Crude Ecklonia cava Flake Extracts Attenuate Inflammation through the Regulation of TLR4 Signaling Pathway in LPS-Induced RAW264.7 Cells
Abstract
:1. Introduction
2. Results
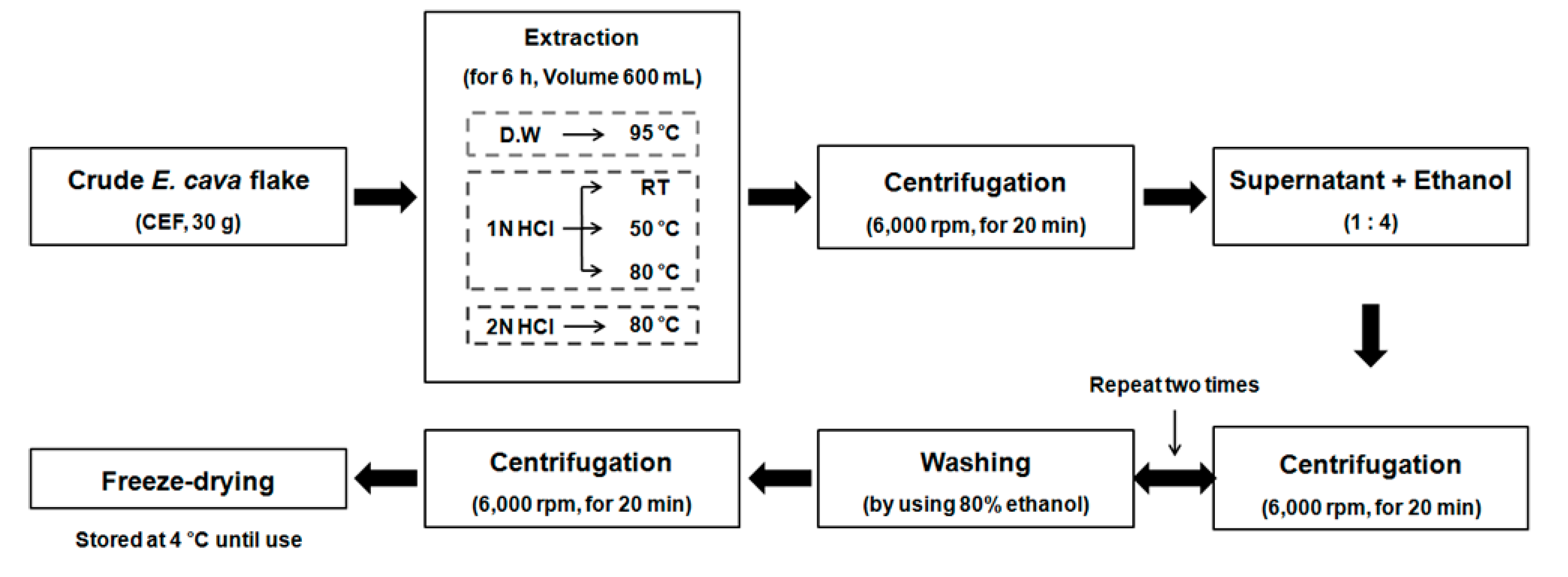
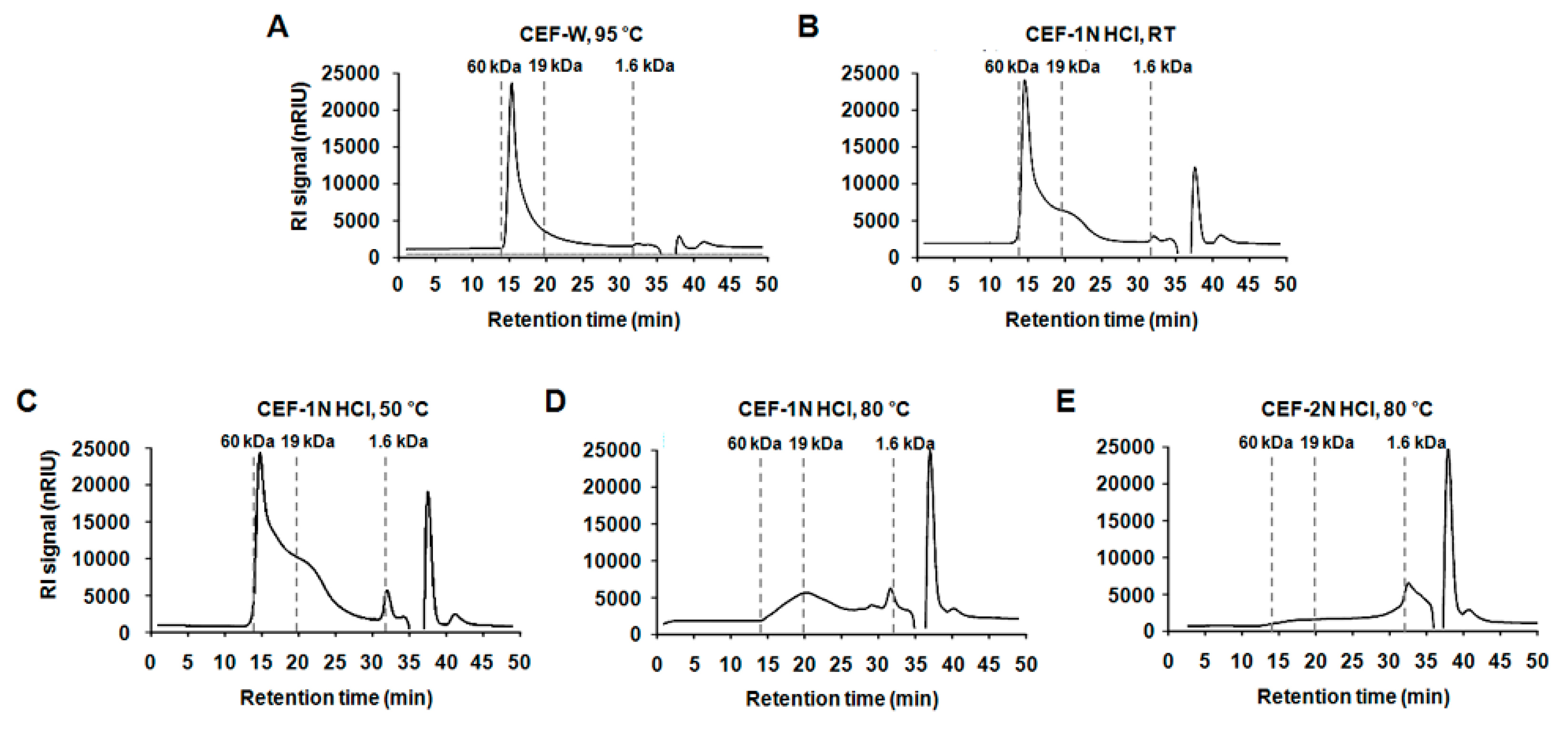
2.1. Preparation of the CEF Extracts with Different Conditions
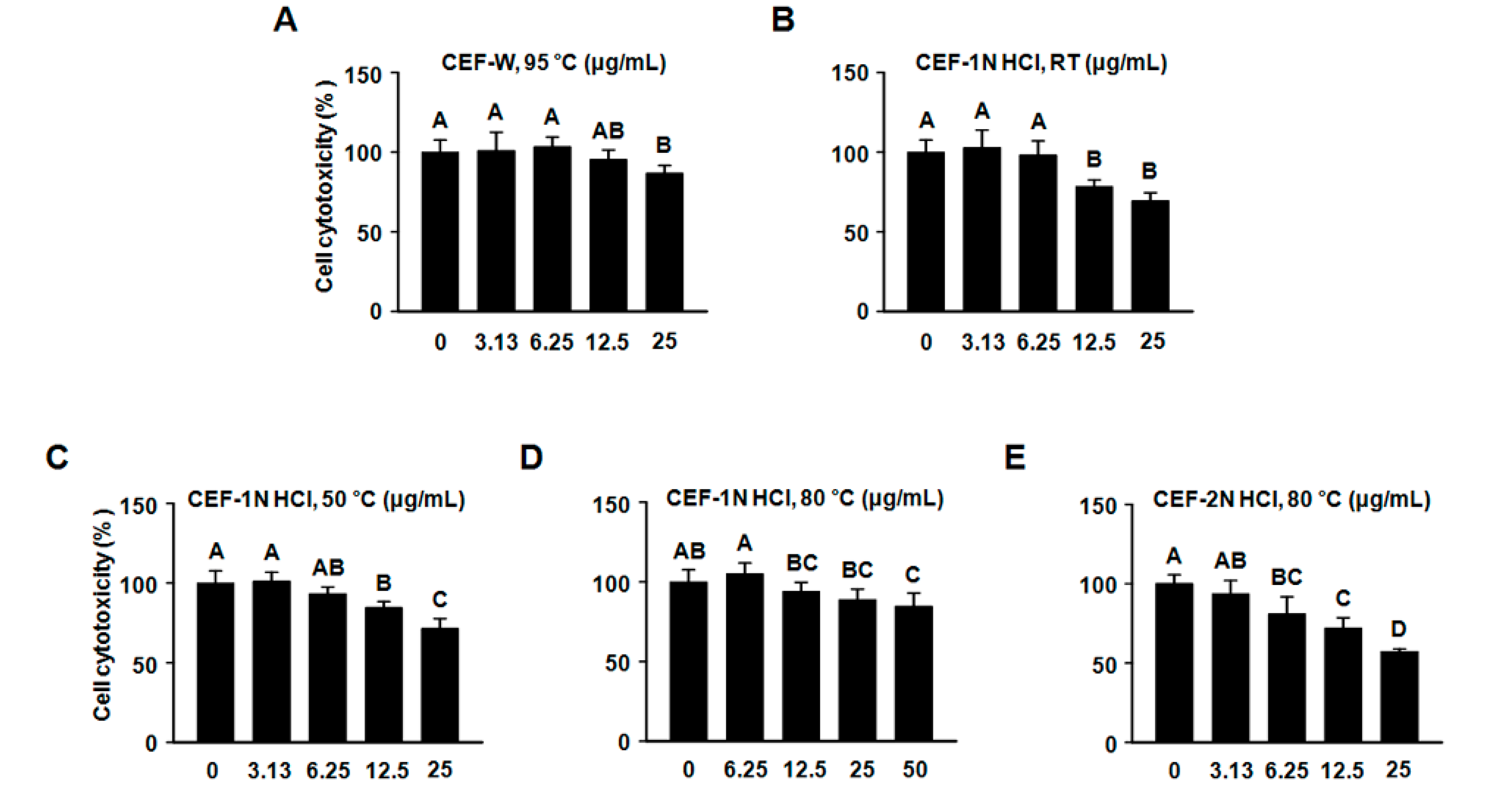
2.2. Evaluation of Cytotoxic Assay of CEF Extracts on RAW264.7 Cells
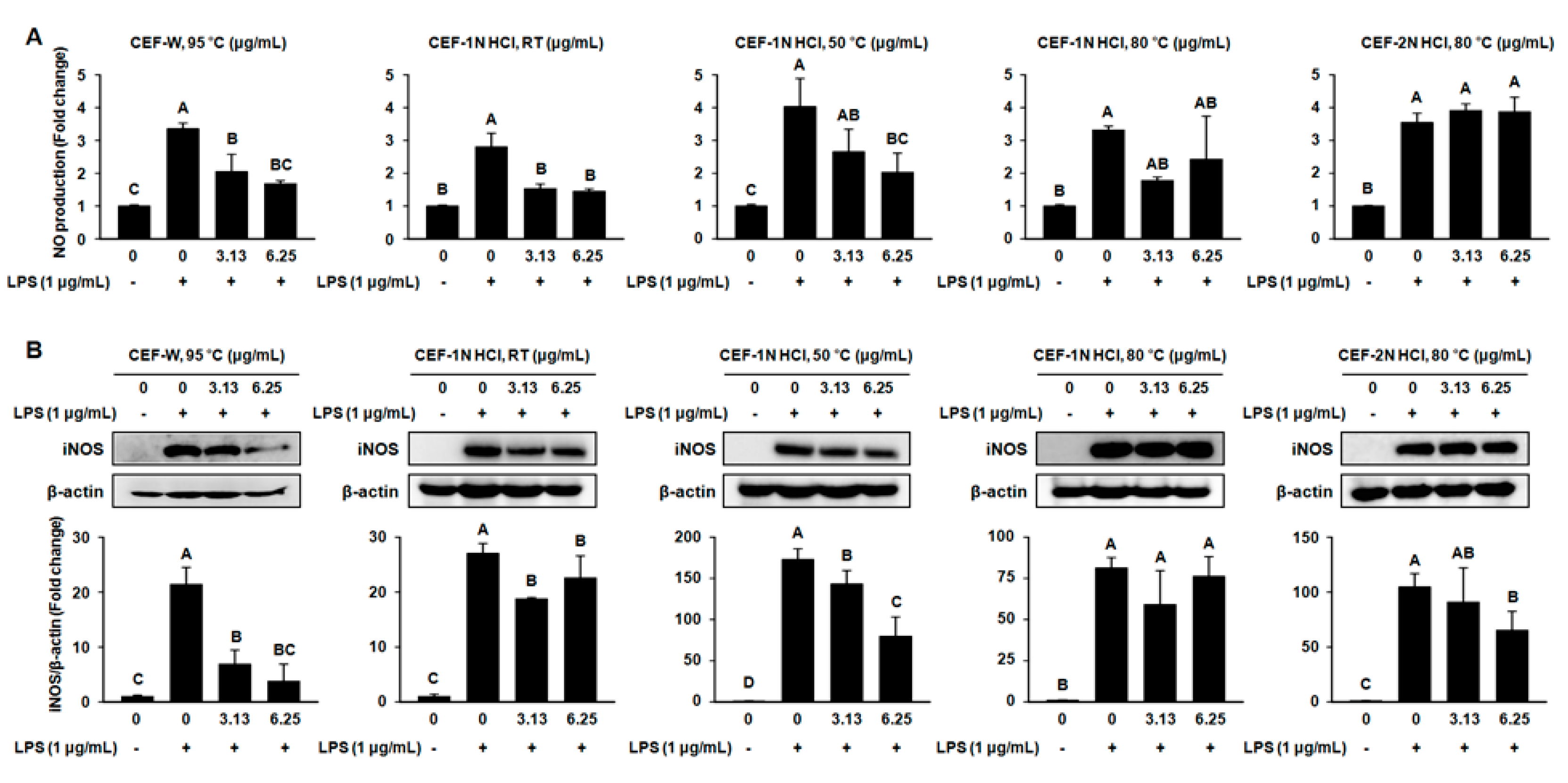
2.3. Effect of CEF Extracts on the Production of Nitric Oxide (NO) and the Protein Expression of Inducible Nitric Oxide Synthase (iNOS) in LPS-Induced RAW264.7 Cells
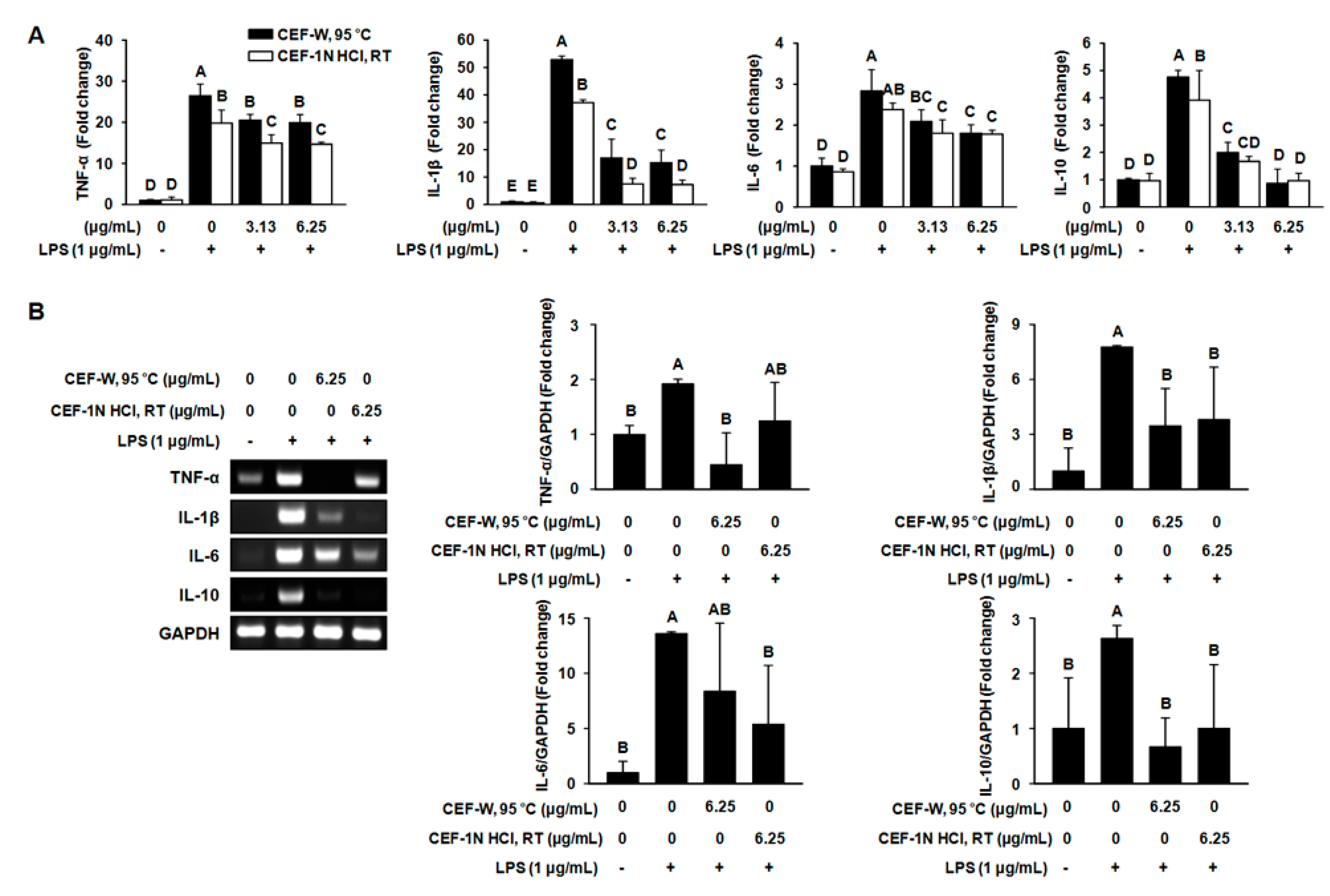
2.4. The Production of Pro-Inflammatory Cytokines such as TNF-α, IL-1β, IL-6, and IL-10 Was Suppressed by CEF-W, 95 °C or CEF-1N HCl, RT in LPS-Induced RAW264.7 Cells
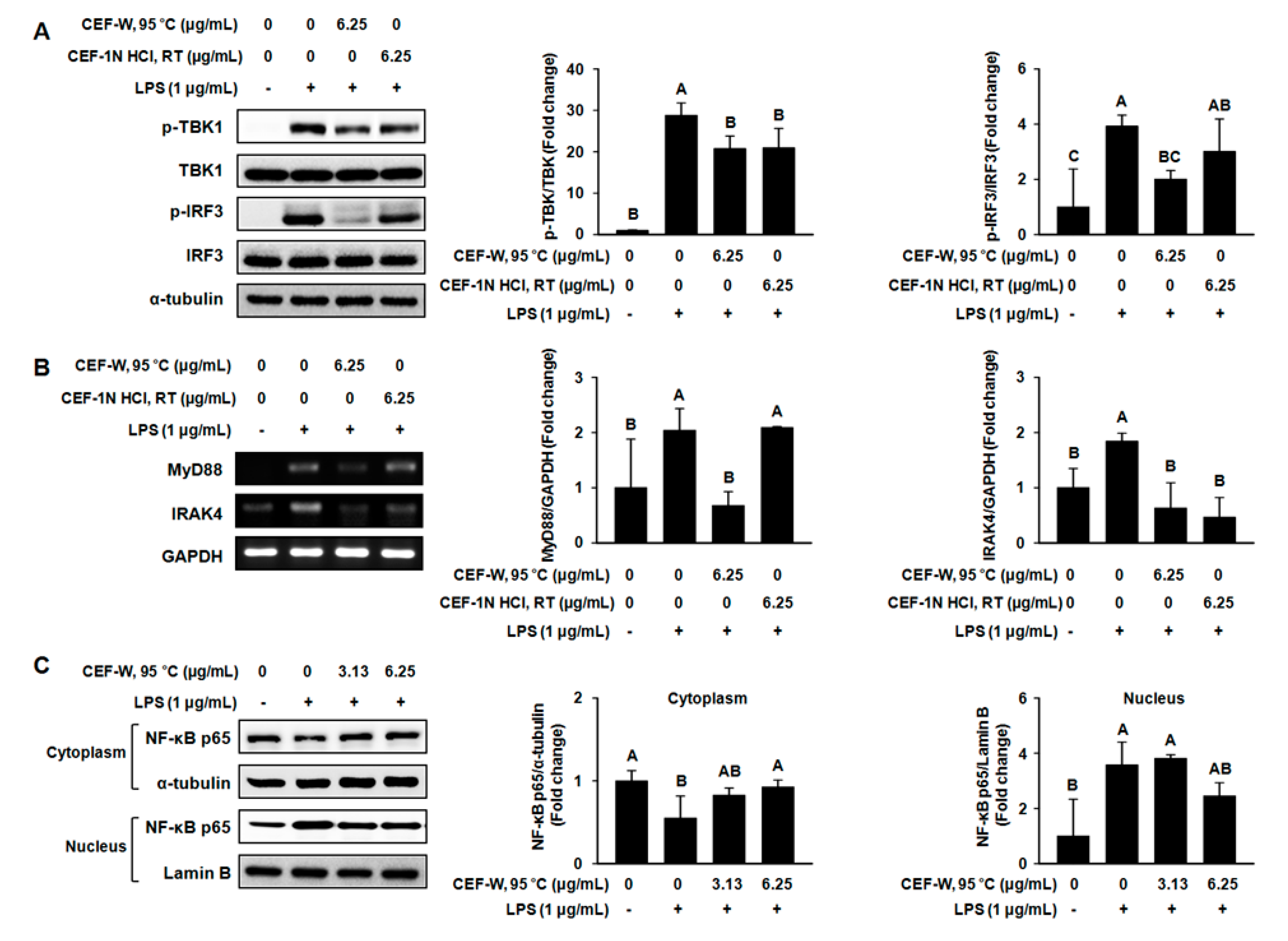
2.5. Effect of CEF-W, 95 °C and CEF-1 N HCl, RT on Downstream Target of the TLR Signaling Pathway
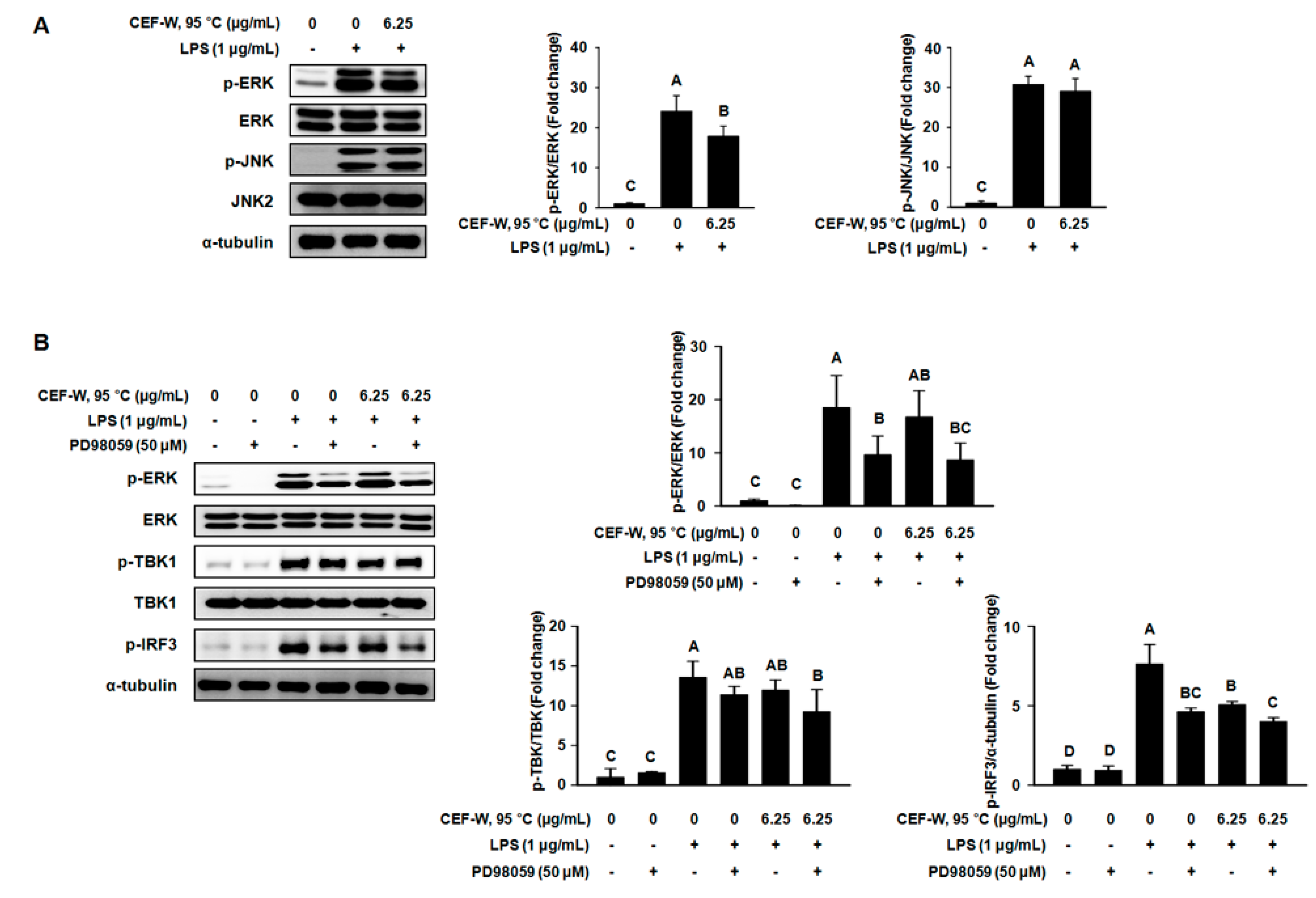
2.6. CEF-W, 95 °C Attenuates a Part of MAPK Signaling Pathway in LPS-Induced RAW264.7 Cells
3. Discussion
4. Materials and Methods
4.1. Materials and Reagents
4.2. Preparation Process of the CEF Extracts
4.3. Analysis of the Molecular Weight of CEF Extracts
4.4. Cell Culture and Viability
4.5. Measurement of the Contents of Nitric Oxide
4.6. Evaluation of the Inflammatory Cytokines Production in RAW264.7 Cells
4.7. RNA Isolation and Reverse Transcription Polymerase Chain Reaction (RT-PCR)
4.8. Western Blot Analysis
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cho, H.J.; Seon, M.R.; Lee, Y.M.; Kim, J.; Kim, J.K.; Kim, S.G.; Park, J.H. 3,3′-Diindolylmethane suppresses the inflammatory response to lipopolysaccharide in murine macrophages. J. Nutr. 2008, 138, 17–23. [Google Scholar] [PubMed]
- Hofseth, L.J.; Ying, L. Identifying and defusing weapons of mass inflammation in carcinogenesis. Biochim. Biophys. Acta 2006, 1765, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, O.; Akira, S. Pattern recognition receptors and inflammation. Cell 2010, 140, 805–820. [Google Scholar] [CrossRef] [PubMed]
- Fukata, M.; Vamadevan, A.S.; Abreu, M.T. Toll-Like receptors (TLRs) and Nod-like receptors (NLRs) in inflammatory disorders. Semin. Immunol. 2009, 21, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Akira, S. TLR signaling. Cell Death Differ. 2006, 13, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, O.; Hoshino, K.; Kawai, T.; Sanjo, H.; Takada, H.; Ogawa, T.; Takeda, K.; Akira, S. Differential roles of TLR2 and TLR4 in recognition of gram-negative and gram-positive bacterial cell wall components. Immunity 1999, 11, 443–451. [Google Scholar] [CrossRef]
- Takeda, K.; Akira, S. TLR signaling pathways. Semin. Immunol. 2004, 16, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Wieland, C.W.; Florquin, S.; Maris, N.A.; Hoebe, K.; Beutler, B.; Takeda, K.; Akira, S.; van der Poll, T. The MyD88-dependent, but not the MyD88-independent, pathway of TLR4 signaling is important in clearing nontypeable haemophilus influenzae from the mouse lung. J. Immunol. 2005, 175, 6042–6049. [Google Scholar] [CrossRef] [PubMed]
- Johnson, G.L.; Lapadat, R. Mitogen-Activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Han, J.; Hui, L. MAPK signaling in inflammation-associated cancer development. Protein Cell 2010, 1, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Wagner, E.F.; Nebreda, A.R. Signal integration by JNK and p38 MAPK pathways in cancer development. Nat. Rev. Cancer 2009, 9, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Manning, A.M.; Davis, R.J. Targeting JNK for therapeutic benefit: From junk to gold? Nat. Rev. Drug Discov. 2003, 2, 554–565. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.H.; Kim, K.J.; Ryu, S.J.; Lee, B.Y. Caffeine prevents LPS-induced inflammatory responses in RAW264.7 cells and zebrafish. Chem. Biol. Interact. 2016, 248, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Kim, K.J.; Kim, B.H.; Koh, E.J.; Seo, M.J.; Lee, B.Y. 6-Gingerol suppresses adipocyte-derived mediators of inflammation in vitro and in high-fat diet-induced obese zebra fish. Planta Med. 2017, 83, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.J.; Seo, Y.J.; Pan, C.H.; Lee, O.H.; Kim, K.J.; Lee, B.Y. Fucoxanthin suppresses lipid accumulation and ROS production during differentiation in 3T3-L1 adipocytes. Phytother. Res. 2016, 30, 1802–1808. [Google Scholar] [CrossRef] [PubMed]
- Jung, W.K.; Ahn, Y.W.; Lee, S.H.; Choi, Y.H.; Kim, S.K.; Yea, S.S.; Choi, I.; Park, S.G.; Seo, S.K.; Lee, S.W.; et al. Ecklonia cava ethanolic extracts inhibit lipopolysaccharide-induced cyclooxygenase-2 and inducible nitric oxide synthase expression in BV2 microglia via the MAP kinase and NF-κB pathways. Food Chem. Toxicol. 2009, 47, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.S.; Chung, H.Y.; Kim, J.Y.; Son, B.W.; Jung, H.A.; Choi, J.S. Inhibitory phlorotannins from the edible brown alga Ecklonia stolonifera on total reactive oxygen species (ROS) generation. Arch. Pharm. Res. 2004, 27, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Kong, C.S. Anti-Adipogenic effect of dioxinodehydroeckol via AMPK activation in 3T3-L1 adipocytes. Chem. Biol. Interact. 2010, 186, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Alassali, A.; Cybulska, I.; Brudecki, G.P.; Farzanah, R.; Thomsen, M.H. Methods for upstream extraction and chemical characterization of secondary metabolites from algae biomass. Adv. Tech. Biol. Med. 2016, 4, 1–16. [Google Scholar] [CrossRef]
- Katsuyama, K.; Shichiri, M.; Marumo, F.; Hirata, Y. NO inhibits cytokine-induced iNOS expression and NF-κB activation by interfering with phosphorylation and degradation of IκB-α. Arteriosclerosis Thrombosis Vasc. Boil. 1998, 18, 1796–1802. [Google Scholar] [CrossRef]
- O’Neill, L.A.; Kaltschmidt, C. NF-κB: A crucial transcription factor for glial and neuronal cell function. Trends Neurosci. 1997, 20, 252–258. [Google Scholar] [CrossRef]
- Vanden Berghe, W.; Plaisance, S.; Boone, E.; De Bosscher, K.; Schmitz, M.L.; Fiers, W.; Haegeman, G. P38 and extracellular signal-regulated kinase mitogen-activated protein kinase pathways are required for nuclear factor-κB p65 transactivation mediated by tumor necrosis factor. J. Biol. Chem. 1998, 273, 3285–3290. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.J.; Yoon, K.Y.; Yoon, H.S.; Oh, S.R.; Lee, B.Y. Brazilein suppresses inflammation through inactivation of IRAK4-NF-κB pathway in LPS-induced RAW264. 7 macrophage cells. Int. J. Mol. Sci. 2015, 16, 27589–27598. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Kim, K.J.; Lee, B.Y. Silibinin suppresses mediators of inflammation through the inhibition of TLR4-TAK1 pathway in LPS-induced RAW264. 7 cells. J. Food Nutr. Res. 2016, 4, 515–521. [Google Scholar]
- Feghali, C.A.; Wright, T.M. Cytokines in acute and chronic inflammation. Front. Biosci. 1997, 2, d12–d26. [Google Scholar] [PubMed]
- Sabroe, I.; Parker, L.C.; Dower, S.K.; Whyte, M.K. The role of TLR activation in inflammation. J. Pathol. 2008, 214, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Bae, J.S. Ecklonia cava extracts inhibit lipopolysaccharide induced inflammatory responses in human endothelial cells. Food Chem. Toxicol. 2010, 48, 1682–1687. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, P.L.; Hatch-Pigott, V.; Mower, H.F.; Cooney, R.V. Mutagenicity of nitric oxide and its inhibition by antioxidants. Mutation Res. 1992, 281, 193–202. [Google Scholar] [CrossRef]
- Molloy, R.G.; Mannick, J.A.; Rodrick, M.L. Cytokines, sepsis and immunomodulation. Br. J. Surg. 1993, 80, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Park, M.Y.; Kwon, H.J.; Sung, M.K. Evaluation of aloin and aloe-emodin as anti-inflammatory agents in aloe by using murine macrophages. Biosci. Biotechnol. Biochem. 2009, 73, 828–832. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.C.; Yeh, W.C.; Ohashi, P.S. LPS/TLR4 signal transduction pathway. Cytokine 2008, 42, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Altfeld, M.; Gale, M., Jr. Innate immunity against HIV-1 infection. Nat. Immunol. 2015, 16, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, I.N.; Chang, A.S.; Teng, C.M.; Chen, C.C.; Yang, C.R. Aciculatin inhibits lipopolysaccharide-mediated inducible nitric oxide synthase and cyclooxygenase-2 expression via suppressing NF-κB and JNK/p38 MAPK activation pathways. J. Biomed. Sci. 2011, 18, 28. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Fazal, F. Blocking NF-κB: An inflammatory issue. Proc. Am. Thoracic Soc. 2011, 8, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Barton, G.M.; Medzhitov, R. Toll-Like receptor signaling pathways. Science 2003, 300, 1524–1525. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Compound | Solvent | Temp. | Recovery (g/30 g) | Yield (%) |
|---|---|---|---|---|
| CEF-W, 95 °C | D.W | 95 °C | 4.49 | 14.96 |
| CEF-1 N HCl, RT | 1 N HCl | RT | 1.53 | 5.10 |
| CEF-1 N HCl, 50 °C | 50 °C | 1.02 | 3.40 | |
| CEF-1 N HCl, 80 °C | 80 °C | 0.38 | 1.27 | |
| CEF-2 N HCl, 80 °C | 2 N HCl | 80 °C | 0.11 | 0.37 |
| Compound | Solvent | Temp. | X > 19 kDa (%) | 19 kDa > X > 1.6 kDa (%) | 1.6 kDa > X (%) |
|---|---|---|---|---|---|
| CEF-W, 95 °C | D.W | 95 °C | 70.46 ± 3.28 A | 19.24 ± 1.19 D | 10.30 ± 1.68 D |
| CEF-1 N HCl, RT | 1 N HCl | RT | 60.66 ± 1.67 B | 22.40 ± 1.73 C,D | 16.93 ± 3.40 C |
| CEF-1 N HCl, 50 °C | 50 °C | 48.15 ± 0.90 C | 32.69 ± 2.34 B | 19.16 ± 0.23 C | |
| CEF-1 N HCl, 80 °C | 80 °C | 14.30 ± 1.29 D | 37.98 ± 3.15 A | 47.73 ± 0.55 B | |
| CEF-2 N HCl, 80 °C | 2 N HCl | 80 °C | 8.25 ± 1.21 E | 26.78 ± 5.82 C | 64.97 ± 4.54 A |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, J.-H.; Kim, K.-J.; Lee, B.-Y. Crude Ecklonia cava Flake Extracts Attenuate Inflammation through the Regulation of TLR4 Signaling Pathway in LPS-Induced RAW264.7 Cells. Molecules 2017, 22, 777. https://doi.org/10.3390/molecules22050777
Hwang J-H, Kim K-J, Lee B-Y. Crude Ecklonia cava Flake Extracts Attenuate Inflammation through the Regulation of TLR4 Signaling Pathway in LPS-Induced RAW264.7 Cells. Molecules. 2017; 22(5):777. https://doi.org/10.3390/molecules22050777
Chicago/Turabian StyleHwang, Ji-Hyun, Kui-Jin Kim, and Boo-Yong Lee. 2017. "Crude Ecklonia cava Flake Extracts Attenuate Inflammation through the Regulation of TLR4 Signaling Pathway in LPS-Induced RAW264.7 Cells" Molecules 22, no. 5: 777. https://doi.org/10.3390/molecules22050777
APA StyleHwang, J.-H., Kim, K.-J., & Lee, B.-Y. (2017). Crude Ecklonia cava Flake Extracts Attenuate Inflammation through the Regulation of TLR4 Signaling Pathway in LPS-Induced RAW264.7 Cells. Molecules, 22(5), 777. https://doi.org/10.3390/molecules22050777





