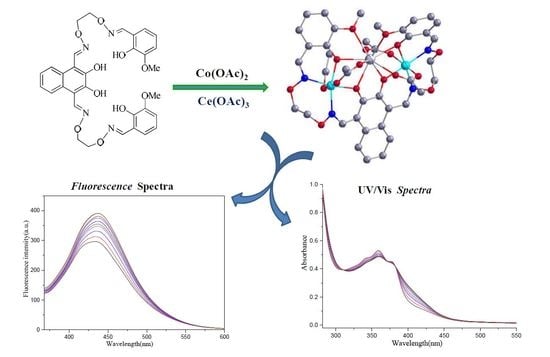
Synthesis and Fluorescence Properties of a New Heterotrinuclear Co(II)-Ce(III)Complex Constructed from a bis(salamo)-Type Tetraoxime Ligand
Abstract
:1. Introduction
2. Results and Discussion
2.1. IR Spectra
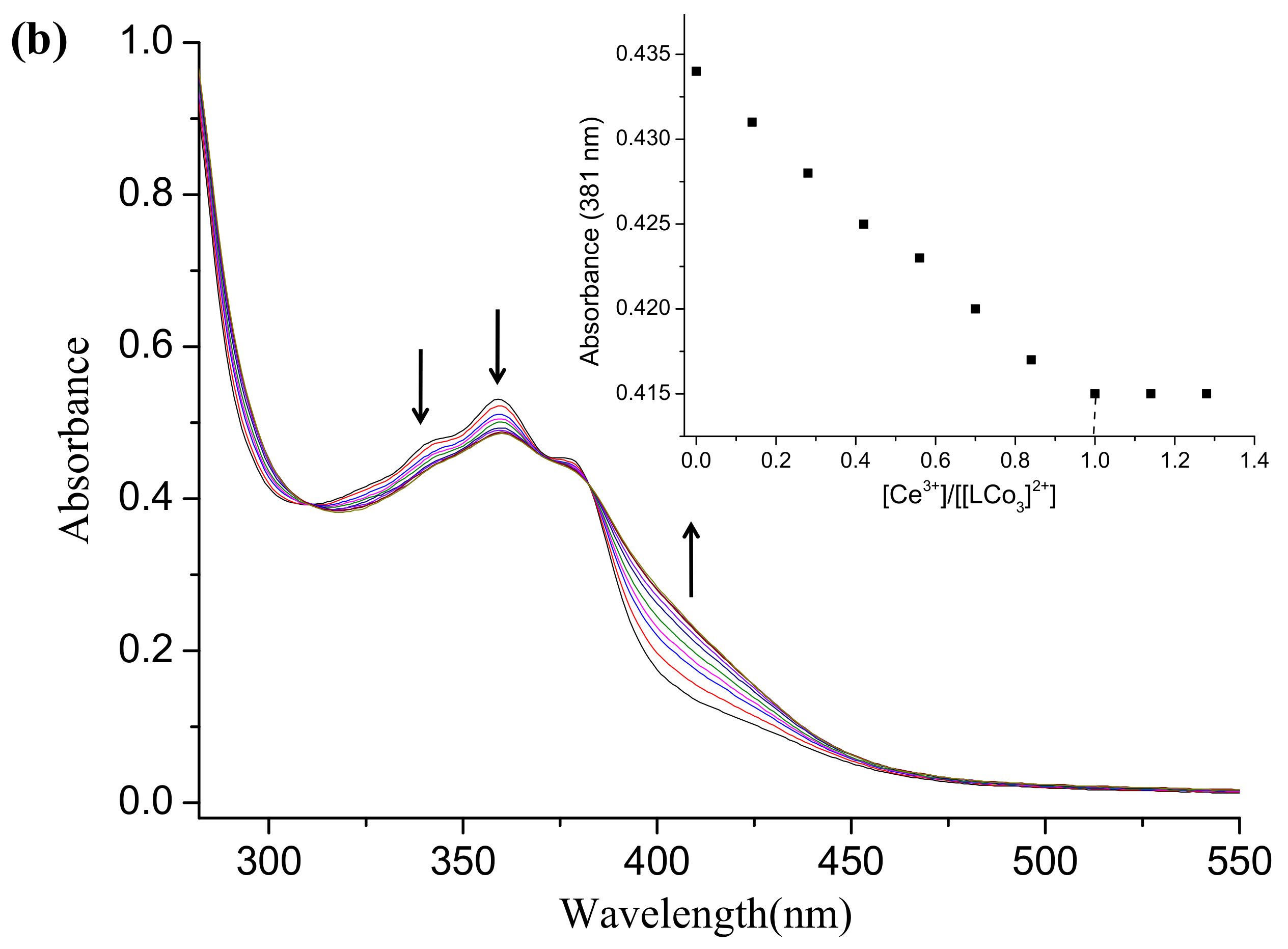
2.2. UV-Vis Absorption Spectra
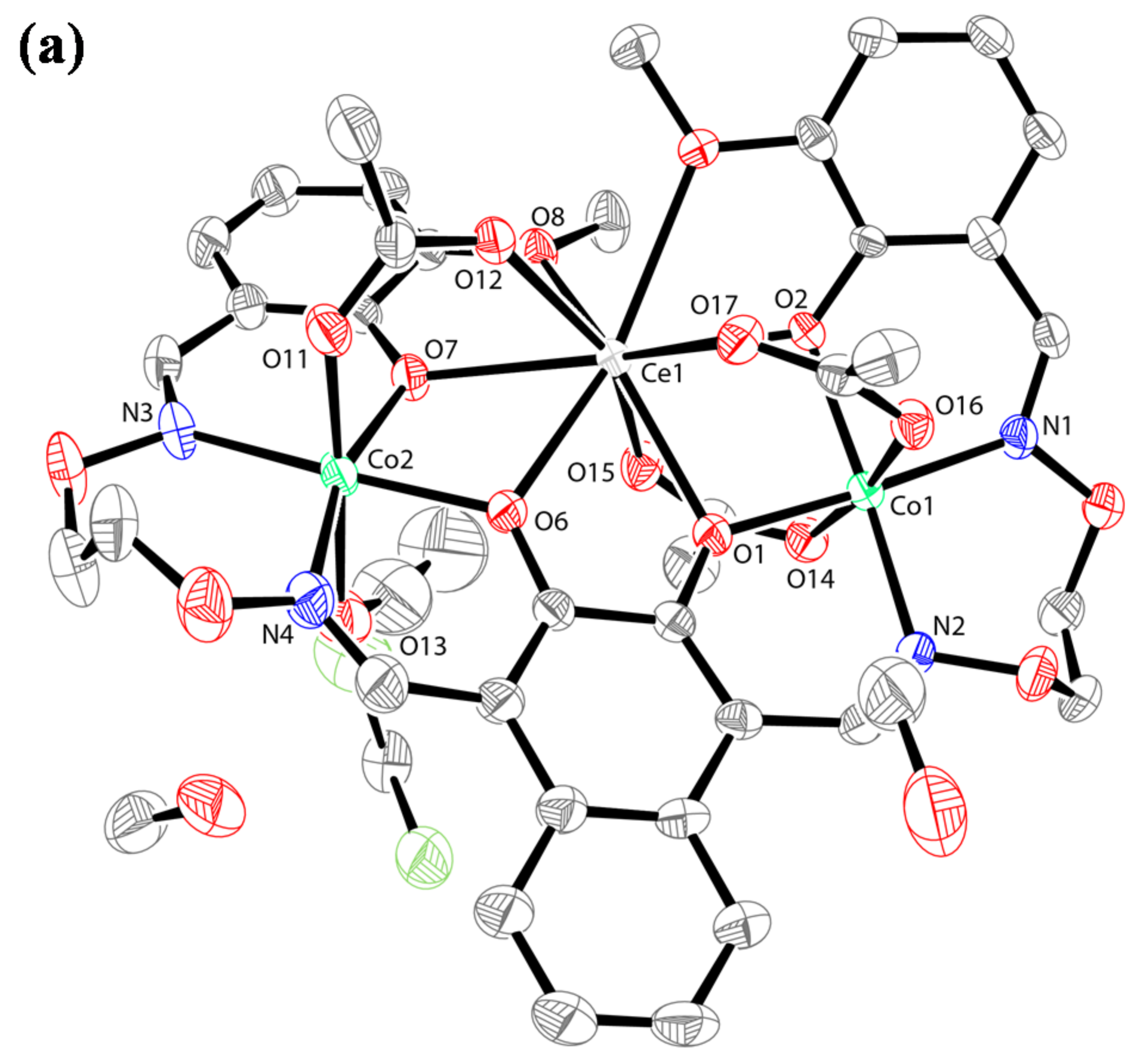
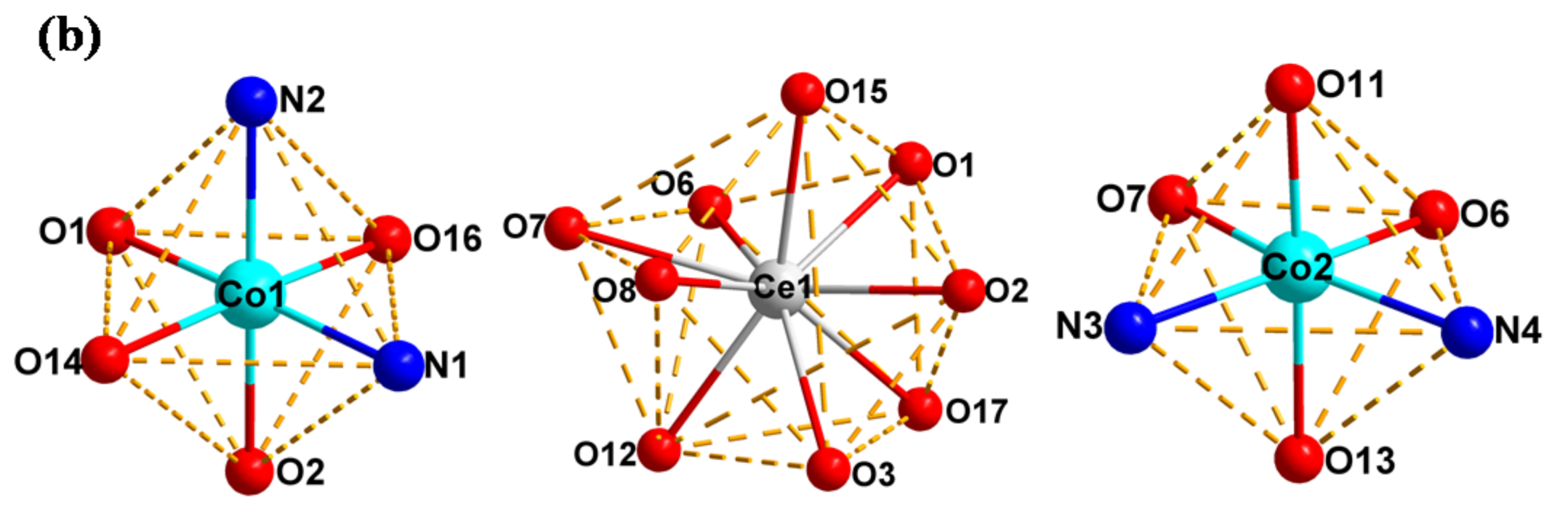
2.3. Crystal Structure Description
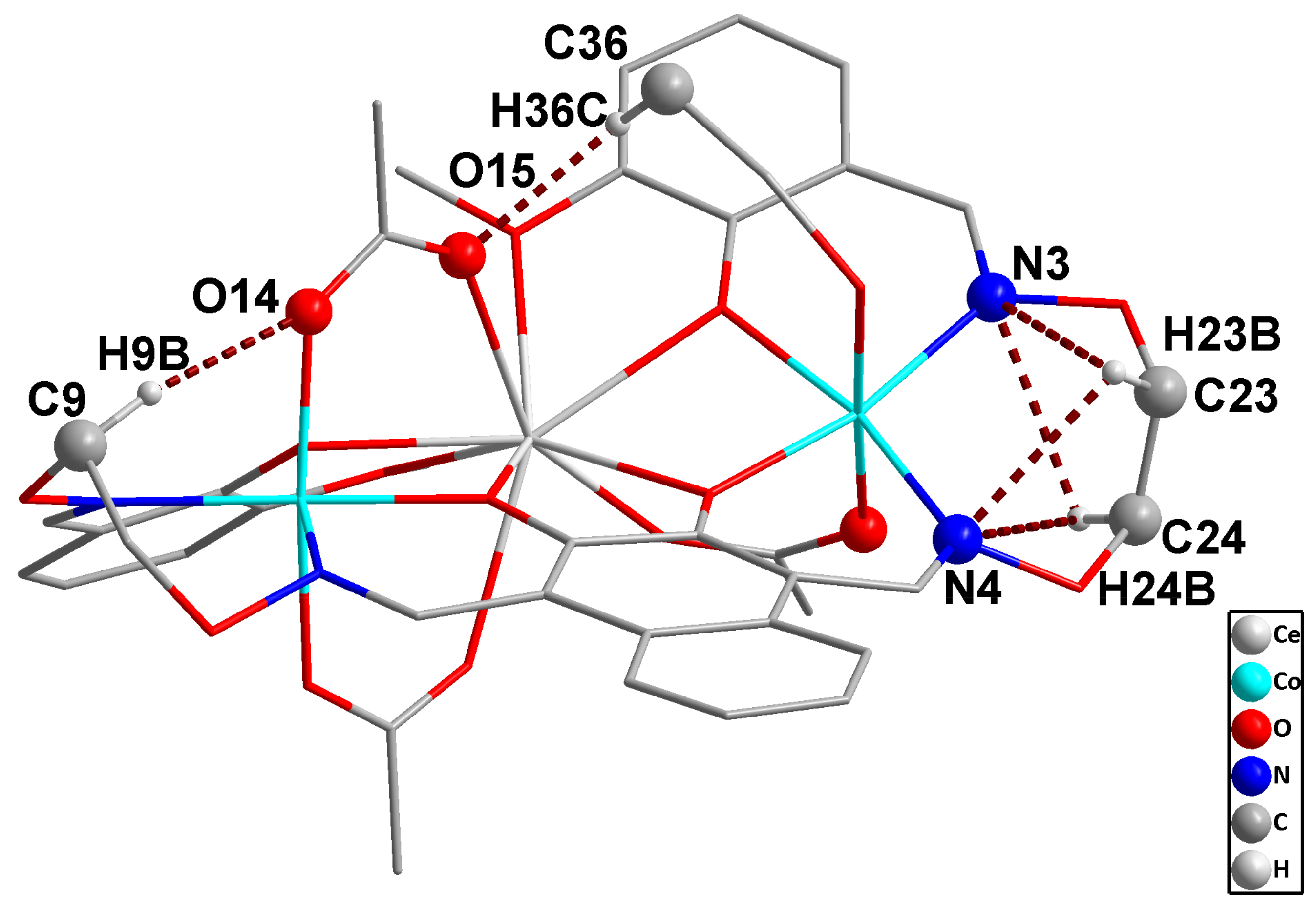
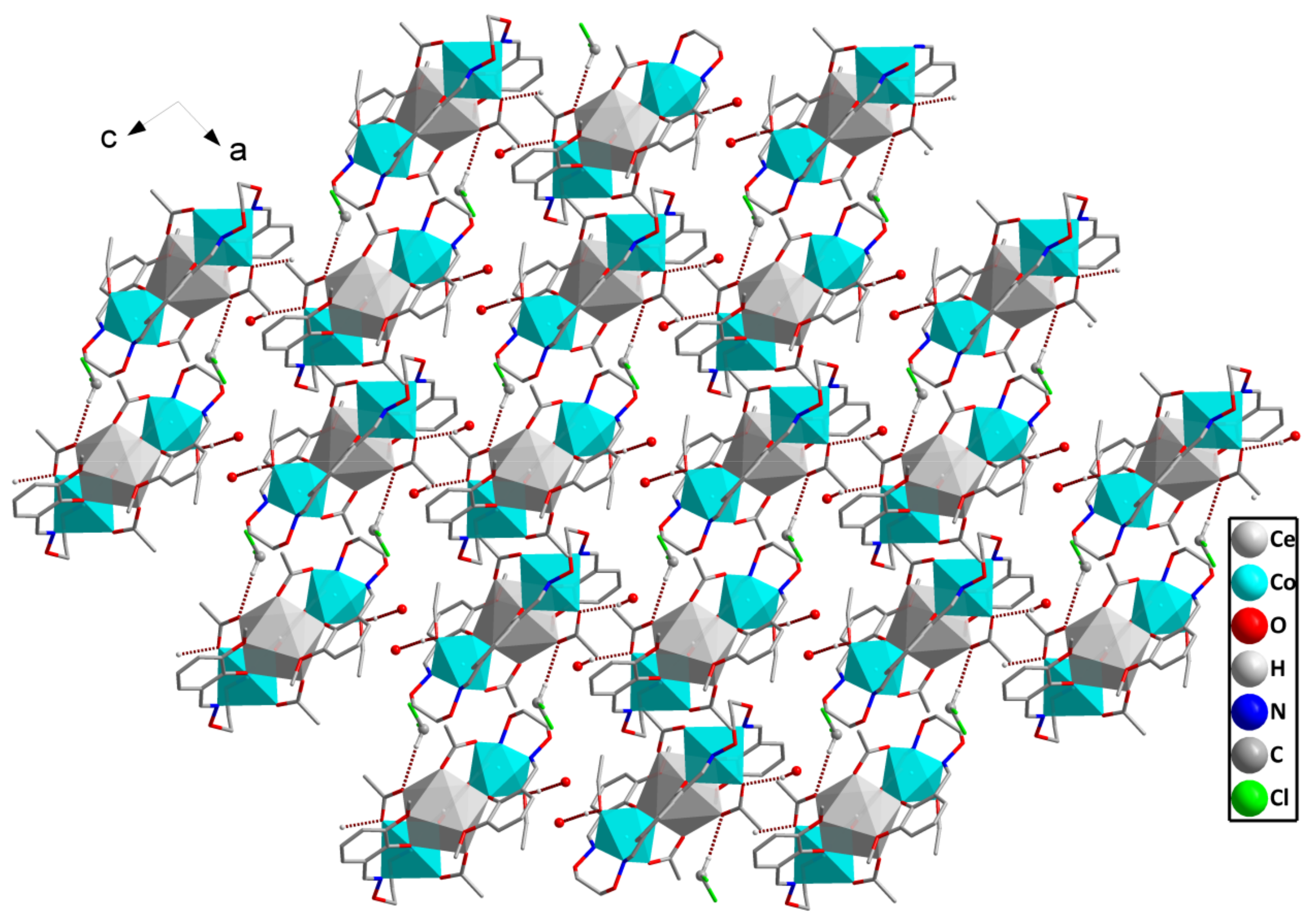
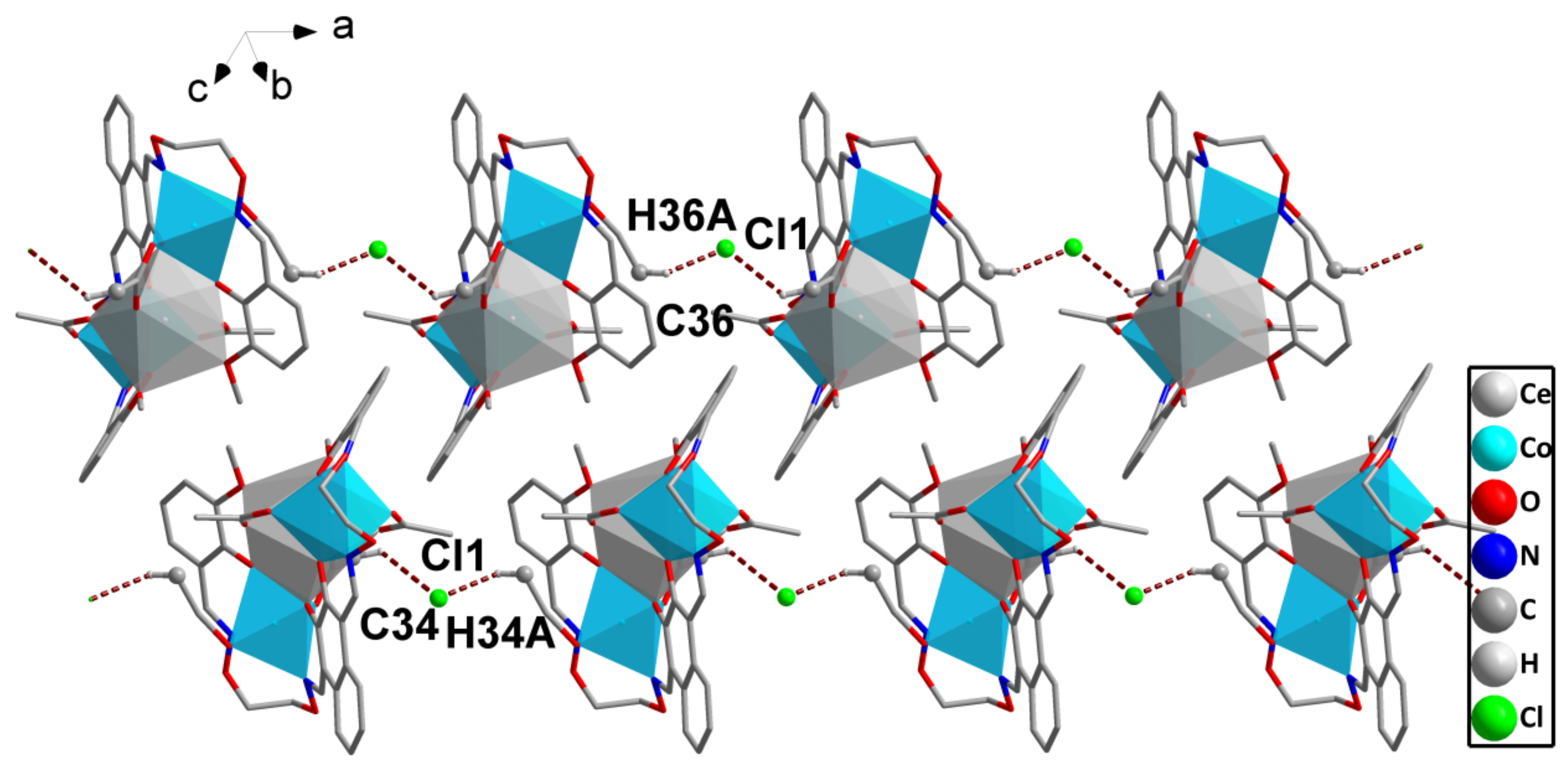
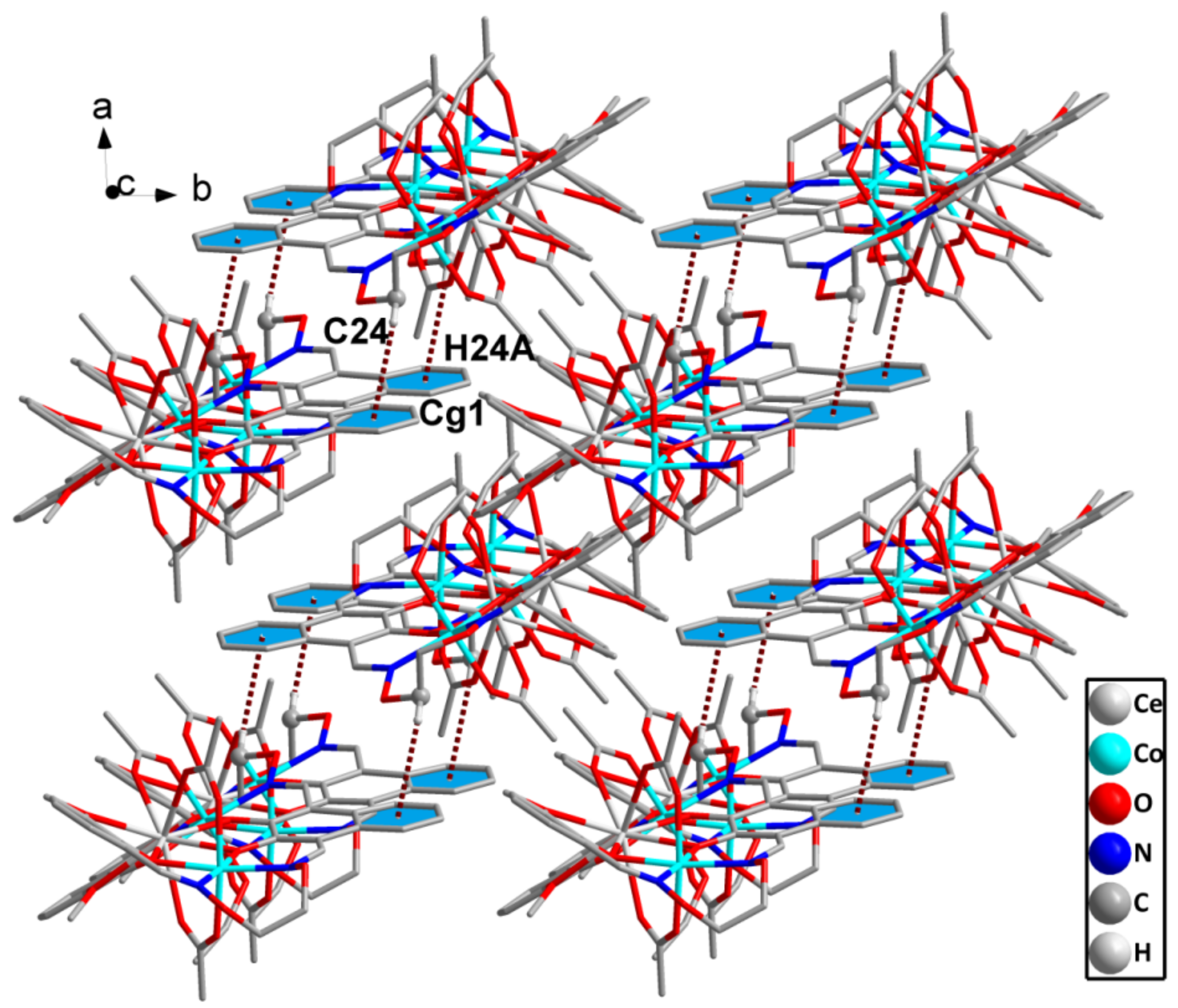
2.4. Supramolecular Interactions
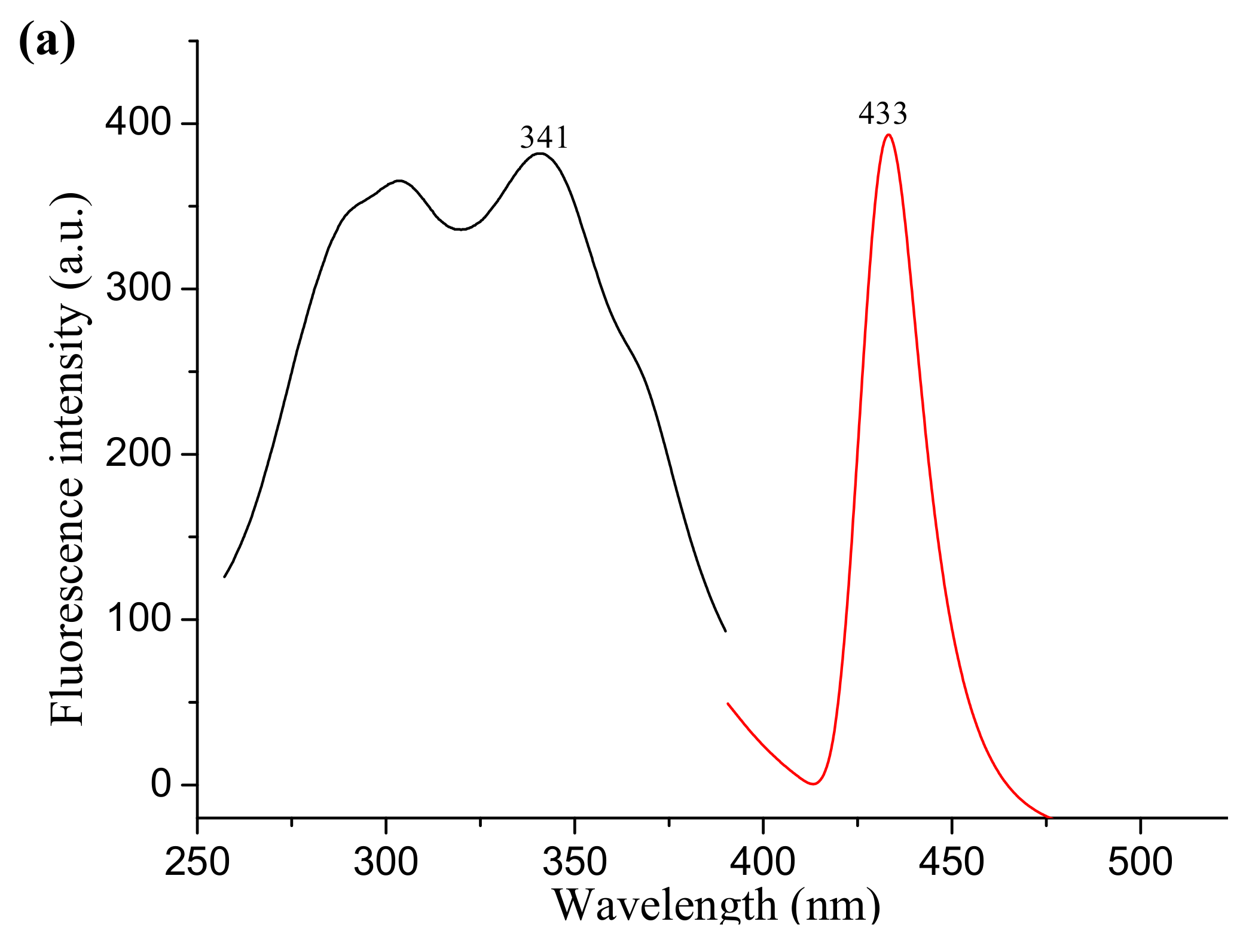
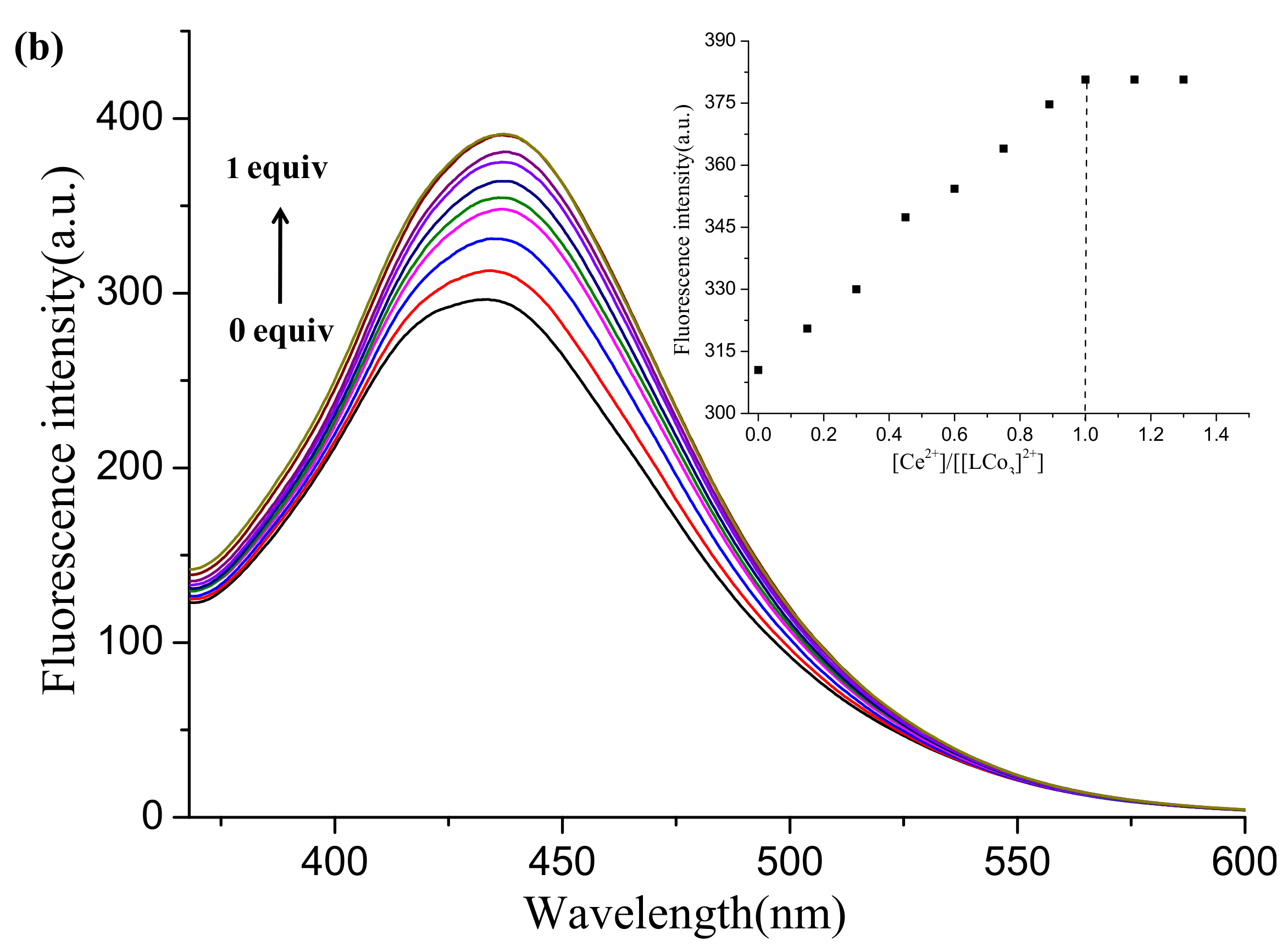
2.5. Fluorescence Tests
3. Experimental Section
3.1. Materials and Methods
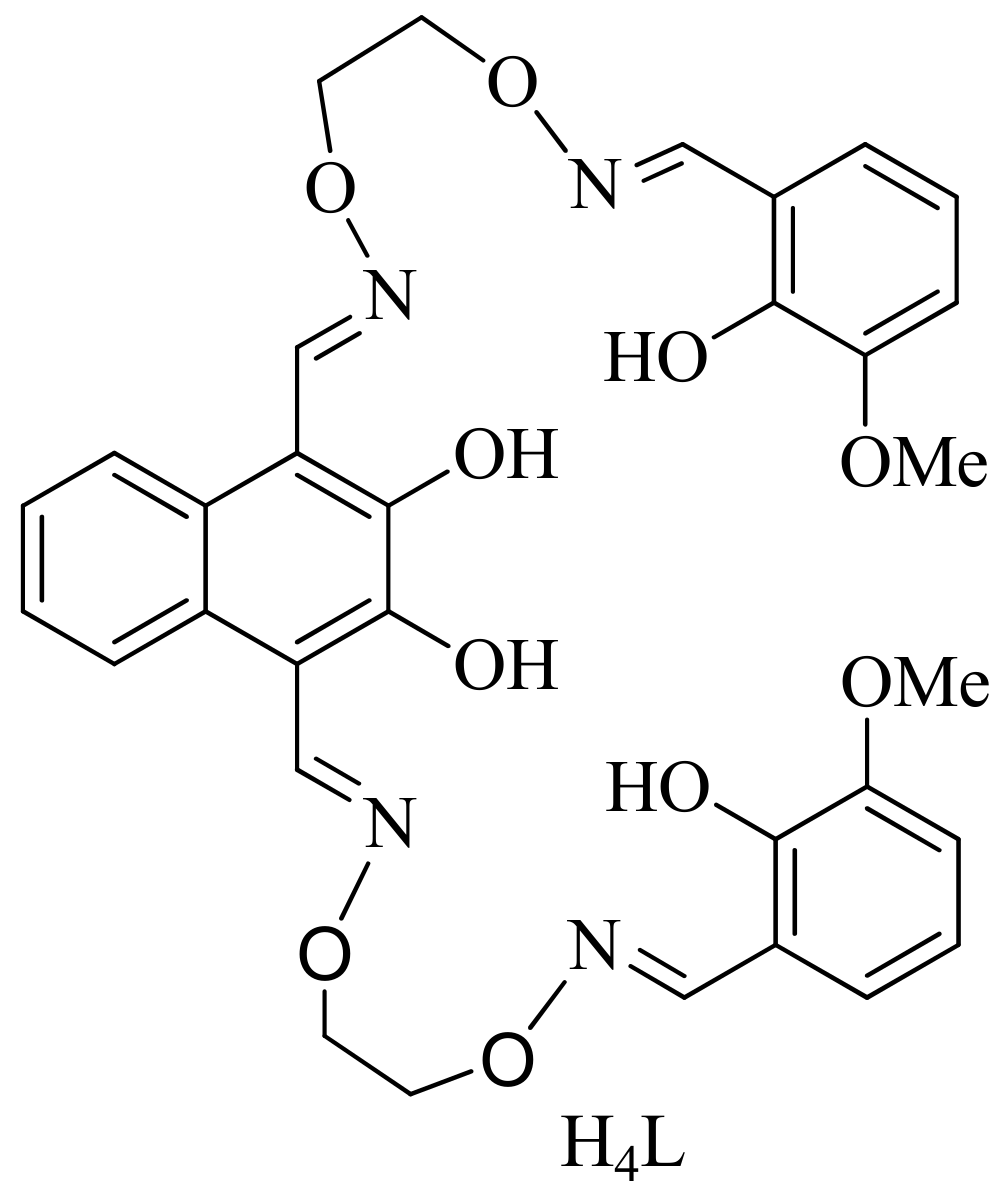
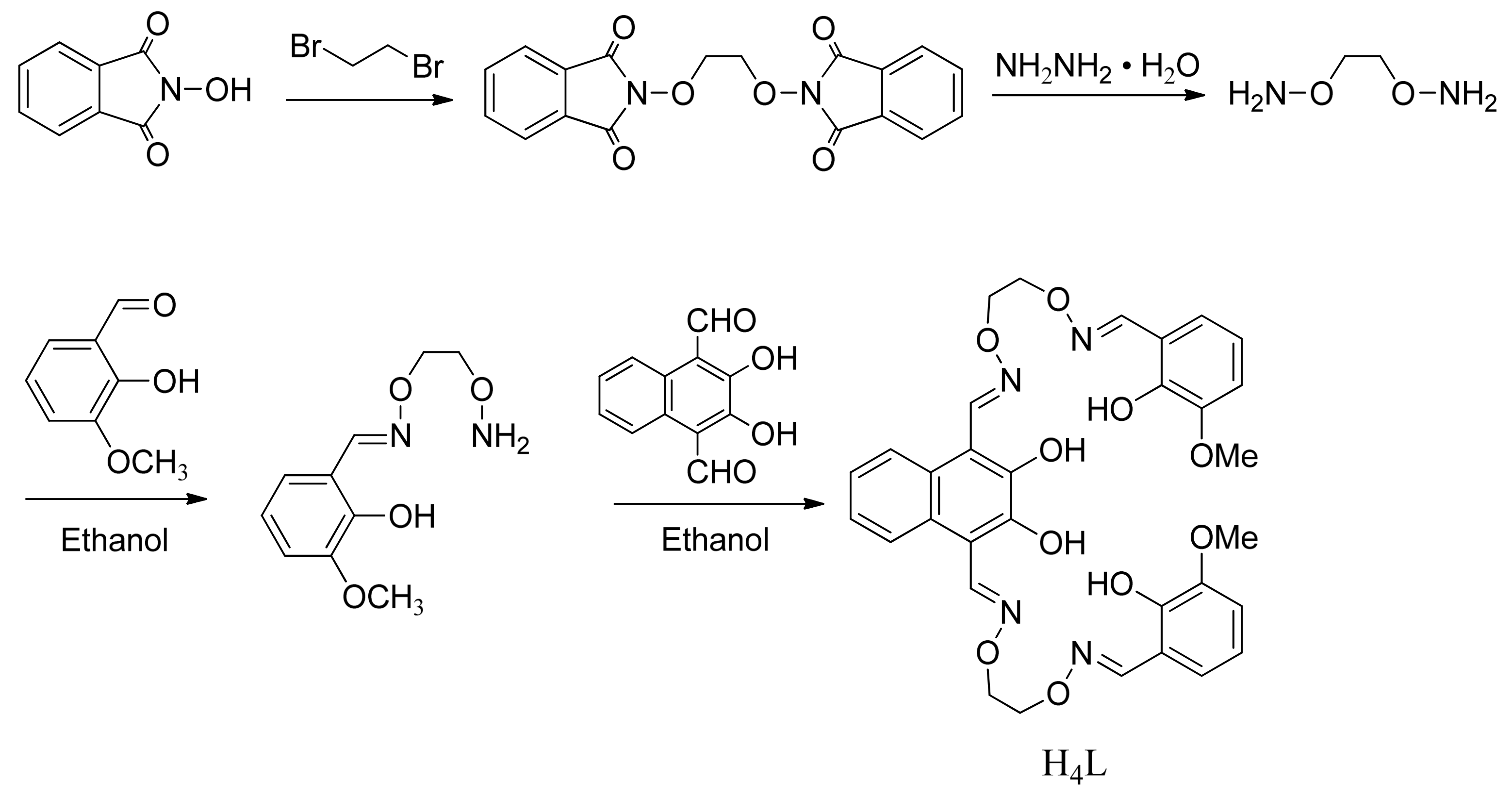
3.2. Synthesis of the Bi(salamo)-Type Ligand H4L
3.3. Synthesis of the Heterotrinuclear Co(II)-Ce(III) Complex
3.4. Crystal Structure Determination and Refinement
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chai, L.Q.; Li, Y.X.; Chen, L.C.; Zhang, J.Y.; Huang, J.J. Synthesis, X-ray structure, spectroscopic, electrochemical properties and DFT calculation of a bridged dinuclear copper(II) complex. Inorg. Chem. Acta 2016, 444, 193–201. [Google Scholar] [CrossRef]
- Ma, X.; Jiao, J.M.; Yang, J.; Huang, X.B.; Cheng, Y.X.; Zhu, C.J. Large stokes shift chiral polymers containing (R,R)-salen-based binuclear boron complex: Synthesis, characterization, and fluorescence properties. Polyhedron 2012, 53, 3894–3899. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, L.; Sun, Y.X.; Dong, W.K.; Tang, X.L.; Gao, X.H. A supramolecular copper(II) complex bearing salen-type bisoxime ligand: Synthesis, structural characterization and thermal property. Synth. React. Inorg. Met-Org. Nano-Met. Chem. 2012, 42, 1303–1308. [Google Scholar] [CrossRef]
- Subramaniam, P.; Anbarasan, S.; Devi, S.S.; Ramdass, A. Modulation of catalytic activity by ligand oxides in the sulfoxidation of phenylmercaptoacetic acids by oxo(salen)chromium(V) complexes. Polyhedron 2016, 119, 14–22. [Google Scholar] [CrossRef]
- Sun, Y.X.; Zhang, S.T.; Ren, Z.L.; Dong, X.Y.; Wang, L. Synthesis, characterization, and crystal structure of a new supramolecular CdII complex with halogen-substituted salen-type bisoxime. Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 2013, 43, 995–1000. [Google Scholar] [CrossRef]
- Shu, Y.B.; Liu, W.S. Luminescent chiral Eu(III) complexes with enantiopure bis(1H-pyridin-2-one)salen ligands. Polyhedron 2015, 102, 293–296. [Google Scholar] [CrossRef]
- Sun, Y.X.; Gao, X.H. Synthesis, characterization, and crystal structure of a new CuII complex with salen-type ligand. Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 2011, 41, 973–978. [Google Scholar] [CrossRef]
- Sun, Y.X.; Xu, L.; Zhao, T.H.; Liu, S.H.; Liu, G.H.; Dong, X.T. Synthesis and crystal structure of a 3D supramolecular copper(II) complex with 1-(3-{[(E)-3-bromo-5-chloro-2-hydroxybenzylidene]amino}phenyl) ethanone oxime. Synth. React. Inorg. Met.-Org. Nano-Met. Chem 2013, 43, 509–513. [Google Scholar] [CrossRef]
- Song, X.Q.; Liu, P.P.; Wang, C.Y.; Liu, Y.A.; Liu, W.S.; Zhang, M. Three sandwich-type zinc(II)–lanthanide(III) clusters: Structures, luminescence and magnetic properties. RSC Adv. 2017, 7, 22692–22698. [Google Scholar] [CrossRef]
- Liu, P.P.; Sheng, L.; Song, X.Q.; Xu, W.Y.; Liu, Y.A. Synthesis, structure and magnetic properties of a new one dimensional manganese coordination polymer constructed by a new asymmetrical ligand. Inorg. Chim. Acta 2015, 434, 252–257. [Google Scholar] [CrossRef]
- Song, X.Q.; Liu, P.P.; Xiao, Z.R.; Li, X.; Liu, Y.A. Four polynuclear complexes based on a versatile salicylamide salen-like ligand: Synthesis, structural variations and magnetic properties. Inorg. Chim. Acta 2015, 438, 232–244. [Google Scholar] [CrossRef]
- Liu, Y.A.; Wang, C.Y.; Zhang, M.; Song, X.Q. Structures and magnetic properties of cyclic heterometallic tetranuclear clusters. Polyhedron 2017, 127, 278–286. [Google Scholar] [CrossRef]
- Zhou, J.J.; Song, X.Q.; Liu, Y.A.; Wang, X.L. Substituent-tuned structure and luminescence sensitizing towards Al3+ based on phenoxy bridged dinuclear EuIII complexes. RSC Adv. 2017, 7, 25549–25559. [Google Scholar] [CrossRef]
- Dong, W.K.; Ma, J.C.; Zhu, L.C.; Zhang, Y. Nine self-assembled nickel(II)–lanthanide(III) heterometallic complexes constructed from a Salamo-type bisoxime and bearing a N- or O-donor auxiliary ligand: Syntheses, structures and magnetic properties. New J. Chem. 2016, 40, 6998–7010. [Google Scholar] [CrossRef]
- Chin, T.K.; Endud, S.; Jamil, S.; Budagumpi, S.; Lintang, H.O. Oxidative dimerization of o-aminophenol by heterogeneous mesoporous material modified with biomimetic salen-type copper(II) complex. Catal. Lett. 2013, 143, 282–288. [Google Scholar] [CrossRef]
- Li, X.Y.; Chen, L.; Gao, L.; Zhang, Y.; Akogun, S.F.; Dong, W.K. Syntheses, crystal structures and catalytic activities of two solvent-induced homotrinuclear Co(II) complexes with a naphthalenediol-based bis(salamo)-type tetraoxime ligand. RSC Adv. 2017, 7, 35905–35916. [Google Scholar] [CrossRef]
- Ömer, S.; Ümmühan, Ö.Ö.; Nurgul, S.; Burcu, A.; Musa, S.; Tuncay, T.; Zeynel, S. A highly selective and sensitive chemosensor derived coumarin-thiazole for colorimetric and fluorimetric detection of CN− ion in DMSO and aqueous solution: Synthesis, sensing ability, Pd(II)/Pt(II) complexes and theoretical studies. Tetrahedron 2016, 72, 5843–5852. [Google Scholar]
- Wang, L.; Li, X.Y.; Zhao, Q.; Li, L.H.; Dong, W.K. Fluorescence properties of heterotrinuclear Zn(II)–M(II) (M = Ca, Sr and Ba) bis(salamo)-type complexes. RSC Adv. 2017, 7, 48730–48737. [Google Scholar] [CrossRef]
- Wu, H.L.; Bai, Y.C.; Zhang, Y.H.; Li, Z.; Wu, M.C.; Chen, C.Y.; Zhang, J.W. Synthesis, crystal structure, antioxidation and DNA-binding properties of a dinuclear copper(II) complex with bis(N-salicylidene)-3-oxapentane-1,5-diamine. J. Coord. Chem. 2014, 67, 3054–3066. [Google Scholar] [CrossRef]
- Chen, C.Y.; Zhang, J.W.; Zhang, Y.H.; Yang, Z.H.; Wu, H.L.; Pan, G.L.; Bai, Y.C. Gadolinium(III) and dysprosium(III) complexes with a Schiff base bis(N-salicylidene)-3-oxapentane-1,5-diamine: Synthesis, characterization, antioxidation, and DNA-binding studies. J. Coord. Chem. 2015, 68, 1054–1071. [Google Scholar] [CrossRef]
- Wu, H.L.; Bai, Y.H.; Zhang, Y.H.; Pan, G.L.; Kong, J.; Shi, F.R.; Wang, X.L. Two lanthanide(III) complexes based on the Schiff base N,N′-Bis(salicylidene)-1,5-diamino-3-oxapentane: Synthesis, characterization, DNA-binding properties, and antioxidation. Z. Anorg. Allg. Chem. 2014, 640, 2062–2071. [Google Scholar] [CrossRef]
- Wu, H.L.; Pan, G.L.; Wang, H.; Wang, X.L.; Bai, Y.C.; Zhang, Y.H. Study on synthesis, crystal structure, antioxidant and DNA-binding of mono-, di- and poly-nuclear lanthanides complexes with bis(N-salicylidene)-3-oxapentane-1,5-diamine. J. Photochem. Photobiol. B Biol. 2014, 135, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.L.; Pan, G.L.; Bai, Y.C.; Wang, H.; Kong, J.; Shi, F.; Zhang, Y.H.; Wang, X.L. Preparation, structure, DNA-binding properties, and antioxidant activities of a homodinuclear erbium(III) complex with a pentadentate Schiff base ligand. J. Chem. Res. 2014, 38, 211–217. [Google Scholar] [CrossRef]
- Wu, H.L.; Wang, H.; Wang, X.L.; Pan, G.L.; Shi, F.R.; Zhang, Y.H.; Bai, Y.C.; Kong, J. V-shaped ligand bis(2-benzimidazolylmethyl)amine containing three copper(II) ternary complexes: Synthesis, structure, DNA-binding properties and antioxidant activity. New J. Chem. 2014, 38, 1052–1061. [Google Scholar] [CrossRef]
- Mao, S.S.; Shen, K.S.; Shi, X.K.; Wu, H.L.; Han, X.T.; Li, C.; Huang, G.Z. Synthesis, crystal structure and biological activity of two binuclear Ag(I) complexes with bis-benzimidazole thioether ligands. Inorg. Chim. Acta 2018, 471, 82–90. [Google Scholar] [CrossRef]
- Wu, H.L.; Pan, G.L.; Bai, Y.C.; Wang, H.; Kong, J.; Shi, F.R.; Zhang, Y.H.; Wang, X.L. Synthesis, structure, antioxidation, and DNA-binding studies of a binuclear ytterbium(III) complex with bis(N-salicylidene)-3-oxapentane-1,5-diamine. Res. Chem. Intermed. 2015, 41, 3375–3388. [Google Scholar] [CrossRef]
- Wang, F.; Xu, Y.L.; Aderinto, S.O.; Peng, H.P.; Zhang, H.; Wu, H.L. A new highly effective fluorescent probe for Al3+ ions and its application in practical samples. J. Photochem. Photobiol. A 2017, 332, 273–282. [Google Scholar] [CrossRef]
- Wu, H.L.; Bai, Y.; Yuan, J.K.; Wang, H.; Pan, G.L.; Fan, X.Y.; Kong, J. A zinc(II) complex with tris(2-(-methyl)benzimidazlylmethyl)amine and salicylate: Synthesis, crystal structure, and DNA-binding. J. Coord. Chem. 2012, 65, 2839–2851. [Google Scholar] [CrossRef]
- Chai, L.Q.; Wang, G.; Sun, Y.X.; Dong, W.K.; Zhao, L.; Gao, X.H. Synthesis, crystal structure, and fluorescence of an unexpected dialkoxo-bridged dinuclear copper(II) complex with bis(salen)-type tetraoxime. J. Coord. Chem. 2012, 65, 1621–1631. [Google Scholar] [CrossRef]
- Chai, L.Q.; Huang, J.J.; Zhang, H.S.; Zhang, Y.L.; Zhang, J.Y.; Li, Y.X. An unexpected cobalt(III) complex containing a Schiff base ligand: Synthesis, crystal structure, spectroscopic behavior, electrochemical property and SOD-like activity. Spectrochim. Acta Part A 2014, 131, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.Q.; Liu, G.; Zhang, J.Y.; Huang, J.J.; Tong, J.F. Synthesis, crystal structure, fluorescence, electrochemical property, and SOD-like activity of an unexpected nickel(II) complex with a quinazoline-type ligand. J. Coord. Chem. 2013, 66, 3926–3938. [Google Scholar] [CrossRef]
- Chai, L.Q.; Zhang, H.S.; Huang, J.J.; Zhang, Y.L. An unexpected Schiff base-type Ni(II) complex: Synthesis, crystal structures, fluorescence, electrochemical property and SOD-like activities. Spectrochim. Acta Part A 2015, 137, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.Q.; Tang, L.J.; Chen, L.C.; Huang, J.J. Structural, spectral, electrochemical and DFT studies of two mononuclear manganese(II) and zinc(II) complexes. Polyhedron 2017, 122, 228–240. [Google Scholar] [CrossRef]
- Chai, L.Q.; Zhang, K.Y.; Tang, L.J.; Zhang, J.Y.; Zhang, H.S. Two mono- and dinuclear Ni(II) complexes constructed from quinazoline-type ligands: Synthesis, X-ray structures, spectroscopic, electrochemical, thermal, and antimicrobial studies. Polyhedron 2017, 130, 100–107. [Google Scholar] [CrossRef]
- Chen, L.; Dong, W.K.; Zhang, H.; Zhang, Y.; Sun, Y.X. Structural variation and luminescence properties of tri- and dinuclear CuII and ZnII complexes constructed from a naphthalenediol-based bis(Salamo)-type ligand. Cryst. Growth Des. 2017, 17, 3636–3648. [Google Scholar] [CrossRef]
- Dong, X.Y.; Li, X.Y.; Liu, L.Z.; Zhang, H.; Ding, Y.J.; Dong, W.K. Tri- and hexanuclear heterometallic Ni(II)–M(II) (M = Ca, Sr and Ba) bis(salamo)-type complexes: Synthesis, structure and fluorescence properties. RSC Adv. 2017, 7, 48394–48403. [Google Scholar] [CrossRef]
- Song, X.Q.; Peng, Y.J.; Chen, G.Q.; Wang, X.R.; Liu, P.P.; Xu, W.Y. Substituted group-directed assembly of Zn(II) coordination complexes based on two new structural related pyrazolone based Salen ligands: Syntheses, structures and fluorescence properties. Inorg. Chim. Acta 2015, 427, 13–21. [Google Scholar] [CrossRef]
- Wang, P.; Zhao, L. An infinite 2D supramolecular cobalt(II) complex based on an asymmetric salamo-type ligand: Synthesis, crystal structure, and spectral properties. Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 2016, 46, 1095–1101. [Google Scholar] [CrossRef]
- Dong, X.Y.; Akogun, S.F.; Zhou, W.M.; Dong, W.K. Tetranuclear Zn(II) complex based on an asymmetrical salamo-type chelating ligand: Synthesis, structural characterization, and fluorescence property. J. Chin. Chem. Soc. 2017, 64, 412–419. [Google Scholar] [CrossRef]
- Tao, C.H.; Ma, J.C.; Zhu, L.C.; Zhang, Y.; Dong, W.K. Heterobimetallic 3d–4f Zn(II)–Ln(III) (Ln = Sm, Eu, Tb and Dy) complexes with a N2O4 bisoxime chelate ligand and a simple auxiliary ligand Py: Syntheses, structures and luminescence properties. Polyhedron 2017, 128, 38–45. [Google Scholar] [CrossRef]
- Dong, Y.J.; Dong, X.Y.; Dong, W.K.; Zhang, Y.; Zhang, L.S. Three asymmetric salamo-type copper(II) and cobalt(II) complexes: Syntheses, structures, fluorescent properties. Polyhedron 2017, 123, 305–315. [Google Scholar] [CrossRef]
- Dong, W.K.; Ma, J.C.; Dong, Y.J.; Zhao, L.; Zhu, L.C.; Sun, Y.X.; Zhang, Y. Two hetero-trinuclear Zn(II)-M(II) (M = Sr, Ba) complexes based on metallohost of mononuclear Zn(II) complex: Syntheses, structures and fluorescence properties. J. Coord. Chem. 2016, 69, 3231–3241. [Google Scholar] [CrossRef]
- Wu, H.L.; Wang, C.P.; Wang, F.; Peng, H.P.; Zhang, H.; Bai, Y.C. A new manganese(III) complex from bis(5-methylsalicylaldehyde)-3-oxapentane-1,5-diamine: Synthesis, characterization, antioxidant activity and luminescence. J. Chin. Chem. Soc. 2015, 62, 1028–1034. [Google Scholar] [CrossRef]
- Wang, L.; Ma, J.C.; Dong, W.K.; Zhu, L.C.; Zhang, Y. A novel Self–assembled nickel(II)–cerium(III) heterotetranuclear dimer constructed from N2O2-type bisoxime and terephthalic acid: Synthesis, structure and photophysical properties. Z. Anorg. Allg. Chem. 2016, 642, 834–839. [Google Scholar] [CrossRef]
- Dong, Y.J.; Li, X.L.; Zhang, Y.; Dong, W.K. A highly selective visual and fluorescent sensor for Pb2+ and Zn2+ and crystal structure of Cu2+ complex based-on a novel single-armed salamotype bisoxime. Supramol. Chem. 2017, 29, 518–527. [Google Scholar] [CrossRef]
- Liu, P.P.; Wang, C.Y.; Zhang, M.; Song, X.Q. Pentanuclear sandwich-type ZnII-LnIII clusters based on a new salen-like salicylamide ligand: Structure, near-infrared emission and magnetic properties. Polyhedron 2017, 129, 133–140. [Google Scholar] [CrossRef]
- Li, G.; Hao, J.; Liu, L.Z.; Zhou, W.M.; Dong, W.K. Syntheses, crystal structures and thermal behaviors of two supramolecular salamo-type cobalt(II) and zinc(II) complexes. Crystals 2017, 7, 217. [Google Scholar]
- Wang, B.J.; Dong, W.K.; Zhang, Y.; Akogun, S.F. A novel relay-sensor for highly sensitive and selective detection of Zn2+/Pic− and fluorescence on/off switch response of H+/OH−. Sens. Actuators B Chem. 2017, 247, 254–264. [Google Scholar] [CrossRef]
- Dong, W.K.; Li, G.; Wang, Z.K.; Dong, X.Y. A novel trinuclear cobalt(II) complex derived from an asymmetric salamo-type N2O3 bisoxime chelate ligand: Synthesis, structure and optical properties. Spectrochim. Acta Part A 2014, 133, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.Y.; Gao, L.; Wang, F.; Zhang, Y.; Dong, W.K. Tri- and mono-nuclear zinc(II) complexes based on half- and mono-salamo chelating ligands. Crystals 2017, 7, 267. [Google Scholar] [CrossRef]
- Yu, B.; Li, C.Y.; Sun, Y.X.; Jia, H.R.; Guo, J.Q.; Li, J. A new azine derivative colorimetric and fluorescent dual-channel probe for cyanide detection. Spectrochim. Acta Part A 2017, 184, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.K.; Lan, P.F.; Zhou, W.M.; Zhang, Y. Salamo-type trinuclear and tetranuclear cobalt (II) complexes based on a new asymmetry salamo-type ligand: Syntheses, crystal structures, and fluorescence. J. Coord. Chem. 2016, 69, 1–22. [Google Scholar] [CrossRef]
- Xu, L.; Zhu, L.C.; Ma, J.C.; Zhang, Y.; Zhang, J.; Dong, W.K. Syntheses, structures and spectral properties of mononuclear CuII and dimeric ZnII complexes based on an asymmetric Salamo-type N2O2 ligand. Z. Anorg. Allg. Chem. 2015, 641, 2520–2524. [Google Scholar] [CrossRef]
- Wang, F.; Gao, L.; Zhao, Q.; Zhang, Y.; Dong, W.K.; Ding, Y.J. A highly selective fluorescent chemosensor for CN− based on a novel bis(salamo)-type tetraoxime ligand. Spectrochim. Acta Part A 2018, 190, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Wang, F.; Zhao, Q.; Zhang, Y.; Dong, W.K. Mononuclear Zn(II) and trinuclear Ni(II) complexes derived from a coumarin-containing N2O2 ligand: Syntheses, crystal structures and fluorescence properties. Polyhedron 2018, 139, 7–16. [Google Scholar] [CrossRef]
- Dong, W.K.; Du, W.; Zhang, X.Y.; Li, G.; Dong, X.Y. Synthesis, crystal structure and spectral properties of a supramolecular trinuclear nickel(II) complex with 5-methoxy-4′-bromo-2,2′-[ethylenedioxybis(nitrilomethylidyne)]diphenol. Spectrochim. Acta Part A 2014, 132, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.K.; Zheng, S.S.; Zhang, J.T.; Zhang, Y.; Sun, Y.X. Luminescent properties of heterotrinuclear 3d–4f complexes constructed from a naphthalenediol-based acyclic bis(salamo)-type ligand. Spectrochim. Acta Part A 2017, 184, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.J.; Ma, J.C.; Zhu, L.C.; Dong, W.K.; Zhang, Y. Four 3d–4f heteromultinuclear zinc(II)–lanthanide(III) complexes constructed from a distinct hexadentate N2O2-type ligand: Syntheses, structures and luminescence properties. J. Coord. Chem. 2017, 70, 103–115. [Google Scholar] [CrossRef]
- Ma, J.C.; Dong, X.Y.; Dong, W.K.; Zhang, Y.; Zhu, L.C.; Zhang, J.T. An unexpected dinuclear Cu(II) complex with a bis(Salamo) chelating ligand: Synthesis, crystal structure, and photophysical properties. J. Coord. Chem. 2016, 69, 149–159. [Google Scholar] [CrossRef]
- Dong, W.K.; Ma, J.C.; Dong, Y.J.; Zhu, L.C.; Zhang, Y. Di- and tetranuclear heterometallic 3d–4f cobalt(II)–lanthanide(III) complexes derived from a hexadentate bisoxime: Syntheses, structures and magnetic properties. Polyhedron 2016, 115, 228–235. [Google Scholar] [CrossRef]
- Dong, W.K.; Ma, J.C.; Zhu, L.C.; Zhang, Y. Self-assembled zinc(II)-lanthanide(III) heteromultinuclear complexes constructed from 3-MeOsalamo ligand: Syntheses, structures and luminescent properties. Cryst. Growth Des. 2016, 16, 6903–6914. [Google Scholar] [CrossRef]
- Zhang, H.; Dong, W.K.; Zhang, Y.; Akogun, S.F. Naphthalenediol-based bis(Salamo)-type homo- and heterotrinuclear cobalt(II) complexes: Syntheses, structures and magnetic properties. Polyhedron 2017, 133, 279–293. [Google Scholar] [CrossRef]
- Li, L.H.; Dong, W.K.; Zhang, Y.; Akogun, S.F.; Xu, L. Syntheses, structures and catecholase activities of homo- and hetero-trinuclear cobalt(II) complexes constructed from an acyclic naphthalenediolbased bis(Salamo)-type ligand. Appl. Organomet. Chem. 2017, 31, e3818. [Google Scholar] [CrossRef]
- Wang, L.; Hao, J.; Zhai, L.X.; Zhang, Y.; Dong, W.K. Synthesis, crystal structure, luminescence, electrochemical and antimicrobial properties of bis(salamo)-based Co(II) complex. Crystals 2017, 7, 277. [Google Scholar] [CrossRef]
- Li, X.Y.; Kang, Q.P.; Liu, L.Z.; Ma, J.C.; Dong, W.K. Trinuclear Co(II) and mononuclear Ni(II) Salamo-type bisoxime coordination compounds. Crystals 2018, 8, 43. [Google Scholar] [CrossRef]
- Peng, Y.D.; Li, X.Y.; Kang, Q.P.; An, G.X.; Zhang, Y.; Dong, W.K. Synthesis and fluorescence properties of asymmetrical Salamo-type tetranuclear zinc(II) complex. Crystals 2018, 8, 107. [Google Scholar] [CrossRef]
- Gao, L.; Liu, C.; Wang, F.; Dong, W.K. Tetra-, penta- and hexa-coordinated transition metal complexes constructed from coumarin-containing N2O2 ligand. Crystals 2018, 8, 77. [Google Scholar] [CrossRef]
- Sun, Y.X.; Li, C.Y.; Yang, C.J.; Zhao, Y.Y.; Guo, J.Q.; Yu, B. Two Cu(II) complexes with Schiff base ligands: Syntheses, crystal structures, spectroscopic properties and substituent effect. Chin. J. Inorg. Chem. 2016, 32, 327–335. [Google Scholar]
- Yu, B.; Sun, Y.X.; Yang, C.J.; Guo, J.Q.; Li, J. Synthesis and crystal structures of an unexpected tetranuclear zinc(II) complex and a benzoquinone compound derived from ZnII- and CdII-promoted reactivity of Schiff base ligands. Z. Anorg. Allg. Chem. 2017, 643, 689–698. [Google Scholar] [CrossRef]
- Sun, Y.X.; Lu, R.E.; Li, X.R.; Zhao, Y.Y.; Li, C.Y. A Schiff base ligand containing oxime group and its Cu(II) complex: Syntheses and supramolecular structures. Chin. J. Inorg. Chem. 2015, 31, 1055–1062. [Google Scholar]
- Guo, J.Q.; Sun, Y.X.; Yu, B.; Li, J.; Jia, H.R. Syntheses, crystal structures and spectroscopic properties of copper(II) and nickel(II) complexes with oxime-type Schiff base ligands. Chin. J. Inorg. Chem. 2017, 33, 1481–1488. [Google Scholar]
- Xu, Y.L.; Mao, S.S.; Shen, K.S.; Shi, X.K.; Wu, H.L.; Tang, X. Different structures of two Cu(I) complexes constructed by bridging 2,2-(1,4-butanediyl)bis-1,3-benzoxazole ligand: Syntheses, structures and properties. Inorg. Chim. Acta 2018, 471, 17–22. [Google Scholar] [CrossRef]
- Sun, Y.X.; Zhao, Y.Y.; Li, C.Y.; Yu, B.; Guo, J.Q.; Li, J. Supramolecular cobalt(II) and copper(II) complexes with Schiff base ligand: Syntheses, characterization and crystal structures. Chin. J. Inorg. Chem. 2016, 32, 913–920. [Google Scholar]
- Jia, H.R.; Li, J.; Sun, Y.X.; Guo, J.Q.; Yu, B.; Wen, N.; Xu, L. Two supramolecular cobalt(II) complexes: Syntheses, crystal structures, spectroscopic behaviors, and counter anion effects. Crystals 2017, 7, 247. [Google Scholar]
- Zheng, S.S.; Dong, W.K.; Zhang, Y.; Chen, L.; Dong, Y.G. Four salamo-type 3d–4f hetero-bimetallic [ZnIILnIII] complexes: Syntheses, crystal structures, and luminescent and magnetic properties. New J. Chem. 2017, 41, 4966–4973. [Google Scholar] [CrossRef]
- Hao, J.; Li, L.H.; Zhang, J.T.; Akogun, S.F.; Wang, L.; Dong, W.K. Four homo- and hetero-bismetallic 3d/3d-2s complexes constructed from a naphthalenediol-based acyclic bis(salamo)-type tetraoxime ligand. Polyhedron 2017, 134, 1–10. [Google Scholar] [CrossRef]
- Akine, S.; Sairenji, S.; Taniguchi, T.; Nabeshima, T. Stepwise helicity inversions by multisequential metal exchange. J. Am. Chem. Soc. 2013, 135, 12948–12951. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Liu, L.Z.; Dong, W.K.; Zhang, J.; Zhang, Y. Three multinuclear Co(II), Zn(II) and Cd(II) complexes based on a single-armed salamo-type bisoxime: Syntheses, structural characterizations and fluorescent properties. J. Coord. Chem. 2017, 70, 1–30. [Google Scholar] [CrossRef]
- Hao, J.; Li, X.Y.; Zhang, Y.; Dong, W.K. A reversible bis(Salamo)-based fluorescence sensor for selective detection of Cd2+ in water-containing systems and food samples. Materials 2018, 11, 523. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
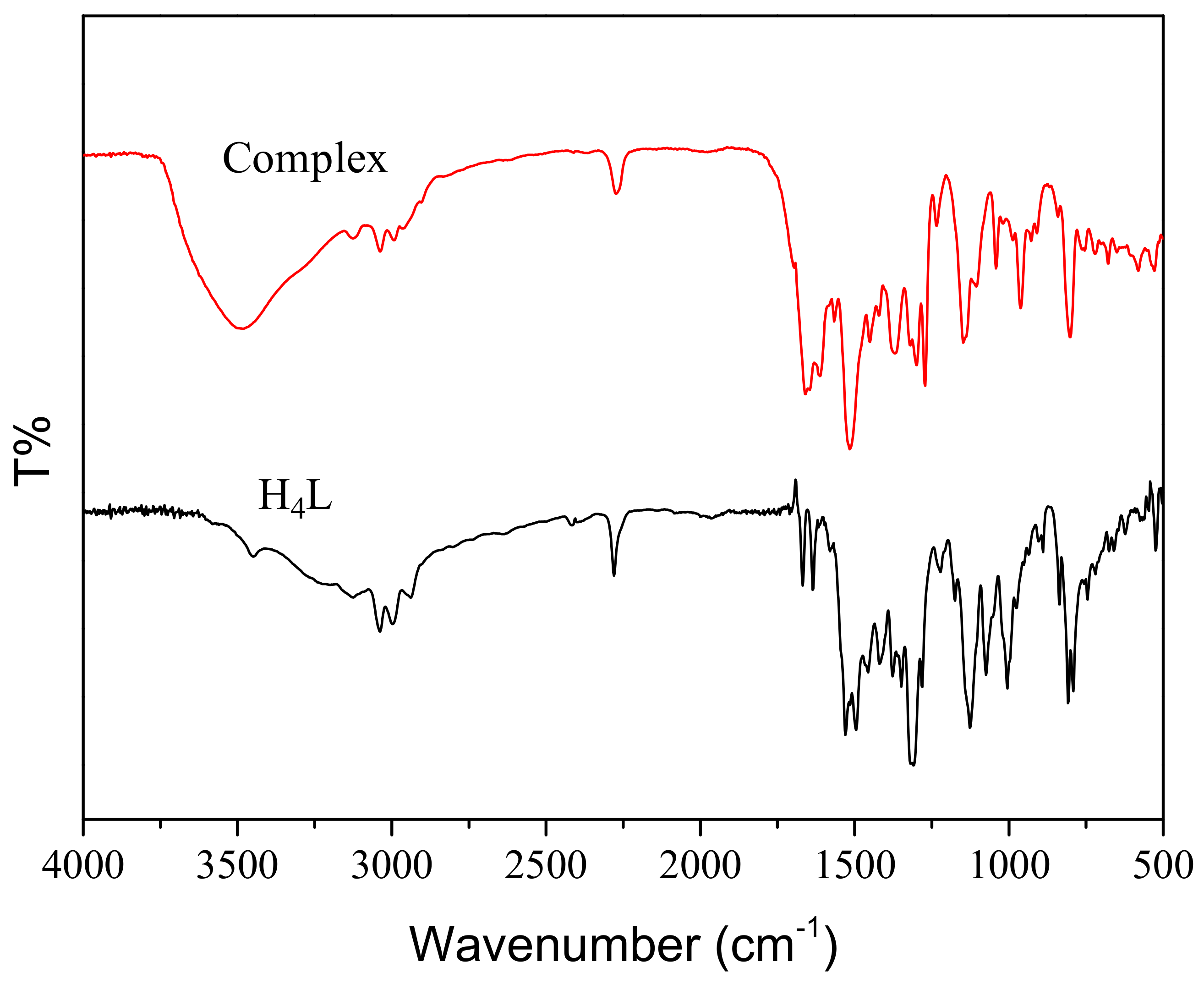


| Compound | ν(O–H) | ν(C=N) | ν(Ar–O) |
|---|---|---|---|
| H4L | 3169 | 1612 | 1255 |
| Complex | – | 1623 | 1239 |
| Formula | C42H50ClCo2CeN4O18.50 |
| Formula weight, g·mol−1 | 1200.29 |
| Temperature, K | 293.66(10) |
| Wavelength, Å | 0.71073 |
| Crystal system | Triclinic |
| Space group | P-1 |
| a, Å | 12.0521(5) |
| b, Å | 13.7804(5) |
| c, Å | 15.2909(4) |
| α, ° | 95.678(3) |
| β, ° | 99.471(3) |
| γ, ° | 90.210(3) |
| Volume, Å3 | 2492.13(15) |
| Z | 2 |
| Calculated density, mg·m−3 | 1.600 |
| Absorption coefficient, mm−1 | 1.685 |
| F (000) | 1214 |
| θ range for data collection, ° | 3.390–25.677° |
| h/k/l(min, max) | −12, 14/−16, 16/−18, 18 |
| Reflections collected | 17180 |
| Independent reflections | 9641 |
| Rint | 0.034 |
| Completeness to θ = 26.32 | 99.78% |
| Data/restraints/parameters | 9641/60/649 |
| Final R indices [I > 2σ (I)] a[I > 2σ(I)] | R1 = 0.0519, wR2 = 0.1377 |
| R indices (all data) b | R1 = 0.0693, wR2 = 0.1537 |
| Goodness-of-fit for F2 c | 1.020 |
| Largest differences peak and hole (e Å−3) | 1.130 and −1.300 |
| Bonds Lengths (Å) | Bonds Lengths (Å) | ||||
| Co1–O1 | 2.018(3) | Co1–O2 | 2.081(4) | Co1–O14 | 2.088(4) |
| Co1–O16 | 2.123(4) | Co1–N1 | 2.095(5) | Co1–N2 | 2.116(4) |
| Co2–O6 | 2.021(4) | Co2–O7 | 2.052(4) | Co2–O11 | 2.058(5) |
| Co2–O13 | 2.168(6) | Co2–N3 | 2.123(5) | Co2–N4 | 2.139(7) |
| Ce1–O1 | 2.421(4) | Ce1–O2 | 2.461(3) | Ce1–O3 | 2.688(4) |
| Ce1–O6 | 2.467(4) | Ce1–O7 | 2.523(3) | Ce1–O8 | 2.655(4) |
| Ce1–O12 | 2.437(5) | Ce1–O15 | 2.528(5) | Ce1–O17 | 2.526(4) |
| Angle (°) | Angle (°) | ||||
| O1–Co1–O2 | 90.51(15) | O1–Co1–O14 | 87.26(16) | O1–Co1–O16 | 88.60(16) |
| O1–Co1–N1 | 176.65(19) | O1–Co1–N2 | 83.73(17) | O2–Co1–O14 | 88.61(15) |
| O2–Co1–O14 | 88.61(15) | O2–Co1–O16 | 85.93(15) | O2–Co1–N1 | 86.51(17) |
| O2–Co1–N2 | 171.58(16) | O14–Co1–O16 | 173.12(17) | O14–Co1–N1 | 94.18(18) |
| O14–Co1–N2 | 97.21(18) | O16–Co1–N1 | 89.67(18) | O16–Co1–N2 | 87.79(18) |
| N1–Co1–N2 | 99.1(2) | O1–Ce1–O2 | 73.22(12) | O1–Ce1–O3 | 128.52(12) |
| O1–Ce1–O6 | 62.41(11) | O1–Ce1–O7 | 115.41(12) | O1–Ce1–O8 | 136.97(13) |
| O1–Ce1–O12 | 129.53(13) | O1–Ce1–O15 | 68.24(13) | O1–Ce1–O17 | 69.66(13) |
| O2–Ce1–O3 | 60.44(13) | O2–Ce1–O6 | 135.35(13) | O2–Ce1–O7 | 147.78(12) |
| O2–Ce1–O8 | 91.55(11) | O2–Ce1–O12 | 130.03(14) | O2–Ce1–O15 | 71.51(13) |
| O2–Ce1–O17 | 72.66(13) | O3–Ce1–O6 | 151.91(15) | O3–Ce1–O7 | 115.69(13) |
| O3–Ce1–O8 | 68.54(13) | O3–Ce1–O12 | 74.57(14) | O3–Ce1–O15 | 112.71(14) |
| O3–Ce1–O17 | 75.97(14) | O6–Ce1–O7 | 64.67(13) | O6–Ce1–O8 | 124.04(12) |
| O6–Ce1–O12 | 79.85(14) | O6–Ce1–O15 | 95.34(15) | O6–Ce1–O17 | 87.10(15) |
| O7–Ce1–O12 | 70.43(14) | O7–Ce1–O8 | 60.38(11) | O7–Ce1–O15 | 82.89(13) |
| O7–Ce1–O17 | 139.42(13) | O8–Ce1–O12 | 91.27(14) | O8–Ce1–O15 | 68.78(14) |
| O8–Ce1–O17 | 144.42(13) | O12–Ce1–O15 | 152.40(15) | O12–Ce1–O17 | 76.59(15) |
| O15–Ce1–O17 | 130.63(14) | ||||
| D–H···A | d(D–H) | d(H···A) | d(D···A) | ∠DHA | Symmetry Code A |
| C9–H9B···O14 | 0.97 | 2.32 | 3.249(8) | 161 | |
| C36–H36C···O15 | 0.96 | 2.20 | 2.879(16) | 126 | |
| C23–H23B···N4 | 0.97 | 2.46 | 2.865(11) | 105 | |
| C24–H24B···N3 | 0.97 | 2.56 | 2.957(11) | 104 | |
| O13–H13···O18 | 0.87 | 1.88 | 2.733(18) | 170 | x, y, 1 + z |
| O19–H19A···O16 | 0.82 | 2.05 | 2.834(9) | 160 | −x, −y, 1 − z |
| C34–H34A···Cl1 | 0.96 | 2.76 | 3.446(16) | 129 | −1 + x, y, z |
| C36–H36A···Cl1 | 0.96 | 2.34 | 2.955(17) | 121 | x, y, 1 + z |
| C41–H41B···O17 | 0.97 | 2.38 | 3.345(16) | 173 | 1 + x, y, z |
| C24–H24A···Cg1 | 2.97 | 3.599(10) | 124 | −x, −y, −z |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pu, L.-M.; Zhao, Q.; Liu, L.-Z.; Zhang, H.; Long, H.-T.; Dong, W.-K. Synthesis and Fluorescence Properties of a New Heterotrinuclear Co(II)-Ce(III)Complex Constructed from a bis(salamo)-Type Tetraoxime Ligand. Molecules 2018, 23, 804. https://doi.org/10.3390/molecules23040804
Pu L-M, Zhao Q, Liu L-Z, Zhang H, Long H-T, Dong W-K. Synthesis and Fluorescence Properties of a New Heterotrinuclear Co(II)-Ce(III)Complex Constructed from a bis(salamo)-Type Tetraoxime Ligand. Molecules. 2018; 23(4):804. https://doi.org/10.3390/molecules23040804
Chicago/Turabian StylePu, Lu-Mei, Qing Zhao, Ling-Zhi Liu, Han Zhang, Hai-Tao Long, and Wen-Kui Dong. 2018. "Synthesis and Fluorescence Properties of a New Heterotrinuclear Co(II)-Ce(III)Complex Constructed from a bis(salamo)-Type Tetraoxime Ligand" Molecules 23, no. 4: 804. https://doi.org/10.3390/molecules23040804
APA StylePu, L.-M., Zhao, Q., Liu, L.-Z., Zhang, H., Long, H.-T., & Dong, W.-K. (2018). Synthesis and Fluorescence Properties of a New Heterotrinuclear Co(II)-Ce(III)Complex Constructed from a bis(salamo)-Type Tetraoxime Ligand. Molecules, 23(4), 804. https://doi.org/10.3390/molecules23040804