Synthesis of Some Novel Thiadiazole Derivative Compounds and Screening Their Antidepressant-Like Activities
Abstract
:1. Introduction
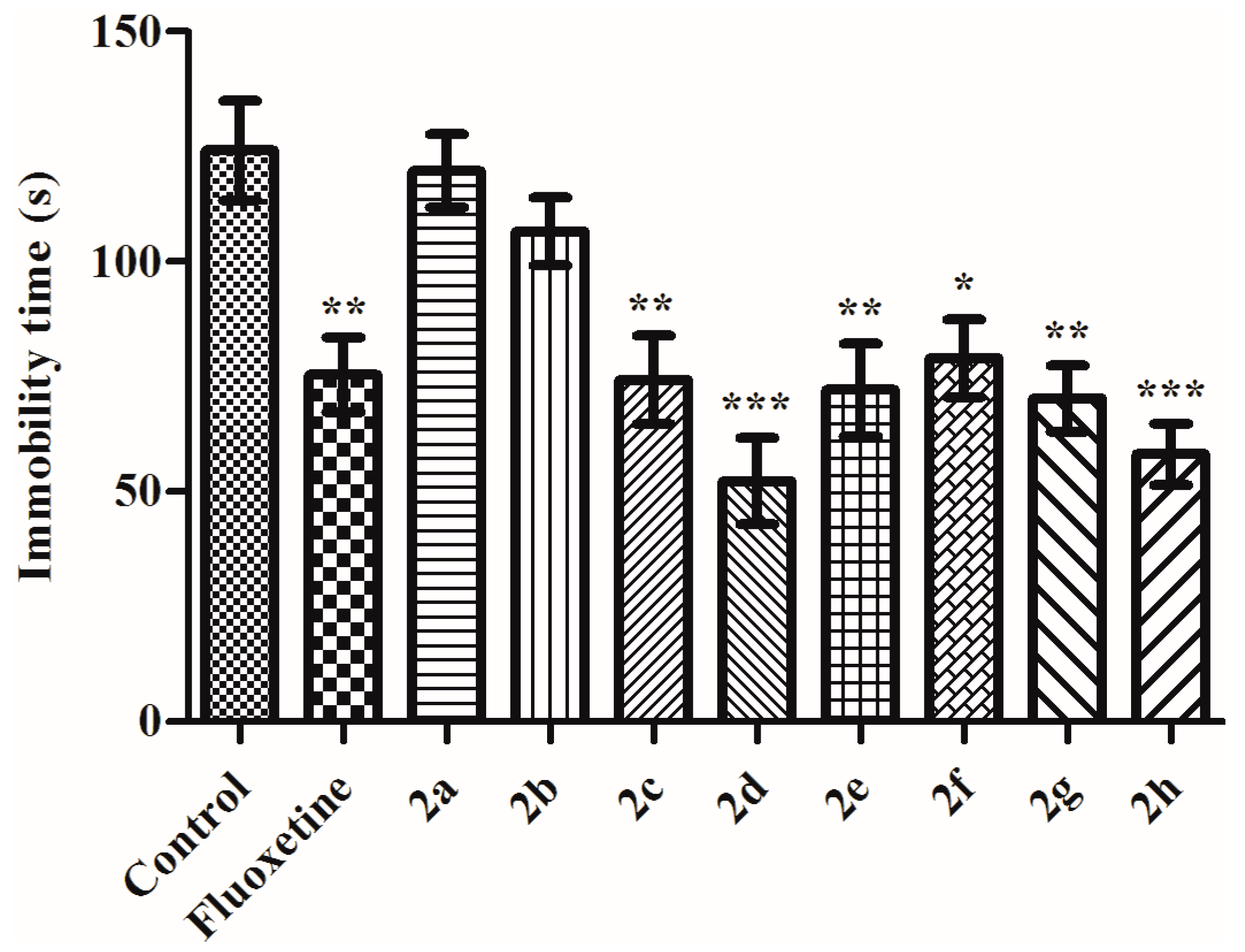
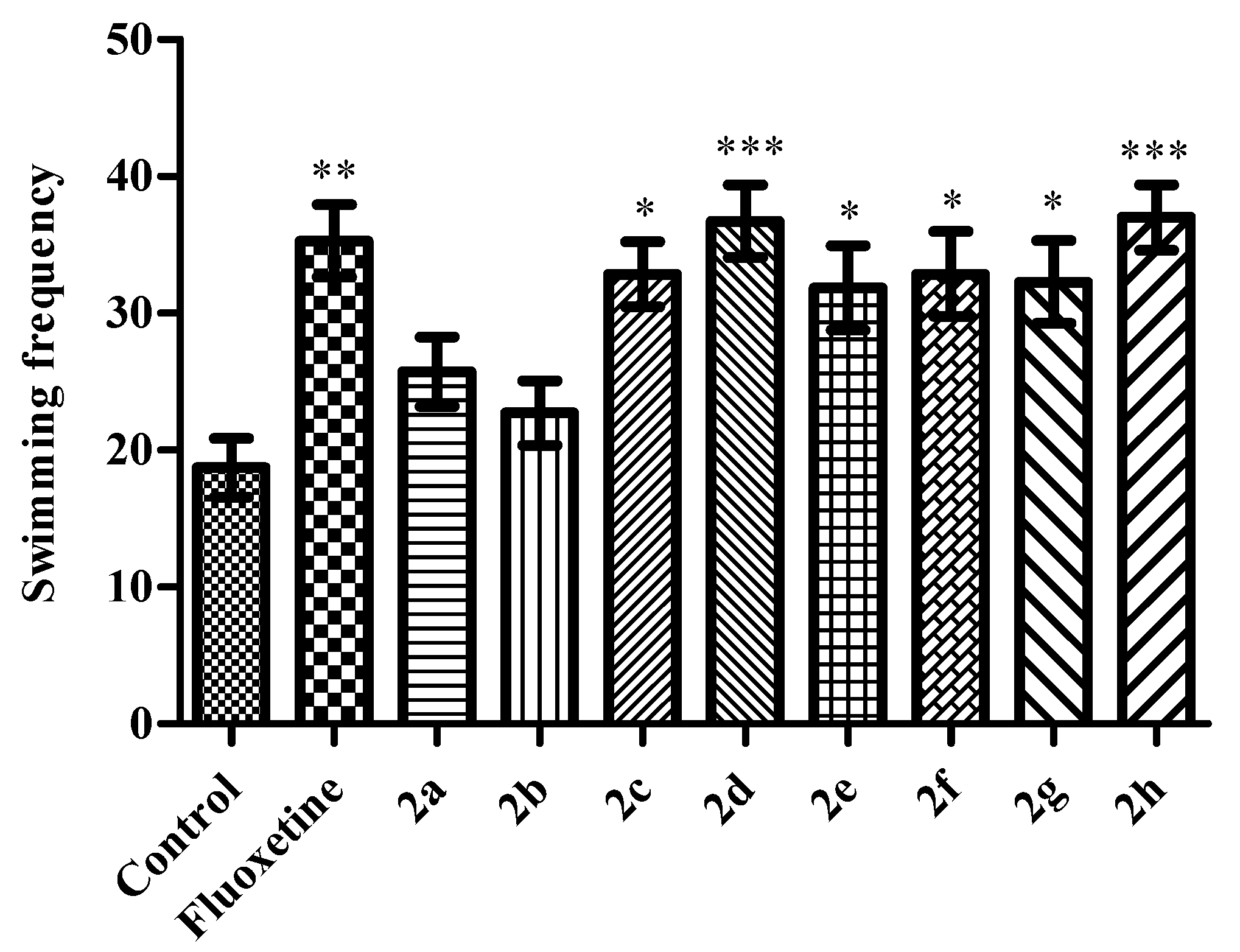
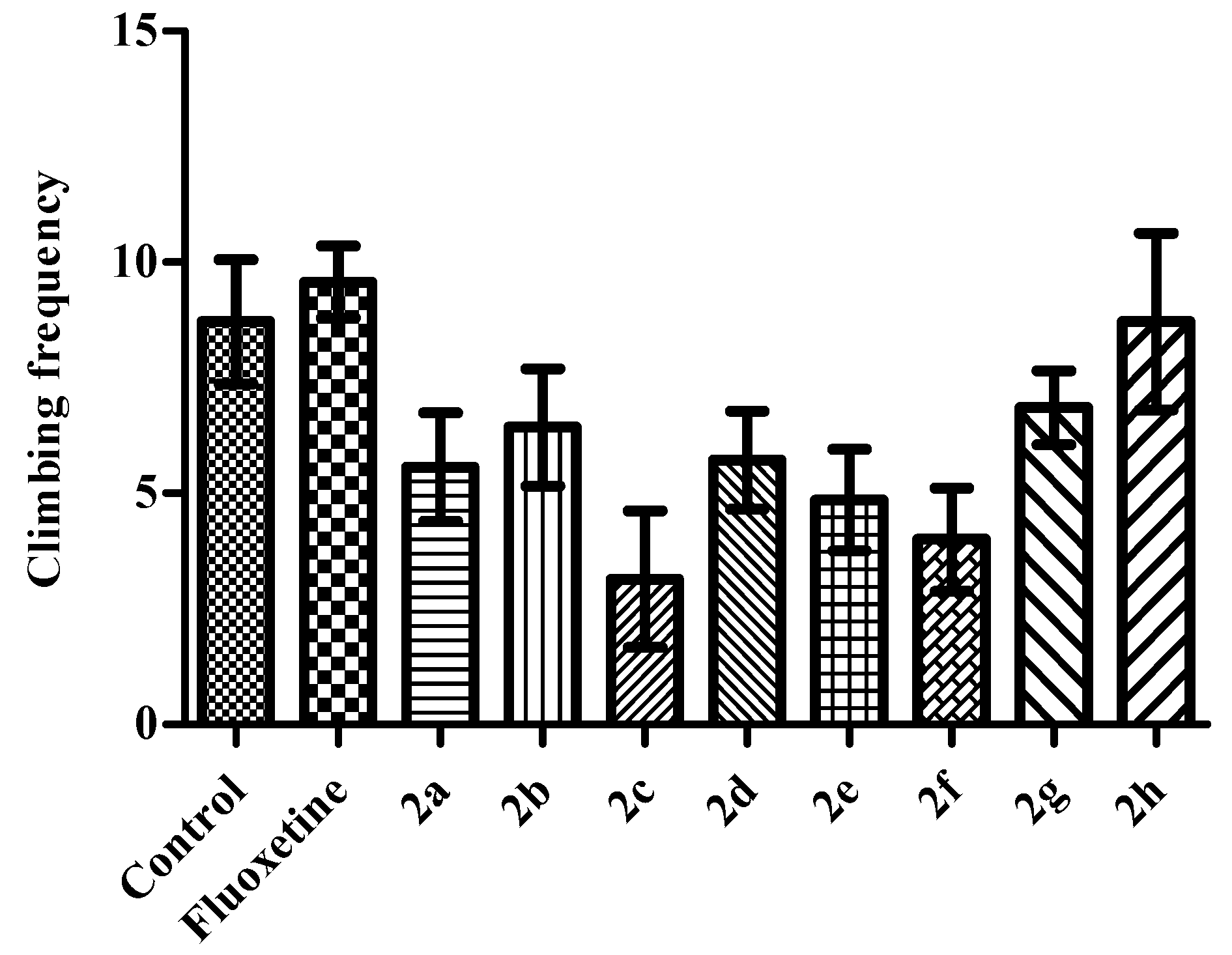
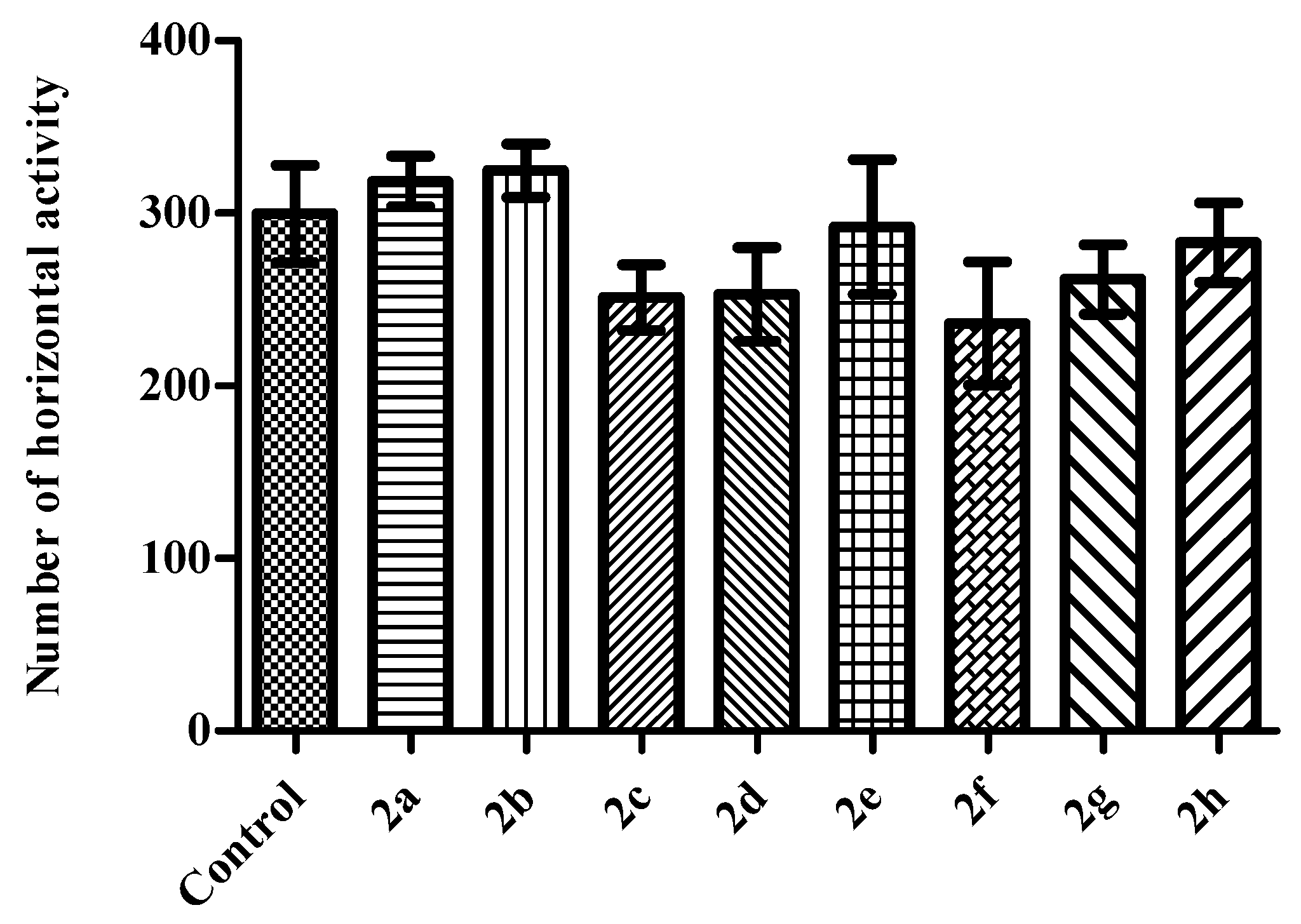
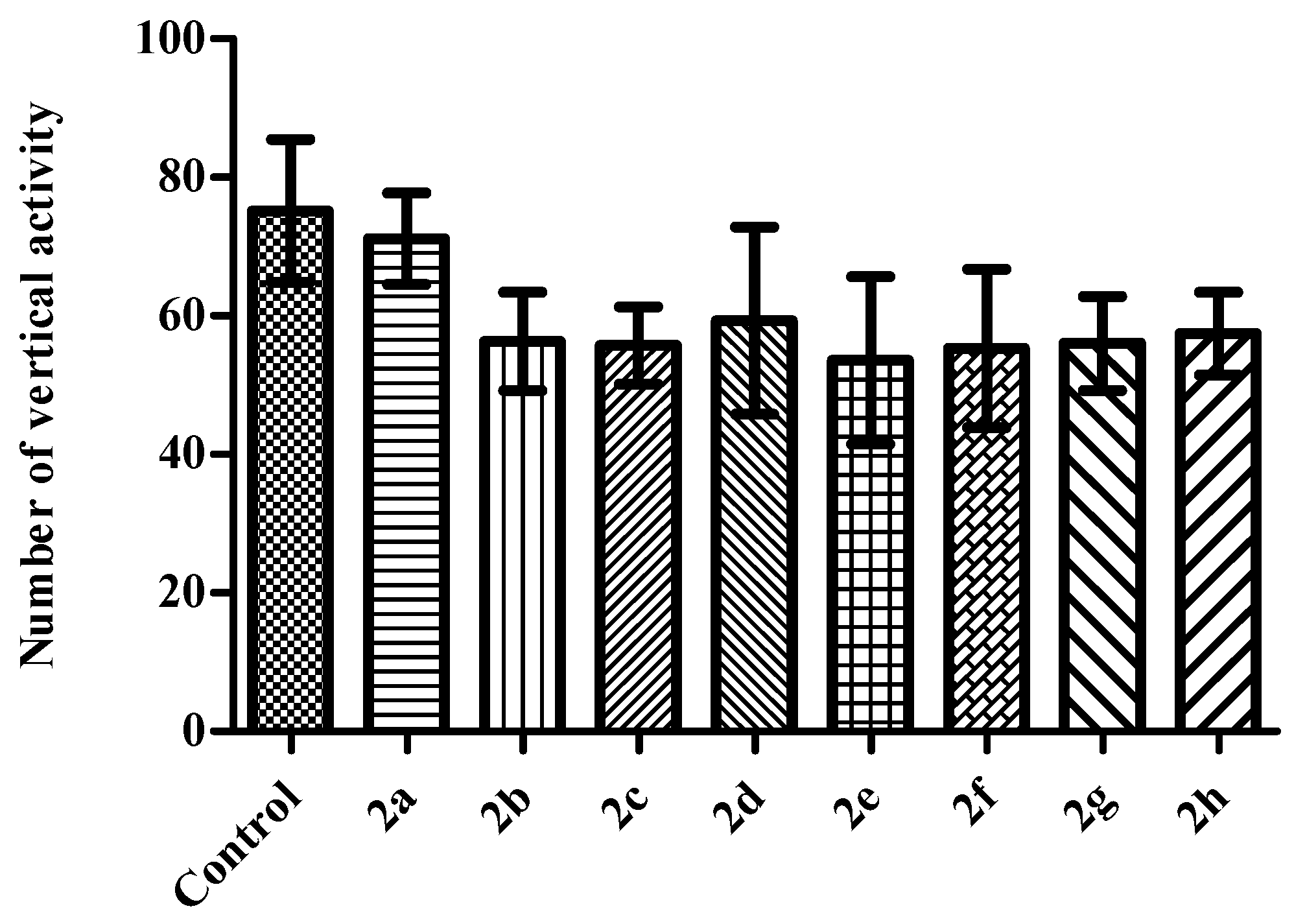
2. Results and Discussion
3. Materials and Methods
3.1. Chemistry
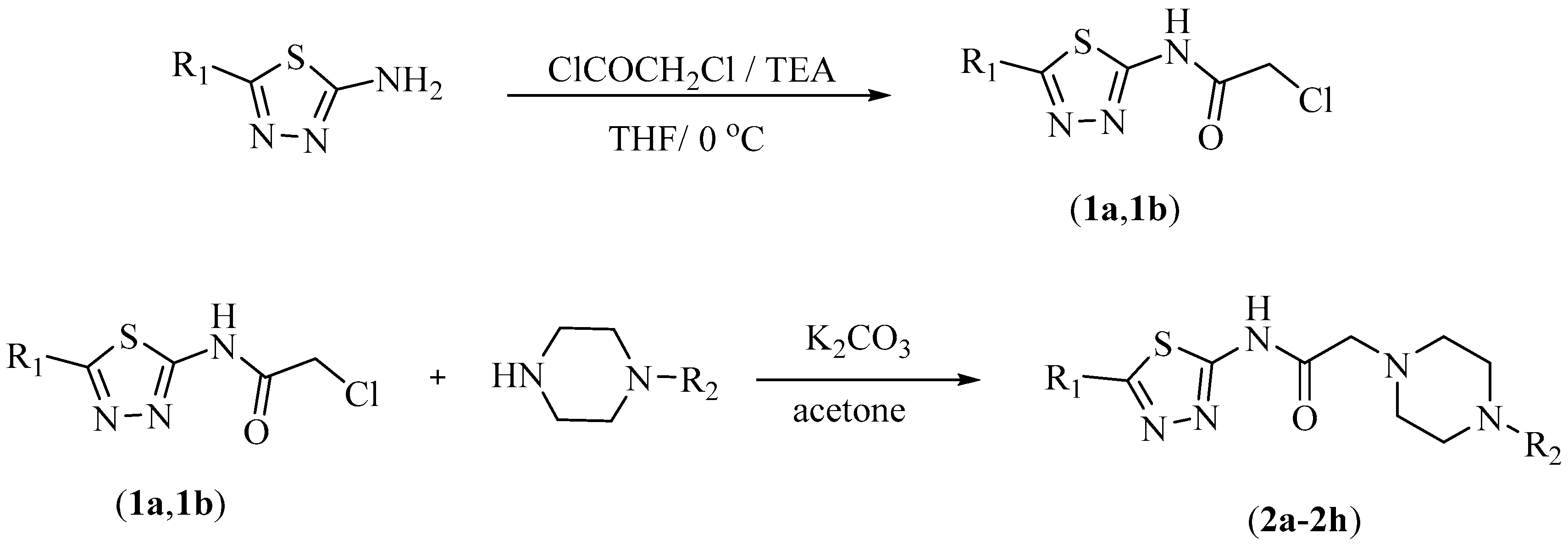
3.1.1. Synthesis of 2-Chloro-N-(5-methyl/ethyl-1,3,4-thiadiazol-2-yl)acetamide (1a, 1b)
3.1.2. Synthesis of 2-(4-Substituepiperazin-1-yl)-N-(5-methyl/ethyl-1,3,4-thiadiazol-2-yl)acetamide (2a–2h)
3.2. Prediction of ADME Parameters and BBB Permeability
3.3. Pharmacology
3.3.1. Animals
3.3.2. Administration of Compounds
3.3.3. Behavioral Tests
Tail Suspension Test
Modified Forced Swimming Test
Activity Cage Test
Statistical Analyses
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- WHO. MhGAP Intervention Guide for Mental, Neurological and Substance Use Disorders in Non-Specialized Health Settings, 2010. Available online: http://apps.who.int/iris/bitstream/10665/44406/1/9789241548069_eng.pdf (accessed on 15 January 2018).
- WHO. Depression: A Global CrisisWorld Mental Health Day, 2012. Available online: http://www.who.int/mental_health/management/depression/wfmh_paper_depression_wmhd_2012.pdf (accessed on 1 February 2018).
- Paschos, K.A.; Veletza, S.; Chatzaki, E. Neuropeptide and sigma receptors as novel therapeutic targets for the pharmacotherapy of depression. CNS Drugs 2009, 23, 755–772. [Google Scholar] [CrossRef]
- Can, O.D.; Altintop, M.D.; Ozkay, U.D.; Uçel, U.I.; Doğruer, B.; Kaplancikli, Z.A. Synthesis of thiadiazole derivatives bearing hydrazone moieties and evaluation of their pharmacological effects on anxiety, depression, and nociception parameters in mice. Arch. Pharm. Res. 2012, 35, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Altıntop, M.D.; Can, Ö.D.; Demir Özkay, Ü.; Kaplancıklı, Z.A. Synthesis and evaluation of new 1,3,4-thiadiazole derivatives as antinociceptive agents. Molecules 2016, 21, 1004. [Google Scholar] [CrossRef]
- Ragab, F.A.; Heiba, H.I.; El-Gazzar, M.G.; Abou-Seri, S.M.; El-Sabbagh, W.A.; El-Hazek, R.M. Anti-inflammatory, analgesic and COX-2 inhibitory activity of novel thiadiazoles in irradiated rats. J. Photochem. Photobiol. B. 2017, 166, 285–300. [Google Scholar] [CrossRef]
- Pandey, A.; Dewangan, D.; Verma, S.; Mishra, A.; Dubey, R.D. Synthesis of Schiff bases of 2-amino-5-aryl-1,3,4-thiadiazole and its analgesic, anti-inflammatory, anti-bacterial and anti-tubercular activity. Int. J. Chem. Tech. Res. 2011, 3, 178–184. [Google Scholar] [CrossRef]
- Harish, K.P.; Mohana, K.N.; Mallesha, L. Synthesis of new 2,5-disubstituted-1,3,4-thiadiazole derivatives and their in vivo anticonvulsant activity. Bioorg. Khim. 2014, 40, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, N.; Ahuja, P.; Malik, S.; Arya, S.K. Design of benzothiazole-1,3,4-thiadiazole conjugates: Synthesis and anticonvulsant evaluation. Arch. Pharm. 2013, 346, 819–831. [Google Scholar] [CrossRef] [PubMed]
- Pattanayak, P.; Sharma, R.; Sahoo, P.K. Synthesis and evaluation of 2-amino-5-sulfanyl-1,3,4-thiadiazoles as antidepressant, anxiolytic, and anticonvulsant agents. Med. Chem. Res. 2009, 18, 351–361. [Google Scholar] [CrossRef]
- Sharma, R.; Misra, G.P.; Sainy, J.; Chaturvedi, S.C. Synthesis and biological evaluation of 2-amino-5-sulfanyl-1,3,4-thiadiazole derivatives as antidepressant, anxiolytics and anticonvulsant agents. Med. Chem. Res. 2011, 20, 245–253. [Google Scholar] [CrossRef]
- Clerici, F.; Pocar, D.; Guido, M.; Loche, A.; Perlini, V.; Brufani, M. Synthesis of 2-amino-5-sulfanyl-1,3,4-thiadiazole derivatives and evaluation of their antidepressant and anxiolytic activity. J. Med. Chem. 2001, 44, 931–936. [Google Scholar] [CrossRef] [PubMed]
- Varvaresou, A.; Siatra-Papastaikoudi, T.; Dalla Tsotinis, A.; Tsantili-Kakoulidou, A.; Vamvakides, A. Synthesis, lipophilicity and biological evaluation of indole-containing derivatives of 1,3,4-thiadiazole and 1,2, 4-triazole. Farmaco 1998, 53, 320–326. [Google Scholar] [CrossRef]
- De Waterbeemd, H.V.; Gifford, E. ADMET in silico modelling: towards prediction paradise? Nat. Rev. Drug Discov. 2013, 2, 192–204. [Google Scholar]
- M. Cheminformatics, Bratislava, Slovak Republic. Available online: http://www.molinspiration.com/services/properties.html (accessed on 17 December 2017).
- Carpenter, T.S.; Kirshner, D.A.; Lau, E.Y.; Wong, S.E.; Nilmeier, J.P.; Lightstone, F.C. A method to predict blood-brain barrier permeability of drug-like compounds using molecular dynamics simulations. Biophys. J. 2014, 107, 630–641. [Google Scholar] [CrossRef]
- Online BBB Predictor. Available online: http://www.cbligand.org/BBB/index.php (accessed on 17 December 2017).
- Cryan, J.F.; Mombereau, C.; Vassout, A. The tail suspension test as a model for assessing antidepressant activity: Review of pharmacological and genetic studies in mice. Neurosci. Biobehav. Rev. 2005, 29, 571–625. [Google Scholar] [CrossRef]
- Slattery, D.A.; Cryan, J.F. Using the rat forced swim test to assess antidepressant-like activity in rodents. Nat. Protoc. 2012, 7, 1009–1014. [Google Scholar] [CrossRef]
- Cryan, J.F.; Markou, A.; Lucki, I. Assessing antidepressant activity in rodents: recent developments and future needs. Trends Pharmacol. Sci. 2002, 23, 238–245. [Google Scholar] [CrossRef]
- Shakya, A.K.; Patnaik, G.K.; Mishra, P. Synthesis and biological evaluation of 2-[substituted acetyl]amino-5-alkyl-1,3,4-thiadiazoles. Eur. J. Med. Chem. 1992, 27, 67–71. [Google Scholar] [CrossRef]
- Can, Ö.D.; Demir Özkay, Ü.; Üçel, U.İ. Anti-depressant-like effect of vitexin in BALB/c mice and evidence for the involvement of monoaminergic mechanisms. Eur. J. Pharmacol. 2013, 699, 250–257. [Google Scholar] [CrossRef]
- Steru, L.; Chermat, R.; Thierry, B.; Simon, P. The tail suspension test: A new method for screening antidepressants in mice. Psychopharmacology 1985, 85, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Can, Ö.D.; Turan, N.; Demir Özkay, Ü.; Öztürk, Y. Antidepressant-like effect of gallic acid in mice: Dual involvement of serotonergic and catecholaminergic systems. Life Sci. 2017, 190, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Palotai, M.; Telegdy, G.; Tanaka, M.; Bagosi, Z.; Jászberényi, M. Neuropeptide AF induces anxiety-like and antidepressant-like behavior in mice. Behav. Brain Res. 2014, 274, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Votava, M.; Hess, L.; Slíva, J.; Krsiak, M.; Agová, V. Dexmedetomidine selectively suppresses dominant behavior in aggressive and sociable mice. Eur. J. Pharmacol. 2005, 523, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Demir Özkay, Ü.; Kaya, C.; Acar Çevik, U.; Can, Ö.D. Synthesis and antidepressant activity profile of some novel benzothiazole derivatives. Molecules 2017. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Compounds | R1 | R2 |
|---|---|---|
| 2a | methyl | methyl |
| 2b | methyl | ethyl |
| 2c | methyl | isopropyl |
| 2d | methyl | phenyl |
| 2e | ethylthio | methyl |
| 2f | ethylthio | ethyl |
| 2g | ethylthio | isopropyl |
| 2h | ethylthio | phenyl |
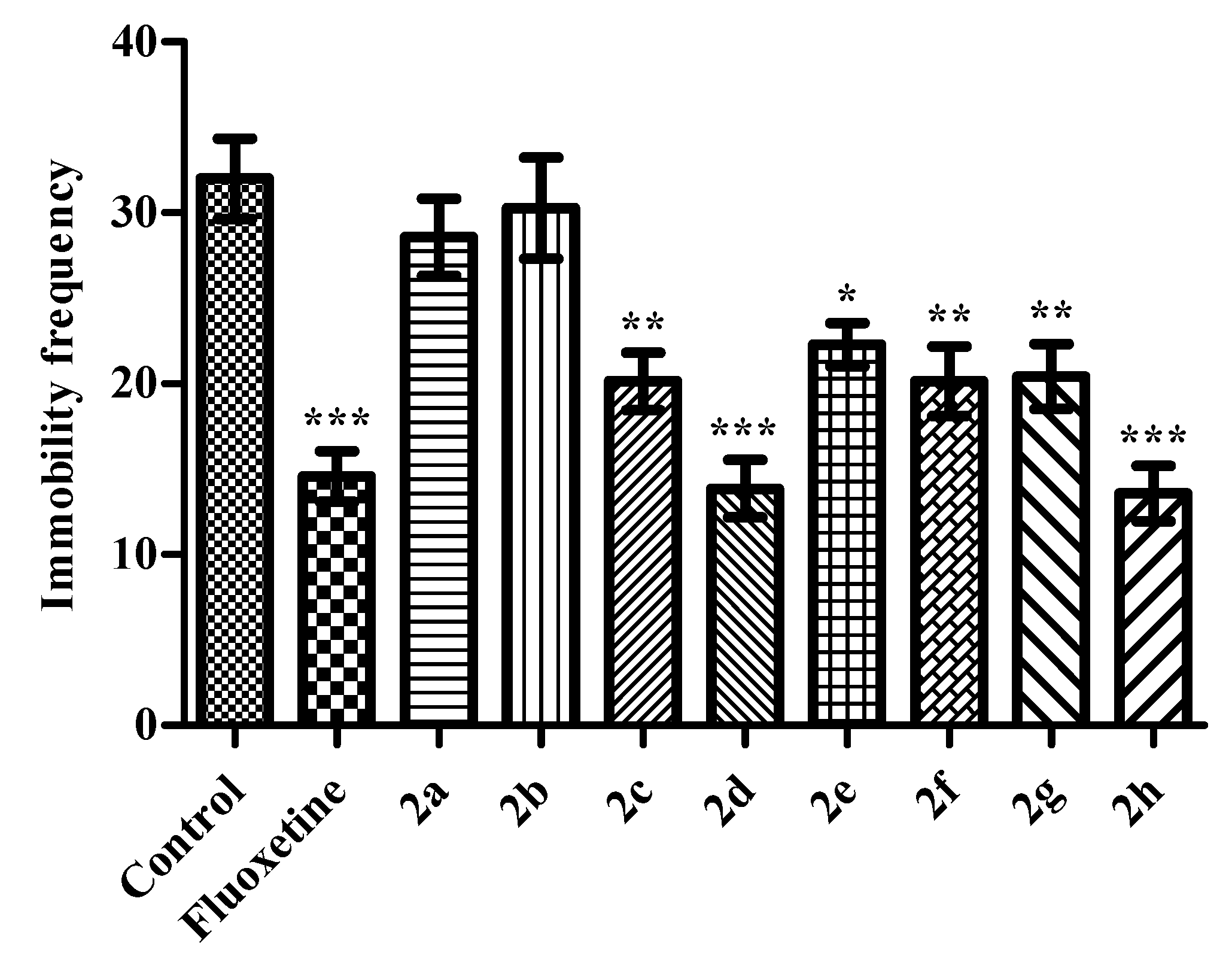
| Comp. | MW | TPSA | Log P | HBA | HBD | MV | Vio | BBB |
|---|---|---|---|---|---|---|---|---|
| 2a | 255.35 | 61.36 | −0.16 | 6 | 1 | 229.69 | 0 | + |
| 2b | 269.37 | 61.36 | 0.21 | 6 | 1 | 246.49 | 0 | + |
| 2c | 283.40 | 61.36 | 0.51 | 6 | 1 | 263.07 | 0 | + |
| 2d | 317.42 | 61.36 | 1.53 | 6 | 1 | 284.53 | 0 | + |
| 2e | 301.44 | 61.36 | 1.22 | 6 | 1 | 264.62 | 0 | + |
| 2f | 315.47 | 61.36 | 1.59 | 6 | 1 | 281.42 | 0 | + |
| 2g | 329.50 | 61.36 | 1.89 | 6 | 1 | 298.00 | 0 | + |
| 2h | 363.51 | 61.36 | 2.92 | 6 | 1 | 319.46 | 0 | + |
| Fluoxetine | 309.33 | 21.26 | 4.53 | 2 | 1 | 275.13 | 0 | + |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Can, N.Ö.; Can, Ö.D.; Osmaniye, D.; Demir Özkay, Ü. Synthesis of Some Novel Thiadiazole Derivative Compounds and Screening Their Antidepressant-Like Activities. Molecules 2018, 23, 716. https://doi.org/10.3390/molecules23040716
Can NÖ, Can ÖD, Osmaniye D, Demir Özkay Ü. Synthesis of Some Novel Thiadiazole Derivative Compounds and Screening Their Antidepressant-Like Activities. Molecules. 2018; 23(4):716. https://doi.org/10.3390/molecules23040716
Chicago/Turabian StyleCan, Nafiz Öncü, Özgür Devrim Can, Derya Osmaniye, and Ümide Demir Özkay. 2018. "Synthesis of Some Novel Thiadiazole Derivative Compounds and Screening Their Antidepressant-Like Activities" Molecules 23, no. 4: 716. https://doi.org/10.3390/molecules23040716
APA StyleCan, N. Ö., Can, Ö. D., Osmaniye, D., & Demir Özkay, Ü. (2018). Synthesis of Some Novel Thiadiazole Derivative Compounds and Screening Their Antidepressant-Like Activities. Molecules, 23(4), 716. https://doi.org/10.3390/molecules23040716





