Abstract
Aminoglycosides are a group of antibiotics used since the 1940s to primarily treat a broad spectrum of bacterial infections. The primary resistance mechanism against these antibiotics is enzymatic modification by aminoglycoside-modifying enzymes that are divided into acetyl-transferases, phosphotransferases, and nucleotidyltransferases. To overcome this problem, new semisynthetic aminoglycosides were developed in the 70s. The most widely used semisynthetic aminoglycoside is amikacin, which is refractory to most aminoglycoside modifying enzymes. Amikacin was synthesized by acylation with the l-(−)-γ-amino-α-hydroxybutyryl side chain at the C-1 amino group of the deoxystreptamine moiety of kanamycin A. The main amikacin resistance mechanism found in the clinics is acetylation by the aminoglycoside 6′-N-acetyltransferase type Ib [AAC(6′)-Ib], an enzyme coded for by a gene found in integrons, transposons, plasmids, and chromosomes of Gram-negative bacteria. Numerous efforts are focused on finding strategies to neutralize the action of AAC(6′)-Ib and extend the useful life of amikacin. Small molecules as well as complexes ionophore-Zn+2 or Cu+2 were found to inhibit the acetylation reaction and induced phenotypic conversion to susceptibility in bacteria harboring the aac(6′)-Ib gene. A new semisynthetic aminoglycoside, plazomicin, is in advance stage of development and will contribute to renewed interest in this kind of antibiotics.
1. A Brief History of Aminoglycoside Antibiotics
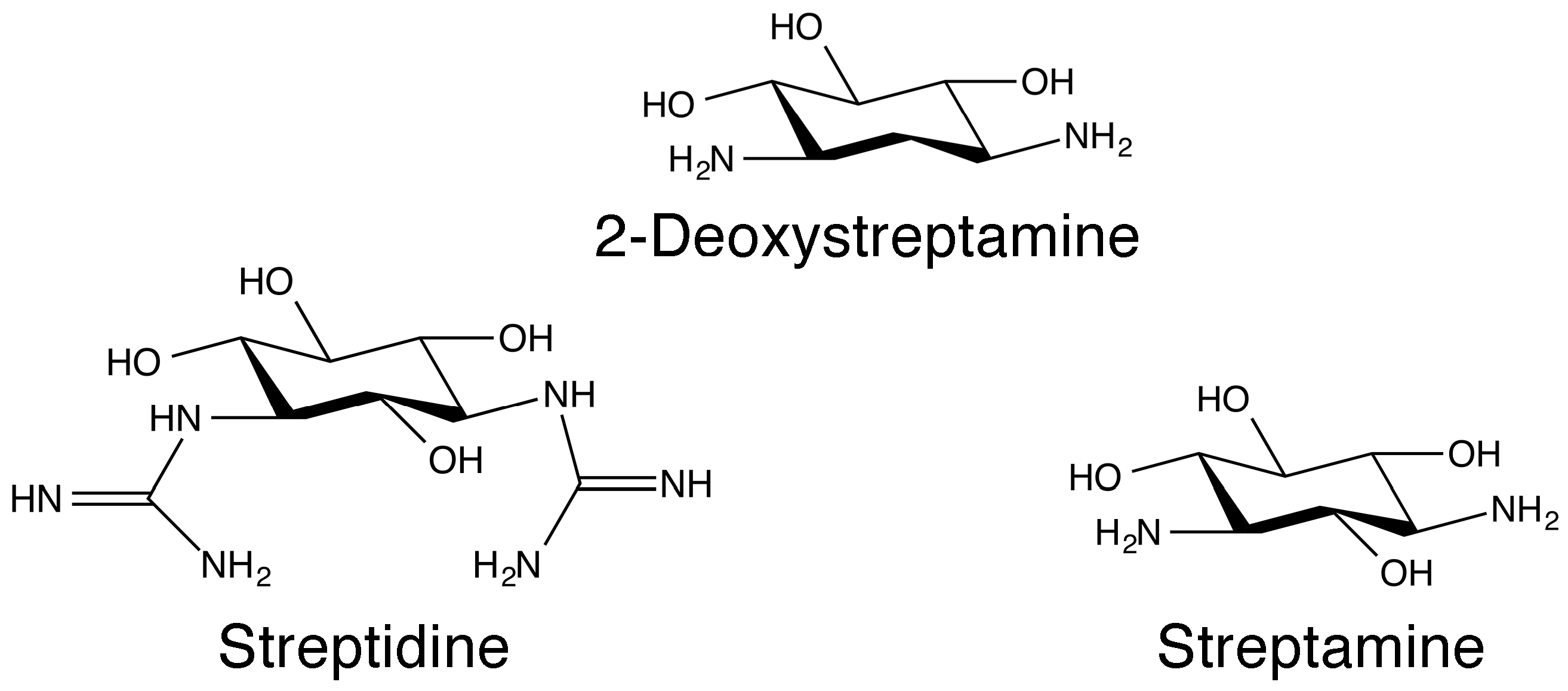
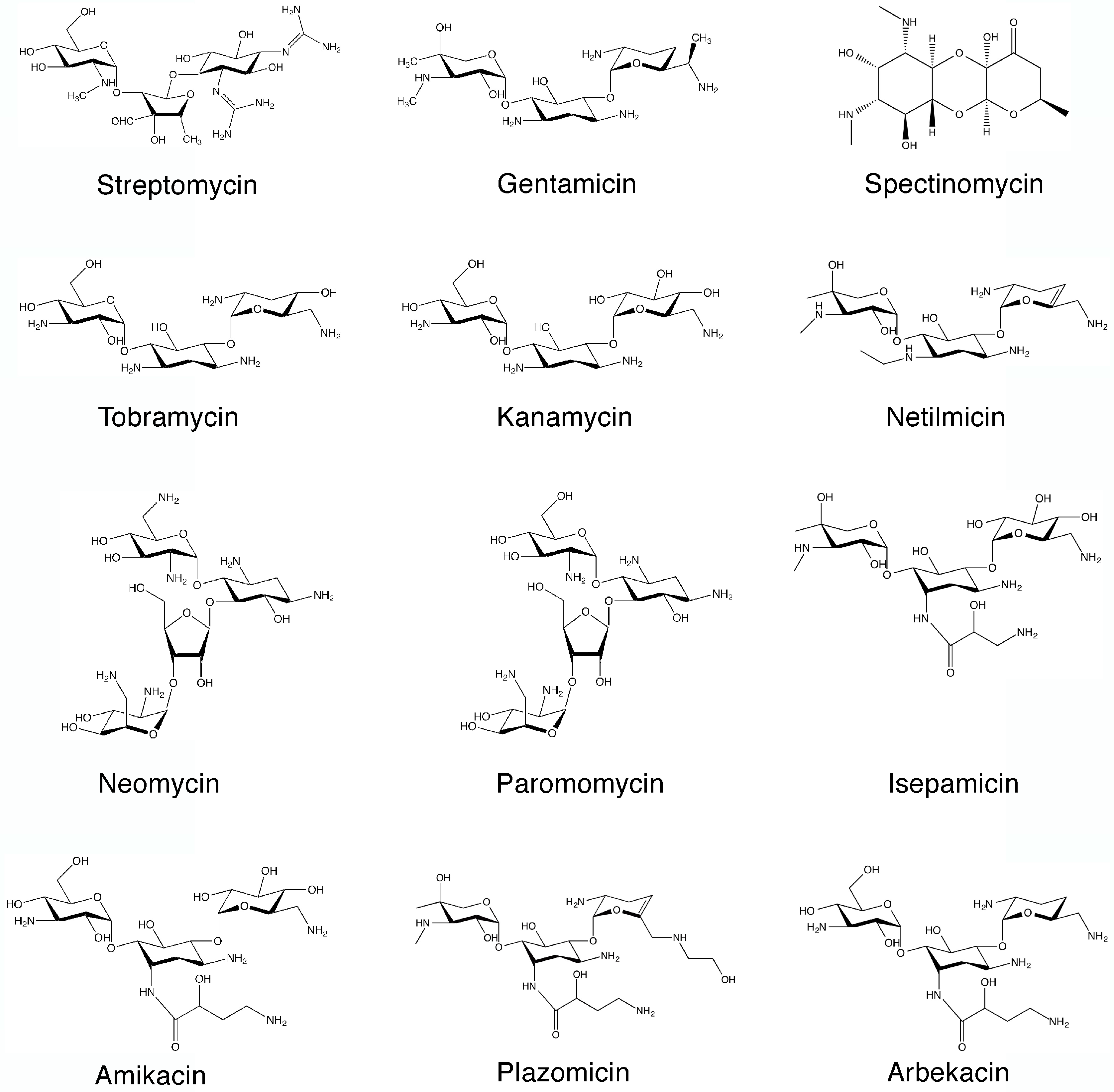
Aminoglycosides are a group of antibiotics primarily used to treat a wide spectrum of bacterial infections [1,2,3,4]. However, modern medicine has found other uses for these agents that include treatments for various genetic disorders and Meniere’s disease [5,6,7,8]. In addition, aminoglycosides are being researched as inhibitors of reproduction of the HIV [3,9]. The general structure of aminoglycosides consists of an aminocyclitol nucleus (streptamine, 2-deoxystreptamine, or streptidine) (Figure 1) linked to amino sugars. In addition, there are few exceptions where the antibiotic is considered an aminoglycoside despite not strictly conforming to this rule such as spectinomycin (Figure 2), which is an aminocyclitol not bound to amino sugars [10]. The first aminoglycoside, streptomycin (Figure 2), was discovered in the early days of the antibiotic era (1944) and it is still in use [11,12]. This discovery was followed by those of neomycin (1949) [13,14], and kanamycin (1957) [15] and gentamicin [16,17] (Figure 2). Following these findings, other natural aminoglycosides such as tobramycin [18] (Figure 2), with robust activity, were discovered. Furthermore, some of them were found to be useful as antifungal and antiparasitic agents [19,20,21,22]. For example, paromomycin (Figure 2), is used in the treatment of cryptosporidiosis, leishmaniasis, and other infections caused by protozoa and cestodes [20]. All natural aminoglycosides in use to date are produced by soil bacteria belonging to the genera Streptomyces or Micromonospora, and the origin of each one of them is identified by the suffix, “-mycin” and “-micin”, respectively [23,24].

Figure 1.
Chemical structures of aminocyclitols.
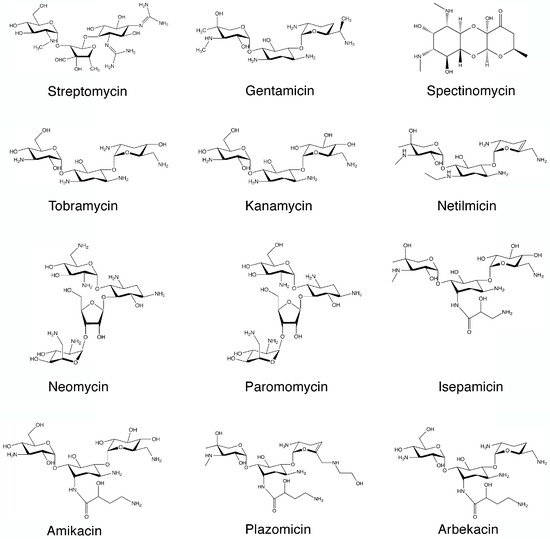
Figure 2.
Chemical structures of representative aminoglycosides.
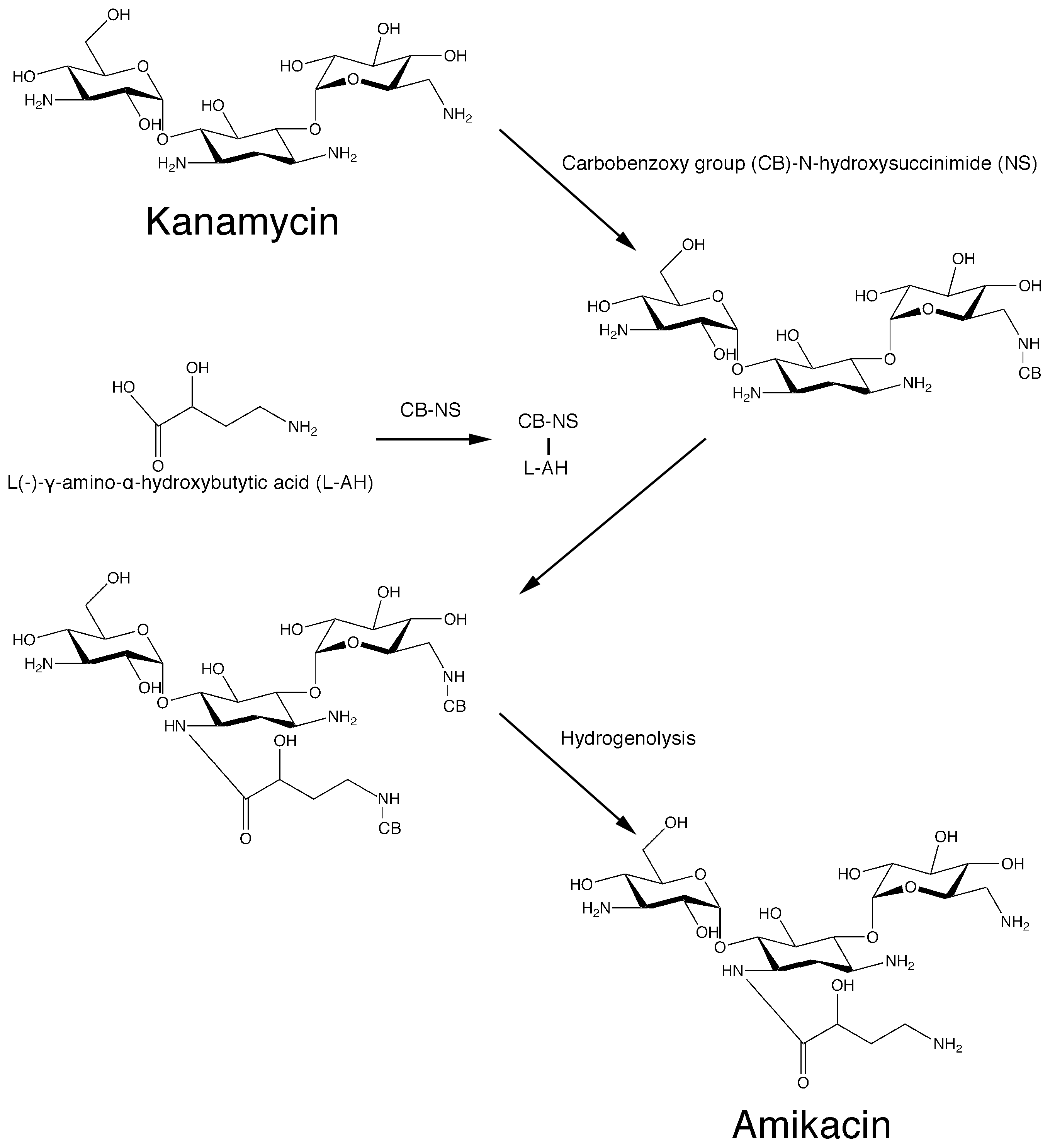
Recent advances have permitted us to understand the biosynthetic pathways of natural aminoglycosides [25]. Unfortunately, as it is the case with all antibiotics, bacteria developed several mechanisms of resistance that threaten the use of these drugs. Aminoglycoside modifying enzymes, which catalyze inactivation by acetylation (aminoglycoside acetyltransferases, AAC), phosphorylation (aminoglycoside phosphotransferases, APH), or adenylylation (aminoglycoside nucleotidyl-transferases, ANT) of the molecule, are the leading cause of the rapid increase and dissemination of resistance [26,27,28,29,30]. The first enzyme of this kind was identified in 1967 in an Escherichia coli strain that possessed an enzyme that could inactivate kanamycin by transferring an acetyl group to the 6′-N position of the antibiotic molecule. The resulting compound, 6′-N-acetylkanamycin, does not have antibiotic properties [31]. Aminoglycoside 6′-N-acetyltransferases belong to the GNAT (GCN5-related N-acetyltransferases) superfamily of enzymes, which includes more than 100,000 members found in prokaryotes, eukaryotes, and archaea [32,33], remain a very significant cause of failure of treatment of numerous severe infections [26,34]. More than hundred aminoglycoside modifying enzymes have been identified to date [26,27]. Among the multiple approaches tried to address the problem of resistance caused by aminoglycoside modifying enzymes, modification of the aminoglycoside molecule was among the most successful [35]. Addition of chemical groups that do not impair the antibiotic activity of the aminoglycoside produced new compounds that are not substrate of most aminoglycoside modifying enzymes [35]. These new molecules derived from natural aminoglycosides are known as semisynthetic. The first aminoglycoside of this kind to be used as an antibacterial was dibekacin, introduced in 1975 and still in use in various countries [36,37]. In the following years, numerous semisynthetic aminoglycosides were synthesized with activity against resistance caused by different aminoglycoside modifying enzymes. Amikacin, one of the most successful semisynthetic aminoglycosides, was synthesized by acylation with the l-(−)-γ-amino-α-hydroxybutyryl side chain at the C-1 amino group of the deoxystreptamine moiety of kanamycin A [38] (Scheme 1).
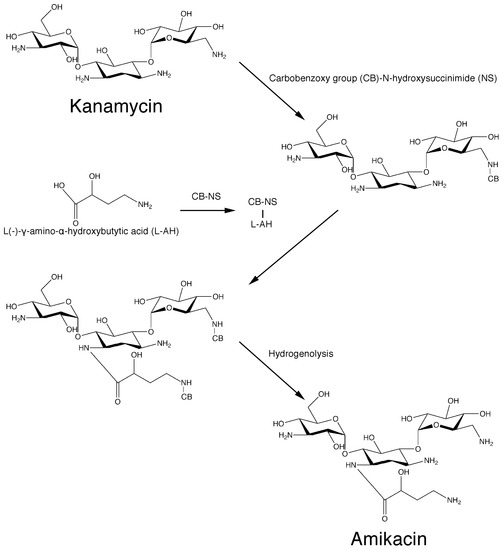
Scheme 1.
Original chemical reactions sequence to obtain amikacin by modification of kanamycin A.
This antibiotic was introduced in 1977, and it is still used with great success to treat a variety of infections although the rise of aminoglycoside 6′-N-acetyltransferases type I are limiting its effectiveness [26,34,39,40,41,42,43]. Other pathways for synthesis of amikacin by modification of kanamycin A were later proposed [44,45]. Netilmicin [46] (Figure 2), isepamicin [47] (Figure 2), and arbekacin [48] (Figure 2), introduced in 1985, 1988, and 1990, respectively, are other semisynthetic aminoglycosides that were successfully used to treat resistant infections. Following these developments, there was a period with relatively few additions to the field of aminoglycosides followed by another period characterized by new approaches that took advantage of the deeper understanding of different aspects of the biology and structure of aminoglycoside modifying enzymes as well as the advances in synthetic chemistry. As a consequence, numerous new generation aminoglycosides, also known as neoglycosides, started to be synthesized [19,49,50,51,52,53,54,55,56,57,58]. Of the several neoglycosides existing in the pipeline, plazomicin (ACHN-490) (Figure 2), which has been granted Breakthrough Therapy designation by the FDA in May 2017, is the one closest to be approved for human use [59,60,61,62]. A New Drug Application for plazomicin was submitted in October 2017 to the U.S. Food and Drug Administration [63]. This antibiotic is active against multidrug resistant Enterobacteriaceae, including problematic carbapenem- and polymyxin-resistant isolates as well as Acinetobacter baumannii [64,65,66,67]. The activity against Pseudomonas is similar to or slightly lower than other aminoglycosides [68]. Plazomicin is also active against methicillin-susceptible and methicillin-resistant Staphylococcus aureus [68,69].
Of the numerous aminoglycosides known to date, five (amikacin, gentamicin, neomycin, streptomycin, and tobramycin) are listed in the British National Formulary for clinical use in the United Kingdom [70] and amikacin, gentamicin, neomycin, streptomycin, kanamycin, paromomycin, and tobramycin are approved by the US Food and Drug Administration (FDA) for clinical use in the United States [19].
2. Mechanism of Action and Side Effects
Due to their polycationic nature, aminoglycosides first bind to the anionic compounds found in the bacterial surface. In the case of Gram-negative bacteria, these compounds are lipopolysaccharide, phospholipids, and outer membrane proteins and in the case of Gram-positives, they are mainly teichoic acids and phospholipids. These interactions produce an increase in permeability that results in penetration of some aminoglycoside molecules into the periplasmic space. This energy-independent mechanism is known as “self-promoted uptake” [71]. Following, in an energy-dependent process, the “energy-dependent phase I”, a small number of molecules of the antibiotic reach the cytoplasm with the participation of a functional electron transport system [72,73,74]. The aminoglycoside molecules inside the cytoplasm produce the antibiotic effect (see below), which results in mistranslated proteins. As a consequence, aberrant cytoplasmic membrane proteins induce damage to the integrity of the cytoplasmic membrane facilitating the entry of aminoglycoside molecules in abundant quantities. This third stage is known as “energy-dependent phase II” [75,76,77,78,79]. The high number of aminoglycoside molecules within the cell produces high levels of errors in protein synthesis leading to more damage in the cytoplasmic membrane permitting a still higher rate of uptake that ultimately results in death of the cell.
Aminoglycosides exert their action through binding to the 30S bacterial ribosome subunit changing the conformation of the A site to one that resembles that one induced by interaction between cognate tRNA and mRNA. As a consequence, proofreading capabilities of the ribosome are reduced, increasing mistranslation [1,76,80,81,82,83,84,85,86,87]. However, although the effect of binding to the ribosome is similar for all aminoglycosides, not all classes of these antimicrobials bind to identical sites of the 16S rRNA. Other effects that may or may not be secondary to RNA binding and protein mistranslation are inhibition of the 30S ribosomal subunit assembly (neomycin and paromomycin) [88,89], ribozyme-like activity resulting in cleavage of RNA molecules [90,91,92] or interference with essential functions dependent on ribozyme activity such inhibition of ribonuclease P [91,93,94].
Aminoglycosides have also been shown to cause other disruptions to bacterial cells when present at subinhibitory concentrations. Goh et al. showed that aminoglycosides at subinhibitory concentrations could modify transcription rates [95] and Possoz et al. found that amikacin at these low concentrations disrupts formation of the Z ring leading to inhibition of cell division [96].
While aminoglycosides were, and continue to be an essential component in the battery of resources to treat severe bacterial infections, their use is not free of side effects. The main toxicity risks are ototoxicity, nephrotoxicity, and rarely neuromuscular blockade [26,97,98,99,100,101,102]. The ototoxicity effects include permanent bilaterally severe, high-frequency sensorineural hearing loss and temporary vestibular hypofunction. The permanent hearing loss occurs as a result of damage caused to the sensory hair cells in the inner ear, in particular, the basal, high-frequency outer hair cells [103,104,105]. Efforts to limit ototoxic effects of aminoglycosides identified several candidates such as free radical scavengers as well as iron chelators [103], salicylate [106], N-acetylcysteine [107], and more recently d-tubocurarine and berbamine as potential otoprotectans [108]. Nephrotoxicity caused by aminoglycosides is usually reversible; its main clinical manifestation is nonoliguric acute kidney injury caused by decreased glomerular filtration [26,97,109,110]. Other manifestations include aminoaciduria, glycosuria, hypomagnesemia, hypocalcemia, and hypokalemia. There have been numerous studies to identify compounds that can prevent aminoglycoside nephrotoxicity. A recent systematic meta-analysis of available data recognized 40 chemicals with nephroprotectant activity [111]. Neuromuscular blockade is a rare aminoglycoside toxic effect [97].
3. Amikacin
Due to its property of being refractory to most aminoglycoside modifying enzymes, amikacin has been successfully used to treat otherwise aminoglycoside resistant infections, and it is the most widely used semisynthetic aminoglycoside [42,112,113,114,115,116,117,118]. Its pharmacokinetics is similar to that of the natural gentamicin and tobramycin, 30–60 min after intravenous administration there is a peak in serum concentration [97]. The optimal antibacterial effects occur when the maximum concentration in serum is 8 to 10 times higher than the minimal inhibitory concentration (MIC) [97]. Amikacin alone or in combination with other antibiotics is used to treat a variety of serious infections caused by aerobic Gram-negative bacteria, as well as mycobacteria and Nocardia [24,114,119,120,121,122,123,124]. This antibiotic is also essential in the treatment of life-threatening infections in neonates [42,115,125,126,127]. Structural studies showed that while amikacin binds the A site of the 16S RNA similarly when compared to kanamycin A, there are specific interactions between the l-(−)-γ-amino-α-hydroxybutyryl group and the RNA at the GC pairs C1404–G1497 and G1405–G1496 [128,129].
Amikacin is mainly administered intravenously, intramuscularly, through nebulization [130,131,132,133,134,135,136,137]. Other routes of administration for specific infections are intrathecal or intraventricular [138,139]. Amikacin is mostly administered as a weight-based dose divided in two to three applications per day or as a once-daily strategy, with this latter strategy being the preferred option [123,140,141,142,143]. Since amikacin exhibits the toxic effects common to aminoglycosides, i.e., ototoxicity and nephrotoxicity, the dose regime to maximize therapeutic outcomes and minimize adverse consequences is of great importance. However, a recent systematic study comparing the information available in the literature was inconclusive concerning optimal dosage regimes [144]. A recent review of the population pharmacokinetic models for amikacin described in critically ill patients contributed information to help optimizing amikacin dosage. In particular, the conclusions point against the “one dose fits all” strategy [114]. Amikacin is used to treat infections in neonates, including preterm neonates [42,125,145,146,147]. Although it has been successful in treating infections caused by multidrug resistant strains [126,148,149] there are still controversies about dosage and pharmacokinetics [126,146]. Unfortunately, the recent rise in resistance to amikacin limits the effectivity of many interventions during outbreaks of infection in neonates [41,42,150].
Amikacin was also researched as a formulation in unilamellar liposomes (MiKasome) [151]. However, in spite of early promising results in treatments of several conditions such as urinary tract infection [152], endocarditis [153], Klebsiella pneumoniae and Mycobacterium infections [154,155,156], the development of the formulation was discontinued in the year 2000.
Among the aminoglycosides currently available for use in humans, amikacin is the most resistant to the action of aminoglycoside modifying enzymes [27,157,158]. However, after it was introduced in the late 70s, resistant strains started to appear in different geographical regions and in some of them it became dangerously high [23,26,27,159,160]. A plasmid-mediated acetyltransferase, now known as AAC(6′)-Ib or AacA4 [26,27,34,161], was first reported in P. aeruginosa that conferred resistance to amikacin besides other aminoglycosides but not gentamicin C1 [160,162,163]. Early work also identified a plasmid-borne phosphotransferase and chromosomal mutations that resulted in resistance to amikacin in non-clinical E. coli strains [164,165,166,167], and a plasmid-mediated adenylyltransferase present in K. pneumoniae, E. coli, Serratia marcescens, and Proteus vulgaris strains that could use amikacin as substrate [168]. Amikacin resistance due to decreased uptake was also reported in K. pneumoniae [169]. The first documented outbreak of hospital infection with amikacin-resistant Enterobacteriaceae in newborn infants occurred in 1978 in the Louisville General Hospital, and three out of 11 neonates infected died [170]. Different Mycobacterium species developed resistance to amikacin through substitutions in the ribosomal RNA [171,172,173,174,175]. M. tuberculosis can also resist amikacin through enzymatic modification mediated by the enhanced intracellular survival (Eis) protein, an acetyltransferase enzyme with a unique structure and properties to acetylate aminoglycosides at multiple positions [29,176,177].
Despite the variety of mechanisms of resistance to amikacin detected, the main one found in the clinics is acetylation of the 6′-N position. The enzymes that act by this mechanism are called AAC(6′)-I followed by a unique identifier and usually confer resistance to aminoglycosides such as amikacin, tobramycin, and kanamycin but not the gentamicin complex [26,27]. However, exceptions have been detected that show an extended spectrum including gentamicin in their resistance profile [178] or, surprisingly, a reduced susceptibility to quinolones [179]. This family of enzymes includes over 50 representatives that are harbored by Gram-positive or Gram-negative bacteria [26]. These enzymes are also found as fusion proteins located adjacent to the N or C location of the accompanying protein [180], which can be an APH, an ANT, or another AAC [181,182,183,184,185,186,187]. Following we describe representative examples AAC(6′)-I enzymes highlighting some characteristics and their genetic environments. For comprehensive listing and description of AAC(6′)-I enzymes, the reader is referred to previous reviews [26,29,34].
3.1. AAC(6′)-I Enzymes of Gram-Positive Bacteria
A small number of 6′-N-acetyltransferases with the AAC(6′)-I profile were found in Gram-positive bacteria [184,188,189]. The AAC(6′)-Ie enzyme is fused to the N-terminal end of the phosphotransferase APH(2″)-Ia, forming a bifunctional enzyme coded for by the aac(6′)-Ie-aph(2″)-Ia fusion gene usually located within Tn4001-like transposons in Gram-positive bacteria [182,183,190,191,192,193,194]. These transposons have been found in plasmids as well as chromosomes of Gram-positive pathogens such as S. aureus, S. epidermidis, or Enterococcus faecalis [195,196,197,198]. These transposons are characterized by their ability to transpose to random location of Gram-positive chromosomes or plasmids and by the presence of the bifunctional aac(6′)-Ie-aph(2″)-Ia gene flanked by copies of IS256 and/or IS257 in their structure [184]. The crystal structure of the APH(2″)-Ia domain has been determined complexed to GTP analogs, guanosine diphosphate, and aminoglycosides [199,200,201].
The enzyme AAC(6′)-Ii was found in E. faecium, its gene is located in the chromosome and confers low levels of resistance, probably as a consequence of the low gene dose [188]. Structural and biochemical characterization of this enzymes permitted to determine that it exists as a homodimer showing subunit cooperativity and the mechanism follows an ordered bi-bi ternary complex with acetyl-CoA binding first [202,203,204].
3.2. AAC(6′)-I Enzymes of Gram-Negative Bacteria
In the case of Gram-negative bacteria, the number of AAC(6′)-I enzymes is large, and it is rapidly growing. There were comprehensive reviews that listed the known enzymes at the time they were written [26,27,29]. The latest enzymes of this kind to be reported are listed in Table 1, which continues where the listing in the review by Ramirez and Tolmasky left off [26]. However, in spite of the numerous AAC(6′)-I variants, AAC(6′)-Ib is the enzyme most often found in Gram-negative isolates from the Acinetobacter genus, and the Enterobacteriaceae, Pseudomonadaceae, and Vibrionaceae [1]. It should be noted that within these groups of bacteria are those Gram-negatives included in the ESKAPE, the bacteria responsible for the majority of antibiotic resistant hospital infections in the United States [205]. This enzyme is found in numerous variants, most of them differing at the N-terminus and some them presenting a few amino acid substitutions that result in enzymes with expanded substrate range. Examples of the later are the AAC(6′)-Ib11, which confers resistance to the gentamicin complex, or the AAC(6′)-Ib-cr, which confers a reduced quinolone susceptibility phenotype to the host [178,179,206]. For a detailed description and comparison of amino acid sequences of AAC(6′)-Ib variants, the reader is referred to a recent review [34].

Table 1.
Newer AAC(6′)-I proteins 1.
The aac(6′)-Ib gene has been found within integrons, transposons, genomic islands, plasmids, and chromosomes [34,39,40,207,208,209,210,211,212,213,214]. It is usually found as a functional, or in some instances deficient, gene cassette that can be located adjacent to the 5′-conserved region or between gene cassettes in the variable region of integrons [215,216,217]. While deficient gene cassettes cannot be mobilized between integrons through the action of the integrase, an alternative mechanism for mobilization of a deficient gene cassette including aac(6′)-Ib mediated by homologous recombination was proposed [218].
The earliest reports about aac(6′)-Ib identified the gene in plasmids from S. marcescens and K. pneumoniae [41,42,219,220,221]. In particular, the K. pneumoniae plasmid, named pJHCMW1, was exhaustively studied [208,222,223,224]. Its study led to the identification of Tn1331, a transposon that harbors four resistance genes, one of them being aac(6′)-Ib [40]. This transposon, as well as derivatives, was later found in numerous plasmids from Gram-negatives bacteria. A transposon named Tn1331.2, isolated from a K. pneumoniae plasmid has a perfect duplication of a 3047-bp DNA segment that includes three resistance genes: the aac(6′)-Ib, ant(3″)-Ia, and blaOXA-9 [39]. Tn6238 is a transposon nearly identical to Tn1331, but instead of the aac(6′)-Ib gene, it harbors a copy of the aac(6′)-Ib-cr variant, product of two point mutations [225]. Tn1332, identified in a multidrug-resistant P. putida strain, is identical to Tn1331 with the insertion of three DNA segments, one of which includes a copy of the blaVIM-2 gene [211]. Another derivative of Tn1331 with a copy of IS26 and a deletion that removed part of the ant(3″)-Ia, all the blaOXA-9, and part of the blaTEM-1 genes was first identified in a 15-kbp plasmid pAAC154 hosted by a carbapenem-resistant ST512 K. pneumoniae clinical strain isolated at the Hadassah Hospital, Jerusalem, Israel [226] and then in other plasmids [227,228]. Other derivatives with insertions in Tn1331 or a deleted version of it added resistance genes to the structure. Insertion of a Tn4401-like transposon added a blaKPC gene that “upgraded” the mobile element to make it able to confer resistance to carbapenem antibiotics [214,227,228,229,230]. In one instance, a copy of Tn1331 with an insertion of a Tn4401-like and an insertion of Tn5387, which includes the fluoroquinolone resistance qnrB19 gene, was identified in K. pneumoniae plasmid [207]. In other cases truncated versions of Tn1331 were also detected [231,232].
A recent report described the intra- and interspecies transfer of the aac(6′)-Ib-cr gene together with blaNDM-1 by secretion of outer membrane vesicles. A clinical A. baumannii strain released vesicles that were purified, and treated with DNase I and proteinase K, before incubation with another A. baumannii strain or E. coli JM109. Both recipient strains acquired the resistance genes showing that formation and secretion of outer membrane vesicles can be one more natural mechanism of dissemination of resistance genes including aac(6′)-Ib [233]. Transfer of plasmids and other cellular components by outer membrane vesicles has been observed before in Gram-negative bacteria [234,235].
3.3. Inhibition of Amikacin-Resistance Mediated by AAC(6′)-Ib
Since AAC(6′)-Ib is the major enzyme causing amikacin resistance in Gram-negative pathogens, it is expected that inhibition of its expression or activity would result in reversal of the resistant phenotype. Inhibition of expression of aac(6′)-Ib has been researched used antisense technologies, which are inspired by natural mechanisms of control of gene expression and DNA replication [236,237,238,239,240]. Several methodologies use different strategies to interfere with gene expression by supplying a short oligonucleotide or oligonucleotide analog that is complementary to a region of the target gene [241,242,243,244,245,246]. Reduction of levels of resistance to amikacin utilizing antisense oligodeoxynucleotides was first demonstrated targeting single-stranded regions in the mRNA that had been identified by RNase H mapping. Although the mechanisms of inhibition of gene expression remain to be confirmed, all evidence indicates that it occurred by eliciting RNase H-mediated degradation of the RNA moiety of the duplex oligodeoxynucleotide-mRNA [247]. Inhibition of resistance to amikacin in a clinical A. baumannii isolate harboring aac(6′)-Ib in its chromosome was achieved using an antisense hybrid oligomer consisting of 2′,4′-bridged nucleic acid-NC and deoxyribonucleotide residues conjugated to a permeabilizer peptide that could penetrate the bacterial cells and targeted the initiation of translation region [248]. Another approach that permitted to overcome amikacin resistance was what is known as External Guide Sequence (EGS) technology. It consists of designing antisense molecules that when interacting with the target mRNA acquire a structure that mimics that of a region of a pre-tRNA and elicits digestion by RNase P [244,249]. Gapmers including deoxyribonucleotide residues flanked by locked nucleic acids were potent inducers of RNase P digestion of the mRNA when forming the duplex at the complementary region, and as a consequence, a reduction of levels of antibiotic resistance was observed [250,251,252].
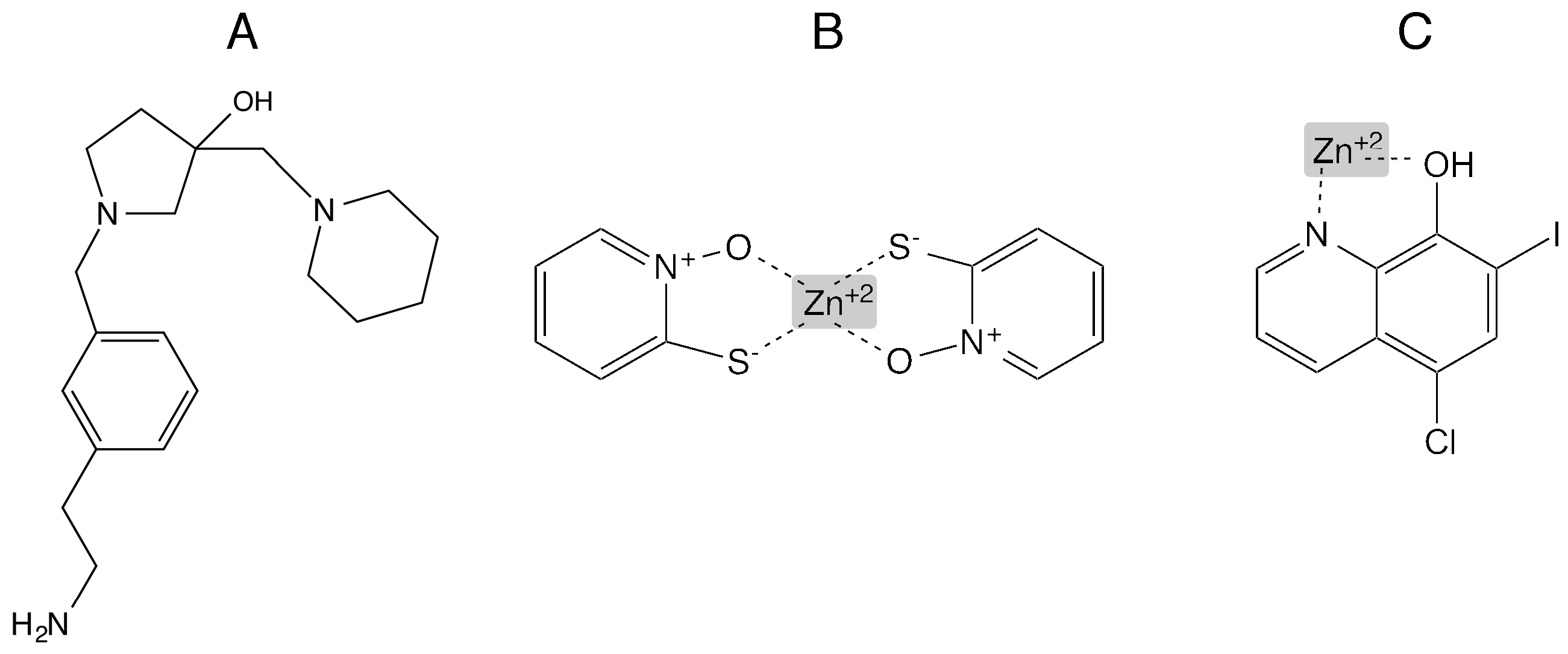
Numerous kinds of compounds with robust inhibitory activity of AAC(6′)-I enzymes have been described and are listed in various comprehensive reviews [26,49,54,253]. An inhibitor of AAC(6′)-Ib was first designed by an NMR-fragment based-approach [254]. Later, inhibitors of the same enzyme were identified using glide [255,256] and Autodock Vina 1.1.2 [257] computer docking programs. Compounds of different chemical nature but that behave as robust inhibitors of the enzymatic acetylation catalyzed by AAC(6′)-Ib. However, only one identified with the glide software, 1-[3-(2-aminoethyl)benzyl]-3-(piperidin-1-ylmethyl)pyrrolidin-3-ol (Figure 3) showed inhibitory activity of resistance to amikacin in cells growing in culture [258,259]. An acetyltransferase responsible for resistance to amikacin and other aminoglycosides present in resistant M. tuberculosis isolates attracted considerable interest in finding inhibitors of the enzymatic inactivation. These efforts resulted in isolation of various inhibitors that reduced the levels of amikacin resistance in growing cells [260,261,262,263]. Another group of compounds that were found to inhibit the acetylation reaction is integrated by Zn+2 and other metal ions [264,265,266]. Although the mechanism of this inhibition is still unknown, an attractive hypothesis is that it occurs through formation of a coordination complex between the substrate aminoglycoside and the cation that is no longer a suitable substrate of the enzyme. The concentrations of metal ions, Zn+2 or Cu+2, necessary for reversing resistance to amikacin in clinical and laboratory A. baumannii, K. pneumoniae, and E. coli strains in liquid cultures are in the low mM levels. However, when the metals are added in complex with some ionophores such as pyrithione or clioquinol, a small hydrophobic molecule also being investigated as a candidate drug to treat tumors and neurodegenerative diseases [267,268], low μM levels are sufficient for phenotypic conversion to susceptibility to amikacin [264,266,269].

Figure 3.
Chemical structures of representative inhibitors of AAC(6′)-Ib-mediated enzymatic acetylation of amikacin. (A) 1-[3-(2-aminoethyl)benzyl]-3-(piperidin-1-ylmethyl)pyrrolidin-3-ol; (B) zinc pyrithione coordination complex; (C) zinc clioquinol coordination complex [267].
4. Final Remarks
Aminoglycosides are one of the first kinds of antibiotics discovered dating back to the 1940s. They are an essential component of the armamentarium against serious infections caused by Gram-negative as well as Gram-positive bacteria; in this latter case, they are usually administered in combination with other antibiotics. Although the first representatives of this family were of natural origin, further research stimulated by the emergence of aminoglycoside modifying enzymes that confer resistance and disseminate very quickly, resulted in the design of a generation of semisynthetic members that are refractory to enzymatic inactivation. Amikacin, introduced in the late 1970s, was and continues to be an essential antibiotic used against numerous infections caused by multidrug-resistant organisms. Unfortunately, the AAC(6′)-I enzymes, and in particular the AAC(6′)-Ib, threaten to reduce the efficacy of amikacin. However, research efforts to design new semisynthetic molecules such as plazomicin or inhibitors of the expression or action of AAC(6′)-Ib, give hope that we will continue to count on aminoglycosides to fight severe multiresistant infections.
Acknowledgments
Authors’ work cited in this review article was funded by Public Health Service Grant 2R15AI047115 from the National Institutes of Health.
Author Contributions
Both authors contributed equally to writing this article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Vakulenko, S.B.; Mobashery, S. Versatility of aminoglycosides and prospects for their future. Clin. Microbiol. Rev. 2003, 16, 430–450. [Google Scholar] [CrossRef] [PubMed]
- Kotra, L.P.; Haddad, J.; Mobashery, S. Aminoglycosides: Perspectives on mechanisms of action and resistance and strategies to counter resistance. Antimicrob. Agents Chemother. 2000, 44, 3249–3256. [Google Scholar] [CrossRef] [PubMed]
- Houghton, J.L.; Green, K.D.; Chen, W.; Garneau-Tsodikova, S. The future of aminoglycosides: The end or renaissance? ChemBioChem 2010, 11, 880–902. [Google Scholar] [CrossRef] [PubMed]
- Serio, A.; Magalaes, M.; Blanchard, J.S.; Connolly, L. Aminoglycosides: Mechanisms of action and resistance. In Antimicrobial Drug Resistance; Mayers, D., Sobel, J., Ouellette, M., Kaye, K., Marchaim, D., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Pullens, B.; van Benthem, P.P. Intratympanic gentamicin for Meniere’s disease or syndrome. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef]
- Richardson, R.; Smart, M.; Tracey-White, D.; Webster, A.R.; Moosajee, M. Mechanism and evidence of nonsense suppression therapy for genetic eye disorders. Exp. Eye Res. 2017, 155, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Keeling, K.M.; Wang, D.; Conard, S.E.; Bedwell, D.M. Suppression of premature termination codons as a therapeutic approach. Crit. Rev. Biochem. Mol. Biol. 2012, 47, 444–463. [Google Scholar] [CrossRef] [PubMed]
- James, P.D.; Raut, S.; Rivard, G.E.; Poon, M.C.; Warner, M.; McKenna, S.; Leggo, J.; Lillicrap, D. Aminoglycoside suppression of nonsense mutations in severe hemophilia. Blood 2005, 106, 3043–3048. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, R.; Waldsich, C.; Wank, H. Modulation of RNA function by aminoglycoside antibiotics. EMBO J. 2000, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Veyssier, P.; Bryskier, A. Aminocyclitol aminoglycosides. In Antimicrobial Agents; Bryskier, A., Ed.; ASM Press: Washington, DC, USA, 2005; pp. 453–469. [Google Scholar]
- Davies, J.E. Aminoglycosides: Ancient and modern. J. Antibiot. 2006, 59, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.; Metzger, H.J.; Schatz, A.; Waksman, S.A. Control of gram-negative bacteria in experimental animals by streptomycin. Science 1944, 100, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Waksman, S.A.; Lechevalier, H.A. Neomycin, a new antibiotic active against streptomycin-resistant bacteria, including tuberculosis organisms. Science 1949, 109, 305–307. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, H.; Tazaki, T.; Okami, Y.; Fukuyama, S. Studies on streptothricin group substances. On streptothricin A and streptothricin B. J. Antibiot. 1949, 3, 232–235. [Google Scholar] [CrossRef]
- Umezawa, H. Kanamycin: Its discovery. Ann. N. Y. Acad. Sci. 1958, 76, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, M.J.; Luedemann, G.M.; Oden, E.M.; Wagman, G.H. Gentamicin, a new broad-spectrum antibiotic complex. Antimicrob. Agents Chemother. 1963, 161, 1–7. [Google Scholar] [PubMed]
- Weinstein, M.J.; Luedemann, G.M.; Oden, E.M.; Wagman, G.H.; Rosselet, J.P.; Marquez, J.A.; Coniglio, C.T.; Charney, W.; Herzog, H.L.; Black, J. Gentamicin, a new antibiotic complex from Micromonospora. J. Med. Chem. 1963, 6, 463–464. [Google Scholar] [CrossRef] [PubMed]
- Higgins, C.E.; Kastner, R.E. Nebramycin, a new broad-spectrum antibiotic complex. II. Description of Streptomyces tenebrarius. Antimicrob. Agents Chemother. 1967, 7, 324–331. [Google Scholar] [PubMed]
- Chandrika, N.T.; Garneau-Tsodikova, S. A review of patents (2011–2015) towards combating resistance to and toxicity of aminoglycosides. MedChemComm 2016, 7, 50–68. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.N.; den Boer, M.; Ritmeijer, K. Paromomycin. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Fosso, M.; AlFindee, M.N.; Zhang, Q.; Nziko Vde, P.; Kawasaki, Y.; Shrestha, S.K.; Bearss, J.; Gregory, R.; Takemoto, J.Y.; Chang, C.W. Structure-activity relationships for antibacterial to antifungal conversion of kanamycin to amphiphilic analogues. J. Org. Chem. 2015, 80, 4398–4411. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.K.; Fosso, M.Y.; Green, K.D.; Garneau-Tsodikova, S. Amphiphilic tobramycin analogues as antibacterial and antifungal agents. Antimicrob. Agents Chemother. 2015, 59, 4861–4869. [Google Scholar] [CrossRef] [PubMed]
- Tolmasky, M.E. Aminoglycoside-modifying enzymes: Characteristics, localization, and dissemination. In Enzyme-Mediated Resistance to Antibiotics: Mechanisms, Dissemination, and Prospects for Inhibition; Bonomo, R.A., Tolmasky, M.E., Eds.; ASM Press: Washington, DC, USA, 2007; pp. 35–52. [Google Scholar]
- Yao, J.; Moellering, R. Antibacterial agents. In Manual of Clinical Microbiology; Murray, P., Baron, E., Jorgensen, J., Landry, M., Pfaller, M., Eds.; American Society for Microbiology Press: Washington, DC, USA, 2007; Volume 1, pp. 1077–1113. [Google Scholar]
- Park, S.R.; Park, J.W.; Ban, Y.H.; Sohng, J.K.; Yoon, Y.J. 2-Deoxystreptamine-containing aminoglycoside antibiotics: Recent advances in the characterization and manipulation of their biosynthetic pathways. Nat. Prod. Rep. 2013, 30, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.S.; Tolmasky, M.E. Aminoglycoside modifying enzymes. Drug Resist. Updat. 2010, 13, 151–171. [Google Scholar] [CrossRef] [PubMed]
- Shaw, K.J.; Rather, P.N.; Hare, R.S.; Miller, G.H. Molecular genetics of aminoglycoside resistance genes and familial relationships of the aminoglycoside-modifying enzymes. Microbiol. Rev. 1993, 57, 138–163. [Google Scholar] [PubMed]
- Umezawa, H.; Kondo, S. Mechanisms of resistance to amino-glycoside antibiotics. In Aminoglycoside Antibiotics; Umezawa, H., Hooper, H., Eds.; Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1982; Volume 62, pp. 267–292. [Google Scholar]
- Garneau-Tsodikova, S.; Labby, K.J. Mechanisms of resistance to aminoglycoside antibiotics: Overview and perspectives. MedChemComm 2016, 7, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Nishino, K.; Roberts, M.C.; Tolmasky, M.; Aminov, R.I.; Zhang, L. Mechanisms of antibiotic resistance. Front. Microbiol. 2015, 6, 34. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, H.; Okanishi, M.; Utahara, R.; Maeda, K.; Kondo, S. Isolation and structure of kanamycin inactivated by a cell free system of kanamycin-resistant E. coli. J. Antibiot. 1967, 20, 136–141. [Google Scholar] [PubMed]
- Favrot, L.; Blanchard, J.S.; Vergnolle, O. Bacterial GCN5-related N-acetyltransferases: From resistance to regulation. Biochemistry 2016, 55, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Salah Ud-Din, A.I.; Tikhomirova, A.; Roujeinikova, A. Structure and functional diversity of GCN5-related N-acetyltransferases (GNAT). Int. J. Mol. Sci. 2016, 17, 1018. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.S.; Nikolaidis, N.; Tolmasky, M.E. Rise and dissemination of aminoglycoside resistance: The aac(6′)-Ib paradigm. Front. Microbiol. 2013, 4, 121. [Google Scholar] [CrossRef] [PubMed]
- Kondo, S.; Hotta, K. Semisynthetic aminoglycoside antibiotics: Development and enzymatic modifications. J. Infect. Chemother. 1999, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Oizumi, K.; Ariji, F.; Kumano, N.; Oka, S.; Konno, K. Action mechanism of 3′,4′-dideoxykanamycin B (DKB) on Klebsiella pneumoniae. Sci. Rep. Res. Inst. Tohoku Univ. Med. 1974, 21, 47–53. [Google Scholar] [PubMed]
- Umezawa, H.; Umezawa, S.; Tsuchiya, T.; Okazaki, Y. 3′,4′-dideoxy-kanamycin B active against kanamycin-resistant Escherichia coli and Pseudomonas aeruginosa. J. Antibiot. 1971, 24, 485–487. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, H.; Naito, T.; Nakagawa, S.; Fujisawa, K.I. BB-K8, a new semisynthetic aminoglycoside antibiotic. J. Antibiot. 1972, 25, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Tolmasky, M.E.; Chamorro, R.M.; Crosa, J.H.; Marini, P.M. Transposon-mediated amikacin resistance in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 1988, 32, 1416–1420. [Google Scholar] [CrossRef] [PubMed]
- Tolmasky, M.E.; Crosa, J.H. Tn1331, a novel multiresistance transposon encoding resistance to amikacin and ampicillin in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 1987, 31, 1955–1960. [Google Scholar] [CrossRef] [PubMed]
- Tolmasky, M.E.; Roberts, M.; Woloj, M.; Crosa, J.H. Molecular cloning of amikacin resistance determinants from a Klebsiella pneumoniae plasmid. Antimicrob. Agents Chemother. 1986, 30, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Woloj, M.; Tolmasky, M.E.; Roberts, M.C.; Crosa, J.H. Plasmid-encoded amikacin resistance in multiresistant strains of Klebsiella pneumoniae isolated from neonates with meningitis. Antimicrob. Agents Chemother. 1986, 29, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Tolmasky, M.E. Bacterial resistance to aminoglycosides and β-lactams: The Tn1331 transposon paradigm. Front. Biosci. 2000, 5, D20–D29. [Google Scholar] [CrossRef] [PubMed]
- Mangia, A.; Giobbio, V.; Ornato, G. Novel Process for the Synthesis of Amikacin. U.S. Patent 4,902,790, 20 February 1990. [Google Scholar]
- Hanessian, S.; Patil, G. Aminoglycoside antibiotics—A method for selective N-acylation based on the temporary protection of amino alcohol functions as copper chelates. Tetrahedron Lett. 1978, 19, 1035–1038. [Google Scholar] [CrossRef]
- Kahlmeter, G. Netilmicin: Clinical pharmacokinetics and aspects on dosage schedules. An overview. Scand. J. Infect. Dis. 1980, 12 (Suppl. 23), 74–81. [Google Scholar]
- Nagabhushan, T.L.; Cooper, A.B.; Tsai, H.; Daniels, P.J.; Miller, G.H. The syntheses and biological properties of 1-N-(S-4-amino-2-hydroxybutyryl)-gentamicin B and 1-N-(S-3-amino-2-hydroxypropionyl)-gentamicin B. J. Antibiot. 1978, 31, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Kondo, S. Development of arbekacin and synthesis of new derivatives stable to enzymatic modifications by methicillin-resistant Staphylococcus aureus. Jpn. J. Antibiot. 1994, 47, 561–574. [Google Scholar] [PubMed]
- Labby, K.J.; Garneau-Tsodikova, S. Strategies to overcome the action of aminoglycoside-modifying enzymes for treating resistant bacterial infections. Future Med. Chem. 2013, 5, 1285–1309. [Google Scholar] [CrossRef] [PubMed]
- Park, J.W.; Ban, Y.H.; Nam, S.J.; Cha, S.S.; Yoon, Y.J. Biosynthetic pathways of aminoglycosides and their engineering. Curr. Opin. Biotechnol. 2017, 48, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Chandrika, N.T.; Green, K.; Houghton, J.L.; Garneau-Tsodikova, S. Synthesis and biological activity of mono- and di-N-acylated aminoglycosides. ACS Med. Chem. Lett. 2015, 6, 1134–1139. [Google Scholar] [CrossRef] [PubMed]
- Jackson, J.; Chen, C.; Buising, K. Aminoglycosides: How should we use them in the 21st century? Curr. Opin. Infect. Dis. 2013, 26, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Dozzo, P.; Moser, H.E. New aminoglycoside antibiotics. Expert Opin. Ther. Pat. 2010, 20, 1321–1341. [Google Scholar] [CrossRef] [PubMed]
- Vong, K.; Auclair, K. Understanding and overcoming aminoglycoside resistance caused by N-6′-acetyltransferase. MedChemComm 2012, 3, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Ye, X.S. Development of aminoglycoside antibiotics effective against resistant bacterial strains. Curr. Top. Med. Chem. 2010, 10, 1898–1926. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, L.; Das, I.; Desire, J.; Sautrey, G.; Barros, R.S.V.; El Khoury, M.; Mingeot-Leclercq, M.P.; Decout, J.L. New broad-spectrum antibacterial amphiphilic aminoglycosides active against resistant bacteria: From neamine derivatives to smaller neosamine analogues. J. Med. Chem. 2016, 59, 9350–9369. [Google Scholar] [CrossRef] [PubMed]
- Fair, R.J.; McCoy, L.S.; Hensler, M.E.; Aguilar, B.; Nizet, V.; Tor, Y. Singly modified amikacin and tobramycin derivatives show increased rRNA A-site binding and higher potency against resistant bacteria. ChemMedChem 2014, 9, 2164–2171. [Google Scholar] [CrossRef] [PubMed]
- You, X.F.; Li, C.R.; Yang, X.Y.; Yuan, M.; Zhang, W.X.; Lou, R.H.; Wang, Y.M.; Li, G.Q.; Chen, H.Z.; Song, D.Q.; et al. In Vivo antibacterial activity of vertilmicin, a new aminoglycoside antibiotic. Antimicrob. Agents Chemother. 2009, 53, 4525–4528. [Google Scholar] [CrossRef] [PubMed]
- Aggen, J.B.; Armstrong, E.S.; Goldblum, A.A.; Dozzo, P.; Linsell, M.S.; Gliedt, M.J.; Hildebrandt, D.J.; Feeney, L.A.; Kubo, A.; Matias, R.D.; et al. Synthesis and spectrum of the neoglycoside ACHN-490. Antimicrob. Agents Chemother. 2010, 54, 4636–4642. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, E.S.; Miller, G.H. Combating evolution with intelligent design: The neoglycoside ACHN-490. Curr. Opin. Microbiol. 2010, 13, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Avial, I.; Pena, I.; Picazo, J.J.; Rodriguez-Avial, C.; Culebras, E. In Vitro activity of the next-generation aminoglycoside plazomicin alone and in combination with colistin, meropenem, fosfomycin or tigecycline against carbapenemase-producing Enterobacteriaceae strains. Int. J. Antimicrob. Agents 2015, 46, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Wright, H.; Bonomo, R.A.; Paterson, D.L. New agents for the treatment of infections with Gram-negative bacteria: Restoring the miracle or false dawn? Clin. Microbiol. Infect. 2017, 23, 704–712. [Google Scholar] [CrossRef] [PubMed]
- Arrington, D. Achaogen submits Plazomicin New Drug Application (NDA) to the U.S. FDA for Treatment of Complicated Urinary Tract Infections and Bloodstream Infections. Available online: http://investors.achaogen.com/releasedetail.cfm?releaseid=1045559 (accessed on 2 November 2017).
- Galani, I.; Souli, M.; Daikos, G.L.; Chrysouli, Z.; Poulakou, G.; Psichogiou, M.; Panagea, T.; Argyropoulou, A.; Stefanou, I.; Plakias, G.; et al. Activity of plazomicin (ACHN-490) against MDR clinical isolates of Klebsiella pneumoniae, Escherichia coli, and Enterobacter spp. from Athens, Greece. J. Chemother. 2012, 24, 191–194. [Google Scholar] [CrossRef] [PubMed]
- Denervaud-Tendon, V.; Poirel, L.; Connolly, L.E.; Krause, K.M.; Nordmann, P. Plazomicin activity against polymyxin-resistant Enterobacteriaceae, including MCR-1-producing isolates. J. Antimicrob. Chemother. 2017, 72, 2787–2791. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kashikar, A.; Bush, K. In Vitro activity of plazomicin against β-lactamase-producing carbapenem-resistant Enterobacteriaceae (CRE). J. Antimicrob. Chemother. 2017, 72, 2792–2795. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Salguero, C.; Rodriguez-Avial, I.; Picazo, J.J.; Culebras, E. Can plazomicin alone or in combination be a therapeutic option against carbapenem-resistant Acinetobacter baumannii? Antimicrob. Agents Chemother. 2015, 59, 5959–5966. [Google Scholar] [CrossRef] [PubMed]
- Walkty, A.; Adam, H.; Baxter, M.; Denisuik, A.; Lagace-Wiens, P.; Karlowsky, J.A.; Hoban, D.J.; Zhanel, G.G. In Vitro activity of plazomicin against 5015 gram-negative and gram-positive clinical isolates obtained from patients in canadian hospitals as part of the CANWARD study, 2011–2012. Antimicrob. Agents Chemother. 2014, 58, 2554–2563. [Google Scholar] [CrossRef] [PubMed]
- Lopez Diaz, M.C.; Rios, E.; Rodriguez-Avial, I.; Simaluiza, R.J.; Picazo, J.J.; Culebras, E. In-Vitro activity of several antimicrobial agents against methicillin-resistant Staphylococcus aureus (MRSA) isolates expressing aminoglycoside-modifying enzymes: Potency of plazomicin alone and in combination with other agents. Int. J. Antimicrob. Agents 2017, 50, 191–196. [Google Scholar] [CrossRef] [PubMed]
- The Royal Pharmaceutical Society of Great Britain, British National Formulary; BMJ Group, RCPCH Publications Ltd.: London, UK; The Royal Pharmaceutical Society of Great Britain: London, UK, 2017.
- Vanhoof, R.; Sonck, P.; Hannecart-Pokorni, E. The role of lipopolysaccharide anionic binding sites in aminoglycoside uptake in Stenotrophomonas (Xanthomonas) maltophilia. J. Antimicrob. Chemother. 1995, 35, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Muir, M.E.; van Heeswyck, R.S.; Wallace, B.J. Effect of growth rate on streptomycin accumulation by Escherichia coli and Bacillus megaterium. J. Gen. Microbiol. 1984, 130, 2015–2022. [Google Scholar] [CrossRef] [PubMed]
- Nichols, W.W.; Young, S.N. Respiration-dependent uptake of dihydrostreptomycin by Escherichia coli. Its irreversible nature and lack of evidence for a uniport process. Biochem. J. 1985, 228, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Bryan, L.; van der Elzen, H. Effects of membrane-energy mutations and cations on streptomycin and gentamicin accumulation by bacteria: A model for entry of streptomycin and gentamicin in susceptible and resistant bacteria. Antimicrob. Agents Chemother. 1977, 12, 163–177. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, C.; Braun, C.B.; Rosano, C.L. Role of ribosome recycling in uptake of dihydrostreptomycin by sensitive and resistant Escherichia coli. Biochim. Biophys. Acta 1981, 652, 168–176. [Google Scholar] [CrossRef]
- Davis, B.D. Non-specific membrane permeability and aminoglycoside action. J. Antimicrob. Chemother. 1989, 24, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Nichols, W.W. The enigma of streptomycin transport. J. Antimicrob. Chemother. 1989, 23, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Nichols, W.W.; Dorrington, S.M.; Slack, M.P.; Walmsley, H.L. Inhibition of tobramycin diffusion by binding to alginate. Antimicrob. Agents Chemother. 1988, 32, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Taber, H.W.; Mueller, J.P.; Miller, P.F.; Arrow, A.S. Bacterial uptake of aminoglycoside antibiotics. Microbiol. Rev. 1987, 51, 439–457. [Google Scholar] [PubMed]
- Bakker, E.P. Aminoglycoside and aminocyclitol antibiotics: Hygromycin B is an atypical bactericidal compound that exerts effects on cells of Escherichia coli characteristics for bacteriostatic aminocyclitols. J. Gen. Microbiol. 1992, 138, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Busse, H.J.; Wostmann, C.; Bakker, E.P. The bactericidal action of streptomycin: Membrane permeabilization caused by the insertion of mistranslated proteins into the cytoplasmic membrane of Escherichia coli and subsequent caging of the antibiotic inside the cells due to degradation of these proteins. J. Gen. Microbiol. 1992, 138, 551–561. [Google Scholar] [PubMed]
- Davis, B.D. Mechanism of bactericidal action of aminoglycosides. Microbiol. Rev. 1987, 51, 341–350. [Google Scholar] [PubMed]
- Magnet, S.; Blanchard, J.S. Molecular insights into aminoglycoside action and resistance. Chem. Rev. 2005, 105, 477–498. [Google Scholar] [CrossRef] [PubMed]
- Degtyareva, N.N.; Gong, C.; Story, S.; Levinson, N.S.; Oyelere, A.K.; Green, K.D.; Garneau-Tsodikova, S.; Arya, D.P. Antimicrobial activity, AME resistance, and A-site binding studies of anthraquinone-neomycin conjugates. ACS Infect. Dis. 2017, 3, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.; Deb, J.K. Molecular targets for design of novel inhibitors to circumvent aminoglycoside resistance. Curr. Drug Targets 2005, 6, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Jana, S.; Deb, J.K. Molecular understanding of aminoglycoside action and resistance. Appl. Microbiol. Biotechnol. 2006, 70, 140–150. [Google Scholar] [CrossRef] [PubMed]
- McCoy, L.S.; Xie, Y.; Tor, Y. Antibiotics that target protein synthesis. Wiley Interdiscip. Rev. RNA 2011, 2, 209–232. [Google Scholar] [CrossRef] [PubMed]
- Foster, C.; Champney, W.S. Characterization of a 30S ribosomal subunit assembly intermediate found in Escherichia coli cells growing with neomycin or paromomycin. Arch. Microbiol. 2008, 189, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.; Champney, W.S. Neomycin and paromomycin inhibit 30S ribosomal subunit assembly in Staphylococcus aureus. Curr. Microbiol. 2003, 47, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Belousoff, M.J.; Graham, B.; Spiccia, L.; Tor, Y. Cleavage of RNA oligonucleotides by aminoglycosides. Org. Biomol. Chem. 2009, 7, 30–33. [Google Scholar] [CrossRef] [PubMed]
- Vourekas, A.; Stamatopoulou, V.; Toumpeki, C.; Tsitlaidou, M.; Drainas, D. Insights into functional modulation of catalytic RNA activity. IUBMB Life 2008, 60, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Herrin, D.L. Mg2+ mimicry in the promotion of group I ribozyme activities by aminoglycoside antibiotics. Biochem. Biophys. Res. Commun. 2006, 344, 1246–1252. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, N.E.; Brannvall, M.; Virtanen, A.; Kirsebom, L.A. Inhibition of RNase P RNA cleavage by aminoglycosides. Proc. Natl. Acad. Sci. USA 1999, 96, 6155–6160. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, S.A.; Sudhahar, C.G.; Hatfield, C.L.; Sun, J.; Behrman, E.J.; Gopalan, V. Studies on the mechanism of inhibition of bacterial ribonuclease P by aminoglycoside derivatives. Nucleic Acids Res. 2008, 36, 697–704. [Google Scholar] [CrossRef] [PubMed]
- Goh, E.B.; Yim, G.; Tsui, W.; McClure, J.; Surette, M.G.; Davies, J. Transcriptional modulation of bacterial gene expression by subinhibitory concentrations of antibiotics. Proc. Natl. Acad. Sci. USA 2002, 99, 17025–17030. [Google Scholar] [CrossRef] [PubMed]
- Possoz, C.; Newmark, J.; Sorto, N.; Sherratt, D.J.; Tolmasky, M.E. Sublethal concentrations of the aminoglycoside amikacin interfere with cell division without affecting chromosome dynamics. Antimicrob. Agents Chemother. 2007, 51, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Radigan, E.A.; Gilchrist, N.A.; Miller, M.A. Management of aminoglycosides in the intensive care unit. J. Intensive Care Med. 2010, 25, 327–342. [Google Scholar] [CrossRef] [PubMed]
- Paradelis, A.G.; Triantaphyllidis, C.; Giala, M.M. Neuromuscular blocking activity of aminoglycoside antibiotics. Methods Find. Exp. Clin. Pharmacol. 1980, 2, 45–51. [Google Scholar] [PubMed]
- Singh, Y.N.; Marshall, I.G.; Harvey, A.L. Some effects of the aminoglycoside antibiotic amikacin on neuromuscular and autonomic transmission. Br. J. Anaesth. 1978, 50, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Wargo, K.A.; Edwards, J.D. Aminoglycoside-induced nephrotoxicity. J. Pharm. Pract. 2014, 27, 573–577. [Google Scholar] [CrossRef] [PubMed]
- Leis, J.A.; Rutka, J.A.; Gold, W.L. Aminoglycoside-induced ototoxicity. CMAJ 2015, 187, E52. [Google Scholar] [CrossRef] [PubMed]
- Prayle, A.; Watson, A.; Fortnum, H.; Smyth, A. Side effects of aminoglycosides on the kidney, ear and balance in cystic fibrosis. Thorax 2010, 65, 654–658. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, T.; Teranishi, M.; Hibi, T.; Kobayashi, M.; Umemura, M. Vestibular and cochlear toxicity of aminoglycosides—A review. Acta Otolaryngol. 2000, 120, 904–911. [Google Scholar] [PubMed]
- Lanvers-Kaminsky, C.; Zehnhoff-Dinnesen, A.A.; Parfitt, R.; Ciarimboli, G. Drug-induced ototoxicity: Mechanisms, pharmacogenetics, and protective strategies. Clin. Pharmacol. Ther. 2017, 101, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Guthrie, O.W. Aminoglycoside induced ototoxicity. Toxicology 2008, 249, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Sha, S.H.; Qiu, J.H.; Schacht, J. Aspirin to prevent gentamicin-induced hearing loss. N. Engl. J. Med. 2006, 354, 1856–1857. [Google Scholar] [CrossRef] [PubMed]
- Feldman, L.; Efrati, S.; Eviatar, E.; Abramsohn, R.; Yarovoy, I.; Gersch, E.; Averbukh, Z.; Weissgarten, J. Gentamicin-induced ototoxicity in hemodialysis patients is ameliorated by N-acetylcysteine. Kidney Int. 2007, 72, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, N.K.; O’Reilly, M.; Derudas, M.; Kenyon, E.J.; Huckvale, R.; van Netten, S.M.; Ward, S.E.; Richardson, G.P.; Kros, C.J. d-Tubocurarine and berbamine: Alkaloids that are permeant blockers of the hair cell’s mechano-electrical transducer channel and protect from aminoglycoside toxicity. Front. Cell. Neurosci. 2017, 11, 262. [Google Scholar] [CrossRef] [PubMed]
- Mingeot-Leclercq, M.P.; Tulkens, P.M. Aminoglycosides: Nephrotoxicity. Antimicrob. Agents Chemother. 1999, 43, 1003–1012. [Google Scholar] [PubMed]
- McWilliam, S.J.; Antoine, D.J.; Smyth, R.L.; Pirmohamed, M. Aminoglycoside-induced nephrotoxicity in children. Pediatr. Nephrol. 2017, 32, 2015–2025. [Google Scholar] [CrossRef] [PubMed]
- Vicente-Vicente, L.; Casanova, A.G.; Hernandez-Sanchez, M.T.; Pescador, M.; Lopez-Hernandez, F.J.; Morales, A.I. A systematic meta-analysis on the efficacy of pre-clinically tested nephroprotectants at preventing aminoglycoside nephrotoxicity. Toxicology 2017, 377, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Gerding, D.N.; Larson, T.A.; Hughes, R.A.; Weiler, M.; Shanholtzer, C.; Peterson, L.R. Aminoglycoside resistance and aminoglycoside usage: Ten years of experience in one hospital. Antimicrob. Agents Chemother. 1991, 35, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Gad, G.F.; Mohamed, H.A.; Ashour, H.M. Aminoglycoside resistance rates, phenotypes, and mechanisms of Gram-negative bacteria from infected patients in upper Egypt. PLoS ONE 2011, 6, e17224. [Google Scholar] [CrossRef] [PubMed]
- Marsot, A.; Guilhaumou, R.; Riff, C.; Blin, O. Amikacin in critically ill patients: A review of population pharmacokinetic studies. Clin. Pharmacokinet. 2017, 56, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Pacifici, G.; Marchini, G. Clinical pharmacokinetics of amikacin in neonates. Int. J. Pediatr. 2017, 5, 4407–4428. [Google Scholar]
- Yu, V.L.; Rhame, F.S.; Pesanti, E.L.; Axline, S.G. Amikacin therapy. Use against infections caused by gentamicin- and tobramycin-resistant organisms. JAMA 1977, 238, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Sklaver, A.R.; Greenman, R.L.; Hoffman, T.A. Amikacin therapy of gram-negative bacteremia and meningitis. Treatment in diseases due to multiple resistant bacilli. Arch. Intern. Med. 1978, 138, 713–716. [Google Scholar] [CrossRef] [PubMed]
- Ristuccia, A.M.; Cunha, B.A. An overview of amikacin. Ther. Drug Monit. 1985, 7, 12–25. [Google Scholar] [CrossRef] [PubMed]
- Tamma, P.D.; Cosgrove, S.E.; Maragakis, L.L. Combination therapy for treatment of infections with gram-negative bacteria. Clin. Microbiol. Rev. 2012, 25, 450–470. [Google Scholar] [CrossRef] [PubMed]
- Ambrosioni, J.; Lew, D.; Garbino, J. Nocardiosis: Updated clinical review and experience at a tertiary center. Infection 2010, 38, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.M. Mycobacterial endocarditis: A comprehensive review. Rev. Bras. Cir. Cardiovasc. 2015, 30, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Caminero, J.A.; Sotgiu, G.; Zumla, A.; Migliori, G.B. Best drug treatment for multidrug-resistant and extensively drug-resistant tuberculosis. Lancet Infect. Dis. 2010, 10, 621–629. [Google Scholar] [CrossRef]
- White, B.P.; Lomaestro, B.; Pai, M.P. Optimizing the initial amikacin dosage in adults. Antimicrob. Agents Chemother. 2015, 59, 7094–7096. [Google Scholar] [CrossRef] [PubMed]
- MacDougall, C.; Chambers, H. Aminogylcosides. In The Pharmacological Basis of Therapeutics; Brunton, L., Chamber, B., Knollman, B., Eds.; Mc Graw Hill: New York, NY, USA, 2011; pp. 1507–1517. [Google Scholar]
- Sherwin, C.M.; Svahn, S.; Van der Linden, A.; Broadbent, R.S.; Medlicott, N.J.; Reith, D.M. Individualised dosing of amikacin in neonates: A pharmacokinetic/pharmacodynamic analysis. Eur. J. Clin. Pharmacol. 2009, 65, 705–713. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, A.; Khan, D.A.; Khan, F.A.; Razzaq, A. Therapeutic drug monitoring of amikacin in preterm and term infants. Singap. Med. J. 2009, 50, 486–489. [Google Scholar]
- Tayman, C.; El-Attug, M.N.; Adams, E.; Van Schepdael, A.; Debeer, A.; Allegaert, K.; Smits, A. Quantification of amikacin in bronchial epithelial lining fluid in neonates. Antimicrob. Agents Chemother. 2011, 55, 3990–3993. [Google Scholar] [CrossRef] [PubMed]
- Kondo, J.; Francois, B.; Russell, R.J.; Murray, J.B.; Westhof, E. Crystal structure of the bacterial ribosomal decoding site complexed with amikacin containing the γ-amino-α-hydroxybutyryl (haba) group. Biochimie 2006, 88, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.J.; Murray, J.B.; Lentzen, G.; Haddad, J.; Mobashery, S. The complex of a designer antibiotic with a model aminoacyl site of the 30S ribosomal subunit revealed by X-ray crystallography. J. Am. Chem. Soc. 2003, 125, 3410–3411. [Google Scholar] [CrossRef] [PubMed]
- Quon, B.S.; Goss, C.H.; Ramsey, B.W. Inhaled antibiotics for lower airway infections. Ann. Am. Thorac. Soc. 2014, 11, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Sime, F.B.; Johnson, A.; Whalley, S.; Santoyo-Castelazo, A.; Montgomery, A.B.; Walters, K.A.; Lipman, J.; Hope, W.W.; Roberts, J.A. Pharmacodynamics of aerosolized fosfomycin and amikacin against resistant clinical isolates of Pseudomonas aeruginosa and Klebsiella pneumoniae in a hollow-fiber infection model: Experimental basis for combination therapy. Antimicrob. Agents Chemother. 2017, 61, e01763-16. [Google Scholar] [CrossRef] [PubMed]
- Hassan, N.A.; Awdallah, F.F.; Abbassi, M.M.; Sabry, N.A. Nebulized versus IV amikacin as adjunctive antibiotic for hospital and ventilator-acquired pneumonia postcardiac surgeries: A randomized controlled trial. Crit. Care Med. 2017. [Google Scholar] [CrossRef] [PubMed]
- Yagi, K.; Ishii, M.; Namkoong, H.; Asami, T.; Iketani, O.; Asakura, T.; Suzuki, S.; Sugiura, H.; Yamada, Y.; Nishimura, T.; et al. The efficacy, safety, and feasibility of inhaled amikacin for the treatment of difficult-to-treat non-tuberculous mycobacterial lung diseases. BMC Infect. Dis. 2017, 17, 558. [Google Scholar] [CrossRef] [PubMed]
- Ghannam, D.E.; Rodriguez, G.H.; Raad, I.I.; Safdar, A. Inhaled aminoglycosides in cancer patients with ventilator-associated Gram-negative bacterial pneumonia: Safety and feasibility in the era of escalating drug resistance. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.K.; Kao, P.N.; Jacobs, S.S.; Ruoss, S.J. Aerosolized amikacin for treatment of pulmonary Mycobacterium avium infections: An observational case series. BMC Pulm. Med. 2007, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Olivier, K.N.; Shaw, P.A.; Glaser, T.S.; Bhattacharyya, D.; Fleshner, M.; Brewer, C.C.; Zalewski, C.K.; Folio, L.R.; Siegelman, J.R.; Shallom, S.; et al. Inhaled amikacin for treatment of refractory pulmonary nontuberculous mycobacterial disease. Ann. Am. Thorac. Soc. 2014, 11, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Malinin, V.; Neville, M.; Eagle, G.; Gupta, R.; Perkins, W.R. Pulmonary deposition and elimination of liposomal amikacin for inhalation and effect on macrophage function after administration in rats. Antimicrob. Agents Chemother. 2016, 60, 6540–6549. [Google Scholar] [CrossRef] [PubMed]
- Tsimogianni, A.; Alexandropoulos, P.; Chantziara, V.; Vassi, A.; Micha, G.; Lagiou, F.; Chinou, E.; Michaloudis, G.; Georgiou, S. Intrathecal or intraventricular administration of colistin, vancomycin and amikacin for central nervous system infections in neurosurgical patients in an intensive care unit. Int. J. Antimicrob. Agents 2017, 49, 389–390. [Google Scholar] [CrossRef] [PubMed]
- Berning, S.E.; Cherry, T.A.; Iseman, M.D. Novel treatment of meningitis caused by multidrug-resistant Mycobacterium tuberculosis with intrathecal levofloxacin and amikacin: Case report. Clin. Infect. Dis. 2001, 32, 643–646. [Google Scholar] [CrossRef] [PubMed]
- Nicolau, D.P.; Freeman, C.D.; Belliveau, P.P.; Nightingale, C.H.; Ross, J.W.; Quintiliani, R. Experience with a once-daily aminoglycoside program administered to 2184 adult patients. Antimicrob. Agents Chemother. 1995, 39, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Sohn, Y.M.; Ko, J.Y.; Lee, S.Y.; Jhun, B.W.; Park, H.Y.; Jeon, K.; Kim, D.H.; Kim, S.Y.; Choi, J.E.; et al. Once-daily dosing of amikacin for treatment of Mycobacterium abscessus lung disease. Int. J. Tuberc. Lung Dis. 2017, 21, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Sima, M.; Hartinger, J.; Cikankova, T.; Slanar, O. Estimation of once-daily amikacin dose in critically ill adults. J. Chemother. 2017, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Tulkens, P.M. Pharmacokinetic and toxicological evaluation of a once-daily regimen versus conventional schedules of netilmicin and amikacin. J. Antimicrob. Chemother. 1991, 27 (Suppl. C), 49–61. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, A.; Thomson, A.H.; Brown, N.M.; Semple, Y.; Sluman, C.; MacGowan, A.; Lovering, A.M.; Wiffen, P.J. Amikacin use and therapeutic drug monitoring in adults: Do dose regimens and drug exposures affect either outcome or adverse events? A systematic review. J. Antimicrob. Chemother. 2016, 71, 2754–2759. [Google Scholar] [CrossRef] [PubMed]
- Bleyzac, N.; Varnier, V.; Labaune, J.M.; Corvaisier, S.; Maire, P.; Jelliffe, R.W.; Putet, G.; Aulagner, G. Population pharmacokinetics of amikacin at birth and interindividual variability in renal maturation. Eur. J. Clin. Pharmacol. 2001, 57, 499–504. [Google Scholar] [PubMed]
- Labaune, J.M.; Bleyzac, N.; Maire, P.; Jelliffe, R.W.; Boutroy, M.J.; Aulagner, G.; Putet, G. Once-a-day individualized amikacin dosing for suspected infection at birth based on population pharmacokinetic models. Biol. Neonate 2001, 80, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Howard, J.B.; McCraken, G.H., Jr.; Trujillo, H.; Mohs, E. Amikacin in newborn infants: Comparative pharmacology with kanamycin and clinical efficacy in 45 neonates with bacterial diseases. Antimicrob. Agents Chemother. 1976, 10, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, A.; Karamat, K.A.; Butt, T. Neonatal sepsis: High antibiotic resistance of the bacterial pathogens in a neonatal intensive care unit in Karachi. J. Pak. Med. Assoc. 2002, 52, 348–350. [Google Scholar] [PubMed]
- Hughes, K.M.; Johnson, P.N.; Anderson, M.P.; Sekar, K.C.; Welliver, R.C.; Miller, J.L. Comparison of amikacin pharmacokinetics in neonates following implementation of a new dosage protocol. J. Pediatr. Pharmacol. Ther. 2017, 22, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Friedland, I.R.; Funk, E.; Khoosal, M.; Klugman, K.P. Increased resistance to amikacin in a neonatal unit following intensive amikacin usage. Antimicrob. Agents Chemother. 1992, 36, 1596–1600. [Google Scholar] [CrossRef] [PubMed]
- Schiffelers, R.; Storm, G.; Bakker-Woudenberg, I. Liposome-encapsulated aminoglycosides in pre-clinical and clinical studies. J. Antimicrob. Chemother. 2001, 48, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Krieger, J.; Childs, S.; Klimberg, I. Urinary tract infection treatment using liposomal amikacin (MiKasome). In Proceedings of the Ninth European Congress of Clinical Microbiology and Infectious Diseases, Berlin, Germany, 21–24 March 1999; Elsevier: Amsterdam, The Netherlands, 1999; Volume 5S3, p. 136. [Google Scholar]
- Xiong, Y.Q.; Kupferwasser, L.I.; Zack, P.M.; Bayer, A.S. Comparative efficacies of liposomal amikacin (MiKasome) plus oxacillin versus conventional amikacin plus oxacillin in experimental endocarditis induced by Staphylococcus aureus: Microbiological and echocardiographic analyses. Antimicrob. Agents Chemother. 1999, 43, 1737–1742. [Google Scholar] [PubMed]
- Leitzke, S.; Bucke, W.; Borner, K.; Muller, R.; Hahn, H.; Ehlers, S. Rationale for and efficacy of prolonged-interval treatment using liposome-encapsulated amikacin in experimental Mycobacterium avium infection. Antimicrob. Agents Chemother. 1998, 42, 459–461. [Google Scholar] [PubMed]
- Duzgunes, N.; Perumal, V.K.; Kesavalu, L.; Goldstein, J.A.; Debs, R.J.; Gangadharam, P.R. Enhanced effect of liposome-encapsulated amikacin on Mycobacterium avium-M. intracellulare complex infection in beige mice. Antimicrob. Agents Chemother. 1988, 32, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- Schiffelers, R.M.; Storm, G.; Ten Kate, M.T.; Bakker-Woudenberg, I.A. Therapeutic efficacy of liposome-encapsulated gentamicin in rat Klebsiella pneumoniae pneumonia in relation to impaired host defense and low bacterial susceptibility to gentamicin. Antimicrob. Agents Chemother. 2001, 45, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Price, K.E.; DeFuria, M.D.; Pursiano, T.A. Amikacin, an aminoglycoside with marked activity against antibiotic-resistant clinical isolates. J. Infect. Dis. 1976, 134, S249–S261. [Google Scholar] [CrossRef] [PubMed]
- Price, K.E.; Pursiano, T.A.; DeFuria, M.D. Activity of BB-K8 (amikacin) against clinical isolates resistant to one or more aminoglycoside antibiotics. Antimicrob. Agents Chemother. 1974, 5, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.H.; Sabatelli, F.J.; Hare, R.S.; Glupczynski, Y.; Mackey, P.; Shlaes, D.; Shimizu, K.; Shaw, K.J. The most frequent aminoglycoside resistance mechanisms—Changes with time and geographic area: A reflection of aminoglycoside usage patterns? Clin. Infect. Dis. 1997, 24 (Suppl. 1), S46–S62. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. Properties of an R plasmid in Pseudomonas aeruginosa producing amikacin (BB-K8), butirosin, kanamycin, tobramycin, and sisomicin resistance. Antimicrob. Agents Chemother. 1974, 6, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Novick, R.P.; Clowes, R.C.; Cohen, S.N.; Curtiss, R.; Datta, N.; Falkow, S. Uniform nomenclature for bacterial plasmids: A proposal. Bacteriol. Rev. 1976, 40, 168–189. [Google Scholar] [PubMed]
- Kawabe, H.; Kondo, S.; Umezawa, H.; Mitsuhashi, S. R factor-mediated aminoglycoside antibiotic resistance in Pseudomonas aeruginosa: A new aminoglycoside 6′-N-acetyltransferase. Antimicrob. Agents Chemother. 1975, 7, 494–499. [Google Scholar] [CrossRef] [PubMed]
- Kawabe, H.; Naito, T.; Mitsuhashi, S. Acetylation of amikacin, a new semisynthetic antibiotic, by Pseudomonas aeruginosa carrying an R factor. Antimicrob. Agents Chemother. 1975, 7, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Hull, R.; Klinger, J.D.; Moody, E.E. Isolation and characterization of mutants of Escherichia coli K12 resistant to the new aminoglycoside antibiotic, amikacin. J. Gen. Microbiol. 1976, 94, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Perlin, M.H.; Lerner, S.A. Amikacin resistance associated with a plasmid-borne aminoglycoside phosphotransferase in Escherichia coli. Antimicrob. Agents Chemother. 1979, 16, 598–604. [Google Scholar] [CrossRef] [PubMed]
- Perlin, M.H.; Lerner, S.A. High-level amikacin resistance in Escherichia coli due to phosphorylation and impaired aminoglycoside uptake. Antimicrob. Agents Chemother. 1986, 29, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Bongaerts, G.P.; Kaptijn, G.M. Aminoglycoside phosphotransferase-II-mediated amikacin resistance in Escherichia coli. Antimicrob. Agents Chemother. 1981, 20, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Coombe, R.G.; George, A.M. New plasmid-mediated aminoglycoside adenylyltransferase of broad substrate range that adenylylates amikacin. Antimicrob. Agents Chemother. 1981, 20, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Murray, B.E.; Moellering, R.C., Jr. In-vivo acquisition of two different types of aminoglycoside resistance by a single strain of Klebsiella pneumoniae causing severe infection. Ann. Intern. Med. 1982, 96, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Cook, L.N.; Davis, R.S.; Stover, B.H. Outbreak of amikacin-resistant Enterobacteriaceae in an intensive care nursery. Pediatrics 1980, 65, 264–268. [Google Scholar] [PubMed]
- Prammananan, T.; Sander, P.; Brown, B.A.; Frischkorn, K.; Onyi, G.O.; Zhang, Y.; Bottger, E.C.; Wallace, R.J., Jr. A single 16S ribosomal RNA substitution is responsible for resistance to amikacin and other 2-deoxystreptamine aminoglycosides in Mycobacterium abscessus and Mycobacterium chelonae. J. Infect. Dis. 1998, 177, 1573–1581. [Google Scholar] [CrossRef] [PubMed]
- Jugheli, L.; Bzekalava, N.; de Rijk, P.; Fissette, K.; Portaels, F.; Rigouts, L. High level of cross-resistance between kanamycin, amikacin, and capreomycin among Mycobacterium tuberculosis isolates from Georgia and a close relation with mutations in the rrs gene. Antimicrob. Agents Chemother. 2009, 53, 5064–5068. [Google Scholar] [CrossRef] [PubMed]
- Sirgel, F.A.; Tait, M.; Warren, R.M.; Streicher, E.M.; Bottger, E.C.; van Helden, P.D.; Gey van Pittius, N.C.; Coetzee, G.; Hoosain, E.Y.; Chabula-Nxiweni, M.; et al. Mutations in the rrs A1401G gene and phenotypic resistance to amikacin and capreomycin in Mycobacterium tuberculosis. Microb. Drug Resist. 2012, 18, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.; Dai, G.; Long, Q.; Yu, X.; Dong, L.; Huang, H.; Xie, J. Mycobacterium tuberculosis rrs A1401G mutation correlates with high-level resistance to kanamycin, amikacin, and capreomycin in clinical isolates from mainland China. Diagn. Microbiol. Infect. Dis. 2013, 77, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Kambli, P.; Ajbani, K.; Nikam, C.; Sadani, M.; Shetty, A.; Udwadia, Z.; Georghiou, S.B.; Rodwell, T.C.; Catanzaro, A.; Rodrigues, C. Correlating rrs and eis promoter mutations in clinical isolates of Mycobacterium tuberculosis with phenotypic susceptibility levels to the second-line injectables. Int. J. Mycobacteriol. 2016, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tsodikov, O.V.; Green, K.D.; Garneau-Tsodikova, S. A random sequential mechanism of aminoglycoside acetylation by Mycobacterium tuberculosis Eis protein. PLoS ONE 2014, 9, e92370. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Biswas, T.; Porter, V.R.; Tsodikov, O.V.; Garneau-Tsodikova, S. Unusual regioversatility of acetyltransferase Eis, a cause of drug resistance in XDR-TB. Proc. Natl. Acad. Sci. USA 2011, 108, 9804–9808. [Google Scholar] [CrossRef] [PubMed]
- Casin, I.; Hanau-Bercot, B.; Podglajen, I.; Vahaboglu, H.; Collatz, E. Salmonella enterica serovar Typhimurium bla(PER-1)-carrying plasmid pSTI1 encodes an extended-spectrum aminoglycoside 6′-N-acetyltransferase of type Ib. Antimicrob. Agents Chemother. 2003, 47, 697–703. [Google Scholar] [CrossRef] [PubMed]
- Robicsek, A.; Strahilevitz, J.; Jacoby, G.A.; Macielag, M.; Abbanat, D.; Park, C.H.; Bush, K.; Hooper, D.C. Fluoroquinolone-modifying enzyme: A new adaptation of a common aminoglycoside acetyltransferase. Nat. Med. 2006, 12, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Fisher, J.F.; Mobashery, S. The bifunctional enzymes of antibiotic resistance. Curr. Opin. Microbiol. 2009, 12, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Centron, D.; Roy, P.H. Presence of a group II intron in a multiresistant Serratia marcescens strain that harbors three integrons and a novel gene fusion. Antimicrob. Agents Chemother. 2002, 46, 1402–1409. [Google Scholar] [CrossRef] [PubMed]
- Boehr, D.D.; Daigle, D.M.; Wright, G.D. Domain-domain interactions in the aminoglycoside antibiotic resistance enzyme AAC(6′)-APH(2″). Biochemistry 2004, 43, 9846–9855. [Google Scholar] [CrossRef] [PubMed]
- Ferretti, J.J.; Gilmore, K.S.; Courvalin, P. Nucleotide sequence analysis of the gene specifying the bifunctional 6′-aminoglycoside acetyltransferase 2″-aminoglycoside phosphotransferase enzyme in Streptococcus faecalis and identification and cloning of gene regions specifying the two activities. J. Bacteriol. 1986, 167, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Culebras, E.; Martinez, J.L. Aminoglycoside resistance mediated by the bifunctional enzyme 6′-N-aminoglycoside acetyltransferase-2″-O-aminoglycoside phosphotransferase. Front. Biosci. 1999, 4, D1–D8. [Google Scholar] [CrossRef] [PubMed]
- Dubois, V.; Poirel, L.; Marie, C.; Arpin, C.; Nordmann, P.; Quentin, C. Molecular characterization of a novel class 1 integron containing bla(GES-1) and a fused product of aac(3)-Ib/aac(6″)-Ib″ gene cassettes in Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2002, 46, 638–645. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.; Toleman, M.; Ribeiro, J.; Sader, H.; Jones, R.; Walsh, T. Integron carrying a novel metallo-β-lactamase gene, blaIMP-16, and a fused form of aminoglycoside-resistance gene aac(6′)-30/aac(6′)-Ib′: Report from the SENTRY antimicrobial surveillance program. Antimicrob. Agents Chemother. 2004, 48, 4693–4702. [Google Scholar] [CrossRef] [PubMed]
- Li, C.R.; Yang, X.Y.; Lou, R.H.; Zhang, W.X.; Wang, Y.M.; Yuan, M.; Li, Y.; Chen, H.Z.; Hong, B.; Sun, C.H.; et al. In vitro antibacterial activity of vertilmicin and its susceptibility to modifications by the recombinant AAC6′-APH2″ enzyme. Antimicrob. Agents Chemother. 2008, 52, 3875–3882. [Google Scholar] [CrossRef] [PubMed]
- Costa, Y.; Galimand, M.; Leclercq, R.; Duval, J.; Courvalin, P. Characterization of the chromosomal aac(6′)-Ii gene specific for Enterococcus faecium. Antimicrob. Agents Chemother. 1993, 37, 1896–1903. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.W.; Kak, V.; You, I.; Kao, S.J.; Petrin, J.; Clewell, D.B.; Lerner, S.A.; Miller, G.H.; Shaw, K.J. Aminoglycoside resistance genes aph(2″)-Ib and aac(6′)-Im detected together in strains of both Escherichia coli and Enterococcus faecium. Antimicrob. Agents Chemother. 2001, 45, 2691–2694. [Google Scholar] [CrossRef] [PubMed]
- Rouch, D.A.; Byrne, M.E.; Kong, Y.C.; Skurray, R.A. The aacA-aphD gentamicin and kanamycin resistance determinant of Tn4001 from Staphylococcus aureus: Expression and nucleotide sequence analysis. J. Gen. Microbiol. 1987, 133, 3039–3052. [Google Scholar] [CrossRef] [PubMed]
- Zarrilli, R.; Tripodi, M.F.; Di Popolo, A.; Fortunato, R.; Bagattini, M.; Crispino, M.; Florio, A.; Triassi, M.; Utili, R. Molecular epidemiology of high-level aminoglycoside-resistant enterococci isolated from patients in a university hospital in southern Italy. J. Antimicrob. Chemother. 2005, 56, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, S.Y.; Garneau-Tsodikova, S. Expanding aminoglycoside resistance enzyme regiospecificity by mutation and truncation. Biochemistry 2016, 55, 5726–5737. [Google Scholar] [CrossRef] [PubMed]
- Sadowy, E.; Sienko, A.; Gawryszewska, I.; Bojarska, A.; Malinowska, K.; Hryniewicz, W. High abundance and diversity of antimicrobial resistance determinants among early vancomycin-resistant Enterococcus faecium in Poland. Eur. J. Clin. Microbiol. Infect. Dis. 2013, 32, 1193–1203. [Google Scholar] [CrossRef] [PubMed]
- Chow, V.C.; Hawkey, P.M.; Chan, E.W.; Chin, M.L.; Au, T.K.; Fung, D.K.; Chan, R.C. High-level gentamicin resistance mediated by a Tn4001-like transposon in seven nonclonal hospital isolates of Streptococcus pasteurianus. Antimicrob. Agents Chemother. 2007, 51, 2508–2513. [Google Scholar] [CrossRef] [PubMed]
- Lyon, B.R.; Gillespie, M.T.; Byrne, M.E.; May, J.W.; Skurray, R.A. Plasmid-mediated resistance to gentamicin in Staphylococcus aureus: The involvement of a transposon. J. Med. Microbiol. 1987, 23, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, M.T.; Lyon, B.R.; Messerotti, L.J.; Skurray, R.A. Chromosome- and plasmid-mediated gentamicin resistance in Staphylococcus aureus encoded by Tn4001. J. Med. Microbiol. 1987, 24, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Hodel-Christian, S.L.; Murray, B.E. Characterization of the gentamicin resistance transposon Tn5281 from Enterococcus faecalis and comparison to staphylococcal transposons Tn4001 and Tn4031. Antimicrob. Agents Chemother. 1991, 35, 1147–1152. [Google Scholar] [CrossRef] [PubMed]
- Thomas, W.D., Jr.; Archer, G.L. Mobility of gentamicin resistance genes from staphylococci isolated in the United States: Identification of Tn4031, a gentamicin resistance transposon from Staphylococcus epidermidis. Antimicrob. Agents Chemother. 1989, 33, 1335–1341. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, S.J.; Huang, Y.; Berghuis, A.M. Antibiotic binding drives catalytic activation of aminoglycoside kinase APH(2″)-Ia. Structure 2016, 24, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Toth, M.; Bhattacharya, M.; Frase, H.; Vakulenko, S.B. Structure of the phosphotransferase domain of the bifunctional aminoglycoside-resistance enzyme AAC(6′)-Ie-APH(2″)-Ia. Acta Crystallogr. D Biol. Crystallogr. 2014, 70, 1561–1571. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Toth, M.; Weiss, T.M.; Frase, H.; Vakulenko, S.B. Structure of the bifunctional aminoglycoside-resistance enzyme AAC(6′)-Ie-APH(2″)-Ia revealed by crystallographic and small-angle X-ray scattering analysis. Acta Crystallogr. D Biol. Crystallogr. 2014, 70, 2754–2764. [Google Scholar] [CrossRef] [PubMed]
- Burk, D.L.; Ghuman, N.; Wybenga-Groot, L.E.; Berghuis, A.M. X-ray structure of the AAC(6′)-Ii antibiotic resistance enzyme at 1.8 A resolution; examination of oligomeric arrangements in GNAT superfamily members. Protein Sci. 2003, 12, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Burk, D.L.; Xiong, B.; Breitbach, C.; Berghuis, A.M. Structures of aminoglycoside acetyltransferase AAC(6′)-Ii in a novel crystal form: Structural and normal-mode analyses. Acta Crystallogr. D Biol. Crystallogr. 2005, 61, 1273–1279. [Google Scholar] [CrossRef] [PubMed]
- Draker, K.A.; Northrop, D.B.; Wright, G.D. Kinetic mechanism of the GCN5-related chromosomal aminoglycoside acetyltransferase AAC(6′)-Ii from Enterococcus faecium: Evidence of dimer subunit cooperativity. Biochemistry 2003, 42, 6565–6574. [Google Scholar] [CrossRef] [PubMed]
- Boucher, H.W.; Talbot, G.H.; Bradley, J.S.; Edwards, J.E.; Gilbert, D.; Rice, L.B.; Scheld, M.; Spellberg, B.; Bartlett, J. Bad bugs, no drugs: No ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis. 2009, 48, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Pourreza, A.; Witherspoon, M.; Fox, J.; Newmark, J.; Bui, D.; Tolmasky, M.E. Mutagenesis analysis of a conserved region involved in acetyl coenzyme A binding in the aminoglycoside 6′-N-acetyltransferase type Ib encoded by plasmid pJHCMW1. Antimicrob. Agents Chemother. 2005, 49, 2979–2982. [Google Scholar] [CrossRef] [PubMed]
- Rice, L.B.; Carias, L.L.; Hutton, R.A.; Rudin, S.D.; Endimiani, A.; Bonomo, R.A. The KQ element, a complex genetic region conferring transferable resistance to carbapenems, aminoglycosides, and fluoroquinolones in Klebsiella pneumoniae. Antimicrob. Agents Chemother. 2008, 52, 3427–3429. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.S.; Traglia, G.M.; Lin, D.L.; Tran, T.; Tolmasky, M.E. Plasmid-mediated antibiotic resistance and virulence in Gram-negatives: The Klebsiella pneumoniae paradigm. Microbiol. Spectr. 2014, 2, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, E.; Saenz, Y.; Zarazaga, M.; Rocha-Gracia, R.; Martinez-Martinez, L.; Arlet, G.; Torres, C. qnr, aac(6′)-Ib-cr and qepA genes in Escherichia coli and Klebsiella spp.: Genetic environments and plasmid and chromosomal location. J. Antimicrob. Chemother. 2012, 67, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Arivett, B.A.; Fiester, S.E.; Ream, D.C.; Centron, D.; Ramirez, M.S.; Tolmasky, M.E.; Actis, L.A. Draft genome of the multidrug-resistant Acinetobacter baumannii strain A155 clinical isolate. Genome Announc. 2015, 3, e00212–e00215. [Google Scholar] [CrossRef] [PubMed]
- Poirel, L.; Cabanne, L.; Collet, L.; Nordmann, P. Class II transposon-borne structure harboring metallo-β-lactamase gene blaVIM-2 in Pseudomonas putida. Antimicrob. Agents Chemother. 2006, 50, 2889–2891. [Google Scholar] [CrossRef] [PubMed]
- Chamorro, R.M.; Actis, L.A.; Crosa, J.H.; Tolmasky, M.E. Dissemination of plasmid-mediated amikacin resistance among pathogenic Klebsiella pneumoniae. Medicina 1990, 50, 543–547. [Google Scholar] [PubMed]
- Woodford, N.; Carattoli, A.; Karisik, E.; Underwood, A.; Ellington, M.J.; Livermore, D.M. Complete nucleotide sequences of plasmids pEK204, pEK499, and pEK516, encoding CTX-M enzymes in three major Escherichia coli lineages from the United Kingdom, all belonging to the international O25:H4-ST131 clone. Antimicrob. Agents Chemother. 2009, 53, 4472–4482. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Chavda, K.D.; Al Laham, N.; Melano, R.G.; Jacobs, M.R.; Bonomo, R.A.; Kreiswirth, B.N. Complete nucleotide sequence of a blaKPC-harboring IncI2 plasmid and its dissemination in New Jersey and New York hospitals. Antimicrob. Agents Chemother. 2013, 57, 5019–5025. [Google Scholar] [CrossRef] [PubMed]
- Gillings, M.R. Class 1 integrons as invasive species. Curr. Opin. Microbiol. 2017, 38, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Escudero, J.A.; Loot, C.; Nivina, A.; Mazel, D. The integron: Adaptation on demand. Microbiol. Spectr. 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, M.S.; Parenteau, T.R.; Centron, D.; Tolmasky, M.E. Functional characterization of Tn1331 gene cassettes. J. Antimicrob. Chemother. 2008, 62, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Zong, Z.; Partridge, S.R.; Iredell, J.R. A blaVEB-1 variant, blaVEB-6, associated with repeated elements in a complex genetic structure. Antimicrob. Agents Chemother. 2009, 53, 1693–1697. [Google Scholar] [CrossRef] [PubMed]
- Van Nhieu, G.T.; Collatz, E. Primary structure of an aminoglycoside 6′-N-acetyltransferase AAC(6′)-4, fused in vivo with the signal peptide of the Tn3-encoded β-lactamase. J. Bacteriol. 1987, 169, 5708–5714. [Google Scholar] [CrossRef]
- Van Nhieu, G.T.; Goldstein, F.W.; Pinto, M.E.; Acar, J.F.; Collatz, E. Transfer of amikacin resistance by closely related plasmids in members of the family Enterobacteriaceae isolated in Chile. Antimicrob. Agents Chemother. 1986, 29, 833–837. [Google Scholar] [CrossRef] [PubMed]
- Nobuta, K.; Tolmasky, M.E.; Crosa, L.M.; Crosa, J.H. Sequencing and expression of the 6′-N-acetyltransferase gene of transposon Tn1331 from Klebsiella pneumoniae. J. Bacteriol. 1988, 170, 3769–3773. [Google Scholar] [CrossRef] [PubMed]
- Sarno, R.; McGillivary, G.; Sherratt, D.J.; Actis, L.A.; Tolmasky, M.E. Complete nucleotide sequence of Klebsiella pneumoniae multiresistance plasmid pJHCMW1. Antimicrob. Agents Chemother. 2002, 46, 3422–3427. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Lamothe, R.; Tran, T.; Meas, D.; Lee, L.; Li, A.M.; Sherratt, D.J.; Tolmasky, M.E. High-copy bacterial plasmids diffuse in the nucleoid-free space, replicate stochastically and are randomly partitioned at cell division. Nucleic Acids Res. 2014, 42, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Trigueros, S.; Tran, T.; Sorto, N.; Newmark, J.; Colloms, S.D.; Sherratt, D.J.; Tolmasky, M.E. mwr Xer site-specific recombination is hypersensitive to DNA supercoiling. Nucleic Acids Res. 2009, 37, 3580–3587. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, M.P.; Orman, B.; Errecalde, L.; Kaufman, S.; Centron, D. Characterization of Tn6238 with a new allele of aac(6′)-Ib-cr. Antimicrob. Agents Chemother. 2015, 59, 2893–2897. [Google Scholar] [CrossRef] [PubMed]
- Warburg, G.; Hidalgo-Grass, C.; Partridge, S.R.; Tolmasky, M.E.; Temper, V.; Moses, A.E.; Block, C.; Strahilevitz, J. A carbapenem-resistant Klebsiella pneumoniae epidemic clone in Jerusalem: Sequence type 512 carrying a plasmid encoding aac(6′)-Ib. J. Antimicrob. Chemother. 2012, 67, 898–901. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Chandler, M.; Varani, A.M.; Hickman, A.B.; Dekker, J.P.; Dyda, F. Mechanisms of evolution in high-consequence drug resistance plasmids. MBio 2016, 7, e01987-16. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Hickman, A.B.; Varani, A.M.; Siguier, P.; Chandler, M.; Dekker, J.P.; Dyda, F. Insertion sequence IS26 reorganizes plasmids in clinically isolated multidrug-resistant bacteria by replicative transposition. MBio 2015, 6, e00762. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Mathema, B.; Chavda, K.D.; DeLeo, F.R.; Bonomo, R.A.; Kreiswirth, B.N. Carbapenemase-producing Klebsiella pneumoniae: Molecular and genetic decoding. Trends Microbiol. 2014, 22, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Chavda, K.D.; Chen, L.; Jacobs, M.R.; Bonomo, R.A.; Kreiswirth, B.N. Molecular diversity and plasmid analysis of KPC-producing Escherichia coli. Antimicrob. Agents Chemother. 2016, 60, 4073–4081. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Deng, H.; Li, L.; Chen, M.Y.; Fang, L.X.; Yang, Q.E.; Liu, Y.H.; Liao, X.P. Complete nucleotide sequence of cfr-carrying IncX4 plasmid pSD11 from Escherichia coli. Antimicrob. Agents Chemother. 2015, 59, 738–741. [Google Scholar] [CrossRef] [PubMed]
- Deleo, F.R.; Chen, L.; Porcella, S.F.; Martens, C.A.; Kobayashi, S.D.; Porter, A.R.; Chavda, K.D.; Jacobs, M.R.; Mathema, B.; Olsen, R.J.; et al. Molecular dissection of the evolution of carbapenem-resistant multilocus sequence type 258 Klebsiella pneumoniae. Proc. Natl. Acad. Sci. USA 2014, 111, 4988–4993. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Mondal, A.; Mitra, S.; Basu, S. Acinetobacter baumannii transfers the blaNDM-1 gene via outer membrane vesicles. J. Antimicrob. Chemother. 2017, 72, 2201–2207. [Google Scholar] [CrossRef] [PubMed]
- Rumbo, C.; Fernandez-Moreira, E.; Merino, M.; Poza, M.; Mendez, J.A.; Soares, N.C.; Mosquera, A.; Chaves, F.; Bou, G. Horizontal transfer of the OXA-24 carbapenemase gene via outer membrane vesicles: A new mechanism of dissemination of carbapenem resistance genes in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2011, 55, 3084–3090. [Google Scholar] [CrossRef] [PubMed]
- Yaron, S.; Kolling, G.L.; Simon, L.; Matthews, K.R. Vesicle-mediated transfer of virulence genes from Escherichia coli O157:H7 to other enteric bacteria. Appl. Environ. Microbiol. 2000, 66, 4414–4420. [Google Scholar] [CrossRef] [PubMed]
- Saberi, F.; Kamali, M.; Najafi, A.; Yazdanparast, A.; Moghaddam, M.M. Natural antisense RNAs as mRNA regulatory elements in bacteria: A review on function and applications. Cell. Mol. Biol. Lett. 2016, 21, 6. [Google Scholar] [CrossRef] [PubMed]
- Thomason, M.K.; Storz, G. Bacterial antisense RNAs: How many are there, and what are they doing? Annu. Rev. Genet. 2010, 44, 167–188. [Google Scholar] [CrossRef] [PubMed]
- Waldbeser, L.S.; Tolmasky, M.E.; Actis, L.A.; Crosa, J.H. Mechanisms for negative regulation by iron of the fatA outer membrane protein gene expression in Vibrio anguillarum 775. J. Biol. Chem. 1993, 268, 10433–10439. [Google Scholar] [PubMed]
- Tolmasky, M.E.; Actis, L.A.; Crosa, J.H. Plasmid DNA replication. In Encyclopedia of Industrial Biotechnology: Bioprocess, Bioseparation, and Cell Technology; Flickinger, M., Ed.; John Wiley and Sons, Inc.: New York, NY, USA, 2010; Volume 6, pp. 3931–3953. [Google Scholar]
- Brantl, S. Plasmid Replication Control by Antisense RNAs. In Plasmids: Biology and Impact in Biotiechnology and Discovery; Tolmasky, M.E., Alonso, J., Eds.; ASM Press: Washignton, DC, USA, 2015; pp. 83–104. [Google Scholar]
- Goyal, N.; Narayanaswami, P. Making sense of antisense oligonucleotides: A narrative review. Muscle Nerve 2017. [Google Scholar] [CrossRef] [PubMed]
- Woodford, N.; Wareham, D.W. Tackling antibiotic resistance: A dose of common antisense? J. Antimicrob. Chemother. 2009, 63, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, L.C.; Sperling-Petersen, H.U.; Mortensen, K.K. Hitting bacteria at the heart of the central dogma: Sequence-specific inhibition. Microb. Cell Fact. 2007, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Davies-Sala, C.; Soler-Bistue, A.; Bonomo, R.A.; Zorreguieta, A.; Tolmasky, M.E. External guide sequence technology: A path to development of novel antimicrobial therapeutics. Ann. N. Y. Acad. Sci. 2015, 1354, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Dinan, A.M.; Loftus, B.J. (Non-)translational medicine: Targeting bacterial RNA. Front. Genet. 2013, 4, 230. [Google Scholar] [CrossRef] [PubMed]
- Sully, E.K.; Geller, B.L. Antisense antimicrobial therapeutics. Curr. Opin. Microbiol. 2016, 33, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Sarno, R.; Ha, H.; Weinsetel, N.; Tolmasky, M.E. Inhibition of aminoglycoside 6′-N-acetyltransferase type Ib-mediated amikacin resistance by antisense oligodeoxynucleotides. Antimicrob. Agents Chemother. 2003, 47, 3296–3304. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.; Arivett, B.A.; Actis, L.A.; Tolmasky, M.E. Inhibition of AAC(6′)-Ib-mediated resistance to amikacin in Acinetobacter baumannii by an antisense peptide-conjugated 2’,4’-bridged nucleic acid-NC-DNA hybrid oligomer. Antimicrob. Agents Chemother. 2015, 59, 5798–5803. [Google Scholar] [CrossRef] [PubMed]
- Lundblad, E.W.; Altman, S. Inhibition of gene expression by RNase P. New Biotechnol. 2010, 27, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.; Jani, S.; Sala, C.D.; Soler-Bistue, A.J.; Zorreguieta, A.; Tolmasky, M.E. Assessment of configurations and chemistries of bridged nucleic acids-containing oligomers as external guide sequences: A methodology for inhibition of expression of antibiotic resistance genes. Biol. Methods Protoc. 2016, 1. [Google Scholar] [CrossRef] [PubMed]
- Soler Bistue, A.J.; Ha, H.; Sarno, R.; Don, M.; Zorreguieta, A.; Tolmasky, M.E. External guide sequences targeting the aac(6′)-Ib mRNA induce inhibition of amikacin resistance. Antimicrob. Agents Chemother. 2007, 51, 1918–1925. [Google Scholar] [CrossRef] [PubMed]
- Bistue, A.J.S.; Martin, F.A.; Vozza, N.; Ha, H.; Joaquin, J.C.; Zorreguieta, A.; Tolmasky, M.E. Inhibition of aac(6′)-Ib-mediated amikacin resistance by nuclease-resistant external guide sequences in bacteria. Proc. Natl. Acad. Sci. USA 2009, 106, 13230–13235. [Google Scholar] [CrossRef] [PubMed]
- Tolmasky, M.E. Strategies to prolong the useful life of existing antibiotics and help overcoming the antibiotic resistance crisis. In Frontiers in Clinical Drug Research-Anti Infectives; Atta-ur-Rhaman, Ed.; Bentham Books: Sharjah, UAE, 2017; Volume 1, pp. 1–27. [Google Scholar]
- Lombes, T.; Begis, G.; Maurice, F.; Turcaud, S.; Lecourt, T.; Dardel, F.; Micouin, L. NMR-guided fragment-based approach for the design of AAC(6′)-Ib ligands. ChemBioChem 2008, 9, 1368–1371. [Google Scholar] [CrossRef] [PubMed]
- Friesner, R.A.; Banks, J.L.; Murphy, R.B.; Halgren, T.A.; Klicic, J.J.; Mainz, D.T.; Repasky, M.P.; Knoll, E.H.; Shelley, M.; Perry, J.K.; et al. Glide: A new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J. Med. Chem. 2004, 47, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Halgren, T.A.; Murphy, R.B.; Friesner, R.A.; Beard, H.S.; Frye, L.L.; Pollard, W.T.; Banks, J.L. Glide: A new approach for rapid, accurate docking and scoring. 2. Enrichment factors in database screening. J. Med. Chem. 2004, 47, 1750–1759. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Chiem, K.; Jani, S.; Fuentes, B.; Lin, D.L.; Rasche, M.E.; Tolmasky, M.E. Identification of an inhibitor of the aminoglycoside 6′-N-acetyltransferase type Ib [AAC(6′)-Ib] by glide molecular docking. MedChemComm 2016, 7, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.L.; Tran, T.; Adams, C.; Alam, J.Y.; Herron, S.R.; Tolmasky, M.E. Inhibitors of the aminoglycoside 6′-N-acetyltransferase type Ib [AAC(6′)-Ib] identified by in silico molecular docking. Bioorg. Med. Chem. Lett. 2013, 23, 5694–5698. [Google Scholar] [CrossRef] [PubMed]
- Green, K.D.; Chen, W.; Garneau-Tsodikova, S. Identification and characterization of inhibitors of the aminoglycoside resistance acetyltransferase Eis from Mycobacterium tuberculosis. ChemMedChem 2012, 7, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Garzan, A.; Willby, M.J.; Green, K.D.; Gajadeera, C.S.; Hou, C.; Tsodikov, O.V.; Posey, J.E.; Garneau-Tsodikova, S. Sulfonamide-based Inhibitors of aminoglycoside acetyltransferase Eis abolish resistance to kanamycin in Mycobacterium tuberculosis. J. Med. Chem. 2016, 59, 10619–10628. [Google Scholar] [CrossRef] [PubMed]
- Garzan, A.; Willby, M.J.; Ngo, H.X.; Gajadeera, C.S.; Green, K.D.; Holbrook, S.Y.; Hou, C.; Posey, J.E.; Tsodikov, O.V.; Garneau-Tsodikova, S. Combating enhanced intracellular survival (Eis)-mediated kanamycin resistance of Mycobacterium tuberculosis by novel pyrrolo[1,5-a]pyrazine-based Eis inhibitors. ACS Infect. Dis. 2017, 3, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Garzan, A.; Willby, M.J.; Green, K.D.; Tsodikov, O.V.; Posey, J.E.; Garneau-Tsodikova, S. Discovery and optimization of two Eis inhibitor families as kanamycin adjuvants against drug-resistant M. tuberculosis. ACS Med. Chem. Lett. 2016, 7, 1219–1221. [Google Scholar] [CrossRef] [PubMed]
- Chiem, K.; Fuentes, B.A.; Lin, D.L.; Tran, T.; Jackson, A.; Ramirez, M.S.; Tolmasky, M.E. Inhibition of aminoglycoside 6′-N-acetyltransferase type Ib-mediated amikacin resistance in Klebsiella pneumoniae by zinc and copper pyrithione. Antimicrob. Agents Chemother. 2015, 59, 5851–5853. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Green, K.D.; Johnson, B.R.; Garneau-Tsodikova, S. Inhibition of aminoglycoside acetyltransferase resistance enzymes by metal salts. Antimicrob. Agents Chemother. 2015, 59, 4148–4156. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.L.; Tran, T.; Alam, J.Y.; Herron, S.R.; Ramirez, M.S.; Tolmasky, M.E. Inhibition of aminoglycoside 6′-N-acetyltransferase type Ib by zinc: Reversal of amikacin resistance in Acinetobacter baumannii and Escherichia coli by a zinc ionophore. Antimicrob. Agents Chemother. 2014, 58, 4238–4241. [Google Scholar] [CrossRef] [PubMed]
- Barnham, K.J.; Bush, A.I. Biological metals and metal-targeting compounds in major neurodegenerative diseases. Chem. Soc. Rev. 2014, 43, 6727–6749. [Google Scholar] [CrossRef] [PubMed]
- Bareggi, S.R.; Cornelli, U. Clioquinol: Review of its mechanisms of action and clinical uses in neurodegenerative disorders. CNS Neurosci. Ther. 2012, 18, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Chiem, K.; Hue, F.; Magallon, J.; Tolmasky, M.E. Inhibition of aminoglycoside 6′-N-acetyltransferase type Ib-mediated amikacin resistance by zinc complexed to clioquinol, an ionophore active against tumors and neurodegenerative diseases. Int. J. Antimicrob. Agents 2017. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Hayashi, I.; Kouda, S.; Kato, F.; Fujiwara, T.; Kayama, S.; Hirakawa, H.; Itaha, H.; Ohge, H.; Gotoh, N.; et al. Identification and characterization of a novel aac(6′)-Iag associated with the blaIMP-1-integron in a multidrug-resistant Pseudomonas aeruginosa. PLoS ONE 2013, 8, e70557. [Google Scholar] [CrossRef] [PubMed]
- Xiong, J.; Hawkey, P.; Roy, P.H. Novel Class 1 Integrons on Large Plasmids in Multiresistant Pseudomonas aeruginosa Isolated from a Multicenter Survey in Guangzhou, PRC. Available online: https://www.ncbi.nlm.nih.gov/nuccore/208436664 (accessed on 11 November 2017).
- Tada, T.; Miyoshi-Akiyama, T.; Shimada, K.; Shimojima, M.; Kirikae, T. Novel 6′-N-aminoglycoside acetyltransferase AAC(6′)-Iaj from a clinical isolate of Pseudomonas aeruginosa. Antimicrob. Agents Chemother. 2013, 57, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Tada, T.; Miyoshi-Akiyama, T.; Dahal, R.K.; Mishra, S.K.; Shimada, K.; Ohara, H.; Kirikae, T.; Pokhrel, B.M. Identification of a novel 6′-N-aminoglycoside acetyltransferase, AAC(6′)-Iak, from a multidrug-resistant clinical isolate of Stenotrophomonas maltophilia. Antimicrob. Agents Chemother. 2014, 58, 6324–6327. [Google Scholar] [CrossRef] [PubMed]
- Tada, T.; Miyoshi-Akiyama, T.; Shimada, K.; Dahal, R.K.; Mishra, S.K.; Ohara, H.; Kirikae, T.; Pokhrel, B.M. A novel 6′-N-aminoglycoside acetyltransferase, AAC(6′)-Ial, from a clinical isolate of Serratia marcescens. Microb. Drug Resist. 2016, 22, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Wachino, J.; Kimura, K.; Yamada, K.; Arakawa, Y. New plasmid-mediated aminoglycoside 6′-N-acetyltransferase, AAC(6′)-Ian, and ESBL, TLA-3, from a Serratia marcescens clinical isolate. J. Antimicrob. Chemother. 2015, 70, 1331–1337. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).