Synthesis and Evaluation of the Anticonvulsant Activities of 4-(2-(Alkylthio)benzo[d]oxazol-5-yl)-2,4-dihydro-3H-1,2,4-triazol-3-ones
Abstract
1. Introduction
2. Results and Discussion
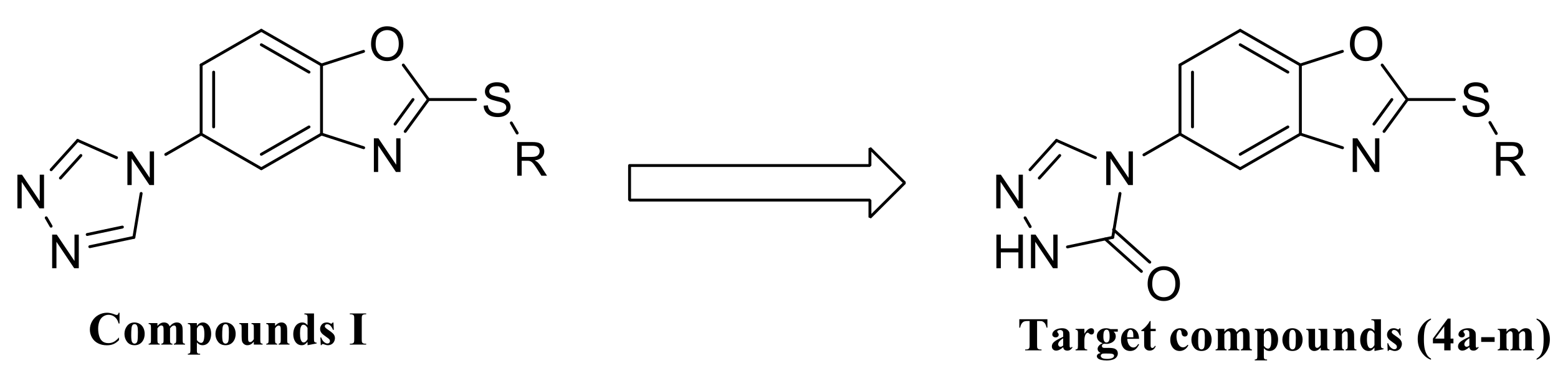
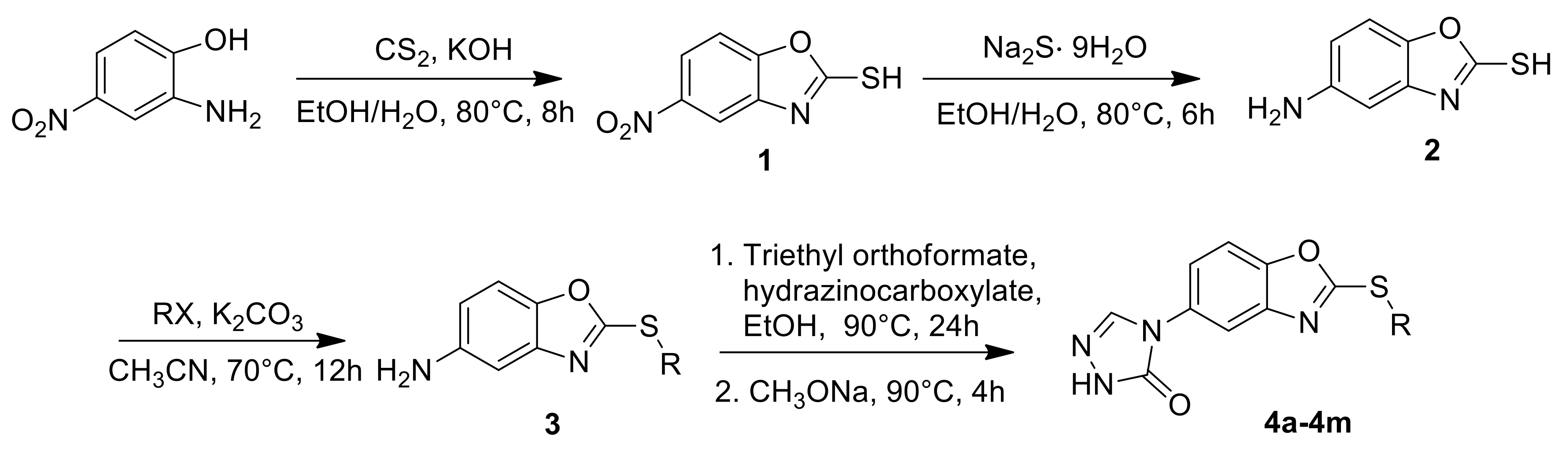
2.1. Chemistry
2.2. Anticonvulsant Activity in MES and scPTZ Models and Neurotoxicity in the Rota-Rod Test
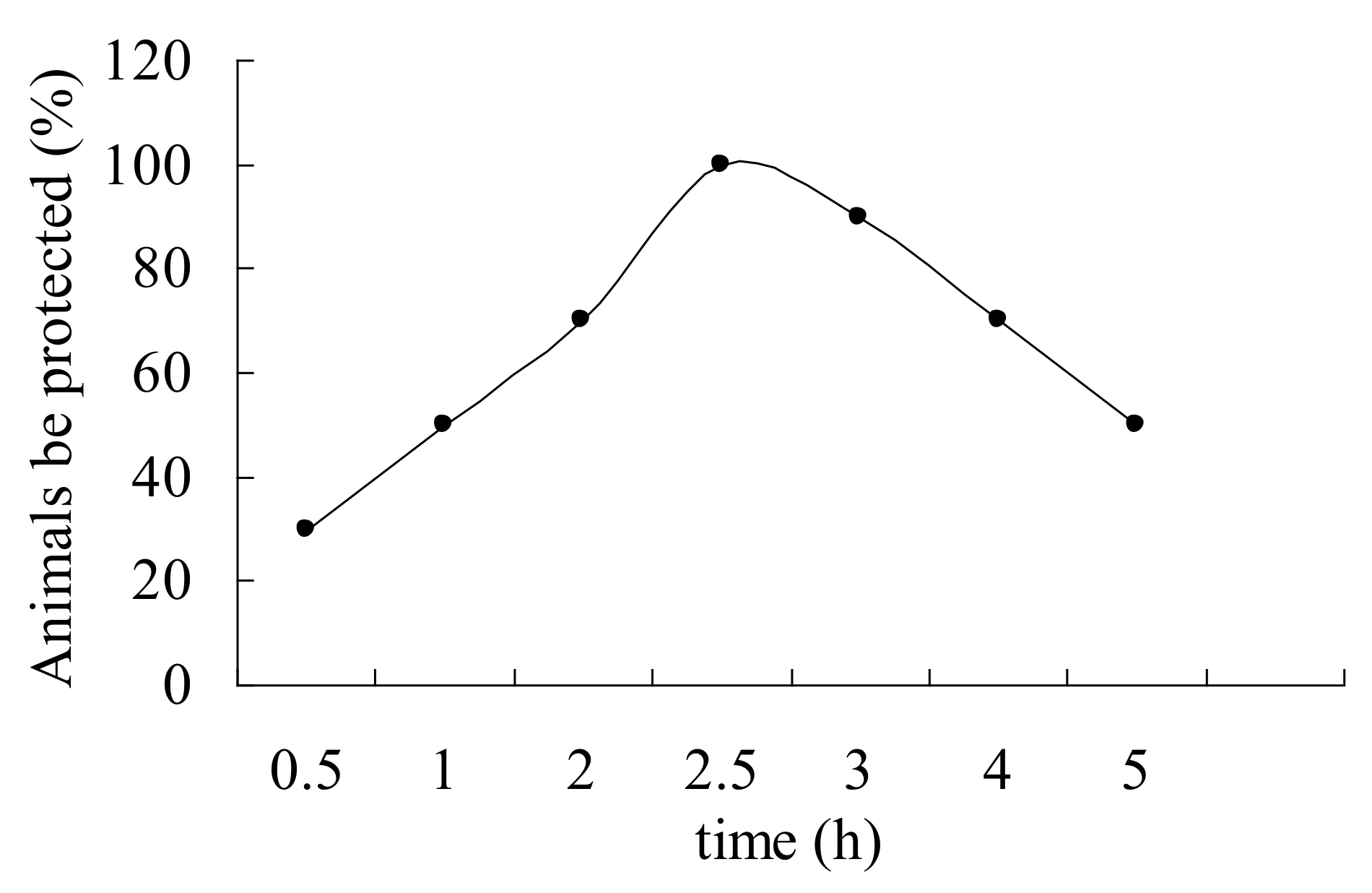
2.3. Anticonvulsant Activity Evaluation of Compound 4g in Other Seizure Models
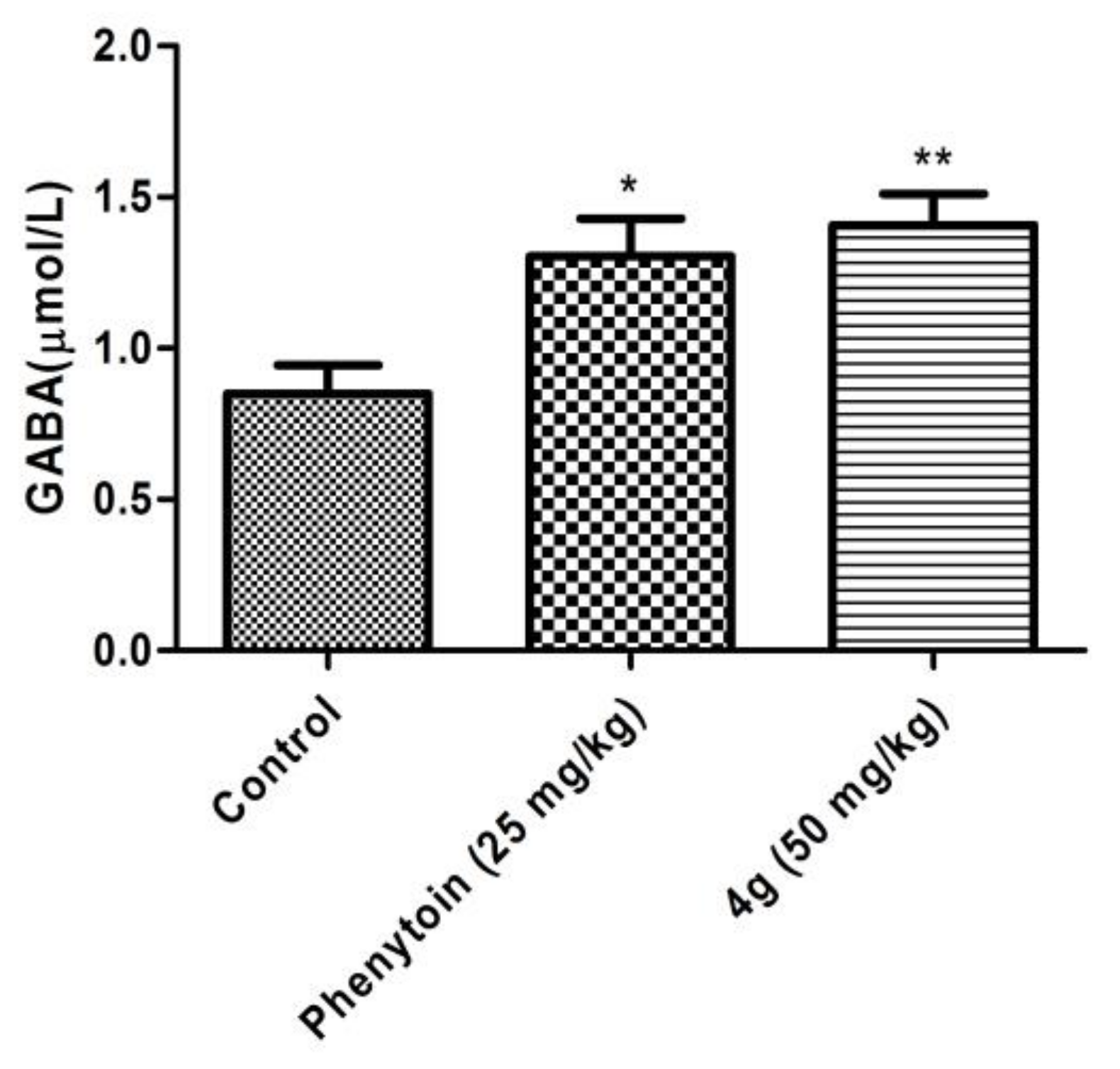
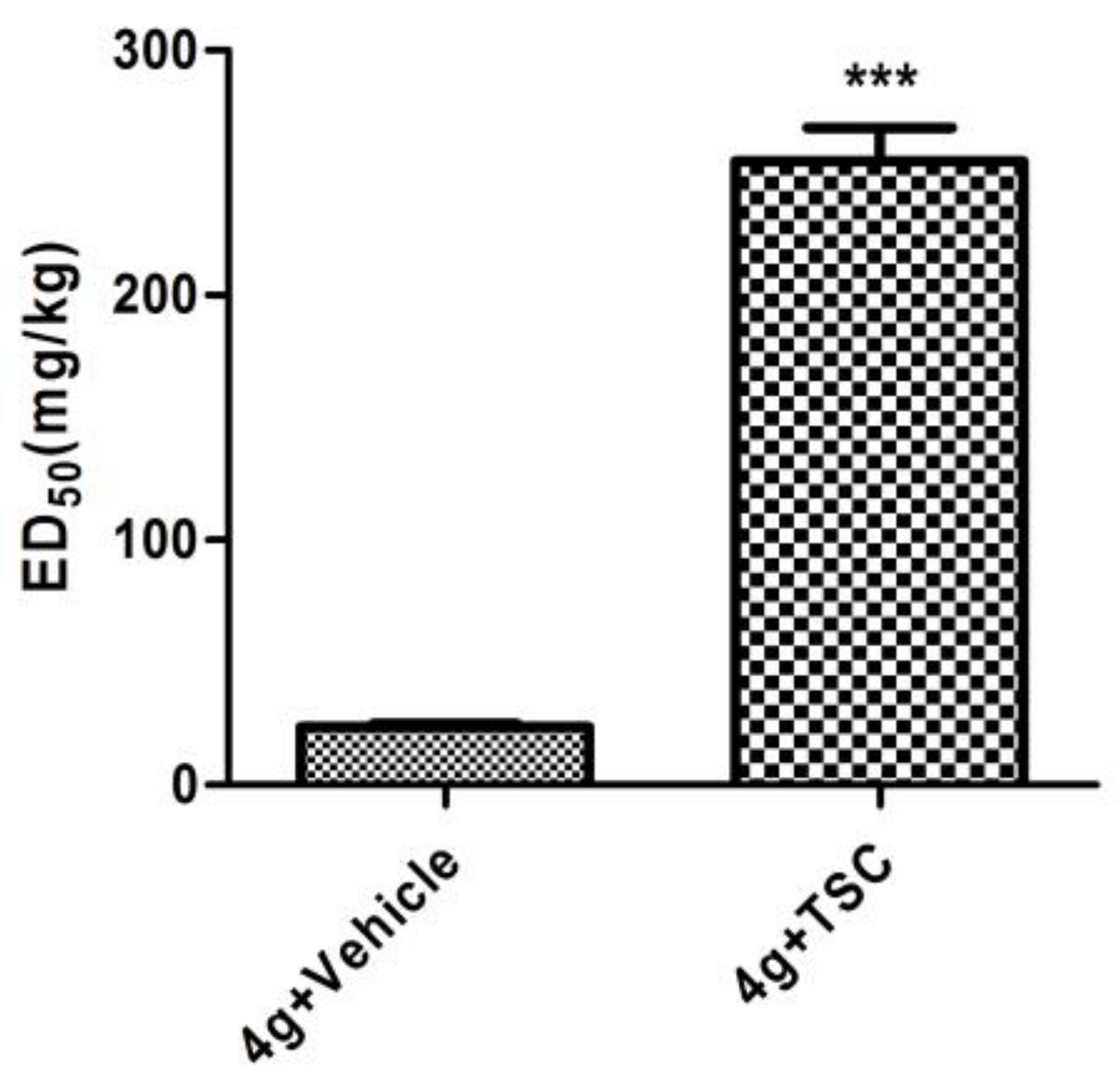
2.4. The Role of the GABAergic System in the Anticonvulsant Mechanism of 4g
3. Materials and Methods
3.1. Instruments and Reagents
3.2. Synthesis Method and Spectral Data
3.2.1. General Procedure for the Preparation of Intermediates 1, 2 and 3
3.2.2. General Procedure for the Preparation of Target Compounds 4a–m
3.2.3. Spectral Data
3.3. Pharmacology
3.3.1. Animals and Experimental Conditions
3.3.2. The Maximal Electroshock (MES) Test
3.3.3. Subcutaneous Pentylenetetrazole Induced Seizures Test (scPTZ)
3.3.4. Neurotoxicity Screening
3.3.5. Quantification Trials
3.3.6. 3-Mercaptopropionic Acid-Induced Seizures Test
3.3.7. Bicuculline-Induced Seizures Test
3.3.8. Determination of Brain GABA Concentrations by ELISA
3.3.9. Test for the Effects of Thiosemicarbazide on the Anti-MES Action of 4g
3.3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Beghi, E. Addressing the burden of epilepsy: Many unmet needs. Pharmacol. Res. 2016, 107, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Sankaraneni, R.; Antony, A.R. Evidence-based guidelines for the management of epilepsy. Neurol. India 2017, 65, S6–S11. [Google Scholar] [PubMed]
- Mcphee, S.J.; Hammer, G.D. Pathophysiology of Disease: An Introduction to Clinical Medicine, 6th ed.; Open University Press: Maidenhead, UK, 2010. [Google Scholar]
- Gaitatzis, A.; Sander, J.W. The long-term safety of antiepileptic drugs. CNS Drugs 2013, 27, 435–455. [Google Scholar] [CrossRef] [PubMed]
- Błaszczyk, B.; Lasoń, W.; Czuczwar, S. Antiepileptic drugs and adverse skin reactions: An update. Pharmacol. Rep. 2015, 67, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, P.; Ballarín, E.; Sabaté, M.; Vidal, X.; Rottenkolber, M.; Amelio, J.; Hasford, J.; Schmiedl, S.; Ibáñez, L. Antiepileptic drugs and suicide: A systematic review of adverse effects. Neuroepidemiology 2014, 42, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.D.; He, X.Y.; Rao, B.Q.; Cheng, B.B.; Song, M.X.; Deng, X.Q. Synthesis and Evaluation of the Anticonvulsant Activities of Triazole-Containing Benzo[d]oxazoles. Chin. J. Org. Chem. 2016, 36, 2449–2455. [Google Scholar] [CrossRef][Green Version]
- Cui, L.J.; Xie, Z.F.; Piao, H.R.; Li, G.; Chai, K.Y.; Quan, Z.S. Synthesis and anticonvulsant activity of 1-substituted-7-benzyloxy-4,5-dihydro-[1,2,4]triazolo[4,3-a]quinoline. Biol. Pharm. Bull. 2005, 28, 1216–1220. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.G.; Sun, X.Y.; Chai, K.Y.; Piao, H.R.; Quan, Z.S. Anticonvulsant and toxicity evaluation of some 7-alkoxy-4,5-dihydro-[1,2,4]triazolo[4,3-a]quinoline-1(2H)-ones. Bioorg. Med. Chem. 2006, 14, 6868–6873. [Google Scholar] [CrossRef] [PubMed]
- Dawidowski, M.; Lewandowski, W.; Turło, J. Synthesis of new perhydropyrrolo[1,2-a]pyrazine derivatives and their evaluation in animal models of epilepsy. Molecules 2014, 19, 15955–15981. [Google Scholar] [CrossRef] [PubMed]
- Loscher, W. Critical review of current animal models of seizures and epilepsy used in the discovery and development of new antiepileptic drugs. Seizure 2011, 20, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Katz, L.; Cohen, M.S. Benzoxazole derivatives. I. 2-mercaptobenzoxazoles. J. Org. Chem. 1954, 19, 758–766. [Google Scholar] [CrossRef]
- Shu, B.; Zheng, Y.; Wang, S.B.; Deng, X.Q.; Quan, Z.S. Design, synthesis, and anticonvulsant activity evaluation of 4-(3-alkoxy-phenyl)-2,4-dihydro-[1,2,4]triazol-3-ones. Arch. Pharm. 2013, 346, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Crick, E.W.; Osorio, I.; Frei, M.; Mayer, A.P.; Lunte, C.E. Correlation of 3-mercaptopropionic acid induced seizures and changes in striatal neurotransmitters monitored by microdialysis. Eur. J. Pharm. Sci. 2014, 57, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.; Rubio-Osornio, M.; Retana-Márquez, S.; Verónica Custodio, M.L.; Paz, C. In vivo experimental models of epilepsy. Cent. Nerv. Syst. Agents Med. Chem. 2010, 10, 298–309. [Google Scholar] [CrossRef] [PubMed]
- Collins, G.S. Effect of aminooxyacetic acid, thiosemicarbazide and haloperidol on the metabolism and half-lives of glutamate and GABA in rat brain. Biochem. Pharmacol. 1973, 22, 101–111. [Google Scholar] [CrossRef]
- Krall, R.L.; Penry, J.K.; White, B.G.; Kupferberg, H.J.; Swinyard, E.A. Antiepileptic drug development: II. Anticonvulsant drug screening. Epilepsia 1978, 19, 409–428. [Google Scholar] [CrossRef] [PubMed]
- Porter, R.J. Antiepileptic drug development program. Prog. Clin. Biol. Res. 1983, 127, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.J.; Shen, Q.K.; Jin, C.M.; Quan, Z.S. Synthesis and Pharmacological Evaluation of New 3,4-Dihydroisoquinolin Derivatives Containing Heterocycle as Potential Anticonvulsant Agents. Molecules 2016, 21, 1635. [Google Scholar] [CrossRef] [PubMed]
- White, H.S.; Woodhead, J.H.; Wilcox, K.S.; Stables, J.P.; Kupferberg, H.J.; Wolf, H.H. Antiepileptic Drugs; Levy, R.H., Mattson, R.H., Meldrum, B.S., Perucca, E., Eds.; Lippincott Williams & Wilkins Publishers: New York, NY, USA, 2002; pp. 36–48. [Google Scholar]
- Huisman, H.; Wynveen, P.; Nichkova, M.; Kellermann, G. Novel ELISAs for screening of the biogenic amines GABA, glycine, beta-phenylethylamine, agmatine, and taurine using one derivatization procedure of whole urine samples. Anal. Chem. 2010, 82, 6526–6533. [Google Scholar] [CrossRef] [PubMed]
- Song, M.X.; Xiao, F.; Yu, H.H.; Liu, B.; Deng, X.Q. The Anticonvulsive Activities of Venlafaxine and its Interactions with Some Antiepileptic Drugs. Lat. Am. J. Pharm. 2016, 35, 1959–1965. [Google Scholar]
Sample Availability: Samples of the compounds (4a–m) are available from the authors. |
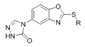
| Compds. | R | Intraperitioneal Injection in Mice a | |||||
|---|---|---|---|---|---|---|---|
| MES Screening b | scPTZ Screening c | NT Screening d | |||||
| 0.5 h | 4 h | 0.5 h | 4 h | 0.5 h | 4 h | ||
| 4a | C3H7 | 100 | 300 | 300 | - | - | - |
| 4b | C5H9 | 300 | - | - | - | - | - |
| 4c | C7H15 | - | - | - | - | - | - |
| 4d | CH2C6H5 | - | - | - | - | - | - |
| 4e | CH2C6H4(2-F) | 100 | - | 100 | 100 | - | - |
| 4f | CH2C6H4(3-F) | 100 | - | 100 | 100 | - | - |
| 4g | CH2C6H4(4-F) | 100 | 30 | 30 | 30 | 300 | 300 |
| 4h | CH2C6H4(2-Cl) | 100 | 100 | - | 300 | - | - |
| 4i | CH2C6H4(3-Cl) | 100 | 100 | - | - | - | - |
| 4j | CH2C6H4(4-Cl) | 300 | - | - | - | - | - |
| 4k | CH2C6H3(2,4-Cl2) | 300 | - | - | - | - | - |
| 4l | CH2C6H4(4-CH3) | 300 | 300 | - | 300 | - | - |
| 4m | CH2C6H4(4-OCH3) | - | - | - | - | - | - |
| Compounds | ED50 a | TD50 b | PI c | ||
|---|---|---|---|---|---|
| MES | scPTZ | MES | scPTZ | ||
| 4g | 23.7 (21.5–26.0) | 18.9 (17.2–20.8) | 284.0 (258.2–312.4) | 12.0 | 15.0 |
| Carbamazepine | 9.8 (8.9–10.8) | >100 | 44.0 (40.2–48.1) | 4.5 | <0.44 |
| Valproate | 264 (247–338) | 149 (123–177) | 418 (369–450) | 1.6 | 2.8 |
| Compound | Doses (mg/kg) | Test Time (h) | Clonic Seizures (%) | Tonic Seizures (%) | Lethality (%) |
|---|---|---|---|---|---|
| DMSO | - | 0.5 | 100 | 100 | 60 |
| Carbamazepine | 30 | 0.5 | 100 | 0 *** | 0 ** |
| 4g | 30 | 0.5 | 100 | 20 ** | 30 * |
| Compound | Doses (mg/kg) | Test Time (h) | Clonic Seizures (%) | Tonic Seizures (%) | Lethality (%) |
|---|---|---|---|---|---|
| DMSO | - | 0.5 | 100 | 100 | 100 |
| Carbamazepine | 30 | 0.5 | 100 | 0 *** | 20 ** |
| 4g | 30 | 0.5 | 100 | 0 *** | 30 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, M.-X.; Wang, Z.-Y.; He, S.-H.; Yu, S.-W.; Chen, S.-L.; Guo, D.-F.; Zhao, W.-H.; Deng, X.-Q. Synthesis and Evaluation of the Anticonvulsant Activities of 4-(2-(Alkylthio)benzo[d]oxazol-5-yl)-2,4-dihydro-3H-1,2,4-triazol-3-ones. Molecules 2018, 23, 756. https://doi.org/10.3390/molecules23040756
Song M-X, Wang Z-Y, He S-H, Yu S-W, Chen S-L, Guo D-F, Zhao W-H, Deng X-Q. Synthesis and Evaluation of the Anticonvulsant Activities of 4-(2-(Alkylthio)benzo[d]oxazol-5-yl)-2,4-dihydro-3H-1,2,4-triazol-3-ones. Molecules. 2018; 23(4):756. https://doi.org/10.3390/molecules23040756
Chicago/Turabian StyleSong, Ming-Xia, Zhen-Yuan Wang, Shi-Hui He, Sheng-Wang Yu, Shi-Long Chen, Dong-Fu Guo, Wen-Hao Zhao, and Xian-Qing Deng. 2018. "Synthesis and Evaluation of the Anticonvulsant Activities of 4-(2-(Alkylthio)benzo[d]oxazol-5-yl)-2,4-dihydro-3H-1,2,4-triazol-3-ones" Molecules 23, no. 4: 756. https://doi.org/10.3390/molecules23040756
APA StyleSong, M.-X., Wang, Z.-Y., He, S.-H., Yu, S.-W., Chen, S.-L., Guo, D.-F., Zhao, W.-H., & Deng, X.-Q. (2018). Synthesis and Evaluation of the Anticonvulsant Activities of 4-(2-(Alkylthio)benzo[d]oxazol-5-yl)-2,4-dihydro-3H-1,2,4-triazol-3-ones. Molecules, 23(4), 756. https://doi.org/10.3390/molecules23040756




