Synthesis and Antimicrobial Studies of New Antibacterial Azo-Compounds Active against Staphylococcus aureus and Listeria monocytogenes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
| R1 | R2 | R3 | R4 | R5 | |
| 4a | O(CH2)3CH3 | H | CH3 | OH | CH3 |
| 4b | OCH2CH(CH3)2 | H | CH3 | OH | CH3 |
| 4c | OCH2C(CH3)3 | H | CH3 | OH | CH3 |
| 4d | OCH2CH2CH(CH3)2 | H | CH3 | OH | CH3 |
| 4e | OCH2CH(CH2CH3)CH2(CH2)2CH3 | H | CH3 | OH | CH3 |
| 4f | H | H | CH3 | OCH3 | CH3 |
| 4g | OCH2CH(CH3)2 | OH | H | OH | H |
| 4h | OCH2C(CH3)3 | OH | H | OH | H |
| 4i | OCH2CH2CH(CH3)2 | OH | H | OH | H |
| 4j | OCH2CH(CH2CH3)CH2(CH2)2CH3 | OH | H | OH | H |
| 4k | OH | OH | H | OH | H |
| 4l | CH3 | OH | H | OH | H |
| 4m | OCH3 | OH | H | OH | H |
2.2. Thermal and Optical Properties
2.3. Antibacterial Activity
3. Materials and Methods
3.1. General
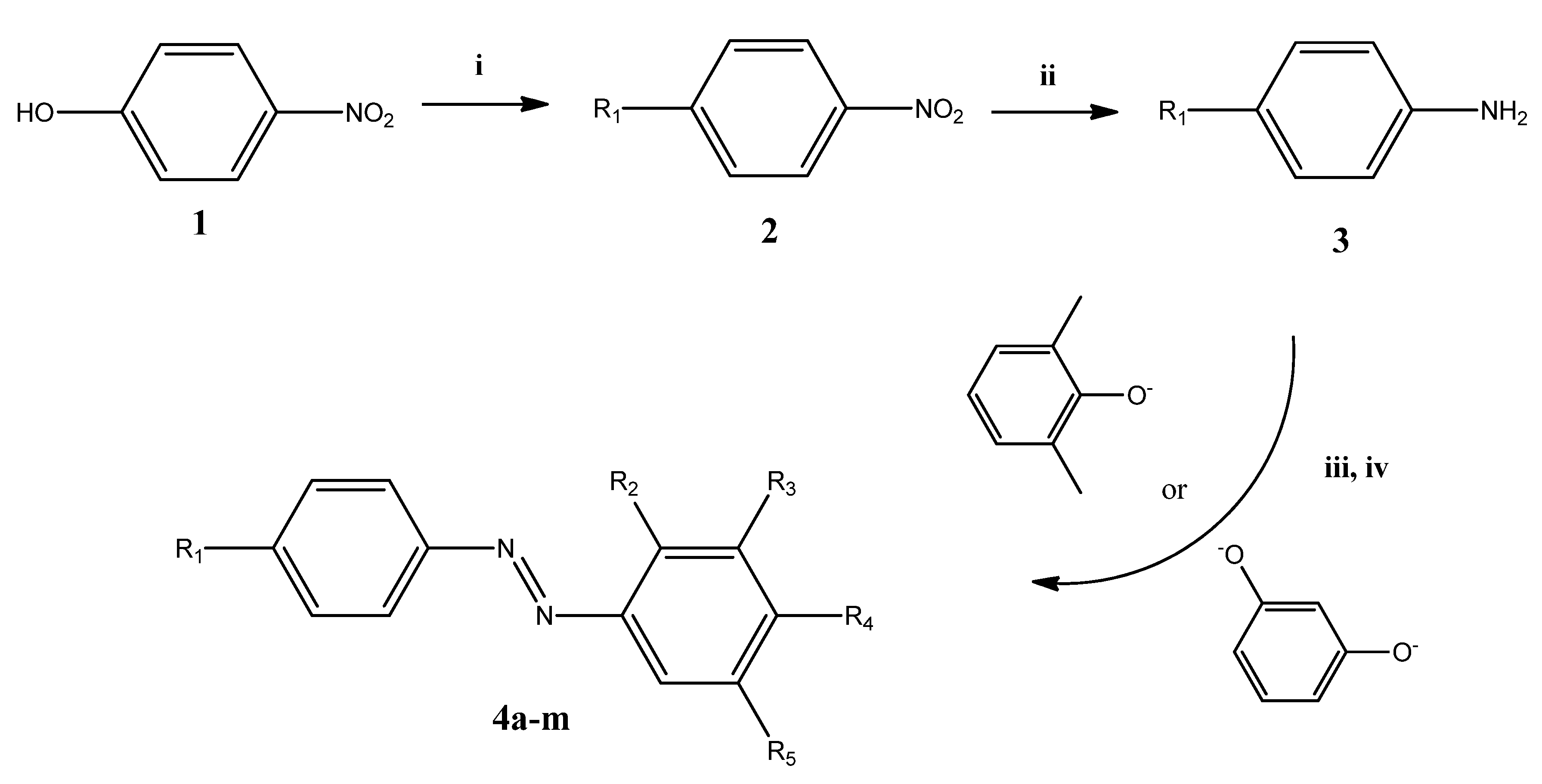
3.2. General Method of the Synthesis of (Phenyl-diazenyl)phenols Derivatives 4a–m
3.2.1. Synthesis of 1-Alkyloxy-4-nitrobenzene (2a–e, 2g–j)
3.2.2. Synthesis of 4-Alkyloxyaniline (3a–e, 3g–j)
3.2.3. Synthesis of Azobenzene Derivatives 4a–m
3.3. Antimicrobial Tests
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pirani, F.; Angelini, A.; Frascella, F.; Rizzo, R.; Ricciardi, S.; Descrovi, E. Light-driven reversible shaping of individual azopolymeric micro-pillars. Sci. Rep. 2016, 6, 31702. [Google Scholar] [CrossRef] [PubMed]
- Angelini, A.; Pirani, F.; Frascella, F.; Ricciardi, S.; Descrovi, E. Light-driven liquid microlenses. In Proceedings of the SPIE—The International Society for Optical Engineering, San Francisco, CA, USA, 28 January 2017. [Google Scholar]
- Pirani, F.; Angelini, A.; Ricciardi, S.; Frascella, F.; Descrovi, E. Laser-induced anisotropic wettability on azopolymeric micro-structures. Appl. Phys. Lett. 2017, 110, 101603. [Google Scholar] [CrossRef]
- Morvillo, P.; Diana, R.; Fontanesi, C.; Ricciardi, R.; Lanzi, M.; Mucci, A.; Tassinari, F.; Schenetti, L.; Minarini, C.; Parenti, F. Low band gap polymers for application in solar cells: Synthesis and characterization of thienothiophene-thiophene copolymers. Polym. Chem. 2014, 5, 2391–2400. [Google Scholar] [CrossRef]
- Parenti, F.; Morvillo, P.; Bobeico, E.; Diana, R.; Lanzi, M.; Fontanesi, C.; Tassinari, F.; Schenetti, L.; Mucci, A. (Alkylsulfanyl) bithiophene-alt-fluorene: Π-conjugated polymers for organic solar cells. Eur. J. Org. Chem. 2011, 2011, 5659–5667. [Google Scholar] [CrossRef]
- Acierno, D.; Amendola, E.; Bugatti, V.; Concilio, S.; Giorgini, L.; Iannelli, P.; Piotto, S.P. Synthesis and characterization of segmented liquid crystalline polymers with the azo group in the main chain. Macromolecules 2004, 37, 6418–6423. [Google Scholar] [CrossRef]
- Caruso, U.; Diana, R.; Panunzi, B.; Roviello, A.; Tingoli, M.; Tuzi, A. Facile synthesis of new pd (II) and cu (II) based metallomesogens from ligands containing thiophene rings. Inorg. Chem. Commun. 2009, 12, 1135–1138. [Google Scholar] [CrossRef]
- Attianese, D.; Petrosino, M.; Vacca, P.; Concilio, S.; Iannelli, P.; Rubino, A.; Bellone, S. Switching device based on a thin film of an azo-containing polymer for application in memory cells. IEEE Electron Device Lett. 2008, 29, 44–46. [Google Scholar] [CrossRef]
- Angiolini, L.; Benelli, T.; Giorgini, L.; Golemme, A.; Mazzocchetti, L.; Termine, R. Effect of a chiral substituent on the photochromic and photoconductive properties of a methacrylic polymer bearing side chain azocarbazole moieties. Dyes Pigment. 2014, 102, 53–62. [Google Scholar] [CrossRef]
- Borbone, F.; Caruso, U.; Causà, M.; Fusco, S.; Panunzi, B.; Roviello, A.; Shikler, R.; Tuzi, A. Series of O, N, O-tridentate ligands zinc (II) complexes with high solid-state photoluminescence quantum yield. Eur. J. Inorg. Chem. 2014, 2014, 2695–2703. [Google Scholar] [CrossRef]
- Caruso, U.; Panunzi, B.; Roviello, A.; Tuzi, A. Fluorescent metallopolymers with Zn (II) in a schiff base/phenoxide coordination environment. Inorg. Chem. Commun. 2013, 29, 138–140. [Google Scholar] [CrossRef]
- Borbone, F.; Carella, A.; Caruso, U.; Roviello, G.; Tuzi, A.; Dardano, P.; Lettieri, S.; Maddalena, P.; Barsella, A. Large second-order nlo activity in poly (4-vinylpyridine) grafted with pdii and cuii chromophoric complexes with tridentate bent ligands containing heterocycles. Eur. J. Inorg. Chem. 2008, 2008, 1846–1853. [Google Scholar] [CrossRef]
- Caruso, U.; Diana, R.; Fort, A.; Panunzi, B.; Roviello, A. Synthesis of Polymers Containing Second Order NLO-Active Thiophene and Thiazole Based Chromophores. In Macromolecular Symposia; Wiley Online Library: Naples, Italy, 2006; Volume 234, pp. 87–93. [Google Scholar] [CrossRef]
- Concilio, S.; Iannelli, P.; Sessa, L.; Olivieri, R.; Porta, A.; De Santis, F.; Pantani, R.; Piotto, S. Biodegradable antimicrobial films based on poly (lactic acid) matrices and active azo compounds. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Piotto, S.; Concilio, S.; Sessa, L.; Iannelli, P.; Porta, A.; Calabrese, E.C.; Galdi, M.R.; Incarnato, L. Novel antimicrobial polymer films active against bacteria and fungi. Polym. Compos. 2013, 34, 1489–1492. [Google Scholar] [CrossRef]
- Piotto, S.; Concilio, S.; Sessa, L.; Porta, A.; Calabrese, E.C.; Zanfardino, A.; Varcamonti, M.; Iannelli, P. Small azobenzene derivatives active against bacteria and fungi. Eur. J. Med. Chem. 2013, 68, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Sessa, L.; Concilio, S.; Iannelli, P.; De Santis, F.; Porta, A.; Piotto, S. Antimicrobial azobenzene compounds and their potential use in biomaterials. In Proceedings of the AIP Conference, Oudeniz, Turkey, 16–19 April 2015. [Google Scholar]
- Farber, J.M. Thermal resistance of listeria monocytogenes in foods. Int. J. Food Microbiol. 1989, 8, 285–291. [Google Scholar] [CrossRef]
- Yousef, A.H.E.; Marth, E.L.H. Inactivation of listeria monocytogenes by ultraviolet energy. J. Food Sci. 1988, 53, 571–573. [Google Scholar] [CrossRef]
- Gill, C.O.; Reichel, M.P. Growth of the cold-tolerant pathogens yersinia enterocolitica, aeromonas hydrophila and listeria monocytogenes on high-pH beef packaged under vacuum or carbon dioxide. Food Microbiol. 1989, 6, 223–230. [Google Scholar] [CrossRef]
- Santos, M.I.; Lima, A.I.; Monteiro, S.A.; Ferreira, R.M.; Pedroso, L.; Sousa, I.; Ferreira, M.A. Preliminary study on the effect of fermented cheese whey on listeria monocytogenes, escherichia coli O157: H7, and salmonella goldcoast populations inoculated onto fresh organic lettuce. Foodborne Pathog. Dis. 2016, 13, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Gahan, C.G.M.; Collins, J.K. Listeriosis: Biology and implications for the food industry. Trends Food Sci. Technol. 1991, 2, 89–93. [Google Scholar] [CrossRef]
- Farber, J.; Peterkin, P. Listeria monocytogenes, a food-borne pathogen. Microbiol. Rev. 1991, 55, 476–511. [Google Scholar] [PubMed]
- Swaminathan, B.; Gerner-Smidt, P. The epidemiology of human listeriosis. Microbes Infect. 2007, 9, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Cossart, P. Listeriology (1926–2007): The rise of a model pathogen. Microbes Infect. 2007, 9, 1143–1146. [Google Scholar] [CrossRef] [PubMed]
- Sessa, L.; Petrone, A.M.; Porta, A.; Diana, R.; Iannelli, P.; Piotto, S. Structure modification of an active azo-compound as a route to new antimicrobial compounds. Molecules 2017, 22, 875. [Google Scholar]
- Wiegand, I.; Hilpert, K.; Hancock, R. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nature Protoc. 2008, 3, 163. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
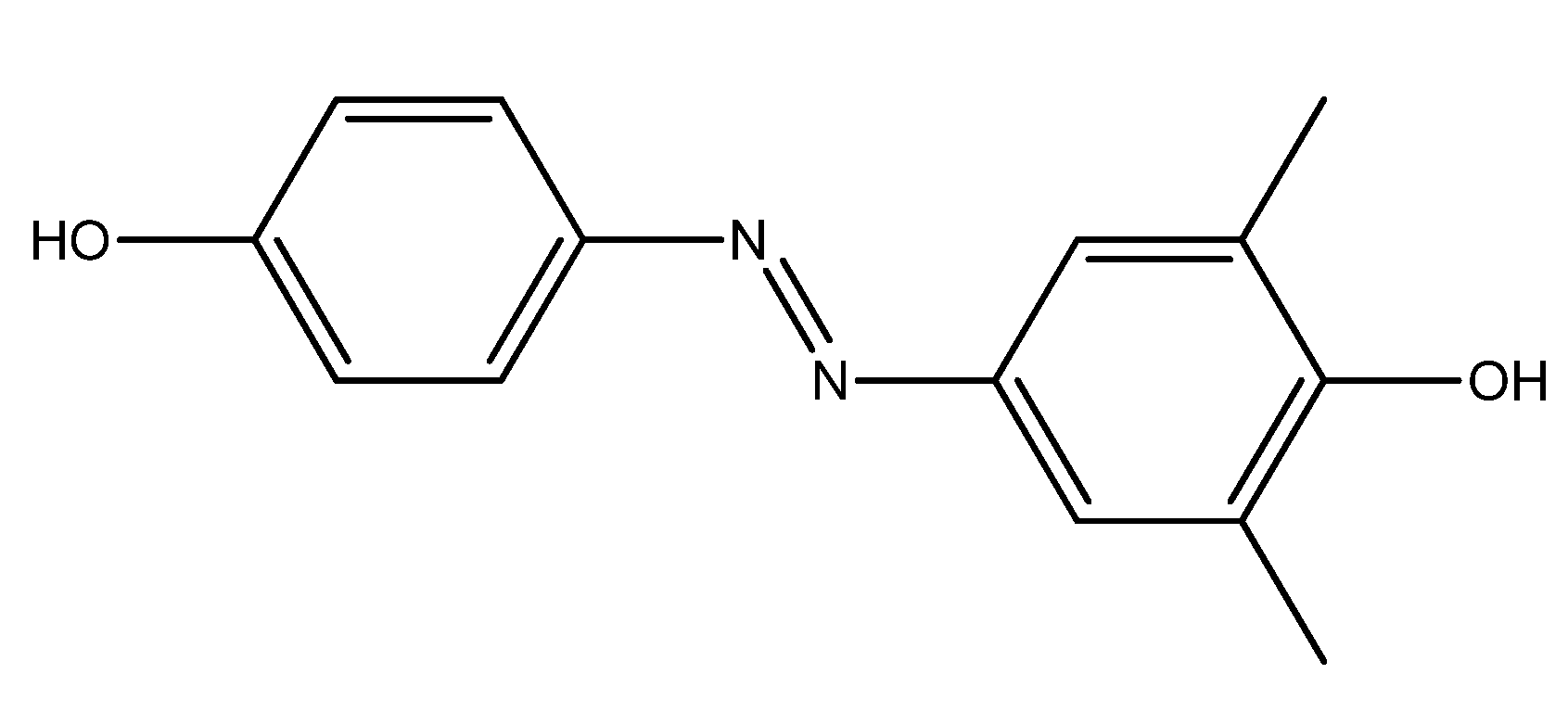
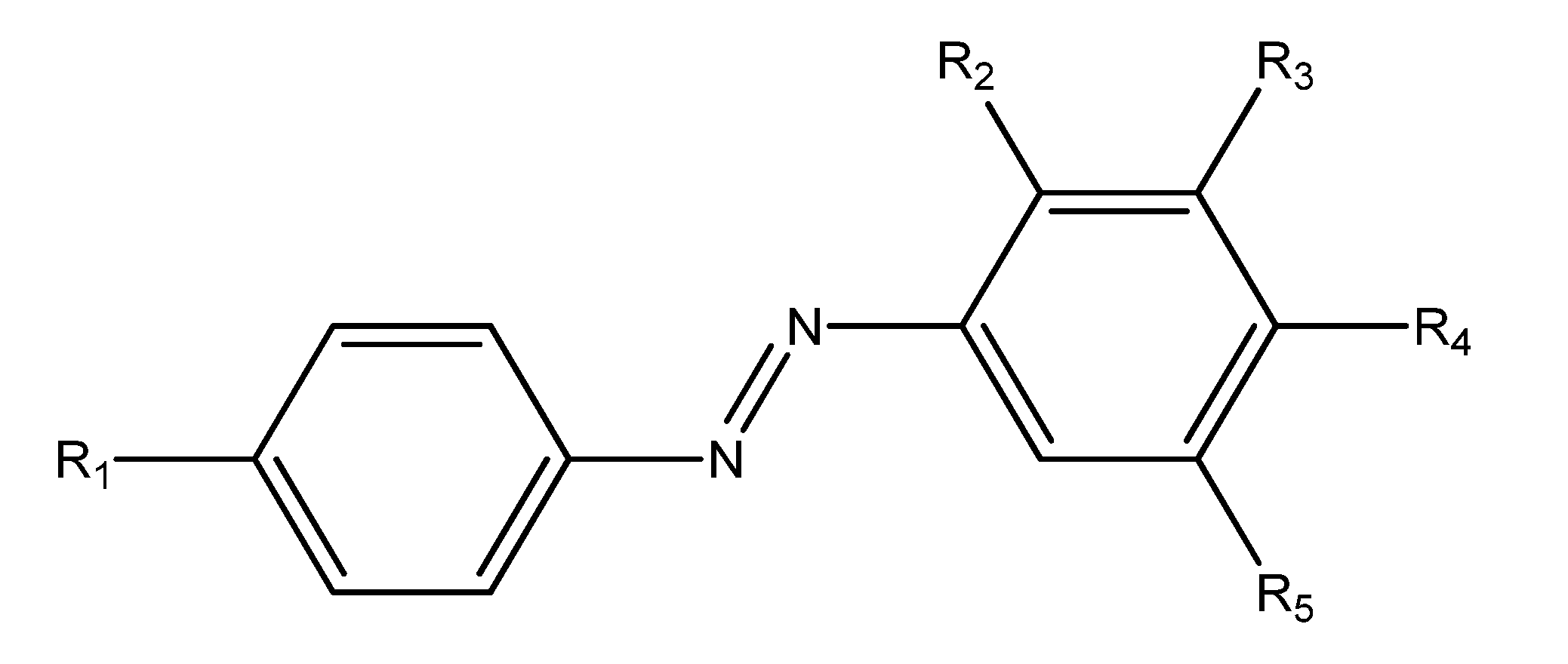
| R1 | R2 | R3 | R4 | R5 | |
| 4a | O(CH2)3CH3 | H | CH3 | OH | CH3 |
| 4b | OCH2CH(CH3)2 | H | CH3 | OH | CH3 |
| 4c | OCH2C(CH3)3 | H | CH3 | OH | CH3 |
| 4d | OCH2CH2CH(CH3)2 | H | CH3 | OH | CH3 |
| 4e | OCH2CH(CH2CH3)CH2(CH2)2CH3 | H | CH3 | OH | CH3 |
| 4f | H | H | CH3 | OCH3 | CH3 |
| 4g | OCH2CH(CH3)2 | OH | H | OH | H |
| 4h | OCH2C(CH3)3 | OH | H | OH | H |
| 4i | OCH2CH2CH(CH3)2 | OH | H | OH | H |
| 4j | OCH2CH(CH2CH3)CH2(CH2)2CH3 | OH | H | OH | H |
| 4k | OH | OH | H | OH | H |
| 4l | CH3 | OH | H | OH | H |
| 4m | OCH3 | OH | H | OH | H |
| Molecule | Thermal Characterization | Optical Characterization | ||||
|---|---|---|---|---|---|---|
| Tm (°C) | ΔHm (J/g) | Tc (°C) | ΔHc (J/g) | λmax (nm) | εmax (L mol−1 cm−1) | |
| 4a | 88.7 | 76.5 | 53.35 | 66.17 | 360 | 26,000 |
| 4b | 126.5 | 74.3 | - | - | 360 | 25,000 |
| 4c | 85.00 | 52.6 | - | - | 360 | 26,100 |
| 116.7 | 19.5 | |||||
| 126.0 | 10.8 | |||||
| 4d | 74.2 | 88.4 | - | - | 361 | 38,000 |
| 4e | 59.2 | 56.1 | - | - | 361 | 33,100 |
| 4f * | - | - | - | - | ||
| 4g | 156.3 | 54.2 | - | - | 384 | 3300 |
| 4h | 179.3 | 80.2 | - | - | 384 | 29,500 |
| 4i | 125.3 | 73.6 | - | - | 384 | 35,700 |
| 4j | 82.6 | 47.2 | - | - | 384 | 3000 |
| 4k | 235 | 75.4 | - | - | 382 | 8300 |
| 4l | 238 | 91.5 | - | - | 447 | 7800 |
| 4m | 160 | 24.8 | - | - | 344 | 4600 |
| MIC100 (μg/mL) after 24 h | ||||
|---|---|---|---|---|
| Staphylococcus aureus | Listeria monocytogenes | Escherichia coli | Pseudomonas aeruginosa | |
| 4a | 32 | >128 | >128 | >128 |
| 4b | 128 | 128 | >128 | >128 |
| 4c | >128 | >128 | >128 | >128 |
| 4d | 4 | 8 | >128 | >128 |
| 4e | 12 | 48 | >128 | >128 |
| 4f | >128 | >128 | >128 | >128 |
| 4g | >128 | 96 | >128 | >128 |
| 4h | 4 | 8 | >128 | >128 |
| 4i | 4 | 8 | >128 | >128 |
| 4j | 16 | 16 | >128 | >128 |
| 4k | >128 | >128 | >128 | >128 |
| 4l | >32 * | >32 * | >32 * | >32 * |
| 4m | 16 | 16 | >128 | >128 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Piotto, S.; Concilio, S.; Sessa, L.; Diana, R.; Torrens, G.; Juan, C.; Caruso, U.; Iannelli, P. Synthesis and Antimicrobial Studies of New Antibacterial Azo-Compounds Active against Staphylococcus aureus and Listeria monocytogenes. Molecules 2017, 22, 1372. https://doi.org/10.3390/molecules22081372
Piotto S, Concilio S, Sessa L, Diana R, Torrens G, Juan C, Caruso U, Iannelli P. Synthesis and Antimicrobial Studies of New Antibacterial Azo-Compounds Active against Staphylococcus aureus and Listeria monocytogenes. Molecules. 2017; 22(8):1372. https://doi.org/10.3390/molecules22081372
Chicago/Turabian StylePiotto, Stefano, Simona Concilio, Lucia Sessa, Rosita Diana, Gabriel Torrens, Carlos Juan, Ugo Caruso, and Pio Iannelli. 2017. "Synthesis and Antimicrobial Studies of New Antibacterial Azo-Compounds Active against Staphylococcus aureus and Listeria monocytogenes" Molecules 22, no. 8: 1372. https://doi.org/10.3390/molecules22081372
APA StylePiotto, S., Concilio, S., Sessa, L., Diana, R., Torrens, G., Juan, C., Caruso, U., & Iannelli, P. (2017). Synthesis and Antimicrobial Studies of New Antibacterial Azo-Compounds Active against Staphylococcus aureus and Listeria monocytogenes. Molecules, 22(8), 1372. https://doi.org/10.3390/molecules22081372











