCRs.
- (3)
- The humanized antibody transgenic mice. Several humanized antibody mouse models have been developed which have the mouse IgH and IgL loci replaced by human ones, and humanized antibodies have been generated [139,140,141,142,143]. The other interesting tool is the human IL6 knock-in mice which allows better engrafting of human cord blood cells to develop into mature human T and B cells. Using OVA as an antigen, Flavell group reported high affinity antibody response and isolated humanized mAb clones in this model [144]. Antibodies generated from mice engrafted with human lymphocyte progenitors serve as a unique tool for answering difficult questions, such as which motif of MUC1 tandem repeat unit is the immune dominant epitope.
8. Outlook
8.1. Possible Breakthrough in Antibody Diagnosis in Next 5 Years
8.2. Possible Breakthrough in Antibody Therapy in Next 5 Years
8.3. Multi-Valent Glycopeptide Vaccine R&D in Next 5 Years
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BCR | B cell receptor |
| CAR | chimeric antigen receptor |
| CDR | complementarity-determining region |
| CTC | circulating tumor cell |
| DM1 | Mertansine |
| FR | framework region |
| IgG | immunoglobulin G |
| IgH | immunoglobulin heavy chain |
| IgL | immunoglobulin light chain |
| KLH | keyhole limpet hemocyanin |
| mAb | monoclonal antibody |
| NK cells | natural killer cells |
| NKT cells | natural killer T cells |
| OVA | Ovalbumin |
| ST antigen | Neu5Ac α3 Gal β3 GalNAc α-Ser/Thr |
| STn antigen | Neu5Ac α6 GalNAcα-Ser/Thr |
| T antigen | Gal β3 GalNAc α-Ser/Thr |
| Tn antigen | GalNAc α-Ser/Thr |
| TR | tandem repeat |
| VH | heavy chain |
| VL | light chain |
References
- Murphy, G.P. The early implementation of the National Cancer Act of 1971. Cancer 1996, 78, 2601–2602. [Google Scholar] [CrossRef]
- Kobata, A.; Amano, J. Altered glycosylation of proteins produced by malignant cells, and application for the diagnosis and immunotherapy of tumours. Immunol. Cell Biol. 2005, 83, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Dausset, J.; Moullec, J.; Bernard, J. Acquired hemolytic anemia with polyagglutinability of red blood cells due to a new factor present in normal human serum (Anti-Tn). Blood 1959, 14, 1079–1093. [Google Scholar] [PubMed]
- Thurnher, M.; Rusconi, S.; Berger, E.G. Persistent repression of a functional allele can be responsible for galactosyltransferase deficiency in Tn syndrome. J. Clin. Investig. 1993, 91, 2103–2110. [Google Scholar] [CrossRef] [PubMed]
- Mi, R.; Song, L.; Wang, Y.; Ding, X.; Zeng, J.; Lehoux, S.; Aryal, R.P.; Wang, J.; Crew, V.K.; van Die, I.; et al. Epigenetic silencing of the chaperone Cosmc in human leukocytes expressing Tn antigen. J. Biol. Chem. 2012, 287, 41523–41533. [Google Scholar] [CrossRef] [PubMed]
- Chia, J.; Goh, G.; Bard, F. Short O-GalNAc glycans: Regulation and role in tumor development and clinical perspectives. Biochim. Biophys. Acta 2016, 1860, 1623–1639. [Google Scholar] [CrossRef] [PubMed]
- Ju, T.; Wang, Y.; Aryal, R.P.; Lehoux, S.D.; Ding, X.; Kudelka, M.R.; Cutler, C.; Zeng, J.; Wang, J.; Sun, X.; et al. Tn and sialyl-Tn antigens, aberrant O-glycomics as human disease markers. Proteom. Clin. Appl. 2013, 7, 618–631. [Google Scholar]
- Springer, G.F. Tn epitope (N-acetyl-d-galactosamine alpha-O-serine/threonine) density in primary breast carcinoma: A functional predictor of aggressiveness. Mol. Immunol. 1989, 26, 1–5. [Google Scholar] [CrossRef]
- Fujita-Yamaguchi, Y. Renewed interest in basic and applied research involving monoclonal antibodies against an oncofetal Tn-antigen. J. Biochem. 2013, 154, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Ju, T.; Aryal, R.P.; Kudelka, M.R.; Wang, Y.; Cummings, R.D. The Cosmc connection to the Tn antigen in cancer. Cancer Biomark. 2014, 14, 63–81. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, L.R.; Carrascal, M.A.; Barbas, A.; Ramalho, J.S.; Novo, C.; Delannoy, P.; Videira, P.A. Challenges in Antibody Development against Tn and Sialyl-Tn Antigens. Biomolecules 2015, 5, 1783–1809. [Google Scholar] [CrossRef] [PubMed]
- Holmberg, L.A.; Sandmaier, B.M. Vaccination with Theratope (STn-KLH) as treatment for breast cancer. Expert Rev. Vaccines 2004, 3, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.G. The oncofetal Thomsen-Friedenreich carbohydrate antigen in cancer progression. Glycoconj. J. 2007, 24, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Baek, M.G. Glycodendrimers: Novel glycotope isosteres unmasking sugar coding. Case study with T-antigen markers from breast cancer MUC1 glycoprotein. J. Biotechnol. 2002, 90, 291–309. [Google Scholar] [CrossRef]
- Springer, G.F. Immunoreactive T and Tn epitopes in cancer diagnosis, prognosis, and immunotherapy. J. Mol. Med. 1997, 75, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Hanisch, F.G.; Baldus, S.E. The Thomsen-Friedenreich (TF) antigen: A critical review on the structural, biosynthetic and histochemical aspects of a pancarcinoma-associated antigen. Histol. Histopathol. 1997, 12, 263–281. [Google Scholar] [PubMed]
- Langkilde, N.C. T-antigens in primary non-invasive and superficially invasive human urinary bladder tumors: The correlation to tumor recurrence and tumor progression. A mini-review. Scand. J. Urol. Nephrol. Suppl. 1995, 172, 45–49. [Google Scholar] [PubMed]
- Malekan, H.; Fung, G.; Thon, V.; Khedri, Z.; Yu, H.; Qu, J.; Li, Y.; Ding, L.; Lam, K.S.; Chen, X. One-pot multi-enzyme (OPME) chemoenzymatic synthesis of sialyl-Tn-MUC1 and sialyl-T-MUC1 glycopeptides containing natural or non-natural sialic acid. Bioorg. Med. Chem. 2013, 21, 4778–4785. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Kinoshita, M.; Hayakawa, T.; Nakaya, S.; Kakehi, K. Comparative studies on the structural features of O-glycans between leukemia and epithelial cell lines. J. Proteome Res. 2009, 8, 521–537. [Google Scholar] [CrossRef] [PubMed]
- Motari, E.; Zheng, X.; Su, X.; Liu, Y.; Kvaratskhelia, M.; Freitas, M.; Wang, P.G. Analysis of Recombinant CD24 Glycans by MALDI-TOF-MS Reveals Prevalence of Sialyl-T Antigen. Am. J. Biomed. Sci. 2009, 1, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rajpert-De Meyts, E.; Poll, S.N.; Goukasian, I.; Jeanneau, C.; Herlihy, A.S.; Bennett, E.P.; Skakkebaek, N.E.; Clausen, H.; Giwercman, A.; Mandel, U. Changes in the profile of simple mucin-type O-glycans and polypeptide GalNAc-transferases in human testis and testicular neoplasms are associated with germ cell maturation and tumour differentiation. Virchows Arch. 2007, 451, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Becker, T.; Dziadek, S.; Wittrock, S.; Kunz, H. Synthetic glycopeptides from the mucin family as potential tools in cancer immunotherapy. Curr. Cancer Drug Targets 2006, 6, 491–517. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, E.V.; Xue, J.; Neelamegham, S.; Matta, K.L. The pattern of glycosyl- and sulfotransferase activities in cancer cell lines: A predictor of individual cancer-associated distinct carbohydrate structures for the structural identification of signature glycans. Carbohydr. Res. 2006, 341, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Pudelko, M.; Lindgren, A.; Tengel, T.; Reis, C.A.; Elofsson, M.; Kihlberg, J. More immunogenic Formation of lactones from sialylated MUC1 glycopeptides. Org. Biomol. Chem. 2006, 4, 713–720. [Google Scholar] [CrossRef] [PubMed]
- Dziadek, S.; Brocke, C.; Kunz, H. Biomimetic synthesis of the tumor-associated (2,3)-sialyl-T antigen and its incorporation into glycopeptide antigens from the mucins MUC1 and MUC4. Chemistry 2004, 10, 4150–4162. [Google Scholar] [CrossRef] [PubMed]
- Bresalier, R.S.; Ho, S.B.; Schoeppner, H.L.; Kim, Y.S.; Sleisenger, M.H.; Brodt, P.; Byrd, J.C. Enhanced sialylation of mucin-associated carbohydrate structures in human colon cancer metastasis. Gastroenterology 1996, 110, 1354–1367. [Google Scholar] [CrossRef] [PubMed]
- Qu, J.; Yu, H.; Li, F.; Zhang, C.; Trad, A.; Brooks, C.; Zhang, B.; Gong, T.; Guo, Z.; Li, Y.; et al. Molecular basis of antibody binding to mucin glycopeptides in lung cancer. Int. J. Oncol. 2016, 48, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Cheever, M.A.; Allison, J.P.; Ferris, A.S.; Finn, O.J.; Hastings, B.M.; Hecht, T.T.; Mellman, I.; Prindiville, S.A.; Viner, J.L.; Weiner, L.M.; et al. The prioritization of cancer antigens: A national cancer institute pilot project for the acceleration of translational research. Clin. Cancer Res. 2009, 15, 5323–5337. [Google Scholar] [CrossRef] [PubMed]
- Finn, O.J. Human Tumor Antigens Yesterday, Today, and Tomorrow. Cancer Immunol. Res. 2017, 5, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Von Mensdorff-Pouilly, S.; Moreno, M.; Verheijen, R.H. Natural and Induced Humoral Responses to MUC1. Cancers 2011, 3, 3073–3103. [Google Scholar] [CrossRef] [PubMed]
- Karsten, U.; Serttas, N.; Paulsen, H.; Danielczyk, A.; Goletz, S. Binding patterns of DTR-specific antibodies reveal a glycosylation-conditioned tumor-specific epitope of the epithelial mucin (MUC1). Glycobiology 2004, 14, 681–692. [Google Scholar] [CrossRef] [PubMed]
- Coltart, D.M.; Royyuru, A.K.; Williams, L.J.; Glunz, P.W.; Sames, D.; Kuduk, S.D.; Schwarz, J.B.; Chen, X.T.; Danishefsky, S.J.; Live, D.H. Principles of mucin architecture: Structural studies on synthetic glycopeptides bearing clustered mono-, di-, tri-, and hexasaccharide glycodomains. J. Am. Chem. Soc. 2002, 124, 9833–9844. [Google Scholar] [CrossRef] [PubMed]
- Lakshminarayanan, V.; Thompson, P.; Wolfert, M.A.; Buskas, T.; Bradley, J.M.; Pathangey, L.B.; Madsen, C.S.; Cohen, P.A.; Gendler, S.J.; Boons, G.J. Immune recognition of tumor-associated mucin MUC1 is achieved by a fully synthetic aberrantly glycosylated MUC1 tripartite vaccine. Proc. Natl. Acad. Sci. USA 2012, 109, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Lohmueller, J.J.; Sato, S.; Popova, L.; Chu, I.M.; Tucker, M.A.; Barberena, R.; Innocenti, G.M.; Cudic, M.; Ham, J.D.; Cheung, W.C.; et al. Antibodies elicited by the first non-viral prophylactic cancer vaccine show tumor-specificity and immunotherapeutic potential. Sci. Rep. 2016, 6, 31740. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, A.L.; Reis, C.A.; Tarp, M.A.; Mandel, U.; Ramachandran, K.; Sankaranarayanan, V.; Schwientek, T.; Graham, R.; Taylor-Papadimitriou, J.; Hollingsworth, M.A.; et al. Chemoenzymatically synthesized multimeric Tn/STn MUC1 glycopeptides elicit cancer-specific anti-MUC1 antibody responses and override tolerance. Glycobiology 2006, 16, 96–107. [Google Scholar] [CrossRef] [PubMed]
- Posey, A.D., Jr.; Schwab, R.D.; Boesteanu, A.C.; Steentoft, C.; Mandel, U.; Engels, B.; Stone, J.D.; Madsen, T.D.; Schreiber, K.; Haines, K.M.; et al. Engineered CAR T Cells Targeting the Cancer-Associated Tn-Glycoform of the Membrane Mucin MUC1 Control Adenocarcinoma. Immunity 2016, 44, 1444–1454. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Delyria, E.S.; Chen, J.; Huang, W.; Lee, J.S.; Mittendorf, E.A.; Ibrahim, N.; Radvanyi, L.G.; Li, Y.; Lu, H.; et al. MUC1 glycopeptide epitopes predicted by computational glycomics. Int. J. Oncol. 2012, 41, 1977–1984. [Google Scholar] [CrossRef] [PubMed]
- Dingjan, T.; Spendlove, I.; Durrant, L.G.; Scott, A.M.; Yuriev, E.; Ramsland, P.A. Structural biology of antibody recognition of carbohydrate epitopes and potential uses for targeted cancer immunotherapies. Mol. Immunol. 2015, 67, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Soliman, C.; Yuriev, E.; Ramsland, P.A. Antibody recognition of aberrant glycosylation on the surface of cancer cells. Curr. Opin. Struct. Biol. 2017, 44, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Thor, A.; Ohuchi, N.; Szpak, C.A.; Johnston, W.W.; Schlom, J. Distribution of oncofetal antigen tumor-associated glycoprotein-72 defined by monoclonal antibody B72.3. Cancer Res. 1986, 46, 3118–3124. [Google Scholar] [PubMed]
- Cao, Y.; Stosiek, P.; Springer, G.F.; Karsten, U. Thomsen-Friedenreich-related carbohydrate antigens in normal adult human tissues: A systematic and comparative study. Histochem. Cell Biol. 1996, 106, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Brooks, C.L.; Schietinger, A.; Borisova, S.N.; Kufer, P.; Okon, M.; Hirama, T.; Mackenzie, C.R.; Wang, L.X.; Schreiber, H.; Evans, S.V. Antibody recognition of a unique tumor-specific glycopeptide antigen. Proc. Natl. Acad. Sci. USA 2010, 107, 10056–10061. [Google Scholar] [CrossRef] [PubMed]
- Schietinger, A.; Philip, M.; Yoshida, B.A.; Azadi, P.; Liu, H.; Meredith, S.C.; Schreiber, H. A mutant chaperone converts a wild-type protein into a tumor-specific antigen. Science 2006, 314, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Raica, M.; Cimpean, A.M.; Ribatti, D. The role of podoplanin in tumor progression and metastasis. Anticancer Res. 2008, 28, 2997–3006. [Google Scholar] [PubMed]
- Klein, F.; Diskin, R.; Scheid, J.F.; Gaebler, C.; Mouquet, H.; Georgiev, I.S.; Pancera, M.; Zhou, T.; Incesu, R.B.; Fu, B.Z.; et al. Somatic mutations of the immunoglobulin framework are generally required for broad and potent HIV-1 neutralization. Cell 2013, 15, 126–138. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, N.; Ogawa, H.; Koizumi, T.; Tsukamoto, K.; Matsumoto-Takasaki, A.; Asanuma, H.; Nakada, H.; Fujita-Yamaguchi, Y. Construction and expression of anti-Tn-antigen-specific single-chain antibody genes from hybridoma producing MLS128 monoclonal antibody. J. Biochem. 2012, 151, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Babino, A.; Pritsch, O.; Oppezzo, P.; Du Pasquier, R.; Roseto, A.; Osinaga, E.; Alzari, P.M. Molecular cloning of a monoclonal anti-tumor antibody specific for the Tn antigen and expression of an active single-chain Fv fragment. Hybridoma 1997, 16, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Price, M.R.; Rye, P.D.; Petrakou, E.; Murray, A.; Brady, K.; Imai, S.; Haga, S.; Kiyozuka, Y.; Schol, D.; Meulenbroek, M.F.; et al. Summary report on the ISOBM TD-4 Workshop: Analysis of 56 monoclonal antibodies against the MUC1 mucin. Tumour Biol. 1998, 19 (Suppl. 1), 1–20. [Google Scholar] [CrossRef] [PubMed]
- Dokurno, P.; Bates, P.A.; Band, H.A.; Stewart, L.M.; Lally, J.M.; Burchell, J.M.; Taylor-Papadimitriou, J.; Snary, D.; Sternberg, M.J.; Freemont, P.S. Crystal structure at 1.95 A resolution of the breast tumour-specific antibody SM3 complexed with its peptide epitope reveals novel hypervariable loop recognition. J. Mol. Biol. 1998, 284, 713–728. [Google Scholar] [PubMed]
- Burchell, J.; Gendler, S.; Taylor-Papadimitriou, J.; Girling, A.; Lewis, A.; Millis, R.; Lamport, D. Development and characterization of breast cancer reactive monoclonal antibodies directed to the core protein of the human milk mucin. Cancer Res. 1987, 47, 5476–5482. [Google Scholar] [PubMed]
- Girling, A.; Bartkova, J.; Burchell, J.; Gendler, S.; Gillett, C.; Taylorpapadimitriou, J. A core protein epitope of the polymorphic epithelial mucin detected by the monoclonal antibody SM-3 is selectively exposed in a range of primary carcinomas. Int. J. Cancer 1989, 43, 1072–1076. [Google Scholar] [CrossRef] [PubMed]
- Möller, H.; Serttas, N.; Paulsen, H.; Burchell, J.M.; Taylor-Papadimitriou, J.; Meyer, B. NMR-based determination of the binding epitope and conformational analysis of MUC-1 glycopeptides and peptides bound to the breast cancer-selective monoclonal antibody SM3. Eur. J. Biochem. 2002, 269, 1444–1455. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sáez, N.; Castro-López, J.; Valero-González, J.; Madariaga, D.; Compañón, I.; Somovilla, V.J.; Salvadó, M.; Asensio, J.L.; Jiménez-Barbero, J.; Avenoza, A.; et al. Deciphering the Non-Equivalence of Serine and Threonine O-Glycosylation Points: Implications for Molecular Recognition of the Tn Antigen by an anti-MUC1 Antibody. Angew. Chem. Int. Ed. Engl. 2015, 54, 9830–9834. [Google Scholar] [CrossRef] [PubMed]
- Barchi, J.J., Jr. Mucin-type glycopeptide structure in solution: Past, present, and future. Biopolymers 2013, 99, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Kinarsky, L.; Suryanarayanan, G.; Prakash, O.; Paulsen, H.; Clausen, H.; Hanisch, F.G.; Hollingsworth, M.A.; Sherman, S. Conformational studies on the MUC1 tandem repeat glycopeptides: Implication for the enzymatic O-glycosylation of the mucin protein core. Glycobiology 2003, 13, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, T.; Ohyabu, N.; Fujitani, N.; Naruchi, K.; Shimizu, H.; Hinou, H.; Nishimura, S. Site-specific conformational alteration induced by sialylation of MUC1 tandem repeating glycopeptides at an epitope region for the anti-KL-6 monoclonal antibody. Biochemistry 2013, 52, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Schuman, J.; Campbell, A.P.; Koganty, R.R.; Longenecker, B.M. Probing the conformational and dynamical effects of O-glycosylation within the immunodominant region of a MUC1 peptide tumor antigen. Chem. Biol. Drug Des. 2003, 61, 91–108. [Google Scholar] [CrossRef]
- Movahedin, M.; Brooks, T.M.; Supekar, N.T.; Gokanapudi, N.; Boons, G.J.; Brooks, C.L. Glycosylation of MUC1 influences the binding of a therapeutic antibody by altering the conformational equilibrium of the antigen. Glycobiology 2017, 27, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.; Schultes, B.C.; Liu, D.; Kuzma, M.; Decker, W.; Madiyalakan, R. Characterization of an anti-MUC1 monoclonal antibody with potential as a cancer vaccine. Hybrid. Hybridomics 2001, 20, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Vella, M.; Pace, D. Glycoconjugate vaccines: An update. Expert Opin. Biol. Ther. 2015, 15, 529–546. [Google Scholar] [CrossRef] [PubMed]
- Perry, C.M. Meningococcal groups C and Y and haemophilus B tetanus toxoid conjugate vaccine (HibMenCY-TT; MenHibrix®): A review. Drugs 2013, 73, 703–713. [Google Scholar] [CrossRef] [PubMed]
- Blanchard-Rohner, G.; Pollard, A.J. Long-term protection after immunization with protein-polysaccharide conjugate vaccines in infancy. Expert Rev. Vaccines 2011, 10, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.K.; Murray, J.L.; Zhou, D.; Mittendorf, E.A.; Sample, D.; Tautchin, M.; Miles, D. Survival Advantage in Patients with Metastatic Breast Cancer Receiving Endocrine Therapy plus Sialyl Tn-KLH Vaccine: Post Hoc Analysis of a Large Randomized Trial. J. Cancer 2013, 4, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Lunn, M.P.; Johnson, L.A.; Fromholt, S.E.; Itonori, S.; Huang, J.; Vyas, A.A.; Hildreth, J.E.; Griffin, J.W.; Schnaar, R.L.; Sheikh, K.A. High-affinity anti-ganglioside IgG antibodies raised in complex ganglioside knockout mice: Reexamination of GD1a immunolocalization. J. Neurochem. 2000, 75, 404–412. [Google Scholar] [CrossRef] [PubMed]
- Pett, C.; Cai, H.; Liu, J.; Palitzsch, B.; Schorlemer, M.; Hartmann, S.; Stergiou, N.; Lu, M.; Kunz, H.; Schmitt, E.; et al. Microarray Analysis of Antibodies Induced with Synthetic Antitumor Vaccines: Specificity against Diverse Mucin Core Structures. Chemistry 2017, 23, 3875–3884. [Google Scholar] [CrossRef] [PubMed]
- Wandall, H.H.; Blixt, O.; Tarp, M.A.; Pedersen, J.W.; Bennett, E.P.; Mandel, U.; Ragupathi, G.; Livingston, P.O.; Hollingsworth, M.A.; Taylor-Papadimitriou, J.; et al. Cancer biomarkers defined by autoantibody signatures to aberrant O-glycopeptide epitopes. Cancer Res. 2010, 70, 1306–1313. [Google Scholar] [CrossRef] [PubMed]
- Szpak, C.A.; Johnston, W.W.; Lottich, S.C.; Kufe, D.; Thor, A.; Schlom, J. Patterns of reactivity of four novel monoclonal antibodies (B72.3, DF3, B1.1 and B6.2) with cells in human malignant and benign effusions. Acta Cytol. 1984, 28, 356–367. [Google Scholar] [PubMed]
- Rixon, M.W.; Gourlie, B.B.; Kaplan, D.A.; Schlom, J.; Mézes, P.S. Preferential use of a H chain V region in antitumor-associated glycoprotein-72 monoclonal antibodies. J. Immunol. 1993, 151, 6559–6568. [Google Scholar] [PubMed]
- Sakai, K.; Yuasa, N.; Tsukamoto, K.; Takasaki-Matsumoto, A.; Yajima, Y.; Sato, R.; Kawakami, H.; Mizuno, M.; Takayanagi, A.; Shimizu, N.; et al. Isolation and characterization of antibodies against three consecutive Tn-antigen clusters from a phage library displaying human single-chain variable fragments. J. Biochem. 2010, 147, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Kato, K.; Denda-Nagai, K.; Hanisch, F.G.; Clausen, H.; Irimura, T. The epitope recognized by the unique anti-MUC1 monoclonal antibody MY.1E12 involves sialyl alpha 2-3galactosyl beta 1-3N-acetylgalactosaminide linked to a distinct threonine residue in the MUC1 tandem repeat. J. Immunol. Methods 2002, 270, 199–209. [Google Scholar] [CrossRef]
- Tsubura, A.; Morii, S.; Hilkens, J.; Hilgers, J. Expression of MAM-3 and MAM-6 antigens in endometrial and endocervical adenocarcinomas. Virchows Arch. A Pathol. Anat. Histopathol. 1985, 407, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Ohyabu, N.; Hinou, H.; Matsushita, T.; Izumi, R.; Shimizu, H.; Kawamoto, K.; Numata, Y.; Togame, H.; Takemoto, H.; Kondo, H.; et al. An essential epitope of anti-MUC1 monoclonal antibody KL-6 revealed by focused glycopeptide library. J. Am. Chem. Soc. 2009, 131, 17102–17109. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, T.; Takada, W.; Igarashi, K.; Naruchi, K.; Miyoshi, R.; Garcia-Martin, F.; Amano, M.; Hinou, H.; Nishimura, S. A straightforward protocol for the preparation of high performance microarray displaying synthetic MUC1 glycopeptides. Biochim. Biophys. Acta 2014, 1840, 1105–1116. [Google Scholar] [CrossRef] [PubMed]
- Saunders, K.O.; Nicely, N.I.; Wiehe, K.; Bonsignori, M.; Meyerhoff, R.R.; Parks, R.; Walkowicz, W.E.; Aussedat, B.; Wu, N.R.; Cai, F.; et al. Vaccine Elicitation of High Mannose-Dependent Neutralizing Antibodies against the V3-Glycan Broadly Neutralizing Epitope in Nonhuman Primates. Cell Rep. 2017, 18, 2175–2188. [Google Scholar] [CrossRef] [PubMed]
- Nuhn, L.; Hartmann, S.; Palitzsch, B.; Gerlitzki, B.; Schmitt, E.; Zentel, R.; Kunz, H. Water-soluble polymers coupled with glycopeptide antigens and T-cell epitopes as potential antitumor vaccines. Angew. Chem. Int. Ed. Engl. 2013, 52, 10652–10656. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, S.; Nuhn, L.; Palitzsch, B.; Glaffig, M.; Stergiou, N.; Gerlitzki, B.; Schmitt, E.; Kunz, H.; Zentel, R. CpG-loaded multifunctional cationic nanohydrogel particles as self-adjuvanting glycopeptide antitumor vaccines. Adv. Healthc. Mater. 2015, 4, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Glaffig, M.; Palitzsch, B.; Hartmann, S.; Schüll, C.; Nuhn, L.; Gerlitzki, B.; Schmitt, E.; Frey, H.; Kunz, H. A fully synthetic glycopeptide antitumor vaccine based on multiple antigen presentation on a hyperbranched polymer. Chemistry 2014, 20, 4232–4236. [Google Scholar] [CrossRef] [PubMed]
- Glaffig, M.; Palitzsch, B.; Stergiou, N.; Schüll, C.; Strassburger, D.; Schmitt, E.; Frey, H.; Kunz, H. Enhanced immunogenicity of multivalent MUC1 glycopeptide antitumour vaccines based on hyperbranched polymers. Org. Biomol. Chem. 2015, 13, 10150–10154. [Google Scholar] [CrossRef] [PubMed]
- Diwan, M.; Elamanchili, P.; Lane, H.; Gainer, A.; Samuel, J. Biodegradable nanoparticle mediated antigen delivery to human cord blood derived dendritic cells for induction of primary T cell responses. J. Drug Target 2003, 11, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Newman, K.D.; Sosnowski, D.L.; Kwon, G.S.; Samuel, J. Delivery of MUC1 mucin peptide by Poly(d,l-lactic-co-glycolic acid) microspheres induces type 1 T helper immune responses. J. Pharm. Sci. 1998, 87, 1421–1427. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Chowdhury, S.; McKay, C.; Baniel, C.; Wright, W.S.; Bentley, P.; Kaczanowska, K.; Gildersleeve, J.C.; Finn, M.G.; BenMohamed, L.; et al. Significant Impact of Immunogen Design on the Diversity of Antibodies Generated by Carbohydrate-Based Anticancer Vaccine. ACS Chem. Biol. 2015, 10, 2364–2372. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Comellas-Aragones, M.; Chowdhury, S.; Bentley, P.; Kaczanowska, K.; Benmohamed, L.; Gildersleeve, J.C.; Finn, M.G.; Huang, X. Boosting immunity to small tumor-associated carbohydrates with bacteriophage qβ capsids. ACS Chem. Biol. 2013, 8, 1253–1262. [Google Scholar] [CrossRef] [PubMed]
- Rivalland, G.; Loveland, B.; Mitchell, P. Update on Mucin-1 immunotherapy in cancer: A clinical perspective. Expert Opin. Biol. Ther. 2015, 15, 1773–1787. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.K.; Wall, K.A. Immunological Evaluation of Recent MUC1 Glycopeptide Cancer Vaccines. Vaccines 2016, 4, 25. [Google Scholar] [CrossRef] [PubMed]
- Scheid, E.; Major, P.; Bergeron, A.; Finn, O.J.; Salter, R.D.; Eady, R.; Yassine-Diab, B.; Favre, D.; Peretz, Y.; Landry, C.; et al. Tn-MUC1 DC Vaccination of Rhesus Macaques and a Phase I/II Trial in Patients with Nonmetastatic Castrate-Resistant Prostate Cancer. Cancer Immunol. Res. 2016, 4, 881–892. [Google Scholar] [CrossRef] [PubMed]
- Cipolla, L.; Rescigno, M.; Leone, A.; Peri, F.; La Ferla, B.; Nicotra, F. Novel Tn antigen-containing neoglycopeptides: Synthesis and evaluation as anti tumor vaccines. Bioorg. Med. Chem. 2002, 10, 1639–1646. [Google Scholar] [CrossRef]
- Corzana, F.; Busto, J.H.; Marcelo, F.; de Luis, M.G.; Asensio, J.L.; Martin-Santamaria, S.; Saenz, Y.; Torres, C.; Jimenez-Barbero, J.; Avenoza, A.; et al. Rational design of a Tn antigen mimic. Chem. Commun. 2011, 47, 5319–5321. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Ocariz, V.; Companon, I.; Aydillo, C.; Castro-Lopez, J.; Jimenez-Barbero, J.; Hurtado-Guerrero, R.; Avenoza, A.; Zurbano, M.M.; Peregrina, J.M.; Busto, J.H.; et al. Design of alpha-S-Neoglycopeptides Derived from MUC1 with a Flexible and Solvent-Exposed Sugar Moiety. J. Org. Chem. 2016, 81, 5929–5941. [Google Scholar] [CrossRef] [PubMed]
- Leiria Campo, V.; Riul, T.B.; Oliveira Bortot, L.; Martins-Teixeira, M.B.; Fiori Marchiori, M.; Iaccarino, E.; Ruvo, M.; Dias-Baruffi, M.; Carvalho, I. A Synthetic MUC1 Glycopeptide Bearing betaGalNAc-Thr as a Tn Antigen Isomer Induces the Production of Antibodies against Tumor Cells. Chembiochem 2017, 18, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann-Roder, A.; Kaiser, A.; Wagner, S.; Gaidzik, N.; Kowalczyk, D.; Westerlind, U.; Gerlitzki, B.; Schmitt, E.; Kunz, H. Synthetic antitumor vaccines from tetanus toxoid conjugates of MUC1 glycopeptides with the Thomsen-Friedenreich antigen and a fluorine-substituted analogue. Angew. Chem. Int. Ed. Engl. 2010, 49, 8498–8503. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Guo, Z. Improving the antigenicity of sTn antigen by modification of its sialic acid residue for development of glycoconjugate cancer vaccines. Bioconjug. Chem. 2006, 17, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Ekanayaka, S.A.; Wu, J.; Zhang, J.; Guo, Z. Synthetic and immunological studies of 5′-N-phenylacetyl sTn to develop carbohydrate-based cancer vaccines and to explore the impacts of linkage between carbohydrate antigens and carrier proteins. Bioconjug. Chem. 2008, 19, 2060–2067. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Guo, Z. Synthetic and Immunological Studies of sTn Derivatives Carrying 5-N-(p-Substituted Phenylacetyl)Sialic Acid for the Development of Effective Cancer Vaccines. ACS Med. Chem. Lett. 2011, 2, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Yu, H.; Lau, K.; Li, Y.; Muthana, S.; Wang, J.; Chen, X. Efficient chemoenzymatic synthesis of sialyl Tn-antigens and derivatives. Chem. Commun. 2011, 47, 8691–8693. [Google Scholar] [CrossRef] [PubMed]
- Huo, C.X.; Zheng, X.J.; Xiao, A.; Liu, C.C.; Sun, S.; Lv, Z.; Ye, X.S. Synthetic and immunological studies of N-acyl modified S-linked STn derivatives as anticancer vaccine candidates. Org. Biomol. Chem. 2015, 13, 3677–3690. [Google Scholar] [CrossRef] [PubMed]
- Tarp, M.A.; Sørensen, A.L.; Mandel, U.; Paulsen, H.; Burchell, J.; Taylor-Papadimitriou, J.; Clausen, H. Identification of a novel cancer-specific immunodominant glycopeptide epitope in the MUC1 tandem repeat. Glycobiology 2007, 17, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.K.; Yariz, K.O.; Bondarenko, I.; Manikhas, A.; Semiglazov, V.; Alyasova, A.; Komisarenko, V.; Shparyk, Y.; Murray, J.L.; Jones, D.; et al. Randomized phase II trial of letrozole plus anti-MUC1 antibody AS1402 in hormone receptor-positive locally advanced or metastatic breast cancer. Clin. Cancer Res. 2011, 17, 6822–6830. [Google Scholar] [CrossRef] [PubMed]
- Fiedler, W.; DeDosso, S.; Cresta, S.; Weidmann, J.; Tessari, A.; Salzberg, M.; Dietrich, B.; Baumeister, H.; Goletz, S.; Gianni, L.; et al. A phase I study of PankoMab-GEX, a humanised glyco-optimised monoclonal antibody to a novel tumour-specific MUC1 glycopeptide epitope in patients with advanced carcinomas. Eur. J. Cancer. 2016, 63, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Verheijen, R.H.; Massuger, L.F.; Benigno, B.B.; Epenetos, A.A.; Lopes, A.; Soper, J.T.; Markowska, J.; Vyzula, R.; Jobling, T.; Stamp, G.; et al. Phase III trial of intraperitoneal therapy with yttrium-90-labeled HMFG1 murine monoclonal antibody in patients with epithelial ovarian cancer after a surgically defined complete remission. J. Clin. Oncol. 2006, 24, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Hughes, O.D.; Bishop, M.C.; Perkins, A.C.; Wastie, M.L.; Denton, G.; Price, M.R.; Frier, M.; Denley, H.; Rutherford, R.; Schubiger, P.A. Targeting superficial bladder cancer by the intravesical administration of copper-67-labeled anti-MUC1 mucin monoclonal antibody C595. J. Clin. Oncol. 2000, 18, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, A.M.; Broadhead, T.J.; Chan, S.Y.; Owen, J.; Farnsworth, A.P.; Sopwith, M.; Coleman, R.E. Phase I open study of the effects of ascending doses of the cytotoxic immunoconjugate CMB-401 (hCTMO1-calicheamicin) in patients with epithelial ovarian cancer. Ann. Oncol. 2000, 11, 735–741. [Google Scholar] [CrossRef] [PubMed]
- Kramer, E.L.; Liebes, L.; Wasserheit, C.; Noz, M.E.; Blank, E.W.; Zabalegui, A.; Melamed, J.; Furmanski, P.; Peterson, J.A.; Ceriani, R.L. Initial clinical evaluation of radiolabeled MX-DTPA humanized BrE-3 antibody in patients with advanced breast cancer. Clin. Cancer Res. 1998, 4, 1679–1688. [Google Scholar] [PubMed]
- Kunkler, R.B.; Bishop, M.C.; Green, D.J.; Pimm, M.V.; Price, M.R.; Frier, M. Targeting of bladder cancer with monoclonal antibody NCRC48—A possible approach for intravesical therapy. Br. J. Urol. 1995, 76, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Kramer, E.L.; DeNardo, S.J.; Liebes, L.; Kroger, L.A.; Noz, M.E.; Mizrachi, H.; Salako, Q.A.; Furmanski, P.; Glenn, S.D.; DeNardo, G.L.; et al. Radioimmunolocalization of metastatic breast carcinoma using indium-111-methyl benzyl DTPA BrE-3 monoclonal antibody: Phase I study. J. Nucl. Med. 1993, 34, 1067–1074. [Google Scholar] [PubMed]
- Ahmad, R.; Alam, M.; Rajabi, H.; Kufe, D. The MUC1-C oncoprotein binds to the BH3 domain of the pro-apoptotic BAX protein and blocks BAX function. J. Biol. Chem. 2012, 287, 20866–20875. [Google Scholar] [CrossRef] [PubMed]
- Uchida, J.; Hamaguchi, Y.; Oliver, J.A.; Ravetch, J.V.; Poe, J.C.; Haas, K.M.; Tedder, T.F. The innate mononuclear phagocyte network depletes B lymphocytes through Fc receptor-dependent mechanisms during anti-CD20 antibody immunotherapy. J. Exp. Med. 2004, 199, 1659–1669. [Google Scholar] [CrossRef] [PubMed]
- Pandey, J.P.; Namboodiri, A.M. Genetic variants of IgG1 antibodies and FcγRIIIa receptors influence the magnitude of antibody-dependent cell-mediated cytotoxicity against prostate cancer cells. Oncoimmunology 2014, 3, e27317. [Google Scholar] [CrossRef] [PubMed]
- Lavrsen, K.; Madsen, C.B.; Rasch, M.G.; Woetmann, A.; Ødum, N.; Mandel, U.; Clausen, H.; Pedersen, A.E.; Wandall, H.H. Aberrantly glycosylated MUC1 is expressed on the surface of breast cancer cells and a target for antibody-dependent cell-mediated cytotoxicity. Glycoconj. J. 2013, 30, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Diéras, V.; Miles, D.; Verma, S.; Pegram, M.; Welslau, M.; Baselga, J.; Krop, I.E.; Blackwell, K.; Hoersch, S.; Xu, J.; et al. Trastuzumab emtansine versus capecitabine plus lapatinib in patients with previously treated HER2-positive advanced breast cancer (EMILIA): A descriptive analysis of final overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2017, 18, 732–742. [Google Scholar] [CrossRef]
- Prince, H.M.; Kim, Y.H.; Horwitz, S.M.; Dummer, R.; Scarisbrick, J.; Quaglino, P.; Zinzani, P.L.; Wolter, P.; Sanches, J.A.; Ortiz-Romero, P.L.; et al. Brentuximab vedotin or physician’s choice in CD30-positive cutaneous T-cell lymphoma (ALCANZA): An international, open-label, randomised, phase 3, multicentre trial. Lancet 2017, 390, 555–566. [Google Scholar] [CrossRef]
- Pollard, J.A.; Loken, M.; Gerbing, R.B.; Raimondi, S.C.; Hirsch, B.A.; Aplenc, R.; Bernstein, I.D.; Gamis, A.S.; Alonzo, T.A.; Meshinchi, S. CD33 Expression and Its Association with Gemtuzumab Ozogamicin Response: Results From the Randomized Phase III Children’s Oncology Group Trial AAML0531. J. Clin. Oncol. 2016, 34, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Roca, C.A.; Boni, V.; Moreno, V.; Morris, J.C.; DeLord, J.-P.; Calvo, E.; Papadopoulos, K.P.; Rixe, O.; Cohen, P.; Tellier, A.; et al. A phase I study of SAR566658, an anti CA6-antibody drug conjugate (ADC), in patients (Pts) with CA6-positive advanced solid tumors (STs) (NCT01156870). J. Clin. Oncol. 2016, 34, 2511. [Google Scholar]
- Prendergast, J.M.; Galvao da Silva, A.P.; Eavarone, D.A.; Ghaderi, D.; Zhang, M.; Brady, D.; Wicks, J.; DeSander, J.; Behrens, J.; Rueda, B.R. Novel anti-Sialyl-Tn monoclonal antibodies and antibody-drug conjugates demonstrate tumor specificity and anti-tumor activity. MAbs 2017, 9, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Carter, P.; Smith, L.; Ryan, M. Identification and validation of cell surface antigens for antibody targeting in oncology. Endocr. Relat. Cancer 2004, 11, 659–687. [Google Scholar] [CrossRef] [PubMed]
- Dokter, W.; Ubink, R.; van der Lee, M.; van der Vleuten, M.; van Achterberg, T.; Jacobs, D.; Loosveld, E.; van den Dobbelsteen, D.; Egging, D.; Mattaar, E.; et al. Preclinical profile of the HER2-targeting ADC SYD983/SYD985: Introduction of a new duocarmycin-based linker-drug platform. Mol. Cancer Ther. 2014, 13, 2618–2629. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.; Wang, Z.; Hao, M.; Li, J. Bispecific antibodies and their applications. J. Hematol. Oncol. 2015, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Yuraszeck, T.; Kasichayanula, S.; Benjamin, J.E. Translation and Clinical Development of Bispecific T-cell Engaging Antibodies for Cancer Treatment. Clin. Pharmacol. Ther. 2017, 101, 634–645. [Google Scholar] [CrossRef] [PubMed]
- Danielczyk, A.; Stahn, R.; Faulstich, D.; Löffler, A.; Märten, A.; Karsten, U.; Goletz, S. PankoMab: A potent new generation anti-tumour MUC1 antibody. Cancer Immunol. Immunother. 2006, 55, 1337–1347. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Lai, Y.; Li, J.; Qin, L.; Xu, Y.; Zhao, R.; Li, B.; Lin, S.; Wang, S.; Wu, Q.; et al. PSCA and MUC1 in non-small-cell lung cancer as targets of chimeric antigen receptor T cells. Oncoimmunology 2017, 6, e1284722. [Google Scholar] [CrossRef] [PubMed]
- Posey, A.D., Jr.; Clausen, H.; June, C.H. Distinguishing Truncated and Normal MUC1 Glycoform Targeting from Tn-MUC1-Specific CAR T Cells: Specificity Is the Key to Safety. Immunity 2016, 45, 947–948. [Google Scholar] [CrossRef] [PubMed]
- Steentoft, C.; Migliorini, D.; King, T.R.; Mandel, U.; June, C.H.; Posey, A.D., Jr. Glycan-directed CAR-t cells. Glycobiology 2018. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.; Wilkie, S.; Davies, D.M.; Arif, S.; Picco, G.; Julien, S.; Foster, J.; Burchell, J.; Taylor-Papadimitriou, J. Targeting of Tumor-Associated Glycoforms of MUC1 with CAR T Cells. Immunity 2016, 45, 945–946. [Google Scholar] [CrossRef] [PubMed]
- You, F.; Jiang, L.; Zhang, B.; Lu, Q.; Zhou, Q.; Liao, X.; Wu, H.; Du, K.; Zhu, Y.; Meng, H.; et al. Phase 1 clinical trial demonstrated that MUC1 positive metastatic seminal vesicle cancer can be effectively eradicated by modified Anti-MUC1 chimeric antigen receptor transduced T cells. Sci. China Life Sci. 2016, 59, 386–397. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Chan, R.; Bajgain, P.; Rambally, S.; Palapattu, G.; Mims, M.; Rooney, C.M.; Leen, A.M.; Brenner, M.K.; Vera, J.F. Combining T-cell immunotherapy and anti-androgen therapy for prostate cancer. Prostate Cancer Prostatic Dis. 2013, 16 (Suppl. 1), 123–131. [Google Scholar] [CrossRef] [PubMed]
- Wilkie, S.; van Schalkwyk, M.C.; Hobbs, S.; Davies, D.M.; van der Stegen, S.J.; Pereira, A.C.; Burbridge, S.E.; Box, C.; Eccles, S.A.; Maher, J. Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling. J. Clin. Immunol. 2012, 32, 1059–1070. [Google Scholar] [CrossRef] [PubMed]
- Wilkie, S.; Picco, G.; Foster, J.; Davies, D.M.; Julien, S.; Cooper, L.; Arif, S.; Mather, S.J.; Taylor-Papadimitriou, J.; Burchell, J.M.; et al. Retargeting of human T cells to tumor-associated MUC1: The evolution of a chimeric antigen receptor. J. Immunol. 2008, 180, 4901–4909. [Google Scholar] [CrossRef] [PubMed]
- Hege, K.M.; Bergsland, E.K.; Fisher, G.A.; Nemunaitis, J.J.; Warren, R.S.; McArthur, J.G.; Lin, A.A.; Schlom, J.; June, C.H.; Sherwin, S.A. Safety, tumor trafficking and immunogenicity of chimeric antigen receptor (CAR)-T cells specific for TAG-72 in colorectal cancer. J. Immunother. Cancer 2017, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Jiang, S.; Fang, C.; Li, H.; Zhang, X.; Zhang, F.; June, C.H.; Zhao, Y. Novel T cells with improved in vivo anti-tumor activity generated by RNA electroporation. Protein Cell 2017, 8, 514–526. [Google Scholar] [CrossRef] [PubMed]
- Beatty, G.L.; Haas, A.R.; Maus, M.V.; Torigian, D.A.; Soulen, M.C.; Plesa, G.; Chew, A.; Zhao, Y.; Levine, B.L.; Albelda, S.M.; et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol. Res. 2014, 2, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Romanski, A.; Uherek, C.; Bug, G.; Seifried, E.; Klingemann, H.; Wels, W.S.; Ottmann, O.G.; Tonn, T. CD19-CAR engineered NK-92 cells are sufficient to overcome NK cell resistance in B-cell malignancies. J. Cell. Mol. Med. 2016, 20, 1287–1294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Burger, M.C.; Jennewein, L.; Genßler, S.; Schönfeld, K.; Zeiner, P.; Hattingen, E.; Harter, P.N.; Mittelbronn, M.; Tonn, T.; et al. ErbB2/HER2-Specific NK Cells for Targeted Therapy of Glioblastoma. J. Natl. Cancer Inst. 2015, 108, djv375. [Google Scholar] [CrossRef] [PubMed]
- Suck, G.; Odendahl, M.; Nowakowska, P.; Seidl, C.; Wels, W.S.; Klingemann, H.G.; Tonn, T. NK-92: An ‘off-the-shelf therapeutic’ for adoptive natural killer cell-based cancer immunotherapy. Cancer Immunol. Immunother. 2016, 65, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Bollino, D.; Webb, T.J. Chimeric antigen receptor-engineered natural killer and natural killer T cells for cancer immunotherapy. Transl. Res. 2017, 187, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Du, S.H.; Li, Z.; Chen, C.; Tan, W.K.; Chi, Z.; Kwang, T.W.; Xu, X.H.; Wang, S. Co-Expansion of Cytokine-Induced Killer Cells and Vγ9Vδ2 T Cells for CAR T-Cell Therapy. PLoS ONE 2016, 11, e0161820. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.; Courtney, A.N.; Jena, B.; Heczey, A.; Liu, D.; Marinova, E.; Guo, L.; Xu, X.; Torikai, H.; Mo, Q.; et al. CD62L+ NKT cells have prolonged persistence and antitumor activity in vivo. J. Clin. Investig. 2016, 126, 2341–2355. [Google Scholar] [CrossRef] [PubMed]
- Heczey, A.; Liu, D.; Tian, G.; Courtney, A.N.; Wei, J.; Marinova, E.; Gao, X.; Guo, L.; Yvon, E.; Hicks, J.; et al. Invariant NKT cells with chimeric antigen receptor provide a novel platform for safe and effective cancer immunotherapy. Blood 2014, 124, 2824–2833. [Google Scholar] [CrossRef] [PubMed]
- DeKosky, B.J.; Ippolito, G.C.; Deschner, R.P.; Lavinder, J.J.; Wine, Y.; Rawlings, B.M.; Varadarajan, N.; Giesecke, C.; Dörner, T.; Andrews, S.F.; et al. High-throughput sequencing of the paired human immunoglobulin heavy and light chain repertoire. Nat. Biotechnol. 2013, 31, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.C.; de Bouteiller, O.; Liu, Y.J.; Potter, K.; Banchereau, J.; Capra, J.D.; Pascual, V. Somatic hypermutation introduces insertions and deletions into immunoglobulin V genes. J. Exp. Med. 1998, 187, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Brüggemann, M.; Caskey, H.M.; Teale, C.; Waldmann, H.; Williams, G.T.; Surani, M.A.; Neuberger, M.S. A repertoire of monoclonal antibodies with human heavy chains from transgenic mice. Proc. Natl. Acad. Sci. USA 1989, 86, 6709–6713. [Google Scholar] [CrossRef] [PubMed]
- Lonberg, N.; Taylor, L.D.; Harding, F.A.; Trounstine, M.; Higgins, K.M.; Schramm, S.R.; Kuo, C.C.; Mashayekh, R.; Wymore, K.; McCabe, J.G.; et al. Antigen-specific human antibodies from mice comprising four distinct genetic modifications. Nature 1994, 368, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, I.C.; Zou, X.; Popov, A.V.; Cook, G.P.; Corps, E.M.; Humphries, S.; Ayling, C.; Goyenechea, B.; Xian, J.; Taussig, M.J.; et al. Antibody repertoires of four- and five-feature translocus mice carrying human immunoglobulin heavy chain and κ and λ light chain yeast artificial chromosomes. J. Immunol. 1999, 163, 6898–6906. [Google Scholar] [PubMed]
- Osborn, M.J.; Ma, B.; Avis, S.; Binnie, A.; Dilley, J.; Yang, X.; Lindquist, K.; Ménoret, S.; Iscache, A.L.; Ouisse, L.H.; et al. High-affinity IgG antibodies develop naturally in Ig-knockout rats carrying germline human IgH/Igκ/Igλ loci bearing the rat CH region. J. Immunol. 2013, 190, 1481–1490. [Google Scholar] [CrossRef] [PubMed]
- Brüggemann, M.; Osborn, M.J.; Ma, B.; Hayre, J.; Avis, S.; Lundstrom, B.; Buelow, R. Human antibody production in transgenic animals. Arch. Immunol. Ther. Exp. 2015, 63, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Borsotti, C.; Schickel, J.N.; Zhu, S.; Strowig, T.; Eynon, E.E.; Frleta, D.; Gurer, C.; Murphy, A.J.; Yancopoulos, G.D.; et al. A novel humanized mouse model with significant improvement of class-switched, antigen-specific antibody production. Blood 2017, 129, 959–969. [Google Scholar] [CrossRef] [PubMed]
- Staubach, S.; Razawi, H.; Hanisch, F.G. Proteomics of MUC1-containing lipid rafts from plasma membranes and exosomes of human breast carcinoma cells MCF-7. Proteomics 2009, 9, 2820–2835. [Google Scholar] [CrossRef] [PubMed]
- Aharon, A.; Sabbah, A.; Ben-Shaul, S.; Berkovich, H.; Loven, D.; Brenner, B.; Bar-Sela, G. Chemotherapy administration to breast cancer patients affects extracellular vesicles thrombogenicity and function. Oncotarget 2017, 8, 63265–63280. [Google Scholar] [CrossRef] [PubMed]
- Osteikoetxea, X.; Benke, M.; Rodriguez, M.; Pálóczi, K.; Sódar, B.W.; Szvicsek, Z.; Szabó-Taylor, K.; Vukman, K.V.; Kittel, Á.; Wiener, Z.; et al. Detection and proteomic characterization of extracellular vesicles in human pancreatic juice. Biochem. Biophys. Res. Commun. 2018, 499, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Skog, J.; Hsu, C.H.; Lessard, R.T.; Balaj, L.; Wurdinger, T.; Carter, B.S.; Breakefield, X.O.; Toner, M.; Irimia, D. Microfluidic isolation and transcriptome analysis of serum microvesicles. Lab Chip 2010, 10, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, S.S.; Dunlay, C.J.; Simeone, D.M.; Nagrath, S. Microfluidic device (ExoChip) for on-chip isolation, quantification and characterization of circulating exosomes. Lab Chip 2014, 14, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Dudani, J.S.; Gossett, D.R.; Tse, H.T.K.; Lamm, R.J.; Kulkarni, R.P.; Carlo, D.D. Rapid inertial solution exchange for enrichment and flow cytometric detection of microvesicles. Biomicrofluidics 2015, 9, 014112. [Google Scholar] [CrossRef] [PubMed]
- Son, K.J.; Rahimian, A.; Shin, D.S.; Siltanen, C.; Patel, T.; Revzin, A. Microfluidic compartments with sensing microbeads for dynamic monitoring of cytokine and exosome release from single cells. Analyst 2016, 141, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.T.; Kim, Y.J.; Bu, J.; Cho, Y.H.; Han, S.W.; Moon, B.I. High-purity capture and release of circulating exosomes using an exosome-specific dual-patterned immunofiltration (ExoDIF) device. Nanoscale 2017, 9, 13495–13505. [Google Scholar] [CrossRef] [PubMed]
- Jett, J.R.; Peek, L.J.; Fredericks, L.; Jewell, W.; Pingleton, W.W.; Robertson, J.F. EarlyCDT(R)-lung, in 1600 patients: An evaluation of its performance in routine clinical practice. Lung Cancer 2014, 83, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Zhang, S.; Jiang, T.; He, Y.; Ma, Z.; Cai, H.; Xu, X.; Li, Y.; Cai, W.; Zhou, J.; et al. Early detection of lung cancer by using an autoantibody panel in Chinese population. Oncoimmunology 2017, 7, e1384108. [Google Scholar] [CrossRef] [PubMed]
- Magarian-Blander, J.; Domenech, N.; Finn, O.J. Specific and effective T-cell recognition of cells transfected with a truncated human mucin cDNA. Ann. N. Y. Acad. Sci. 1993, 690, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Apostolopoulos, V.; Yu, M.; Corper, A.L.; Teyton, L.; Pietersz, G.A.; McKenzie, I.F.; Wilson, I.A.; Plebanski, M. Crystal structure of a non-canonical low-affinity peptide complexed with MHC class I: A new approach for vaccine design. J. Mol. Biol. 2002, 318, 1293–1305. [Google Scholar] [CrossRef]
- Apostolopoulos, V.; Mckenzie, I.F.; Wilson, I.A. Getting into the groove: Unusual features of peptide binding to MHC class I molecules and implications in vaccine design. Front. Biosci. 2001, 6, D1311–D1320. [Google Scholar] [PubMed]
- Soares, M.M.; Mehta, V.; Finn, O.J. Three different vaccines based on the 140-amino acid MUC1 peptide with seven tandemly repeated tumor-specific epitopes elicit distinct immune effector mechanisms in wild-type versus MUC1-transgenic mice with different potential for tumor rejection. J. Immunol. 2001, 166, 6555–6563. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Ginardi, A.R.; Tinder, T.L.; Sterner, C.J.; Gendler, S.J. MUC1-specific cytotoxic T lymphocytes eradicate tumors when adoptively transferred in vivo. Clin. Cancer Res. 2001, 7 (Suppl. 3), 848s–855s. [Google Scholar] [PubMed]
- Pisarev, V.M.; Kinarsky, L.; Caffrey, T.; Hanisch, F.G.; Sanderson, S.; Hollingsworth, M.A.; Sherman, S. T cells recognize PD(N/T)R motif common in a variable number of tandem repeat and degenerate repeat sequences of MUC1. Int. Immunopharmacol. 2005, 5, 315–330. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Tinder, T.L.; Basu, G.D.; Pathangey, L.B.; Chen, L.; Gendler, S.J. Therapeutic efficacy of MUC1-specific cytotoxic T lymphocytes and CD137 co-stimulation in a spontaneous breast cancer model. Breast Dis. 2004, 20, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Koido, S.; Hara, E.; Homma, S.; Torii, A.; Toyama, Y.; Kawahara, H.; Watanabe, M.; Yanaga, K.; Fujise, K.; Tajiri, H.; et al. Dendritic cells fused with allogeneic colorectal cancer cell line present multiple colorectal cancer-specific antigens and induce antitumor immunity against autologous tumor cells. Clin. Cancer Res. 2005, 11, 7891–7900. [Google Scholar] [CrossRef] [PubMed]
- Stepensky, D.; Tzehoval, E.; Vadai, E.; Eisenbach, L. O-glycosylated versus non-glycosylated MUC1-derived peptides as potential targets for cytotoxic immunotherapy of carcinoma. Clin. Exp. Immunol. 2006, 143, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Ninkovica, T.; Kinarskyb, L.; Engelmannc, K.; Pisarevb, V.; Shermanb, S.; Finn, O.J. Identification of O-glycosylated decapeptides within the MUC1 repeat domain as potential MHC class I (A2) binding epitopes. Mol. Immunol. 2009, 47, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.O.; Vlad, A.M.; Islam, K.; Gariépy, J.; Finn, O.J. Tumor-associated MUC1 glycopeptide epitopes are not subject to self-tolerance and improve responses to MUC1 peptide epitopes in MUC1 transgenic mice. Biol. Chem. 2009, 390, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.H.; Shi, L.; Ma, J.W.; Sun, Z.Y.; Cai, H.; Chen, Y.X.; Zhao, Y.F.; Li, Y.M. A totally synthetic, self-assembling, adjuvant-free MUC1 glycopeptide vaccine for cancer therapy. J. Am. Chem. Soc. 2012, 134, 8730–8733. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aal, A.B.; Lakshminarayanan, V.; Thompson, P.; Supekar, N.; Bradley, J.M.; Wolfert, M.A.; Cohen, P.A.; Gendler, S.J.; Boons, G.J. Immune and anticancer responses elicited by fully synthetic aberrantly glycosylated MUC1 tripartite vaccines modified by a TLR2 or TLR9 agonist. Chembiochem 2014, 15, 1508–1513. [Google Scholar] [CrossRef] [PubMed]

| Antibody Binding Site | Examples | Epitopes | VH Usage | References |
|---|---|---|---|---|
| Peptide backbone | DF3 C595 GP1.4 | TRPAPGS RPAP DTRP | Diverse | [48] [48] [48] |
| Sugar | B72.3 4E10, 4G2 | clustered sialyl-Tn three consecutive Tn | IgHV1-78 (mouse) IGHV6-1(human) | [67,68] [69] |
| Both peptide backbone and sugar | BW835 MY.1E12 115D8 KL-6 5E5 | GVT(Galβ1,3GalNAc)SA GVT(Galβ1,3GalNAcS) Sialyl-Tn MUC1 tandem repeat PDT(Neu5Ac α2,3Galβ1,3GalNAc)RPAP GST(Tn)A | Diverse | [70] [70] [71] [72,73] [35] |
| Year Published | Clone/Product Name | Epitope | Active Ingredient | Efficacy | PI and Reference |
|---|---|---|---|---|---|
| 2017 | SAR566658(CA6) | Glycosylated PDTR | Humanized mouse mAb conjugated to maytansinoid | Phase 1 trial; safe; encouraging antitumor activity; selected for further clinical development | Sanofi, [112] |
| CAR TAG72 | STn sugar | CAR T cells | Safe; trafficking of CAR T cells to tumor. | Hege, [127] | |
| CAR SM3 | Glycosylated PDTR | CAR T cells | Remission of tumor (intratumoral injection) | Yang, [123] | |
| 2016 | PankoMab-GEX | Glycosylated PDTR | Humanized mouse mAb | Phase 1 trial; safe, promising efficacy in advanced diseases | Fiedler, [98] |
| 2011 | AS1402(AR20.5) | Glycosylated PDTR | Humanized mouse mAb | Phase 2 trial; no improvement in clinical outcome | Ibrahim, [97] |
| 2006 | R1549(HMFG1) | Glycosylated PDTR | Mouse mAb conjugated with 90Y-l | Phase 3 trial; no improvement in clinical outcome | Verheijen, [99] |
| 2000 | C595-67Cu | Glycosylated PDTR | Mouse mAb conjugated with 67Cu | Phase 1 trial; initial tumor uptake was high after intravesical injection | Hughes, [100] |
| CMB401 | Unknown MUC1 sequence | Humanized mouse mAb conjugated with calicheamicin | Phase 1 trial; safe; reduction in CA125 and tumor bulk observed in some patients | Gillespie, [101] | |
| 1998 | Bre-3 | Unknown MUC1 sequence | Humanized mouse mAb conjugated with 111In and 90Y-l | Phase 1 trial; excellent tumor localization, good tumor dosimetry, and low immunogenicity | Kramer, [102] |
| 1995 | 111In-NCRC48 | Unknown MUC1 sequence | Mouse mAb conjugated with 111In and 90Y-l | Phase 1 trial; uptake by tumor after intravesical injection to treat bladder cancer | Kunkler, [103] |
| 1994 | Bre-3 | Unknown MUC1 sequence | Mouse mAb conjugated with radioisotopes | Phase 1 trial; rapid clearance of antibody due to immunogenicity of mouse mAb | Kramer, [104] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, D.; Xu, L.; Huang, W.; Tonn, T. Epitopes of MUC1 Tandem Repeats in Cancer as Revealed by Antibody Crystallography: Toward Glycopeptide Signature-Guided Therapy. Molecules 2018, 23, 1326. https://doi.org/10.3390/molecules23061326
Zhou D, Xu L, Huang W, Tonn T. Epitopes of MUC1 Tandem Repeats in Cancer as Revealed by Antibody Crystallography: Toward Glycopeptide Signature-Guided Therapy. Molecules. 2018; 23(6):1326. https://doi.org/10.3390/molecules23061326
Chicago/Turabian StyleZhou, Dapeng, Lan Xu, Wei Huang, and Torsten Tonn. 2018. "Epitopes of MUC1 Tandem Repeats in Cancer as Revealed by Antibody Crystallography: Toward Glycopeptide Signature-Guided Therapy" Molecules 23, no. 6: 1326. https://doi.org/10.3390/molecules23061326
APA StyleZhou, D., Xu, L., Huang, W., & Tonn, T. (2018). Epitopes of MUC1 Tandem Repeats in Cancer as Revealed by Antibody Crystallography: Toward Glycopeptide Signature-Guided Therapy. Molecules, 23(6), 1326. https://doi.org/10.3390/molecules23061326