Promising Fungicides from Allelochemicals: Synthesis of Umbelliferone Derivatives and Their Structure–Activity Relationships
Abstract
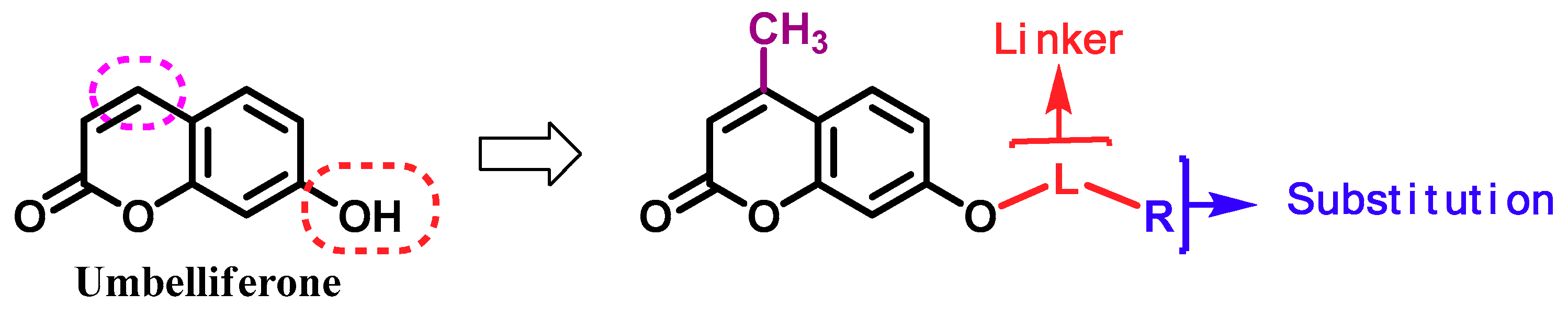
1. Introduction
2. Results and Discussion
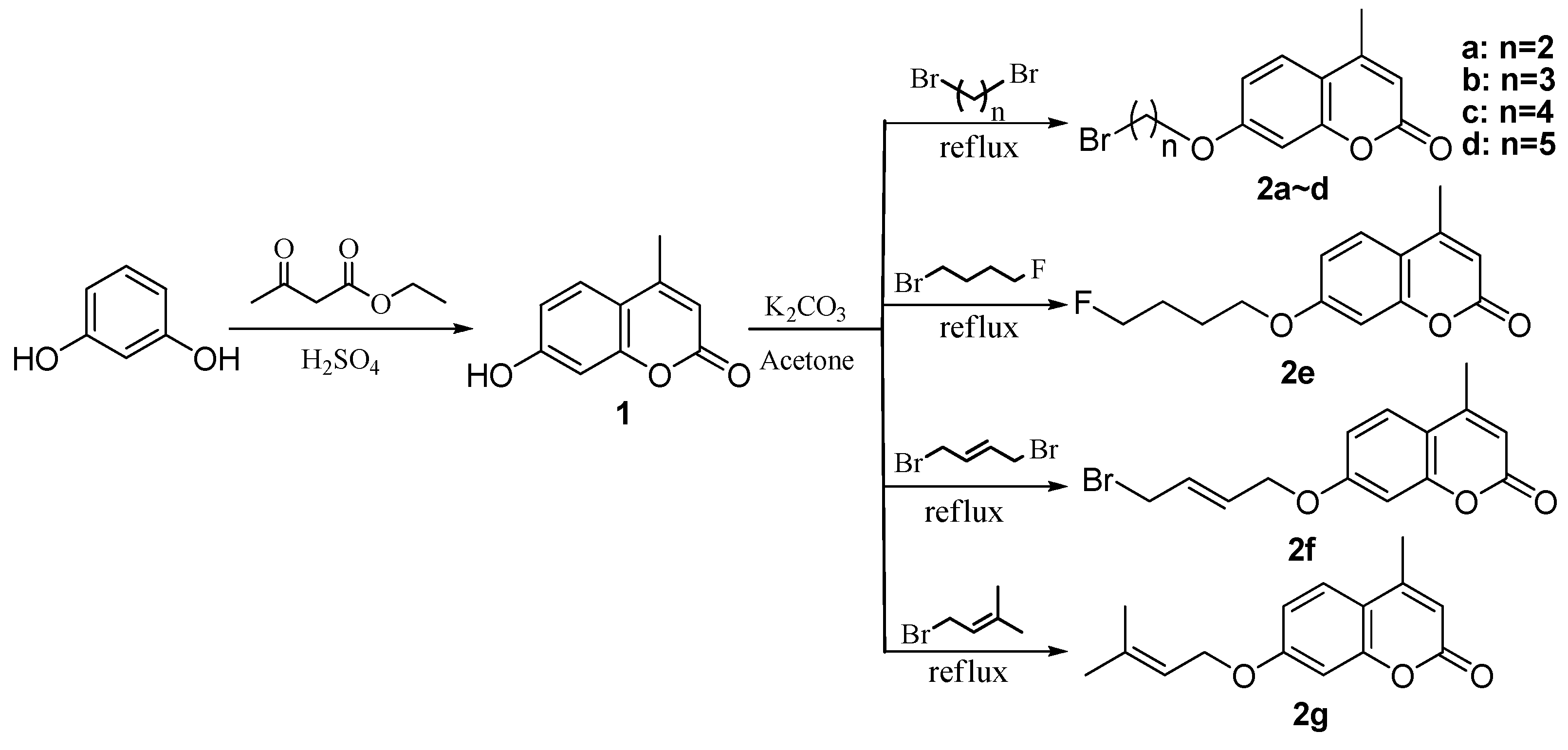
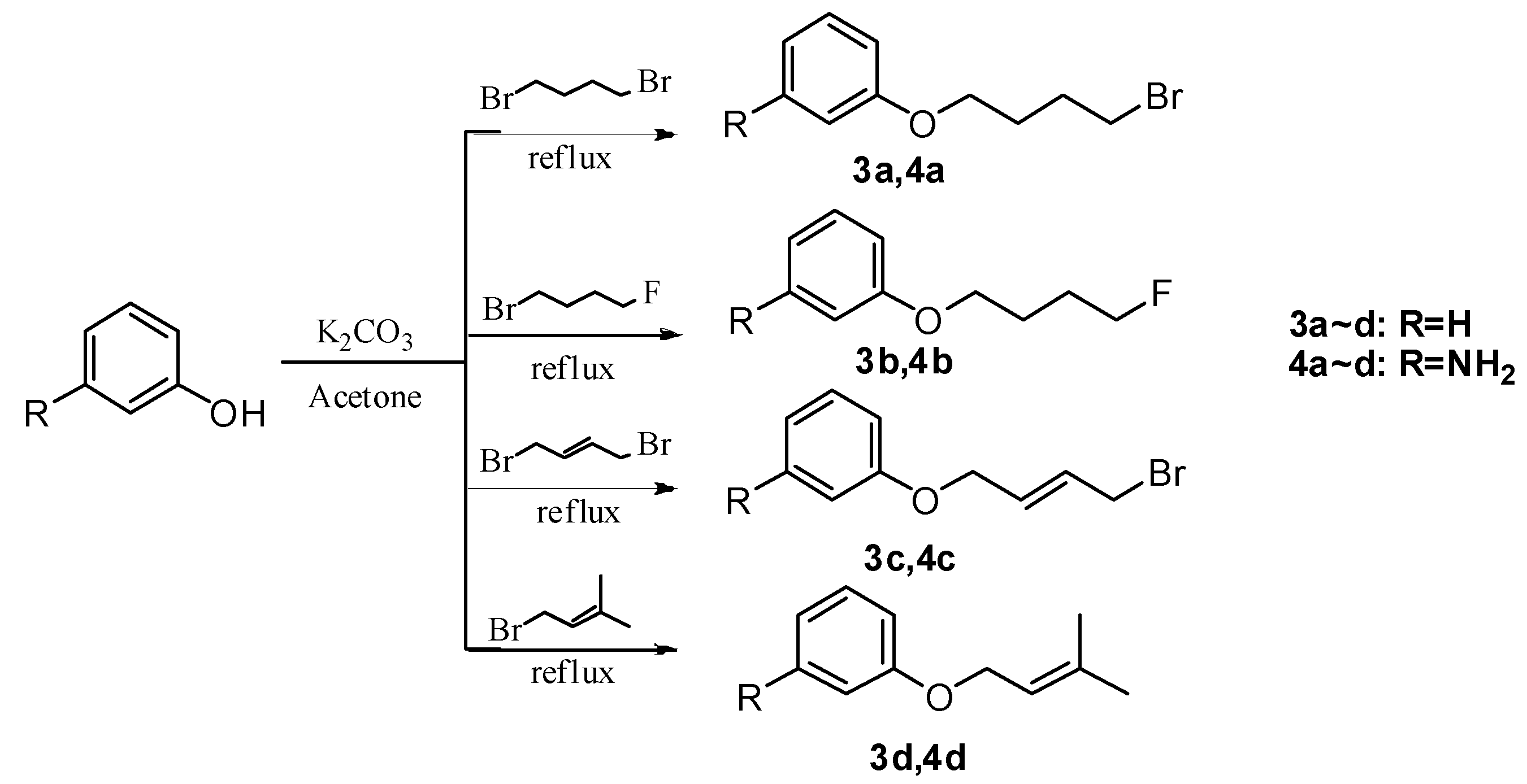
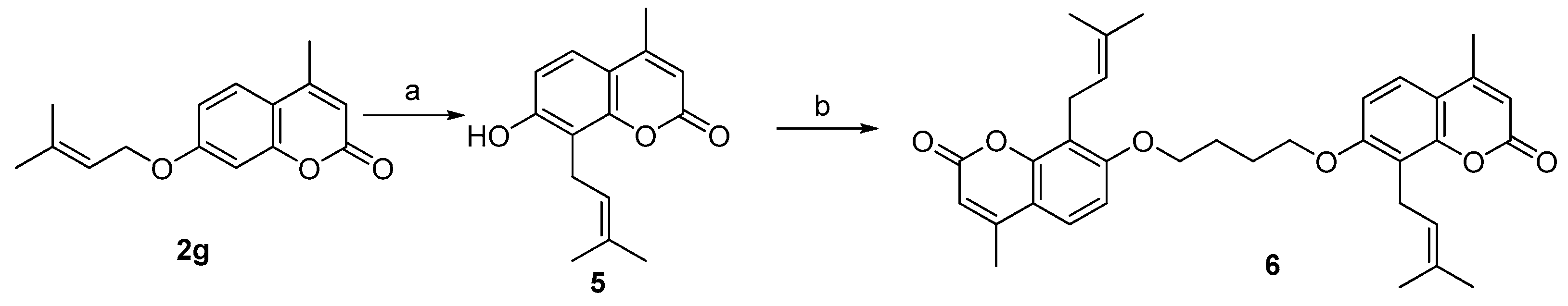
3. Experimental Section
3.1. General Procedure for the Preparation of Compounds 2c–g
3.2. General Procedure for the Preparation of Compounds 3a–d and 4a–d
3.3. General Procedure for the Preparation of Compounds 5 and 6
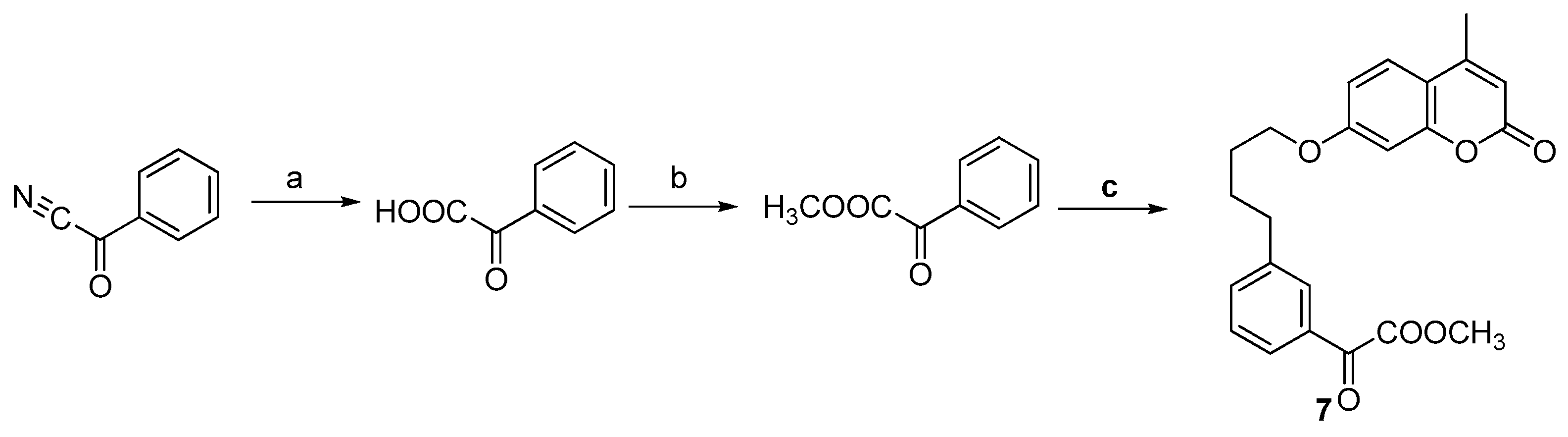
3.4. General Procedure for the Preparation of Compound 7
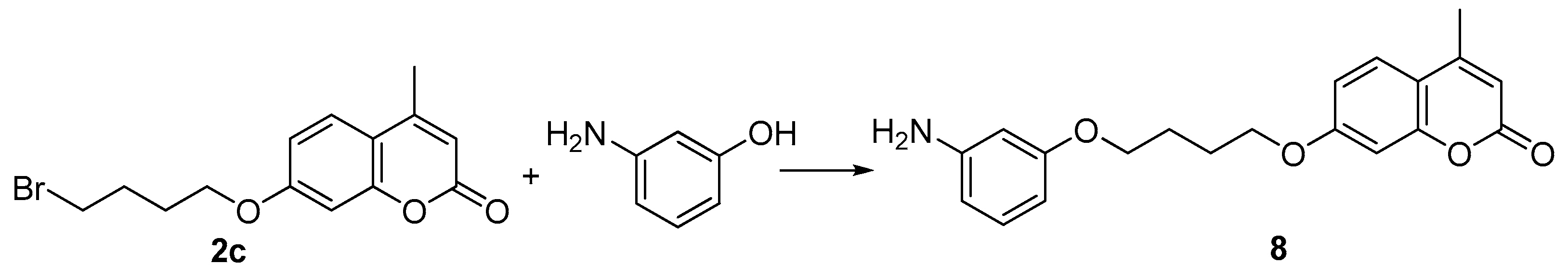
3.5. General Procedure for the Preparation of Compound 8
3.6. General Procedure for the Preparation of Compounds 9 and 10
3.7. Antifungal Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sparks, T.C.; Lorsbach, B.A. Perspectives on the agrochemical industry and agrochemical discovery. Pest. Manag. Sci. 2017, 73, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Bardas, G.A.; Veloukas, T.; Koutita, O.; Karaoglanidis, G.S. Multiple resistance of Botrytis cinerea from kiwifruit to SDHIs, QoIs and fungicides of other chemical groups. Pest. Manag. Sci. 2010, 66, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Halstead, N.T.; Hoover, C.M.; Arakala, A.; Civitello, D.J.; De Leo, G.A.; Gambhir, M. Agrochemicals increase risk of human schistosomiasis by supporting higher densities of intermediate hosts. Nat. Commun. 2018, 9, 837. [Google Scholar] [CrossRef] [PubMed]
- Lamberth, C. Agrochemical lead optimization by scaffold hopping. Pest. Manag. Sci. 2018, 74, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Lahouel, M.; Zini, R.; Zellagui, A.; Rhouati, S.; Carrupt, P.A.; Morin, D. Ferulenol specifically inhibits succinate ubiquinone reductase at the level of the ubiquinone cycle. Biochem. Biophys. Res. Commun. 2007, 355, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Dan, W.J.; Tuong, T.M.L.; Wang, D.C.; Li, D.; Zhang, A.L.; Gao, J.M. Natural products as sources of new fungicides (V), Design and synthesis of acetophenone derivatives against phytopathogenic fungi in vitro and in vivo. Bioorg. Med. Chem. Lett. 2018, 28, 2861–2864. [Google Scholar] [CrossRef] [PubMed]
- Lamberth, C.; Jeanmart, S.; Luksch, T.; Plant, A. Current challenges and trends in the discovery of agrochemicals. Science 2013, 341, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Qasem, J.R. Allelopathy Importance, field application and potential role in pest management. J. Agric. Sci. Technol. 2010, 4, 104–120. [Google Scholar]
- de Oliveira, J.R.F.; de Resende, J.T.V.; Maluf, W.R.; Lucini, T.; de Lima, R.B.; de Lima, I.P.; Nardi, C. Trichomes and Allelochemicals in Tomato Genotypes Have Antagonistic Effects Upon Behavior and Biology of Tetranychus urticae. Front. Plant Sci. 2018, 9, 1132. [Google Scholar] [CrossRef] [PubMed]
- Kong, C.H.; Xu, X.H.; Zhang, M.; Zhang, S.Z. Allelochemical tricin in rice hull and its aurone isomer against rice seedling rot disease. Pest. Manag. Sci. 2010, 66, 1018–1024. [Google Scholar] [CrossRef] [PubMed]
- Nebo, L.; Varela, R.M.; Molinillo, J.M.G.; Sampaio, O.M.; Severino, V.G.P.; Cazal, C.M.; das Graças Fernandes, M.F.; Fernandes, J.B.; Macias, F.A. Phytotoxicity of alkaloids, coumarins and flavonoids isolated from 11 species belonging to the Rutaceae and Meliaceae families. Phytochem. Lett. 2014, 8, 226–232. [Google Scholar] [CrossRef]
- Yan, Z.Q.; Wang, D.D.; Cui, H.Y.; Zhang, D.H.; Sun, Y.H.; Jin, H. Phytotoxicity mechanisms of two coumarin allelochemicals from Stellera chamaejasme in lettuce seedlings. Acta Physiol. Plant. 2016, 38, 248. [Google Scholar] [CrossRef]
- Pan, L.; Li, X.Z.; Yan, Z.Q.; Guo, H.R.; Qin, B. Phytotoxicity of umbelliferone and its analogs, Structure-activity relationships and action mechanisms. Plant Physiol. Biochem. 2015, 97, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Stevens, B.D.; Bungard, C.J.; Nelson, S.G. Strategies for expanding structural diversity available from olefin isomerization-Claisen rearrangement reactions. J. Org. Chem. 2006, 271, 6397–6402. [Google Scholar] [CrossRef] [PubMed]
- Buchman, C.D.; Hurley, T.D. Inhibition of the aldehyde dehydrogenase 1/2 family by psoralen and coumarin derivatives. J. Med. Chem. 2017, 60, 2439–2455. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, S.L.; Xu, X.Q.; Liu, X.; Yu, M.Q.; Zhao, S.; Liu, S.C.; Qiu, Y.L.; Zhang, T.; Liu, B.F.; et al. Synthesis and biological investigation of coumarin piperazine (piperidine) derivatives as potential multireceptor atypical antipsychotics. J. Med. Chem. 2013, 56, 4671–4690. [Google Scholar] [CrossRef] [PubMed]
- Tucaliuc, R.A.; Cotea, V.V.; Niculaua, M.; Tuchilus, C.; Mantu, D.; Mangalagiu, I.I. New pyridazine—Fluorine derivatives, synthesis, chemistry and biological activity. Part II. Eur. J. Med. Chem. 2013, 67, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Berthelot, K.; Estevez, Y.; Quiliano, M.; Baldera-Aguayo, P.A.; Zimic, M.; Pribat, A.; Bakleh, M.E.; Teyssier, E.; Gallusci, P.; Gardrat, C.; et al. HbIDI; SlIDI and EcIDI: A comparative study of isopentenyl diphosphate isomerase activity and structure. Biochimie 2016, 127, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Scholte, A.A.; Eubanks, L.M.; Poulter, C.D.; Vederas, J.C. Synthesis and biological activity of isopentenyl diphosphate analogues. Bioorg. Med. Chem. 2004, 12, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.L.; Wang, F.; Zhang, M.Z.; Liu, Z.M.; Huang, W.; Yang, G.F. Synthesis, fungicidal, and insecticidal activities of beta-methoxyacrylate-containing N-acetyl pyrazoline derivatives. J. Agric. Food Chem. 2008, 56, 10767–10773. [Google Scholar] [CrossRef] [PubMed]
- Herms, S.; Seehaus, K.; Koehle, H.; Conrath, U. Astrobilurin fungicide enhances the resistance of tobacco against tobacco mosaic virus and Pseudomonas syringae pv tabaci. Plant Physiol. 2002, 130, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, H.; Nishida, H.; Imamura, N.; Tomoda, H.; Omura, S.; Bordner, J. The structures of atpenins A4, A5 and B, new antifungal antibiotics produced by Penicillium sp. J. Antibiot. 1990, 43, 1553–1558. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.H.; Ren, Z.L.; Liu, P.; Li, B.X.; Li, Q.S.; Chu, M.J.; Cao, H.Q. Design, synthesis and biological evaluation of novel nicotinamide derivatives bearing a substituted pyrazole moiety as potential SDH inhibitors. Pest Manage. Sci. 2017, 73, 1585–1592. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–10 are available from the authors. |

| Compd. | The Inhibition Rate (%; mean ± SD; N = 3) | ||
|---|---|---|---|
| A. Alternata | B. Cinerea | F. Oxysporum | |
| 1 | 38.5 ± 4.38 | 25.8 ± 3.8 | 41.2 ± 2.5 |
| 2a | 38.1 ± 5.9 | 39.6 ± 6.2 | 33.2 ± 4.4 |
| 2b | 42.9 ± 6.7 | 36.3 ± 5.8 | 28.6 ± 3.4 |
| 2c | 70.6 ± 5.5 | 68.3 ± 4.6 | 60.4 ± 5.3 |
| 2d | 40.3 ± 3.7 | 46.2 ± 5.8 | 35.5 ± 7.2 |
| 2e | 63.7 ± 3.9 | 72.9 ± 3.5 | 62.8 ± 5.6 |
| 2f | 81.5 ± 3.7 | 90.99 ± 2.3 | 84.8 ± 3.2 |
| 2g | 48.4 ± 4.0 | 70.6 ± 5.2 | 53.5 ± 4.2 |
| 3a | 49.6 ± 3.6 | 41.2 ± 5.4 | 50.2 ± 6.3 |
| 3b | 3.8 ± 2.8 | 10.2 ± 2.6 | 8.4 ± 4.1 |
| 3c | 58.2 ± 3.6 | 63.0 ± 7.0 | 61.3 ± 3.8 |
| 3d | 29.6 ± 5.3 | 32.5 ± 6.2 | 26.2 ± 4.9 |
| 4a | 30.5 ± 5.2 | 32.1 ± 4.5 | 28.4 ± 5.8 |
| 4b | 35.3 ± 5.6 | 46.2 ± 3.7 | 33.2 ± 3.1 |
| 4c | 12.6 ± 2.6 | 15..0 ± 6.4 | 13.7 ± 4.2 |
| 4d | 10.2 ± 3.5 | 12.6 ± 4.5 | 9.8 ± 2.6 |
| 5 | 22.4 ± 4.7 | 26.4 ± 2.1 | 32.6 ± 5.6 |
| 6 | 27.6 ± 8.3 | 35.93 ± 1.73 | 29.8 ± 3.8 |
| 7 | 20.8 ± 7.7 | 21.9 ± 3.0 | 18.7 ± 4.1 |
| 8 | 26.6 ± 3.9 | 18.9 ± 8.5 | 25.3 ± 5.2 |
| 9 | 58.5 ± 3.4 | 70.9 ± 1.7 | 77.5 ± 3.4 |
| 10 | 77.3 ± 5.5 | 84.4 ± 3.5 | 80.5 ± 5.7 |
| DMSO (1%) | 0 | 0 | 0 |
| carbendazim | 90.7 ± 8.18 | 94.3 ± 6.4 | 91.4 ± 5.4 |
| Compound | Log P | EC50 (95% CI μmol/L) | ||
|---|---|---|---|---|
| A. Alternata | B. Cinerea | F. Oxysporum | ||
| 1 | 1.61 | 922.4 (827.6–1098.9) | 1037.1 (955.3–1201.1) | 1165.9 (1076.2–1268.1) |
| 2c | 3.26 | 193.5 (145.6–258.7) | 221.1 (137.2–270.9) | 245.2 (180.9–298.2) |
| 2e | 2.62 | 381.6 (282.1–466.7) | 360.4 (274.1–462.7) | 405.9 (311.3–505.9) |
| 2f | 3.22 | 50.5 (28.5–82.2) | 30.7 (15.5–47.5) | 58.9 (30.4–89.3) |
| 2g | 3.11 | 232.1 (158.8–336.9) | 280.8(206.7–350.4) | 207.1 (149.8–279.2) |
| 9 | 2.87 | 185.8 (117.6–254.0) | 130.7 (76.0–195.7) | 182.9 (145.9–248.2) |
| 10 | 3.23 | 105.1 (69.5–176.2) | 66.6 (50.7–115.1) | 60.8 (34.6–120.6) |
| Carbendazim | 1.29 | 18.8 (6.8–30.0) | 20.6 (7.8–32.4) | 33.5 (14.6–49.7) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, L.; Lei, D.; Jin, L.; He, Y.; Yang, Q. Promising Fungicides from Allelochemicals: Synthesis of Umbelliferone Derivatives and Their Structure–Activity Relationships. Molecules 2018, 23, 3002. https://doi.org/10.3390/molecules23113002
Pan L, Lei D, Jin L, He Y, Yang Q. Promising Fungicides from Allelochemicals: Synthesis of Umbelliferone Derivatives and Their Structure–Activity Relationships. Molecules. 2018; 23(11):3002. https://doi.org/10.3390/molecules23113002
Chicago/Turabian StylePan, Le, Dongyu Lei, Lu Jin, Yuan He, and Qingqing Yang. 2018. "Promising Fungicides from Allelochemicals: Synthesis of Umbelliferone Derivatives and Their Structure–Activity Relationships" Molecules 23, no. 11: 3002. https://doi.org/10.3390/molecules23113002
APA StylePan, L., Lei, D., Jin, L., He, Y., & Yang, Q. (2018). Promising Fungicides from Allelochemicals: Synthesis of Umbelliferone Derivatives and Their Structure–Activity Relationships. Molecules, 23(11), 3002. https://doi.org/10.3390/molecules23113002







