Measuring the Transition Rates of Coalescence Events during Double Phase Separation in Microgravity
Abstract
:1. Introduction
2. Experimental Setup
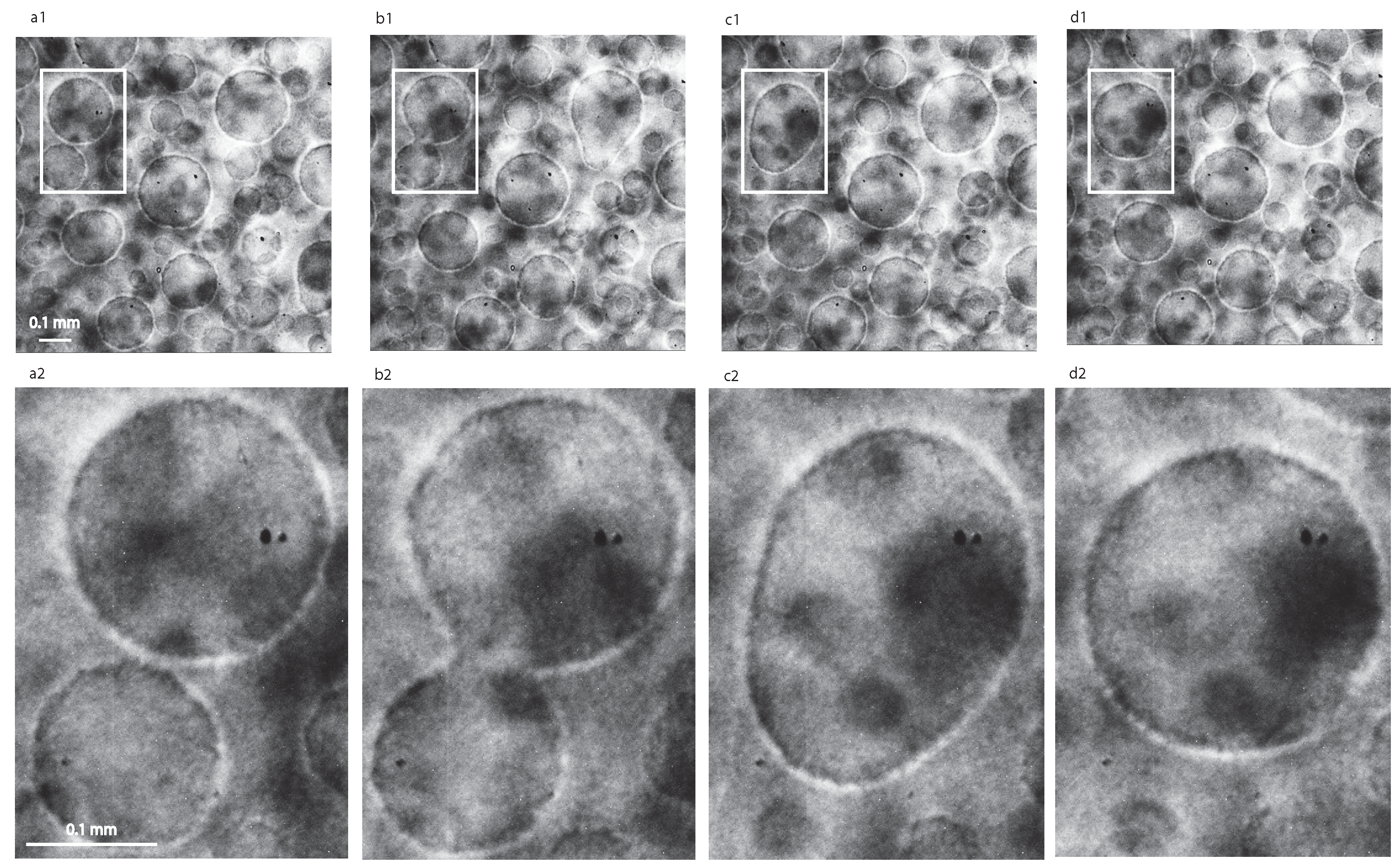
3. Narrow Field of View (NFOV) Image Processing
4. Results
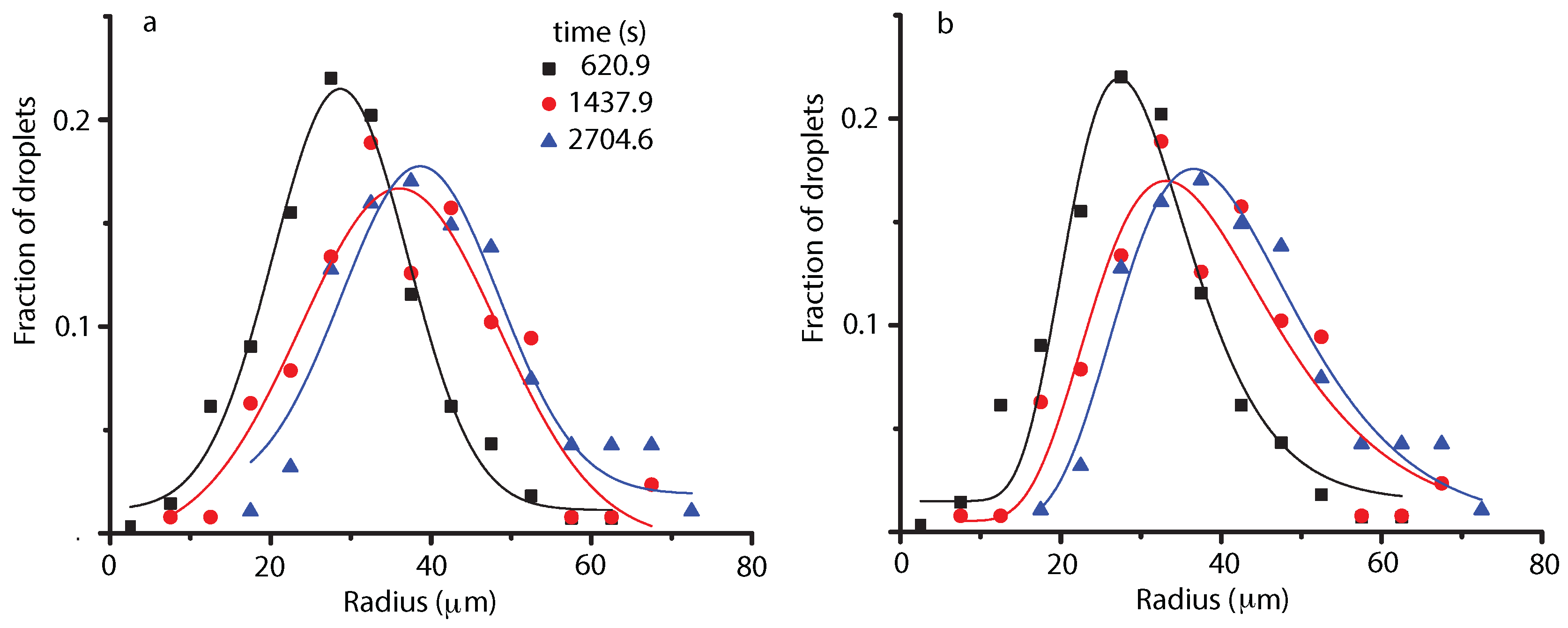
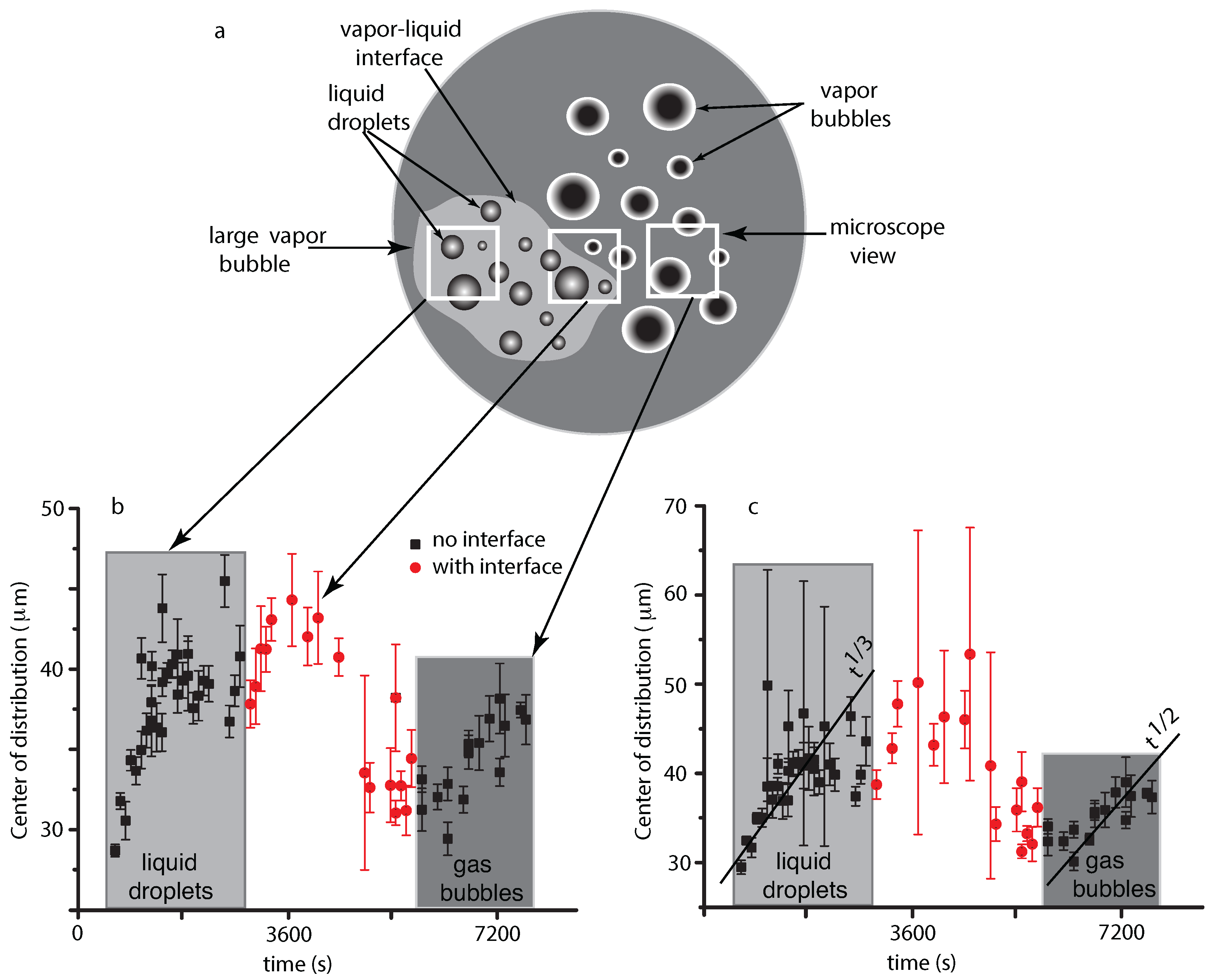
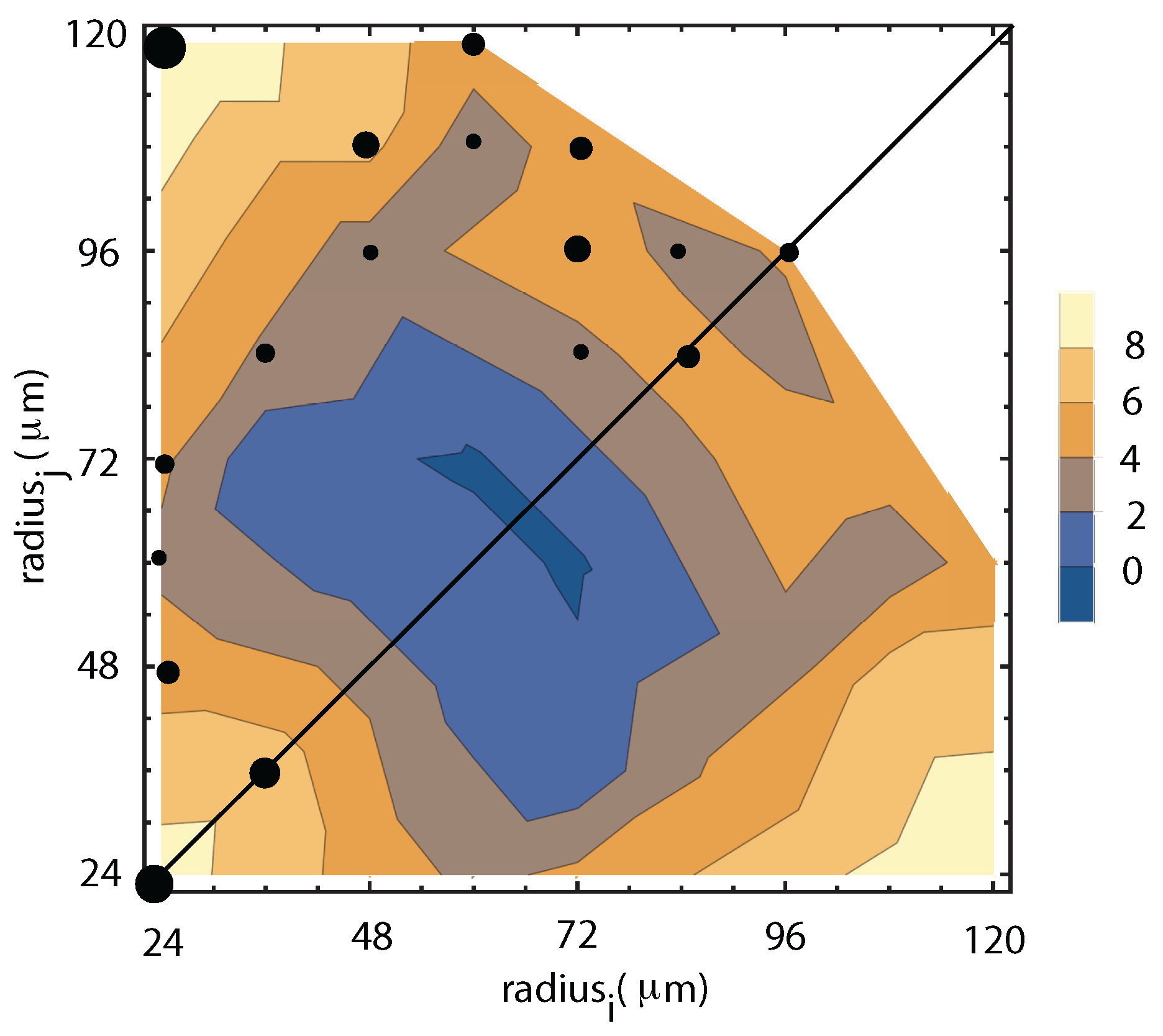
4.1. Liquid Droplets Dynamics from NFOV Images
4.2. Vapor Bubbles Dynamics from NFOV Images
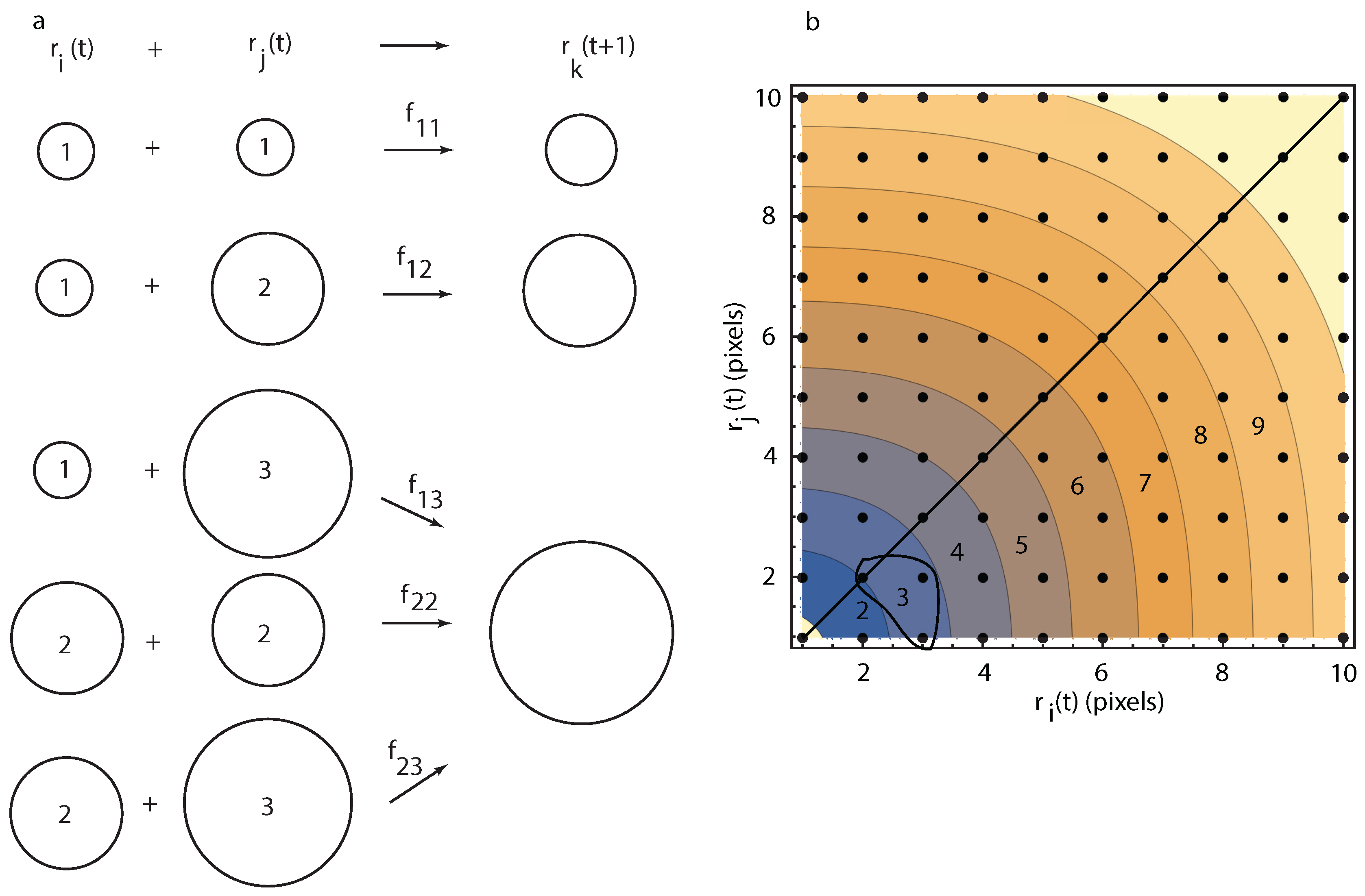
5. Theoretical Modeling
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Akgerman, A. Supercritical Fluids in Environmental Remediation and Pollution Prevention. In Supercritical Fluids; Abraham, M.A., Sunol, A.K., Eds.; American Chemical Society: Washington, DC, USA, 1997; Chapter 15; pp. 208–231. [Google Scholar]
- Bates, F.S.; Wiltzius, P. Spinodal decomposition of a symmetric critical mixture of deuterated and protonated polymer. J. Chem. Phys. 1989, 91, 3258–3274. [Google Scholar] [CrossRef]
- Beysens, D. Critical Phenomena. In Materials Sciences in Space, a Contribution to the Scientific Basis of Space Processing; Feuerbacher, B., Hamacher, H., Naumann, R., Eds.; Springer: Berlin, Germany, 1986; p. 191. [Google Scholar]
- Binder, K. Spinodal Decomposition. In Materials Science and Technology. A Comprehensive Treatment; Cahn, R.W., Haasen, P., Kramer, E., Eds.; Phase Transformations in Materials; Wiley-VCH: Weinheim, Germany, 1991; Chapter 7; Volume 5, pp. 405–471. [Google Scholar]
- Frisch, H.; Lebowitz, J.L. The Equilibrium Theory of Classical Fluids; Benjamin: New York, NY, USA, 1964. [Google Scholar]
- Zappoli, B.; Daniel, B.; Garrabos, Y. Heat Transfers and Related Effects in Supercritical Fluids; Springer: Dordrecht, The Netherlands, 2015; Volume 108. [Google Scholar]
- Abyzov, A.S.; Schmelzer, J.W.P. Nucleation versus spinodal decomposition in confined binary solutions. J. Chem. Phys. 2007, 127, 114504. [Google Scholar] [CrossRef] [PubMed]
- Cumming, A.; Wiltzius, P.; Bates, F.S.; Rosedale, J.H. Light-scattering experiments on phase-separation dynamics in binary fluid mixtures. Phys. Rev. A 1992, 45, 885. [Google Scholar] [CrossRef] [PubMed]
- Perrot, F.; Guenoun, P.; Baumberger, T.; Beysens, D.; Garrabos, Y.; Le Neindre, B. Nucleation and growth of tightly packed droplets in fluids. Phys. Rev. Lett. 1994, 73, 688–691. [Google Scholar] [CrossRef] [PubMed]
- Schmelzer, J.W.P.; Abyzov, A.S.; Moller, J. Nucleation versus spinodal decomposition in phase formation processes in multicomponent solutions. J. Chem. Phys. 2004, 121, 6900–6917. [Google Scholar] [CrossRef] [PubMed]
- Beysens, D.; Straub, J.; Turner, D. Phase Transitions and Near-Critical Phenomena. In Space, a European Perspective; Walter, H., Ed.; Springer: Berlin, Germany, 1987; p. 221. [Google Scholar]
- Domb, C.; Lebowitz, J.L.; Lebowitz, J. Phase Transitions and Critical Phenomena; Academic Press: London, UK, 2001; p. 320. [Google Scholar]
- Onuki, A. Phase Transition Dynamics; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Schmelzer, J.W.P. Comments on the Nucleation Theorem. J. Colloid Interface Sci. 2001, 242, 354–372. [Google Scholar] [CrossRef]
- Schmelzer, J.W.P.; Schmelzer, J.J.; Gutzow, I.S. Reconciling Gibbs and van der Waals: A new approach to nucleation theory. J. Chem. Phys. 2000, 112, 3820. [Google Scholar] [CrossRef]
- Schmelzer, J.W.P.; Schmelzer, J.J. Kinetics of condensation of gases: A new approach. J. Chem. Phys. 2001, 114, 5180. [Google Scholar] [CrossRef]
- Cahn, W.J.; Hilliard, E.J. Free Energy of a Nonuniform System. I. Interfacial Free Energy. J. Chem. Phys. 1958, 29, 258–267. [Google Scholar] [CrossRef]
- Cahn, J.; Hilliard, J. Free energy of a non-uniform system III: Nucleation in a two-component incompressible fluid. J. Chem. Phys. 1959, 31, 688–699. [Google Scholar] [CrossRef]
- Cahn, J. On spinodal decomposition. Acta Metall. 1961, 9, 795–801. [Google Scholar] [CrossRef]
- Granasy, L. Nucleation and spinodal decomposition. Solid State Phenom. 1997, 56, 67. [Google Scholar] [CrossRef]
- Oprisan, A.; Oprisan, S.A.; Hegseth, J.J.; Garrabos, Y.; Lecoutre-Chabot, C.; Beysens, D. Universality in early-stage growth of phase-separating domains near the critical point. Phys. Rev. E 2008, 77, 051118. [Google Scholar] [CrossRef] [PubMed]
- Bonn, D.; Bertrand, E.; Meunier, J.; Blossey, R. Dynamics of Wetting Layer Formation. Phys. Rev. Lett. 2000, 84, 4661–4664. [Google Scholar] [CrossRef] [PubMed]
- Fenistein, D.; Bonn, D.; Rafai, S.; Wegdam, G.; Meunier, J.; Parry, A.; TelodaGama, M. What controls the thickness of wetting layers near bulk criticality? Phys. Rev. Lett. 2002, 89, 096101. [Google Scholar] [CrossRef] [PubMed]
- Guenoun, P.; Beysens, D.; Robert, M. Dynamics of wetting and phase separation. Phys. Rev. Lett. 1990, 65, 2406–2409. [Google Scholar] [CrossRef] [PubMed]
- Hegseth, J.; Garrabos, Y.; Nikolayev, V.S.; Lecoutre-Chabot, C.; Wunenburger, R.; Beysens, D. Gas wets a solid wall in orbit. Int. J. Thermophys. 2002, 23, 89–101. [Google Scholar] [CrossRef]
- Lipowsky, R.; Huse, D.A. Diffusion-Limited Growth of Wetting Layers. Phys. Rev. Lett. 1986, 57, 353. [Google Scholar] [CrossRef] [PubMed]
- Steiner, U.; Klein, J. Growth of Wetting Layers from Liquid Mixtures. Phys. Rev. Lett. 1996, 77, 2526–2529. [Google Scholar] [CrossRef] [PubMed]
- Beysens, D.; Garrabos, Y. The phase transition of gas and liquids. Physica A 2000, 281, 361–380. [Google Scholar] [CrossRef]
- Beysens, D.; Forgacs, G.; Glazier, J.A. Cell sorting is analogous to phase ordering in fluids. Proc. Natl. Acad. Sci. USA 2000, 97, 9467–9471. [Google Scholar] [CrossRef] [PubMed]
- Pont, G.; Barde, S.; Blonde, D.; Zappoli, B.; Garrabos, Y.; Lecoutre, C.; Beysens, D.; Hicks, M.; Hegde, U.; Hahn, I.; et al. DECLIC, soon two years of successful operations. In Proceedings of the 62nd International Astronautical Congress, Cape Town, South Africa, 3–7 October 2011; International Astronautical Federation: Paris, France, 2011; pp. 1–12. [Google Scholar]
- Beysens, D.; Guenoun, P.; Sibille, P.; Kumar, A. Dimple and nose coalescences in phase-separation processes. Phys. Rev. E 1994, 50, 1299–1302. [Google Scholar] [CrossRef]
- Beysens, D. Kinetics and morphology of phase separation in fluids: The role of droplet coalescence. Physica A 1997, 239, 329–339. [Google Scholar] [CrossRef]
- Binder, K. Theory for the dynamics of “clusters.” II. Critical diffusion in binary systems and the kinetics of phase separation. Phys. Rev. B 1977, 15, 4425–4447. [Google Scholar] [CrossRef]
- Hegseth, J.; Nikolayev, V.; Beysens, D.; Garrabos, Y.; Chabot, C. Growth and Morphology of Phase Separating Supercritical Fluids (GMSF), Boiling in Subcritical Fluids, and Critical Fluctuations. In Proceedings of the Fourth Microgravity Fluid Physics and Transport Phenomena Conference, Cleveland, OH, USA, 12–14 August 1998; National Center for Microgravity Research on Fluids and Combustion: Cleveland, OH, USA, 1998; Volume NASA/CP-1999-208526/SUPPL1, pp. 184–189. [Google Scholar]
- Klein, H.; Schmitz, G.; Woermann, D. Temperature propagation in near-critical fluids prior to and during phase separation. Phys. Rev. A 1991, 43, 4562–4563. [Google Scholar] [CrossRef] [PubMed]
- Oprisan, A. Fluctuations, Phase Separation and Wetting Films near Liquid-Gas Critical Point. Ph.D. Thesis, University of New Orleans, New Orleans, LA, USA, 2006. [Google Scholar]
- Perrot, F.; Beysens, D.; Garrabos, Y.; Froumlhlich, T.; Guenoun, P.; Bonetti, M.; Bravais, P. Morphology transition observed in a phase separating fluid. Phys. Rev. E 1999, 59, 3079. [Google Scholar] [CrossRef]
- Roy, S.; Das, S.K. Effects of domain morphology on kinetics of fluid phase separation. J. Chem. Phys. 2013, 139, 044911. [Google Scholar] [CrossRef] [PubMed]
- Sagui, C.; O’Gorman, D.S.; Grant, M. Nucleation, growth and coarsening in phase-separating systems. Scanning Microsc. 1998, 12, 3–8. [Google Scholar]
- Tanaka, H. New mechanisms of droplet coarsening in phase-separating fluid mixtures. J. Chem. Phys. 1997, 107, 3734. [Google Scholar] [CrossRef]
- Oprisan, A.; Garrabos, Y.; Lecoutre, C.; Beysens, D. Pattern Evolution during Double Liquid-Vapor Phase Transitions under Weightlessness. Molecules 2017, 22, 947. [Google Scholar] [CrossRef] [PubMed]
- Marcout, R.; Raymond, G.; Martin, B.C.G.; Zappoli, B.; Duclos, F.; Barde, S.; Beysens, D.; Garrabos, Y.; Lecoutre, C.; Billia, B.; et al. DECLIC: A facility to investigate fluids and transparent materials in microgravity conditions in ISS. In Proceedings of the 57th International Astronautical Congress, Valencia, Spain, 2–6 October 2006; American Institute of Aeronautics and Astronautics: Reston, VA, USA, 2006. [Google Scholar]
- Beysens, D.; Pichavant, G.; Chatain, D.; Nikolayev, V.; Lecoutre, C.; Garrabos, Y. Non Marangoni motion of bubble under temperature gradient. In Proceedings of the 62nd International Astronautical Congress, Cape Town, South Africa, 3–7 October 2011; International Astronautical Federation: Paris, France, 2011. [Google Scholar]
- Beysens, D.; Chatain, D.; Nikolayev, V.S.; Ouazzani, J.; Garrabos, Y. Possibility of long-distance heat transport in weightlessness using supercritical fluids. Phys. Rev. E 2010, 82, 061126. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, R.; Tanaka, H. A novel coarsening mechanism of droplets in immiscible fluid mixtures. Nat. Commun. 2015, 6, 7407. [Google Scholar] [CrossRef] [PubMed]
- Oprisan, A.; Hegseth, J.; Smith, G.; Lecoutre, C.; Garrabos, Y.; Beysens, D.A. Dynamics of wetting layer and Marangoni convection in microgravity. Phys. Rev. E 2011, 84, 021202. [Google Scholar] [CrossRef] [PubMed]
- Martula, D.; Hasegawa, T.; Lloyd, D.; Bonnecaze, R. Coalescence-induced coalescence of inviscid droplets in a viscous fluid. J. Colloid Interface Sci. 2000, 232, 241–253. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oprisan, A.; Garrabos, Y.; Lecoutre, C.; Beysens, D. Measuring the Transition Rates of Coalescence Events during Double Phase Separation in Microgravity. Molecules 2017, 22, 1125. https://doi.org/10.3390/molecules22071125
Oprisan A, Garrabos Y, Lecoutre C, Beysens D. Measuring the Transition Rates of Coalescence Events during Double Phase Separation in Microgravity. Molecules. 2017; 22(7):1125. https://doi.org/10.3390/molecules22071125
Chicago/Turabian StyleOprisan, Ana, Yves Garrabos, Carole Lecoutre, and Daniel Beysens. 2017. "Measuring the Transition Rates of Coalescence Events during Double Phase Separation in Microgravity" Molecules 22, no. 7: 1125. https://doi.org/10.3390/molecules22071125
APA StyleOprisan, A., Garrabos, Y., Lecoutre, C., & Beysens, D. (2017). Measuring the Transition Rates of Coalescence Events during Double Phase Separation in Microgravity. Molecules, 22(7), 1125. https://doi.org/10.3390/molecules22071125





