PTEN Inhibition in Human Disease Therapy
Abstract
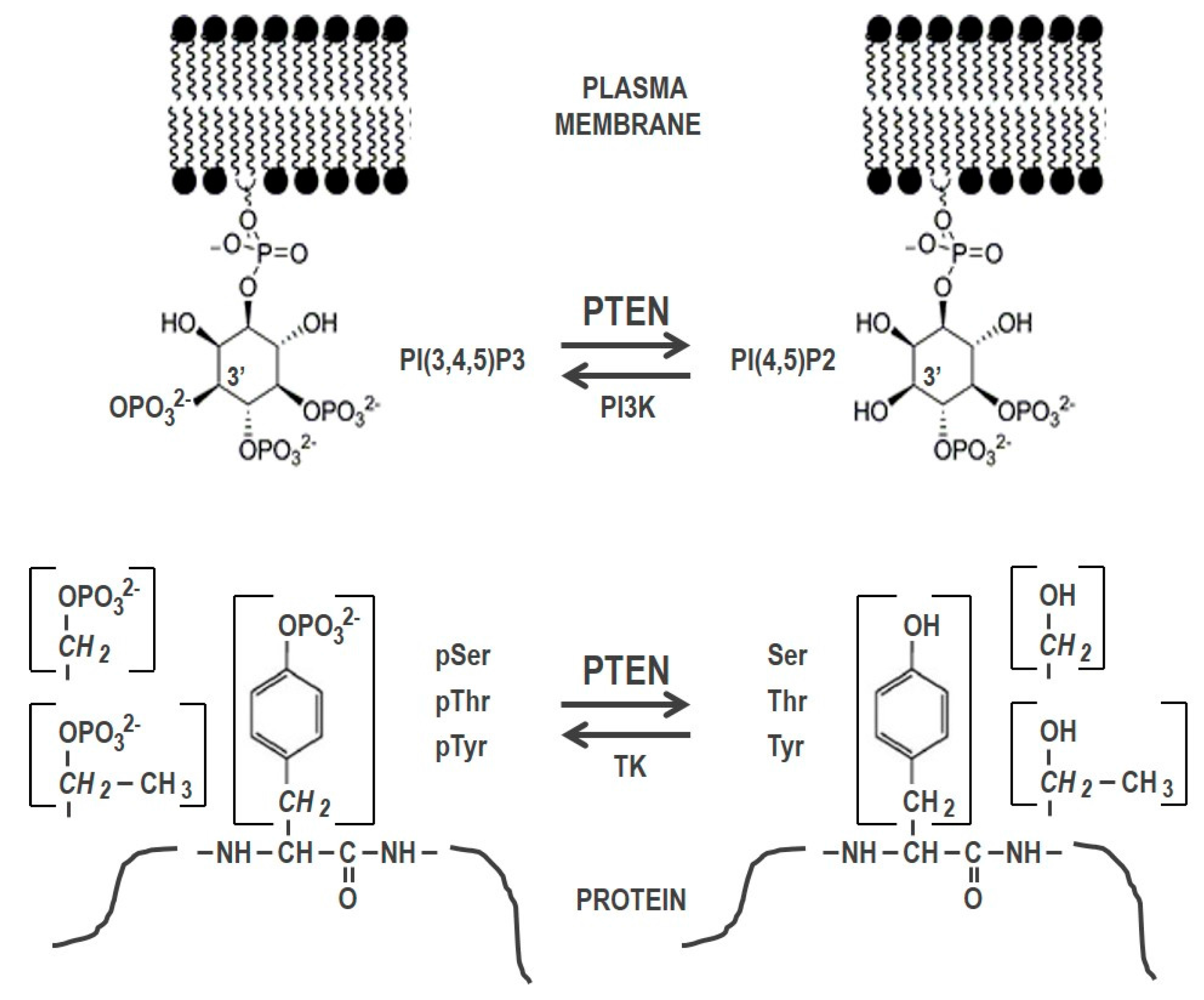
1. Introduction
2. Small Molecule PTEN Inhibitors
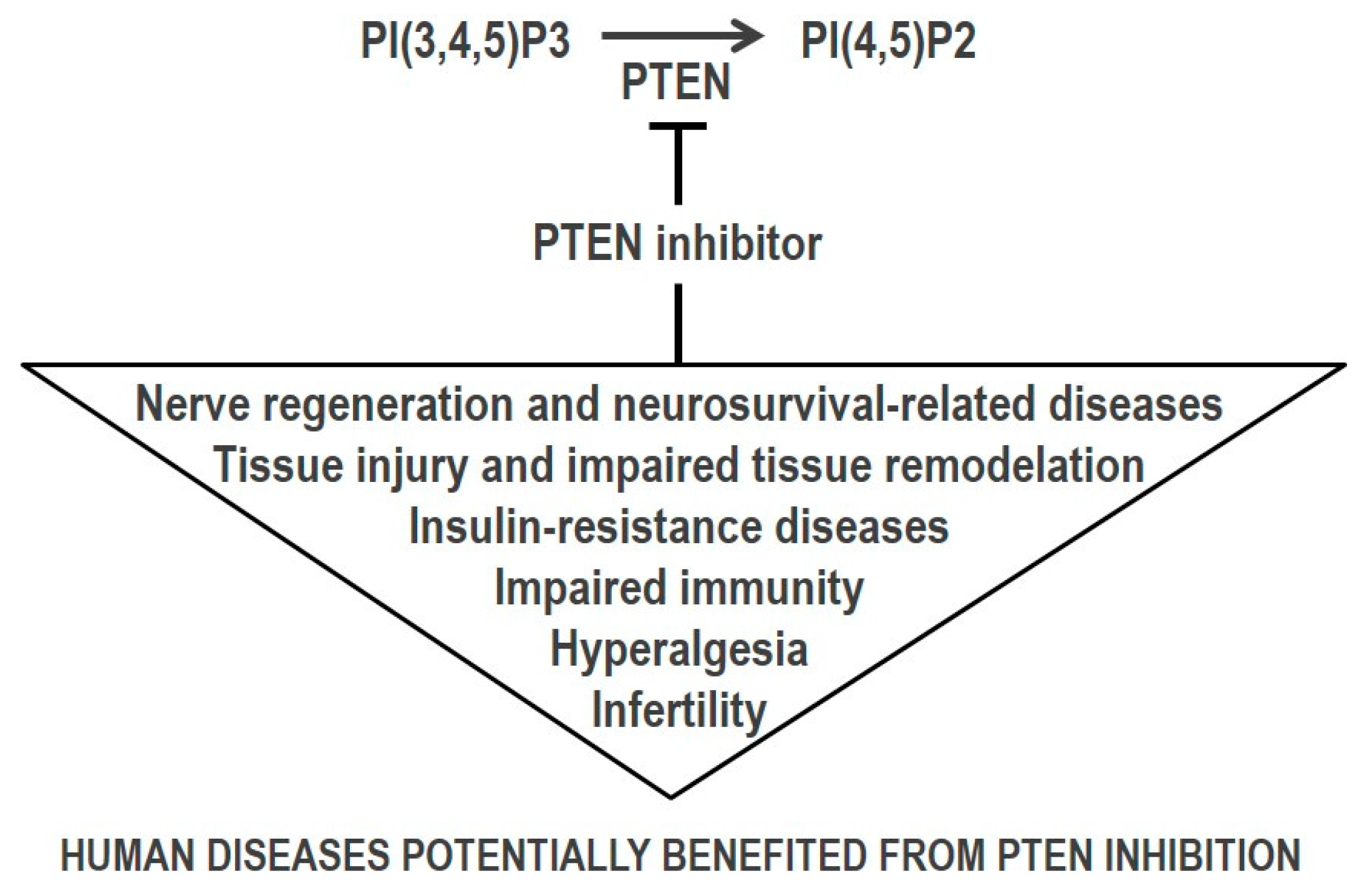
3. PTEN Inhibition by Small Molecules in Human Disease Therapy
3.1. Nerve Regeneration and Neurosurvival-Related Diseases
3.2. Ischemia/Reperfusion Tissue Injury
3.3. Wound Repair and Tissue Maintenance
3.4. Response to Infection
3.5. Infertility
3.6. Stem Cells-Based Therapy
3.7. Cancer-Related Diseases
3.8. Insulin-Resistance Metabolic Diseases
3.9. Pain Relief/Antinociception
4. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Pulido, R. PTEN: A yin-yang master regulator protein in health and disease. Methods 2015, 77–78, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Worby, C.A.; Dixon, J.E. PTEN. Annu. Rev. Biochem. 2014, 83, 641–669. [Google Scholar] [CrossRef] [PubMed]
- Malaney, P.; Uversky, V.N.; Dave, V. PTEN proteoforms in biology and disease. Cell. Mol. Life Sci. CMLS 2017, 74, 2783–2794. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.O.; Yang, H.; Georgescu, M.M.; Di Cristofano, A.; Maehama, T.; Shi, Y.; Dixon, J.E.; Pandolfi, P.; Pavletich, N.P. Crystal structure of the PTEN tumor suppressor: Implications for its phosphoinositide phosphatase activity and membrane association. Cell 1999, 99, 323–334. [Google Scholar] [CrossRef]
- Maehama, T.; Dixon, J.E. PTEN: A tumour suppressor that functions as a phospholipid phosphatase. Trends Cell Biol. 1999, 9, 125–128. [Google Scholar] [CrossRef]
- Malek, M.; Kielkowska, A.; Chessa, T.; Anderson, K.E.; Barneda, D.; Pir, P.; Nakanishi, H.; Eguchi, S.; Koizumi, A.; Sasaki, J.; et al. PTEN Regulates PI(3,4)P2 Signaling Downstream of Class I PI3K. Mol. Cell 2017, 68, 566–580. [Google Scholar] [CrossRef] [PubMed]
- Stambolic, V.; Suzuki, A.; de la Pompa, J.L.; Brothers, G.M.; Mirtsos, C.; Sasaki, T.; Ruland, J.; Penninger, J.M.; Siderovski, D.P.; Mak, T.W. Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell 1998, 95, 29–39. [Google Scholar] [CrossRef]
- Hobert, J.A.; Eng, C. PTEN hamartoma tumor syndrome: An overview. Genet. Med. 2009, 11, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Tilot, A.K.; Frazier, T.W., II; Eng, C. Balancing Proliferation and Connectivity in PTEN-associated Autism Spectrum Disorder. Neurotherapeutics 2015, 12, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Parada, L.F. PTEN signaling in autism spectrum disorders. Curr. Opin. Neurobiol. 2012, 22, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Caffrey, J.J.; Darden, T.; Wenk, M.R.; Shears, S.B. Expanding coincident signaling by PTEN through its inositol 1,3,4,5,6-pentakisphosphate 3-phosphatase activity. FEBS Lett. 2001, 499, 6–10. [Google Scholar] [CrossRef]
- Deleu, S.; Choi, K.; Pesesse, X.; Cho, J.; Sulis, M.L.; Parsons, R.; Shears, S.B. Physiological levels of PTEN control the size of the cellular Ins(1,3,4,5,6)P5 pool. Cell. Signal. 2006, 18, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Orchiston, E.A.; Bennett, D.; Leslie, N.R.; Clarke, R.G.; Winward, L.; Downes, C.P.; Safrany, S.T. PTEN M-CBR3, a versatile and selective regulator of inositol 1,3,4,5,6-pentakisphosphate (Ins(1,3,4,5,6)P5). Evidence for Ins(1,3,4,5,6)P5 as a proliferative signal. J. Biol. Chem. 2004, 279, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Tibarewal, P.; Zilidis, G.; Spinelli, L.; Schurch, N.; Maccario, H.; Gray, A.; Perera, N.M.; Davidson, L.; Barton, G.J.; Leslie, N.R. PTEN protein phosphatase activity correlates with control of gene expression and invasion, a tumor-suppressing phenotype, but not with AKT activity. Sci. Signal. 2012, 5, ra18. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.C.; Piccini, A.; Myers, M.P.; van Aelst, L.; Tonks, N.K. Functional analysis of the protein phosphatase activity of PTEN. Biochem. J. 2012, 444, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Andrés-Pons, A.; Gil, A.; Oliver, M.D.; Sotelo, N.S.; Pulido, R. Cytoplasmic p27Kip1 counteracts the pro-apoptotic function of the open conformation of PTEN by retention and destabilization of PTEN outside of the nucleus. Cell. Signal. 2012, 24, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Rahdar, M.; Inoue, T.; Meyer, T.; Zhang, J.; Vazquez, F.; Devreotes, P.N. A phosphorylation-dependent intramolecular interaction regulates the membrane association and activity of the tumor suppressor PTEN. Proc. Natl. Acad. Sci. USA 2009, 106, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Vazquez, F.; Grossman, S.R.; Takahashi, Y.; Rokas, M.V.; Nakamura, N.; Sellers, W.R. Phosphorylation of the PTEN tail acts as an inhibitory switch by preventing its recruitment into a protein complex. J. Biol. Chem. 2001, 276, 48627–48630. [Google Scholar] [CrossRef] [PubMed]
- Davidson, L.; Maccario, H.; Perera, N.M.; Yang, X.; Spinelli, L.; Tibarewal, P.; Glancy, B.; Gray, A.; Weijer, C.J.; Downes, C.P.; et al. Suppression of cellular proliferation and invasion by the concerted lipid and protein phosphatase activities of PTEN. Oncogene 2010, 29, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Luna, S.; Mingo, J.; Aurtenetxe, O.; Blanco, L.; Amo, L.; Schepens, J.; Hendriks, W.J.; Pulido, R. Tailor-Made Protein Tyrosine Phosphatases: In Vitro Site-Directed Mutagenesis of PTEN and PTPRZ-B. Methods Mol. Biol. 2016, 1447, 79–93. [Google Scholar] [PubMed]
- Myers, M.P.; Pass, I.; Batty, I.H.; Van der Kaay, J.; Stolarov, J.P.; Hemmings, B.A.; Wigler, M.H.; Downes, C.P.; Tonks, N.K. The lipid phosphatase activity of PTEN is critical for its tumor supressor function. Proc. Natl. Acad. Sci. USA 1998, 95, 13513–13518. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, B.D.; Hodakoski, C.; Barrows, D.; Mense, S.M.; Parsons, R.E. PTEN function: The long and the short of it. Trends Biochem. Sci. 2014, 39, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Alimonti, A.; Carracedo, A.; Clohessy, J.G.; Trotman, L.C.; Nardella, C.; Egia, A.; Salmena, L.; Sampieri, K.; Haveman, W.J.; Brogi, E.; et al. Subtle variations in Pten dose determine cancer susceptibility. Nat. Genet. 2010, 42, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Ngeow, J.; He, X.; Mester, J.L.; Lei, J.; Romigh, T.; Orloff, M.S.; Milas, M.; Eng, C. Utility of PTEN protein dosage in predicting for underlying germline PTEN mutations among patients presenting with thyroid cancer and Cowden-like phenotypes. J. Clin. Endocrinol. Metab. 2012, 97, E2320–E2327. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.; Lee, S.R.; Yang, K.S.; Ahn, Y.; Kim, Y.J.; Stadtman, E.R.; Rhee, S.G. Reversible oxidation and inactivation of the tumor suppressor PTEN in cells stimulated with peptide growth factors. Proc. Natl. Acad. Sci. USA 2004, 101, 16419–16424. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.R.; Yang, K.S.; Kwon, J.; Lee, C.; Jeong, W.; Rhee, S.G. Reversible inactivation of the tumor suppressor PTEN by H2O2. J. Biol. Chem. 2002, 277, 20336–20342. [Google Scholar] [CrossRef] [PubMed]
- Leslie, N.R.; Bennett, D.; Lindsay, Y.E.; Stewart, H.; Gray, A.; Downes, C.P. Redox regulation of PI 3-kinase signalling via inactivation of PTEN. EMBO J. 2003, 22, 5501–5510. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.H.; Lindsay, Y.; Safrany, S.T.; Lorenzo, O.; Villa, F.; Toth, R.; Clague, M.J.; Downes, C.P.; Leslie, N.R. Differential redox regulation within the PTP superfamily. Cell. Signal. 2007, 19, 1521–1530. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.H.; Lee, C.H.; Ahn, Y.; Kim, H.; Kim, H.; Ahn, C.Y.; Yang, K.S.; Lee, S.R. Redox regulation of PTEN and protein tyrosine phosphatases in H2O2 mediated cell signaling. FEBS Lett. 2004, 560, 7–13. [Google Scholar] [CrossRef]
- Kim, Y.; Song, Y.B.; Kim, T.Y.; Kim, I.; Han, S.J.; Ahn, Y.; Cho, S.H.; Choi, C.Y.; Chay, K.O.; Yang, S.Y.; et al. Redox regulation of the tumor suppressor PTEN by glutathione. FEBS Lett. 2010, 584, 3550–3556. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Schulte, J.; Knight, A.; Leslie, N.R.; Zagozdzon, A.; Bronson, R.; Manevich, Y.; Beeson, C.; Neumann, C.A. Prdx1 inhibits tumorigenesis via regulating PTEN/AKT activity. EMBO J. 2009, 28, 1505–1517. [Google Scholar] [CrossRef] [PubMed]
- Schwertassek, U.; Haque, A.; Krishnan, N.; Greiner, R.; Weingarten, L.; Dick, T.P.; Tonks, N.K. Reactivation of oxidized PTP1B and PTEN by thioredoxin 1. FEBS J. 2014, 281, 3545–3558. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Chay, K.O.; Kim, I.; Song, Y.B.; Kim, T.Y.; Han, S.J.; Ahn, Y.; Cho, S.H.; Hoe, K.L.; Ahn, B.W.; et al. Redox regulation of the tumor suppressor PTEN by glutaredoxin 5 and Ycp4. Biochem. Biophys. Res. Commun. 2011, 407, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Maehama, T.; Dixon, J.E. The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J. Biol. Chem. 1998, 273, 13375–13378. [Google Scholar] [CrossRef] [PubMed]
- Phadngam, S.; Castiglioni, A.; Ferraresi, A.; Morani, F.; Follo, C.; Isidoro, C. PTEN dephosphorylates AKT to prevent the expression of GLUT1 on plasmamembrane and to limit glucose consumption in cancer cells. Oncotarget 2016, 7, 84999–85020. [Google Scholar] [CrossRef] [PubMed]
- Vogelmann, R.; Nguyen-Tat, M.D.; Giehl, K.; Adler, G.; Wedlich, D.; Menke, A. TGFbeta-induced downregulation of E-cadherin-based cell-cell adhesion depends on PI3-kinase and PTEN. J. Cell Sci. 2005, 118, 4901–4912. [Google Scholar] [CrossRef] [PubMed]
- Serezani, C.H.; Kane, S.; Medeiros, A.I.; Cornett, A.M.; Kim, S.H.; Marques, M.M.; Lee, S.P.; Lewis, C.; Bourdonnay, E.; Ballinger, M.N.; et al. PTEN directly activates the actin depolymerization factor cofilin-1 during PGE2-mediated inhibition of phagocytosis of fungi. Sci. Signal. 2012, 5, ra12. [Google Scholar] [CrossRef] [PubMed]
- Gu, T.; Zhang, Z.; Wang, J.; Guo, J.; Shen, W.H.; Yin, Y. CREB is a novel nuclear target of PTEN phosphatase. Cancer Res. 2011, 71, 2821–2825. [Google Scholar] [CrossRef] [PubMed]
- Kreis, P.; Hendricusdottir, R.; Kay, L.; Papageorgiou, I.E.; van Diepen, M.; Mack, T.; Ryves, J.; Harwood, A.; Leslie, N.R.; Kann, O.; et al. Phosphorylation of the actin binding protein Drebrin at S647 is regulated by neuronal activity and PTEN. PLoS ONE 2013, 8, e71957. [Google Scholar] [CrossRef] [PubMed]
- Brisbin, S.; Liu, J.; Boudreau, J.; Peng, J.; Evangelista, M.; Chin-Sang, I. A role for C. elegans Eph RTK signaling in PTEN regulation. Dev. Cell 2009, 17, 459–469. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Gu, J.; Matsumoto, K.; Aota, S.; Parsons, R.; Yamada, K.M. Inhibition of cell migration, spreading, and focal adhesions by tumor suppressor PTEN. Science 1998, 280, 1614–1617. [Google Scholar] [CrossRef] [PubMed]
- Dey, N.; Crosswell, H.E.; De, P.; Parsons, R.; Peng, Q.; Su, J.D.; Durden, D.L. The protein phosphatase activity of PTEN regulates SRC family kinases and controls glioma migration. Cancer Res. 2008, 68, 1862–1871. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Visser-Grieve, S.; Boudreau, J.; Yeung, B.; Lo, S.; Chamberlain, G.; Yu, F.; Sun, T.; Papanicolaou, T.; Lam, A.; et al. Insulin activates the insulin receptor to downregulate the PTEN tumour suppressor. Oncogene 2014, 33, 3878–3885. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhu, M.; Pan, R.; Fang, T.; Cao, Y.Y.; Chen, S.; Zhao, X.; Lei, C.Q.; Guo, L.; Chen, Y.; et al. The tumor suppressor PTEN has a critical role in antiviral innate immunity. Nat. Immunol. 2016, 17, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Wang, J.; Chandarlapaty, S.; Cross, J.; Thompson, C.; Rosen, N.; Jiang, X. PTEN is a protein tyrosine phosphatase for IRS1. Nat. Struct. Mol. Biol. 2014, 21, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Liang, J.; Li, J.; Li, Y.; Liang, H.; Zhao, X.; McNutt, M.A.; Yin, Y. PTEN Controls the DNA Replication Process through MCM2 in Response to Replicative Stress. Cell Rep. 2015, 13, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- Mahimainathan, L.; Choudhury, G.G. Inactivation of platelet-derived growth factor receptor by the tumor suppressor PTEN provides a novel mechanism of action of the phosphatase. J. Biol. Chem. 2004, 279, 15258–15268. [Google Scholar] [CrossRef] [PubMed]
- Raftopoulou, M.; Etienne-Manneville, S.; Self, A.; Nicholls, S.; Hall, A. Regulation of cell migration by the C2 domain of the tumor suppressor PTEN. Science 2004, 303, 1179–1181. [Google Scholar] [CrossRef] [PubMed]
- Wozniak, D.J.; Kajdacsy-Balla, A.; Macias, V.; Ball-Kell, S.; Zenner, M.L.; Bie, W.; Tyner, A.L. PTEN is a protein phosphatase that targets active PTK6 and inhibits PTK6 oncogenic signaling in prostate cancer. Nat. Commun. 2017, 8, 1508. [Google Scholar] [CrossRef] [PubMed]
- He, J.; de la Monte, S.; Wands, J.R. The p85beta regulatory subunit of PI3K serves as a substrate for PTEN protein phosphatase activity during insulin mediated signaling. Biochem. Biophys. Res. Commun. 2010, 397, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hou, S.Q.; He, J.; Gu, T.; Yin, Y.; Shen, W.H. PTEN regulates PLK1 and controls chromosomal stability during cell division. Cell Cycle 2016, 15, 2476–2485. [Google Scholar] [CrossRef] [PubMed]
- Shinde, S.R.; Maddika, S. PTEN modulates EGFR late endocytic trafficking and degradation by dephosphorylating Rab7. Nat. Commun. 2016, 7, 10689. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Tamura, M.; Pankov, R.; Danen, E.H.; Takino, T.; Matsumoto, K.; Yamada, K.M. Shc and FAK differentially regulate cell motility and directionality modulated by PTEN. J. Cell Biol. 1999, 146, 389–403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, W.C.; Li, P.; Guo, H.; Poh, S.B.; Brady, S.W.; Xiong, Y.; Tseng, L.M.; Li, S.H.; Ding, Z.; et al. Combating trastuzumab resistance by targeting SRC, a common node downstream of multiple resistance pathways. Nat. Med. 2011, 17, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.P.; Zhang, Y.; Van Cleemput, J.; Jiang, W.; Liao, M.; Li, L.; Wan, Q.; Backstrom, J.R.; Zhang, X. Disruption of PTEN coupling with 5-HT2C receptors suppresses behavioral responses induced by drugs of abuse. Nat. Med. 2006, 12, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Dillon, L.M.; Miller, T.W. Therapeutic targeting of cancers with loss of PTEN function. Curr. Drug Targets 2014, 15, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Boosani, C.S.; Agrawal, D.K. PTEN modulators: A patent review. Expert Opin. Ther. Pat. 2013, 23, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Suh, J.; Surh, Y.J.; Na, H.K. Regulation of the tumor suppressor PTEN by natural anticancer compounds. Ann. N. Y. Acad. Sci. 2017, 1401, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Mak, L.H.; Woscholski, R. Targeting PTEN using small molecule inhibitors. Methods 2015, 77–78, 63–68. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, N.M.; Mueller, C.; Grossmann, T.N. The Therapeutic Potential of PTEN Modulation: Targeting Strategies from Gene to Protein. Cell Chem. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Rosivatz, E. Inhibiting PTEN. Biochem. Soc. Trans. 2007, 35, 257–259. [Google Scholar] [CrossRef] [PubMed]
- Gordon, J.A. Use of vanadate as protein-phosphotyrosine phosphatase inhibitor. Methods Enzymol. 1991, 201, 477–482. [Google Scholar] [PubMed]
- Huyer, G.; Liu, S.; Kelly, J.; Moffat, J.; Payette, P.; Kennedy, B.; Tsaprailis, G.; Gresser, M.J.; Ramachandran, C. Mechanism of inhibition of protein-tyrosine phosphatases by vanadate and pervanadate. J. Biol. Chem. 1997, 272, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Posner, B.I.; Faure, R.; Burgess, J.W.; Bevan, A.P.; Lachance, D.; Zhang-Sun, G.; Fantus, I.G.; Ng, J.B.; Hall, D.A.; Lum, B.S.; et al. Peroxovanadium compounds. A new class of potent phosphotyrosine phosphatase inhibitors which are insulin mimetics. J. Biol. Chem. 1994, 269, 4596–4604. [Google Scholar] [PubMed]
- Mak, L.H.; Vilar, R.; Woscholski, R. Characterisation of the PTEN inhibitor VO-OHpic. J. Chem. Biol. 2010, 3, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Rosivatz, E.; Matthews, J.G.; McDonald, N.Q.; Mulet, X.; Ho, K.K.; Lossi, N.; Schmid, A.C.; Mirabelli, M.; Pomeranz, K.M.; Erneux, C.; et al. A small molecule inhibitor for phosphatase and tensin homologue deleted on chromosome 10 (PTEN). ACS Chem. Biol. 2006, 1, 780–790. [Google Scholar] [CrossRef] [PubMed]
- Schmid, A.C.; Byrne, R.D.; Vilar, R.; Woscholski, R. Bisperoxovanadium compounds are potent PTEN inhibitors. FEBS Lett. 2004, 566, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Chen, J.; Han, X.; Zhang, Y.; Liao, H.B.; Lei, R.X.; Zhuang, Y.; Wang, Z.F.; Li, Z.; Chen, J.C.; et al. Bisperoxovandium (pyridin-2-squaramide) targets both PTEN and ERK1/2 to confer neuroprotection. Br. J. Pharmacol. 2017, 174, 641–656. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.U.; Hahne, G.; Hanske, J.; Bange, T.; Bier, D.; Rademacher, C.; Hennig, S.; Grossmann, T.N. Redox Modulation of PTEN Phosphatase Activity by Hydrogen Peroxide and Bisperoxidovanadium Complexes. Angew. Chem. Int. Ed. Engl. 2015, 54, 13796–13800. [Google Scholar] [CrossRef] [PubMed]
- Garlich, J.R.; Durden, D.L.; Georgiadis, T.M.; Su, J.; Peng, X.; Smith, T.C. PTEN Inhibitors. Patent EP1755574 A2, 28 February 2007. [Google Scholar]
- Li, Y.; Prasad, A.; Jia, Y.; Roy, S.G.; Loison, F.; Mondal, S.; Kocjan, P.; Silberstein, L.E.; Ding, S.; Luo, H.R. Pretreatment with phosphatase and tensin homolog deleted on chromosome 10 (PTEN) inhibitor SF1670 augments the efficacy of granulocyte transfusion in a clinically relevant mouse model. Blood 2011, 117, 6702–6713. [Google Scholar] [CrossRef] [PubMed]
- Ajeawung, N.F.; Faure, R.; Jones, C.; Kamnasaran, D. Preclinical evaluation of dipotassium bisperoxo (picolinato) oxovanadate V for the treatment of pediatric low-grade gliomas. Future Oncol. 2013, 9, 1215–1229. [Google Scholar] [CrossRef] [PubMed]
- Faure, R.; Vincent, M.; Dufour, M.; Shaver, A.; Posner, B.I. Arrest at the G2/M transition of the cell cycle by protein-tyrosine phosphatase inhibition: Studies on a neuronal and a glial cell line. J. Cell. Biochem. 1995, 59, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Scrivens, P.J.; Alaoui-Jamali, M.A.; Giannini, G.; Wang, T.; Loignon, M.; Batist, G.; Sandor, V.A. Cdc25A-inhibitory properties and antineoplastic activity of bisperoxovanadium analogues. Mol. Cancer Ther. 2003, 2, 1053–1059. [Google Scholar] [PubMed]
- Spinelli, L.; Lindsay, Y.E.; Leslie, N.R. PTEN inhibitors: An evaluation of current compounds. Adv. Biol. Regul. 2015, 57, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.G.; Wu, D.N.; Han, D.; Zhang, G.Y. Critical role of PTEN in the coupling between PI3K/Akt and JNK1/2 signaling in ischemic brain injury. FEBS Lett. 2007, 581, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Dankowski, A.; Hagg, T. Protein tyrosine phosphatase inhibition reduces degeneration of dopaminergic substantia nigra neurons and projections in 6-OHDA treated adult rats. Eur. J. Neurosci. 2007, 25, 1332–1340. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, S.; Arnold, S.A.; Mahoney, E.T.; Sithu, S.D.; Zhang, Y.P.; D’Souza, S.E.; Shields, C.B.; Hagg, T. Small-molecule protein tyrosine phosphatase inhibition as a neuroprotective treatment after spinal cord injury in adult rats. J. Neurosci. 2008, 28, 7293–7303. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Qu, Y.; Mao, M.; Zhang, X.; Li, J.; Ferriero, D.; Mu, D. Involvement of the PTEN-AKT-FOXO3a pathway in neuronal apoptosis in developing rat brain after hypoxia-ischemia. J. Cereb. Blood Flow Metab. 2009, 29, 1903–1913. [Google Scholar] [CrossRef] [PubMed]
- Christie, K.J.; Webber, C.A.; Martinez, J.A.; Singh, B.; Zochodne, D.W. PTEN inhibition to facilitate intrinsic regenerative outgrowth of adult peripheral axons. J. Neurosci. 2010, 30, 9306–9315. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.D.; OuYang, Y.P.; Shi, J.G.; Liu, Y.; Yuan, W.; Jia, L.S. PTEN deletion prevents ischemic brain injury by activating the mTOR signaling pathway. Biochem. Biophys. Res. Commun. 2011, 404, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Walker, M.J.; Liu, N.K.; Risberg, E.C.; Gao, X.; Chen, J.; Xu, X.M. Systemic bisperoxovanadium activates Akt/mTOR, reduces autophagy, and enhances recovery following cervical spinal cord injury. PLoS ONE 2012, 7, e30012. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Wang, X.; Bullis, C.; Liu, N.K.; Lu, Q.; Fry, C.; Deng, L.; Xu, X.M. Biphasic bisperoxovanadium administration and Schwann cell transplantation for repair after cervical contusive spinal cord injury. Exp. Neurol. 2015, 264, 163–172. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.L.; Xu, X.M. PTEN inhibitor bisperoxovanadium protects oligodendrocytes and myelin and prevents neuronal atrophy in adult rats following cervical hemicontusive spinal cord injury. Neurosci. Lett. 2014, 573, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Qu, Y.; Wu, J.; Cao, M.; Ferriero, D.M.; Zhang, L.; Mu, D. PTEN inhibition prevents rat cortical neuron injury after hypoxia-ischemia. Neuroscience 2013, 238, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Guo, J.; Yuan, Q.; Yuan, F.; Chen, H.; Tian, H. Inhibition of phosphatase and tensin homolog deleted on chromosome 10 decreases rat cortical neuron injury and blood-brain barrier permeability, and improves neurological functional recovery in traumatic brain injury model. PLoS ONE 2013, 8, e80429. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.Y.; Ding, J.; Yuan, F.; Chen, H.; Chen, S.W.; Tian, H.L. Dose-dependent protective effect of bisperoxovanadium against acute cerebral ischemia in a rat model of ischemia/reperfusion injury. Int. J. Mol. Sci. 2013, 14, 12013–12022. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, L.; Long, Z.Y.; Wu, Y.M.; Wan, Q.; Jiang, J.X.; Wang, Z.G. Inhibiting PTEN protects hippocampal neurons against stretch injury by decreasing membrane translocation of AMPA receptor GluR2 subunit. PLoS ONE 2013, 8, e65431. [Google Scholar] [CrossRef] [PubMed]
- Wyatt, L.A.; Filbin, M.T.; Keirstead, H.S. PTEN inhibition enhances neurite outgrowth in human embryonic stem cell-derived neuronal progenitor cells. J. Comp. Neurol. 2014, 522, 2741–2755. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Luo, C.; Zhao, M.; Li, Q.; Hu, R.; Zhang, J.H.; Liu, Z.; Feng, H. Administration of a PTEN inhibitor BPV(pic) attenuates early brain injury via modulating AMPA receptor subunits after subarachnoid hemorrhage in rats. Neurosci. Lett. 2015, 588, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.L.; Hao, D.L.; Mao, X.W.; Xu, Y.F.; Huang, T.T.; Wu, B.N.; Wang, L.H. Neuroprotective effects of bisperoxovanadium on cerebral ischemia by inflammation inhibition. Neurosci. Lett. 2015, 602, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Vuong, P.M.; Nguyen, H.T.; Katano, T.; Matsumura, S.; Saito, A.; Yamada, A.; Furue, H.; Ito, S. Impaired peripheral nerve regeneration in type-2 diabetic mouse model. Eur. J. Neurosci. 2017. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, B.; Xu, W.F.; Liu, R.F.; Yang, J.; Yu, C.X. Effects of PTEN inhibition on regulation of tau phosphorylation in an okadaic acid-induced neurodegeneration model. Int. J. Dev. Neurosci. 2012, 30, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Knafo, S.; Sanchez-Puelles, C.; Palomer, E.; Delgado, I.; Draffin, J.E.; Mingo, J.; Wahle, T.; Kaleka, K.; Mou, L.; Pereda-Perez, I.; et al. PTEN recruitment controls synaptic and cognitive function in Alzheimer’s models. Nat. Neurosci. 2016, 19, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Wang, S.; Wang, Z.; Wang, Z.; Sun, C.; Zhang, Y. Inhibition of PTEN Attenuates Endoplasmic Reticulum Stress and Apoptosis via Activation of PI3K/AKT Pathway in Alzheimer’s Disease. Neurochem. Res. 2017, 42, 3052–3060. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Zhang, L.J.; Chen, Z.; Liu, L.B. The PTEN inhibitor bpV(pic) promotes neuroprotection against amyloid beta-peptide (25-35)-induced oxidative stress and neurotoxicity. Neurol. Res. 2017, 39, 758–765. [Google Scholar] [CrossRef] [PubMed]
- De Paula, M.L.; Cui, Q.L.; Hossain, S.; Antel, J.; Almazan, G. The PTEN inhibitor bisperoxovanadium enhances myelination by amplifying IGF-1 signaling in rat and human oligodendrocyte progenitors. Glia 2014, 62, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Li, F.; Chen, X. Pten Inhibitor-bpV Ameliorates Early Postnatal Propofol Exposure-Induced Memory Deficit and Impairment of Hippocampal LTP. Neurochem. Res. 2015, 40, 1593–1599. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, W.; Zhang, J.; Li, S.; Zhao, Y.; Tan, L.; Luo, A. Involvement of caspase-3/PTEN signaling pathway in isoflurane-induced decrease of self-renewal capacity of hippocampal neural precursor cells. Brain Res. 2015, 1625, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Chen, X.; Wang, W.; Zhang, J.; Li, S.; Zhao, Y.; Wang, J.; Luo, A. Pharmacological inhibition of PTEN attenuates cognitive deficits caused by neonatal repeated exposures to isoflurane via inhibition of NR2B-mediated tau phosphorylation in rats. Neuropharmacology 2017, 114, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Grande, V.; Manassero, G.; Vercelli, A. Neuroprotective and Anti-Inflammatory Roles of the Phosphatase and Tensin Homolog Deleted on Chromosome Ten (PTEN) Inhibition in a Mouse Model of Temporal Lobe Epilepsy. PLoS ONE 2014, 9, e114554. [Google Scholar] [CrossRef] [PubMed]
- Sury, M.D.; Vorlet-Fawer, L.; Agarinis, C.; Yousefi, S.; Grandgirard, D.; Leib, S.L.; Christen, S. Restoration of Akt activity by the bisperoxovanadium compound bpV(pic) attenuates hippocampal apoptosis in experimental neonatal pneumococcal meningitis. Neurobiol. Dis. 2011, 41, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Keyes, K.T.; Xu, J.; Long, B.; Zhang, C.; Hu, Z.; Ye, Y. Pharmacological inhibition of PTEN limits myocardial infarct size and improves left ventricular function postinfarction. Am. J. Physiol. Heart Circ. Physiol. 2010, 298, H1198–H1208. [Google Scholar] [CrossRef] [PubMed]
- Zu, L.; Shen, Z.; Wesley, J.; Cai, Z.P. PTEN inhibitors cause a negative inotropic and chronotropic effect in mice. Eur. J. Pharmacol. 2011, 650, 298–302. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Jia, J.; Zhou, X.; Xiao, Y.; Wang, Y.; Mao, X.; Zhen, X.; Guan, Y.; Alkayed, N.J.; Cheng, J. Delayed administration of a PTEN inhibitor BPV improves functional recovery after experimental stroke. Neuroscience 2013, 231, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Shao, Z.H.; Li, C.; Li, J.; Zhong, Q.; Learoyd, J.; Meliton, A.; Meliton, L.; Leff, A.R.; Vanden Hoek, T.L. TAT-protein blockade during ischemia/reperfusion reveals critical role for p85 PI3K-PTEN interaction in cardiomyocyte injury. PLoS ONE 2014, 9, e95622. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, H.; Zhong, Q.; Zhu, X.; Chen, S.J.; Qian, Y.; Costakis, J.; Bunney, G.; Beiser, D.G.; Leff, A.R.; et al. A novel pharmacological strategy by PTEN inhibition for improving metabolic resuscitation and survival after mouse cardiac arrest. Am. J. Physiol. Heart Circ. Physiol. 2015, 308, H1414–H1422. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Lei, S.; Xia, Z.Y.; Wu, Y.; Meng, Q.; Zhan, L.; Su, W.; Liu, H.; Xu, J.; Liu, Z.; et al. Selective inhibition of PTEN preserves ischaemic post-conditioning cardioprotection in STZ-induced Type 1 diabetic rats: Role of the PI3K/Akt and JAK2/STAT3 pathways. Clin. Sci. 2016, 130, 377–392. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Daoud, A.; Shang, J. Effects of bpV(pic) and bpV(phen) on H9c2 cardiomyoblasts during both hypoxia/reoxygenation and H2O2-induced injuries. Mol. Med. Rep. 2012, 5, 852–858. [Google Scholar] [PubMed]
- Dal Ponte, C.; Alchera, E.; Follenzi, A.; Imarisio, C.; Prat, M.; Albano, E.; Carini, R. Pharmacological postconditioning protects against hepatic ischemia/reperfusion injury. Liver Transpl. 2011, 17, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.P.; Dalton, J.T.; Knoell, D.L. Phosphatase and tensin homologue deleted on chromosome ten (PTEN) as a molecular target in lung epithelial wound repair. Br. J. Pharmacol. 2007, 152, 1172–1184. [Google Scholar] [CrossRef] [PubMed]
- Mihai, C.; Bao, S.; Lai, J.P.; Ghadiali, S.N.; Knoell, D.L. PTEN inhibition improves wound healing in lung epithelia through changes in cellular mechanics that enhance migration. Am. J. Physiol. Lung Cell. Mol. Physiol. 2012, 302, L287–L299. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.P.; Bao, S.; Davis, I.C.; Knoell, D.L. Inhibition of the phosphatase PTEN protects mice against oleic acid-induced acute lung injury. Br. J. Pharmacol. 2009, 156, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, S.; Wang, X.; Zhou, J.; Cao, Y.; Wang, F.; Duan, E. The PI3K-Akt pathway inhibits senescence and promotes self-renewal of human skin-derived precursors in vitro. Aging Cell 2011, 10, 661–674. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Graue-Hernandez, E.O.; Tran, V.; Reid, B.; Pu, J.; Mannis, M.J.; Zhao, M. Downregulation of PTEN at corneal wound sites accelerates wound healing through increased cell migration. Investig. Ophthalmol. Vis. Sci. 2011, 52, 2272–2278. [Google Scholar] [CrossRef] [PubMed]
- Dimchev, G.A.; Al-Shanti, N.; Stewart, C.E. Phospho-tyrosine phosphatase inhibitor Bpv(Hopic) enhances C2C12 myoblast migration in vitro. Requirement of PI3K/AKT and MAPK/ERK pathways. J. Muscle Res. Cell Motil. 2013, 34, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.; Sun, X. Reactivation of the PI3K/Akt Signaling Pathway by the Bisperoxovanadium Compound bpV(pic) Attenuates Photoreceptor Apoptosis in Experimental Retinal Detachment. Investig. Ophthalmol. Vis. Sci. 2015, 56, 5519–5532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Ma, J.; Yu, P.; Tang, G.J.; Li, C.J.; Yu, D.M.; Zhang, Q.M. Reduced beta 2 glycoprotein I prevents high glucose-induced cell death in HUVECs through miR-21/PTEN. Am. J. Transl. Res. 2017, 9, 3935–3949. [Google Scholar] [PubMed]
- Canetti, C.; Serezani, C.H.; Atrasz, R.G.; White, E.S.; Aronoff, D.M.; Peters-Golden, M. Activation of phosphatase and tensin homolog on chromosome 10 mediates the inhibition of FcgammaR phagocytosis by prostaglandin E2 in alveolar macrophages. J. Immunol. 2007, 179, 8350–8356. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, L.L.; Wilke, C.A.; White, E.S.; Moore, B.B. PTEN limits alveolar macrophage function against Pseudomonas aeruginosa after bone marrow transplantation. Am. J. Respir. Cell Mol. Biol. 2011, 45, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Kawamura, K.; Cheng, Y.; Liu, S.; Klein, C.; Liu, S.; Duan, E.K.; Hsueh, A.J. Activation of dormant ovarian follicles to generate mature eggs. Proc. Natl. Acad. Sci. USA 2010, 107, 10280–10284. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, D.; Gorre, N.; Risal, S.; Zhao, Z.; Zhang, H.; Shen, Y.; Liu, K. The safe use of a PTEN inhibitor for the activation of dormant mouse primordial follicles and generation of fertilizable eggs. PLoS ONE 2012, 7, e39034. [Google Scholar] [CrossRef] [PubMed]
- Morohaku, K.; Hoshino, Y.; Sasada, H.; Sato, E. Incorporation of phosphatase inhibitor in culture prompts growth initiation of isolated non-growing oocytes. PLoS ONE 2013, 8, e77533. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, K.; Cheng, Y.; Suzuki, N.; Deguchi, M.; Sato, Y.; Takae, S.; Ho, C.H.; Kawamura, N.; Tamura, M.; Hashimoto, S.; et al. Hippo signaling disruption and Akt stimulation of ovarian follicles for infertility treatment. Proc. Natl. Acad. Sci. USA 2013, 110, 17474–17479. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, N.; Yoshioka, N.; Takae, S.; Sugishita, Y.; Tamura, M.; Hashimoto, S.; Morimoto, Y.; Kawamura, K. Successful fertility preservation following ovarian tissue vitrification in patients with primary ovarian insufficiency. Hum. Reprod. 2015, 30, 608–615. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, M.; Kinnell, H.L.; Anderson, R.A.; Telfer, E.E. Inhibition of phosphatase and tensin homologue (PTEN) in human ovary in vitro results in increased activation of primordial follicles but compromises development of growing follicles. Mol. Hum. Reprod. 2014, 20, 736–744. [Google Scholar] [CrossRef] [PubMed]
- Novella-Maestre, E.; Herraiz, S.; Rodriguez-Iglesias, B.; Diaz-Garcia, C.; Pellicer, A. Short-Term PTEN Inhibition Improves In Vitro Activation of Primordial Follicles, Preserves Follicular Viability, and Restores AMH Levels in Cryopreserved Ovarian Tissue from Cancer Patients. PLoS ONE 2015, 10, e0127786. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Marumoto, T.; Yamaguchi, S.; Okano, S.; Takeda, N.; Sakamoto, C.; Kawano, H.; Nii, T.; Miyamato, S.; Nagai, Y.; et al. Inhibition of PTEN tumor suppressor promotes the generation of induced pluripotent stem cells. Mol. Ther. 2013, 21, 1242–1250. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, T.; Wu, Y.; Tang, Z. Role and mechanism of PTEN in adiponectin-induced osteogenesis in human bone marrow mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2017, 483, 712–717. [Google Scholar] [CrossRef] [PubMed]
- Alimonti, A.; Nardella, C.; Chen, Z.; Clohessy, J.G.; Carracedo, A.; Trotman, L.C.; Cheng, K.; Varmeh, S.; Kozma, S.C.; Thomas, G.; et al. A novel type of cellular senescence that can be enhanced in mouse models and human tumor xenografts to suppress prostate tumorigenesis. J. Clin. Investig. 2010, 120, 681–693. [Google Scholar] [CrossRef] [PubMed]
- Augello, G.; Puleio, R.; Emma, M.R.; Cusimano, A.; Loria, G.R.; McCubrey, J.A.; Montalto, G.; Cervello, M. A PTEN inhibitor displays preclinical activity against hepatocarcinoma cells. Cell Cycle 2016, 15, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.D.; Shinde, R.; McGaha, T.L.; Huang, L.; Holmgaard, R.B.; Wolchok, J.D.; Mautino, M.R.; Celis, E.; Sharpe, A.H.; Francisco, L.M.; et al. The PTEN pathway in Tregs is a critical driver of the suppressive tumor microenvironment. Sci. Adv. 2015, 1, e1500845. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.R.; Zaytseva, Y.Y.; Jackson, L.N.; Lee, E.Y.; Weiss, H.L.; Bowen, K.A.; Townsend, C.M., Jr.; Evers, B.M. The effect of PTEN on serotonin synthesis and secretion from the carcinoid cell line BON. Anticancer Res. 2011, 31, 1153–1160. [Google Scholar] [PubMed]
- Bulger, D.A.; Conley, J.; Conner, S.H.; Majumdar, G.; Solomon, S.S. Role of PTEN in TNFalpha induced insulin resistance. Biochem. Biophys. Res. Commun. 2015, 461, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.F.; Yang, H.J.; Gu, J.Q.; Cao, Y.L.; Meng, X.; Wang, X.L.; Lin, Y.C.; Gao, M. Suppression of phosphatase and tensin homolog protects insulin-resistant cells from apoptosis. Mol. Med. Rep. 2015, 12, 2695–2700. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, R.M.; Neves, K.B.; Mestriner, F.L.; Louzada-Junior, P.; Bruder-Nascimento, T.; Tostes, R.C. TNF-alpha induces vascular insulin resistance via positive modulation of PTEN and decreased Akt/eNOS/NO signaling in high fat diet-fed mice. Cardiovasc. Diabetol. 2016, 15, 119. [Google Scholar] [CrossRef] [PubMed]
- Suo, Z.W.; Yang, X.; Li, L.; Liu, Y.N.; Shi, L.; Hu, X.D. Inhibition of protein tyrosine phosphatases in spinal dorsal horn attenuated inflammatory pain by repressing Src signaling. Neuropharmacology 2013, 70, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Shiwarski, D.J.; Tipton, A.; Giraldo, M.D.; Schmidt, B.F.; Gold, M.S.; Pradhan, A.A.; Puthenveedu, M.A. A PTEN-Regulated Checkpoint Controls Surface Delivery of delta Opioid Receptors. J. Neurosci. 2017, 37, 3741–3752. [Google Scholar] [CrossRef] [PubMed]
- Christie, K.J.; Zochodne, D. Peripheral axon regrowth: New molecular approaches. Neuroscience 2013, 240, 310–324. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Junco-Clemente, P.; Golshani, P. PTEN: A master regulator of neuronal structure, function, and plasticity. Commun. Integr. Biol. 2014, 7, e28358. [Google Scholar] [CrossRef] [PubMed]
- Knafo, S.; Esteban, J.A. PTEN: Local and Global Modulation of Neuronal Function in Health and Disease. Trends Neurosci. 2017, 40, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, Y.; Hayat, U.; Li, S. PTEN inhibition and axon regeneration and neural repair. Neural Regen. Res. 2015, 10, 1363–1368. [Google Scholar] [PubMed]
- Park, K.K.; Liu, K.; Hu, Y.; Kanter, J.L.; He, Z. PTEN/mTOR and axon regeneration. Exp. Neurol. 2010, 223, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Zhang, L.; Chen, Y.; Zhan, L.; Gao, Z. Therapeutic Targets for Cerebral Ischemia Based on the Signaling Pathways of the GluN2B C Terminus. Stroke 2015, 46, 2347–2353. [Google Scholar] [CrossRef] [PubMed]
- Park, K.K.; Liu, K.; Hu, Y.; Smith, P.D.; Wang, C.; Cai, B.; Xu, B.; Connolly, L.; Kramvis, I.; Sahin, M.; et al. Promoting axon regeneration in the adult CNS by modulation of the PTEN/mTOR pathway. Science 2008, 322, 963–966. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Lu, Y.; Lee, J.K.; Samara, R.; Willenberg, R.; Sears-Kraxberger, I.; Tedeschi, A.; Park, K.K.; Jin, D.; Cai, B.; et al. PTEN deletion enhances the regenerative ability of adult corticospinal neurons. Nat. Neurosci. 2010, 13, 1075–1081. [Google Scholar] [CrossRef] [PubMed]
- Danilov, C.A.; Steward, O. Conditional genetic deletion of PTEN after a spinal cord injury enhances regenerative growth of CST axons and motor function recovery in mice. Exp. Neurol. 2015, 266, 147–160. [Google Scholar] [CrossRef] [PubMed]
- Du, K.; Zheng, S.; Zhang, Q.; Li, S.; Gao, X.; Wang, J.; Jiang, L.; Liu, K. Pten Deletion Promotes Regrowth of Corticospinal Tract Axons 1 Year after Spinal Cord Injury. J. Neurosci. 2015, 35, 9754–9763. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Liu, Y.; Sun, F.; Wang, X.; Liu, X.; He, Z. Restoration of skilled locomotion by sprouting corticospinal axons induced by co-deletion of PTEN and SOCS3. Nat. Commun. 2015, 6, 8074. [Google Scholar] [CrossRef] [PubMed]
- Kurimoto, T.; Yin, Y.; Omura, K.; Gilbert, H.Y.; Kim, D.; Cen, L.P.; Moko, L.; Kugler, S.; Benowitz, L.I. Long-distance axon regeneration in the mature optic nerve: Contributions of oncomodulin, cAMP, and pten gene deletion. J. Neurosci. 2010, 30, 15654–15663. [Google Scholar] [CrossRef] [PubMed]
- Lewandowski, G.; Steward, O. AAVshRNA-mediated suppression of PTEN in adult rats in combination with salmon fibrin administration enables regenerative growth of corticospinal axons and enhances recovery of voluntary motor function after cervical spinal cord injury. J. Neurosci. 2014, 34, 9951–9962. [Google Scholar] [CrossRef] [PubMed]
- Little, D.; Valori, C.F.; Mutsaers, C.A.; Bennett, E.J.; Wyles, M.; Sharrack, B.; Shaw, P.J.; Gillingwater, T.H.; Azzouz, M.; Ning, K. PTEN depletion decreases disease severity and modestly prolongs survival in a mouse model of spinal muscular atrophy. Mol. Ther. 2015, 23, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Ning, K.; Drepper, C.; Valori, C.F.; Ahsan, M.; Wyles, M.; Higginbottom, A.; Herrmann, T.; Shaw, P.; Azzouz, M.; Sendtner, M. PTEN depletion rescues axonal growth defect and improves survival in SMN-deficient motor neurons. Hum. Mol. Genet. 2010, 19, 3159–3168. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Singh, V.; Krishnan, A.; Koshy, K.; Martinez, J.A.; Cheng, C.; Almquist, C.; Zochodne, D.W. Regeneration of diabetic axons is enhanced by selective knockdown of the PTEN gene. Brain 2014, 137, 1051–1067. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Park, K.K.; Belin, S.; Wang, D.; Lu, T.; Chen, G.; Zhang, K.; Yeung, C.; Feng, G.; Yankner, B.A.; et al. Sustained axon regeneration induced by co-deletion of PTEN and SOCS3. Nature 2011, 480, 372–375. [Google Scholar] [CrossRef] [PubMed]
- Zukor, K.; Belin, S.; Wang, C.; Keelan, N.; Wang, X.; He, Z. Short hairpin RNA against PTEN enhances regenerative growth of corticospinal tract axons after spinal cord injury. J. Neurosci. 2013, 33, 15350–15361. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.S.; Stutzmann, G.E. Inhibition of Glycogen Synthase Kinase-3: An Emerging Target in the Treatment of Traumatic Brain Injury. J. Neurotrauma 2016, 33, 2065–2076. [Google Scholar] [CrossRef] [PubMed]
- Griffin, R.J.; Moloney, A.; Kelliher, M.; Johnston, J.A.; Ravid, R.; Dockery, P.; O’Connor, R.; O’Neill, C. Activation of Akt/PKB, increased phosphorylation of Akt substrates and loss and altered distribution of Akt and PTEN are features of Alzheimer’s disease pathology. J. Neurochem. 2005, 93, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Rickle, A.; Bogdanovic, N.; Volkmann, I.; Zhou, X.; Pei, J.J.; Winblad, B.; Cowburn, R.F. PTEN levels in Alzheimer’s disease medial temporal cortex. Neurochem. Int. 2006, 48, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Sonoda, Y.; Mukai, H.; Matsuo, K.; Takahashi, M.; Ono, Y.; Maeda, K.; Akiyama, H.; Kawamata, T. Accumulation of tumor-suppressor PTEN in Alzheimer neurofibrillary tangles. Neurosci. Lett. 2010, 471, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Li, F.; Bulloj, A.; Zhang, Y.W.; Tong, G.; Zhang, Z.; Liao, F.F.; Xu, H. Tumor-suppressor PTEN affects tau phosphorylation, aggregation, and binding to microtubules. FASEB J. 2006, 20, 1272–1274. [Google Scholar] [CrossRef] [PubMed]
- Kerr, F.; Rickle, A.; Nayeem, N.; Brandner, S.; Cowburn, R.F.; Lovestone, S. PTEN, a negative regulator of PI3 kinase signalling, alters tau phosphorylation in cells by mechanisms independent of GSK-3. FEBS Lett. 2006, 580, 3121–3128. [Google Scholar] [CrossRef] [PubMed]
- Marciniak, E.; Leboucher, A.; Caron, E.; Ahmed, T.; Tailleux, A.; Dumont, J.; Issad, T.; Gerhardt, E.; Pagesy, P.; Vileno, M.; et al. Tau deletion promotes brain insulin resistance. J. Exp. Med. 2017, 214, 2257–2269. [Google Scholar] [CrossRef] [PubMed]
- Jurado, S.; Benoist, M.; Lario, A.; Knafo, S.; Petrok, C.N.; Esteban, J.A. PTEN is recruited to the postsynaptic terminal for NMDA receptor-dependent long-term depression. EMBO J. 2010, 29, 2827–2840. [Google Scholar] [CrossRef] [PubMed]
- Fraser, M.M.; Bayazitov, I.T.; Zakharenko, S.S.; Baker, S.J. Phosphatase and tensin homolog, deleted on chromosome 10 deficiency in brain causes defects in synaptic structure, transmission and plasticity, and myelination abnormalities. Neuroscience 2008, 151, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Goebbels, S.; Oltrogge, J.H.; Kemper, R.; Heilmann, I.; Bormuth, I.; Wolfer, S.; Wichert, S.P.; Mobius, W.; Liu, X.; Lappe-Siefke, C.; et al. Elevated phosphatidylinositol 3,4,5-trisphosphate in glia triggers cell-autonomous membrane wrapping and myelination. J. Neurosci. 2010, 30, 8953–8964. [Google Scholar] [CrossRef] [PubMed]
- Harrington, E.P.; Zhao, C.; Fancy, S.P.; Kaing, S.; Franklin, R.J.; Rowitch, D.H. Oligodendrocyte PTEN is required for myelin and axonal integrity, not remyelination. Ann. Neurol. 2010, 68, 703–716. [Google Scholar] [CrossRef] [PubMed]
- Maire, C.L.; Ramkissoon, S.; Hayashi, M.; Haidar, S.; Ramkissoon, L.; DiTomaso, E.; Ligon, K.L. Pten loss in Olig2 expressing neural progenitor cells and oligodendrocytes leads to interneuron dysplasia and leukodystrophy. Stem Cells 2014, 32, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Goebbels, S.; Wieser, G.L.; Pieper, A.; Spitzer, S.; Weege, B.; Yan, K.; Edgar, J.M.; Yagensky, O.; Wichert, S.P.; Agarwal, A.; et al. A neuronal PI(3,4,5)P3-dependent program of oligodendrocyte precursor recruitment and myelination. Nat. Neurosci. 2017, 20, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Sahin, E.; Brunner, J.S.; Kral, J.B.; Kuttke, M.; Hanzl, L.; Datler, H.; Paar, H.; Neuwinger, N.; Saferding, V.; Zinser, E.; et al. Loss of Phosphatase and Tensin Homolog in APCs Impedes Th17-Mediated Autoimmune Encephalomyelitis. J. Immunol. 2015, 195, 2560–2570. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Li, L.; Yuan, W. Neonatal anesthetic neurotoxicity: Insight into the molecular mechanisms of long-term neurocognitive deficits. Biomed. Pharmacother. 2017, 87, 196–199. [Google Scholar] [CrossRef] [PubMed]
- Adachi, T.; Takigawa, H.; Nomura, T.; Watanabe, Y.; Kowa, H. Cowden Syndrome with a Novel PTEN Mutation Presenting with Partial Epilepsy Related to Focal Cortical Dysplasia. Intern. Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.M.; Lam, C.W.; Chan, Y.K.; Siu, W.K.; Yong, L. Atypical focal cortical dysplasia in a patient with Cowden syndrome. Hong Kong Med. J. 2014, 20, 165–167. [Google Scholar] [CrossRef] [PubMed]
- Child, N.D.; Cascino, G.D. Mystery case: Cowden syndrome presenting with partial epilepsy related to focal cortical dysplasia. Neurology 2013, 81, e98–e99. [Google Scholar] [CrossRef] [PubMed]
- Conti, S.; Condo, M.; Posar, A.; Mari, F.; Resta, N.; Renieri, A.; Neri, I.; Patrizi, A.; Parmeggiani, A. Phosphatase and tensin homolog (PTEN) gene mutations and autism: Literature review and a case report of a patient with Cowden syndrome, autistic disorder, and epilepsy. J. Child Neurol. 2012, 27, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Elia, M.; Amato, C.; Bottitta, M.; Grillo, L.; Calabrese, G.; Esposito, M.; Carotenuto, M. An atypical patient with Cowden syndrome and PTEN gene mutation presenting with cortical malformation and focal epilepsy. Brain Dev. 2012, 34, 873–876. [Google Scholar] [CrossRef] [PubMed]
- Citraro, R.; Leo, A.; Constanti, A.; Russo, E.; De Sarro, G. mTOR pathway inhibition as a new therapeutic strategy in epilepsy and epileptogenesis. Pharmacol. Res. 2016, 107, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Kalogeris, T.; Baines, C.P.; Krenz, M.; Korthuis, R.J. Cell biology of ischemia/reperfusion injury. Int. Rev. Cell Mol. Biol. 2012, 298, 229–317. [Google Scholar] [PubMed]
- Mocanu, M.M.; Yellon, D.M. PTEN, the Achilles’ heel of myocardial ischaemia/reperfusion injury? Br. J. Pharmacol. 2007, 150, 833–838. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Semenza, G.L. PTEN activity is modulated during ischemia and reperfusion: Involvement in the induction and decay of preconditioning. Circ. Res. 2005, 97, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- Parajuli, N.; Yuan, Y.; Zheng, X.; Bedja, D.; Cai, Z.P. Phosphatase PTEN is critically involved in post-myocardial infarction remodeling through the Akt/interleukin-10 signaling pathway. Basic Res. Cardiol. 2012, 107, 248. [Google Scholar] [CrossRef] [PubMed]
- Siddall, H.K.; Warrell, C.E.; Yellon, D.M.; Mocanu, M.M. Ischemia-reperfusion injury and cardioprotection: Investigating PTEN, the phosphatase that negatively regulates PI3K, using a congenital model of PTEN haploinsufficiency. Basic Res. Cardiol. 2008, 103, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Crackower, M.A.; Oudit, G.Y.; Kozieradzki, I.; Sarao, R.; Sun, H.; Sasaki, T.; Hirsch, E.; Suzuki, A.; Shioi, T.; Irie-Sasaki, J.; et al. Regulation of myocardial contractility and cell size by distinct PI3K-PTEN signaling pathways. Cell 2002, 110, 737–749. [Google Scholar] [CrossRef]
- Oudit, G.Y.; Kassiri, Z.; Zhou, J.; Liu, Q.C.; Liu, P.P.; Backx, P.H.; Dawood, F.; Crackower, M.A.; Scholey, J.W.; Penninger, J.M. Loss of PTEN attenuates the development of pathological hypertrophy and heart failure in response to biomechanical stress. Cardiovasc. Res. 2008, 78, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Roe, N.D.; Xu, X.; Kandadi, M.R.; Hu, N.; Pang, J.; Weiser-Evans, M.C.; Ren, J. Targeted deletion of PTEN in cardiomyocytes renders cardiac contractile dysfunction through interruption of Pink1-AMPK signaling and autophagy. Biochim. Biophys. Acta 2015, 1852, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Yue, S.; Rao, J.; Zhu, J.; Busuttil, R.W.; Kupiec-Weglinski, J.W.; Lu, L.; Wang, X.; Zhai, Y. Myeloid PTEN deficiency protects livers from ischemia reperfusion injury by facilitating M2 macrophage differentiation. J. Immunol. 2014, 192, 5343–5353. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Fan, Y.; Tang, S.; Wu, H.; Zhong, J.; Huang, Z.; Yang, C.; Chen, H. Inhibition of PTEN activity aggravates cisplatin-induced acute kidney injury. Oncotarget 2017, 8, 103154–103166. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Jia, L.; Hu, Z.; Wang, Y. Pharmacological Inhibition of PTEN Aggravates Acute Kidney Injury. Sci. Rep. 2017, 7, 9503. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Wang, H.; Lee, I.H.; Modi, S.; Wang, X.; Du, J.; Mitch, W.E. PTEN inhibition improves muscle regeneration in mice fed a high-fat diet. Diabetes 2010, 59, 1312–1320. [Google Scholar] [CrossRef] [PubMed]
- Squarize, C.H.; Castilho, R.M.; Bugge, T.H.; Gutkind, J.S. Accelerated wound healing by mTOR activation in genetically defined mouse models. PLoS ONE 2010, 5, e10643. [Google Scholar] [CrossRef] [PubMed]
- Tiozzo, C.; de Langhe, S.; Yu, M.; Londhe, V.A.; Carraro, G.; Li, M.; Li, C.; Xing, Y.; Anderson, S.; Borok, Z.; et al. Deletion of Pten expands lung epithelial progenitor pools and confers resistance to airway injury. Am. J. Respir. Crit. Care Med. 2009, 180, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Tsugawa, K.; Jones, M.K.; Akahoshi, T.; Moon, W.S.; Maehara, Y.; Hashizume, M.; Sarfeh, I.J.; Tarnawski, A.S. Abnormal PTEN expression in portal hypertensive gastric mucosa: A key to impaired PI 3-kinase/Akt activation and delayed injury healing? FASEB J. 2003, 17, 2316–2318. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Song, B.; Pu, J.; Wada, T.; Reid, B.; Tai, G.; Wang, F.; Guo, A.; Walczysko, P.; Gu, Y.; et al. Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-gamma and PTEN. Nature 2006, 442, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Flodby, P.; Liebler, J.M.; Sunohara, M.; Castillo, D.R.; McConnell, A.M.; Krishnaveni, M.S.; Banfalvi, A.; Li, M.; Stripp, B.; Zhou, B.; et al. Region-specific role for Pten in maintenance of epithelial phenotype and integrity. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 312, L131–L142. [Google Scholar] [CrossRef] [PubMed]
- Leslie, N.R.; Maccario, H.; Spinelli, L.; Davidson, L. The significance of PTEN’s protein phosphatase activity. Adv. Enzyme Regul. 2009, 49, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Wei, G.; Fang, H.; Guo, J.; Weinstein, M.; Marsh, C.B.; Ostrowski, M.C.; Tridandapani, S. The inositol 3-phosphatase PTEN negatively regulates Fc gamma receptor signaling, but supports Toll-like receptor 4 signaling in murine peritoneal macrophages. J. Immunol. 2004, 172, 4851–4857. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Jia, Y.; Pichavant, M.; Loison, F.; Sarraj, B.; Kasorn, A.; You, J.; Robson, B.E.; Umetsu, D.T.; Mizgerd, J.P.; et al. Targeted deletion of tumor suppressor PTEN augments neutrophil function and enhances host defense in neutropenia-associated pneumonia. Blood 2009, 113, 4930–4941. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, K.K.; Jia, Y.; Zhu, D.; Simms, B.T.; Jo, H.; Hattori, H.; You, J.; Mizgerd, J.P.; Luo, H.R. Tumor suppressor PTEN is a physiologic suppressor of chemoattractant-mediated neutrophil functions. Blood 2007, 109, 4028–4037. [Google Scholar] [CrossRef] [PubMed]
- Jagarlamudi, K.; Liu, L.; Adhikari, D.; Reddy, P.; Idahl, A.; Ottander, U.; Lundin, E.; Liu, K. Oocyte-specific deletion of Pten in mice reveals a stage-specific function of PTEN/PI3K signaling in oocytes in controlling follicular activation. PLoS ONE 2009, 4, e6186. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.; Liu, L.; Adhikari, D.; Jagarlamudi, K.; Rajareddy, S.; Shen, Y.; Du, C.; Tang, W.; Hamalainen, T.; Peng, S.L.; et al. Oocyte-specific deletion of Pten causes premature activation of the primordial follicle pool. Science 2008, 319, 611–613. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.Y.; Liu, Z.; Cahill, N.; Richards, J.S. Targeted disruption of Pten in ovarian granulosa cells enhances ovulation and extends the life span of luteal cells. Mol. Endocrinol. 2008, 22, 2128–2140. [Google Scholar] [CrossRef] [PubMed]
- Lerer-Serfaty, G.; Samara, N.; Fisch, B.; Shachar, M.; Kossover, O.; Seliktar, D.; Ben-Haroush, A.; Abir, R. Attempted application of bioengineered/biosynthetic supporting matrices with phosphatidylinositol-trisphosphate-enhancing substances to organ culture of human primordial follicles. J. Assist. Reprod. Genet. 2013, 30, 1279–1288. [Google Scholar] [CrossRef] [PubMed]
- Lan, Z.J.; Krause, M.S.; Redding, S.D.; Li, X.; Wu, G.Z.; Zhou, H.X.; Bohler, H.C.; Ko, C.; Cooney, A.J.; Zhou, J.; et al. Selective deletion of Pten in theca-interstitial cells leads to androgen excess and ovarian dysfunction in mice. Mol. Cell. Endocrinol. 2017, 444, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Ciuffreda, L.; Falcone, I.; Incani, U.C.; Del Curatolo, A.; Conciatori, F.; Matteoni, S.; Vari, S.; Vaccaro, V.; Cognetti, F.; Milella, M. PTEN expression and function in adult cancer stem cells and prospects for therapeutic targeting. Adv. Biol. Regul. 2014, 56, 66–80. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.; Wu, H. PTEN, stem cells, and cancer stem cells. J. Biol. Chem. 2009, 284, 11755–11759. [Google Scholar] [CrossRef] [PubMed]
- Alva, J.A.; Lee, G.E.; Escobar, E.E.; Pyle, A.D. Phosphatase and tensin homolog regulates the pluripotent state and lineage fate choice in human embryonic stem cells. Stem Cells 2011, 29, 1952–1962. [Google Scholar] [CrossRef] [PubMed]
- Kuijk, E.W.; van Mil, A.; Brinkhof, B.; Penning, L.C.; Colenbrander, B.; Roelen, B.A. PTEN and TRP53 independently suppress Nanog expression in spermatogonial stem cells. Stem Cells Dev. 2010, 19, 979–988. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Cao, Y.; Wang, L.; Han, Y.; Zhong, X.; Zhou, G.; Cai, Y.; Zhang, H.; Gao, P. Human fibroblast reprogramming to pluripotent stem cells regulated by the miR19a/b-PTEN axis. PLoS ONE 2014, 9, e95213. [Google Scholar] [CrossRef] [PubMed]
- Burgers, T.A.; Hoffmann, M.F.; Collins, C.J.; Zahatnansky, J.; Alvarado, M.A.; Morris, M.R.; Sietsema, D.L.; Mason, J.J.; Jones, C.B.; Ploeg, H.L.; et al. Mice lacking pten in osteoblasts have improved intramembranous and late endochondral fracture healing. PLoS ONE 2013, 8, e63857. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Bruxvoort, K.J.; Zylstra, C.R.; Liu, J.; Cichowski, R.; Faugere, M.C.; Bouxsein, M.L.; Wan, C.; Williams, B.O.; Clemens, T.L. Lifelong accumulation of bone in mice lacking Pten in osteoblasts. Proc. Natl. Acad. Sci. USA 2007, 104, 2259–2264. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Trotman, L.C.; Shaffer, D.; Lin, H.K.; Dotan, Z.A.; Niki, M.; Koutcher, J.A.; Scher, H.I.; Ludwig, T.; Gerald, W.; et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 2005, 436, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Kim, B.C.; Park, M.J.; Lee, Y.S.; Kim, Y.N.; Lee, B.L.; Lee, J.S. PTEN status switches cell fate between premature senescence and apoptosis in glioma exposed to ionizing radiation. Cell Death Differ. 2011, 18, 666–677. [Google Scholar] [CrossRef] [PubMed]
- Huynh, A.; DuPage, M.; Priyadharshini, B.; Sage, P.T.; Quiros, J.; Borges, C.M.; Townamchai, N.; Gerriets, V.A.; Rathmell, J.C.; Sharpe, A.H.; et al. Control of PI(3) kinase in Treg cells maintains homeostasis and lineage stability. Nat. Immunol. 2015, 16, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Yang, K.; Guy, C.; Vogel, P.; Neale, G.; Chi, H. Treg cells require the phosphatase PTEN to restrain TH1 and TFH cell responses. Nat. Immunol. 2015, 16, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Richards, J.A.; Gupta, R.; Aung, P.P.; Emley, A.; Kluger, Y.; Dogra, S.K.; Mahalingam, M.; Wajapeyee, N. PTEN functions as a melanoma tumor suppressor by promoting host immune response. Oncogene 2014, 33, 4632–4642. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.R.; Bowen, K.A.; Rychahou, P.G.; Jackson, L.N.; Weiss, H.L.; Lee, E.Y.; Townsend, C.M., Jr.; Evers, B.M. VEGFR-2 expression in carcinoid cancer cells and its role in tumor growth and metastasis. Int. J. Cancer. 2011, 128, 1045–1056. [Google Scholar] [CrossRef] [PubMed]
- Lazar, D.F.; Saltiel, A.R. Lipid phosphatases as drug discovery targets for type 2 diabetes. Nat. Rev. Drug Discov. 2006, 5, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Li, A.; Qiu, M.; Zhou, H.; Wang, T.; Guo, W. PTEN, Insulin Resistance and Cancer. Curr. Pharm. Des. 2017, 23, 3667–3676. [Google Scholar] [CrossRef] [PubMed]
- Kurlawalla-Martinez, C.; Stiles, B.; Wang, Y.; Devaskar, S.U.; Kahn, B.B.; Wu, H. Insulin hypersensitivity and resistance to streptozotocin-induced diabetes in mice lacking PTEN in adipose tissue. Mol. Cell. Biol. 2005, 25, 2498–2510. [Google Scholar] [CrossRef] [PubMed]
- Stiles, B.L.; Kuralwalla-Martinez, C.; Guo, W.; Gregorian, C.; Wang, Y.; Tian, J.; Magnuson, M.A.; Wu, H. Selective deletion of Pten in pancreatic beta cells leads to increased islet mass and resistance to STZ-induced diabetes. Mol. Cell. Biol. 2006, 26, 2772–2781. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, Y.; Lu, S.Y.; Nguyen, K.T.; Schroer, S.A.; Suzuki, A.; Mak, T.W.; Gaisano, H.; Woo, M. Deletion of Pten in pancreatic ss-cells protects against deficient ss-cell mass and function in mouse models of type 2 diabetes. Diabetes 2010, 59, 3117–3126. [Google Scholar] [CrossRef] [PubMed]
- Wijesekara, N.; Konrad, D.; Eweida, M.; Jefferies, C.; Liadis, N.; Giacca, A.; Crackower, M.; Suzuki, A.; Mak, T.W.; Kahn, C.R.; et al. Muscle-specific Pten deletion protects against insulin resistance and diabetes. Mol. Cell. Biol. 2005, 25, 1135–1145. [Google Scholar] [CrossRef] [PubMed]
- Butler, M.G.; Dasouki, M.J.; Zhou, X.P.; Talebizadeh, Z.; Brown, M.; Takahashi, T.N.; Miles, J.H.; Wang, C.H.; Stratton, R.; Pilarski, R.; et al. Subset of individuals with autism spectrum disorders and extreme macrocephaly associated with germline PTEN tumour suppressor gene mutations. J. Med. Genet. 2005, 42, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Lo, Y.T.; Tzeng, T.F.; Liu, I.M. Role of tumor suppressor PTEN in tumor necrosis factor alpha-induced inhibition of insulin signaling in murine skeletal muscle C2C12 cells. Horm. Metab. Res. 2007, 39, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Qin, G.; Fan, X.; Chen, L.; Shen, C.; Gui, B.; Tan, G.; Zhou, J. Preventive effects of AdR-siPTEN through the regulation of NMDA receptor NR2B subunit in trigeminal ganglia of migraine rats. Neurol. Res. 2012, 34, 998–1006. [Google Scholar] [CrossRef] [PubMed]
- Qin, G.; Xie, J.; Chen, L.; Wu, B.; Gui, B.; Zhou, J. PTEN inhibition preserves trigeminal nucleus caudalis neuron activation through tyrosine phosphorylation of the NR2B subunit at Tyr1472 of the NMDA receptor in a rat model of recurrent migraine. Neurol. Res. 2016, 38, 320–326. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.Y.; Sung, C.S.; Chen, W.F.; Chen, C.H.; Feng, C.W.; Yang, S.N.; Hung, H.C.; Chen, N.F.; Lin, P.R.; Chen, S.C.; et al. Involvement of phosphatase and tensin homolog deleted from chromosome 10 in rodent model of neuropathic pain. J. Neuroinflamm. 2015, 12, 59. [Google Scholar] [CrossRef] [PubMed]
- Pouliot, P.; Camateros, P.; Radzioch, D.; Lambrecht, B.N.; Olivier, M. Protein tyrosine phosphatases regulate asthma development in a murine asthma model. J. Immunol. 2009, 182, 1334–1340. [Google Scholar] [CrossRef] [PubMed]
- Kwak, Y.G.; Song, C.H.; Yi, H.K.; Hwang, P.H.; Kim, J.S.; Lee, K.S.; Lee, Y.C. Involvement of PTEN in airway hyperresponsiveness and inflammation in bronchial asthma. J. Clin. Investig. 2003, 111, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- Lan, H.; Zhong, H.; Gao, Y.; Ren, D.; Chen, L.; Zhang, D.; Lai, W.; Xu, J.; Luo, Y. The PTEN tumor suppressor inhibits human airway smooth muscle cell migration. Int. J. Mol. Med. 2010, 26, 893–899. [Google Scholar] [PubMed]
- Lee, K.S.; Kim, S.R.; Park, S.J.; Lee, H.K.; Park, H.S.; Min, K.H.; Jin, S.M.; Lee, Y.C. Phosphatase and tensin homolog deleted on chromosome 10 (PTEN) reduces vascular endothelial growth factor expression in allergen-induced airway inflammation. Mol. Pharmacol. 2006, 69, 1829–1839. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.S.; Park, S.J.; Hwang, P.H.; Yi, H.K.; Song, C.H.; Chai, O.H.; Kim, J.S.; Lee, M.K.; Lee, Y.C. PPAR-gamma modulates allergic inflammation through up-regulation of PTEN. FASEB J. 2005, 19, 1033–1035. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Tang, J.; Cai, Z.; Yang, W.; Zhang, L.; Chen, Q.; Zhang, L.; Wang, X. A new pathway of glucocorticoid action for asthma treatment through the regulation of PTEN expression. Respir. Res. 2011, 12, 47. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.Q.; Ouyang, M.; Brandmaier, A.; Hao, H.; Shen, W.H. PTEN in the maintenance of genome integrity: From DNA replication to chromosome segregation. BioEssays 2017, 39. [Google Scholar] [CrossRef] [PubMed]
| Substrate | Functional Outcome of Dephosphorylation | References |
|---|---|---|
| Phosphatydilinositol/Inositol substrates (3′ specificity) | ||
| PI(3,4,5)P3, PI(3,4)P2 2 | Inhibition of PI3K/AKT/mTOR pathway | [6,7,34] |
| I(1,3,4,5,6)P5 3 | Inhibition of cell proliferation | [11,12,13] |
| Protein substrate 4 | ||
| AKT (Thr) | AKT inactivation | [35] |
| b-catenin (Tyr) | Maintenance of cell-cell adhesion | [36] |
| Cofilin-1 (Ser) | Activation of actin depolymerization | [37] |
| CREB (Ser) | CREB inactivation | [38] |
| Drebrin (Ser) | Changes in neuronal actin dynamics | [39] |
| EphR (Tyr) (C. elegans) | EphR inactivation | [40] |
| FAK (Tyr) | Inhibition of directional cell migration | [41] |
| FYN (Tyr) | FYN inactivation | [42] |
| IR (Tyr) | IR inactivation | [43] |
| IRF3 (Ser) | Inhibition of IRF3 nuclear import | [44] |
| IRS1 (Tyr) | Inhibition of insulin and IGF signaling | [45] |
| MCM2 (Ser) | Inhibition of replication fork progression | [46] |
| PDGFR (Tyr) | PDGFR inactivation | [47] |
| PTEN (Ser, Thr) | PTEN conformational opening and activation | [14,15,48] |
| PTK6 (Tyr) | PTK6 inactivation | [49] |
| p85b (Tyr, Thr) | AKT inactivation | [50] |
| Plk1 (Thr) | Plk1 inactivation | [51] |
| Rab7 (Tyr, Ser) | Promotion of late endosomal maturation | [52] |
| SHC (Tyr) | Inhibition of random cell migration | [53] |
| SRC (Tyr) | SRC inactivation | [54] |
| 5-HT2cR | 5-HT2cR inactivation | [55] |
| Inhibitor | Chemical Name | IC50 | Chemical Structure | Reference |
|---|---|---|---|---|
| bpV(phen) | bisperoxovanadium 1,10-phenantroline | 38 nM |  | [67] |
| bpV(pic) | bisperoxovanadium 5-hydroxipyridine | 31 nM | 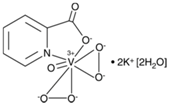 | [67] |
| bpV(HOpic) | bisperoxovanadium 5-hydroxipyridine-2-carboxylic acid | 14 nM |  | [67] |
| bpV(pis) | bisperoxovanadium pyridin-2-squaramide | 39 nM | 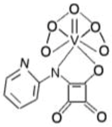 | [68] |
| VO-OHpic | hydroxyl(oxo)vanadium 3-hydroxypiridine-2-carboxylic acid | 35 nM |  | [66] |
| SF1670 | N-(9,10-dioxo-9,10-dihydrophenanthren-2-yl) pivalamide | 2 μM | 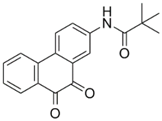 | [70] |
| Disease/Condition | Experimental Model | Inhibitor | Reference |
|---|---|---|---|
| Nerve regeneration and neurosurvival-related diseases | |||
| Central nervous system (CNS) and peripheral nervous system (PNS) injury | - Vertebral arteries occlusion ischemia rat model | - bpV(pic) | - [76] |
| - 6-hydroxydopamine midbrain injection rat model of dopaminergic neurons degeneration | - bpV(phen) | - [77] | |
| - Thoracic spinal cord contusion injury rat model | - bpV(phen) | - [78] | |
| - Carotid artery ligation ischemia postnatal rat model | - bpV(pic) | - [79] | |
| - Rat sensory neurons; sciatic nerve cut rat model | - bpV(pic) | - [80] | |
| - Middle cerebral artery occlusion (MCAO) ischemia rat model | - bpV(pic) | - [81] | |
| - Hemicontusive spinal cord injury (SCI) rat model | - bpV(pic) | - [82,83,84] | |
| - Cortical neuron oxygen/glucose deprivation (OGD)-rat model cultures | - bpV(pic) | - [85] | |
| - Fluid percussion-traumatic brain injury rat model | - bpV(pic) | - [86] | |
| - MCAO ischemia rat model | - bpV(HOpic) | - [87] | |
| - Hippocampal neuron culture stretch injury rat model | - bpV(HOpic) | - [88] | |
| - Human embrionic stem cells (hESC)-derived neuronal progenitors | - bpV(pic) | - [89] | |
| - Subarachnoid hemorrhage (SAH)-early brain injury rat model | - bpV(pic) | - [90] | |
| - MCAO ischemia rat model | - bpV(phen) | - [91] | |
| - SH-SY5Y human neuroblastoma cells; cortical neuron OGD rat cultures; MCAO ischemia rat model | - bpV(pis) | - [68] | |
| - Sciatic nerve transection-regeneration type 2 diabetic mouse model | - SF1670 | - [92] | |
| Alzheimer’s disease (AD) | - SH-SY5Y human neuroblastoma cells | - bpV(pic) | - [93] |
| - App/Psen1 Tg mice model of AD | - bpV(HOpic), VO-OHPic | - [94] | |
| - App/Psen1 Tg mice model of AD | - bpV(HOpic) | - [95] | |
| - SH-SY5Y human neuroblastoma cells | - bpV(pic) | - [96] | |
| Multiple sclerosis (MS) | - Rat newborn and human fetal olygodendrocyte (OLG) progenitors; rat dorsal root ganglion neuron cultures | - bpV(phen) | - [97] |
| Cognitive dysfunction associated to postnatal anesthesia exposure | - Neonatal propofol-exposure mice model | - bpV(phen) | - [98] |
| - Rat hippocampal neural precursor cells | - bpV(phen) | - [99] | |
| - Postnatal isoflurane-exposure rat model | - bpV(pic) | - [100] | |
| Epilepsy | - Temporal lobe epilepsy mouse model | - bpV(pic) | - [101] |
| Pneumococcal meningitis (PM)-induced neuronal death | - Infant rat model of PM | - bpV(pic) | - [102] |
| Ischemia/reperfusion (I/R) tissue injury | |||
| Ischemia/reperfusion (I/R) cardiac injury; myocardial infarct | - Rat cardiomyocytes; carotid artery occlusion I/R rat model | - bpV(HOpic) | - [103] |
| - Isolated mouse hearts; carotid artery occlusion I/R mouse model | - VO-OHpic | - [104] | |
| - MCAO ischemia mouse model | - bpV(phen) | - [105] | |
| - Mouse cardiomyocytes | - VO-OHpic | - [106] | |
| - KCl-sudden cardiac arrest-resuscitation mouse model | - VO-OHpic | - [107] | |
| - Streptozotocin-induced type 1 diabetic rats subjected to myocardial I/R | - bpV(HOpic) | - [108] | |
| - I/R and H2O2-induced injury models in H9c2 rat cardiomyoblasts | - bpV(phen), bpV(pic) | - [109] | |
| I/R hepatic injury | - Rat hepatocytes, and liver warm I/R rat models. | - bpV(HOpic) | - [110] |
| Wound repair and tissue maintenance | |||
| Lung epithelium injury; acute lung injury | - Primary human upper airway epithelia; BEAS-2B human bronchial epithelium cells | - bpV(phen), bpV(pic) | - [111,112] |
| - Oleic acid-induced acute lung injury mice model | - bpV(phen) | - [113] | |
| Skin renewal | - Human foreskin-derived precursors | - bpV(pic) | - [114] |
| Corneal wound healing | - Human corneal epithelial (HCE) cells; cornea scratch wound rat model | - bpV(pic) | - [115] |
| Muscle regeneration | - C2C12 mice myoblasts | - bpV(HOpic) | - [116] |
| Retinal detachment (RD) | - Retinotomy-RD rat model | - bpV(pic) | - [117] |
| Vascular endotelial injury associated with hyperglicemia | - Human umbilical vein endothelial cells (HUVECs) | - bpV(phen) | - [118] |
| Response to infection | |||
| Pneumonia/deficit in phagocytic response | - Rat alveolar macrophages (AM) | - bpV(pic) | - [119] |
| - AM from syngeneic bone marrow (BM) transplantation mouse model | - bpV(pic) | - [120] | |
| Neutropenia-related infections | - Peritonitis and neutropenia mouse models | - SF1670 | - [71] |
| Infertility | |||
| In vitro fertilization | - Mouse neonatal ovarian follicles | - bpV(pic) | - [121] |
| - Mouse neonatal ovarian follicles | - bpV(HOpic) | - [122] | |
| - Mouse non-growing oocytes | - bpV(HOpic) | - [123] | |
| - Human ovarian follicles | - bpV(HOpic) | - [124,125] | |
| - Human ovarian follicles | - bpV(HOpic) | - [126] | |
| - Human ovarian cortex | - bpV(pic) | - [127] | |
| Stem cells-based therapy | |||
| Generation of induced pluripotent stem cells (iPSCs) | - Mouse embryonic fibroblasts (MEFs) | - bpV(HOpic) | - [128] |
| Bone regeneration | - Human BM stem cells | - SF1670 | - [129] |
| Cancer-related diseases | |||
| Cancers with PTEN low expression | - MDA PCa-2b human prostate cancer cells | - VO-OHpic | - [130] |
| - Hep3b human hepatocarcinoma cells | - VO-OHpic | - [131] | |
| Cancers under immunotherapy | - Mice with established melanoma and lymphoma tumors | - VO-OHpic | - [132] |
| Carcinoid syndrome | - BON human carcinoid cells; carcinoid syndrome mouse model | - bpV(HOpic) | - [133] |
| Insulin-resistance metabolic diseases | |||
| Type 2 Diabetes | - H-411E rat liver cells TNFα-induced insulin resistance model | - VO-OHpic | - [134] |
| - Glucosamine-induced insulin-resistant rat skeletal muscle cells | - bpV(HOpic) | - [135] | |
| - Mouse mesenteric arteries | - VO-OHpic | - [136] | |
| Pain relief/Antinociception | |||
| Inflammatory pain; chronic migraine | - NMDA-triggered nociceptive rat model | - bpV(phen) | - [137] |
| Antinociception mediated by δ opioid receptors | - PC12 rat pheochromocytoma cells, mouse trigeminal ganglia (TG) neurons | - bpV(phen), bpV(HOpic), SF1670 | - [138] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pulido, R. PTEN Inhibition in Human Disease Therapy. Molecules 2018, 23, 285. https://doi.org/10.3390/molecules23020285
Pulido R. PTEN Inhibition in Human Disease Therapy. Molecules. 2018; 23(2):285. https://doi.org/10.3390/molecules23020285
Chicago/Turabian StylePulido, Rafael. 2018. "PTEN Inhibition in Human Disease Therapy" Molecules 23, no. 2: 285. https://doi.org/10.3390/molecules23020285
APA StylePulido, R. (2018). PTEN Inhibition in Human Disease Therapy. Molecules, 23(2), 285. https://doi.org/10.3390/molecules23020285





