Microwave-Assisted Synthesis of some Novel Azoles and Azolopyrimidines as Antimicrobial Agents
Abstract
:1. Introduction
2. Results and Discussion
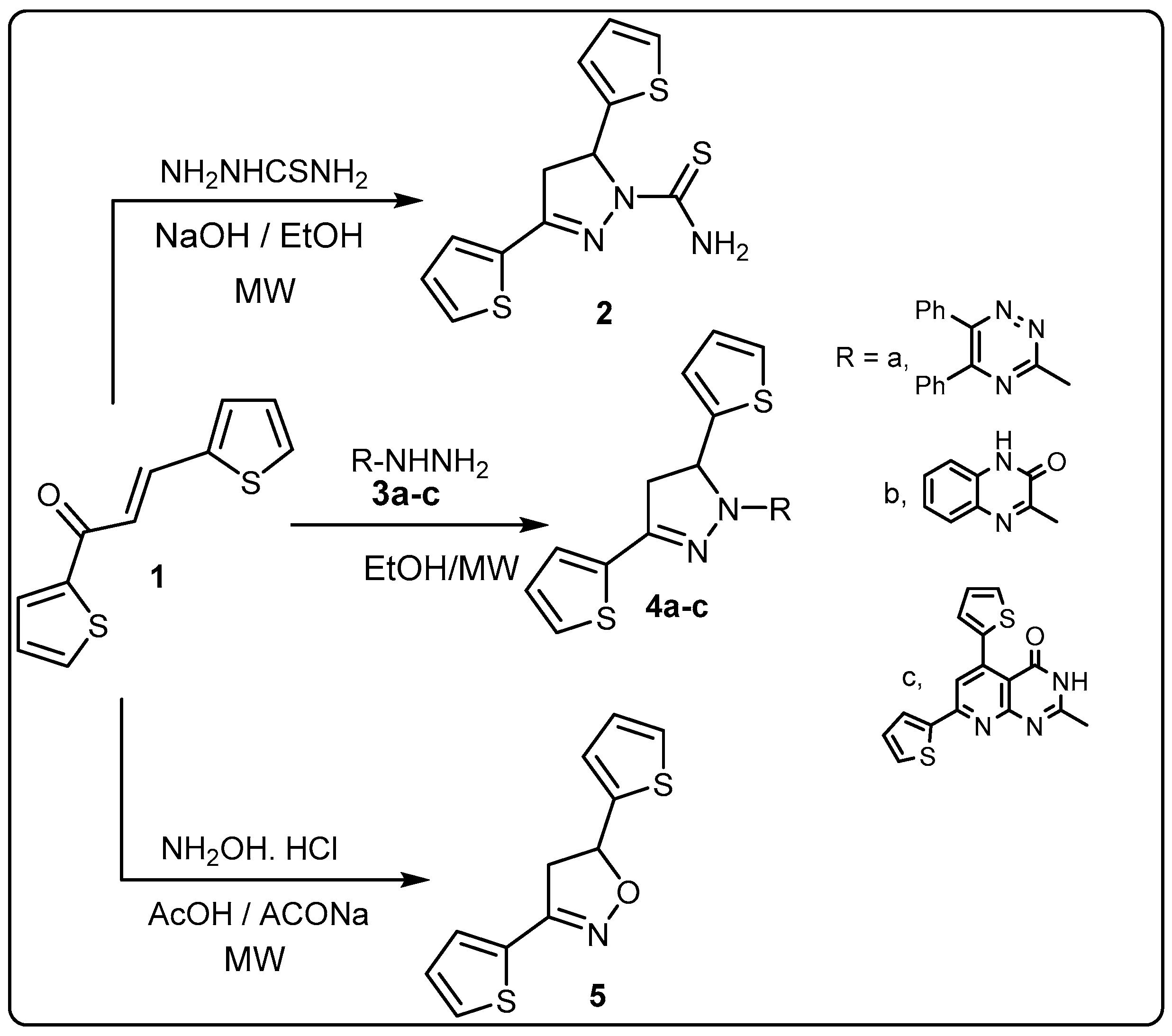
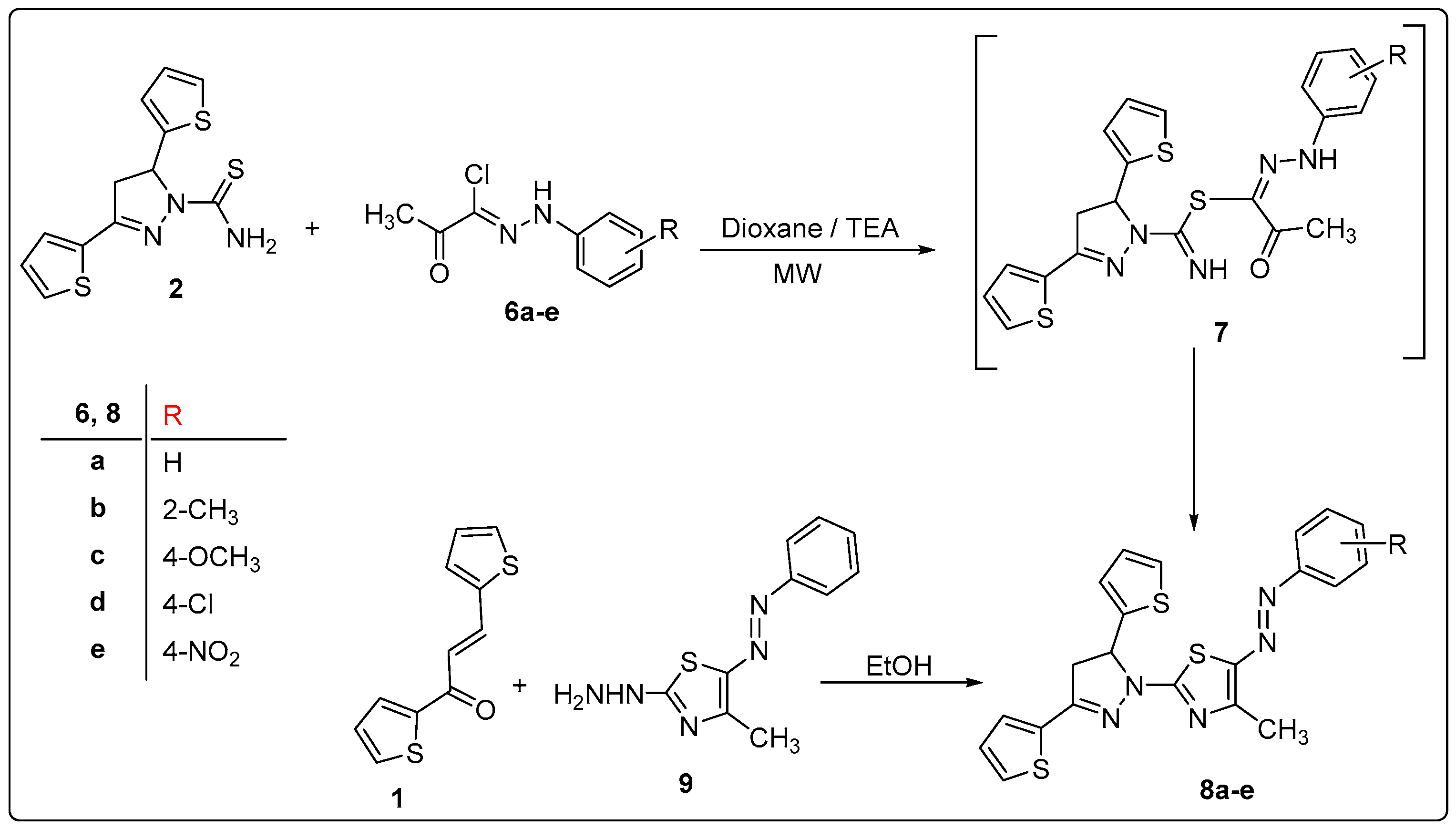
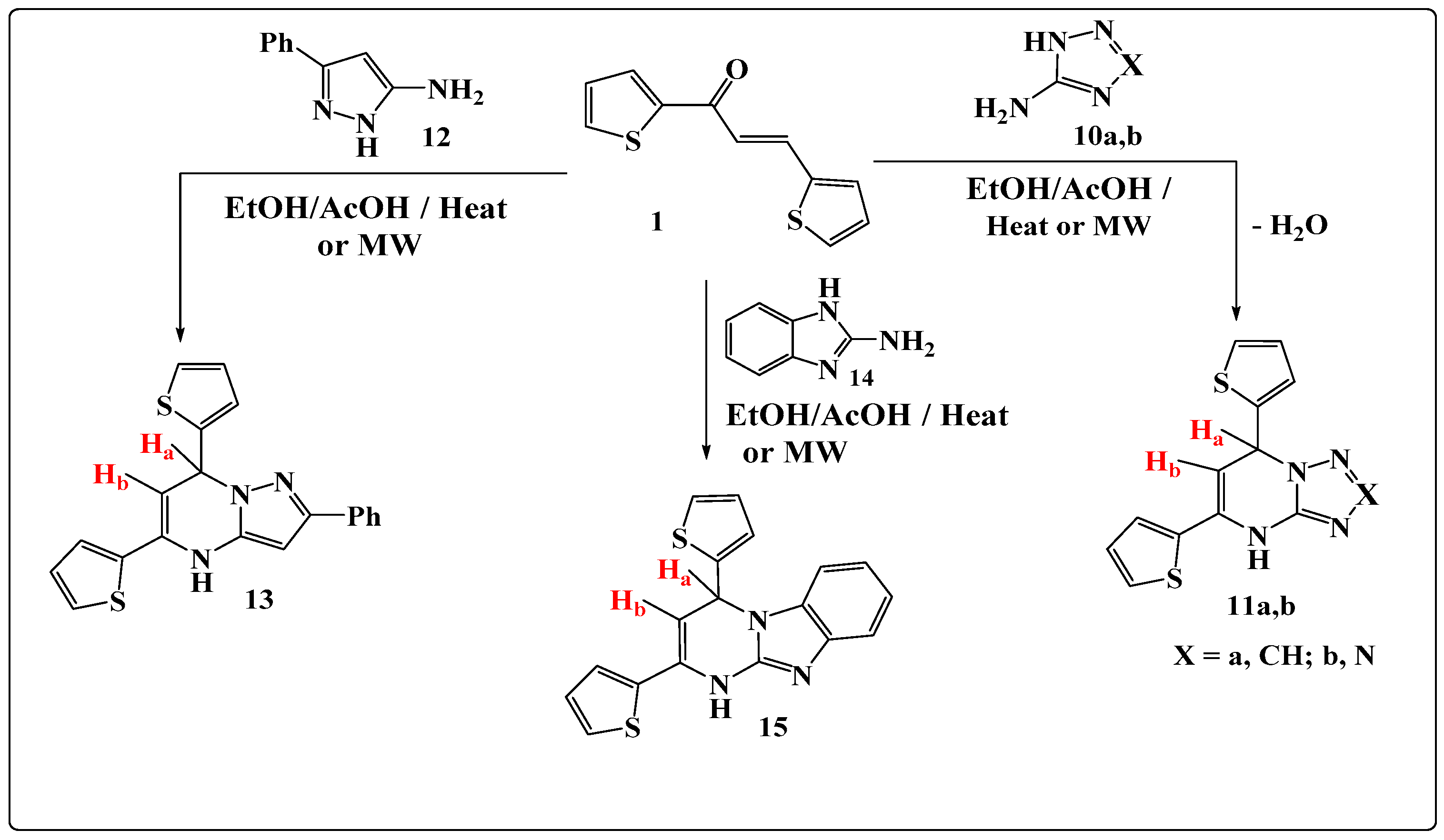
2.1. Synthesis
2.2. Antimicrobial Activity
3. Materials and Methods
3.1. General Experimental Procedures
3.2. Synthesis of Pyrazoline Derivatives 4a–c
3.3. 3,5-Di(thiophen-2-yl)-4,5-dihydroisoxazole (5)
3.4. Synthesis of 2-(3,5-Di(thiophen-2-yl)-4,5-dihydro-1H-pyrazol-1-yl)-4-methyl-5-(aryldiazenyl)thiazoles 8a–e
3.5. Alternate Synthesis of 8a
3.6. General Method for Synthesis of Compounds 11a,b, 13, and 15
3.7. Biological Activity
3.7.1. Antimicrobial Activity
3.7.2. Media Used
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mongevega, A.; Aldama, I.; Robbani, M.M.; Fernandez, E. The synthesis of 11H-1,2,4-triazolo[4,3-b]pyridazino[4,5-b]indoles,11H-tetrazolo[4,5-b]pyridazino[4,5-b]indoles and 1,2,4-triazolo[3,4-f]-1,2,4-triazino[4,5-a]indoles. J. Heterocycl. Chem. 1980, 17, 77–80. [Google Scholar] [CrossRef]
- Russe, L.R.K.; Press, J.B.; Rampulla, R.A. Thiophene systems. 9. Thienopyrimidinedione derivatives as potential antihypertensive agents. J. Med. Chem. 1988, 31, 1786–1793. [Google Scholar] [CrossRef]
- Abdelhamid, A.O. Convenient synthesis of some new pyrazolo[1,5-a]pyrimidine, pyridine, thieno[2,3-b]pyridine, and isoxazolo[3,4-d]pyridazine derivatives containing benzofuran moiety. J. Heterocycl. Chem. 2009, 46, 680–686. [Google Scholar] [CrossRef]
- Rashad, A.E.; Shamroukh, A.H.; Abdel-Megeid, R.E.; Mostafa, A.; El-Shesheny, R.; Kandeil, A.; Ali, M.A.; Banert, K. Synthesis and screening of some novel fused thiophene and thienopyrimidine derivatives for anti-avian influenza virus (H5N1) activity. Eur. J. Med. Chem. 2010, 45, 5251–5257. [Google Scholar] [CrossRef] [PubMed]
- Deka, S.; Mohan, S.; Saravanan, J.; Kakati, M.; Talukdar, A.; Sahariah, B.J.; Dey, B.K.; Sarma, R.K. Syntheses, characterization and in vitro anti-Inflammatory activity of some novel thiophenes. Maced. J. Med. Sci. 2012, 5, 159–163. [Google Scholar]
- Ghorab, M.M.; Bashandy, M.S.; Alsaid, M.S. Novel thiophene derivatives with sulfonamide, isoxazole, benzothiazole, quinoline and anthracene moieties as potential anticancer agents. Acta Pharm. 2014, 64, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Saad, H.A.; Youssef, M.M.; Mosselhi, M.A. Microwave assisted synthesis of some new fused 1,2,4-triazines bearing thiophene moieties with expected pharmacological activity. Molecules 2011, 16, 4937–4957. [Google Scholar] [CrossRef] [PubMed]
- Dawood, D.H.; Batran, R.Z.; Farghaly, T.A.; Khedr, M.A.; Abdulla, M.M. New Coumarin Derivatives as Potent Selective COX-2 Inhibitors; Synthesis, Anti-inflammatory, QSAR and Molecular Modeling Studies. Arch. Pharm. Chem. Life Sci. 2015, 348, 875–888. [Google Scholar] [CrossRef] [PubMed]
- Tewari, A.K.; Singh, V.P.; Yadav, P.; Gupta, G.; Singh, A.; Goel, R.K.; Shinde, P.; Mohan, G.C. Synthesis, biological evaluation and molecular modeling study of pyrazole derivatives as selective COX-2 inhibitors and anti-inflammatory agents. Bioorg. Chem. 2014, 56, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Ningaiah, S.; Bhadraiah, U.K.; Doddaramappa, S.D.; Keshavamurthy, S.; Javarasetty, C. Novel pyrazole integrated 1,3,4-oxadiazoles: Synthesis, characterization and antimicrobial evaluation. Bioorg. Med. Chem. Lett. 2014, 24, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Farghaly, T.A.; Gomha, S.M.; Sayed, A.R.; Khedr, M.A. Hydrazonoyl Halides as Precursors for Synthesis of Bioactive Thiazole and Thiadiazole Derivatives: Synthesis, Molecular Docking and Pharmacological Study. Curr. Org. Syn. 2016, 13, 445–455. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Abdallah, M.A.; Masaret, G.S.; Muhammad, Z.A. New and efficient approach for synthesis of novel bioactive [1,3,4]thiadiazoles incorporated with 1,3-thiazole moiety. Eur. J. Med. Chem. 2015, 97, 320–333. [Google Scholar] [CrossRef] [PubMed]
- Mert, S.; Kasimogullari, R.; Ica, T.; Colak, F.; Altun, A.; Ok, S. Synthesis, structure–activity relationships, and in vitro antibacterial and antifungal activity evaluations of novel pyrazole carboxylic and dicarboxylic acid derivatives. Eur. J. Med. Chem. 2014, 78, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.; Zhang, Z.; Zhuang, X.; Luo, J.; Cao, X.; Li, H.; Tu, Z.; Lu, X.; Ren, X.; Ding, K. New thiazolecarboxamides as potent inhibitors of Akt kinases. Bioorg. Med. Chem. Lett. 2012, 22, 1208–1212. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhao, B. Progress of the synthesis of condensed pyrazole derivatives (from 2010 to mid-2013). Eur. J. Med. Chem. 2014, 85, 311–340. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Wang, M.; Miller, K.D.; Zheng, Q.H. Synthesis and preliminary in vitro biological evaluation of new carbon-11-labeled celecoxib derivatives as candidate PET tracers for imaging of COX-2 expression in cancer. Eur. J. Med. Chem. 2011, 46, 4760–4767. [Google Scholar] [CrossRef] [PubMed]
- Meena, D.R.; Maiti, B.; Chanda, K. Cu(I) catalyzed microwave assisted telescopic synthesis of 3,5-disubstituted isoxazoles in green media. Tetrahedron Lett. 2016, 57, 5514–5517. [Google Scholar] [CrossRef]
- Balwe, S.G.; Shinde, V.V.; Jeong, Y.T. Iron-catalyzed microwave-promoted expeditious one-pot synthesis of benzo[b][1,4]thiazine-4-carbonitrile under solvent-free condition. Tetrahedron Lett. 2016, 57, 5074–5078. [Google Scholar] [CrossRef]
- Abbas, E.M.H.; Gomha, S.M.; Farghaly, T.A. Multicomponent Reactions for Synthesis of Bioactive Polyheterocyclic Ring Systems under Controlled Microwave Irradiation. Arabian J. Chem. 2014, 7, 623–629. [Google Scholar] [CrossRef]
- Gomha, S.M.; Riyadh, S.M. Synthesis of triazolo[4,3-b][1,2,4,5]tetrazines and triazolo[3,4-b][1,3,4]thiadiazines using chitosan as ecofriendly catalyst under microwave irradiation. ARKIVOC 2009, 2009, 58–68. [Google Scholar]
- Gomha, S.M.; Eldebss, T.M.A.; Badrey, M.G.; Abdulla, M.M.; Mayhoub, A.S. Novel 4-Heteroaryl-Antipyrines as DPP-IV Inhibitors. Chem. Biol. Drug Des. 2015, 86, 1292–1303. [Google Scholar] [CrossRef] [PubMed]
- Awad, H.M.; Fayad, W.; El-Hallouty, S.M.; Farghaly, T.A.; Abdallah, M.M. Anticancer Activity of Some [1,2,4]Triazepino[2,3-a]Quinazoline Derivatives: Monolayer and Multicellular Spheroids In Vitro Models. Med. Chem. Res. 2016, 25, 1952–1957. [Google Scholar] [CrossRef]
- Gomha, S.M.; Farghaly, T.A.; Sayed, A.R.; Abdalla, M.M. Synthesis of pyrazolyl-pyrazoles and pyrazolyl-[1,2,4]-triazolo[3,4-d][1,5]benzothiazepines as p53 activators using hydrazonoyl chlorides. J. Heterocycl. Chem. 2016, 53, 1503–1509. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Gomha, S.M.; Mousa, E.K.; Elaasser, M. Hydrazonoyl chlorides in synthesis of pyrazolo[5,1-c][1,2,4]triazole derivatives and their biological activities. J. Chem. Res. 2016, 40, 467–470. [Google Scholar] [CrossRef]
- Abbas, E.M.H.; Gomha, S.M.; Farghaly, T.A.; Abdalla, M.M. Synthesis of new thiazole derivatives as antitumor agents. Curr. Org. Syn. 2016, 13, 456–465. [Google Scholar] [CrossRef]
- Farghaly, T.A.; Gomha, S.M.; Abbas, E.M.H.; Abdalla, M.M. Hydrazonoyl halides as precursors for new fused heterocycles of 5 α-reductase inhibitors. Arch. Pharm. 2012, 345, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Khloya, P.; Kumar, S.; Kaushik, P.; Surain, P.; Kaushik, D.; Sharma, P.K. Synthesis and biological evaluation of pyrazolylthiazole carboxylic acids as potent anti-inflammatory-antimicrobial agents. Bioorg. Med. Chem. Lett. 2015, 25, 1177–1181. [Google Scholar] [CrossRef] [PubMed]
- Turan, Z.G.; Chevallet, P.; Kilic, T.S.; Erolic, K. Synthesis of some thiazolyl-pyrazoline derivatives and preliminary investigation of their hypotensive acivity. Eur. J. Med. Chem. 2000, 35, 635–641. [Google Scholar] [CrossRef]
- Nishida, S.; Maruoka, H.; Yoshimura, Y.; Goto, T.; Tomita, R.; Masumoto, E.; Okabe, F.; Yamagata, K.; Fiyioka, T. Synthesis and biological activities of some new thizolidine derivatives containing pyrazole ring system. J. Heterocycl. Chem. 2012, 49, 303–309. [Google Scholar] [CrossRef]
- Ozdemir, Z.; Kandilci, H.B.; Gumusel, B.; Calis, U.; Bilgin, A.A. Synthesis and Studies on Antidepressant and Anticonvulsant Activities of Some 3-(2-Thienyl)pyrazoline Derivatives. Arch. Pharm. 2008, 341, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Eweiss, N.F.; Osman, A. Synthesis of heterocycles. Part II new routes to acetylthiadiazolines and alkylazothiazoles. J. Heterocycl. Chem. 1980, 17, 1713–1717. [Google Scholar] [CrossRef]
- Abdelhamid, A.O.; Sayed, A.R. Reaction of Hydrazonoyl Halides 52: Synthesis and Antimicrobial Activity of Some New Pyrazolines and 1,3,4-Thiadiazolines. Phosphorus Sulfur Silicon Relat. Elem. 2007, 182, 1767–1777. [Google Scholar] [CrossRef]
- Hossain, M.A.; Shah, M.D.; Sang, S.V.; Sakari, M. Chemical composition and antibacterial properties of the essential oils and crude extracts of Merremiaborneensis. J. King Saud Univ. Sci. 2012, 24, 243–249. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 2, 4, 5, 6, 8, 11, 13 and 15 are available from the authors.
| Compound No. | Reaction Times | Reaction Yields (%) | ||
|---|---|---|---|---|
| Conventional Methods | Microwave | Conventional Methods | Microwave | |
| 2 | 2 h [30] | 3 min | 66 [30] | 84 |
| 4a | 4 h | 5 min | 70 | 85 |
| 4b | 5 h | 8 min | 73 | 90 |
| 4c | 5h | 10 min | 69 | 88 |
| 5 | 6 h | 10 min | 67 | 82 |
| 8a | 6 h | 8 min | 74 | 95 |
| 8b | 8 h | 10 min | 76 | 90 |
| 8c | 10 h | 12 min | 68 | 92 |
| 8d | 8 h | 9 min | 72 | 93 |
| 8e | 10 h | 13 min | 75 | 90 |
| 11a | 10 h | 12 min | 70 | 89 |
| 11b | 15 h | 15 min | 60 | 81 |
| 13 | 10 h | 15 min | 67 | 88 |
| 15 | 13 h | 20 min | 60 | 85 |
| Compound Number | Fungi | Gram Positive Bacteria | Gram Negative Bacteria | ||
|---|---|---|---|---|---|
| ASP | CA | SA | BS | EC | |
| 2 | NA. | NA. | 21 | 19 | 23 |
| 4a | N.A. | N.A. | 18 | 20 | 15 |
| 4b | N.A. | 20 | 17 | 18 | 18 |
| 4c | N.A. | 23 | 22 | 20 | 17 |
| 5 | N.A. | 9 | 19 | 12 | 17 |
| 8a | N.A | 10 | 12 | 18 | 11 |
| 8b | N.A. | 8 | 21 | 18 | 12 |
| 8c | N.A. | 24 | 18 | 15 | 23 |
| 8d | N.A. | N.A. | 22 | 12 | 17 |
| 8e | N.A. | 8 | 18 | 14 | 13 |
| 11a | N.A. | 9 | N.A. | N.A. | 10 |
| 11b | N.A. | 25 | 19 | 17 | 11 |
| 13 | N.A. | 12 | 14 | 11 | 8 |
| 15 | N.A. | 11 | 21 | 19 | 15 |
| Chloramphenicol | 29 | 25 | 30 | 24 | 29 |
| Trimethoprim/sulphamethoxazole | 2.4 | 13 | 20 | 23 | 24 |
| DMSO | N.A. | N.A. | N.A. | N.A. | N.A. |
High activity  Moderate activity Moderate activity  Low activity Low activity  N.A. (No activity) N.A. (No activity)  | |||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomha, S.M.; Farghaly, T.A.; Mabkhot, Y.N.; Zayed, M.E.M.; Mohamed, A.M.G. Microwave-Assisted Synthesis of some Novel Azoles and Azolopyrimidines as Antimicrobial Agents. Molecules 2017, 22, 346. https://doi.org/10.3390/molecules22030346
Gomha SM, Farghaly TA, Mabkhot YN, Zayed MEM, Mohamed AMG. Microwave-Assisted Synthesis of some Novel Azoles and Azolopyrimidines as Antimicrobial Agents. Molecules. 2017; 22(3):346. https://doi.org/10.3390/molecules22030346
Chicago/Turabian StyleGomha, Sobhi M., Thoraya A. Farghaly, Yahia Nasser Mabkhot, Mohie E. M. Zayed, and Amany M. G. Mohamed. 2017. "Microwave-Assisted Synthesis of some Novel Azoles and Azolopyrimidines as Antimicrobial Agents" Molecules 22, no. 3: 346. https://doi.org/10.3390/molecules22030346
APA StyleGomha, S. M., Farghaly, T. A., Mabkhot, Y. N., Zayed, M. E. M., & Mohamed, A. M. G. (2017). Microwave-Assisted Synthesis of some Novel Azoles and Azolopyrimidines as Antimicrobial Agents. Molecules, 22(3), 346. https://doi.org/10.3390/molecules22030346







