The Anti-Inflammatory Properties of Citrus wilsonii Tanaka Extract in LPS-Induced RAW 264.7 and Primary Mouse Bone Marrow-Derived Dendritic Cells
Abstract
1. Introduction
2. Results
2.1. Profile of the ZQE
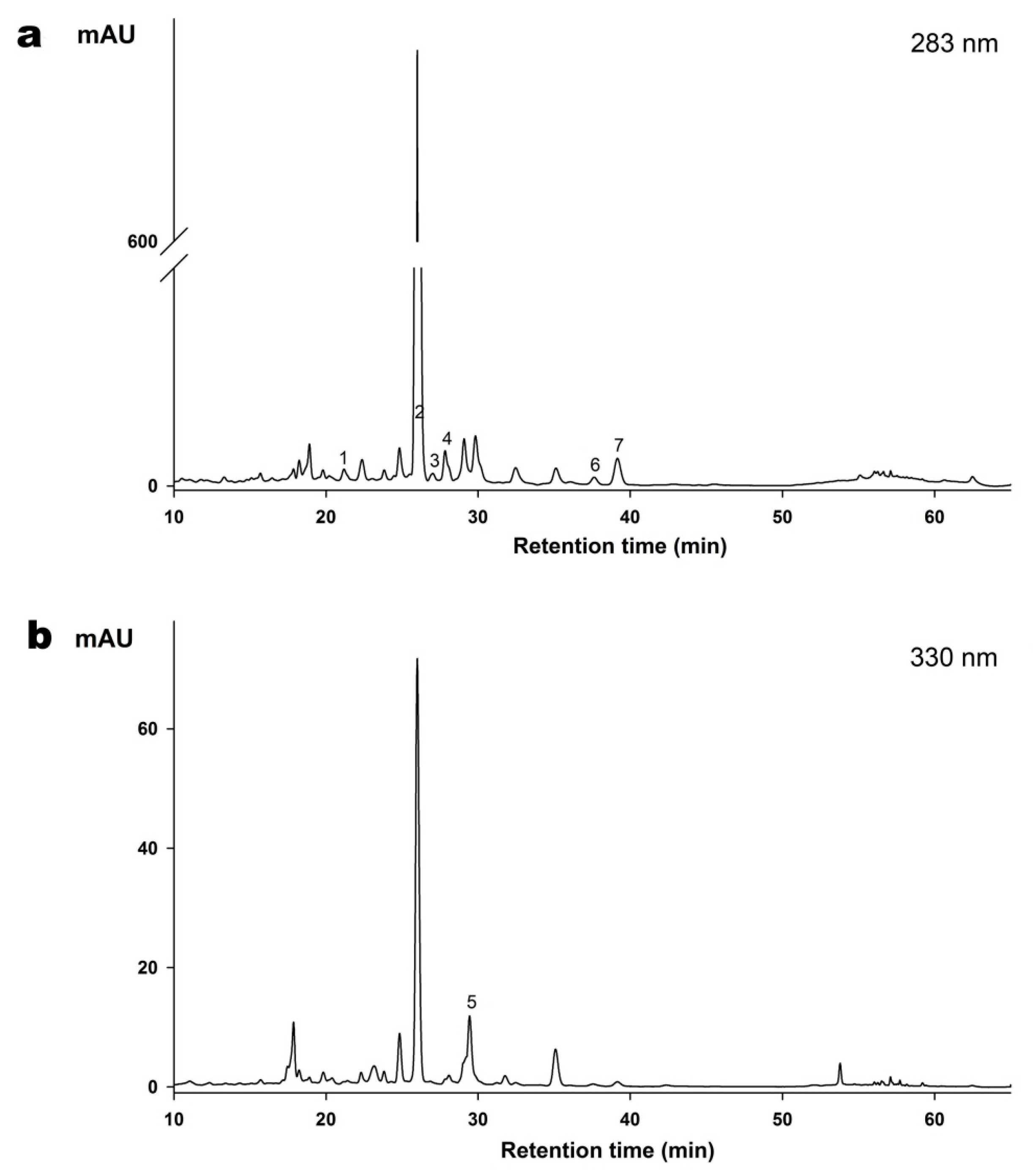
2.2. Determination of Flavonoids Composition of the ZQE by HPLC
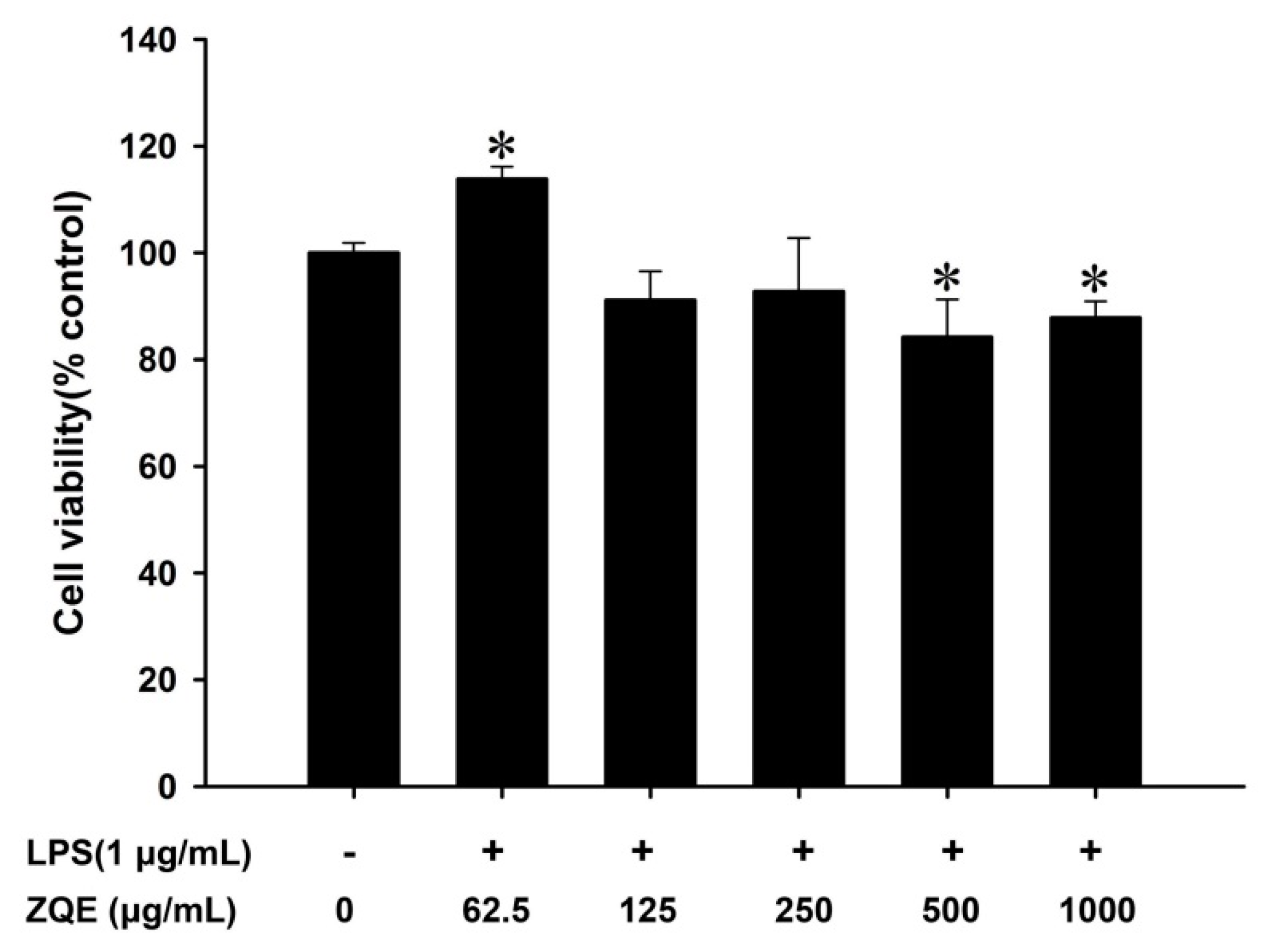
2.3. Effects of ZQE on Cell Viability in RAW 264.7 Macrophages
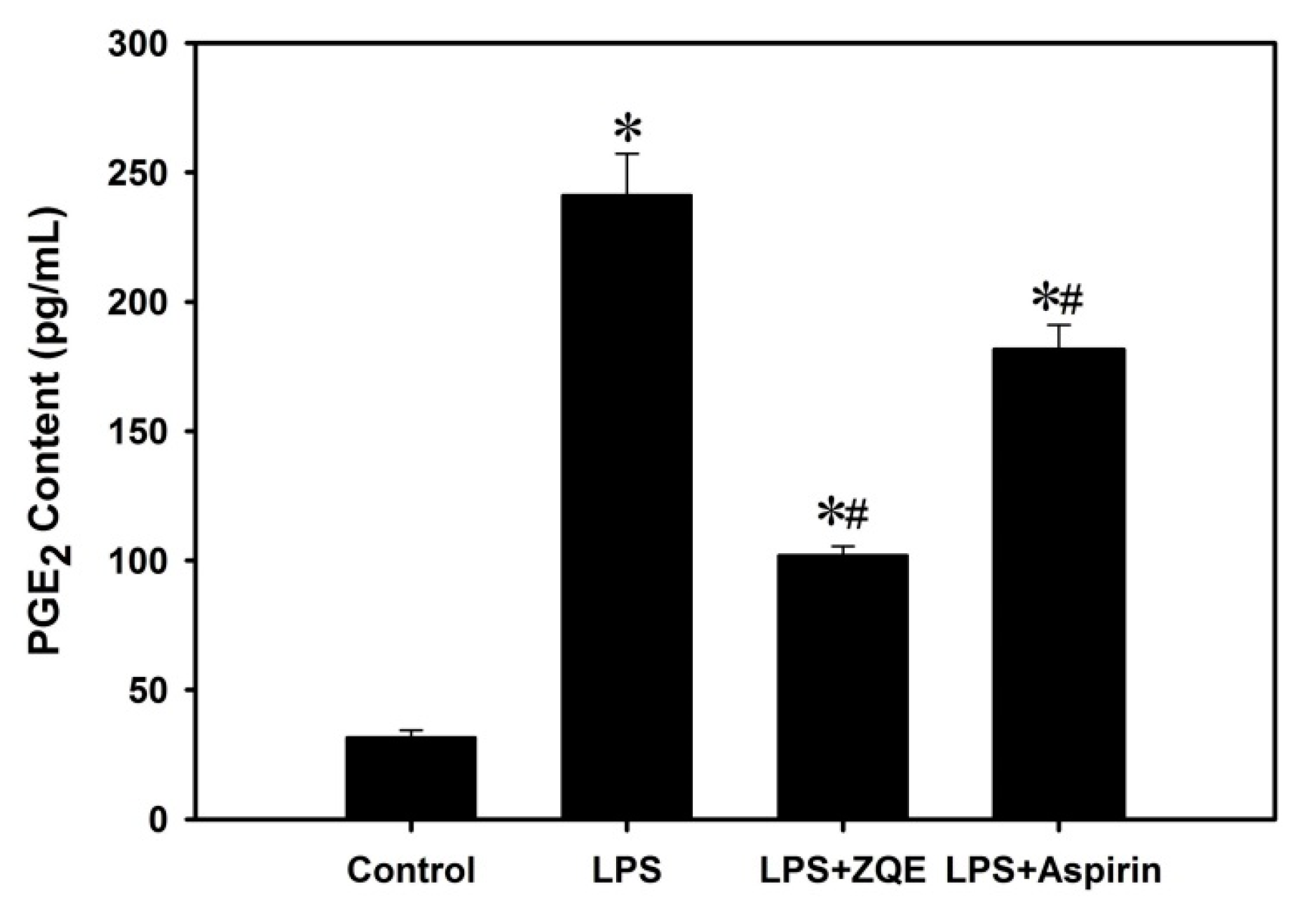
2.4. Effects of ZQE on the Production of PGE2 in LPS-Induced RAW 264.7 Macrophages
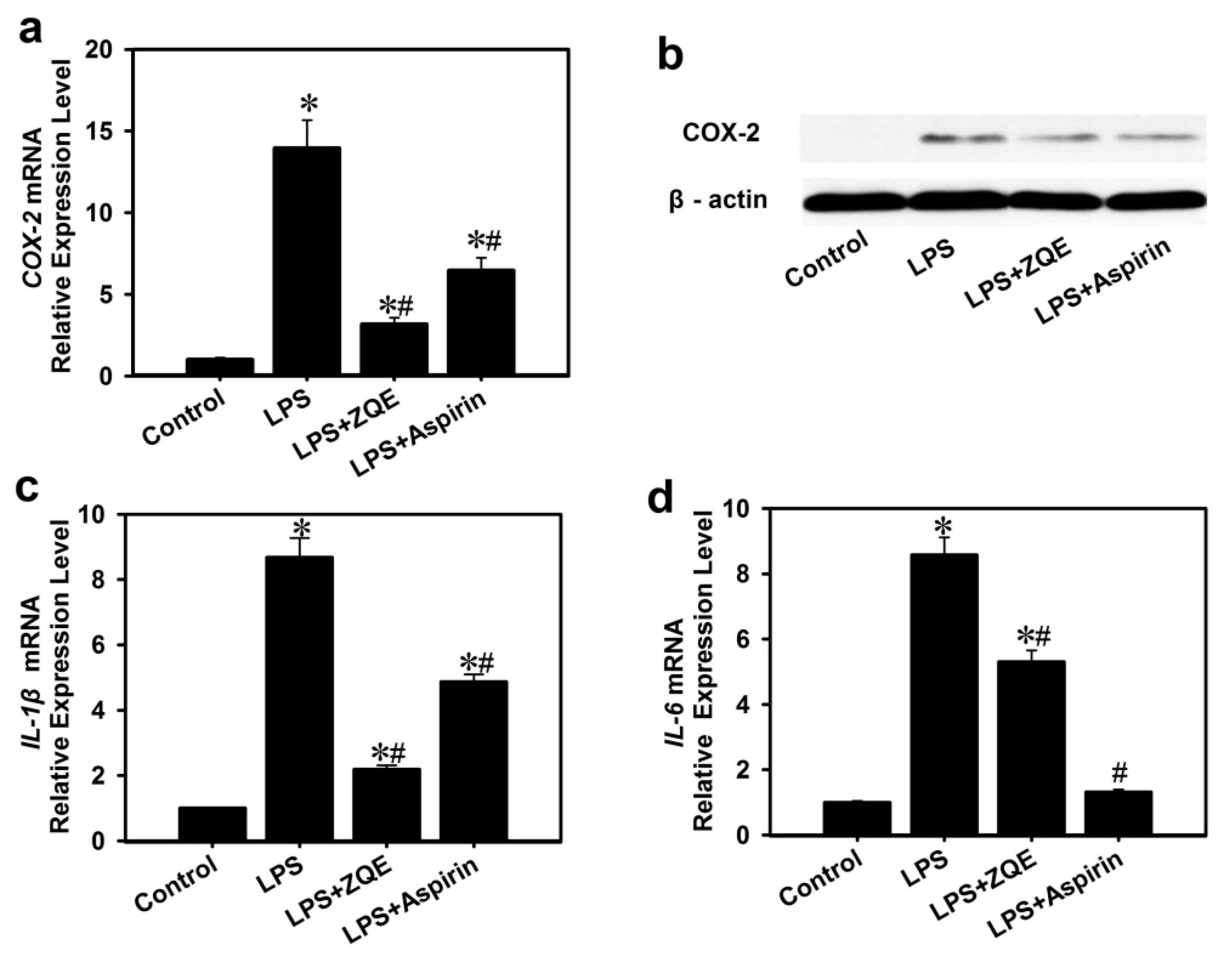
2.5. Effects of ZQE on Expression of Pro-Inflammatory Cytokines in RAW 264.7 Macrophages
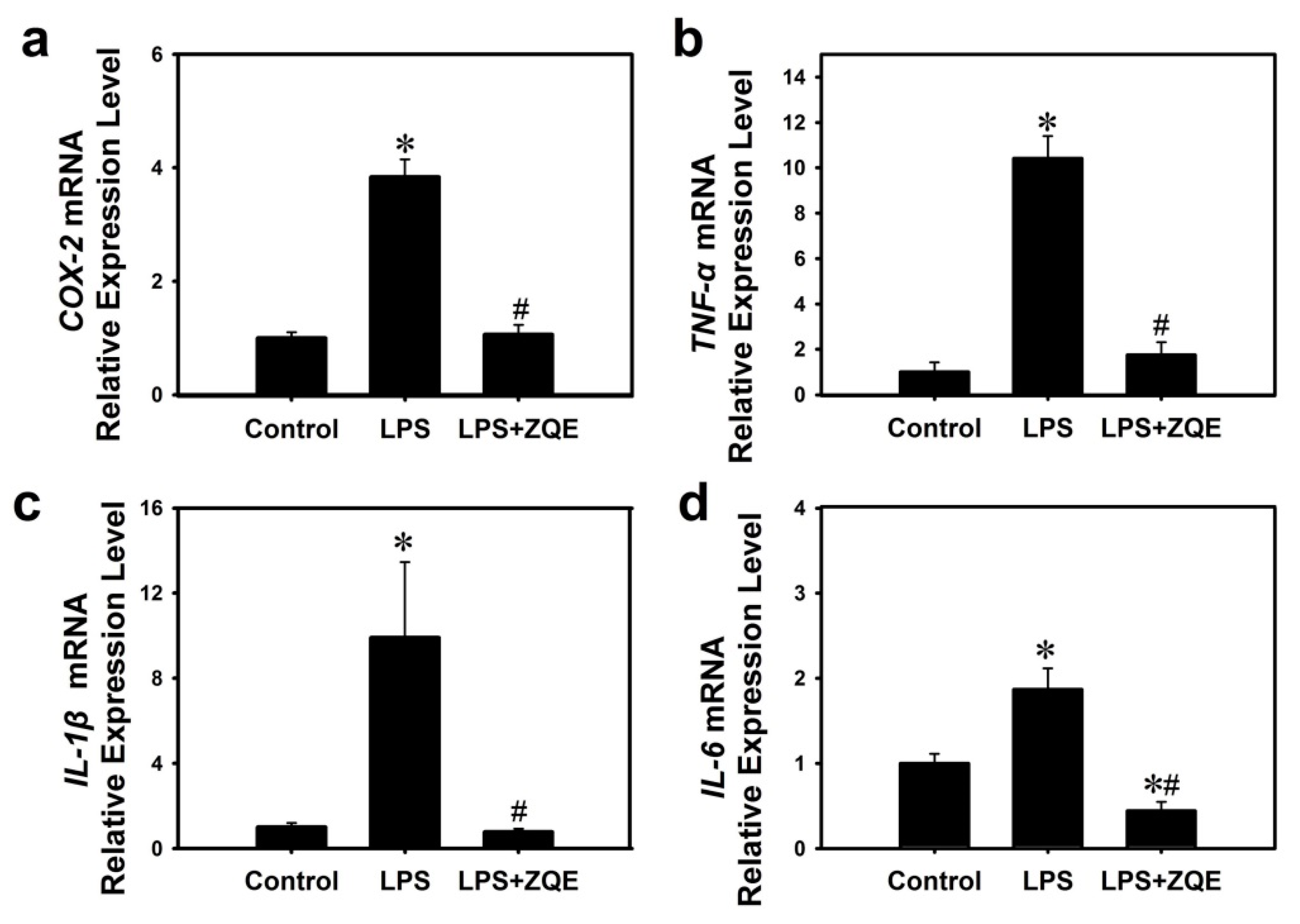
2.6. Effects of ZQE on the Gene Expression of Inflammatory Mediators and Cytokines in LPS-Induced Primary BMDCs
3. Discussion
4. Materials and Methods
4.1. Reagents
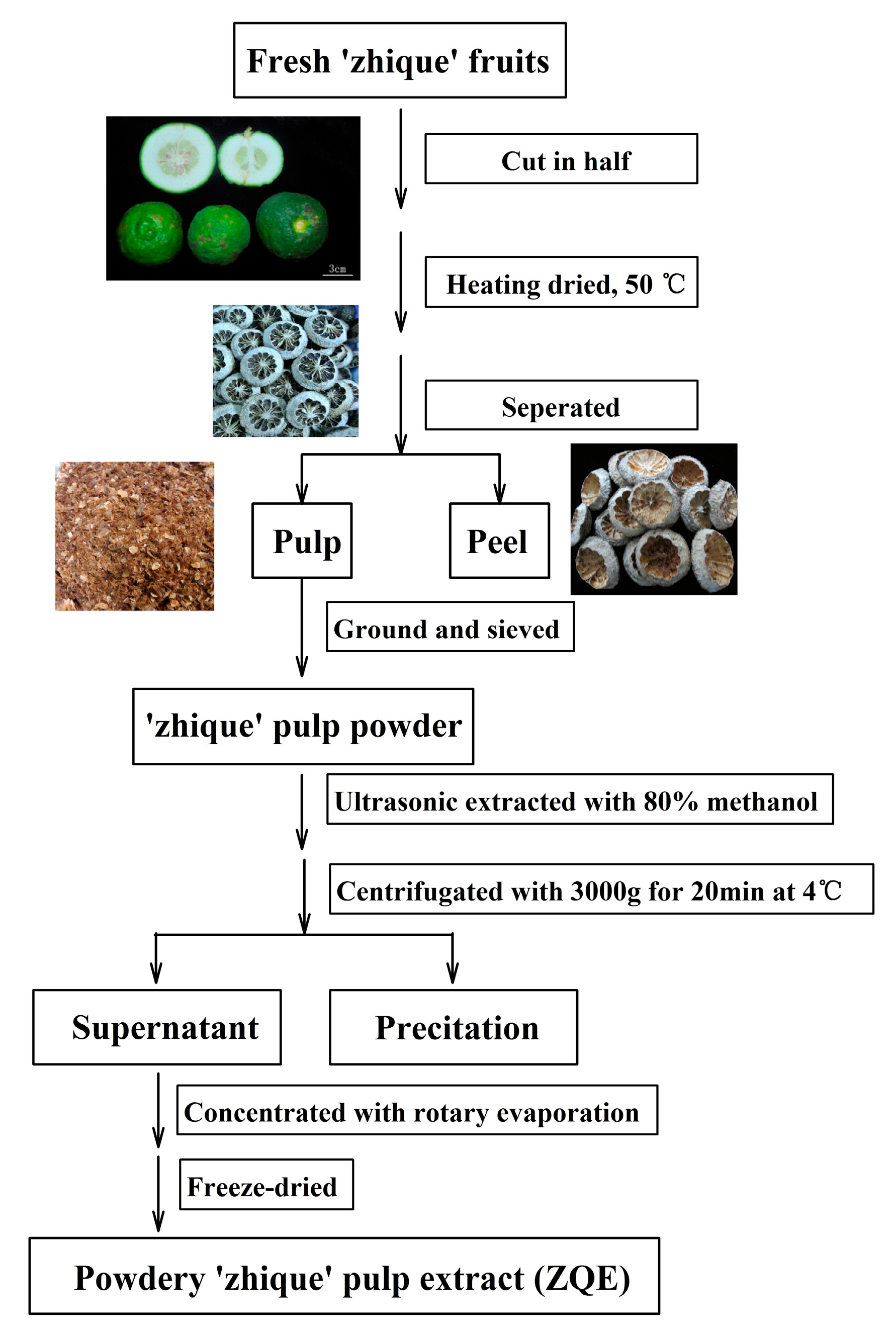
4.2. Extraction of ‘Zhique’ Pulp Extract
4.3. Analysis of the ZQE Profile by LC-ESI-MS/MS Analysis
4.4. Analysis of Flavonoids in ZQE
4.5. Cell Culture
4.6. Measurement of Cell Viability
4.7. Measurement of PGE2
4.8. RNA Isolation and qPCR Analysis
4.9. Western Blot Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zhou, K.L.; Ye, Y.M. Chinese Fruit Trees-CITRUS; China Forestry Press: Beijing, China, 2010; p. 101. [Google Scholar]
- Ding, D.K. Citrus; Shaanxi Publishing Media Group Sanqin Press: Xi’an, China, 2014; p. 56. [Google Scholar]
- Chinese Pharmacopoeia Commission. Pharmacopeia of the People’s Republic of China; Chemical Industry Press: Beijing, China, 2010; p. 242. [Google Scholar]
- Dan, B.; Andrew, G. Chinese Herbal Medicine, 8th ed.; Eastland Press: Seattle, WA, USA, 1986; pp. 334–335. [Google Scholar]
- Irwin, R.S.; Ownbey, R.; Cagle, P.T.; Baker, S.; Fraire, A.E. Interpreting the histopathology of chronic cough: A prospective, controlled, comparative study. Chest 2006, 130, 362–370. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.G.; Lin, X.C.; Diao, J.X.; Yu, Z.L.; Li, K. Pi (Spleen)-deficiency syndrome in tumor microenvironment is the pivotal pathogenesis of colorectal cancer immune escape. Chin. J. Integr. Med. 2016, 22, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.Y.; Gao, W.Y.; Gao, Y.; Man, S.L.; Huang, L.Q.; Liu, C.X. Identification of chemical constituents in extracts and rat plasma from Fructus Aurantii by UPLC-PDA-Q-TOF/MS. Phytochem. Anal. 2011, 22, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Guo, L.; Liu, K.; Liu, E.H.; Li, P. Characterization and classification of seven Citrus herbs by liquid chromatography-quadrupole time-of-flight mass spectrometry and genetic algorithm optimized support vector machines. J. Chromatogr. A 2014, 1339, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Duan, L.; Guo, L.; Dou, L.L.; Dong, X.; Zhou, P.; Liu, E.H. Chemical and biological comparison of the fruit extracts of Citrus wilsonii Tanaka and Citrus medica L. Food Chem. 2015, 173, 54–60. [Google Scholar] [CrossRef]
- Alam, M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef] [PubMed]
- García-Lafuente, A.; Guillamón, E.; Villares, A.; Rostagno, M.A.; Martínez, J.A. Flavonoids as anti-inflammatory agents: Implications in cancer and cardiovascular disease. Inflamm. Res. 2009, 58, 537–552. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.M.; Ahmed, O.M.; Ashour, M.B.; Abdel-Moneim, A. In vivo and in vitro antidiabetic effects of citrus flavonoids; a study on the mechanism of action. Int. J. Diabetes Dev. Ctries. 2015, 35, 250–263. [Google Scholar] [CrossRef]
- Kang, S.R.; Park, K.I.; Park, H.S.; Lee, D.H.; Kim, J.A.; Nagappan, A.; Kim, E.H.; Lee, W.S.; Shin, S.C.; Park, M.K. Anti-inflammatory effect of flavonoids isolated from Korea Citrus aurantium L. on lipopolysaccharide-induced mouse macrophage RAW 264.7 cells by blocking of nuclear factor-κB (NF-κB) and mitogen-activated protein kinase (MAPK) signalling pathways. Food Chem. 2011, 129, 1721–1728. [Google Scholar] [CrossRef]
- Mukherjee, N.; Kumar, A.P.; Ghosh, R. DNA Methylation and Flavonoids in Genitourinary Cancers. Curr. Pharmacol. Rep. 2015, 1, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Ben, L.; Dudonne, A.; Desjardins, S.Y.; Grenier, D. Wild Blueberry (Vaccinium angustifolium Ait.) Polyphenols Target Fusobacterium nucleatum and the Host Inflammatory Response: Potential Innovative Molecules for Treating Periodontal Diseases. J. Agric. Food Chem. 2015, 63, 6999–7008. [Google Scholar] [CrossRef]
- Hasturk, H.; Kantarci, A.; Van Dyke, T.E. Oral inflammatory diseases and systemic inflammation: Role of the macrophage. Front. Immunol. 2012, 3, 118. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Jobin, C. The flavonoid luteolin prevents lipopolysaccharide-induced NF-κB signalling and gene expression by blocking IκB kinase activity in intestinal epithelial cells and bone-marrow derived dendritic cells. Immunology 2005, 115, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Le, D.D.; Thao, N.P.; Tai, B.H.; Luyen, B.T.T.; Kim, S.; Koo, J.E.; Koh, Y.S.; Cuong, N.T.; Thanh, N.V.; Cuong, N.X. Chemical constituents from Kandelia candel with their inhibitory effects on pro-inflammatory cytokines production in LPS-stimulated bone marrow-derived dendritic cells (BMDCs). Bioorg. Med. Chem. Lett. 2015, 25, 1412–1416. [Google Scholar] [CrossRef]
- Liu, Y.; Su, W.W.; Wang, S.; Li, P.B. Naringin inhibits chemokine production in an LPS-induced RAW 264.7 macrophage cell line. Mol. Med. Rep. 2012, 6, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.S.; Lee, J.H.; Seok, J.H.; Hong, J.H.; Lee, Y.S.; Lim, J.H.; Kim, Y.M.; Hur, G.M. Acetaminophen inhibits iNOS gene expression in RAW 264.7 macrophages: Differential regulation of NF-κB by acetaminophen and salicylates. Biochem. Biophys. Res. Commun. 2000, 272, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Park, J.W.; Kwon, O.K.; Kim, J.H.; Oh, S.R.; Lee, H.K.; Bach, T.T.; Quang, B.H.; Ahn, K.S. Anti-inflammatory activity of a methanol extract from Ardisia tinctoria on mouse macrophages and paw edema. Mol. Med. Rep. 2014, 9, 1388–1394. [Google Scholar] [CrossRef] [PubMed]
- Balan, T.; Sani, M.H.; Mumtaz Ahmad, S.H.; Suppaiah, V.; Mohtarrudin, N.; Zakaria, Z.A. Antioxidant and anti-inflammatory activities contribute to the prophylactic effect of semi-purified fractions obtained from the crude methanol extract of Muntingia calabura leaves against gastric ulceration in rats. J. Ethnopharmacol. 2015, 164, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Song, Q.; Wang, L.; Xie, T.Z.; Wu, X.; Wang, P.; Yin, G.; Ye, W.C.; Wang, T.J. Antitussive, expectorant and anti-inflammatory activities of different extracts from Exocarpium Citri grandis. J. Ethnopharmacol. 2014, 156, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Ke, Z.L.; Pan, Y.; Xu, X.D.; Nie, C.; Zhou, Z.Q. Citrus flavonoids and human Cancers. J. Food Nutr. Res. 2015, 3, 341–351. [Google Scholar] [CrossRef]
- Burkina, V.; Zlabek, V.; Halsne, R.; Ropstad, E.; Zamaratskaia, G. In vitro effects of the citrus flavonoids diosmin, naringenin and naringin on the hepatic drug-metabolizing CYP3A enzyme in human, pig, mouse and fish. Biochem. Pharmacol. 2016, 110, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Buscemi, S.; Rosafio, G.; Arcoleo, G.; Mattina, A.; Canino, B.; Montana, M.; Verga, S.; Rini, G. Effects of red orange juice intake on endothelial function and inflammatory markers in adult subjects with increased cardiovascular risk. Am. J. Clin. Nutr. 2012, 95, 1089–1095. [Google Scholar] [CrossRef] [PubMed]
- Kandhare, A.D.; Alam, J.; Patil, M.V.; Sinha, A.; Bodhankar, S.L. Wound healing potential of naringin ointment formulation via regulating the expression of inflammatory, apoptotic and growth mediators in experimental rats. Pharm. Biol. 2015, 54, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wu, H.; Nie, Y.C.; Chen, J.L.; Su, W.W.; Li, P.B. Naringin attenuates acute lung injury in LPS-treated mice by inhibiting NF-κB pathway. Int. Immunopharmacol. 2011, 11, 1606–1612. [Google Scholar] [CrossRef] [PubMed]
- Nie, Y.C.; Wu, H.; Li, P.B.; Luo, Y.L.; Long, K.; Xie, L.M.; Shen, J.G.; Su, W.W. Anti-inflammatory effects of naringin in chronic pulmonary neutrophilic inflammation in cigarette smoke-exposed rats. J. Med. Food. 2012, 15, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.F.; Attia, S.M.; Bakheet, S.A.; Zoheir, K.M.; Ansari, M.A.; Korashy, H.M.; Abdel-Hamied, H.E.; Ashour, A.E.; Abd-Allah, A.R. Naringin attenuates the development of carrageenan-induced acute lung inflammation through inhibition of NF-κb, STAT3 and pro-inflammatory mediators and enhancement of IκBα and anti-inflammatory cytokines. Inflammation 2015, 38, 846–857. [Google Scholar] [CrossRef] [PubMed]
- Jung, U.J.; Leem, E.; Kim, S.R. Naringin: A protector of the nigrostriatal dopaminergic projection. Exp. Neurobiol. 2014, 23, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Bibbins-Domingo, K. Aspirin use for the primary prevention of cardiovascular disease and colorectal cancer: U.S. preventive services task force recommendation statement. Ann. Intern. Med. 2016, 164, 836–845. [Google Scholar] [CrossRef] [PubMed]
- Guirguis-Blake, J.M.; Evans, C.V.; Senger, C.A.; O’Connor, E.A.; Whitlock, E.P. Aspirin for the primary prevention of cardiovascular events: A systematic evidence review for the U.S. preventive services task force. Ann. Intern. Med. 2016, 164, 804–813. [Google Scholar] [CrossRef] [PubMed]
- Med, A.I. Aspirin for the primary prevention of cardiovascular events: Recommendation and rationale. Ann. Intern. Med. 2002, 136, 157–160. [Google Scholar]
- Grilli, M.; Pizzi, M.; Memo, M.; Spano, P. Neuroprotection by aspirin and sodium salicylate through blockade of NF-KappaB activation. Science 1996, 274, 1383–1385. [Google Scholar] [CrossRef] [PubMed]
- Kopp, E.; Ghosh, S. Inhibition of NF-κB by sodium salicylate and aspirin. Science 1994, 265, 956–959. [Google Scholar] [CrossRef] [PubMed]
- Wallace, J.L.; Vong, L. NSAID-induced gastrointestinal damage and the design of GI-sparing NSAIDs. Curr. Opin. Investig. Drugs 2008, 9, 1151–1156. [Google Scholar] [PubMed]
- Scott, M.J.; Billiar, T.R. β2-integrin-induced p38 MAPK activation is a key mediator in the CD14/TLR4/MD2-dependent uptake of lipopolysaccharide by hepatocytes. J. Biol. Chem. 2008, 283, 29433–29446. [Google Scholar] [CrossRef] [PubMed]
- Bognar, E.; Sarszegi, Z.; Szabo, A.; Debreceni, B.; Kalman, N.; Tucsek, Z.; Sumegi, B.; Gallyas, F., Jr. Antioxidant and anti-inflammatory effects in RAW264.7 macrophages of malvidin, a major red wine polyphenol. PLoS ONE 2013, 8, e65355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Yang, Y.J.; Yan, S.X.; Liu, J.T.; Xu, Z.M.; Yu, J.P.; Song, Y.J.; Zhang, A.D.; Jin, M.L. A novel pro-inflammatory protein of Streptococcus suis 2 induces the Toll-like receptor 2-dependent expression of pro-inflammatory cytokines in RAW 264.7 macrophages via activation of ERK1/2 pathway. Front. Microbiol. 2015, 6, 178. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.L.; Hu, X.R.; Yan, H.Q.; Ma, Z.C.; Deng, X.X. Pro-inflammatory effects of a litchi protein extract in murine RAW264.7 macrophages. Hortic. Res. 2016, 3, 16017. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.K.; Iwasaki, A. Innate control of adaptive immunity: Dendritic cells and beyond. Semin. Immunol. 2007, 19, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Qi, Y.; Xu, W.H.; Liu, Y.; Qiu, L.; Wang, K.Q.; Hu, H.G.; He, Z.G.; Zhang, J.P. Matrine derivate MASM suppresses LPS-induced phenotypic and functional maturation of murine bone marrow-derived dendritic cells. Int. Immunopharmacol. 2016, 36, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Fallarino, F.; Pallotta, M.T.; Matino, D.; Gargaro, M.; Orabona, C.; Vacca, C.; Mondanelli, G.; Allegrucci, M.; Boon, L.; Romani, R. LPS-conditioned dendritic cells confer endotoxin tolerance contingent on tryptophan catabolism. Immunobiology 2014, 220, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Gasparini, C.; Foxwell, B.M.; Feldmann, M. RelB/p50 regulates TNF production in LPS-stimulated dendritic cells and macrophages. Cytokine 2013, 61, 736–740. [Google Scholar] [CrossRef] [PubMed]
- Fu, L.N.; Chai, L.J.; Ding, D.K.; Pan, Z.Y.; Peng, S.A. A novel citrus rootstock tolerant to iron deficiency in calcareous soil. J. Am. Soc. Hortic. Sci. 2016, 141, 112–118. [Google Scholar]
- Yan, H.Q.; Ma, Z.C.; Peng, S.A.; Deng, X.X. Anti-inflammatory effect of auraptene extracted from trifoliate orange (Poncirus trifoliate) on LPS-stimulated RAW 264.7 cells. Inflammation 2013, 36, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.D.; Zhang, M.; Zhang, M.X.; Ren, Y.J.; Jin, J.; Zhao, Q.Y.; Pan, Z.S.; Wu, M.; Shu, H.B.; Dong, C.; et al. Induction of USP25 by viral infection promotes innate antiviral responses by mediating the stabilization of TRAF3 and TRAF6. Proc. Natl. Acad. Sci. USA 2015, 112, 11324–11329. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Compounds Classification | Numbers |
|---|---|
| Amino acids | 28 |
| Amino acid derivatives | 22 |
| Alkaloids | 10 |
| Anthocyanin | 2 |
| Carbohydrates | 6 |
| Catechin derivatives | 3 |
| Cholines | 7 |
| Coumarins and their derivatives | 2 |
| Flavonoids | 65 |
| Indoles and their derivatives | 4 |
| Lipids-fatty acid | 8 |
| Lipids-glycerides | 2 |
| Lipids-glycerophospholipid | 17 |
| Nucleotides and their derivatives | 29 |
| Organic acids and their derivatives | 11 |
| Terpenoids | 4 |
| Tryptamines and their derivatives | 12 |
| Phenolamides | 13 |
| Phenylpropanoids | 14 |
| Proanthocyanidins | 2 |
| Phytohormones | 6 |
| Vitamin and Vitamin-related | 16 |
| Compounds | ZQE |
|---|---|
| Naringin (mg/g) | 79.59 ± 0.21 |
| Eriocitrin (μg/g) | 2197.54 ± 18.29 |
| Hespiridin (μg/g) | 3348.21 ± 204.00 |
| Neohesperidin (μg/g) | 1909.71 ± 2.81 |
| Rhoifolin (μg/g) | 2818.0± 18.00 |
| Naringenin (μg/g) | 1117.82 ± 21.78 |
| Poncirin (μg/g) | 2114.42 ± 91.72 |
| Gene Name | Forward/Reverse Primer Sequence (5′–3′) |
|---|---|
| β-actin | TGAAGGGCATCTTGGGCTACAC TGGGTGGTCCAGGGTTTCTTAC |
| COX-2 | ATCTGGCTTCGGGAGCACAAC GAGGCAATGCGGTTCTGATACTG |
| IL-1β | GTTGACGGACCCCAAAAGAT CCTCATCCTGGAAGGTCCAC |
| IL-6 | ACAAAGCCAGAGTCCTTCAGA TCCTTAGCCACTCCTTCTGT |
| TNF-α | ACTGAACTTCGGGGTGATCG TCTTTGAGATCCATGCCGTTG |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, L.; Ren, Y.; Lin, D.; Peng, S.; Zhong, B.; Ma, Z. The Anti-Inflammatory Properties of Citrus wilsonii Tanaka Extract in LPS-Induced RAW 264.7 and Primary Mouse Bone Marrow-Derived Dendritic Cells. Molecules 2017, 22, 1213. https://doi.org/10.3390/molecules22071213
Cheng L, Ren Y, Lin D, Peng S, Zhong B, Ma Z. The Anti-Inflammatory Properties of Citrus wilsonii Tanaka Extract in LPS-Induced RAW 264.7 and Primary Mouse Bone Marrow-Derived Dendritic Cells. Molecules. 2017; 22(7):1213. https://doi.org/10.3390/molecules22071213
Chicago/Turabian StyleCheng, Liping, Yujie Ren, Dingbo Lin, Shu’ang Peng, Bo Zhong, and Zhaocheng Ma. 2017. "The Anti-Inflammatory Properties of Citrus wilsonii Tanaka Extract in LPS-Induced RAW 264.7 and Primary Mouse Bone Marrow-Derived Dendritic Cells" Molecules 22, no. 7: 1213. https://doi.org/10.3390/molecules22071213
APA StyleCheng, L., Ren, Y., Lin, D., Peng, S., Zhong, B., & Ma, Z. (2017). The Anti-Inflammatory Properties of Citrus wilsonii Tanaka Extract in LPS-Induced RAW 264.7 and Primary Mouse Bone Marrow-Derived Dendritic Cells. Molecules, 22(7), 1213. https://doi.org/10.3390/molecules22071213





