Synthesis, Characterization and Antibacterial Studies of N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide and Its Neodymium(III) and Thallium(III) Complexes
Abstract
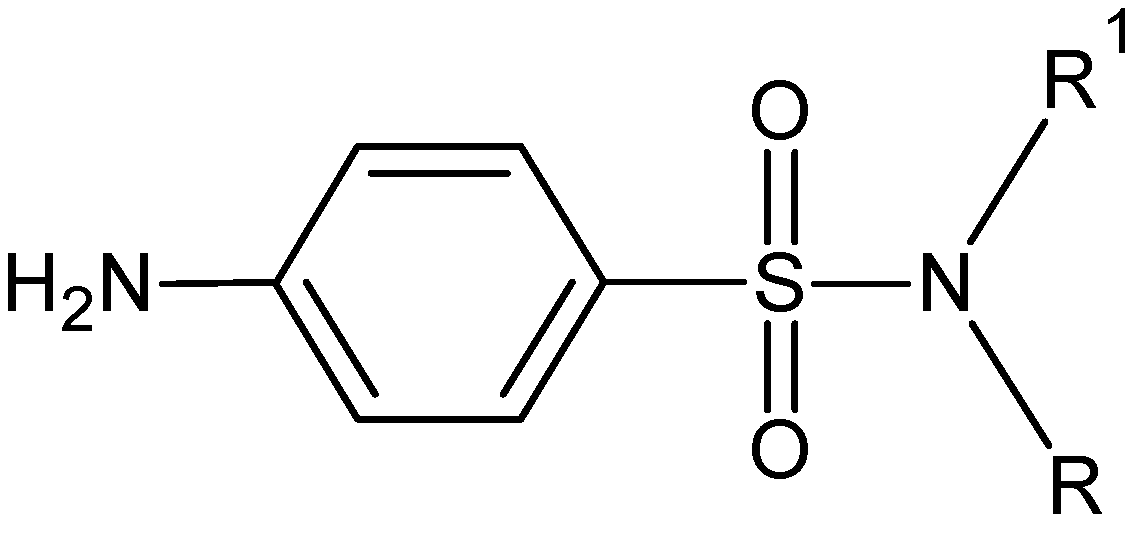
:1. Introduction
2. Results and Discussion
2.1. Electronic Spectra
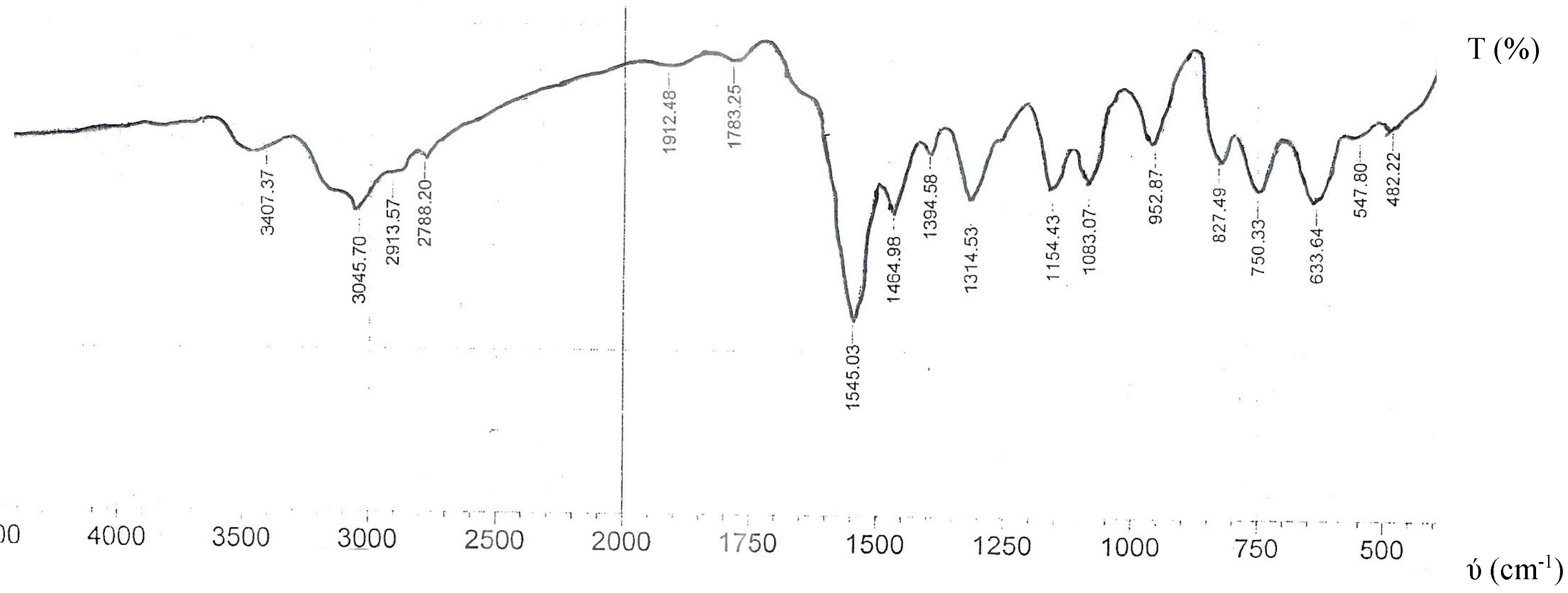
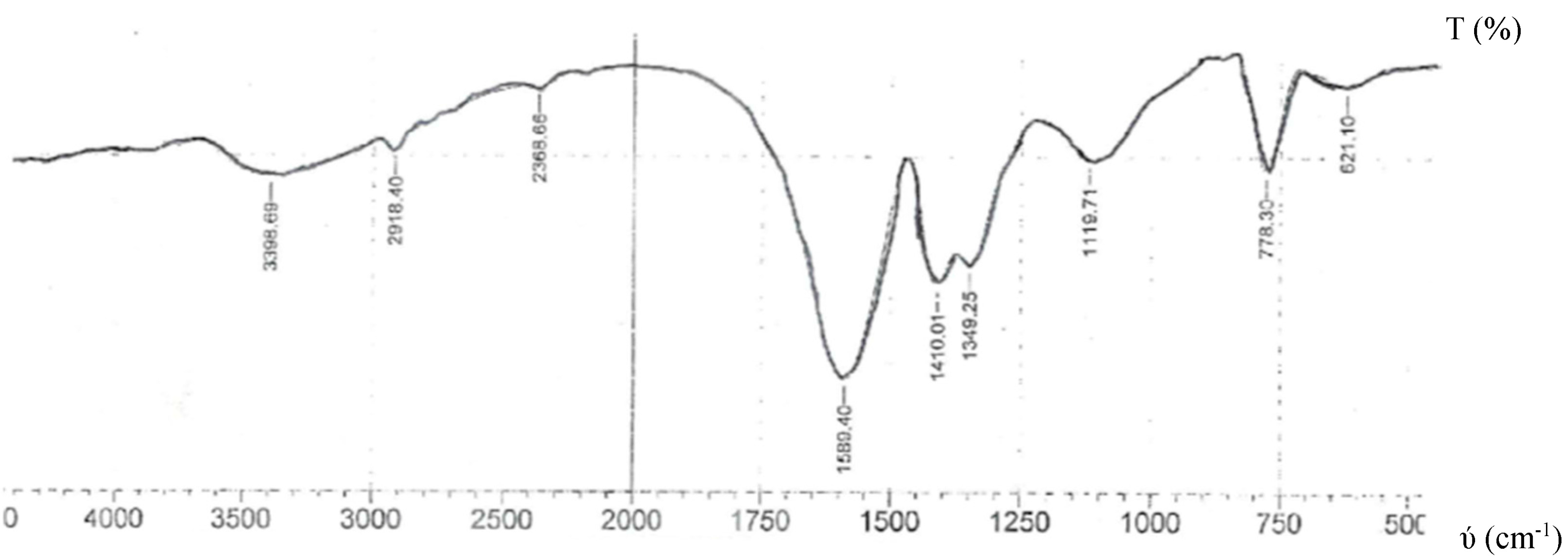
2.2. Infrared Spectra
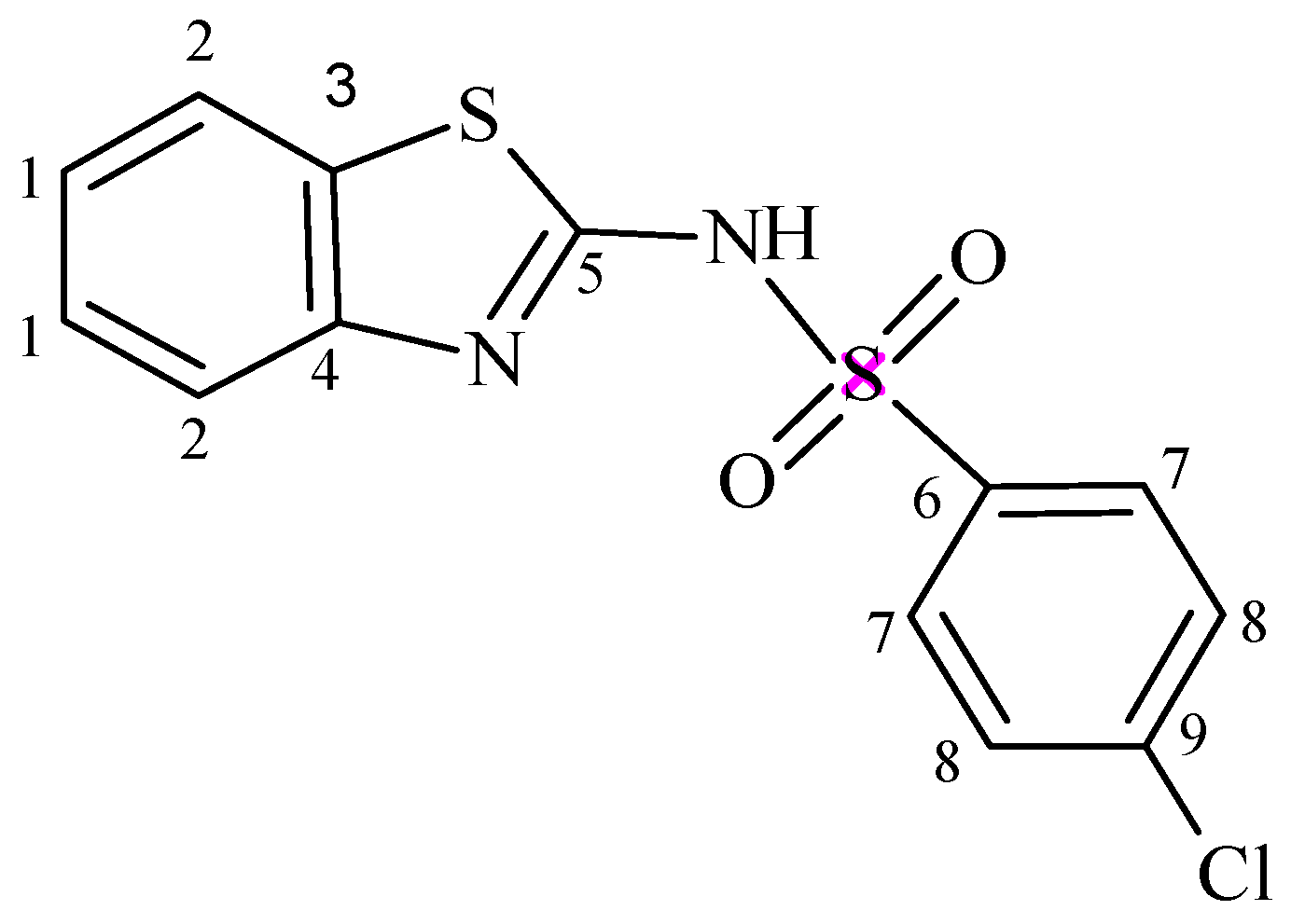
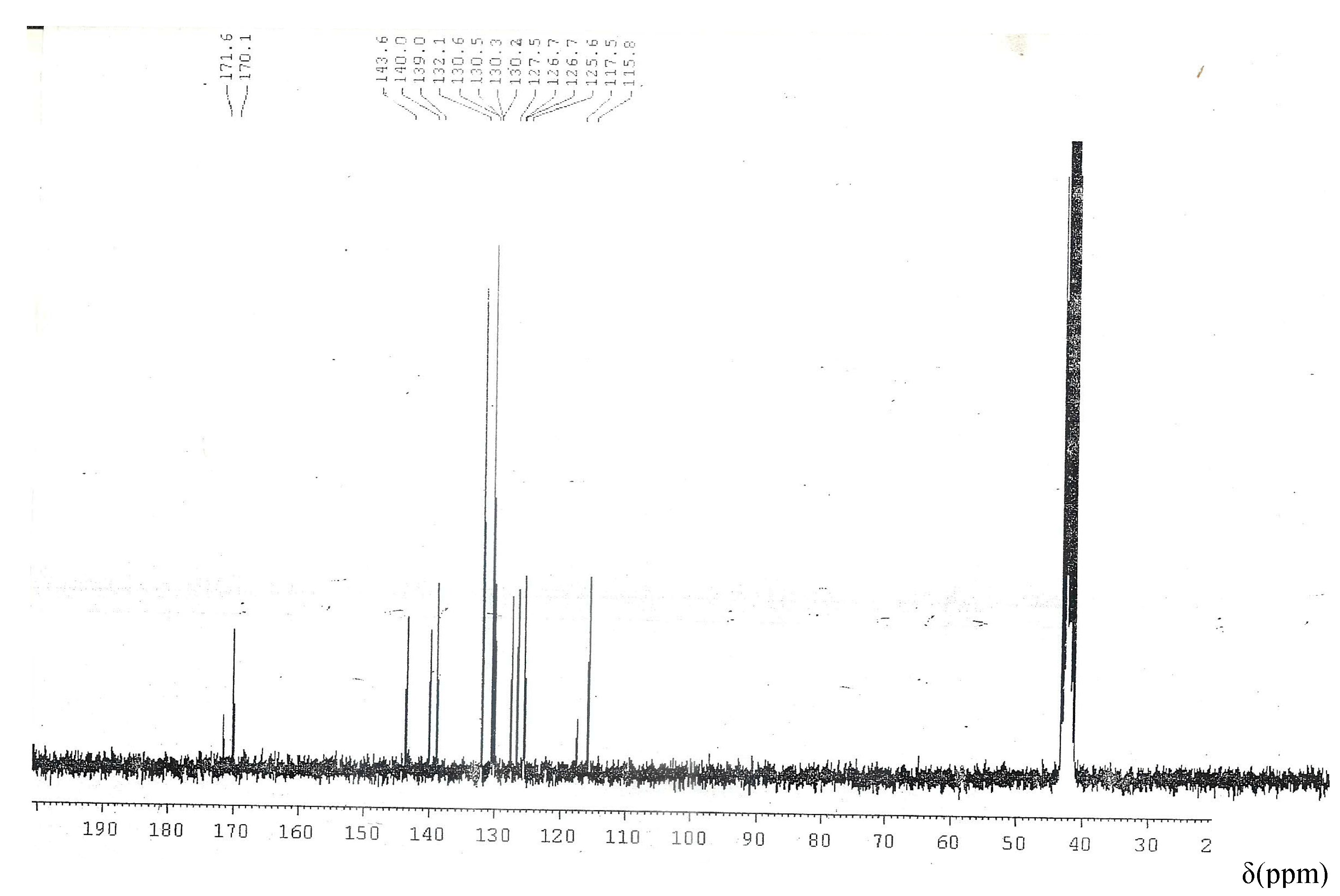
2.3. 1H-NMR and 13C-NMR Spectra
2.4. Antibacterial Activity
3. Materials and Methods
3.1. Materials
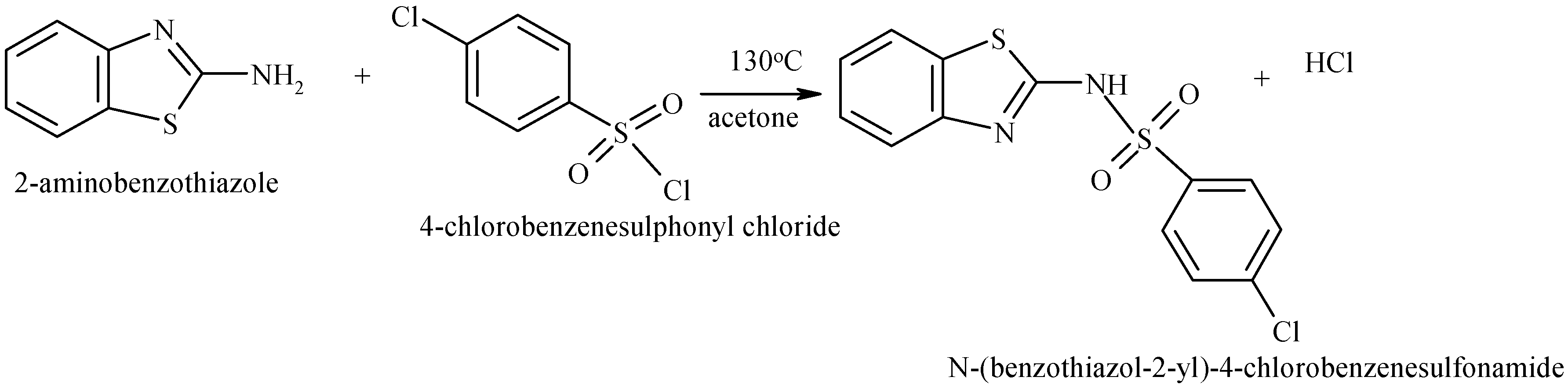
3.2. Synthesis of N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide (NBTCS)
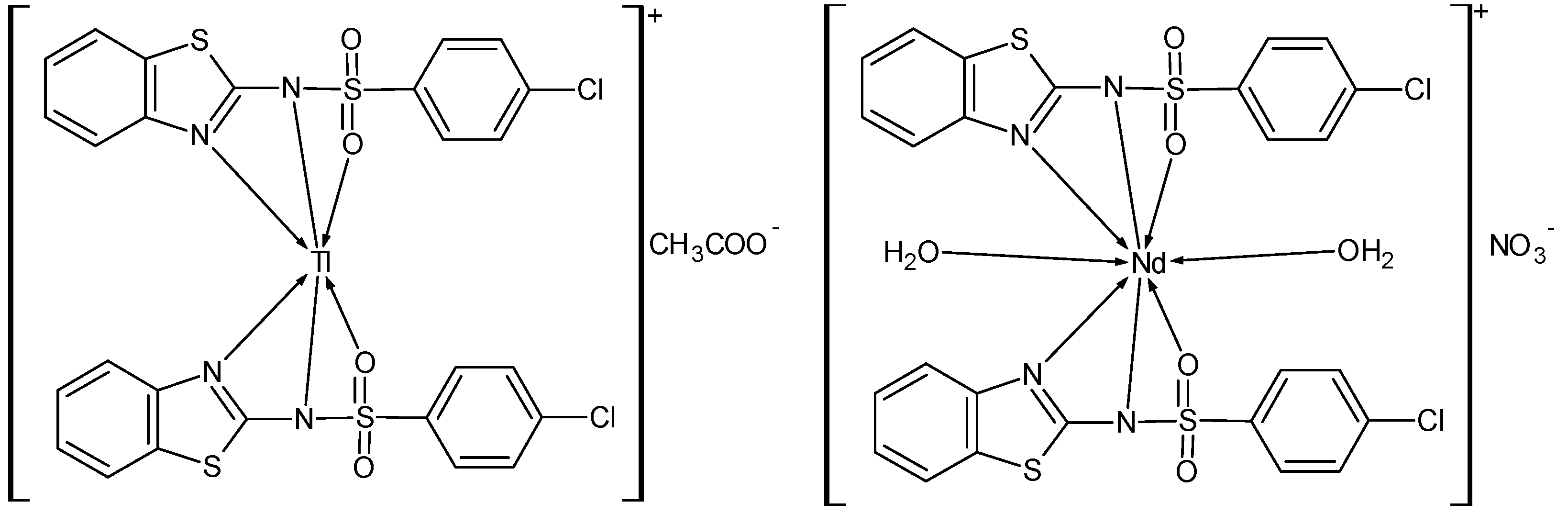
3.3. Synthesis of [N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide] Neodymium(III) Complex, [Nd(NBTCS)2(H2O)2]NO3
3.4. Synthesis of [N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide] Thallium(III) Complex, [Tl(NBTCS)2]CH3COO
3.5. Antibacterial Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yun, M.-K.; Wu, Y.; Li, Z.; Zao, Y.; Waddell, M.B.; Ferreira, A.M.; Lee, R.E.; Bashford, D.; White, S.W. Catalysis and Sulpha Drugs Resistance in Dihydropteroate Synthase. Science 2012, 335, 1110–1114. [Google Scholar] [CrossRef] [PubMed]
- Bhat, M.A.; Imran, M.; Khan, S.A.; Siddioui, N. Biological activities of Sulfonamides. Indian J. Pharm. Sci. 2005, 67, 151–159. [Google Scholar]
- Guar, A.; Gupta, R.K.; Shrivastava, B. Colorimetric Estimation of Sulphacetamide Sodium in bulk and in Formulation. Indian J. Pharm. Sci. 2007, 69, 464–467. [Google Scholar] [CrossRef]
- Genç, Y.; Özkanca, R.; Bekdemir, Y. Antimicrobial activity of some sulphonamide derivatives on clinical isolates of Staphylococus aureus. Ann. Clin. Microbiol. Antimicrob. 2008, 7, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Ozbek, N.; Katircioğlu, H.; Karacan, N.; Baykal, T. Synthesis, characterization and antimicrobial activity of new aliphatic sulfonamide. Bioorg. Med. Chem. 2007, 15, 5105–5109. [Google Scholar] [CrossRef] [PubMed]
- Kaur, I.P.; Smitha, R.; Aggarwal, D.; Kapil, M. Acetazolamide: Future Perspective in Topical Glaucoma Therapeutics. Int. J. Pharm. 2002, 248, 1–14. [Google Scholar] [CrossRef]
- Arnold, P.J. The Chemistry of metal Complexes with Selenolate and Tellurolate Ligands. Prog. Inorg. Chem. 1995, 43, 353–417. [Google Scholar]
- Ghorab, M.M.; Ragab, F.A.; Hamed, M.M. Design, synthesis and anticancer evaluation of Novel tetrahydroquinoline derivatives containing sulfonamide moiety. Eur. J. Med. Chem. 2009, 44, 4211–4217. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, N.S.; El-Bendary, E.R.; El-Ashry, S.M.; El-Kerdawy, M.M. Synthesis and antitumora ctivity of new sulphonamide derivatives of thiadiazolo[3,2-a]pyrimidines. Eur. J. Med. Chem. 2011, 46, 3714–3720. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Fuld, A.D.; Rigas, J.R.; Hagey, A.E.; Gordon, G.B.; Dmitrovsky, E.; Dragnev, K.H. A Phase I Trial and in vitro Studies Combining ABT-751 with Carboplatin in Previously Treated Non-Small Cell Lung Cancer Patients. Chemotherapy 2012, 58, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Borne, R.F.; Peden, R.L.; Waters, I.W.; Weiner, M.; Jordan, R.; Coats, E.A. Anti-inflammatory activity of para-substituted N-benzenesulfonyl derivatives of anthranilic acid. J. Pharm. Sci. 1974, 63, 615–617. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.; Thorley, M. Pharmaceutical Substances, 3rd ed.; Kleeman, A., Engel, J., Kutscher, B., Reichert, D., Eds.; Thieme: Stuttgart, Germany, 1999; p. 27. [Google Scholar]
- Kanda, Y.; Kawanishi, Y.; Oda, K.; Sakata, T.; Mihara, S.; Asakura, K.; Kanemasa, T.; Ninomiya, M.; Fujimoto, M.; Kanoike, T. Synthesis and structure-activity relationships of potent and orally active sulfonamide ETB selective antagonists. Bioorg. Med. Chem. 2001, 9, 897–907. [Google Scholar] [CrossRef]
- De Clercq, E. New developments in anti-HIV chemotherapy. Curr. Med. Chem. 2001, 8, 1543–1572. [Google Scholar] [CrossRef] [PubMed]
- Dekker, M. Protease Inhibitors in AIDS Therapy; Ogden, R.C., Flexner, C.W., Eds.; NY Bagel: New York, NY, USA, 2001; p. 112. [Google Scholar]
- Kent, M. Advanced Biology; Oxford University Press: Oxford, UK, 2000; p. 46. [Google Scholar]
- Brackett, C.C.; Singh, H.; Block, J.H. Likelihood and Mechanisms of Cross-allergenicity between Sulfonamide antibiotics and other drugs containing a Sulfonamide functional group. Pharmacotherapy 2004, 24, 856–870. [Google Scholar] [CrossRef] [PubMed]
- Slatore, C.G.; Tilles, S. Sulfonamide hypersensitivity. Immunol. Allergy Clin. N. Am. 2004, 24, 477–490. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, H.R.; Kazi, A.; Luo, Y.; Kendig, R.; Ge, Y.; Jain, S.; Daniel, K.; Santiago, D.; Guida, W.C.; Sebti, S.M. Synthesis and biological evaluation of naphthoquinone analogs as a novel class of proteasome inhibitors. Bioorg. Med. Chem. 2010, 18, 5576–5592. [Google Scholar] [CrossRef] [PubMed]
- Obasi, L.N.; Okoye, C.O.B.; Ukoha, P.O.; Anaga, A.O. Syntheses, Characterization, and Antimicrobial Screening of N-(benzothiazol-2-yl) benzenesulphonamide and its Cu(I), Ni(II), Mn(II), Co(II), and Zn(II) Complexes. E-J. Chem. 2012, 9, 2354–2370. [Google Scholar] [CrossRef]
- Obasi, L.N.; Okoye, C.O.B.; Ukoha, P.O.; Chah, K.F. Synthesis, Characterization and Antimicrobial Screening of N-(benzothiazol-2-yl)-2,5-dichlorobenzenesulphonamide and its Cu(I), Ni(II), Mn(II), Co(III) and Zn(II) Complexes. Asian J. Chem. 2013, 25, 2199–2207. [Google Scholar]
- Obasi, L.N.; Okoye, C.O.B.; Ukoha, P.O.; Chah, K.F. Synthesis, characterization and Antimicrobial Screening of N-(benzothiazol-2-yl)-4-methylbenzenesulphonamide and its Cu(I), Ni(II), Mn(II), Co(II) and Zn(II) Complexes. Asian J. Chem. 2013, 25, 2369–2376. [Google Scholar]
- Caira, M.R. Molecular complexes of sulfonamides. 2.1:1 complexes between drug molecules: Sulfadimidine-acetylsalicylic acid and sulfadimidine-4-aminosalicylic acid. J. Chem. Crystallogr. 1992, 22, 193–200. [Google Scholar] [CrossRef]
- Chohan, H.Z.; Mahmood-ul-Hassan; Khan, K.M.; Supuran, C.T. In-vitro antibacterial, antifungal and cytotoxic properties of Sulfonamide-derived Schiff’s bases and their metal complexes. J. Enzym. Inhib. Med. Chem. 2005, 20, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Obaleye, J.A.; Adediji, J.F.; Adebayo, M.A. Synthesis and Biological Activities on Metal Complexes of 2,5-diamino-1,3,4-thiadiazole derived from Semicarbazide Hydrochloride. Molecules 2011, 16, 5861–5874. [Google Scholar] [CrossRef] [PubMed]
- Issa, R.M.; Azim, S.A.; Khedr, A.M.; Draz, D.F. Synthesis, Characterization, thermal and antimicrobial studies of binuclear metal complexes of Sulfa-guanidine Schiff bases. J. Coord. Chem. 2009, 62, 1859–1870. [Google Scholar] [CrossRef]
- Narang, K.; Gupta, J.K. Copper(II) acetate and Picrate Complexes of Sulfa drugs. Transit. Met. Chem. 1977, 2, 83–86. [Google Scholar] [CrossRef]
- Jitianu, A.; Llies, M.A.; Scozzafava, A.; Supuran, C.T. Synthesis and Carbonic Anhydrase Inhibitory activities of 5-Benzoylamido- and 5-(3-nitrobenzoylamido)-1,3,4-thiadiazole-2-sulfonamide and their metal complexes. Main Group Met. Chem. 1997, 20, 151–156. [Google Scholar] [CrossRef]
- Oruma, U.S.; Ukoha, P.O.; Asegbeloyin, J.N. Synthesis, Characterization and Biological Studies of S-1,3-Benzothiazol-2-ylthiophene-2-carbothioate and Its Ce(IV) and Nd(III) Complexes. Asian J. Chem. 2014, 26, 7622–7626. [Google Scholar]
- Rinehart, J.D.; Long, J.R. Slow Magnetic Relaxation in Homoleptic Trispyrazolylborate Complexes of Neodymium(III) and Uranium(III). Dalton Trans. 2012, 14, 13572–13574. [Google Scholar] [CrossRef] [PubMed]
- Khorasani-Motlagh, M.; Norozifar, M.; Niroomand, S.; Saffari, J. Synthesis and Spectroscopy Studies of new Neodymium(III) Complexes with Cyanamide Derivatives as N-Donor Ligands. J. Iran. Chem. Soc. 2010, 4, 807–813. [Google Scholar] [CrossRef]
- Kostova, I.; Manolov, I.; Momekov, G. Cytotoxic Activity of new Neodymium(III) Complexes of Bis-Coumarins. Eur. J. Med. Chem. 2004, 39, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Romanov, A.S.; Muratov, D.V.; Timofeeva, T.V.; Kudinov, A.R. Triple-decker complex CpCo(µ-C3B2Me5)Rh(C2H4)2: Synthesis, structure and bonding. Inorg. Chim. Acta 2014, 415, 120–123. [Google Scholar] [CrossRef]
- Kumar, S.; PalSharma, R.; Saini, A.; Venugopalan, P.; Starynowicz, P. Synthesis, characterization, single crystal X-ray, structure determination and packing analysis of thallium(I)anthracene-9-carboxylate, [Tl4(H2O2)(anthracene-9-carboxylate)4]. J. Mol. Struct. 2015, 1079, 291–297. [Google Scholar] [CrossRef]
- Sivagurunathan, G.S.; Ramalingam, K.; Rizzoli, C. Nanothallium(III)sulfide from dithiocarbamate precursors: Synthesis. Single crystal X-ray structures and characterization. Polyhedron 2013, 65, 316–321. [Google Scholar] [CrossRef]
- Gharib, F.; Feizabadi, M.; Soltani, L. Equilibrium studies of thallium(I) complexes with cytidine 5′-monophosphate in different aqueous solutions of methanol. J. Mol. Liquids 2013, 182, 64–69. [Google Scholar] [CrossRef]
- Rana, S.; Sharma, N.; Ojha, H.; Shivkumar, H.G.; Sultana, S.; Sharma, R.K. p-Tertbutylcalix[4]arenenanoemulsion: Preparation, characterization and comparative evaluation of its decontamination efficacy against Technetium-99m, Iodine 131 and Thallium-201. Colloids Surf. B Biointerfaces 2014, 117, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Mimouni, M.; Khardli, F.Z.; Warad, I.; Ahmad, M.; Mubarak, M.S.; Sultana, S.; Hadda, T.B. Antimicrobial Activity of Naturally occurring Antibiotics Monensin, Lasalocid and their Metal Complexes. J. Mater. Environ. Sci. 2014, 5, 207–214. [Google Scholar]
- Marc, A.; Daniel, H. Crystal structure of the thallium salt of the antibiotics Grisorixin. J. Chem. Soc. Chem. Commun. 1972, 175–176. [Google Scholar] [CrossRef]
- Rutkowski, J.; Brzezinski, B. Structures and Properties of Naturally Occurring Polyether Antibiotics. BioMed Res. Int. 2013, 2013, 162513. [Google Scholar] [CrossRef] [PubMed]
- Imran, A.; Waseem, A.W.; Kishwar, S. Empirical Fomulae to Molecular Structures of Metal Complexes by Molar Conductance. Synth. React. Inorg. Metal-Organ. Nano-Met. Chem. 2013, 43, 1162–1170. [Google Scholar]
- Lekha, L.; Raja, K.K.; Rajagopal, G.; Easwaramoorthy, D. Synthesis, Spectroscopic Characterization and antibacterial studies of lanthanide(III) Schiff base complexes containing N, O donor atoms. J. Mol. Struct. 2014, 1056–1057, 307–313. [Google Scholar] [CrossRef]
- Mamdouh, S.M.; Ekram, A.K.; Ahmad, M.R.; Yousry, M.G.; Amr, S. Spectral, electrical conductivity and biological activity properties of some new azopyrimidine derivatives and their complexes. Spectrochim. Acta A 2007, 67, 669–677. [Google Scholar]
- Obasi, L.N.; Ukoha, P.O.; Chah, K.F. Synthesis, Spectroscopic Characterization and Antimicrobial Screening of N-(thiazol-2-yl)-4-chlorobenzenesulphonamide and its Ni(II) and Co(II) Complexes. J. Chem. Soc. Niger. 2011, 36, 102–109. [Google Scholar]
- Dawood, Z.F.; Al-Moula, F.J.A. Preparation and Characterization of Cr(III) and Cu(II) Complexes With Mixed Ligand Containing Isatinazine and Semicarbazones. J. Univ. Anbar Pure Sci. 2009, 3, 21–31. [Google Scholar]
- Robert, M.; Francis, X.; David, J. Spectrometric Identification of Organic Compounds, 7th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; pp. 89, 95–97, 200. [Google Scholar]
- Ummathur, M.B.; Sayudevi, P.; Krishnankutty, K. Schiff bases of 3-[2-(1,3-benzothiazol-2-yl)hydrazinylidene]pentane-2,4-dione with aliphatic diamines and their metal complexes. J. Argent. Chem. Soc. 2009, 97, 31–39. [Google Scholar]
- Collins, C.; Lynes, P.; Grange, J. Microbiological Methods, 7th ed.; Butterwort-Heinemann Ltd.: Oxford, UK, 1995; pp. 175–190. [Google Scholar]
- Galbraith, A.; Bullock, S.; Manias, E.; Hunt, E.; Richard, A. Fundamentals of Pharmacology. An Applied Approach for Nursing and Health, 2nd ed.; Pearson Education Limited: Harlow, UK, 2007; pp. 723–725, 741. [Google Scholar]
- Jadhav, S.M.; Munde, A.S.; Shankarwar, S.G.; Patharkar, V.R.; Shelke, V.A.; Chondhekar, T.K. Synthesis, Potentiometric, Spectral Characterization and Microbial Studies of Transition Metal Complexes with Tridentate Ligand. J. Korean Chem. Soc. 2010, 54, 515–522. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
| Compound | Molar Weight (g/mol) | Cal. (Exp., %) | Molar Cond. (Ω−1·cm2·mol−1) | Colour | Yield (%) | |||
|---|---|---|---|---|---|---|---|---|
| C | H | N | S | |||||
| NBTCS | 324.5 | 48.07 | 2.77 | 8.63 | 19.72 | 15.6 | White | 40.83 |
| (48.11) | (2.94) | (8.75) | (19.89) | |||||
| [Nd(NBTCS)2(H2O)2]NO3 | 891 | 35.02 | 2.47 | 7.86 | 14.37 | 72 | Yellow | 32.24 |
| (34.99) | (2.36) | (7.60) | (14.20) | |||||
| [Tl(NBTCS)2]CH3COO | 912 | 36.84 | 2.30 | 6.14 | 14.04 | 88 | Brown | 34.97 |
| (36.40) | (2.80) | (6.07) | (13.96) | |||||
| Compound | λmax (nm) | ῦ (cm-1) | ε (L·mol-1·cm-1) | Assignment |
|---|---|---|---|---|
| NBTCS | 272, 282 | 29,239, 30,314 | 6925, 6860 | π–π* |
| 305 | 32,787 | 7363 | n–π* | |
| [Nd(NBTCS)2(H2O)2]NO3 | 230, 237, 250 | 24,725, 25,477, 26,875 | 8237,8333,8333 | π–π* |
| [Tl(NBTCS)2]CH3COO | 241, 248, 261 | 25,907, 26,659, 28,057 | 8333,8333,8333 | π–π* |
| Compound | νN–H (cm−1) | νO–H (cm−1) | νC–H (cm−1) | νC=N (cm−1) | νSO2 (cm−1) | νNO3 (ionic) (cm−1) | δC–H (cm−1) | δO–H (cm−1) | νM–O (cm−1) | νM–N (cm−1) |
|---|---|---|---|---|---|---|---|---|---|---|
| NBTCS | 3407 | - | - | 1545 1465 | 1154 | 953, 827, 750 | - | - | - | |
| [Nd(NBTCS)2(H2O)2]NO3 | - | 3399 | - | 1589 1410 | 1120 | 1349 | - | 778 | 621 | 392 |
| [Tl(NBTCS)2]CH3COO | - | - | 3142 | 1548 | 1128 | - | 845 | - | 420 |
| IZD Values (mm) | |||||
|---|---|---|---|---|---|
| Compounds | E. coli 6 | E. coli 13 | S. aureus | P. aeruginosa | Proteus species |
| NBTCS | 13 | 10 | 0 | 0 | 12 |
| [Nd(NBTCS)2(H2O)2]NO3 | 15 | 14 | 17 | 12 | 16 |
| [Tl(NBTCS)2]CH3COO | 12 | 12 | 15 | 0 | 12 |
| MIC Values (μg/mL) | |||||
|---|---|---|---|---|---|
| Compounds | E. coli 6 | E. coli 13 | S. aureus | P. aeruginosa | Proteus species |
| NBTCS | 3.16 | 4.57 | 0.00 | 0.00 | 3.39 |
| [Nd(NBTCS)2(H2O)2]NO3 | 1.78 | 2.19 | 1.45 | 3.31 | 1.26 |
| [Tl(NBTCS)2]CH3COO | 3.47 | 3.47 | 1.26 | 0.00 | 3.47 |
| Ciprofloxacin | 20.00 | 20.00 | 0.63 | 10.00 | 0.16 |
| Gentamicin | 100.00 | 100.00 | 2.50 | 20.00 | 5.00 |
| Co-trimoxazole | 100.00 | 100.00 | 50.00 | 170.00 | 30.00 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Obasi, L.N.; Oruma, U.S.; Al-Swaidan, I.A.; Ramasami, P.; Ezeorah, C.J.; Ochonogor, A.E. Synthesis, Characterization and Antibacterial Studies of N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide and Its Neodymium(III) and Thallium(III) Complexes. Molecules 2017, 22, 153. https://doi.org/10.3390/molecules22020153
Obasi LN, Oruma US, Al-Swaidan IA, Ramasami P, Ezeorah CJ, Ochonogor AE. Synthesis, Characterization and Antibacterial Studies of N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide and Its Neodymium(III) and Thallium(III) Complexes. Molecules. 2017; 22(2):153. https://doi.org/10.3390/molecules22020153
Chicago/Turabian StyleObasi, Lawrence Nnamdi, Uchechukwu Susan Oruma, Ibrahim Abdulrazak Al-Swaidan, Ponnadurai Ramasami, Chigozie Julius Ezeorah, and Alfred Ezinna Ochonogor. 2017. "Synthesis, Characterization and Antibacterial Studies of N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide and Its Neodymium(III) and Thallium(III) Complexes" Molecules 22, no. 2: 153. https://doi.org/10.3390/molecules22020153
APA StyleObasi, L. N., Oruma, U. S., Al-Swaidan, I. A., Ramasami, P., Ezeorah, C. J., & Ochonogor, A. E. (2017). Synthesis, Characterization and Antibacterial Studies of N-(Benzothiazol-2-yl)-4-chlorobenzenesulphonamide and Its Neodymium(III) and Thallium(III) Complexes. Molecules, 22(2), 153. https://doi.org/10.3390/molecules22020153






