An Overview of l-Amino Acid Oxidase Functions from Bacteria to Mammals: Focus on the Immunoregulatory Phenylalanine Oxidase IL4I1
Abstract
1. Introduction
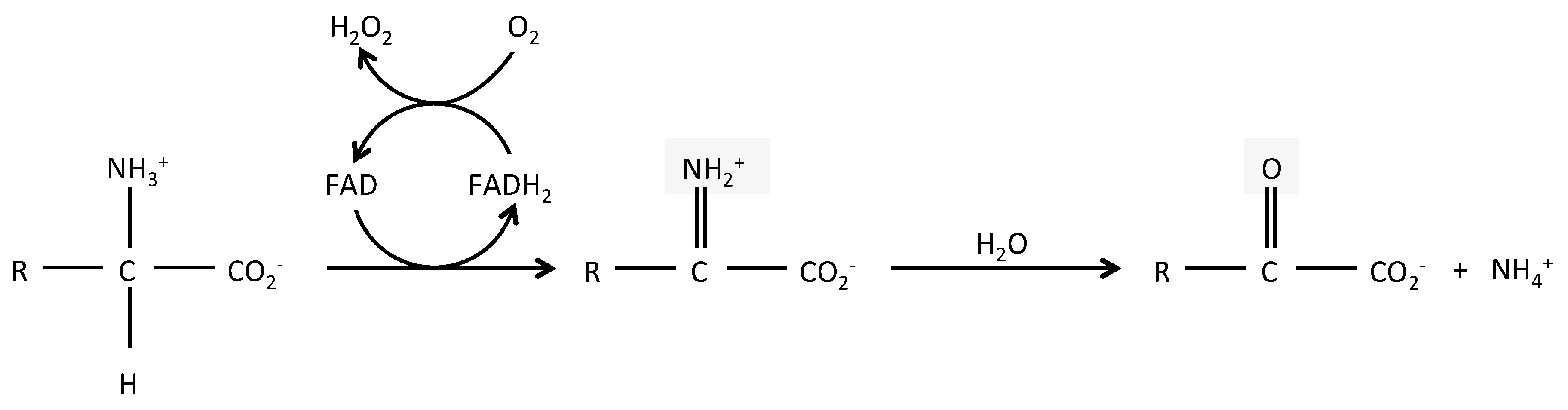
2. LAAO Structure and Reaction
3. LAAO Expression and Functions
4. Mammalian LAAOs
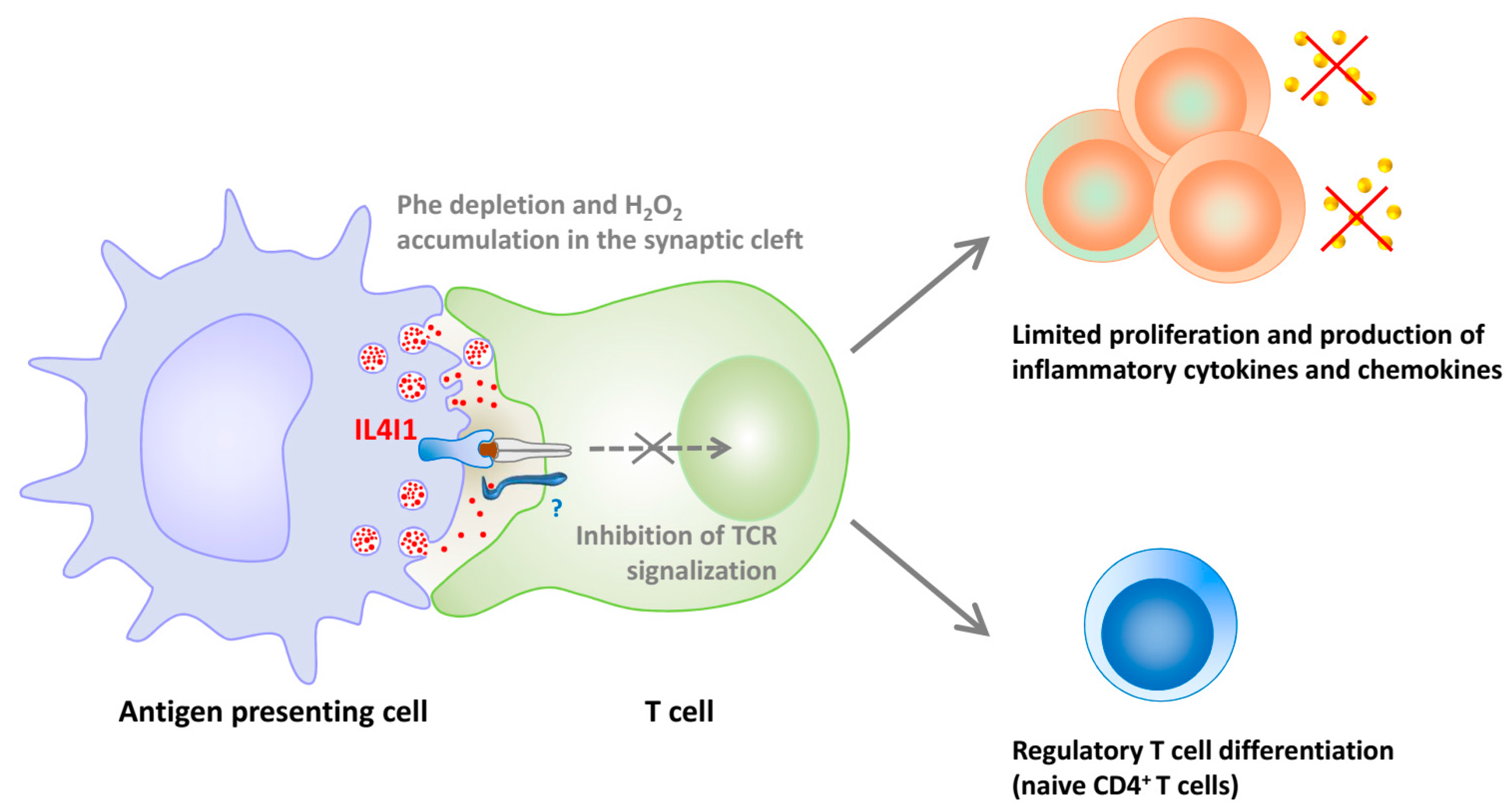
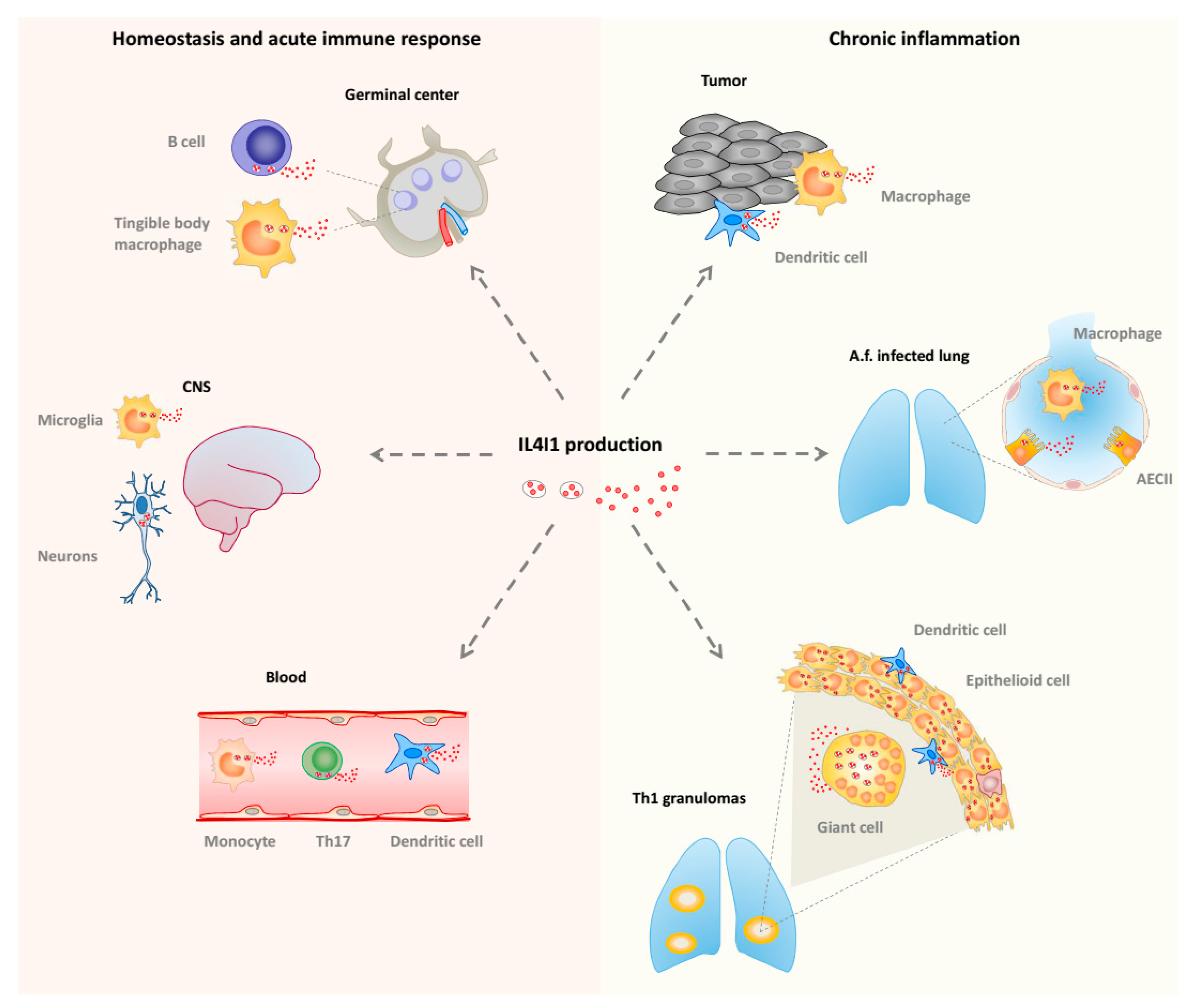
5. IL4I1, an LAAO Implicated in Immune Regulation
6. Conclusions
Acknowledgments
Conflicts of Interest
Abbreviations
| LAAOs | l-amino acid oxidases |
| DAAOs | d-amino acid oxidases |
| FAD | Flavin Adenine Dinucleotide |
| FMN | Flavin Mononucleotide |
| FoxP3 | Forkhead box P3 |
| IFN | Interferon |
| IL | Interleukin |
| IL4I1 | Interleukin-4 Induced gene 1 |
| NFκB | Nuclear factor-kappa B |
| RORγT | Retinoic acid receptor-related orphan receptor gamma |
| STAT | Signal Transducers and Activators of Transcription |
| Th | T helper |
| TNF | Tumor necrosis factor |
References
- Blanchard, M.; Green, D.E.; Nocito, V.; Ratner, S. Isolation of l-amino acid oxidase. J. Biol. Chem. 1945, 161, 583–597. [Google Scholar] [PubMed]
- Izidoro, L.F.; Sobrinho, J.C.; Mendes, M.M.; Costa, T.R.; Grabner, A.N.; Rodrigues, V.M.; da Silva, S.L.; Zanchi, F.B.; Zuliani, J.P.; Fernandes, C.F.; et al. Snake venom l-amino acid oxidases: Trends in pharmacology and biochemistry. Biomed. Res. Int. 2014, 2014, 196754. [Google Scholar] [CrossRef] [PubMed]
- Campillo-Brocal, J.C.; Lucas-Elio, P.; Sanchez-Amat, A. Distribution in Different Organisms of Amino Acid Oxidases with FAD or a Quinone As Cofactor and Their Role as Antimicrobial Proteins in Marine Bacteria. Mar. Drugs 2015, 13, 7403–7418. [Google Scholar] [CrossRef] [PubMed]
- Faust, A.; Niefind, K.; Hummel, W.; Schomburg, D. The structure of a bacterial l-amino acid oxidase from Rhodococcus opacus gives new evidence for the hydride mechanism for dehydrogenation. J. Mol. Biol. 2007, 367, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, P.R.; Rustiguel, J.K.; Soares, R.O.; Sampaio, S.V.; Cristina Nonato, M. Crystal structure and molecular dynamics studies of l-amino acid oxidase from Bothrops atrox. Toxicon 2017, 128, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Moustafa, I.M.; Foster, S.; Lyubimov, A.Y.; Vrielink, A. Crystal structure of LAAO from Calloselasma rhodostoma with an l-phenylalanine substrate: Insights into structure and mechanism. J. Mol. Biol. 2006, 364, 991–1002. [Google Scholar] [CrossRef] [PubMed]
- Pawelek, P.D.; Cheah, J.; Coulombe, R.; Macheroux, P.; Ghisla, S.; Vrielink, A. The structure of l-amino acid oxidase reveals the substrate trajectory into an enantiomerically conserved active site. EMBO J. 2000, 19, 4204–4215. [Google Scholar] [CrossRef] [PubMed]
- Ullah, A.; Coronado, M.; Murakami, M.T.; Betzel, C.; Arni, R.K. Crystallization and preliminary X-ray diffraction analysis of an l-amino-acid oxidase from Bothrops jararacussu venom. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2012, 68(Pt. 2), 211–213. [Google Scholar] [PubMed]
- Ullah, A.; Masood, R.; Spencer, P.J.; Murakami, M.T.; Arni, R.K. Crystallization and preliminary X-ray diffraction studies of an l-amino-acid oxidase from Lachesis muta venom. Acta Crystallogr. F Struct. Biol. Commun. 2014, 70(Pt. 11), 1556–1559. [Google Scholar] [CrossRef] [PubMed]
- Curti, B.; Massey, V.; Zmudka, M. Inactivation of snake venom l-amino acid oxidase by freezing. J. Biol. Chem. 1968, 243, 2306–2314. [Google Scholar] [PubMed]
- Molinier-Frenkel, V.; Mestivier, D.; Castellano, F. Alterations of the immunosuppressive IL4I1 enzyme activity induced by naturally occurring SNP/mutations. Genes Immun. 2016, 17, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Geyer, A.; Fitzpatrick, T.B.; Pawelek, P.D.; Kitzing, K.; Vrielink, A.; Ghisla, S.; Macheroux, P. Structure and characterization of the glycan moiety of l-amino-acid oxidase from the Malayan pit viper Calloselasma rhodostoma. Eur. J. Biochem. 2001, 268, 4044–4053. [Google Scholar] [CrossRef] [PubMed]
- Bregge-Silva, C.; Nonato, M.C.; de Albuquerque, S.; Ho, P.L.; Junqueira de Azevedo, I.L.; Vasconcelos Diniz, M.R.; Lomonte, B.; Rucavado, A.; Diaz, C.; Gutierrez, J.M.; et al. Isolation and biochemical, functional and structural characterization of a novel l-amino acid oxidase from Lachesis muta snake venom. Toxicon 2012, 60, 1263–1276. [Google Scholar] [CrossRef] [PubMed]
- Torii, S.; Yamane, K.; Mashima, T.; Haga, N.; Yamamoto, K.; Fox, J.W.; Naito, M.; Tsuruo, T. Molecular cloning and functional analysis of apoxin I, a snake venom-derived apoptosis-inducing factor with l-amino acid oxidase activity. Biochemistry 2000, 39, 3197–3205. [Google Scholar] [CrossRef] [PubMed]
- Torii, S.; Naito, M.; Tsuruo, T. Apoxin I, a novel apoptosis-inducing factor with l-amino acid oxidase activity purified from Western diamondback rattlesnake venom. J. Biol. Chem. 1997, 272, 9539–9542. [Google Scholar] [CrossRef] [PubMed]
- Boulland, M.L.; Marquet, J.; Molinier-Frenkel, V.; Moller, P.; Guiter, C.; Lasoudris, F.; Copie-Bergman, C.; Baia, M.; Gaulard, P.; Leroy, K.; et al. Human IL4I1 is a secreted l-phenylalanine oxidase expressed by mature dendritic cells that inhibits T-lymphocyte proliferation. Blood 2007, 110, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Ande, S.R.; Kommoju, P.R.; Draxl, S.; Murkovic, M.; Macheroux, P.; Ghisla, S.; Ferrando-May, E. Mechanisms of cell death induction by l-amino acid oxidase, a major component of ophidian venom. Apoptosis 2006, 11, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- De Kok, A.; Veeger, C. Studies on l-amino-acid oxidase. I. Effects of pH and competitive inhibitors. Biochim. Biophys. Acta 1968, 167, 35–47. [Google Scholar] [CrossRef]
- Bhattacharjee, P.; Bera, I.; Chakraborty, S.; Ghoshal, N.; Bhattacharyya, D. Aristolochic acid and its derivatives as inhibitors of snake venom l-amino acid oxidase. Toxicon 2017, 138, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Bhattacharyya, D. Two l-amino acid oxidase isoenzymes from Russell’s viper (Daboia russelli russelli) venom with different mechanisms of inhibition by substrate analogs. FEBS J. 2008, 275, 2078–2095. [Google Scholar] [CrossRef] [PubMed]
- Mitra, J.; Bhattacharyya, D. Irreversible inactivation of snake venom l-amino acid oxidase by covalent modification during catalysis of l-propargylglycine. FEBS Open Bio 2013, 3, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.O.; Seo, J.W.; Kwon, O.; Seong, S.I.; Kim, I.H.; Kim, C.H. Expression and characterization of a second l-amino acid deaminase isolated from Proteus mirabilis in Escherichia coli. J. Basic Microbiol. 2011, 51, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.; Chen, W.; Shi, W.; Qi, F.; Dong, X. SO-LAAO, a novel l-amino acid oxidase that enables Streptococcus oligofermentans to outcompete Streptococcus mutans by generating H2O2 from peptone. J. Bacteriol. 2008, 190, 4716–4721. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.A.; Askin, M.C.; Hynes, M.J. Amino acid catabolism by an areA-regulated gene encoding an l-amino acid oxidase with broad substrate specificity in Aspergillus nidulans. Appl. Environ. Microbiol. 2005, 71, 3551–3555. [Google Scholar] [CrossRef] [PubMed]
- Nuutinen, J.T.; Timonen, S. Identification of nitrogen mineralization enzymes, l-amino acid oxidases, from the ectomycorrhizal fungi Hebeloma spp. and Laccaria bicolor. Mycol. Res. 2008, 112(Pt. 12), 1453–1464. [Google Scholar] [CrossRef] [PubMed]
- Stasyk, T.; Lutsik-Kordovsky, M.; Wernstedt, C.; Antonyuk, V.; Klyuchivska, O.; Souchelnytskyi, S.; Hellman, U.; Stoika, R. A new highly toxic protein isolated from the death cap Amanita phalloides is an l-amino acid oxidase. FEBS J. 2010, 277, 1260–1269. [Google Scholar] [CrossRef] [PubMed]
- Pislar, A.; Sabotic, J.; Slenc, J.; Brzin, J.; Kos, J. Cytotoxic l-amino-acid oxidases from Amanita phalloides and Clitocybe geotropa induce caspase-dependent apoptosis. Cell Death Discov. 2016, 2, 16021. [Google Scholar] [CrossRef] [PubMed]
- Derby, C.D. Escape by inking and secreting: Marine molluscs avoid predators through a rich array of chemicals and mechanisms. Biol. Bull. 2007, 213, 274–289. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.K.; Mai, A.; Iwamoto, M.; Arizono, N.; Fujimoto, D.; Sakamaki, K.; Yonehara, S. Purification and cloning of an apoptosis-inducing protein derived from fish infected with Anisakis simplex, a causative nematode of human anisakiasis. J. Immunol. 2000, 165, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Wang, J.; Luo, H.L.; Lu, G.L.; Li, Y.W.; Li, A.X. l-amino acid oxidase expression profile and biochemical responses of rabbitfish (Siganus oramin) after exposure to a high dose of Cryptocaryon irritans. Fish Shellfish Immunol. 2017, 69, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Kamio, M.; Ko, K.C.; Zheng, S.; Wang, B.; Collins, S.L.; Gadda, G.; Tai, P.C.; Derby, C.D. The chemistry of escapin: Identification and quantification of the components in the complex mixture generated by an l-amino acid oxidase in the defensive secretion of the sea snail Aplysia californica. Chemistry 2009, 15, 1597–1603. [Google Scholar] [CrossRef] [PubMed]
- Morais, I.C.; Pereira, G.J.; Orzaez, M.; Jorge, R.J.; Bincoletto, C.; Toyama, M.H.; Monteiro, H.S.; Smaili, S.S.; Perez-Paya, E.; Martins, A.M. l-Aminoacid Oxidase from Bothrops leucurus Venom Induces Nephrotoxicity via Apoptosis and Necrosis. PLoS ONE 2015, 10, e0132569. [Google Scholar] [CrossRef] [PubMed]
- Burin, S.M.; Berzoti-Coelho, M.G.; Cominal, J.G.; Ambrosio, L.; Torqueti, M.R.; Sampaio, S.V.; de Castro, F.A. The l-amino acid oxidase from Calloselasma rhodostoma snake venom modulates apoptomiRs expression in Bcr-Abl-positive cell lines. Toxicon 2016, 120, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.K.; Saviola, A.J.; Burns, P.D.; Mackessy, S.P. Apoptosis induction in human breast cancer (MCF-7) cells by a novel venom l-amino acid oxidase (Rusvinoxidase) is independent of its enzymatic activity and is accompanied by caspase-7 activation and reactive oxygen species production. Apoptosis 2015, 20, 1358–1372. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, T.L.; Oliveira Silva, V.A.; da Cunha, D.B.; Polettini, F.L.; Thomaz, C.D.; Pianca, A.A.; Zambom, F.L.; da Silva Leitao Mazzi, D.P.; Reis, R.M.; Mazzi, M.V. Isolation, characterization and screening of the in vitro cytotoxic activity of a novel l-amino acid oxidase (LAAOcdt) from Crotalus durissus terrificus venom on human cancer cell lines. Toxicon 2016, 119, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Costa, T.R.; Menaldo, D.L.; Zoccal, K.F.; Burin, S.M.; Aissa, A.F.; Castro, F.A.; Faccioli, L.H.; Greggi Antunes, L.M.; Sampaio, S.V. CR-LAAO, an l-amino acid oxidase from Calloselasma rhodostoma venom, as a potential tool for developing novel immunotherapeutic strategies against cancer. Sci. Rep. 2017, 7, 42673. [Google Scholar] [CrossRef] [PubMed]
- Pontes, A.S.; da, S.S.S.; Xavier, C.V.; Lacouth-Silva, F.; Kayano, A.M.; Pires, W.L.; Nery, N.M.; Boeri de Castro, O.; da Silva, S.D.; Calderon, L.A.; et al. Effect of l-amino acid oxidase from Calloselasma rhodosthoma snake venom on human neutrophils. Toxicon 2014, 80, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Y.; Yu, T.F.; Lian, E.C. Purification and characterization of l-amino acid oxidase from king cobra (Ophiophagus hannah) venom and its effects on human platelet aggregation. Toxicon 1994, 32, 1349–1358. [Google Scholar] [CrossRef]
- Zhong, S.R.; Jin, Y.; Wu, J.B.; Jia, Y.H.; Xu, G.L.; Wang, G.C.; Xiong, Y.L.; Lu, Q.M. Purification and characterization of a new l-amino acid oxidase from Daboia russellii siamensis venom. Toxicon 2009, 54, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, Y.; Shima, M.; Matsumoto, T.; Takatsuka, H.; Nishiya, K.; Kasuda, S.; Fujimura, Y.; Yoshioka, A. Anticoagulant activity of M-LAO, l-amino acid oxidase purified from Agkistrodon halys blomhoffii, through selective inhibition of factor IX. Biochim. Biophys. Acta 2003, 1649, 51–57. [Google Scholar] [CrossRef]
- Fujisawa, D.; Yamazaki, Y.; Morita, T. Re-evaluation of M-LAO, l-amino acid oxidase, from the venom of Gloydius blomhoffi as an anticoagulant protein. J. Biochem. 2009, 146, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.A.; Stoeva, S.; Abbasi, A.; Alam, J.M.; Kayed, R.; Faigle, M.; Neumeister, B.; Voelter, W. Isolation, structural, and functional characterization of an apoptosis-inducing l-amino acid oxidase from leaf-nosed viper (Eristocophis macmahoni) snake venom. Arch Biochem. Biophys. 2000, 384, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Perumal Samy, R.; Gopalakrishnakone, P.; Thwin, M.M.; Chow, T.K.; Bow, H.; Yap, E.H.; Thong, T.W. Antibacterial activity of snake, scorpion and bee venoms: A comparison with purified venom phospholipase A2 enzymes. J. Appl. Microbiol. 2007, 102, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Liu, S.; Dong, P.; Zhao, D.; Wang, C.; Tao, Z.; Sun, M.Z. Akbu-LAAO exhibits potent anti-tumor activity to HepG2 cells partially through produced H2O2 via TGF-β signal pathway. Sci. Rep. 2015, 5, 18215. [Google Scholar] [CrossRef] [PubMed]
- Skarnes, R.C. l-amino-acid oxidase, a bactericidal system. Nature 1970, 225, 1072–1073. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.K.; Ler, S.G.; Gunaratne, J.; Bay, B.H.; Ponnampalam, G. In vitro cytotoxicity of l-amino acid oxidase from the venom of Crotalus mitchellii pyrrhus. Toxicon 2017, 139, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Aitken, J.B.; Naumovski, N.; Curry, B.; Grupen, C.G.; Gibb, Z.; Aitken, R.J. Characterization of an l-amino acid oxidase in equine spermatozoa. Biol. Reprod. 2015, 92, 125. [Google Scholar] [CrossRef] [PubMed]
- Shannon, P.; Curson, B. Site of aromatic l-amino acid oxidase in dead bovine spermatozoa and determination of between-bull differences in the percentage of dead spermatozoa by oxidase activity. J. Reprod. Fertil. 1982, 64, 469–473. [Google Scholar] [CrossRef] [PubMed]
- Upreti, G.C.; Jensen, K.; Munday, R.; Duganzich, D.M.; Vishwanath, R.; Smith, J.F. Studies on aromatic amino acid oxidase activity in ram spermatozoa: Role of pyruvate as an antioxidant. Anim. Reprod. Sci. 1998, 51, 275–287. [Google Scholar] [CrossRef]
- Murthy, S.N.; Janardanasarma, M.K. Identification of l-amino acid/l-lysine α-amino oxidase in mouse brain. Mol. Cell. Biochem. 1999, 197, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Wiemann, S.; Kolb-Kokocinski, A.; Poustka, A. Alternative pre-mRNA processing regulates cell-type specific expression of the IL4l1 and NUP62 genes. BMC Biol. 2005, 3, 16. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Nonobe, E.; Kobayashi, Y.; Kuraishi, T.; Aoki, F.; Yamamoto, K.; Sakai, S. Characterization and expression of l-amino acid oxidase of mouse milk. J. Biol. Chem. 2002, 277, 19080–19086. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.C.; Paul, W.E. Fig1, an interleukin 4-induced mouse B cell gene isolated by cDNA representational difference analysis. Proc. Natl. Acad. Sci. USA 1997, 94, 2507–2512. [Google Scholar] [CrossRef] [PubMed]
- Nagaoka, K.; Zhang, H.; Arakuni, M.; Taya, K.; Watanabe, G. Low expression of the antibacterial factor l-amino acid oxidase in bovine mammary gland. Anim. Sci. J. 2014, 85, 976–980. [Google Scholar] [CrossRef] [PubMed]
- Castellano, F.; Molinier-Frenkel, V. IL4I1: An Emerging Target to Reinvigorate Antitumor Immune Responses. Immunother. Open Access 2017, 3, 132–136. [Google Scholar] [CrossRef]
- Chavan, S.S.; Tian, W.; Hsueh, K.; Jawaheer, D.; Gregersen, P.K.; Chu, C.C. Characterization of the human homolog of the IL-4 induced gene-1 (Fig1). Biochim. Biophys. Acta 2002, 1576, 70–80. [Google Scholar] [CrossRef]
- Chu, C.C.; Paul, W.E. Expressed genes in interleukin-4 treated B cells identified by cDNA representational difference analysis. Mol. Immunol. 1998, 35, 487–502. [Google Scholar] [CrossRef]
- Aubatin, A.; Sako, N.; Decrouy, X.; Donnadieu, E.; Molinier-Frenkel, V.; Castellano, F. IL4-induced gene 1 is secreted at the immune synapse and modulates TCR activation independently of its enzymatic activity. Eur. J. Immunol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Seddigh, P.; Bracht, T.; Molinier-Frenkel, V.; Castellano, F.; Kniemeyer, O.; Schuster, M.; Weski, J.; Hasenberg, A.; Kraus, A.; Poschet, G.; et al. Quantitative Analysis of Proteome Modulations in Alveolar Epithelial Type II Cells in Response to Pulmonary Aspergillus fumigatus Infection. Mol. Cell. Proteom. 2017. [Google Scholar] [CrossRef] [PubMed]
- Marquet, J.; Lasoudris, F.; Cousin, C.; Puiffe, M.; Martin-Garcia, N.; Baud, V.; Chéreau, F.; Farcet, J.; Molinier-Frenkel, V.; Castellano, F. Dichotomy between factors inducing the immunosuppressive enzyme IL4I1 in B lymphocytes and mononuclear phagocytes. Eur. J. Immunol. 2010, 40, 2557–2568. [Google Scholar] [CrossRef] [PubMed]
- Psachoulia, K.; Chamberlain, K.A.; Heo, D.; Davis, S.E.; Paskus, J.D.; Nanescu, S.E.; Dupree, J.L.; Wynn, T.A.; Huang, J.K. IL4I1 augments CNS remyelination and axonal protection by modulating T cell driven inflammation. Brain 2016, 139, 3121–3136. [Google Scholar] [CrossRef] [PubMed]
- Puiffe, M.L.; Lachaise, I.; Molinier-Frenkel, V.; Castellano, F. Antibacterial Properties of the Mammalian l-Amino Acid Oxidase IL4I1. PLoS ONE 2013, 8, e54589. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Huang, W.; Liang, J.; Guo, J.; Ji, J.; Yao, Y.; Zheng, M.; Cai, Z.; Lu, L.; Wang, J. IL4I1 Is a Novel Regulator of M2 Macrophage Polarization That Can Inhibit T Cell Activation via l-Tryptophan and Arginine Depletion and IL-10 Production. PLoS ONE 2015, 10, e0142979. [Google Scholar] [CrossRef] [PubMed]
- Caron, G.; Le Gallou, S.; Lamy, T.; Tarte, K.; Fest, T. CXCR4 expression functionally discriminates centroblasts versus centrocytes within human germinal center B cells. J. Immunol. 2009, 182, 7595–7602. [Google Scholar] [CrossRef] [PubMed]
- Victora, G.D.; Schwickert, T.A.; Fooksman, D.R.; Kamphorst, A.O.; Meyer-Hermann, M.; Dustin, M.L.; Nussenzweig, M.C. Germinal center dynamics revealed by multiphoton microscopy with a photoactivatable fluorescent reporter. Cell 2010, 143, 592–605. [Google Scholar] [CrossRef] [PubMed]
- Bod, L.; Lengagne, R.; Douguet, L.; Rondeau, E.; Auffray, C.; Molinier-Frenkel, V.; Castellano, F.; Richard, Y.; Prévost-Blondel, A. IL4-induced gene 1: A negative immune checkpoint controlling B cell differentiation and activation. J. Immunol. 2017, in press. [Google Scholar]
- Copie-Bergman, C.; Boulland, M.L.; Dehoulle, C.; Moller, P.; Farcet, J.P.; Dyer, M.J.; Haioun, C.; Romeo, P.H.; Gaulard, P.; Leroy, K. Interleukin 4-induced gene 1 is activated in primary mediastinal large B-cell lymphoma. Blood 2003, 101, 2756–2761. [Google Scholar] [CrossRef] [PubMed]
- Carbonnelle-Puscian, A.; Copie-Bergman, C.; Baia, M.; Martin-Garcia, N.; Allory, Y.; Haioun, C.; Cremades, A.; Abd-Alsamad, I.; Farcet, J.P.; Gaulard, P.; et al. The novel immunosuppressive enzyme IL4I1 is expressed by neoplastic cells of several B-cell lymphomas and by tumor-associated macrophages. Leukemia 2009, 23, 952–960. [Google Scholar] [CrossRef] [PubMed]
- Lasoudris, F.; Cousin, C.; Prevost-Blondel, A.; Martin-Garcia, N.; Abd-Alsamad, I.; Ortonne, N.; Farcet, J.P.; Castellano, F.; Molinier-Frenkel, V. IL4I1: An inhibitor of the CD8(+) antitumor T-cell response in vivo. Eur. J. Immunol. 2011, 41, 1629–1638. [Google Scholar] [CrossRef] [PubMed]
- Bod, L.; Lengagne, R.; Wrobel, L.; Ramspott, J.P.; Kato, M.; Avril, M.F.; Castellano, F.; Molinier-Frenkel, V.; Prevost-Blondel, A. IL4-induced gene 1 promotes tumor growth by shaping the immune microenvironment in melanoma. Oncoimmunology 2017, 6, e1278331. [Google Scholar] [CrossRef] [PubMed]
- Santarlasci, V.; Maggi, L.; Mazzoni, A.; Capone, M.; Querci, V.; Rossi, M.C.; Beltrame, L.; Cavalieri, D.; De Palma, R.; Liotta, F.; et al. IL-4-induced gene 1 maintains high Tob1 expression that contributes to TCR unresponsiveness in human T helper 17 cells. Eur. J. Immunol. 2014, 44, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Santarlasci, V.; Maggi, L.; Capone, M.; Querci, V.; Beltrame, L.; Cavalieri, D.; D’Aiuto, E.; Cimaz, R.; Nebbioso, A.; Liotta, F.; et al. Rarity of human T helper 17 cells is due to retinoic acid orphan receptor-dependent mechanisms that limit their expansion. Immunity 2012, 36, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Scarlata, C.M.; Celse, C.; Pignon, P.; Ayyoub, M.; Valmori, D. Differential expression of the immunosuppressive enzyme IL4I1 induce Aiolos, but not natural Helios, FOXP3 Treg cells. Eur. J. Immunol. 2014, 45, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Cousin, C.; Aubatin, A.; Le Gouvello, S.; Apetoh, L.; Castellano, F.; Molinier-Frenkel, V. The immunosuppressive enzyme IL4I1 promotes FoxP3 regulatory T lymphocyte differentiation. Eur. J. Immunol. 2015, 45, 1772–1782. [Google Scholar] [CrossRef] [PubMed]
- Finak, G.; Bertos, N.; Pepin, F.; Sadekova, S.; Souleimanova, M.; Zhao, H.; Chen, H.; Omeroglu, G.; Meterissian, S.; Omeroglu, A.; et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat. Med. 2008, 14, 518–527. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castellano, F.; Molinier-Frenkel, V. An Overview of l-Amino Acid Oxidase Functions from Bacteria to Mammals: Focus on the Immunoregulatory Phenylalanine Oxidase IL4I1. Molecules 2017, 22, 2151. https://doi.org/10.3390/molecules22122151
Castellano F, Molinier-Frenkel V. An Overview of l-Amino Acid Oxidase Functions from Bacteria to Mammals: Focus on the Immunoregulatory Phenylalanine Oxidase IL4I1. Molecules. 2017; 22(12):2151. https://doi.org/10.3390/molecules22122151
Chicago/Turabian StyleCastellano, Flavia, and Valérie Molinier-Frenkel. 2017. "An Overview of l-Amino Acid Oxidase Functions from Bacteria to Mammals: Focus on the Immunoregulatory Phenylalanine Oxidase IL4I1" Molecules 22, no. 12: 2151. https://doi.org/10.3390/molecules22122151
APA StyleCastellano, F., & Molinier-Frenkel, V. (2017). An Overview of l-Amino Acid Oxidase Functions from Bacteria to Mammals: Focus on the Immunoregulatory Phenylalanine Oxidase IL4I1. Molecules, 22(12), 2151. https://doi.org/10.3390/molecules22122151



