Preparation of Cyano-Substituted Tetraphenylethylene Derivatives and Their Applications in Solution-Processable OLEDs
Abstract
:1. Introduction
2. Results and Discussion
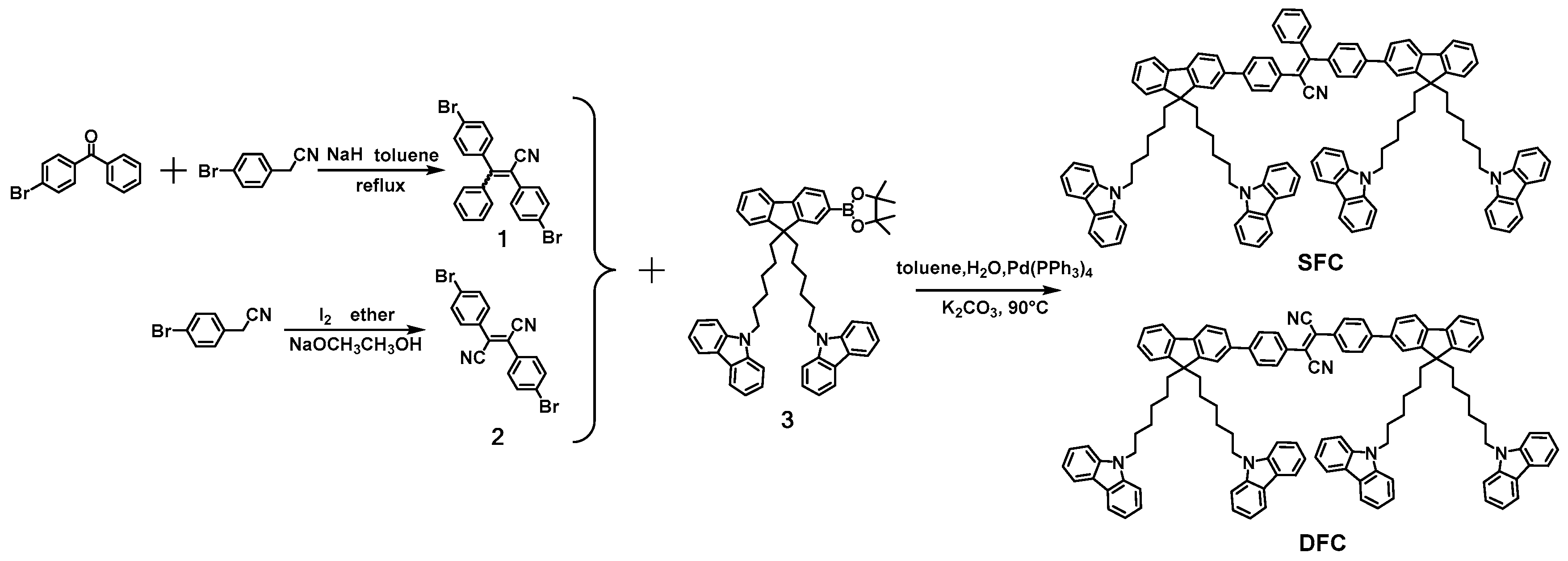
2.1. Synthesis and Characterization
2.2. Thermal Stablity
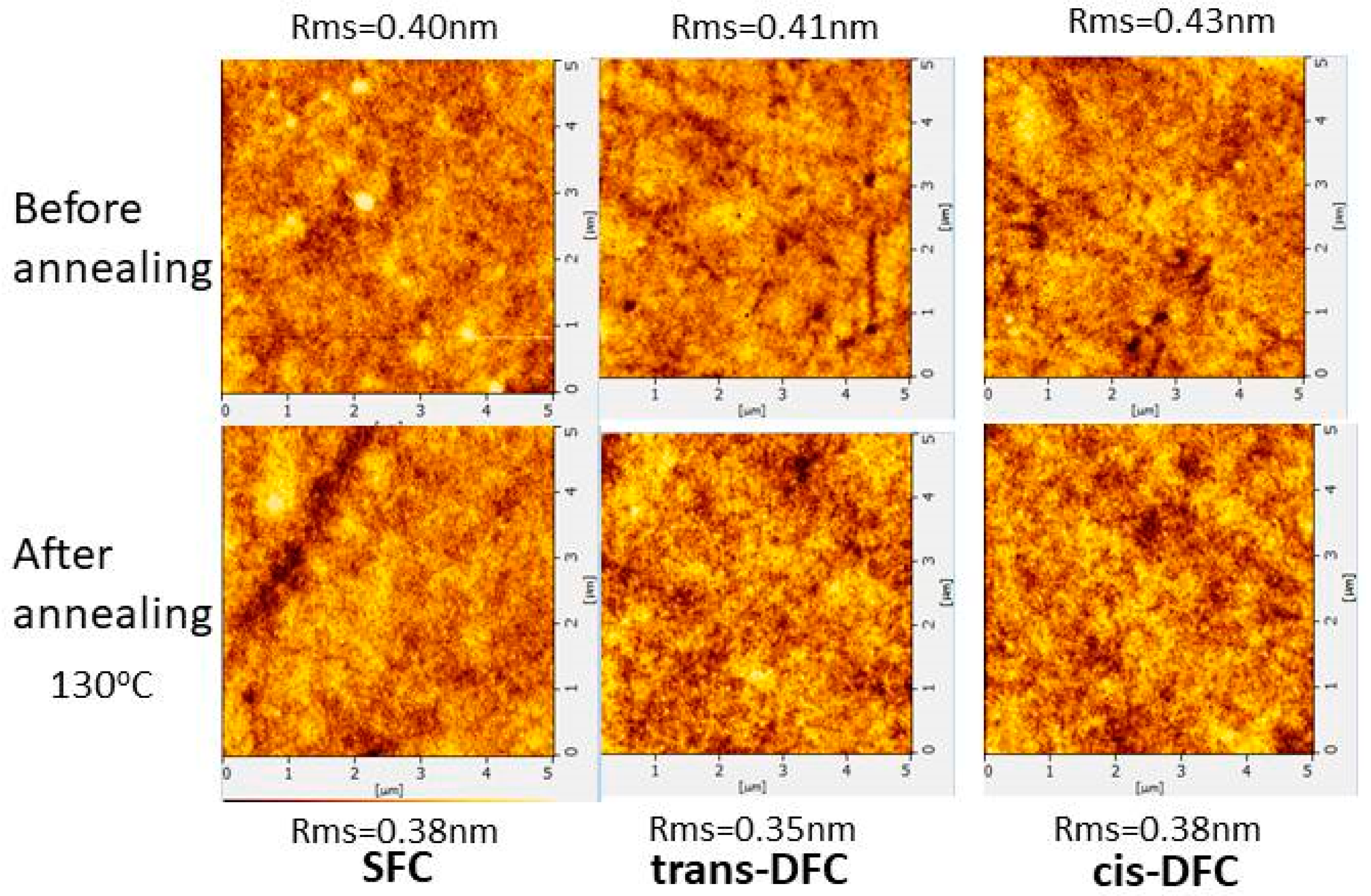
2.3. Morphological Stablity
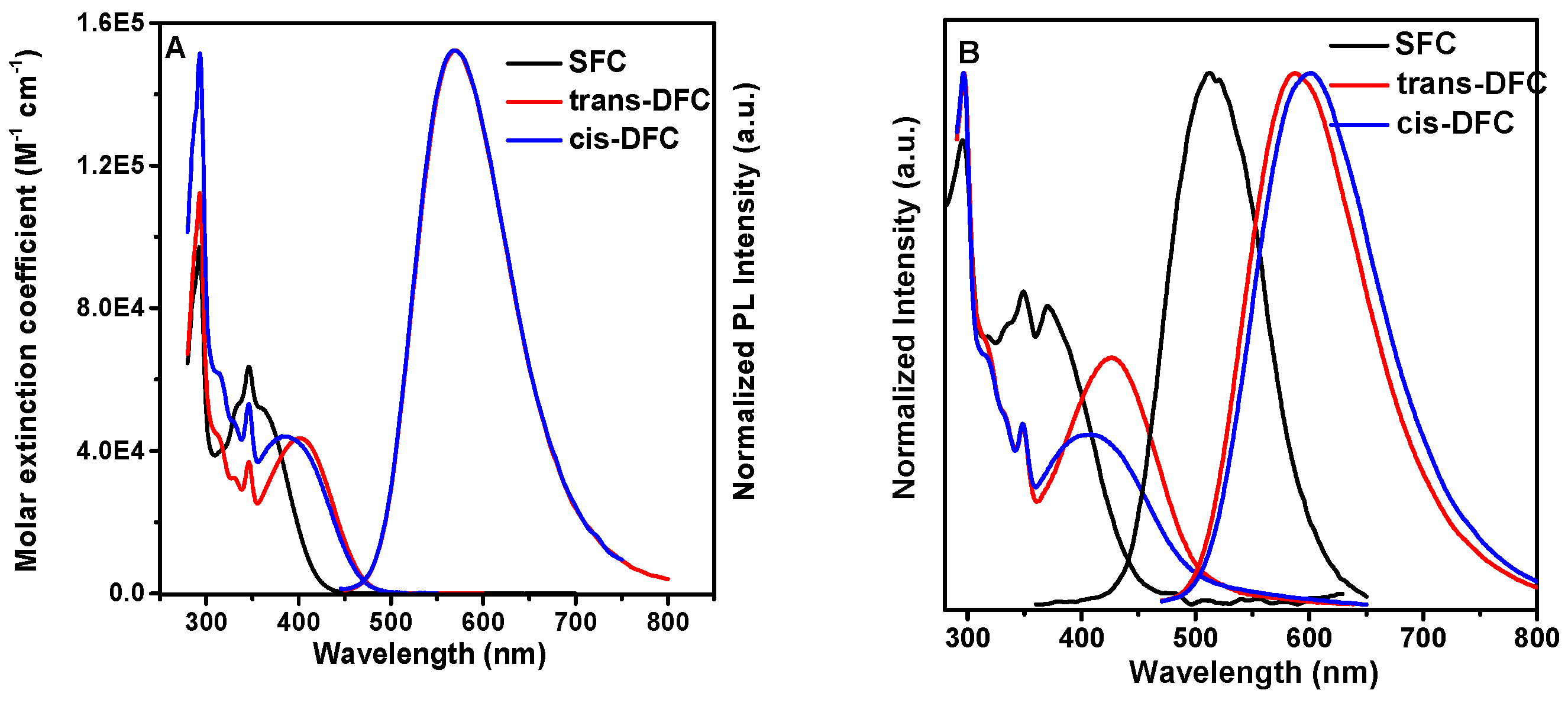
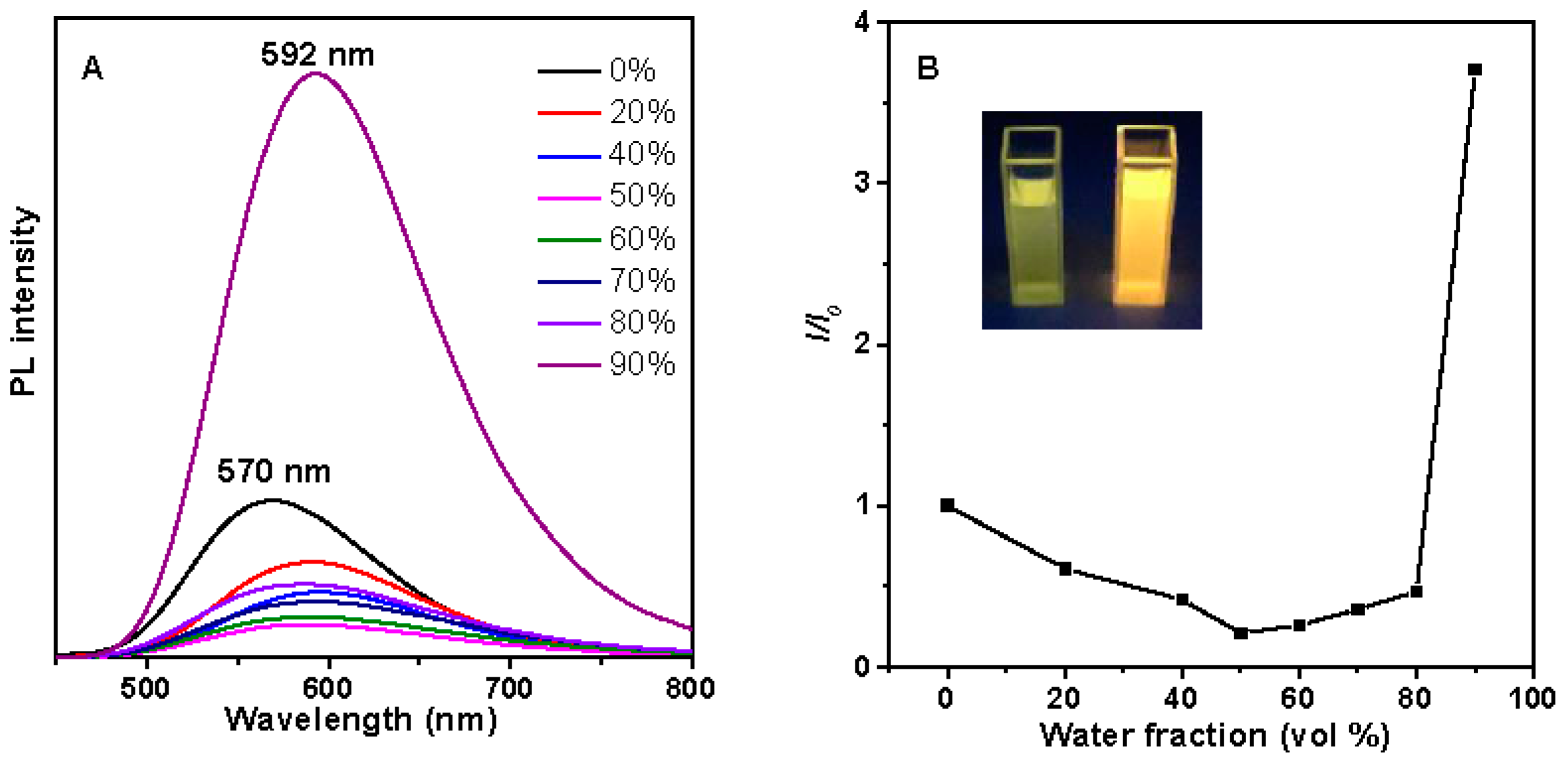
2.4. Photophysical Property
2.5. Electrochemical Properties
2.6. Electroluminescence Properties
3. Materials and Methods
3.1. Instrumentation
3.2. Device Fabrication
3.3. Experimental Section
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tang, C.W.; VanSlyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- Yeh, H.-C.; Chan, L.-H.; Wu, W.-C.; Chen, C.-T. Non-doped red organic light-emitting diodes. J. Mater. Chem. 2004, 14, 1293–1298. [Google Scholar] [CrossRef]
- Wu, K.C.; Ku, P.J.; Lin, C.S.; Shih, H.T.; Wu, F.I.; Huang, M.J.; Lin, J.J.; Chen, I.C.; Cheng, C.H. The Photophysical Properties of Dipyrenylbenzenes and Their Application as Exceedingly Efficient Blue Emitters for Electroluminescent Devices. Adv. Funct. Mater. 2008, 18, 67–75. [Google Scholar] [CrossRef]
- Im, Y.; Byun, S.Y.; Kim, J.H.; Lee, D.R.; Oh, C.S.; Yook, K.S.; Lee, J.Y. Recent Progress in High-Efficiency Blue-Light-Emitting Materials for Organic Light-Emitting Diodes. Adv. Funct. Mater. 2017, 27. [Google Scholar] [CrossRef]
- Nikolka, M.; Nasrallah, I.; Rose, B.; Ravva, M.K.; Broch, K.; Sadhanala, A.; Harkin, D.; Charmet, J.; Hurhangee, M.; Brown, A.; et al. High operational and environmental stability of high-mobility conjugated polymer field-effect transistors through the use of molecular additives. Nat. Mater. 2017, 16, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Guo, Y.; Liu, Y. 25th anniversary article: Recent advances in n-type and ambipolar organic field-effect transistors. Adv. Mater. 2013, 25, 5372–5391. [Google Scholar] [CrossRef] [PubMed]
- Lei, T.; Cao, Y.; Fan, Y.; Liu, C.J.; Yuan, S.C.; Pei, J. High-performance air-stable organic field-effect transistors: Isoindigo-based conjugated polymers. J. Am. Chem. Soc. 2011, 133, 6099–6101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Han, Y.; Zhu, X.; Fei, Z.; Feng, Y.; Treat, N.D.; Faber, H.; Stingelin, N.; McCulloch, I.; Anthopoulos, T.D.; et al. A Novel Alkylated Indacenodithieno[3,2-b]thiophene-Based Polymer for High-Performance Field-Effect Transistors. Adv. Mater. 2016, 28, 3922–3927. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.; Ramos-Ortíz, G.; Alcalá-Salas, M.I.; Maldonado, J.L.; López-Varela, K.A.; López, Y.; Domínguez, O.; Meneses-Nava, M.A.; Barbosa-García, O.; Santillan, R.; et al. One-pot synthesis and characterization of novel boronates for the growth of single crystals with nonlinear optical properties. Dyes Pigments 2010, 87, 76–83. [Google Scholar] [CrossRef]
- Li, J.; Yim, D.; Jang, W.D.; Yoon, J. Recent progress in the design and applications of fluorescence probes containing crown ethers. Chem. Soc. Rev. 2017, 46, 2437–2458. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Yasuda, T.; Komiyama, H.; Lee, J.; Adachi, C. Thermally Activated Delayed Fluorescence Polymers for Efficient Solution-Processed Organic Light-Emitting Diodes. Adv. Mater. 2016, 28, 4019–4024. [Google Scholar] [CrossRef] [PubMed]
- Birks, J.B. (Ed.) Photophysics of Aromatic Molecules; Wiley: London, UK, 1970. [Google Scholar]
- Luo, J.; Xie, Z.; Lam, J.W.Y.; Cheng, L.; Tang, B.Z.; Chen, H.; Qiu, C.; Kwok, H.S.; Zhan, X.; Liu, Y.; et al. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole. Chem. Commun. 2001, 1740–1741. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.; Kwok, R.T.; Lam, J.W.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Qin, W.; Leung, N.L.C.; Arseneault, M.; Lam, J.W.Y.; Liang, G.; Sung, H.H.Y.; Williams, I.D.; Tang, B.Z. A mechanistic study of AIE processes of TPE luminogens: Intramolecular rotation vs. configurational isomerization. J. Mater. Chem. C 2016, 4, 99–107. [Google Scholar] [CrossRef]
- Mei, J.; Hong, Y.; Lam, J.W.; Qin, A.; Tang, Y.; Tang, B.Z. Aggregation-induced emission: The whole is more brilliant than the parts. Adv. Mater. 2014, 26, 5429–5479. [Google Scholar] [CrossRef] [PubMed]
- Kwok, R.T.; Leung, C.W.; Lam, J.W.; Tang, B.Z. Biosensing by luminogens with aggregation-induced emission characteristics. Chem. Soc. Rev. 2015, 44, 4228–4238. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Bai, Q.; Yao, L.; Liu, H.; Gao, Y.; Li, J.; Liu, L.; Liu, Y.; Li, X.; Lu, P.; et al. Highly Efficient Solid-State Near-Infrared Emitting Material Based on Triphenylamine and Diphenylfumaronitrile with an EQE of 2.58% in Nondoped Organic Light-Emitting Diode. Adv. Funct. Mater. 2015, 25, 7521–7529. [Google Scholar] [CrossRef]
- Tang, S.; Liu, M.R.; Lu, P.; Xia, H.; Li, M.; Xie, Z.Q.; Shen, F.Z.; Gu, C.; Wang, H.P.; Yang, B.; et al. A Molecular Glass for Deep-Blue Organic Light-Emitting Diodes Comprising a 9,9′-Spirobifluorene Core and Peripheral Carbazole Groups. Adv. Funct. Mater. 2007, 17, 2869–2877. [Google Scholar] [CrossRef]
- Liu, H.; Cheng, G.; Hu, D.; Shen, F.; Lv, Y.; Sun, G.; Yang, B.; Lu, P.; Ma, Y. A Highly Efficient, Blue-Phosphorescent Device Based on a Wide-Bandgap Host/FIrpic: Rational Design of the Carbazole and Phosphine Oxide Moieties on Tetraphenylsilane. Adv. Funct. Mater. 2012, 22, 2830–2836. [Google Scholar] [CrossRef]
- Yao, L.; Xue, S.; Wang, Q.; Dong, W.; Yang, W.; Wu, H.; Zhang, M.; Yang, B.; Ma, Y. RGB small molecules based on a bipolar molecular design for highly efficient solution-processed single-layer OLEDs. Chem. Eur. J. 2012, 18, 2707–2714. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Lam, J.W.; Tang, B.Z. Aggregation-induced emission: Phenomenon, mechanism and applications. Chem. Commun. 2009, 4332–4353. [Google Scholar] [CrossRef] [PubMed]
- Ellinger, S.; Graham, K.R.; Shi, P.; Farley, R.T.; Steckler, T.T.; Brookins, R.N.; Taranekar, P.; Mei, J.; Padilha, L.A.; Ensley, T.R.; et al. Donor–Acceptor–Donor-based π-Conjugated Oligomers for Nonlinear Optics and Near-IR Emission. Chem. Mater. 2011, 23, 3805–3817. [Google Scholar] [CrossRef]
- Li, J.; Han, X.; Bai, Q.; Shan, T.; Lu, P.; Ma, Y. Electropolymerized AIE-active polymer film with high quantum efficiency and its application in OLED. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 707–715. [Google Scholar] [CrossRef]
- Gu, C.; Huang, N.; Wu, Y.; Xu, H.; Jiang, D. Design of Highly Photofunctional Porous Polymer Films with Controlled Thickness and Prominent Microporosity. Angew. Chem. Int. Ed. Engl. 2015, 54, 11540–11544. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds SFC, trans-DFC, and cis-DFC are available from the authors. |

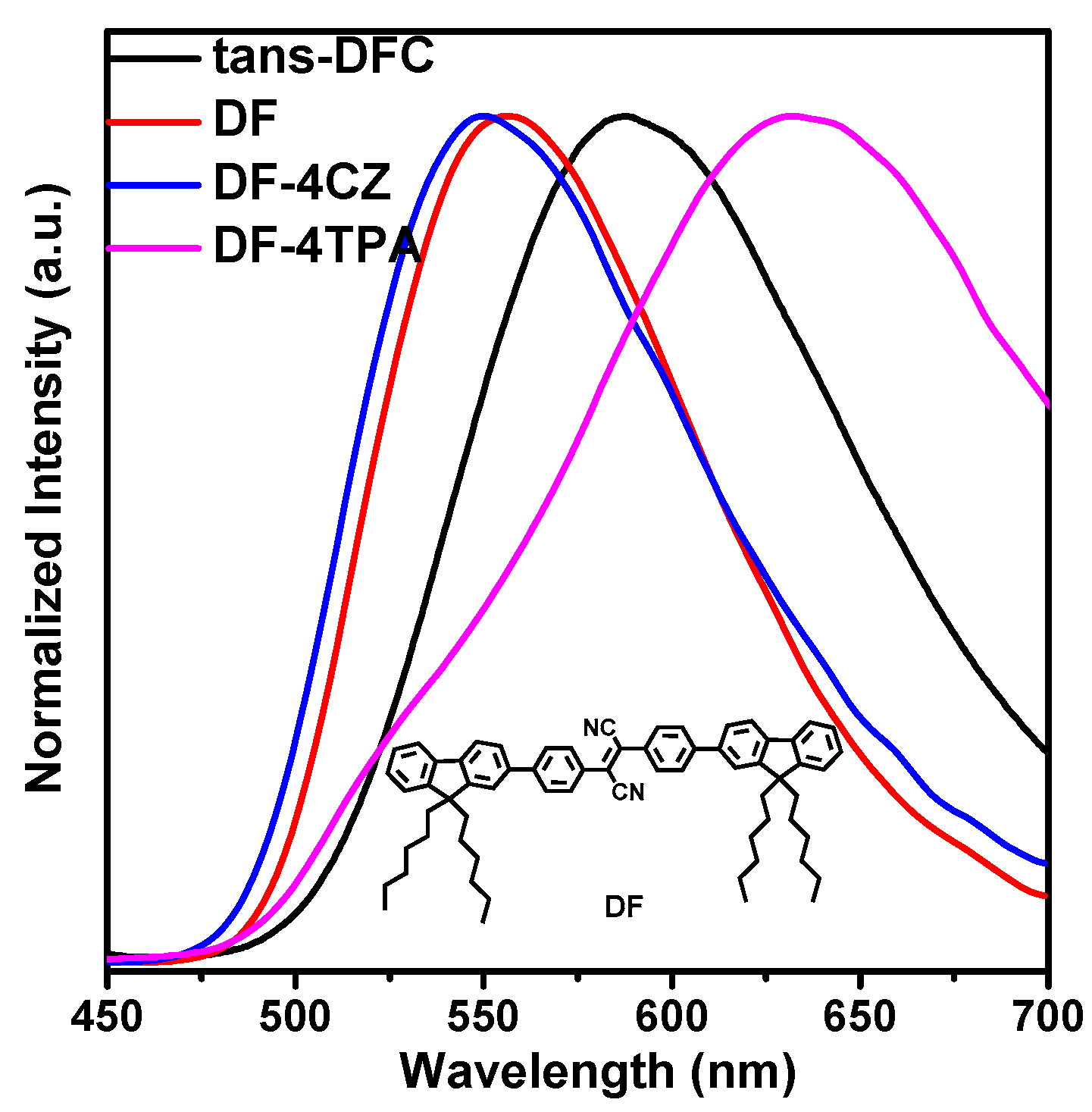
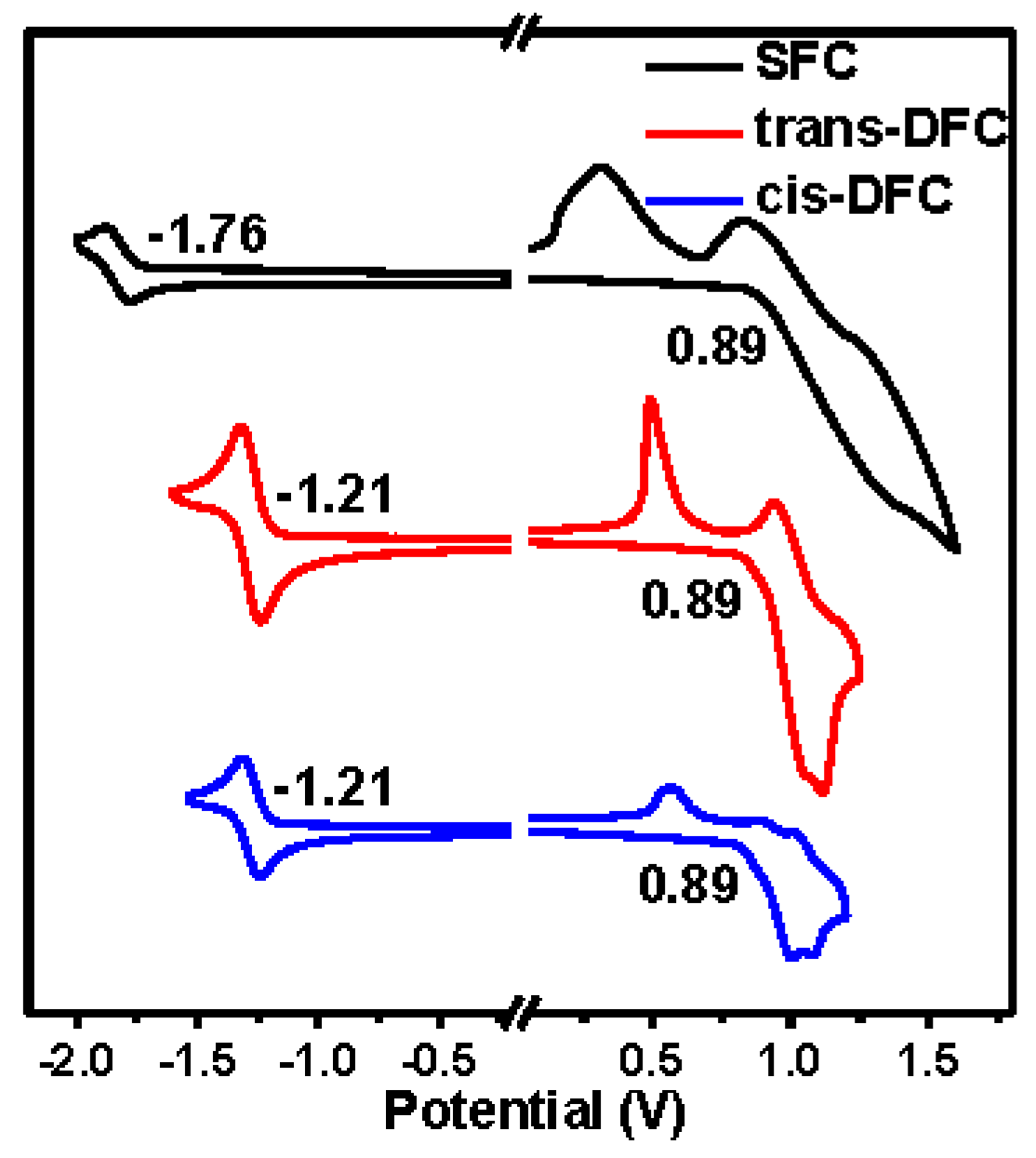
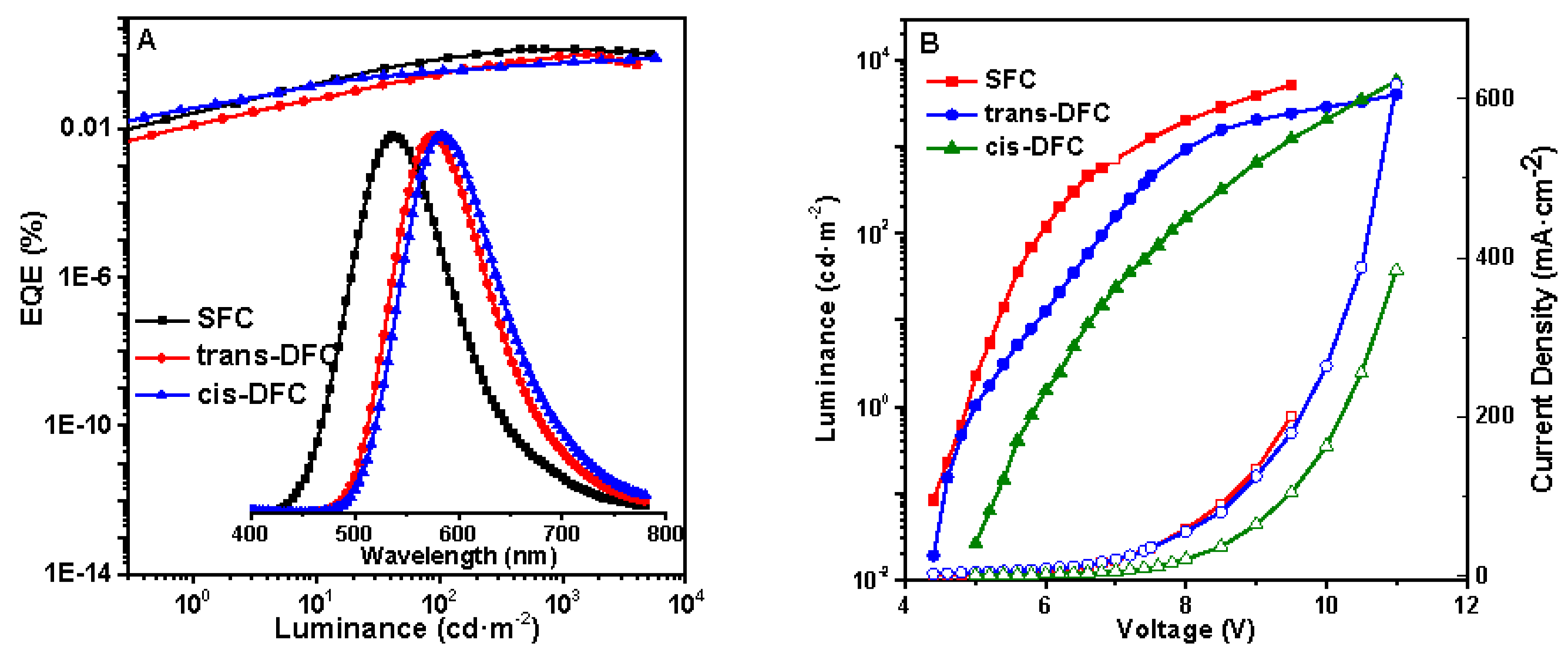
| Compounds | Td/Tg a (°C) | HOMO/LUMO b (eV) | λabsmax c (nm) | λemmax d (nm) | φfilm/powder e (%) |
|---|---|---|---|---|---|
| SFC | 442/94 | −5.48/−2.97 | 346/371 | --/513 | 51/46 |
| trans-DFC | 438/90 | −5.48/−3.51 | 400/426 | 570/587 | 40/54 |
| cis-DFC | 435/90 | −5.48/−3.52 | 387/406 | 568/600 | 44/55 |
| Compounds | Von a (V) | Lmax b (cd m−2) | LEmax c (cd A−1) | λELmax (nm) | EQE d (%) | CIE (x, y) |
|---|---|---|---|---|---|---|
| SFC | 5.0 | 5201 | 3.67 | 536 | 1.37 | (0.36,0.52) |
| trans-DFC | 5.0 | 4025 | 1.97 | 576 | 0.99 | (0.49,0.50) |
| cis-DFC | 6.0 | 5716 | 1.49 | 584 | 0.79 | (0.51,0.48) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, X.; Zhao, L.; Han, X.; Liu, H.; Gao, Y.; Tao, Y.; Zhang, H.; Yang, B.; Lu, P. Preparation of Cyano-Substituted Tetraphenylethylene Derivatives and Their Applications in Solution-Processable OLEDs. Molecules 2018, 23, 190. https://doi.org/10.3390/molecules23010190
Sun X, Zhao L, Han X, Liu H, Gao Y, Tao Y, Zhang H, Yang B, Lu P. Preparation of Cyano-Substituted Tetraphenylethylene Derivatives and Their Applications in Solution-Processable OLEDs. Molecules. 2018; 23(1):190. https://doi.org/10.3390/molecules23010190
Chicago/Turabian StyleSun, Xiaoyi, Lele Zhao, Xiao Han, Hui Liu, Yu Gao, Yanchun Tao, Haiquan Zhang, Bing Yang, and Ping Lu. 2018. "Preparation of Cyano-Substituted Tetraphenylethylene Derivatives and Their Applications in Solution-Processable OLEDs" Molecules 23, no. 1: 190. https://doi.org/10.3390/molecules23010190
APA StyleSun, X., Zhao, L., Han, X., Liu, H., Gao, Y., Tao, Y., Zhang, H., Yang, B., & Lu, P. (2018). Preparation of Cyano-Substituted Tetraphenylethylene Derivatives and Their Applications in Solution-Processable OLEDs. Molecules, 23(1), 190. https://doi.org/10.3390/molecules23010190




