Modulation of HO-1 by Ferulic Acid Attenuates Adipocyte Differentiation in 3T3-L1 Cells
Abstract
:1. Introduction
2. Results
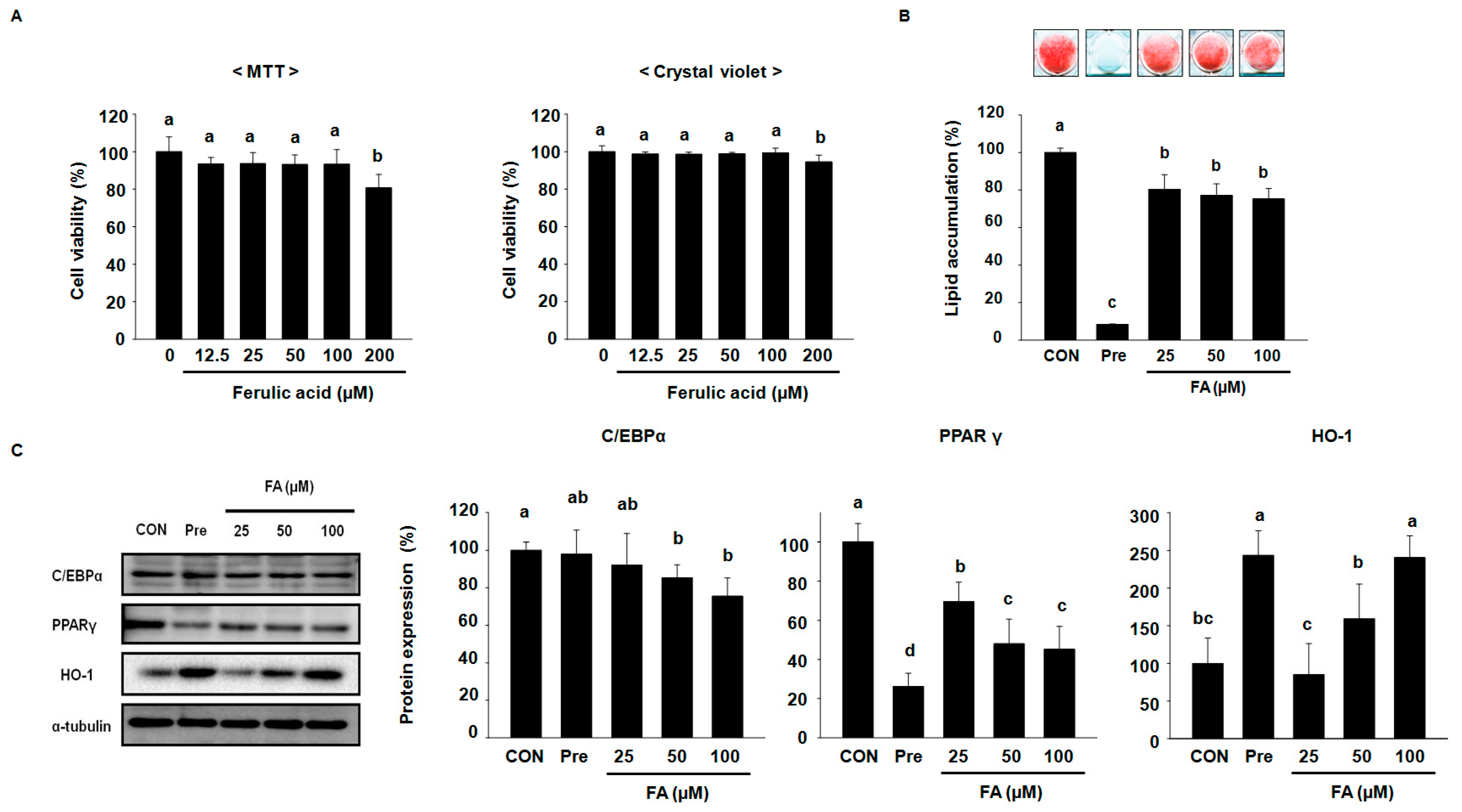
2.1. FA Suppresses Adipocyte Differentiation Factors and Intracellular Lipid Accumulation in 3T3-L1
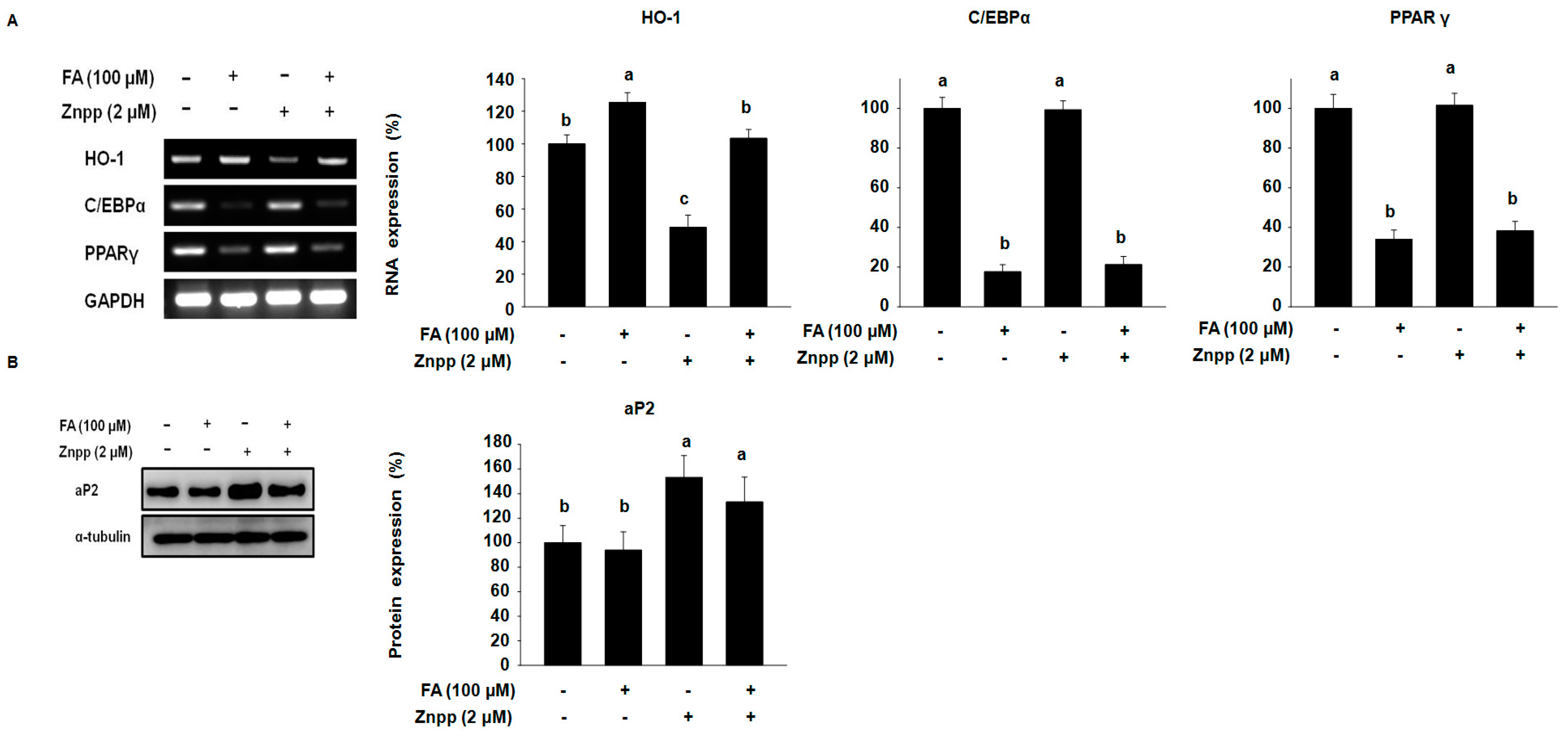
2.2. FA Increases the Expression of HO-1 in 3T3-L1 Treated with a HO-1 Antagonist
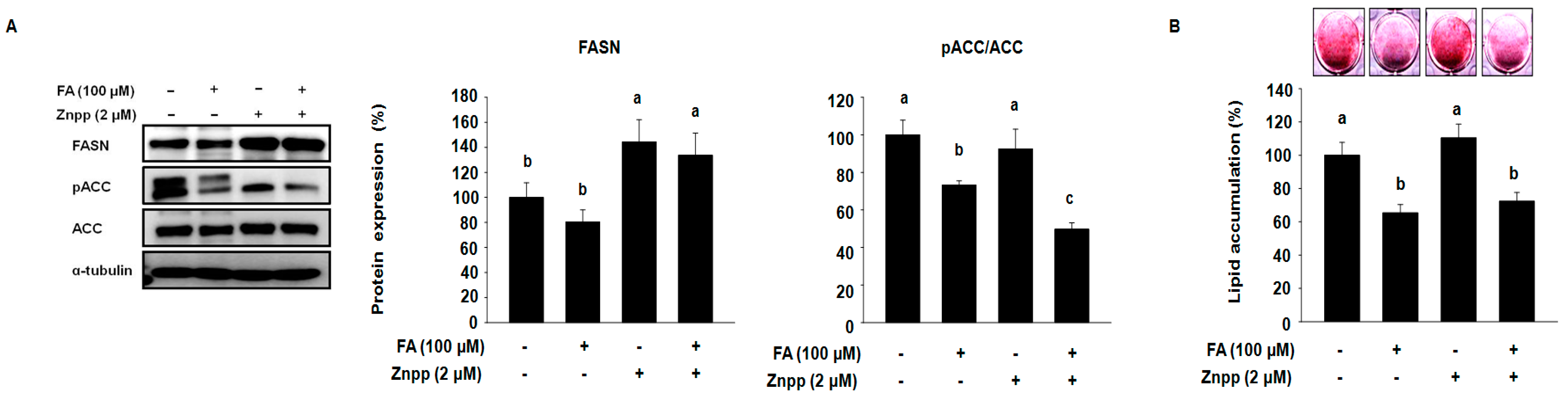
2.3. FA Attenuates the Expression of Fatty Acid Synthases and Intracellular Lipid Accumulation in 3T3-L1 Treated with the Presence of ZnPP
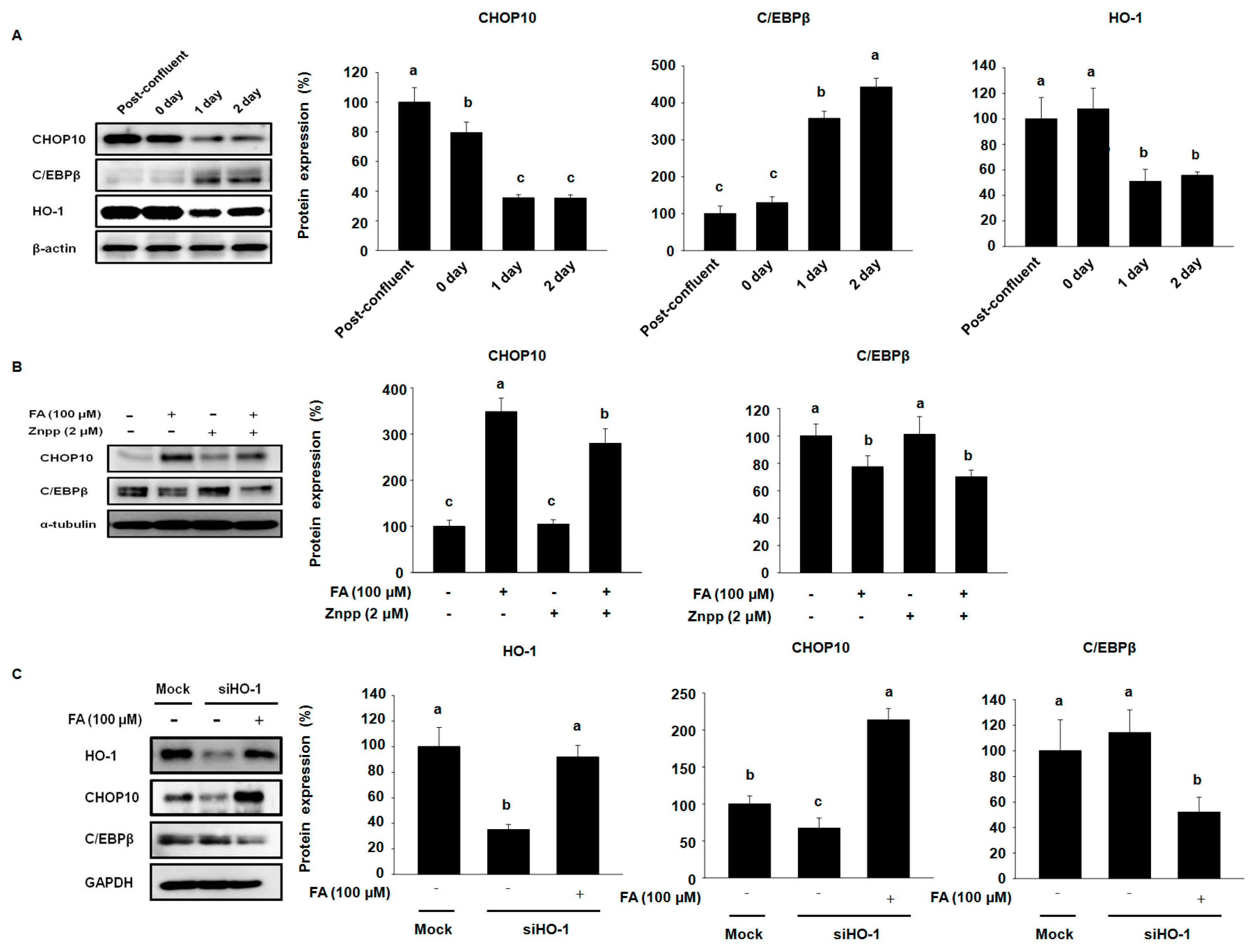
2.4. Effects of FA on Early Stage Adipocyte Differentiation Factors by Activating HO-1 in 3T3-L1 Cells
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Cytotoxicity Assay
4.4. Oil Red O Staining
4.5. Transfection (siRNA)
4.6. Isolation of RNA and RT-PCR
4.7. Western Blot Analysis
4.8. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Huttunen, R.; Syrjanen, J. Obesity and the risk and outcome of infection. Int. J. Obes. 2013, 37, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, K.; Barofsky, I. Obesity and health-related quality of life. Obes. Rev. 2001, 2, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Wellen, K.E.; Hotamisligil, G.S. Obesity-induced inflammatory changes in adipose tissue. J. Clin. Investig. 2003, 112, 1785. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, A.; Virbasius, J.V.; Puri, V.; Czech, M.P. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Atashi, F.; Modarressi, A.; Pepper, M.S. The role of reactive oxygen species in mesenchymal stem cell adipogenic and osteogenic differentiation: A review. Stem Cells Dev. 2015, 24, 1150–1163. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, Y.J.; Choi, H.; Ko, E.H.; Kim, J.W. Reactive oxygen species facilitate adipocyte differentiation by accelerating mitotic clonal expansion. J. Biol. Chem. 2009, 284, 10601–10609. [Google Scholar] [CrossRef] [PubMed]
- Giudice, A.; Arra, C.; Turco, M.C. Review of molecular mechanisms involved in the activation of the nrf2-are signaling pathway by chemopreventive agents. Methods Mol. Biol. 2010, 647, 37–74. [Google Scholar] [PubMed]
- Waldman, M.; Bellner, L.; Vanella, L.; Schragenheim, J.; Sodhi, K.; Singh, S.P.; Lin, D.; Lakhkar, A.; Li, J.; Hochhauser, E.; et al. Epoxyeicosatrienoic acids regulate adipocyte differentiation of mouse 3t3 cells, via pgc-1alpha activation, which is required for ho-1 expression and increased mitochondrial function. Stem. Cells Dev. 2016, 25, 1084–1094. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Kim, D.H.; Tsenovoy, P.L.; Peterson, S.J.; Rezzani, R.; Rodella, L.F.; Aronow, W.S.; Ikehara, S.; Abraham, N.G. Treatment of obese diabetic mice with a heme oxygenase inducer reduces visceral and subcutaneous adiposity, increases adiponectin levels, and improves insulin sensitivity and glucose tolerance. Diabetes 2008, 57, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Sacerdoti, D.; Olszanecki, R.; Li Volti, G.; Colombrita, C.; Scapagnini, G.; Abraham, N.G. Heme oxygenase overexpression attenuates glucose-mediated oxidative stress in quiescent cell phase: Linking heme to hyperglycemia complications. Curr. Neurovasc. Res. 2005, 2, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Rangwala, S.M.; Lazar, M.A. Transcriptional control of adipogenesis. Annu. Rev. Nutr. 2000, 20, 535–559. [Google Scholar] [CrossRef] [PubMed]
- Cristancho, A.G.; Lazar, M.A. Forming functional fat: A growing understanding of adipocyte differentiation. Nat. Rev. Mol. Cell Biol. 2011, 12, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Farmer, S.R. Transcriptional control of adipocyte formation. Cell Metab. 2006, 4, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Cases, S.; Smith, S.J.; Zheng, Y.W.; Myers, H.M.; Lear, S.R.; Sande, E.; Novak, S.; Collins, C.; Welch, C.B.; Lusis, A.J.; et al. Identification of a gene encoding an acyl coa: Diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc. Natl. Acad. Sci. USA 1998, 95, 13018–13023. [Google Scholar] [CrossRef] [PubMed]
- Ranganathan, G.; Unal, R.; Pokrovskaya, I.; Yao-Borengasser, A.; Phanavanh, B.; Lecka-Czernik, B.; Rasouli, N.; Kern, P.A. The lipogenic enzymes dgat1, fas, and lpl in adipose tissue: Effects of obesity, insulin resistance, and tzd treatment. J. Lipid Res. 2006, 47, 2444–2450. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Moghadasian, M.H. Chemistry, natural sources, dietary intake and pharmacokinetic properties of ferulic acid: A review. Food Chem. 2008, 109, 691–702. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, C.; Santangelo, R. Ferulic acid: Pharmacological and toxicological aspects. Food Chem. Toxicol. 2014, 65, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.C.; Hong, Q.; Wang, Y.G.; Liang, Q.D.; Tan, H.L.; Xiao, C.R.; Tang, X.L.; Shao, S.; Zhou, S.S.; Gao, Y. Ferulic acid induces heme oxygenase-1 via activation of erk and nrf2. Drug Discov. Ther. 2011, 5, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.L.; Yen, G.C. Effects of flavonoids and phenolic acids on the inhibition of adipogenesis in 3t3-l1 adipocytes. J. Agric. Food Chem. 2007, 55, 8404–8410. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, M.R.; Satyanarayana, M.N. Influence of capsaicin, curcumin and ferulic acid in rats fed high fat diets. J. Biosci. 1987, 12, 143–152. [Google Scholar] [CrossRef]
- Cao, Y. Adipose tissue angiogenesis as a therapeutic target for obesity and metabolic diseases. Nat. Rev. Drug Discov. 2010, 9, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Pruthi, V. Potential applications of ferulic acid from natural sources. Biotechnol. Rep. 2014, 4, 86–93. [Google Scholar] [CrossRef]
- Vanella, L.; Kim, D.H.; Asprinio, D.; Peterson, S.J.; Barbagallo, I.; Vanella, A.; Goldstein, D.; Ikehara, S.; Kappas, A.; Abraham, N.G. Ho-1 expression increases mesenchymal stem cell-derived osteoblasts but decreases adipocyte lineage. Bone 2010, 46, 236–243. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| Name | Forward (5′–3′) | Reverse (5′–3′) |
|---|---|---|
| HO-1 | CTGTGTAACCTCTGCTGTTCC | CCACACTACCTGAGTCTACC |
| PPARγ | CTGTGAGACCAACAGCCTGA | AATGCGAGTGGTCTTCCATC |
| C/EBPα | TGAAGGAACTTGAAGCACAA | TCAGAGCAAAACCAAAACAA |
| GAPDH | AACTTTGGCATTGTGGAAGG | ACACATTGGGGGTAGGA |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koh, E.-J.; Kim, K.-J.; Seo, Y.-J.; Choi, J.; Lee, B.-Y. Modulation of HO-1 by Ferulic Acid Attenuates Adipocyte Differentiation in 3T3-L1 Cells. Molecules 2017, 22, 745. https://doi.org/10.3390/molecules22050745
Koh E-J, Kim K-J, Seo Y-J, Choi J, Lee B-Y. Modulation of HO-1 by Ferulic Acid Attenuates Adipocyte Differentiation in 3T3-L1 Cells. Molecules. 2017; 22(5):745. https://doi.org/10.3390/molecules22050745
Chicago/Turabian StyleKoh, Eun-Jeong, Kui-Jin Kim, Young-Jin Seo, Jia Choi, and Boo-Yong Lee. 2017. "Modulation of HO-1 by Ferulic Acid Attenuates Adipocyte Differentiation in 3T3-L1 Cells" Molecules 22, no. 5: 745. https://doi.org/10.3390/molecules22050745
APA StyleKoh, E.-J., Kim, K.-J., Seo, Y.-J., Choi, J., & Lee, B.-Y. (2017). Modulation of HO-1 by Ferulic Acid Attenuates Adipocyte Differentiation in 3T3-L1 Cells. Molecules, 22(5), 745. https://doi.org/10.3390/molecules22050745





