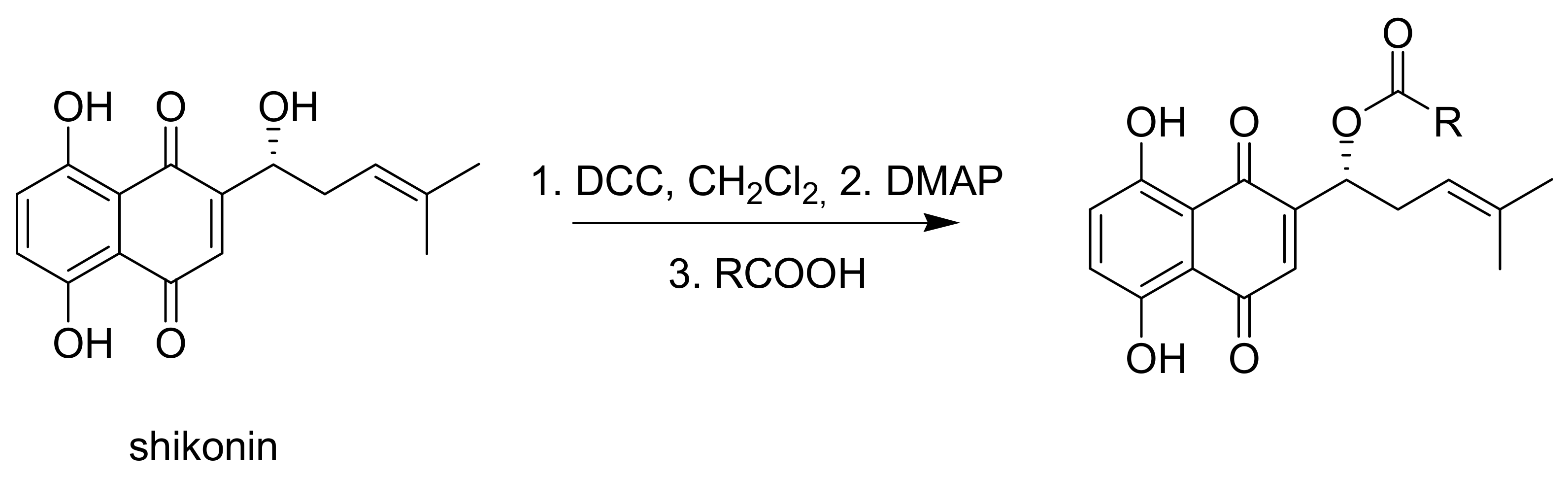
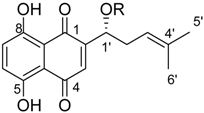
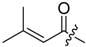
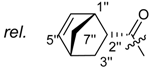
3.1.3. General Procedure for the Acylation of Shikonin
A solution of shikonin in abs. CH2Cl2 (0.1 mmol/5 mL) was cooled to 0 °C under argon atmosphere and DCC was added. DMAP was added after stirring for 15 min. After further stirring for 15 min, the corresponding acid was added and stirring was continued for 14 h to 5 days with slowly warming up to room temperature. After addition of 1 mL cyclohexane/0.1 mmol shikonin, the mixture was concentrated under reduced pressure at room temperature to approx. 0.5 mL/0.1 mmol shikonin. The mixture was filtered over 3 mm silica and 2 mm celite® (eluent: petroleum ether/CH2Cl2 = 1:0 to 1:2). Product containing fractions were evaporated and submitted to flash CC and/or repeated PTLC (cyclohexane/CH2Cl2 mixtures). Due to the rapid decomposition of raw acylshikonin mixtures on evaporation to higher concentrations (c > approx. 0.2 M acylshikonin) and degradation of acylshikonins on prolonged contact with silica, intermediate solutions were not to be concentrated to dryness and all isolation and purification steps had be performed at a good pace.
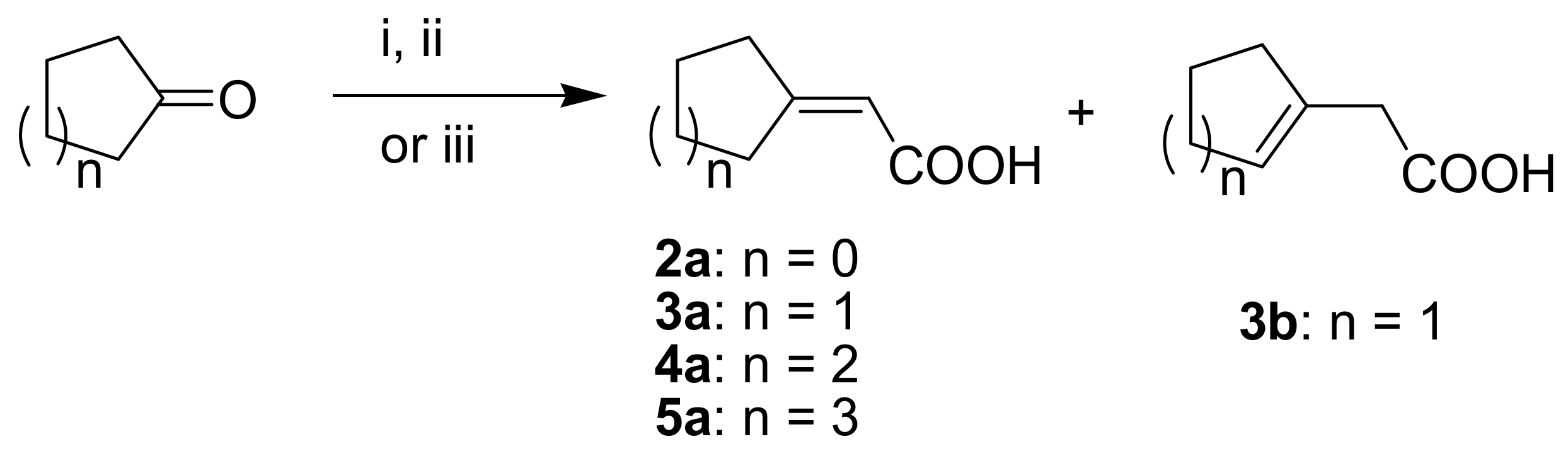
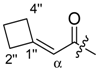
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl 2-cyclobutylideneacetate (2): 0.1 mmol Shikonin, 0.20 mmol DCC, 30 µmol DMAP, and 0.1 mmol 2-cyclobutylideneacetic acid (2a); reaction time 5 days; PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1 and twice with cyclohexane/CH2Cl2 = 1:2) and PTLC on silica with cyclohexane/CH2Cl2 = 2:1 (three times developed), 2, yield: 3%. 2: Rf = 0.31 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2957 (w), 2919 (m), ≈ 2950 (br), 2851 (w), 1716 (m), 1668 (m), 1609 (m), 1568 (m), 1454 (m), 1263 (s), 1202 (s), 1170 (s), 1108 (m), 703 (w) cm−1; 1H-NMR (CDCl3): δ 1.58 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 2.12 (quint, J = 7.9 Hz, 2H, H-3′′), 2.49 (dtm, J = 14.9, 7.4 Hz, 1H, H-2′), 2.62 (dtm, J = 14.9, 5.5 Hz, 1H, H-2′), 2.88 (t, J = 7.6 Hz, 2H, H-2′′, H-4′′), 3.14 (qm, J = 7.7 Hz, 2H, H-2′′, H-4′′), 5.14 (tm, J = 7.1 Hz, 1H, H-3’), 5.68 (quint, 1H, J = 2.1 Hz, H-α), 6.02 (dd, J = 7.1, 4.4 Hz, 1H, H-1’), 6.96 (d, J = 0.9 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.44 (s, 1H, C5-OH), 12.59 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 17.6 (C-3′′), 18.0 (C-6’), 25.8 (C-5’), 32.4 (C-2′′*), 32.9 (C-2’), 34.1 C-4′′*), 69.0 (C-1’), 111.6 (C-4a, C-α), 111.8 (C-8a), 118.1 (C-3’), 131.6 (C-3), 132.5 (C-6, C-7), 135.9 (C-4’), 148.9 (C-2), 165.1 (COO), 166.2 (C-5), 166.8 (C-8), 169.8 (C-1′′), 177.4 (C-1), 179.0 (C-4); MS (ESI−) m/z (%): 819 (16) [2(M − H) + Cl + Na]−, 785 (37) [2(M − H) + Na]−, 417 and 415 (14) [M − 2H + Cl]−, 382.11 (100) [M]−; 381.21 (53) [M − H]−, 137 (44); [M]− calculated for C22H22O6: 382.1416.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl 2-cyclopentylideneacetate (3): 0.1 mmol Shikonin, 0.20 mmol DCC, 25 µmol DMAP, and 0.11 mmol 2-cyclopentylideneacetic acid (3a); reaction time 16 h; PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1 and twice with cyclohexane/CH2Cl2 = 1:2) and PTLC on silica with cyclohexane/CH2Cl2 = 2:1 (four times developed), 3, yield: 5%. 3: Rf = 0.37 (silica, CH2Cl2); IR (ATR): 2959 (w), 2918 (w), ≈2950 (br), 2850 (w), 1717 (m), 1648 (m), 1609 (vs), 1569 (m), 1453 (m), 1230 (m), 1188 (vs), 1114 (s) cm−1; 1H-NMR (CDCl3): 1.58 (s, 3H, H-6’), 1.68 (m, 2H, H-4′′), 1.69 (s, 3H, H-5’), 1.75 (m, 2H, H-3′′), 2.48 (m, 3H, H-2’ and H-2′′), 2.63 (dtm, J = 15.3, 5.4 Hz, 1H, H-2’), 2.75 (m, 2H, H-5′′), 5.15 (tm, J = 7.2 Hz, 1H, H-3’), 5.90 (quint, 1H, J = 1.9 Hz, H-α), 6.02 (dd, J = 7.4, 4.7 Hz, 1H, H-1’), 6.97 (d, J = 0.8 Hz, 1H, H-3), 7.18 (s, 2H, H-6 and H-7), 12.43 (s, 1H, C5-OH), 12.60 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6’), 25.5 (C-4′′), 25.8 (C-5’), 26.4 (C-3′′), 32.9, 33.0 (C-2’, C-2′′), 36.2 (C-5′′), 68.7 (C-1’), 110.9 (C-α), 111.7 (C-4a), 111.8 (C-8a), 118.1 (C-3’), 131.6 (C-3), 132.4 (C-7), 133.5 (C-6), 135.8 (C-4’), 149.2 (C-2), 165.5 (COO), 166.1 (C-5), 166.7 (C-8), 171.5 (C-1′′), 177.7 (C-1), 179.1 (C-4); MS (ESI−) m/z (%): 813 (10) [2(M − H)+Na]−, 395.30 (100) [M − H]−; [M − H]− calculated for C23H24O6: 395.1495.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl 2-cyclohexylideneacetate (4): 0.1 mmol Shikonin, 0.20 mmol DCC, 25 µmol DMAP, and 0.11 mmol 2-cyclohexylideneacetic acid (4a); reaction time 16 h; Flash-CC on silica (3 g) with petroleum ether/CH2Cl2 = 0:1 to 1:0) and PTLC on silica each developed twice with cyclohexane/CH2Cl2 = 2:1 and twice with cyclohexane/CH2Cl2 = 1:2); 4, yield: 13%. 4: Rf = 0.31 (silica, CH2Cl2); IR (ATR): 2919 (m), ≈2950 (br) (OH), 2851 (w), 1720 (m) (C=O), 1643 (m), 1610 (s), 1570 (m), 1451 (m), 1266 (m), 1233 (m), 1201 (s) (COC), 1147 (s), 777 (w) cm−1; 1H-NMR (CDCl3): 1.58 (s, 3H, H-6’), 1.59-1.70 (m, 6H, H-3′′, H-4′′, H-5′′), 1.69 (s, 3H, H-5’), 1.75 (m, 2H, H-3′′), 2.23 (t, J = 6.5 Hz, 2H, H-2′′), 2.48 (dt, J = 15.0, 7.3 Hz, 1H, H-2’), 2.62 (dtm, J = 15.2, 5.4 Hz, 1H, H-2’), 2.75 (t, J = 5.5 Hz, 2H, H-6′′), 5.15 (tm, J = 7.2 Hz, 1H, H-3’), 5.70 (s, 1H, H-α), 6.02 (dd, J = 7.4, 4.7 Hz, 1H, H-1’), 6.98 (d, J = 0.8 Hz, 1H, H-3), 7.18 (s, 2H, H-6 and H-7), 12.44 (s, 1H, C5-OH), 12.60 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6’), 25.8 (C-5’), 26.2 (C-4′′), 27.8 (C-5′′), 28.6 (C-3′′), 30.0 (C-6′′), 32.9 (C-2’), 38.1 (C-2′′), 68.6 (C-1’), 111.6 (C-4a), 111.9 (C-8a), 112.1 (C-α), 118.0 (C-3’), 131.7 (C-3), 132.4 (C-7), 132.6 (C-6), 135.9 (C-4’), 149.1 (C-2), 165.4 (COO), 166.0 (C-1′′), 166.3 (C-5), 166.8 (C-8), 177.6 (C-1), 179.1 (C-4); MS (ESI−) m/z (%): 841 (11) [2(M − H) + Na]−, 410.23 (47) [M]−, 409.23 (100) [M − H]−; [M]− calculated for C24H26O6: 410.1729.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl 2-cycloheptylideneacetate (5): 0.1 mmol Shikonin, 0.20 mmol DCC, 25 µmol DMAP, and 0.11 mmol 2-cycloheptylideneacetic acid (5a); reaction time 16 h; PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1 and twice with cyclohexane/CH2Cl2 = 1:2) and PTLC on silica with cyclohexane/CH2Cl2 = 2:1 (four times developed), 5, yield: 10%. 5: Rf = 0.40 (silica, CH2Cl2); IR (ATR): 2918 (m), ≈2950 (br), 2850 (w), 1717 (m), 1609 (s), 1569 (m), 1560 (m), 1453 (m), 1201 (m), 1136 (s), 779 (w) cm−1; 1H-NMR (CDCl3): 1.52–1.55 (m, 2H, H-4′′, H-5′′), 1.55 (s, 3H, H-6′), 1.62-1.69 (m, 2H, H-3′′, H-6′′), 1.69 (s, 3H, H-5′), 2.42 (m, 2H, H-2′′), 2.49 (dtm, J = 14.8, 7.3 Hz, 1H, H-2′), 2.61 (dtm, J = 14.9, 5.0 Hz, 1H, H-2′), 2.85 (m, 2H, H-7′′), 5.15 (tm, J = 7.4 Hz, 1H, H-3′), 5.77 (s, 1H, H-α), 6.01 (ddd, J = 7.2, 4.6, 0.8 Hz, 1H, H-1′), 6.98 (d, J = 0.8 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.44 (s, 1H, C5-OH), 12.60 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 25.8 (C-5′), 26.6 (C-6′′), 27.9 (C-3′′), 29.0 (C-4′′), 29.8 (C-5′′), 32.3 (C-7′′), 33.0 (C-2′), 39.2 (C-2′′), 68.5 (C-1′), 111.6 (C-4a), 111.9 (C-8a), 114.7 (C-α), 118.1 (C-3′), 131.7 (C-3), 132.4 (C-7), 133.5 (C-6), 135.8 (C-4′), 149.2 (C-2), 165.2 (COO), 166.1 (C-5), 166.7 (C-8), 169.2 (C-1′′), 177.7 (C-1), 179.2 (C-4); MS (ESI−) m/z (%): 869 (15) [2(M − H) + Na]−, 424.25 (45) [M]−, 423.22 (100) [M − H]− 159 (31), 137 (39); [M]− calculated for C25H28O6: 424.1886.
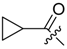
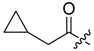
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclopropylacetate (6): 0.2 mmol Shikonin, 0.4 mmol DCC, 80 µmol DMAP, and 0.25 mmol cyclopropylacetic acid (6a); reaction time 16 h; CC on silica (7 g) with cyclohexane/CH2Cl2 = 2:1 to 1:4; 6, yield 26%. 6: Rf = 0.29 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 3080 (w), 2972 (w), ≈2950 (br), 2916 (w), 2857 (w), 1742 (s), 1610 (s), 1570 (m), 1454 (m), 1231 (m), 1204 (s), 784 (m) cm−1; 1H-NMR (CDCl3): 0.17–0.24 (m, 2H, H-2′′, H-3′′), 0.56–0.63 (m, 2H, H-2′′, H-3′′), 1.02–1.13 (m, 1H, H-1′′), 1.58 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 2.23–2.37 (m, 2H, H-α), 2.49 (dtm, J = 15.0, 7.5 Hz, 1H, H-2′), 2.63 (dtm, J = 15.0, 5.5 Hz, 1H, H-2′), 5.14 (tm, J = 7.1 Hz, 1H, H-3′), 6.05 (ddd, J = 7.3, 4.5, 1.0 Hz, 1H, H-1′), 7.01 (d, J = 1.0 Hz, 1H, H-3), 7.19 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.59 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 4.4, 4.5 (C-2′′, C-3′′), 7.0 (C-1′′), 18.0 (C-6’), 25.8 (C-5’), 32.9 (C-2’), 39.5 (C-α), 69.4 (C-1’), 111.6 (C-4a), 111.8 (C-8a), 117.8 (C-3’), 131.5 (C-3), 132.7 (C-7), 132.8 (C-6), 136.0 (C-4’), 148.4 (C-2), 166.8 (C-5), 167.4 (C-8), 172.0 (COO), 176.8 (C-1), 178.3 (C-4); MS (ESI−) m/z (%):1165 (5) [3M − 3H + Na + K]−, 1147 (7) [3M − 2H + K]−, 1131 (11) [3M − 2H + Na]−, 761 (1) [2(M − H) + Na]−, 637 (2) [2M − 3H − RCOOH]−, 370.22 (32) [M]−, 369.25 (100) [M − H]−; [M]− calculated for C21H22O6: 370.1416.
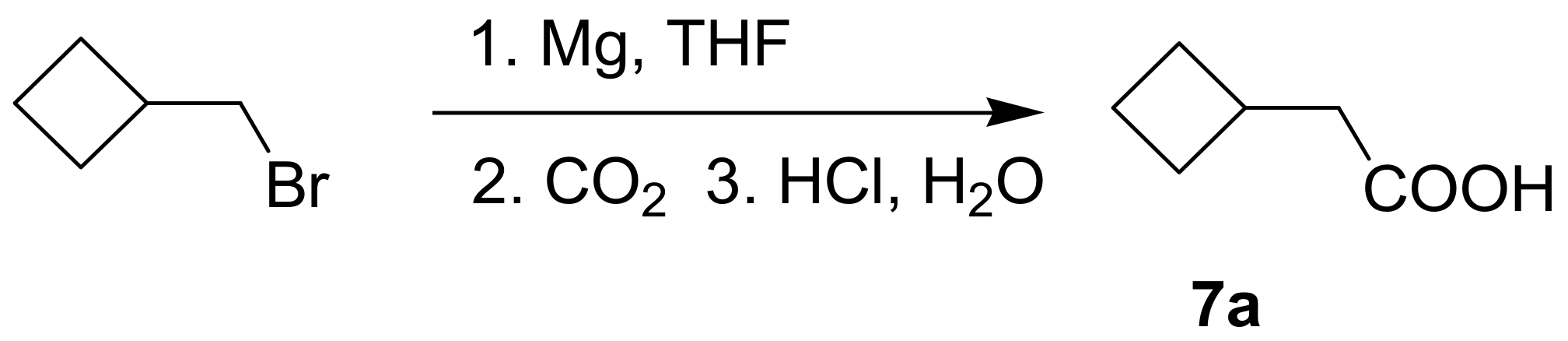
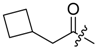
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclobutylacetate (7): 50 µmol Shikonin, 0.15 mmol DCC, 30 µmol DMAP, and 53 µmol cyclobutylacetic acid (7a); reaction time 16 h; PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1 and one time with cyclohexane/CH2Cl2 = 1:2) and an additional PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1); 7, yield: 21%. 7: Rf = 0.21 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2959 (w), 2918 (m), ≈2950 (br), 2851 (w), 1737 (m), 1609 (s), 1568 (m), 1453 (m), 1264 (m), 1229 (m), 1202 (s), 1158 (m), 777 (w) cm−1; 1H-NMR (CDCl3): 1.58 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 1.70–1.78 (m, 2H, trans H-2′′, trans H-4′′), 1.81–1.97 (m, 2H, H-3′′), 2.09–2.20 (m, 2H, cis H-2′′, cis H-4′′), 2.46 (dtm, J = 15.0, 7.5 Hz, 1H, H-2′), 2.49 (d, J = 7.5 Hz, 2H, H-α), 2.60 (dtm, J = 14.7, 5.9 Hz, 1H, H-2′), 2.71 (quint, J = 7.8 Hz, 1H, H-1′′), 5.11 (tm, J = 7.3 Hz, 1H, H-3’), 6.00 (ddd, J = 7.7, 4.2, 0.9 Hz, 1H, H-1’), 6.96 (d, J = 1.0 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.42 (s, 1H, C5-OH), 12.57 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6’), 18.5 (C-3′′), 25.8 (C-5’), 28.2, 28.3 (C-2′′, C-4′′), 32.2 (C-1′′), 32.9 (C-2’), 41.2 (C-α), 69.2 (C-1’), 111.6 (C-4a), 111.8 (C-8a), 117.8 (C-3’), 131.4 (C-3), 132.7 (C-7), 132.9 (C-6), 136.0 (C-4’), 148.5 (C-2), 166.9 (C-5), 167.4 (C-8), 171.5 (COO), 176.8 (C-1), 178.3 (C-4); MS (ESI−) m/z (%): 789.37 (8) [2(M − H) + Na]−, 384.21 [M]− (52), 383.26 (100) [M − H]−; [M]− calculated for C22H24O6: 384.1573.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclopentylacetate (8): 0.1 mmol Shikonin, 0.15 mmol DCC, 25 µmol DMAP, and 0.1 mmol cyclopentylacetic acid (8a); reaction time 17 h; CC on silica (7 g) with cyclohexane/CH2Cl2 = 2:5 and PTLC on silica with cyclohexane/CH2Cl2 = 2:5 (three times developed); 8, yield 10%. 8: Rf = 0.26 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2951 (m, br), 2866 (w), 1738 (s), 1608 (s), 1569 (m), 1452 (m), 1230 (s), 1200 (s), 1111 (s), 777 (m) cm−1; 1H-NMR (CDCl3): 1.12–1.23 (m, 2H, H-2′′, H-5′′), 1.58 (s, 3H, H-6′), 1.52–1.68 (m, 4H, H-3′′, H-4′′), 1.69 (s, 3H, H-5′), 1.78–1.88 (m, 2H, H-2′′, H-5′′), 2.25 (sept, J = 7.7 Hz, 1H, H-1′′), 2.37–2.43 (m, 2H, H-α), 2.47 (dtm, J = 15.1, 7.5 Hz, 1H, H-2′), 2.61 (dtm, J = 15.0, 5.7 Hz, 1H, H-2′), 5.13 (tm, J = 7.3 Hz, 1H, H-3′), 6.03 (ddd, J = 7.3, 4.4, 0.8 Hz, 1H, H-1′), 6.99 (d, J = 1.0 Hz, 1H, H-3), 7.19 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.59 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 25.0 (C-3′′, C-4′′), 25.8 (C-5’), 32.4, 32.5 (C-2′′, C-5′′), 33.0 (C-2′), 36.5 (C-1′′), 40.4 (C-α), 69.2 (C-1′), 111.6 (C-4a), 111.8 (C-8a), 117.9 (C-3′), 131.5 (C-3), 132.7 (C-7), 132.8 (C-6), 136.0 (C-4′), 148.5 (C-2), 166.8 (C-5), 167.4 (C-8), 172.1 (COO), 176.8 (C-1), 178.3 (C-4); MS (ESI−) m/z (%): 1231 (9) [3M − 2H + K]−, 1215 (8) [3M − 2H + Na]−, 398.23 (34) [M]−, 397.30 (100) [M − H]−; [M]− calculated for C23H26O6: 398.1729.
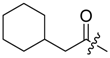
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclohexylacetate (9): 50 µmol Shikonin, 0.15 mmol DCC, 30 µmol DMAP, and 53 µmol cyclohexylacetic acid (9a); reaction time 19 h; flash CC on silica (8 g) with cyclohexane/CH2Cl2 = 2:1 to cyclohexane/CH2Cl2 = 0:1) and an additional PTLC on silica (developed three times with cyclohexane/CH2Cl2 = 2:1 and one time with cyclohexane/CH2Cl2 = 1:2); 9, yield: 36%. 9: Rf = 0.21 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2920 (s), ≈2950 (br), 2850 (m), 1738 (s), 1610 (s), 1570 (m), 1450 (s), 1230 (s), 1204 (s), 1160 (m), 1111 (m), 782 (w) cm−1; 1H-NMR (CDCl3): 0.92–1.05 (m, 2H, H-2′′ax, H-6′′ax), 1.09–1.22 (m, 1H, H-4′′ax), 1.21-1.33 (m, 2H, H-3′′ax, H-5′′ax), 1.59 (s, 3H, H-6′), 1.62-1.76 (m, 5H, H-2′′eq, H-3′′eq, H-4′′eq, H-5′′eq, H-6′′eq), 1.76–1.86 (m, 1H, H-1′′), 2.24 (dd, 12.4, 4.7 Hz, 1H, H-α), 2.29 (dd, 12.4, 5.0 Hz, 1H, H-α), 2.47 (dtm, J = 14.8, 7.5 Hz, 1H, H-2′), 2.61 (dtm, J = 15.1, 5.7 Hz, 1H, H-2′), 5.12 (tm, J = 7.2 Hz, 1H, H-3′), 6.04 (ddd, J = 7.3, 4.3, 0.8 Hz, 1H, H-1′), 6.99 (d, J = 0.8 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.42 (s, 1H, C5-OH), 12.58 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 25.7 (C-5′), 2 × 26.0, 26.1 (C-3′′, C-4′′, C-5′′), 3 × 33.0 (C-2′, C-2′′, C-6′′), 35.0 (C-1′′), 42.1 (C-α), 69.2 (C-1′), 111.6 (C-4a), 111.8 (C-8a), 117.9 (C-3′), 131.5 (C-3), 132.7 (C-7), 132.8 (C-6), 136.0 (C-4′), 148.5 (C-2), 166.9 (C-5), 167.4 (C-8), 171.8 (COO), 176.8 (C-1), 178.3 (C-4); MS (ESI−) m/z (%): 845 (12) [2(M − H)+Na]−, 412.29 (75) [M]−, 411.12 (100) [M − H]−; [M]− calculated for C24H28O6: 412.1886.
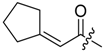
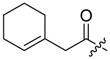
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl 1-cyclohexen-1-ylacetate (10): 50 µmol Shikonin, 0.12 mmol DCC, 15 µmol DMAP, and 53 µmol 1-cyclohexen-1-ylacetic acid (11a); reaction time 13 h; PTLC on silica with cyclohexane/CH2Cl2 = 1:1 (three times developed); 10, yield 5%. 10: Rf = 0.48 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2926 (m), 2857 (w), 2837 (w), 1738 (m), 1608 (s), 1569 (m), 1436 (m), 1409 (m), 1335 (m), 1229 (s), 1202 (s), 1147 (m), 1112 (m), 777 (m) cm−1; 1H-NMR (CDCl3): 1.51–1.68 (m, 4H, H-4′′, H-5′′), 1.55 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 1.97–2.03 (m, 2H, H-6′′), 2.03–2.09 (m, 2H, H-3′′), 2.47 (dtm, J = 15.1, 7.6 Hz, 1H, H-2′), 2.62 (dtm, J = 15.0, 5.7 Hz, 1H, H-2′), 3.01 (s, 2H, H-α), 5.13 (tm, J = 7.2 Hz, 1H, H-3′), 5.62 (s br, 1H, H-2′′), 6.01 (ddd, J = 6.5, 3.5, 0.9 Hz, 1H, H-1’), 6.97 (d, J = 1.0 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.42 (s, 1H, C5-OH), 12.57 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6’), 21.9 (C-5′′), 22.7 (C-4′′), 25.3 (C-3′′), 25.8 (C-5’), 28.6 (C-6’), 32.9 (C-2’), 43.6 (C-α), 69.5 (C-1’), 111.6 (C-4a), 111.9 (C-8a), 117.9 (C-3’), 126.5 (C-2′′), 130.6 (C-1′′), 131.4 (C-3), 132.7 (C-7), 132.9 (C-6), 136.0 (C-4’), 148.4 (C-2), 167.0 (C-5), 167.5 (C-8), 170.7 (COO), 176.7 (C-1), 178.2 (C-4); MS (ESI−) m/z (%): 841 (5) [2(M − H) + Na]−, 410.23 (35) [M]−, 409.33 (100) [M − H]−; [M]− calculated for C24H26O6: 410.1729.
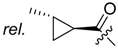
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclopropanecarboxylate (
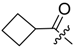
11): 0.1 mmol Shikonin, 0.15 mmol DCC, 25 µmol DMAP, and 0.1 mmol cyclopropanecarboxylic acid (
11a); reaction time 22 h; CC on silica (7 g) with cyclohexane/CH
2Cl
2 = 1:1 to 1:4;
11, yield 20%.
11: R
f = 0.28 (silica, cyclohexane/CH
2Cl
2 = 1:4); NMR data fit with literature [
20].
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclobutanecarboxylate (12): 0.1 mmol Shikonin, 0.15 mmol DCC, 25 µmol DMAP, and 0.1 mmol cyclobutane carboxylic acid (12a); reaction time 22 h; double PTLC on silica with cyclohexane/CH2Cl2 = 1:4; 12, yield 40%. 12: Rf = 0.35 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2980 (w), ≈2950 (vbr), 2947 (m), 2866 (w), 1736 (s), 1611 (vs), 1570 (m), 1455 (s), 1204 (s), 1162 (s), 787 (w) cm−1; 1H-NMR (CDCl3): 1.58 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 1.87–2.07 (m, 2H, H-3′′), 2.19–2.36 (m, 4H, H-2′′, H-4′′), 2.47 (dtm, J = 14.7, 7.7 Hz, 1H, H-2′), 2.61 (dtm, J = 14.9, 5.7 Hz, 1H, H-2′), 3.22 (quintd, J = 8.5, 0.7 Hz, 1H, H-1′′), 5.12 (tm, J = 7.3 Hz, 1H, H-3′), 6.02 (ddd, J = 7.4, 4.5, 0.9 Hz, 1H, H-1′), 6.96 (d, J = 1.0 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.59 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 17.9 (C-6′), 18.4 (C-3′′), 25.1, 25.2 (C-2′′, C-4′′), 25.8 (C-5’), 32.9 (C-2’), 38.0 (C-1′′), 69.0 (C-1’), 111.6 (C-4a), 111.8 (C-8a), 117.8 (C-3’), 131.4 (C-3), 132.6 (C-7), 132.8 (C-6), 136.0 (C-4’), 148.5 (C-2), 166.7 (C-5), 167.2 (C-8), 174.1 (COO), 176.9 (C-1), 178.4 (C-4); MS (ESI−) m/z (%): 1165 (3) [3M − 3H + Na + K]−, 1147 (6) [3M − 2H + K]−, 1231 (8) [3M − 2H + Na]−, 370.11 (42) [M]−, 369.17 (100) [M − H]−; [M]− calculated for C21H22O6: 370.1416.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclopentanecarboxylate (13): 0.1 mmol Shikonin, 0.15 mmol DCC, 25 µmol DMAP, and 0.1 mmol cyclopentanecarboxylic acid (13a); reaction time 17 h; double PTLC on silica with cyclohexane/CH2Cl2 = 1:4; 13, yield 41%. 13: Rf = 0.32 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 3050 (w), 2954 (m), 2919 (w), 2870 (w), 1737 (s), 1608 (s), 1570 (m), 1452 (m), 1205 (s), 1141 (s), 767 (m) cm−1; 1H-NMR (CDCl3): 1.58 (s, 3H, H-6′), 1.57–1.66 (m, 2H, H-3′′, H-4′′), 1.66–1.75 (m, 2H, H-3′′, H-4′′), 1.69 (s, 3H, H-5′), 1.75–1.87 (m, 2H, H-2′′, H-5′′), 1.87–1.99 (m, 2H, H-2′′, H-5′′), 2.47 (dtm, J = 14.7, 7.5 Hz, 1H, H-2′), 2.62 (dtm, J = 14.8, 5.7 Hz, 1H, H-2′), 2.82 (tt, J = 7.3, 8.4 Hz, 1H, H-1′′), 5.12 (tm, J = 7.3 Hz, 1H, H-3′), 6.02 (ddd, J = 7.3, 4.4, 0.8 Hz, 1H, H-1′), 6.98 (d, J = 0.9 Hz, 1H, H-3), 7.19 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.59 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 2 × 25.8 (C-5′, C-3′′, C-4′′), 29.9, 30.0 (C-2′′, C-5′′), 33.0 (C-2′), 43.8 (C-1′′), 69.0 (C-1′), 111.6 (C-4a), 111.8 (C-8a), 117.8 (C-3′), 131.4 (C-3), 132.6 (C-7), 132.8 (C-6), 135.9 (C-4′), 148.7 (C-2), 166.7 (C-5), 167.2 (C-8), 175.4 (COO), 176.9 (C-1), 178.5 (C-4); MS (ESI−) m/z (%): 1189 (7) [3M − 2H + K]−, 384.13 (62) [M−], 383.28 (100) [M − H]−; [M]− calculated for C22H24O6: 384.1573.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclohexanecarboxylate (14): 50 µmol Shikonin, 0.12 mmol DCC, 15 µmol DMAP, and 53 µmol cyclohexanecarboxylic acid (14a); reaction time 16 h; PTLC on silica with cyclohexane/CH2Cl2 = 1:4 (twice developed) and an additional PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 1:1 and twice with cyclohexane/CH2Cl2 = 1:2); 14, yield 10%. 14: Rf = 0.24 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2927 (m), ≈2950 (br), 2852 (w), 1736 (s), 1609 (s), 1570 (m), 1451 (m), 1230 (m), 1203 (m), 1162 (m), 1159 (s) cm−1; 1H-NMR (CDCl3): 1.18–1.38 (m, 3H, H-3′′ax, H-4′′ax, H-5′′ax), 1.40-1.54 (m, 2H, H-2′′ax, H-6′′ax), 1.58 (s, 3H, H-6′), 1.62–1.69 (m, 1H, H-4′′eq), 1.69 (s, 3H, H-5′), 1.74–1.81 (m, 2H, H-3′′eq, H-5′′eq), 1.94 (dm, J = 11.9 Hz, 2H, H-2′′eq, H-6′′eq), 2.38 (tt, J = 11.2, 3.7 Hz, 1H, H-1′′), 2.47 (dtm, J = 15.0, 7.3 Hz, 1H, H-2′), 2.61 (dtm, J = 14.8, 5.8 Hz, 1H, H-2′), 5.12 (tm, J = 7.3 Hz, 1H, H-3′), 6.03 (ddd, J = 7.3, 4.5, 0.9 Hz, 1H, H-1′), 6.97 (d, J = 1.0 Hz, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.58 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 2 × 25.4 (C-3′′, C-5′′), 25.7 (C-4′′), 26.9 (C-5′), 28.9, 29.0 (C-2′′, C-6′′), 33.0 (C-2’), 43.3 (C-1′′), 68.9 (C-1’), 111.6 (C-4a), 111.9 (C-8a), 117.8 (C-3’), 131.4 (C-3), 132.6 (C-7), 132.8 (C-6), 135.9 (C-4’), 148.7 (C-2), 166.7 (C-5), 167.3 (C-8), 174.7 (COO), 177.0 (C-1), 178.5 (C-4); MS (ESI−) m/z (%): 817 (5) [2(M − H) + Na]−, 398.16 (51) [M]−, 397.28 (100) [M − H]−; [M]− calculated for C23H26O6: 398.1729.
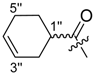
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclohex-1-enecarboxylate (15): 0.1 mmol Shikonin, 0.17 mmol DCC, 25 µmol DMAP, and 0.1 mmol cyclohex-1-enecarboxylic acid (15a); reaction time 16 h; PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1 and one time with cyclohexane/CH2Cl2 = 1:2) and an additional PTLC on silica (developed twice with cyclohexane / CH2Cl2 = 2:1); 15, yield 13%. 15: Rf = 0.37 (silica, cyclohexane / CH2Cl2 = 1:4); IR (ATR): 2917 (m), ≈2950 (br), 2890 (w), 1712 (m), 1609 (s), 1569 (m), 1452 (m), 1229 (s), 1203 (s), 1077 (m) cm−1; 1H-NMR (CDCl3): 1.57 (s, 3H, H-6′), 1.60–1.69 (m, 4H, H-4′′, H-5′′), 1.69 (s, 3H, H-5′), 2.20–2.32 (m, 4H, H-3′, H-6′′), 2.52 (dtm, J = 15.0, 7.2 Hz, 1H, H-2′), 2.64 (dtm, J = 15.1, 5.4 Hz, 1H, H-2′), 5.14 (tm, J = 7.2 Hz, 1H, H-3′), 6.03 (ddd, J = 7.1, 4.6, 0.8 Hz, 1H, H-1′), 6.96 (d, J = 1.0 Hz, 1H, H-3), 7.09 (m, 1H, H-2′′), 7.19 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.59 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 21.4, 22.0 (C-5′′, C-4′′), 24.1 (C-3′′*), 25.8 (C-5′), 25.9 (C-6′′*), 32.9 (C-2′), 69.2 (C-1′), 111.6 (C-4a), 111.9 (C-8a), 117.8 (C-3′), 129.9 (C-1′′), 131.5 (C-3), 132.5 (C-7), 132.6 (C-6), 135.9 (C-4′), 141.1 (C-2′′), 148.9 (C-2), 166.1 (COO), 166.3 (C-5), 166.9 (C-8), 177.4 (C-1), 178.9 (C-4); MS (ESI−) m/z (%): 813 (11) [2(M − H) + Na]−, 396.24 (44) [M]−, 395.29 (100) [M − H]−; [M]− calculated for C23H24O6: 396.1573.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl cyclohex-3-enecarboxylate (16): 0.1 mmol Shikonin, 0.20 mmol DCC, 30 µmol DMAP, and 0.1 mmol cyclohex-3-enecarboxylic acid (16a); reaction time 16 h; two subsequent PTLC on silica each one developed twice with cyclohexane/CH2Cl2 = 2:1 and twice with cyclohexane/CH2Cl2 = 1:2); 16, yield: 29%. 16: 1:1 mixture of diastereomers; Rf = 0.28 (silica, CH2Cl2); IR (ATR): 3025 (vw), 2917 (m), ≈2950 (br), 2849 (w), 1735 (s), 1608 (s), 1568 (m), 1452 (m), 1220 (s), 1201 (s), 1155 (s) cm−1; 1H-NMR (CDCl3): 1.59 (s, 3H, H-6′), 1.70 (s, 3H, H-5′), 1.68–1.81 (m, 1H, H-6′′), 2.00–2.08 (m, 1H, H-6′′), 2.10–2.17 (m, 2H, H-5′′), 2.26–2.32 (m, 2H, H-2′′), 2.49 (dtm, J = 15.0, 7.3 Hz, 1H, H-2′), 2.59–2.71 (m, 2H, H-2′ and H-1′′), 5.13 (tm, J = 6.6 Hz, 1H, H-3′), 5.66–5.76 (m, 2H, H-3′′, H-4′′), 6.05 (m, 1H, H-1′), 6.98 (2 d, 1H, J ≈1 Hz, H-3), 7.19 (s, 2H, H-6, H-7), 12.42 (s, 1H, C5-OH), 12.58 (2 s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 24.2 and 24.3 (C-5′′), 24.9 and 25.0 (C-6′′), 25.8 (C-5′), 27.3, 27.4 (C-2′′), 33.0 (C-2′), 2 × 39.3 (C-1′′), 69.1, 69.2 (C-1′), 111.6 (C-4a), 111.8 (C-8a), 117.7, 117.8 (C-3′), 124.9, 125.0 (C-3′′), 2 × 126.8 (C-4′′), 131.3 (C-3), 2 × 132.7 (C-7), 2 × 132.9 (C-6), 136.0 (C-4′), 2 × 148.5 (C-2), 2 × 167.0 (C-5), 167.5, 167.6 (C-8), 174.4, 174.5 (COO), 176.6, 176.7 (C-1), 178.1, 178.2 (C-4); MS (ESI−) m/z (%): 813.41 (7) [2(M − H) + Na]−, 395.16 [M − H]−; [M − H]− calculated for C23H24O6: 395.1495.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl trans 2-methylcyclopropanecarboxylate (17): 0.1 mmol Shikonin, 0.2 mmol DCC, 82 µmol DMAP, and 0.11 mmol 2-methyl-cyclopropane carboxylic acid (cis/trans mixture ca. 1:3,5) (17a); reaction time 15 h; CC on silica (7 g) with cyclohexane/CH2Cl2 = 2:1 to 0:1 and PTLC on silica with cyclohexane/CH2Cl2 = 2:1 (five times developed); two diastereomers of 17, yield 3%. 17: Rf = 0.26 (silica, CH2Cl2); IR (ATR): 2960 (w), 2919 (w), ≈2950 (br), 2856 (w), 1732 (s), 1609 (s), 1569 (m), 1559 (m), 1454 (m), 1406 (m), 1205 (m), 1177 (m), 1159 (s), 781 (m) cm−1; 1H-NMR (CDCl3): 0.70–0.79 (m, 2H, 2 × H-3′′), 1.12–1.20 (m, 7H, 2 × cyclopropyl-CH3, 1 × H-3′′), 1.20–1.26 (m, 1H, H-3′′), 1.36–1.48 (m, 4H, 2 H-1′′, 2 × H-2′′), 1.58 (s, 6H, H-6′), 1.70 (s, 6H, H-5′), 2.42-2.53 (m, 2H, 2 × H-2′), 2.57–2.66 (m, 2H, 2 × H-2′), 5.08–5.16 (m, 2H, 2 × H-3′), 6.00 (tm, J = 5.9 Hz, 2H, H-1′), 6.99 (d, J = 0.9 Hz, 1H, H-3), 7.00 (d, J = 0.9 Hz, 1H, H-3), 7.18 (s, 4H, H-6, H-7), 12.44 (2s, 2H, C5-OH), 12.58 (s, 2H, C8-OH); 13C-NMR (CDCl3): δ 2 × 17.2 (C-3′′), 2 × 17.7 (C-2′′), 3 × 17.9 (cyclopropyl-CH3, C-6′), 2 × 21.2 (C-1′′), 25.8 (C-5′), 32.9 (C-2′), 69.2, 69.3 (C-1′), 111.6 (C-4a), 111.8 (C-8a), 117.7 (C-3′), 2 × 131.6 (C-3), 132.6 (C-7), 132.7 (C-6), 136.0 (C-4′), 148.6 (C-2), 166.5 (C-5), 167.0 (C-8), 2 × 173.3 (COO), 2 × 177.2 (C-1), 178.7, 178.8 (C-4); MS (ESI−) m/z (%): 761.32 (12) [2(M − H)+Na]−, 370.14 (46) [M]−, 369.26 (100) [M − H]−; [M]− calculated for C21H22O6: 370.1416.
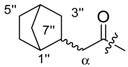
Exo-(R)-1-(1,4-dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-3-yl)-4-methylpent-3-enyl bicyclo[2 .2.1]hept-5-ene-2-carboxylate (18) and endo-(R)-1-(1,4-dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-3-yl)-4-methylpent-3-enyl bicyclo[2.2.1]hept-5-ene-2-carboxylate (19): 0.1 mmol Shikonin, 0.20 mmol DCC, 25 µmol DMAP, and 0.11 mmol bicyclo[2.2.1]hept-5-ene-2-carboxylic acid (Exo (18a)/Endo (19a)/ = 1.0:4.1); reaction time 16 h; PTLC on silica (developed three times with cyclohexane/CH2Cl2 = 2:1 and one time with cyclohexane/CH2Cl2 = 1:2) and PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 2:1 and three times with cyclohexane/CH2Cl2 = 1:2), yield: 18, 6% and 19 10%. 18: 1:1 mixture of diastereomers, Rf = 0.31 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2972 (w), 2917 (m), ≈2950 (br), 2850 (w), 1734 (m), 1609 (s), 1568 (m), 1453 (m), 1230 (m), 1203 (m), 1163 (s), 1147 (m) cm−1; 1H-NMR (CDCl3): δ 1.37–1.51 (m, 4H, H-3′′, H-7′′a, H-7′′b), 1.58 (s, 3H, H-6′), 1.60 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 1.70 (s, 3H, H-5′), 1.88–1.95 (m, 2H, H-3′′), 2.28-2.34 (m, 2H, H-2′′), 2.48 (dt, J = 14.8, 2H, 7.7 Hz, H-2′), 2.62 (dt, J = 15.0, 2H, 5.7 Hz, H-2′); 2.94 (s, br, 2H, H-4′′), 3.06 (s, br, 1H, H-1′′), 3.08 (s, br, 1H, H-1′′), 5.13 (tm, J = 7.3 Hz, 1H, H-3’), 5.15 (tm, J = 7.3 Hz, 1H, H-3’), 6.04 (td, J = 4.3, 0.9 Hz, 1H, H-1’), 6.06 (td, J = 4.4, 0.9 Hz, 1H, H-1’), 6.12–6.20 (m, 4H, H-5′′, H-6′′), 7.00 (d, J = 1.0 Hz, 1H, H-3), 7.01 (d, J = 1.0 Hz, 1H, H-3), 6.12–6.19 (m, 4H, H-5′′, H-6′′), 7.18, 7.19 (2s, 4H, H-6, H-7), 12.43 (2s, 2H, C5-OH), 12.59 (2s, 2H, C8-OH); 13C-NMR (CDCl3): δ 2 × 18.0 (C-6’), 25.8 (C-5’), 30.3, 30.5 (C-3′′), 33.0, 33.1 (C-2’), 41.6, 41.7 (C-4′′), 43.1, 43.3 (C-2′′), 2 × 46.4 (C-7′′), 46.5, 46.7 (C-1′′), 69.2 (C-1’), 111.6 (C-4a), 111.9 (C-8a), 117.8, 117.9 (C-3’), 2 × 131.4 (C-3), 2 × 132.7 (C-7), 2 × 132.9 (C-6), 135.6 (C-6′′), 2 × 136.0 (C-4’), 138.2, 138.3 (C-5′′), 148.6 (C-2), 2 × 166.9, 167.0 (C-5), 2 × 167.5 (C-8), 174.9, 175.0 (COO), 176.7, 176.8 (C-1), 2 × 178.3 (C-4); MS (ESI−) m/z (%): 837 (5) [2(M − H) + Na]−, 408.18 (42) [M]−, 407.22 (100) [M − H]−; [M]− calculated for C24H24O6: 408.1573. 19: 1:1 mixture of diastereomers, Rf = 0.26 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2973 (w), 2917 (m), ≈2950 (br), 2850 (w), 1733 (m), 1609 (s), 1569 (m), 1452 (m), 1231 (m), 1202 (m), 1165 (s), 1108 (m), 709 (w) cm−1; 1H-NMR (CDCl3): 1.32 (d, J = 8.3 Hz, 2H, H-7′′a), 1.39–1.51 (m, 4H, H-3′′, H-7′′b), 1.58 (s, 3H, H-6′), 1.61 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 1.72 (s, 3H, H-5′), 1.88–2.00 m, 2H, H-3′′), 2.46 (dt, J = 14.9, 7.5 Hz, 2H, H-2′), 2.59 (dt, J = 15.0, 5.3 Hz, 2H, H-2′), 2.93 (s, br, 2H, H-4′′), 3.04 (dtd, J = 8.9, 4.0, 1.1 Hz, 2H, H-2′′), 3.28 (s, br, 2H, H-1′′), 5.11 (tm, J = 7.1 Hz, 1H, H-3′), 5.16 (tm, J = 7.3 Hz, 1H, H-3′), 5.88 (dd, J = 5.7, 2.8 Hz, 1H, H-6′′), 5.90 (dd, J = 5.7, 2.8 Hz, 1H, H-6′′), 5.94 (ddd, J = ≈7.5, 4.2, 0.8 Hz, 1H, H-1′), 5.96 (ddd, J = ≈7.5, 4.6, 0.6 Hz, 2H, H-1′), 6.19 (dd, J = 5.7, 3.0 Hz, 1H, H-5′′), 6.22 (dd, J = 5.7, 3.1 Hz, 1H, H-5′′), 6.96 (d, J = 0.9 Hz, 1H, H-3), 6.99 (d, J = 0.9 Hz, 1H, H-3), 7.18 (s, 4H, H-6, H-7), 12.42 (s, 2H, C5-OH), 12.57 (2s, 1H, C8-OH); 13C-NMR (CDCl3): δ 2 × 18.0 (C-6′), 2 × 25.8 (C-5′), 29.0), 29.3 (C-3′′), 32.9, 33.0 (C-2′), 2 × 42.6 (C-4′′), 2 × 43.4 (C-2′′), 46.0 (C-1′), 49.7, 49.9 (C-7′′), 69.2, 69. 4 (C-1′), 2 × 111.6 (C-4a), 111.9 (C-8a), 117.9, 118.1 (C-3′), 131.3, 131.4 (C-3), 132.0, 132.2 (C-6′′), 132.6, 2 × 132.8, 132.9 (C-6, C-7), 135.8, 135.9 (C-4′), 138.0, 138.1 (C-5′′), 148.6 (C-2), 167.0 (C-5), 167.7 (C-8), 173.6 (COO), 176.5 (COO, C-1), 176.7 (C-1), 178.2 (C-4); more polar diastereomer: MS (ESI−) m/z (%): 408.25 (33) [M]−, 407.31 (100) [M − H]−; less polar diastereomer: MS (ESI−) m/z (%): 408.17 (43) [M]−, 407.21 (100) [M − H]−; [M]− calculated for C24H24O6: 408.1573.
(R)-1-(1,4-Dihydro-5,8-dihydroxy-1,4-dioxonaphthalen-2-yl)-4-methylpent-3-enyl bicyclo[2.2.1]heptane-2-ylacetate (20): 50 µmol Shikonin, 0.12 mmol DCC, 15 µmol DMAP, and 53 µmol bicyclo[2.2.1]heptane-2-ylacetic acid (20a); reaction time 16 h; PTLC on silica with cyclohexane/CH2Cl2 = 1:4 (twice developed) and an additional PTLC on silica (developed twice with cyclohexane/CH2Cl2 = 1 : 1 and twice with cyclohexane/CH2Cl2 = 1:2); 20, yield 19% (sum of isomers). 20: Rf = 0.24 (silica, cyclohexane/CH2Cl2 = 1:4); IR (ATR): 2948 (m), 2917 (m), ≈2950 (br), 2868 (w), 1738 (s), 1609 (s), 1570 (m), 1453 (m), 1204 (s), 1173 (m), 1159 (s), 783 (m) cm−1; 1H-NMR (CDCl3): 1.06 (m, 1H, H-3′′), 1.13 (m, 1H, H-7′′), 1.15 (m, 1H, H-5′′), 1.24 (m, 1H, H-6′′), 1.32 (dquint, J = 9.9, 1.5 Hz, 1H, H-7′′), 1.48 (m, 1H, H-5′′), 1.51 (m, 1H, H-6′′), 1.53 (m, 1H, H-3′′), 1.58 (s, 3H, H-6′), 1.69 (s, 3H, H-5′), 1.91 (quint, J = 6.5 Hz, 1H, H-2′′), 1.98 (s, br, 1H, H-1′′), 2.20 (m, 1H, H-α), 2.23 (m, 1H, H-4′′), 2.34 (dd, J = 15.2, 7.9 Hz, 1H, H-α), 2.46 (dt, J = 15.0, 7.5 Hz, 1H, H-2′), 2.60 (dt, J = 15.5, 5.2 Hz, 1H, H-2′), 5.12 (t, J = 7.2 Hz, 1H, H-3′), 6.02 (dd, J = 7.4, 4.7 Hz, 1H, H-1′), 6.98 (s, 1H, H-3), 7.18 (s, 2H, H-6, H-7), 12.43 (s, 1H, C5-OH), 12.58 (s, 1H, C8-OH); 13C-NMR (CDCl3): δ 18.0 (C-6′), 25.8 (C-5′), 28.5 (C-5′′), 29.8 (C-6′′), 2 × 33.0 (C-2′), 2 × 35.2 (C-7′′), 36.8 (C-4′′), 37.8, 37.9 (C-3′′), 38.5, 38.6 (C-2′′), 41.1, 41.3 (C-1′′), 2 × 41.3 (C-α), 69.2, 69.3 (C-1’), 111.6 (C-4a), 111.9 (C-8a), 117.9 (C-3’), 131.4, 131.5 (C-3), 132.7 (C-7), 132.8 (C-6), 136.0 (C-4’), 148.5 (C-2), 2 × 166.9 (C-5), 2 × 167.4 (C-8), 171.9 (COO), 2 × 176.8 (C-1), 2 × 178.3 (C-4); MS (ESI−) m/z (%): 869.37 (9) [2(M − H) + Na]−, 424.23 (49) [M]−, 423.25 (100) [M − H]−, [M]− calculated for C25H28O6: 424.1886.