Nitrogen-Doped Carbon Quantum Dots as Fluorescent Probes for Sensitive and Selective Detection of Nitrite
Abstract
1. Introduction
2. Results and Discussion
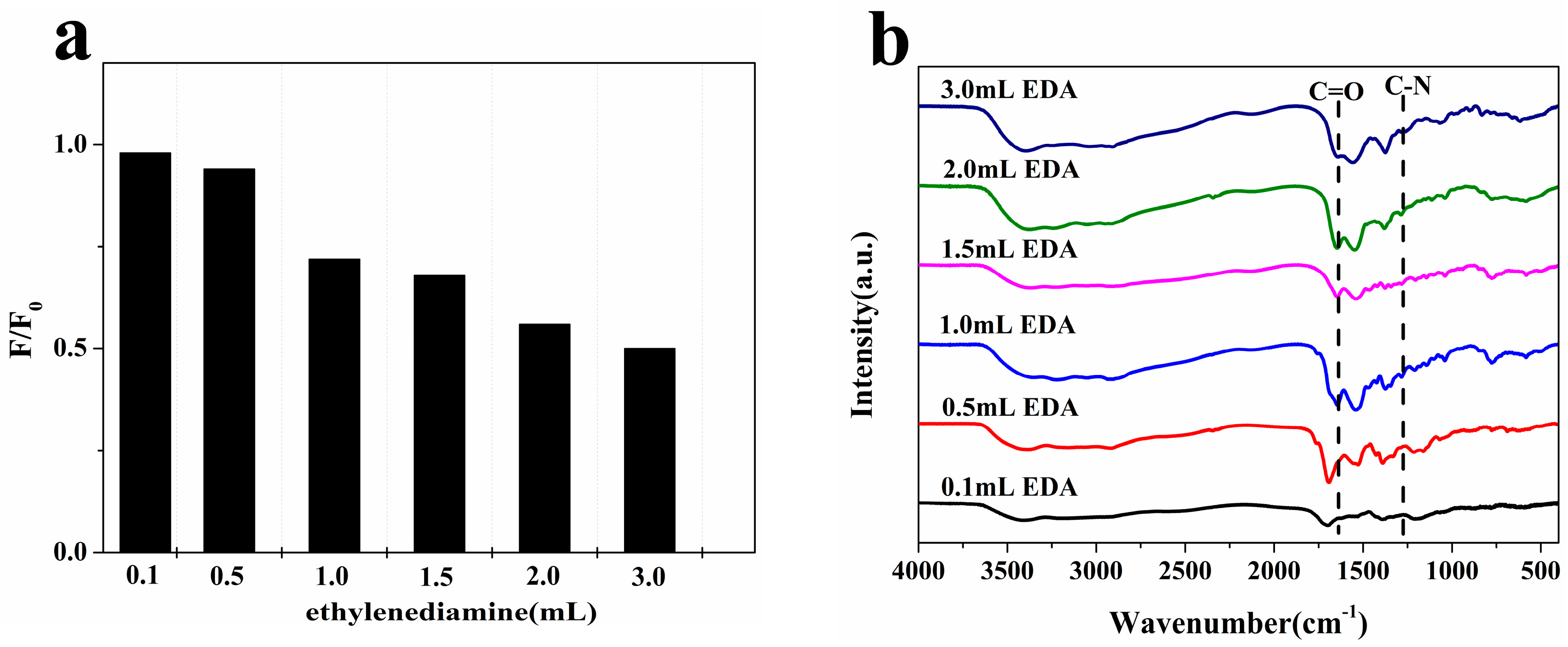
2.1. Optimization of N-CQD Synthesis Conditions
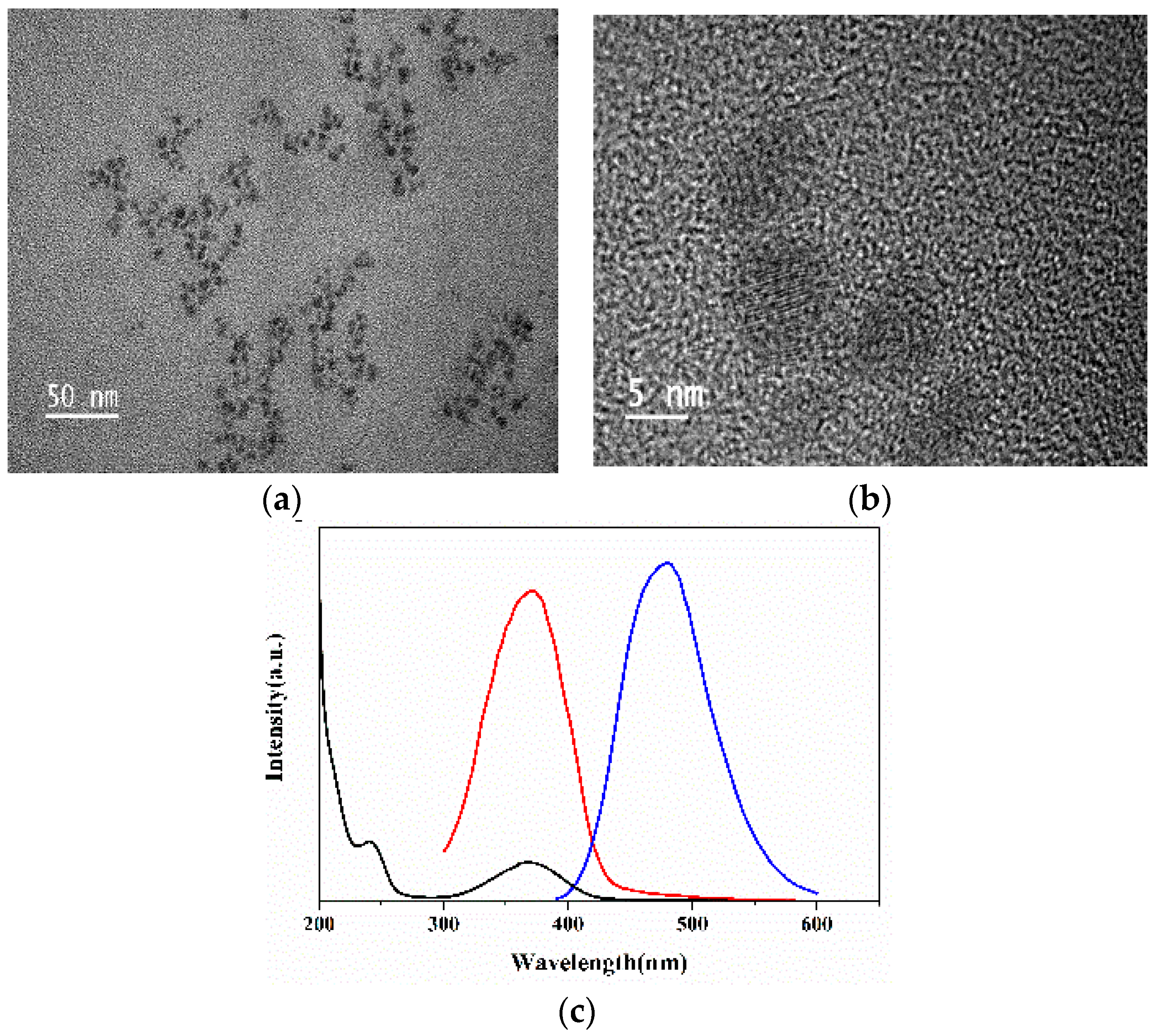
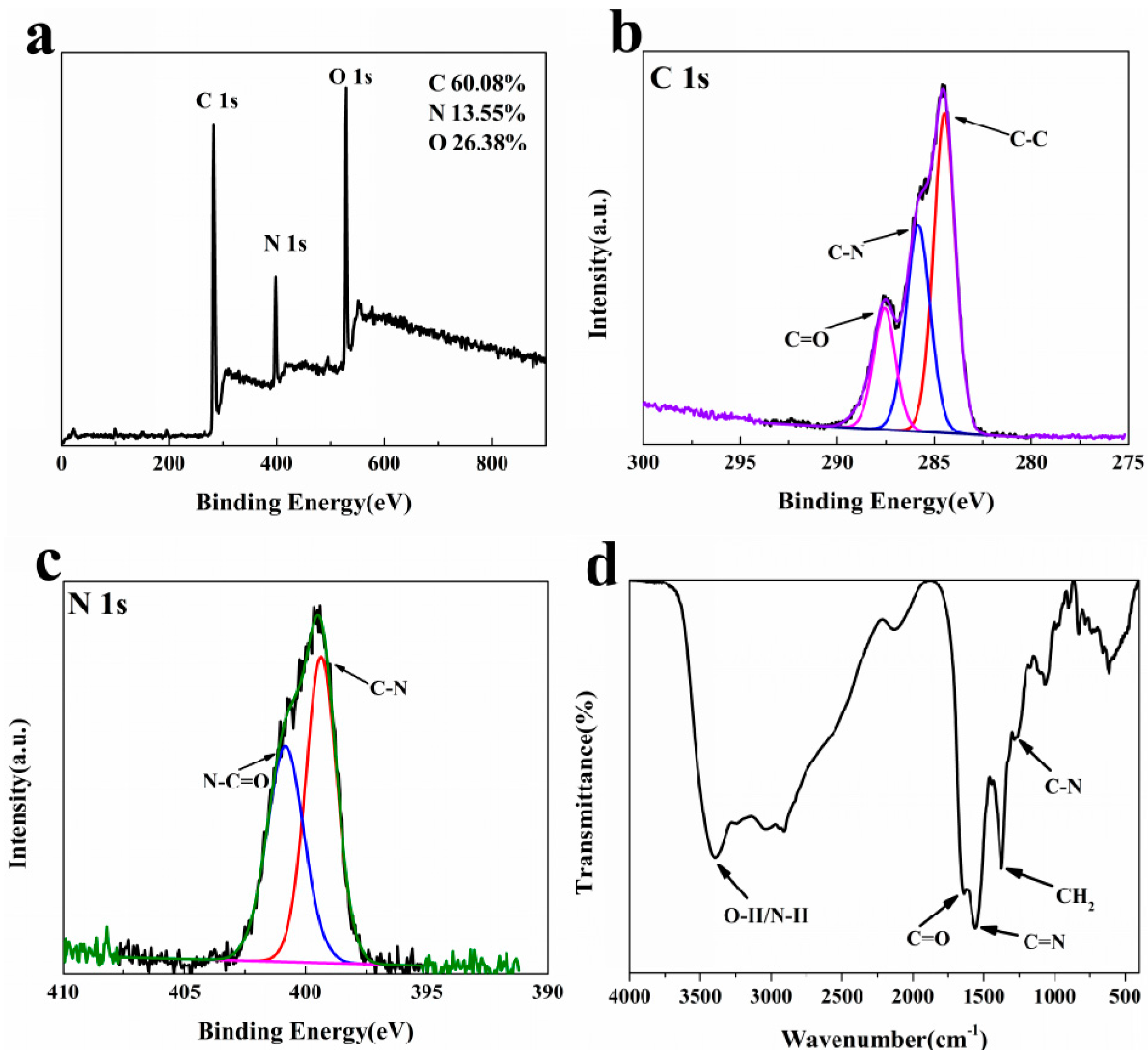
2.2. Characterization of the N-CQDs
2.3. Feasibility of N-CQDs Based Sensor for NO2−
2.3.1. Effects of pH on NO2− Detection and N-CQDs
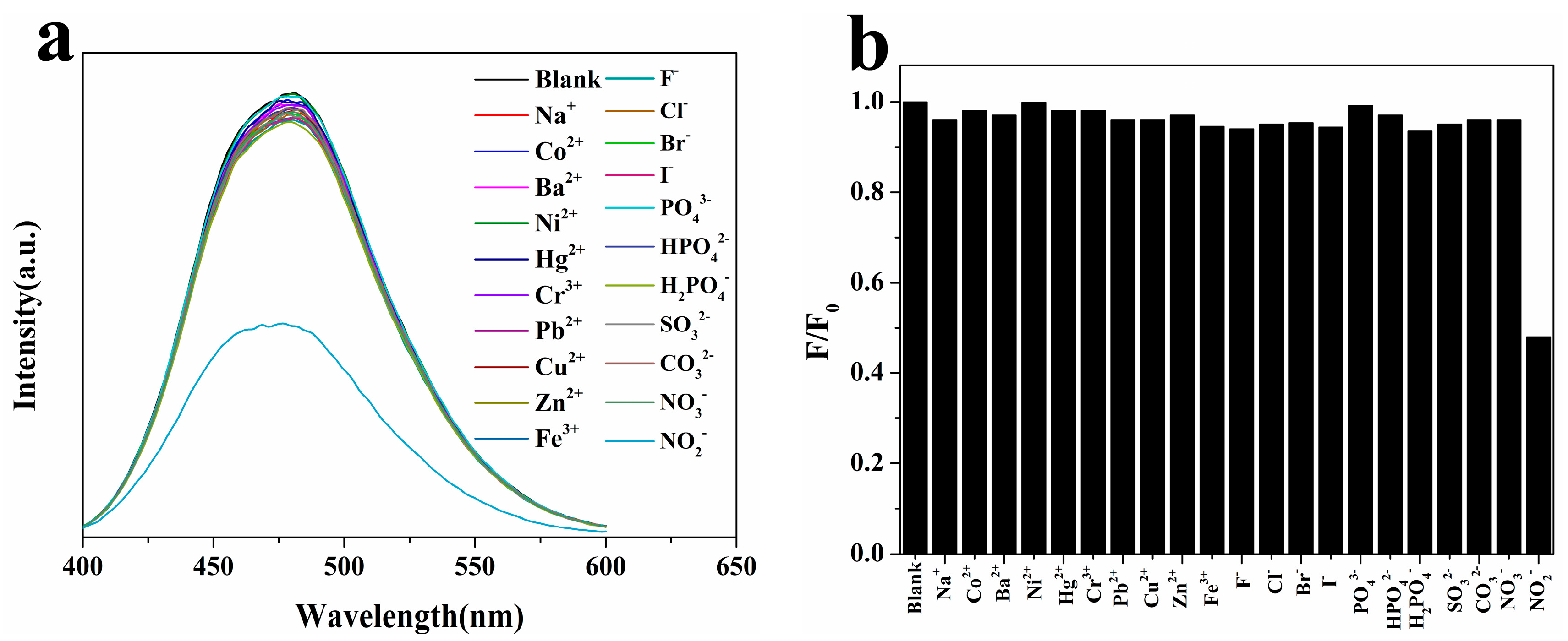
2.3.2. Selectivity of N-CQDs Detection of NO2−
2.3.3. Sensitivity of N-CQD Detection of NO2−
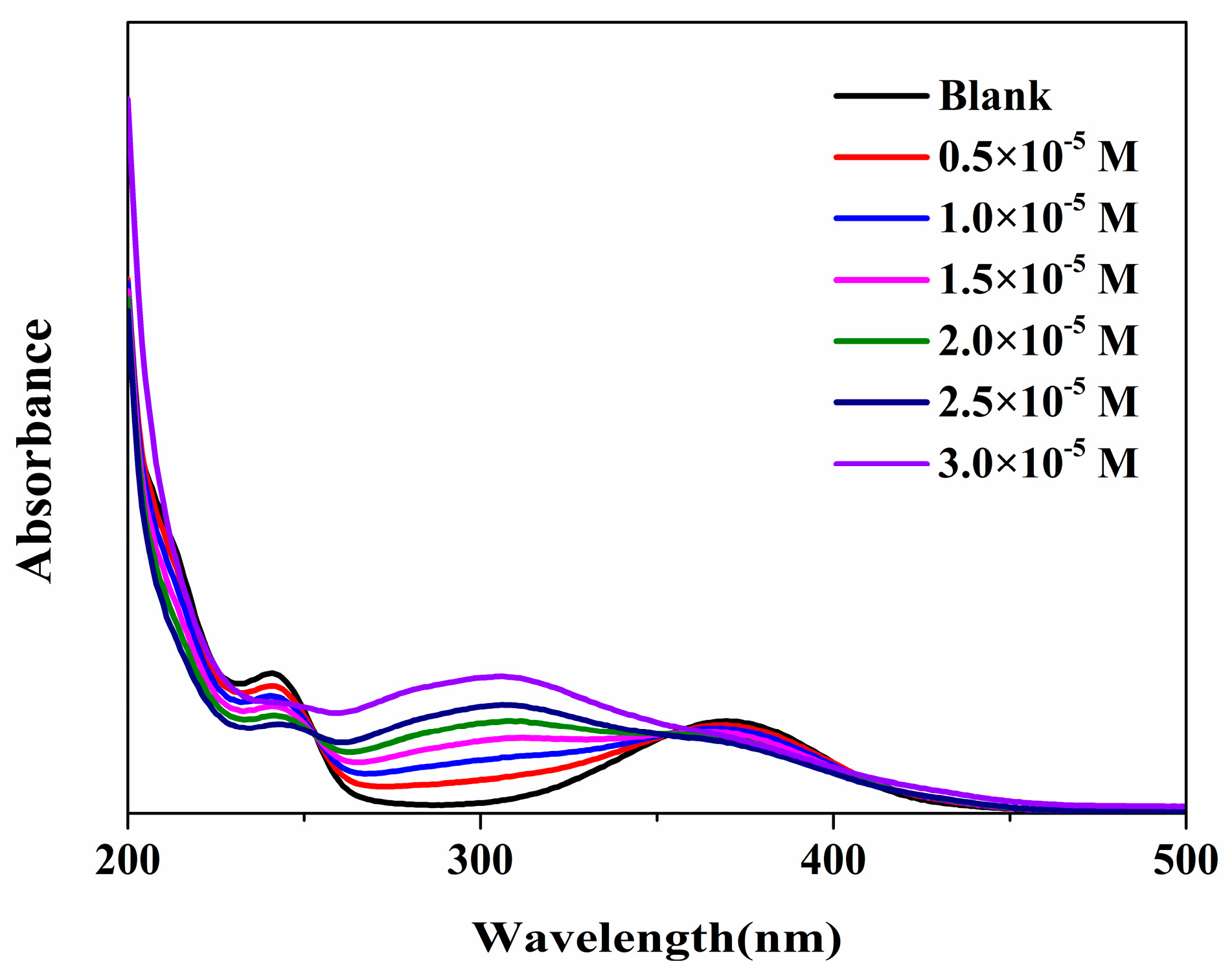
2.3.4. Mechanism of Quenching
2.4. Analytical Applications
3. Materials and Methods
3.1. Chemicals/Reagents
3.2. Apparatus
3.3. Preparation of N-CQDs
3.4. NO2− Determination
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Olajos, E.J.; Coulston, F. Comparative toxicology of n-nitroso compounds and their carcinogenic potential to man. Ecotoxicol. Environ. Saf. 1978, 2, 317–367. [Google Scholar] [CrossRef]
- Fan, A.M. Nitrate and nitrite in drinking water: A toxicological review. In Encyclopedia of Environmental Health; Nriagu, J.O., Ed.; Elsevier: Amsterdam, The Netherlands, 2011; Volume 1, pp. 137–145. [Google Scholar]
- Griess, P. Bemerkungen zu der abhandlung der hh. Weselsky und benedikt & bdquo;ueber einige azoverbindungen & rdquo. Eur. J. Inorg. Chem. 2010, 12, 426–428. [Google Scholar] [CrossRef]
- Cox, R.D.; Frank, C.W. Determination of nitrate and nitrite in blood and urine by chemiluminescence. J. Anal. Toxicol. 1982, 6, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Garside, C. A chemiluminescent technique for the determination of nanomolar concentrations of nitrate and nitrite in seawater. Mar. Chem. 1982, 11, 159–167. [Google Scholar] [CrossRef]
- He, D.; Zhang, Z.; Huang, Y.; Hu, Y. Chemiluminescence microflow injection analysis system on a chip for the determination of nitrite in food. Food Chem. 2007, 101, 667–672. [Google Scholar] [CrossRef]
- Badea, M.; Amine, A.; Palleschi, G.; Moscone, D.; Volpe, G.; Curulli, A. New electrochemical sensors for detection of nitrites and nitrates. J. Electroanal. Chem. 2001, 509, 66–72. [Google Scholar] [CrossRef]
- Zhu, N.; Xu, Q.; Li, S.; Gao, H. Electrochemical determination of nitrite based on poly(amidoamine) dendrimer-modified carbon nanotubes for nitrite oxidation. Electrochem. Commun. 2009, 11, 2308–2311. [Google Scholar] [CrossRef]
- Liu, Q.H.; Yan, X.L.; Guo, J.C.; Wang, D.H.; Lei, L.; Yan, F.Y.; Chen, L.G. Spectrofluorimetric determination of trace nitrite with a novel fluorescent probe. Spectrochim. Acta 2009, 73, 789–793. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M. Resorcinol as fluorimetric reagent for the determination of nitrate. Anal. Lett. 2006, 13, 771–779. [Google Scholar] [CrossRef]
- Rider, B.F.; Mellon, M.G. Colorimetric determination of nitrites. Ind. Eng. Chem. Anal. Ed. 1946, 18, 96–99. [Google Scholar] [CrossRef]
- Armijo, F.; Goya, M.C.; Reina, M.; Canales, M.J.; Arévalo, M.C.; Aguirre, M.J. Electrocatalytic oxidation of nitrite to nitrate mediated by Fe(iii) poly-3-aminophenyl porphyrin grown on five different electrode surfaces. J. Mol. Catal. A 2007, 268, 148–154. [Google Scholar] [CrossRef]
- Milhano, C.; Pletcher, D. The electrodeposition and electrocatalytic properties of copper–palladium alloys. J. Electroanal. Chem. 2008, 614, 24–30. [Google Scholar] [CrossRef]
- Silva, S.M.D.; Mazo, L.H. Differential pulse voltammetric determination of nitrite with gold ultramicroelectrode. Electroanalysis 2015, 10, 1200–1203. [Google Scholar] [CrossRef]
- Cox, R.D. Determination of nitrate and nitrite at the parts per billion level by chemiluminescence. Anal. Chem. 1980, 52, 332–335. [Google Scholar] [CrossRef]
- Lin, Z.; Xue, W.; Chen, H.; Lin, J.M. Peroxynitrous-acid-induced chemiluminescence of fluorescent carbon dots for nitrite sensing. Anal. Chem. 2011, 83, 8245–8251. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.H.; Yu, L.J.; Liu, Y.; Lin, L.; Lu, R.G.; Zhu, J.P.; He, L.; Lu, Z.L. Methods for the detection and determination of nitrite and nitrate: A review. Talanta 2017, 165, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, H.D.; Engel, N.A. Fluorometric determination of subnanogram levels of nitrite using 5-aminofluorescein. Anal. Chem. 1975, 47, 922–924. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.H.; Geng, X.; Zhang, L.; Huang, Z.M.; Ge, J.; Li, Z.H. Nitrogen-doped carbon dots mediated fluorescent on-off assay for rapid and highly sensitive pyrophosphate and alkaline phosphatase detection. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ray, R.; Gu, Y.; Ploehn, H.J.; Gearheart, L.; Raker, K.; Scrivens, W.A. Electrophoretic analysis and purification of fluorescent single-walled carbon nanotube fragments. J. Am. Chem. Soc. 2004, 126, 12736–12737. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Guo, S.; Li, D.; Zhu, C.; Ren, W.; Dong, S.; Wang, E. Easy synthesis and imaging applications of cross-linked green fluorescent hollow carbon nanoparticles. ACS Nano 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Kang, Z.; Liu, Y.; Lee, S.T. Carbon nanodots: Synthesis, properties and applications. J. Mater. Chem. 2012, 22, 24230–24253. [Google Scholar] [CrossRef]
- Kong, B.; Zhu, A.; Ding, C.; Zhao, X.; Li, B.; Tian, Y. Carbon dot-based inorganic-organic nanosystem for two-photon imaging and biosensing of pH variation in living cells and tissues. Adv. Mater. 2012, 24. [Google Scholar] [CrossRef] [PubMed]
- Qiang, Q.; Zhu, A.; Shao, X.; Shi, G.; Yang, T. Development of a carbon quantum dots-based fluorescent Cu2+ probe suitable for living cell imaging. Chem. Commun. 2012, 48, 5473–5475. [Google Scholar] [CrossRef]
- Yang, S.T.; Cao, L.; Luo, P.G.; Lu, F.; Wang, X.; Wang, H.; Meziani, M.J.; Liu, Y.; Qi, G.; Sun, Y.P. Carbon dots for optical imaging in vivo. J. Am. Chem. Soc. 2009, 131. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.X.; Liu, L.Q.; Liu, Z.D.; Wang, Y.; Zhao, X.J.; Huang, C.Z. Highly selective detection of phosphate in very complicated matrixes with an off-on fluorescent probe of europium-adjusted carbon dots. Chem. Commun. 2011, 47, 2604–2606. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhang, Y.; Wang, L.; Tian, J.; Sun, X. Nucleic acid detection using carbon nanoparticles as a fluorescent sensing platform. Chem. Commun. 2010, 47, 961–963. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Zhang, L.; Zhang, S.; Yang, Y.; Chen, X.; Zhang, M. Fluorescent carbon nanoparticles for the fluorescent detection of metal ions. Biosens. Bioelectron. 2015, 63. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cao, L.; Lu, F.; Meziani, M.J.; Li, H.; Qi, G.; Zhou, B.; Harruff, B.A.; Kermarrec, F.; Sun, Y.P. Photoinduced electron transfers with carbon dots. Chem. Commun. 2009, 46. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Lin, Y.; Huang, Z.; Ren, J.; Qu, X. Carbon nanodots as fluorescence probes for rapid, sensitive, and label-free detection of Hg2+ and biothiols in complex matrices. Chem. Commun. 2012, 48. [Google Scholar] [CrossRef] [PubMed]
- Doroodmand, M.M.; Askari, M. Synthesis of a novel nitrogen-doped carbon dot by microwave-assisted carbonization method and its applications as selective probes for optical ph (acidity) sensing in aqueous/nonaqueous media, determination of nitrate/nitrite, and optical recognition of nox gas. Anal. Chim. Acta 2017, 968, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Zhai, X.; Zhang, P.; Liu, C.; Bai, T.; Li, W.; Dai, L.; Liu, W. Highly luminescent carbon nanodots by microwave-assisted pyrolysis. Chem. Commun. 2012, 48. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yang, J.; Tian, J.; Yu, J.S. How do nitrogen-doped carbon dots generate from molecular precursors? An investigation of formation mechanism and a solution-based large-scale synthesis. J. Mater. Chem. B 2015, 3, 5608–5614. [Google Scholar] [CrossRef]
- Zhai, Y.; Zhu, Z.; Zhu, C.; Ren, J.; Wang, E.; Dong, S. Multifunctional water-soluble luminescent carbon dots for imaging and Hg2+ sensing. J. Mater. Chem. B 2014, 2, 6995–6999. [Google Scholar] [CrossRef]
- Cai, Q.Y.; Li, J.; Ge, J.; Zhang, L.; Hu, Y.L.; Li, Z.H.; Qu, L.B. A rapid fluorescence “switch-on” assay for glutathione detection by using carbon dots-MnO2 nanocomposites. Biosens. Bioelectron. 2015, 72, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Qu, S.; Wang, X.; Lu, Q.; Liu, X.; Wang, L. A biocompatible fluorescent ink based on water-soluble luminescent carbon nanodots. Angew. Chem. 2012, 51. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.Q.; Pang, H.C.; Yang, H.B.; Guo, C.X.; Shao, J.W.; Chi, Y.W.; Li, C.M.; Yu, T. Carbon-based dots co-doped with nitrogen and sulfur for high quantum yield and excitation-independent emission. Angew. Chem. Int. Ed. Engl. 2013, 52, 7800–7804. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Iijima, K.; Moriya, A.; Mcelroy, K.; Scobie, G.; Fyfe, V.; Mccoll, K.E. Conditions for acid catalysed luminal nitrosation are maximal at the gastric cardia. Gut 2003, 52. [Google Scholar] [CrossRef]
- Zhu, S.; Meng, Q.; Wang, L.; Zhang, J.; Song, Y.; Jin, H.; Zhang, K.; Sun, H.; Wang, H.; Yang, B. Highly photoluminescent carbon dots for multicolor patterning, sensors, and bioimaging. Angew. Chem. 2013, 52. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Dou, X.; Li, H.; Ma, Y.; Lin, J.M. Nitrite sensing based on the carbon dots-enhanced chemiluminescence from peroxynitrous acid and carbonate. Talanta 2015, 132, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.B.; March, J. March’s Advanced Organic Chemistry; John Wiley and Sons: New York, NY, USA, 2001. [Google Scholar]
- Eftink, M.R. Fluorescence Quenching: Theory and Applications. In Topics in Fluorescence Spectroscopy; Lakowicz, J.R., Ed.; Springer: Boston, MA, USA, 2002; Volume 2, pp. 53–126. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |

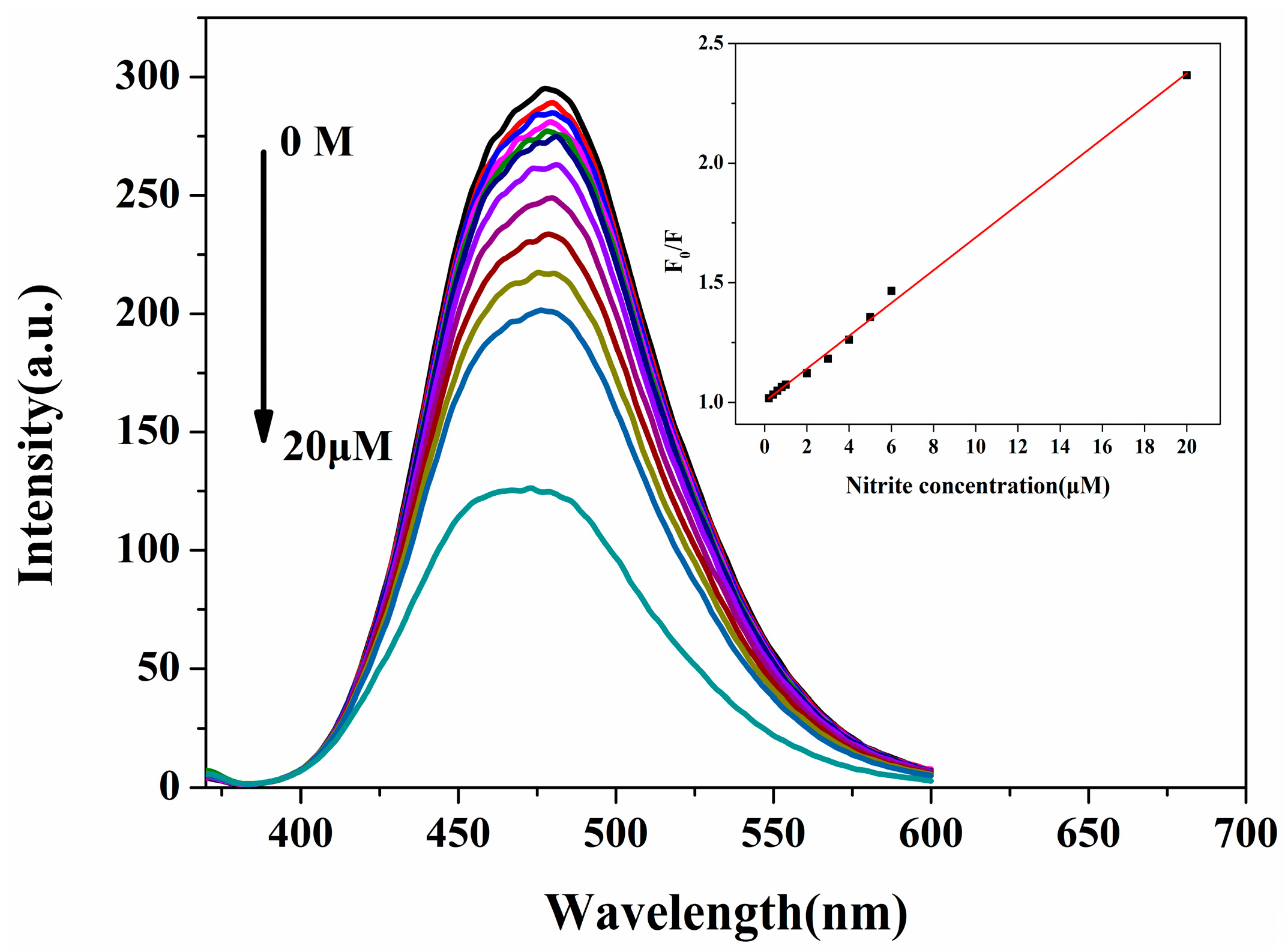
| Fluorescent Probe | LOD (nM) | Linear Range (μM) | Ref. |
|---|---|---|---|
| CQDs-NaNO2-H2O2 | 53 | 0.1–10 | [16] |
| CQDs-NaNO2-H2O2-Na2CO3 | 5 | 0.1–10 | [40] |
| N-CQDs | 25 | 0.1–75 | [31] |
| N-CQDs | 40 | 0.2–20 | [This work] |
| Added/μM | Found/μM | Recovery/% |
|---|---|---|
| 1.00 | 1.03 | 103 |
| 2.00 | 2.08 | 104 |
| 3.00 | 2.96 | 98.7 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, Z.; Li, Z.; Zhang, X.; Shi, Y.; Zhou, N. Nitrogen-Doped Carbon Quantum Dots as Fluorescent Probes for Sensitive and Selective Detection of Nitrite. Molecules 2017, 22, 2061. https://doi.org/10.3390/molecules22122061
Feng Z, Li Z, Zhang X, Shi Y, Zhou N. Nitrogen-Doped Carbon Quantum Dots as Fluorescent Probes for Sensitive and Selective Detection of Nitrite. Molecules. 2017; 22(12):2061. https://doi.org/10.3390/molecules22122061
Chicago/Turabian StyleFeng, Zhibiao, Zeliang Li, Xingwei Zhang, Yanping Shi, and Nan Zhou. 2017. "Nitrogen-Doped Carbon Quantum Dots as Fluorescent Probes for Sensitive and Selective Detection of Nitrite" Molecules 22, no. 12: 2061. https://doi.org/10.3390/molecules22122061
APA StyleFeng, Z., Li, Z., Zhang, X., Shi, Y., & Zhou, N. (2017). Nitrogen-Doped Carbon Quantum Dots as Fluorescent Probes for Sensitive and Selective Detection of Nitrite. Molecules, 22(12), 2061. https://doi.org/10.3390/molecules22122061







