Chemical and Biological Significance of Oenothein B and Related Ellagitannin Oligomers with Macrocyclic Structure
Abstract
1. Introduction
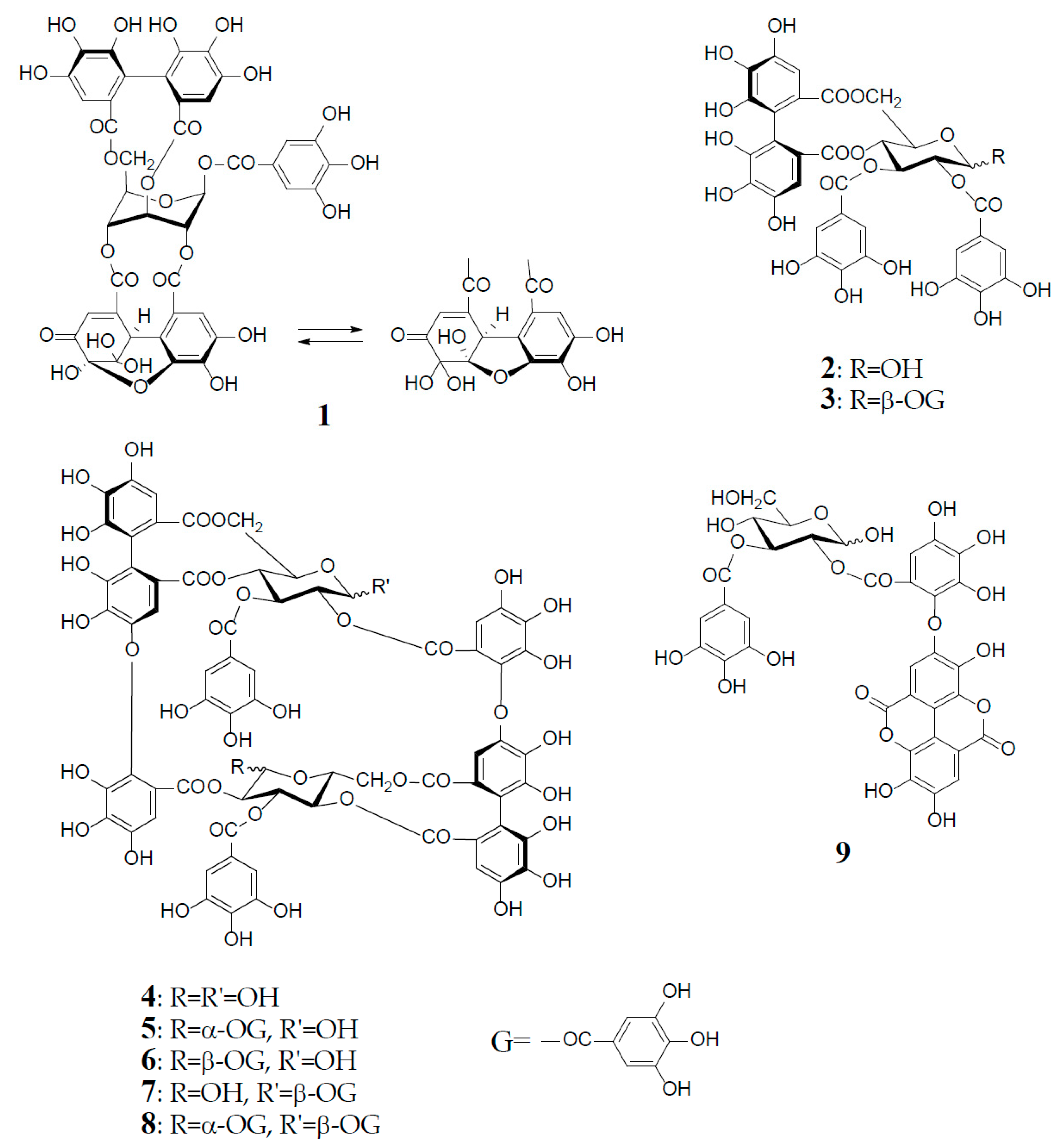
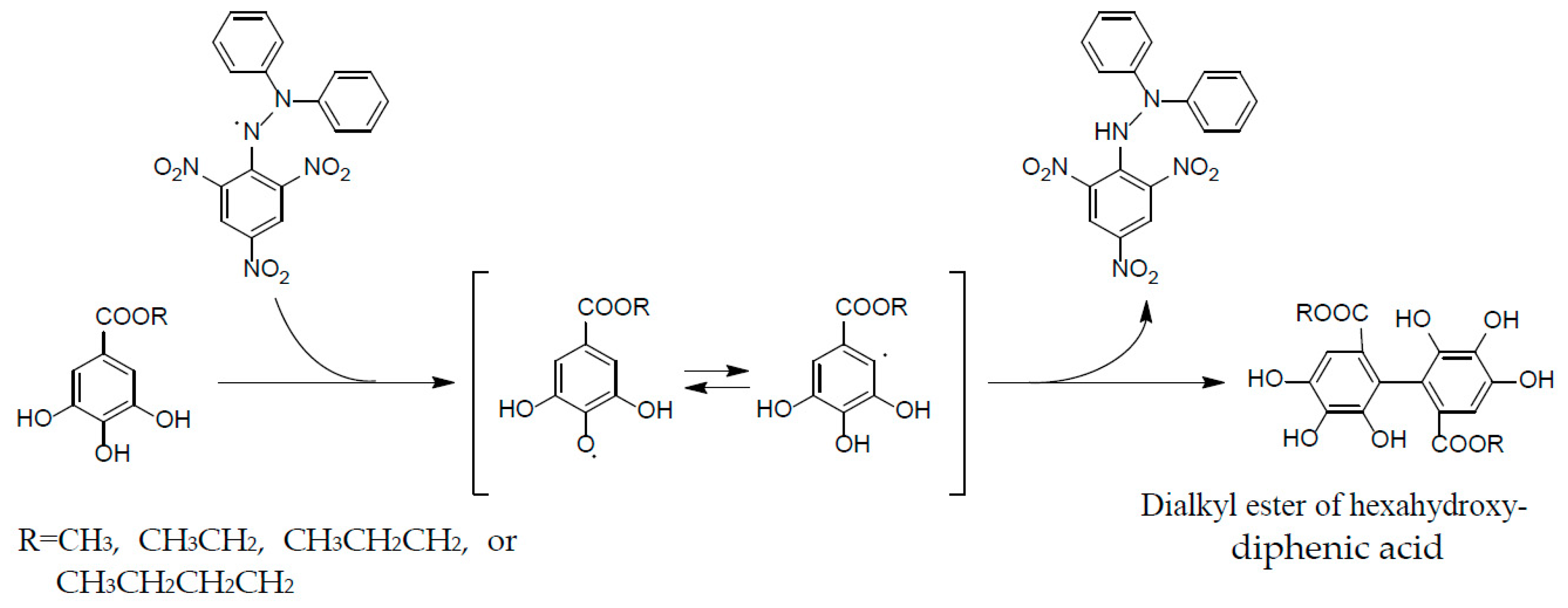
2. Structural Characteristics of Oenothein B
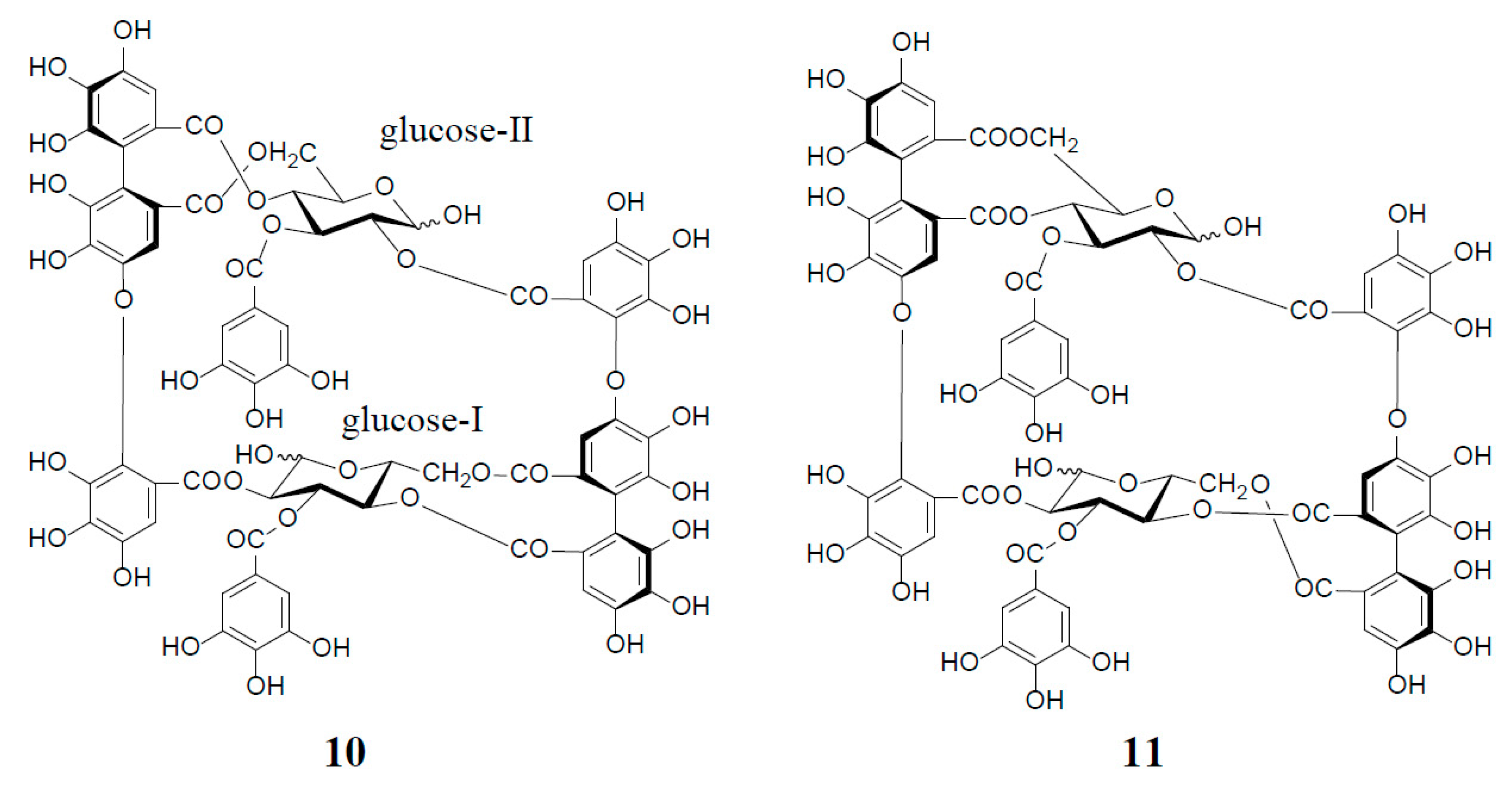
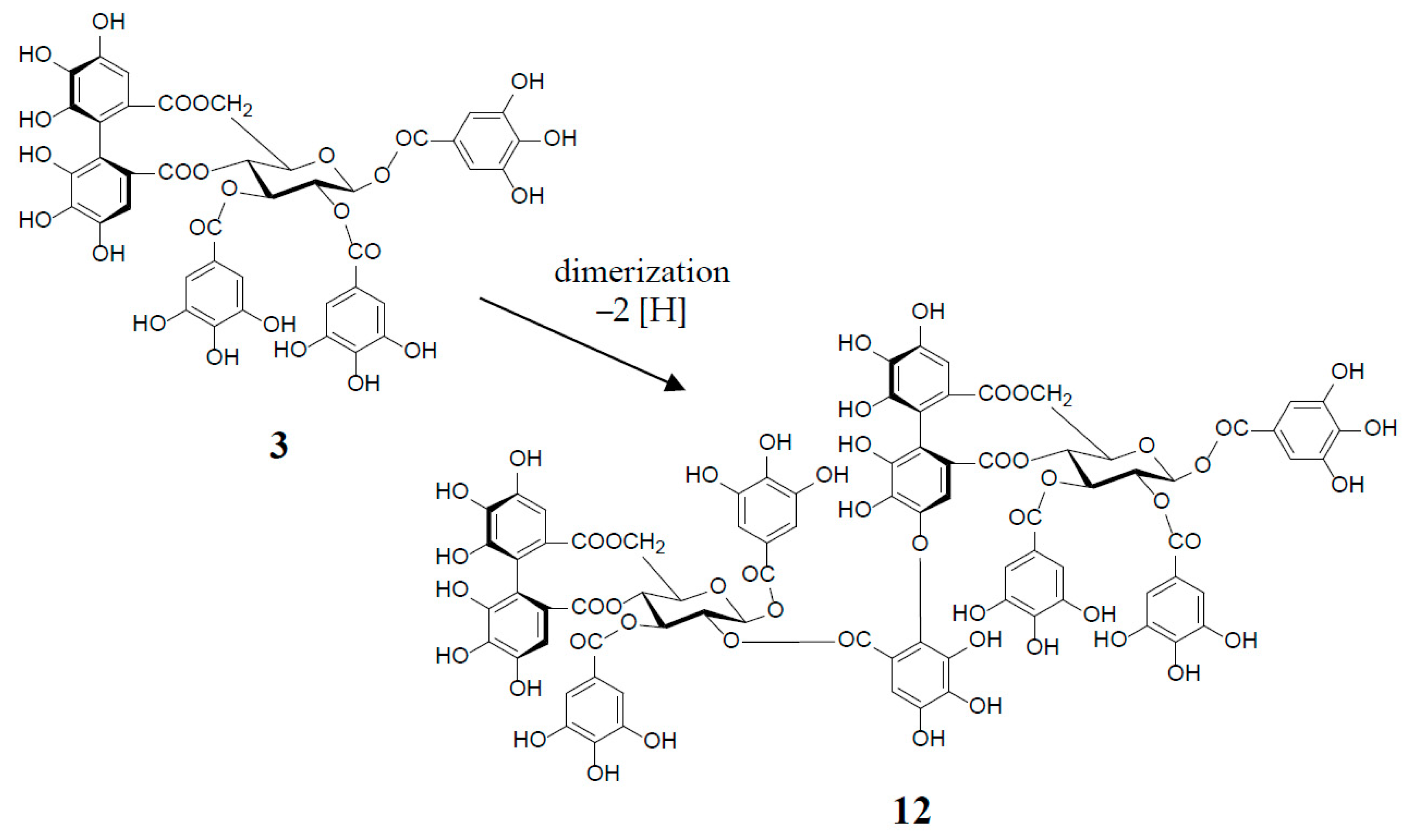
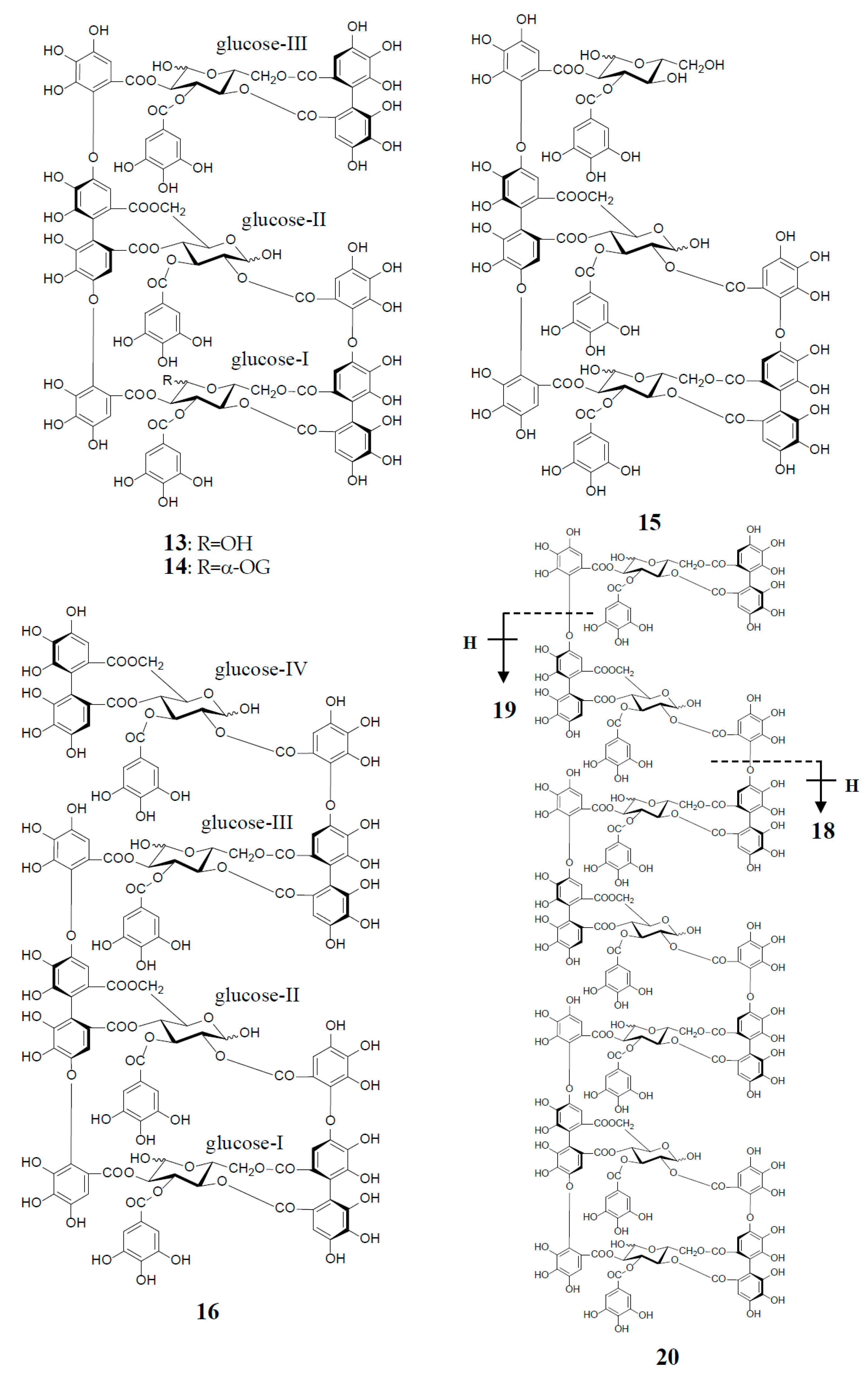
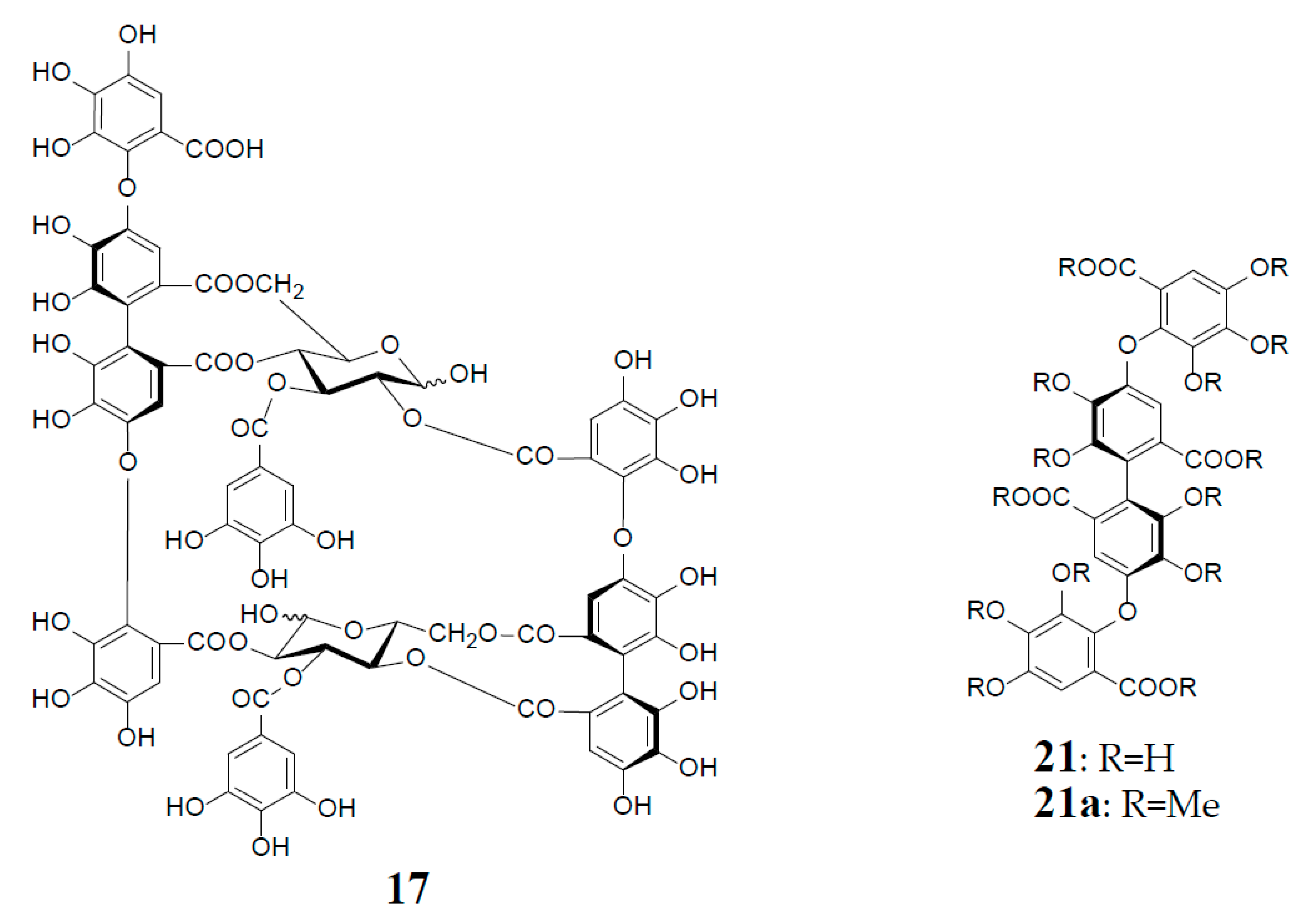
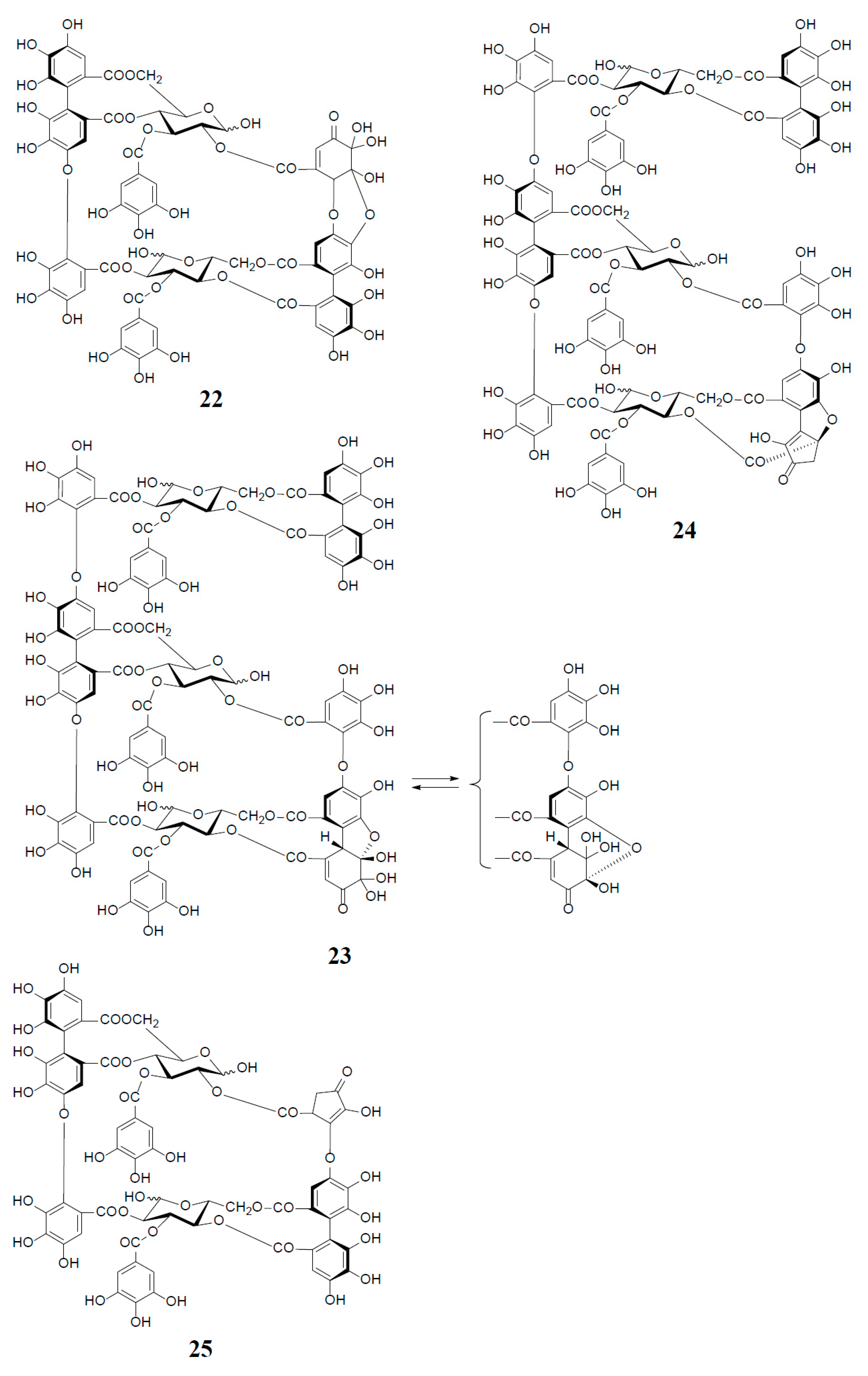
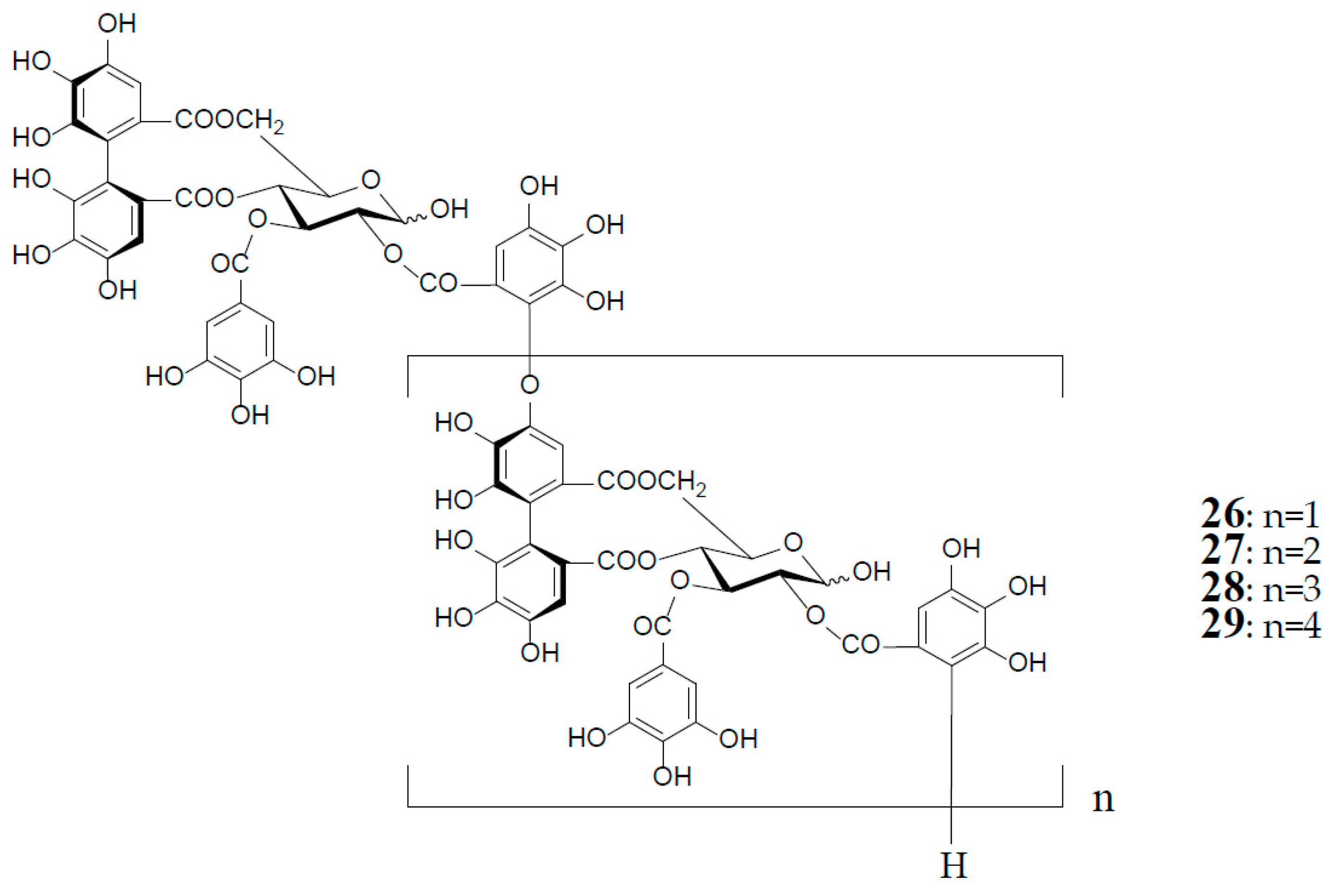
3. Oxidized Metabolites (Dimers and Oligomers) of Oenothein B
4. Distribution of Oenothein B and Its Analogs
5. Biological Activities of Oenothein B and Related Oligomers
5.1. Antioxidant and Anti-Inflammation Activity
5.2. Antitumor Effect
5.2.1. Cytotoxicity against Tumor Cell Lines
5.2.2. Antitumor Effect Caused by Tumor-Related Enzyme Inhibition
5.2.3. Host Mediated Antitumor Activity
5.3. Immunomodulatory Effect
5.4. Antimicrobial Effects
6. Conclusions
Author Contributions
Conflicts of Interest
References
- Haslam, E. The metabolism of gallic and hexahydroxydiphenic acid in higher plant. Fortschr. Chem. Org. Naturstoffe 1982, 41, 1–46. [Google Scholar]
- Okuda, T.; Yoshida, T.; Hatano, T. Geraniin, a new ellagitannin from Geranium thunbergii. Tetrahedron Lett. 1976, 17, 3721–3724. [Google Scholar] [CrossRef]
- Okuda, T.; Yoshida, T.; Hatano, T. Constituents of Geranium thunbergii Sieb. et Zucc. Part 12. Hydrated stereostructure and equilibration of geraniin. J. Chem. Soc. Perkin Trans. 1 1982, 9–14. [Google Scholar] [CrossRef]
- Okuda, T.; Yoshida, T.; Hatano, T. Hydrolyzable tannins and related polyphenols. Prog. Chem. Organ. Nat. Prod. 1995, 66, 1–117. [Google Scholar]
- Yoshida, T.; Amakura, Y.; Yoshimura, M. Structural Features and Biological Properties of Ellagitannins in the Myrtales. Int. J. Mol. Sci. 2010, 11, 79–106. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Hatano, T.; Ito, H.; Okuda, T. Highly Oxidized Ellagitannins and Their Biological Activity. In Plant Polyphenols 2; Gross, G.G., Hemingway, R.W., Yoshida., T., Eds.; Kluwer/Plenum: New York, NY, USA, 1999; pp. 127–144. [Google Scholar]
- Okuda, T.; Yoshida, T.; Hatano, T.; Ito, H. Ellagitannins Renewed the Concept of Tannins. In Chemistry and Biology of Ellagitannins; Quideau, S., Ed.; World Scientific: Hackensack, NJ, USA, 2009; pp. 1–54. [Google Scholar]
- Okuda, T.; Yoshida, T.; Kuwahara, M.; Memon, M.; Shingu, T. Agrimoniin and potentillin, ellagitannin dimer and monomer having α-glucose cores. J. Chem. Commun. 1982, 163–164. [Google Scholar] [CrossRef]
- Hatano, T.; Yasuhara, T.; Matsuda, M.; Yazaki, K.; Yoshida, T.; Okuda, T. Oenothein B, a Dimeric, hydrolysable tannin with macrocyclic structure, and accompanying tannins from Oenothera erythrosepala. J. Chem. Soc. Perkin Trans. 1 1990, 2735–2743. [Google Scholar] [CrossRef]
- Ducrey, B.; Marston, A.; Göhring, S.; Hartmann, R.W.; Hostettmann, K. Inhibition of 5α-reductase and aromatase by the ellagitannins oenothein A and oenothein B from Epilobium species. Planta Med. 1997, 63, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Tóth, B.H.; Blazics, B.; Kéry, A. Polyphenol composition and antioxidant capacity of Epilobium species. J. Pharm. Biomed. Anal. 2009, 49, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Chou, T.; Shingu, T.; Okuda, T. Oenotheins D, F and G, Hydrolysable Tannin Dimers from Oenothera laciniata. Phytochemistry 1995, 40, 555–561. [Google Scholar] [CrossRef]
- Yoshida, T.; Chou, T.; Nitta, A.; Miyamoto, K.; Koshiura, R.; Okuda, T. Woodfordin C, a macro-ring hydrolyzable tannin dimer with antitumor activity, and accompanying dimers from Woodfordia fruticosa flowers. Chem. Pharm. Bull. 1990, 38, 1211–1217. [Google Scholar] [CrossRef] [PubMed]
- Kadota, S.; Takamori, Y.; Nyein, K.N.; Kikuchi, T.; Tanaka, K.; Ekimoto, H. Constituents of the leaves of Woodfordia fruticosa Kurz. I. Isolation, structure, and proton and carbon-13 nuclear magnetic resonance signal assignments of woodfruticosin (woodfordin C), an inhibitor of deoxyribonucleic acid topoisomerase II. Chem. Pharm. Bull. 1990, 38, 2687–2697. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Nishimoto, S.; Yang, L.L.; Yen, K.Y.; Hatano, T.; Yoshida, T.; Okuda, T. Two macrocyclic hydrolysable tannin dimers from Eugenia uniflora. Phytochemistry 1997, 44, 1343–1349. [Google Scholar]
- Chen, L.G.; Yen, K.Y.; Yang, L.L.; Hatano, T.; Okuda, T.; Yoshida, T. Macrocyclic ellagitannin dimers, cuphiins D1 and D2, and accompanying tannins from Cuphea hyssopifolia. Phytochemistry 1999, 50, 307–312. [Google Scholar] [CrossRef]
- Haddock, E.A.; Gupta, R.K.; Al-Shafi, S.M.K.; Layden, K.; Haslam, E.; Magnolato, D. The Metabolism of gallic acid and hexahydroxydiphenic acid in plants; biogenetic and molecular taxonomic considerations. Phytochemistry 1982, 21, 1049–1062. [Google Scholar] [CrossRef]
- Niemetz, R.; Schilling, G.; Gross, G.G. Ellagitannin Biosynthesis: Oxidation of pentagalloylglucose to tellimagrandin II by an emzyme from Tellima grandiflora leaves. Chem. Commun. 2001, 1, 35–36. [Google Scholar] [CrossRef]
- Niemetz, R.; Schilling, G.; Gross, G.G. Biosynthesis of dimeric ellagitannin, cornusiin E, in Tellima grandiflora. Phytochemistry 2003, 64, 109–114. [Google Scholar] [CrossRef]
- Yoshida, T.; Chou, T.; Matsuda, M.; Yasuhara, T.; Yazaki, K.; Hatano, T.; Nitta, A.; Okuda, T. Woodfordin D and oenothein A, trimeric hydrolyzable tannins of macro-ring structure with antitumor activity. Chem. Pharm. Bull. 1991, 39, 1157–1162. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Chou, T.; Nitta, A.; Okuda, T. Tannins and related polyphenols of lythraceous plants. III. Hydrolyzable tannin oligomers with macrocyclic structure and accompanying tannins from Woodfordia fruticosa KURTZ. Chem. Pharm. Bull. 1992, 40, 2023–2030. [Google Scholar] [CrossRef]
- Baert, N.; Karonen, M.; Salminen, J.P. Isolation, characterization and quantification of the main oligomeric macrocyclic ellagitannins in Epilobium angustifolium by ultra-high performance chromatography with diode array detection and electrospray tandem mass spectrometry. J. Chromatogr. A 2015, 1419, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Baert, N.; Kim, J.; Karonen, M.; Salminen, J.P. Inter-population and inter-organ distribution of the main polyphenolic compounds of Epilobium angustifolium. Phytochemistry 2017, 134, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.J.; Wang, Z.; Li, H.X.; Wu, R.C.; Liu, Y.Z.; Wu, Q.Y. Mitochondrial dysfunction as an early event in the process of apoptosis induced by woodfordin I in human leukemia K562 cells. Toxicol. Appl. Pharmacol. 2004, 194, 141–155. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, S.; Imayoshi, Y.; Yabu-uchi, R.; Ito, H.; Hatano, T.; Yoshida, T. A macrocyclic ellagitannin trimer, oenotherin T1, from Oenothera species. Phytochemistry 2002, 59, 191–195. [Google Scholar] [CrossRef]
- Taniguchi, S.; Imayoshi, Y.; Yoshida, T.; Hatano, T. A new trimeric hydrolysable tannin, oenotherin T2, isolated from aerial parts of Oenothera tetraptera Cav. Heterocyles 2009, 79, 617–626. [Google Scholar]
- Yoshida, T.; Hatano, T.; Ito, H. Chemistry and function of vegetable polyphenols with high molecular weights. BioFactors 2000, 13, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Amakura, Y.; Tokuhara, M.; Yoshida, T. Polyphenolic compounds isolated from the leaves of Myrtus communis. J. Nat. Med. 2008, 62, 366–368. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, S.; Nakamura, N.; Nose, M.; Takeda, S.; Yabu-uchi, R.; Ito, H.; Yoshida, T.; Yazaki, K. Production of macrocyclic ellagitannin oligomers by Oenothera laciniata callus cultures. Phytochemistry 1998, 48, 981–985. [Google Scholar] [CrossRef]
- Amakura, Y.; Yoshimura, M.; Sugimoto, N.; Yamazaki, T.; Yoshida, T. Marker constituents of the natural antioxidant Eucalyptus leaf extract for the evaluation of food additives. Biosci. Biotechnol. Biochem. 2009, 73, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Okuda, T.; Yoshida, T.; Hatano, T.; Yazaki, K.; Kira, R.; Ikeda, Y. Chromatography of tannins II: Preparative fractionation of hydrolyzable tannins by centrifugal partition chromatography. J. Chromatogr. A 1986, 362, 375–381. [Google Scholar] [CrossRef]
- Santos, S.C.; Waterman, P.G. Polyphenols from Eucalyptus consideniana and Eucalyptus viminalis. Fitoterapia 2001, 72, 95–97. [Google Scholar] [CrossRef]
- Yoshida, T.; Maruyama, T.; Nitta, A.; Okuda, T. Eucalbanins A, B and C, monomeric and dimeric hydrolyzable tannins from Eucalyptus alba REINW. Chem. Pharm. Bull. 1992, 40, 1750–1754. [Google Scholar] [CrossRef]
- Ito, H.; Li, P.; Koreishi, M.; Nagatomo, A.; Nishida, N.; Yoshida, T. Ellagitannin oligomers and a neolignan from pomegranate arils and their inhibitory effects on the formation of advanced glycation end products. Food Chem. 2014, 152, 323–330. [Google Scholar] [CrossRef] [PubMed]
- The Angiosperm Phylogeny Group. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants. APG III. Bot. J. Linn. Soc. 2009, 161, 105–121. [Google Scholar]
- Graham, S.A.; Hall, J.; Sytsma, K.; Shi, S. Phylogenetic analysis of the Lythraceae based on four gene regions and morphology. Int. J. Plant Sci. 2005, 166, 995–1017. [Google Scholar] [CrossRef]
- Granica, S.; Czerwińska, M.E.; Piwowarski, J.P.; Ziaja, M.; Kiss, A.K. Chemical composition, antioxidative and anti-inflammatory activity of extracts prepared from aerial parts of Oenothera biennis L. and Oenothera paradoxa Hudziok obtained after seeds cultivation. J. Agric. Food Chem. 2013, 30, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Kiss, A.K.; Kapłon-Cieślicka, A.; Filipiak, K.J.; Opolski, G.; Naruszewicz, M. Ex vivo effects of an Oenothera paradoxa extract on the reactive oxygen species generation and neutral endopeptidase activity in neutrophils from patients after acute myocardial infarction. Phytother. Res. 2012, 26, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Granica, S.; Piwowarski, J.P.; Czerwińska, E.M.; Kiss, A.K. Phytochemistry, pharmacology and traditional uses of different Epilobium species (Onagraceae): A review. J. Ethnopharmacol. 2014, 156, 316–346. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.H.; Chiou, J.F.; Yen, K.Y.; Yang, L.L. EBV DNA polymerase inhibition of 4 tannins from Eugenia uniflora. Cancer Lett. 2000, 54, 131–136. [Google Scholar] [CrossRef]
- Yoshida, T.; Hatano, T.; Ito, H.; Okuda, T. Structural diversity and antimicrobial activities of ellagitannins. In Chemistry and Biology of Ellagitannins; Quideau, S., Ed.; World Scientific: Hackensack, NJ, USA, 2009; pp. 55–93. [Google Scholar]
- Dobreva, M.A.; Green, R.J.; Muller-Harvey, I.; Salminen, J.P.; Howlin, B.J.; Frazier, R.A. Size and molecular flexibility affect the binding of ellagitannins to bovine serum albumin. J. Agric. Food Chem. 2014, 62, 9186–9194. [Google Scholar] [CrossRef] [PubMed]
- Schepetkin, I.A.; Ramstead, A.G.; Kirpotina, L.N.; Voyich, J.M.; Jutila, M.A.; Quinn, M.T. Therapeutic potential of polyphenols from Epilobium angustifolium (Fireweed). Phytother. Res. 2016, 30, 1287–1297. [Google Scholar] [CrossRef] [PubMed]
- Jassen, Y.M.V.; Van Houten, B.; Borm, P.J.A.; Mossman, B.T. Cell and tissue responses to oxidative damage. Lab. Investig. 1993, 69, 261–274. [Google Scholar]
- Haslam, E. Natural polyphenols (vegetable tannins) as drugs: possible models of action. J. Nat. Prod. 1996, 59, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Okuda, T.; Yoshida, T.; Hatano, T. Pharmacologically active tannins isolated from medicinal plants. In Plant Polyphenols; Hemingway, R.W., Lacks, P.E., Eds.; Plenum Press: New York, NY, USA, 1992; pp. 530–569. [Google Scholar]
- Fujita, Y.; Komagoe, K.; Sasaki, Y.; Uehara, I.; Okuda, T.; Yoshida, T. Inhibition mechanism of tannins on Cu(II)-catalyzed autoxidation of ascorbic acid. Yakugaku Zasshi 1987, 107, 17–22. [Google Scholar] [CrossRef]
- Yoshida, T.; Mori, K.; Hatano, T.; Okumura, T.; Uehara, I.; Komagoe, K.; Fujita, Y.; Okuda, T. Radical-scavenging effects of tannins and related polyphenols on 1,1-diphenyl-2-picrylhydrazyl radical. Chem. Pharm. Bull. 1989, 37, 1919–1921. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Riedl, K.M.; Rice, R.E. Tannins as biological antioxidants. In Plant Polyphenols 2; Gross, G.G., Hemingway, R.W., Yoshida, T., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 1999; pp. 495–505. [Google Scholar]
- Hagerman, A.E.; Riedl, K.M.; Jones, G.A.; .Sovik, K.N.; Ritchard, N.T.; Hartzfeld, P.W.; Riechel, T.L. High molecular weight plant polyphenolics (Tannins) as biological antioxidants. J. Agric. Food Chem. 1998, 46, 1887–1892. [Google Scholar] [CrossRef] [PubMed]
- Kiss, A.K.; Bazylko, A.; Filipek, A.; Granica, S.; Jaszewska, E.; Kiarszys, U.; Kośmider, A.; Piwowarski, J.P. Oenothein B’s contribution to the anti-inflammatory and antioxidant activity of Epilobium sp. Phytomedicine 2011, 18, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.; Chen, L.G.; Yang, L.L. Antitumor activity of four macrocyclic ellagitannins from Cuphea hyssopifolia. Cancer Lett. 1999, 140, 195–200. [Google Scholar] [CrossRef]
- Wang, C.C.; Chen, L.G.; Yang, L.L. Cytotoxic effects of cuphiin D1 on the growth of human cervical carcinoma and normal cells. Anticancer Res. 2002, 22, 2677–2684. [Google Scholar] [PubMed]
- Sakagami, H.; Jiang, Y.; Kusama, K.; Atsumi, T.; Ueha, T.; Toguchi, M.; Iwakura, I.; Satoh, K.; Ito, H.; Hatano, T.; et al. Cytotoxic activity of hydrolyzable tannins against human oral tumor cell lines---A possible mechanism. Phytomedicine 2000, 7, 39–47. [Google Scholar] [CrossRef]
- Kiss, A.; Kowalski, J.; Melzig, M.F. Induction of neutral endopeptidase activity in PC-3 cell by aqueous extract of Epilobium angustifolium and Oenothein B. Phytomedicine 2006, 13, 284–289. [Google Scholar] [CrossRef] [PubMed]
- Piwowarski, J.P.; Bobrowska-Korczak, B.; Stanislawska, I.; Bielecki, W.; Wrzesien, R.; Granica, S.; Krupa, K.; Kiss, A.N. Evaluation of the effect of Eepilobium angustifolium aqueous extract on LNCap cell proliferation in in vitro and in vivo medels. Planta Med. 2017, 83, 1159–1168. [Google Scholar] [PubMed]
- Piwowarski, J.P.; Granica, S.; Stefarska, J.; Kiss, A.K. Differences in metabolism of ellagitannins by human gut microbiota ex vivo cultures. J. Nat. Prod. 2016, 79, 3022–3030. [Google Scholar] [CrossRef] [PubMed]
- Kuramochi-Motegi, A.; Kuramochi, H.; Kobayashi, F.; Ekimoto, H.; Takahashi, K.; Kadota, S.; Takamori, Y.; Kikuchi, T. Woodfruticosin (woodfordin C), a new inhibitor of DNA topoisomerase II. Experimental antitumor activity. Biochem. Pharmacol. 1992, 44, 1961–1965. [Google Scholar] [PubMed]
- Tanuma, S.; Johnson, L.D.; Johnson, G.S. ADP-ribosylation of chromosomal proteins and mouse mammary tumor virus gene expression. J. Biol. Chem. 1983, 258, 15371–15375. [Google Scholar] [PubMed]
- Aoki, K.; Nishimura, K.; Abe, H.; Maruta, H.; Sakagame, H.; Hatano, T.; Okuda, T.; Yoshida, T.; Tsai, Y.J.; Uchiumi, F.; et al. A novel inhibitors of poly(ADP-ribose) glycohydrolase. Biochim. Biophys. Acta 1993, 1158, 251–256. [Google Scholar] [CrossRef]
- Yoshida, T.; Haba, K.; Arata, R.; Nakata, F.; Shingu, T.; Okuda, T. Tannins and related polyphenols of Melastomataceous plants. VII. Nobotanins J and K, trimeric and tetrameric hydrolyzable tanins from Heterocentron roseum. Chem. Pharm. Bull. 1995, 43, 1101–1106. [Google Scholar] [CrossRef]
- Aoki, K.; Maruta, H.; Uchiumi, F.; Hatano, T.; Yoshida, T.; Tanuma, S. A macrocircular ellagitannin, oenothein B, suppresses mouse mammary tumor gene expressionvia inhibition of poly(ADP-ribose) glycohydrolase. Biochim. Biophys. Res. Commun. 1995, 210, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Hanai, S.; Kanai, M.; Ohashi, S.; Okamoto, K.; Yamada, M.; Takahashi, H.; Miwa, M. Loss of poly(ADP-ribose) glycohydrolase causes progressive neurodegeneration in Drosophila melanogaster. Proc. Natl. Acad. Sci. USA 2004, 101, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Koh, D.W. Roles of poly(ADP-ribose) glycohydrolase in DNA damage and apoptosis. Int. Rev. Cell Mol. Biol. 2013, 304, 227–281. [Google Scholar] [PubMed]
- Miyamoto, K.; Kishi, N.; Koshiura, R.; Yoshida, T.; Hatano, T.; Okuda, T. Relationship between the structures and the antitumor activities of tannins. Chem. Pharm. Bull. 1987, 35, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, K.; Nomura, M.; Sasakura, M.; Matsui, E.; Koshiura, R.; Murayama, T.; Furukawa, T.; Hatano, T.; Yoshida, T.; Okuda, T. Antitumor activity of oenothein B, a unique macrocyclic ellagitannin. Jpn. J. Cancer Res. 1993, 84, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Murayama, T.; Kishi, N.; Koshiura, R.; Takagi, K.; Furukawa, T.; Miyamoto, K. Agrimoniin, an antitumor tannin of Agrimonia pilosa Ledeb., induces interleukin-1. Anticancer Res. 1992, 12, 1471–1474. [Google Scholar] [PubMed]
- Murayama, T.; Natsuume-Sakai, S.; Ryoyama, K.; Koshiura, S. Studies on the properties of a streptococcal preparation, OK-432 (NSC-B116209), as an immunopotentiator. II. Mechanism of macrophage activation by OK-432. Cancer Immunol. Immunother. 1982, 12, 141–146. [Google Scholar] [CrossRef]
- Miyamoto, K.; Murayama, T.; Nomura, M.; Hatano, T.; Yoshida, T.; Furukawa, T.; Koshiura, R.; Okuda, T. Antitumor activity and interleukin-1 induction by tannins. Anticancer Res. 1993, 13, 37–42. [Google Scholar] [PubMed]
- Wang, C.C.; Chen, L.G.; Yang, L.L. in vitro immunomodulatory effects of cuphiin D1 on human mononuclear cells. Anticancer Res. 2002, 22, 4233–42336. [Google Scholar] [PubMed]
- Schepetkin, I.A.; Kirpotina, L.N.; Jarissa, L.; Khlebnikov, A.I.; Blaskovich, C.L.; Jutila, M.A.; Quinn, M.T. Immunomodulatory activity of oenothein B isolated from Epilobium angustifolium. J. Immun. 2009, 183, 6754–6766. [Google Scholar] [CrossRef] [PubMed]
- Schmid, D.; Gruber, M.; Piskaty, C.; Woehs, F.; Renner, A.; Nagy, Z.; Kaltenboeck, A.; Wasserscheid, T.; Bazylko, A.; Kiss, A.K. Inhibition of NF-κB-dependent cytokine and inducible nitric oxide synthesis by the macrocyclic ellagtannin oenothein B in TLR-stimulated RAW 264.7 macrophages. J. Nat. Prod. 2012, 75, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yang, L.L.; Lee, T.J. Oroxylin A inhibition of lipopolysaccharide-induced iNOS and COX-2 gene expression via suppression of nuclear factor-kappaB activation. Biochem. Pharmacol. 2000, 59, 1445–1457. [Google Scholar] [CrossRef]
- Ramstead, A.G.; Schepetkin, I.A.; Quinn, M.T.; Jutila, M.A. Oenothein B, a cyclic dimeric ellagitannin isolated from Epilobium angustifolium, enhances IFNγ production by lymphocytes. PLoS ONE 2012, 7, e50546. [Google Scholar] [CrossRef] [PubMed]
- Ramstead, A.G.; Schepetkin, I.A.; Todd, K.; Loeffelholz, J.; Berardinelli, J.G.; Quinn, M.T.; Jutila, M.A. Aging influences the response of T cells to stimulation by the ellagitannin, oenothein B. Int. Immunopharmacol. 2015, 26, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, M.; Akiyama, H.; Kondo, K.; Sakata, K.; Matsuoka, H.; Amakura, Y.; Teshima, R.; Yoshida, T. Immunological effects of oenothein B, an ellagitannin dimer, on dendritic cells. Int. J. Mol. Sci. 2013, 14, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.W.; Wang, H.; Poitras, M.F.; Coombs, C.; Bowers, W.J.; Federoff, H.J.; Poirier, G.G.; Dawson, T.M.; Dawson, V.L. Mediation of poly(ADP-ribose) polymerase-1-dependent cell death by apoptosis-inducing factor. Science 2002, 297, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Rios, L.Y.; Bennett, R.N.; Lazarus, S.A.; Rémésy, C.; Scalbert, A.; Williamson, G. Cocoa procyanidins are stable during gastric transit in humans. Am. J. Clin. Nutr. 2002, 76, 1106–1110. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, S.; Makihata, N.; Yoshimura, M.; Amakura, Y.; Yoshida, T.; Nakajima, M.; Furukawa, Y. Oenothein B suppresses lipopolysaccharide (LPS)-induced inflammation in the mouse brain. Int. J. Mol. Sci. 2013, 14, 9767–9778. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, J.C.; Lawson, M.A.; André, C.; Moreau, M.; Lestage, J.; Castanon, N.; Kelley, K.W.; Dantzer, R. Lipopolysaccharide-induced depressive-like behavior is mediated by indoleamine 2,3-dioxygenase activation in mice. Mol. Psychiatry 2009, 14, 511–522. [Google Scholar] [CrossRef] [PubMed]
- Ito, H.; Iguchi, A.; Hatano, T. Identification of urinary and intestinal bacterial metabolites of ellagitannin geraniin in rats. J. Agric. Food Chem. 2008, 56, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Bastida, J.A.; Larrosa, M.; González-Sarrías, A.; Tomás-Barberán, F.; Espín, J.C.; García-Conesa, M.T. Intestinal ellagitannin metabolites ameliorate cytokine-induced inflammation and associated molecular markers in human colon fibroblasts. J. Agric. Food Chem. 2012, 60, 8866–8876. [Google Scholar] [CrossRef] [PubMed]
- Kolodziej, H.; Kayser, O.; Latte, K.P.; Kiderlen, A.F. Enhancement of antimicrobial activity of tannins and related compounds by immune modulatory effects. In Plant Polyphenols 2; Gross, G.G., Hemingway, R.W., Yoshida, T., Eds.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 1999; pp. 575–594. [Google Scholar]
- Kolodziej, H.; Kayser, O.; Latte, K.P.; Kiderlen, A.F.; Ito, H.; Hatano, T.; Yoshida, T.; Foo, L.Y. Antileishmanial activity of hydrolysable tannins and their modulatory effects on nitric oxide and tumor necrosis factor-a release in macrophages in vitro. Planta Med. 2001, 67, 825–832. [Google Scholar] [CrossRef] [PubMed]
- Hatano, T.; Tsugawa, M.; Ohyabu, T.; Kusuda, M.; Shiota, S.; Tsuchiya, T.; Yoshida, T. Effects of polyphenols in tea and foods on methicillin-resistant Staphylococcus aureus and the sustainability of the antibacterial effects in the presence of food additives. Assoc. J. Jpn. Soc. Med. Use Funct. Foods 2006, 4, 43–48. [Google Scholar]
- Ishimoto, H.; Shibata, M.; Myojin, Y.; Ito, H.; Sugimoto, Y.; Tai, A.; Hatano, T. in vivo anti-inflammatory and antioxidant properties of ellagitannin metabolite urolithin A. Bioorg. Med. Chem. Lett. 2011, 21, 5901–5904. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Barberan, F.A.; Espin, J.C.; Garcia-Conesa, M.T. Bioavailability and metabolism of ellagic acid and ellagitannins. In Chemisty and Biology of Ellagitannins; Quideau, S., Ed.; World Scientific: Hackensack, NJ, USA, 2009; pp. 273–297. [Google Scholar]
- Torronen, R. Sources and health effects of dietry ellagitannins. In Chemisty and Biology of Ellagitannins; Quideau, S., Ed.; World Scientific: Hackensack, NJ, USA, 2009; pp. 298–319. [Google Scholar]
- Lan, Y.; Wang, L.; Cao, S.; Zhong, Y.; Li, Y.; Cao, Y.; Zhao, L. Rational design of food-grade polyelectrolyte complex coacervate for encapsulation and enhanced oral delivery of oenothein B. Food Funct. 2017, 8, 4070–4080. [Google Scholar] [CrossRef] [PubMed]
| Family Species | Tannins | Ref. |
|---|---|---|
| Oenotheraceae | ||
| Oenothera erythrosepala Bordas | oenothein B | [9] |
| O. biennis L. | oenotheins A, B | [20,37,38] |
| O. laciniata Hill. | oenotheins A, B, D, F, G | [12] |
| O. tetraptera Cav. | oenotheins A, B, oenotherins T1, T2 | [25,26] |
| O. paradoxa Hudziok | oenothein B | [38] |
| Epilobium capense Buch. | oenotheins A, B | [10,11] |
| E. angustifolium L. | oenotheins A, B, woodfordin I, tetramer–heptamer | [10,11,22,24,39] |
| E. pyrricholophum Franch. et Sav. | oenotheins B | [39] |
| E. hirsutum L. | oenotheins B | [10,39] |
| E. palustre L. | oenotheins A, B | [39] |
| E. dodonoei Vill. | oenothein B (HPLC) * | [10] |
| E. stereophyllum Fres. | oenothein B (HPLC) | [10] |
| E. salignum Hausskn. | oenothein B (HPLC) | [10] |
| E. parviflorum Schreb. | oenothein B (HPLC) | [10] |
| E. roseum Schreb. | oenothein B (HPLC), (LC/MS) * | [10,11] |
| E. tetragonum L. | oenothein B (LC/MS) | [11] |
| E. montanum L. | oenothein B (HPLC), (LC/MS) | [10,11] |
| Lythraceae | ||
| Lythrum anceps Makino | oenothein B | [31] |
| Woodfordia fruticosa Kurz. | oenotheins A, B, woodfordins C, D, E, F, I | [13,14,20,21] |
| Cuphea hyssopifolia Humb. | oenotheins A, B, woodfordin C, cuphiins D1, D2 | [16] |
| Punica granatum L. | oenothein B | [34] |
| Myrtaceae | ||
| Eugenia uniflora L. | oenothein B, eugeniflorins D1, D2 | [15,40] |
| Melaleuca leucadendron L. | oenothein B | [5] |
| Myrtus communis L. | oenothein B, eugeniflorin D2 | [28] |
| Eucalyptus alba Reinw. Ex Blume | oenothein B | [33] |
| E. robusta Sm. | oenothein B, eugeniflorin D2, eurobustin C | [27] |
| E. cypellocarpa LAS Johnson | oenothein B, eugeniflorin D2 | [27] |
| E. globulus Labill. | oenothein B | [30] |
| E. consideniana Maiden | oenothein B | [32] |
| E. viminalis Labill. | oenothein B | [32] |
| E. pulverulenta Sims. | oenothein B (HPLC) * | [30] |
| E. nicholii Box Hill. Merbourne | oenothein B (HPLC) | [30] |
| E. camaldulensis Dehnh. | oenothein B (HPLC) | [30] |
| Myrtus communis var. microphylla Willk. | oenothein B (HPLC) | [30] |
| Austromyrtus dulcis L.S. Sm. | oenothein B (HPLC) | [30] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshida, T.; Yoshimura, M.; Amakura, Y. Chemical and Biological Significance of Oenothein B and Related Ellagitannin Oligomers with Macrocyclic Structure. Molecules 2018, 23, 552. https://doi.org/10.3390/molecules23030552
Yoshida T, Yoshimura M, Amakura Y. Chemical and Biological Significance of Oenothein B and Related Ellagitannin Oligomers with Macrocyclic Structure. Molecules. 2018; 23(3):552. https://doi.org/10.3390/molecules23030552
Chicago/Turabian StyleYoshida, Takashi, Morio Yoshimura, and Yoshiaki Amakura. 2018. "Chemical and Biological Significance of Oenothein B and Related Ellagitannin Oligomers with Macrocyclic Structure" Molecules 23, no. 3: 552. https://doi.org/10.3390/molecules23030552
APA StyleYoshida, T., Yoshimura, M., & Amakura, Y. (2018). Chemical and Biological Significance of Oenothein B and Related Ellagitannin Oligomers with Macrocyclic Structure. Molecules, 23(3), 552. https://doi.org/10.3390/molecules23030552




