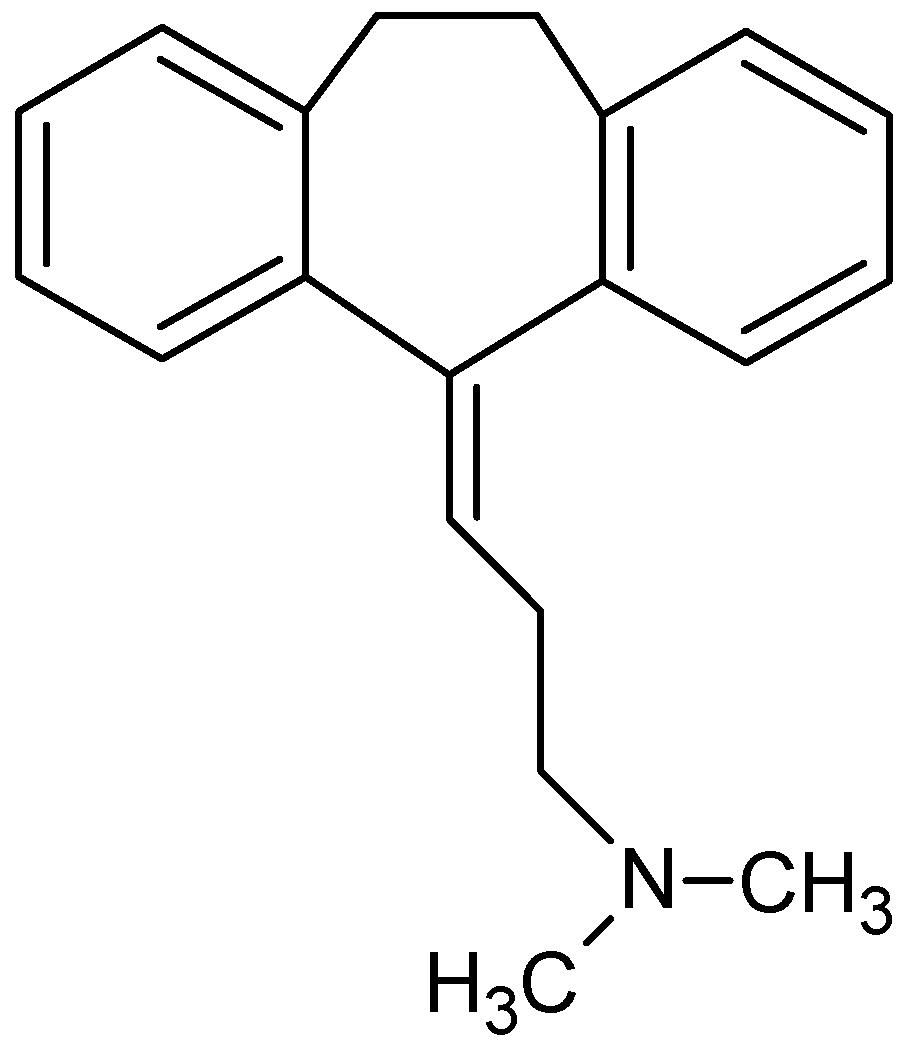
Direct Modification of Microcrystalline Cellulose with Ethylenediamine for Use as Adsorbent for Removal Amitriptyline Drug from Environment
Abstract
:1. Introduction
2. Results and Discussion
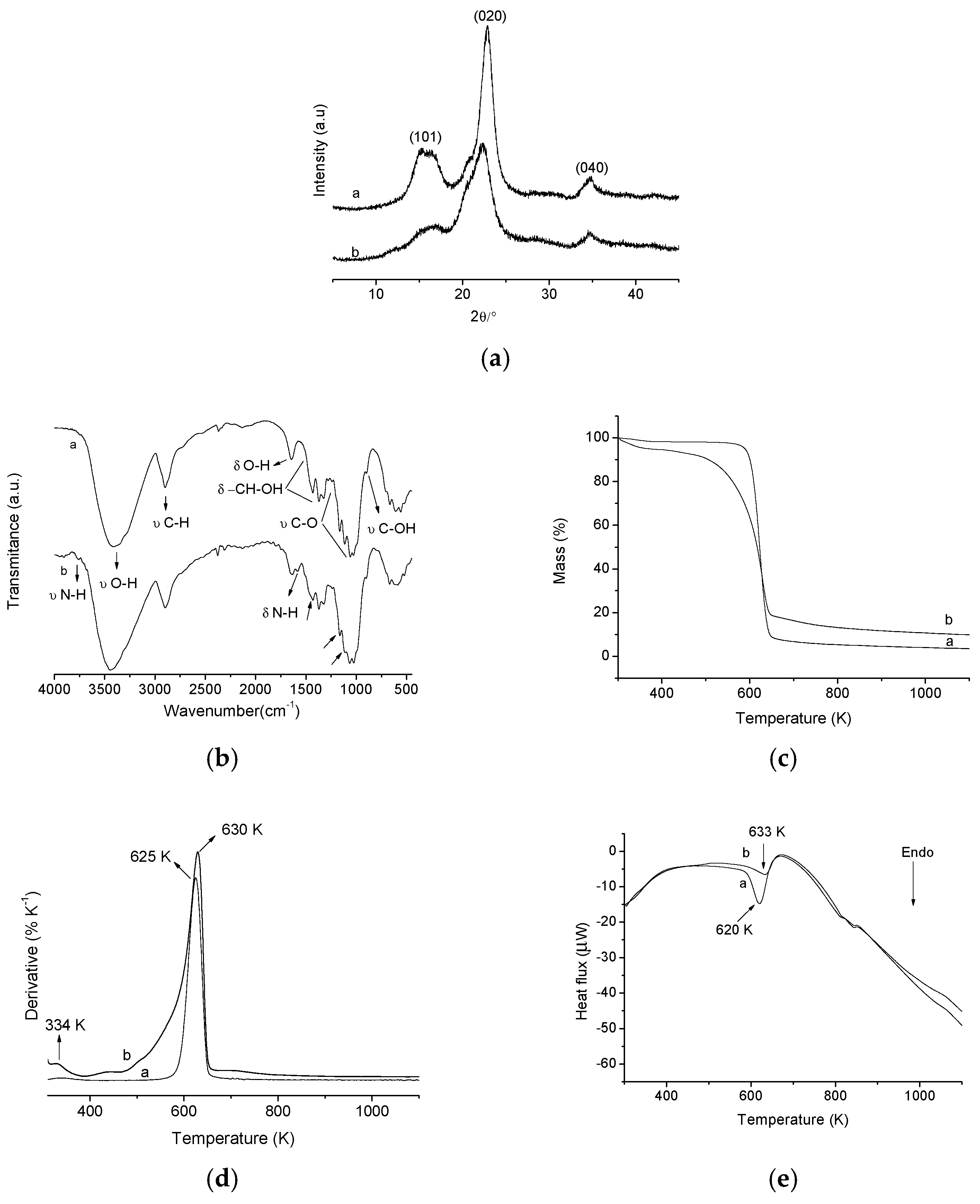
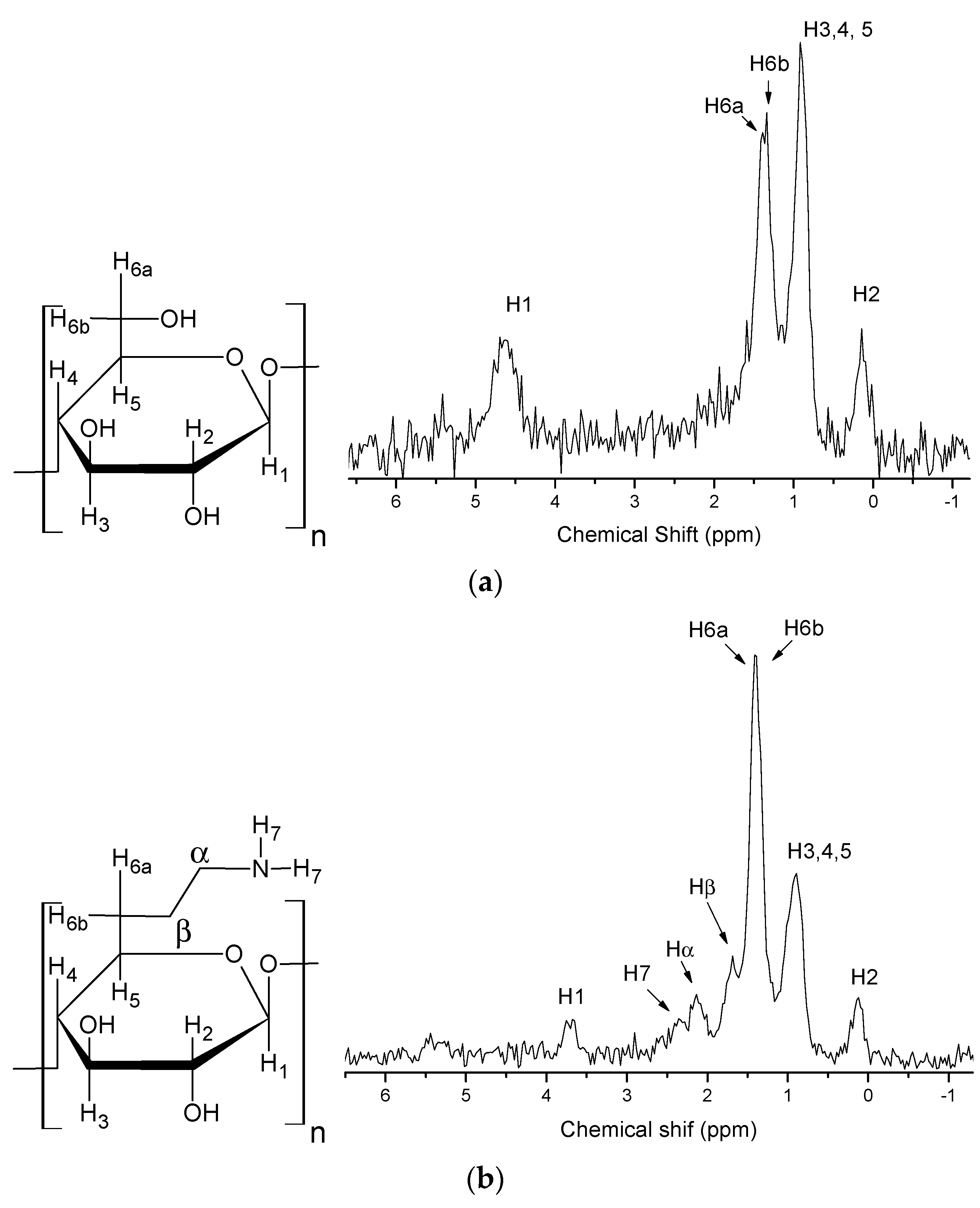
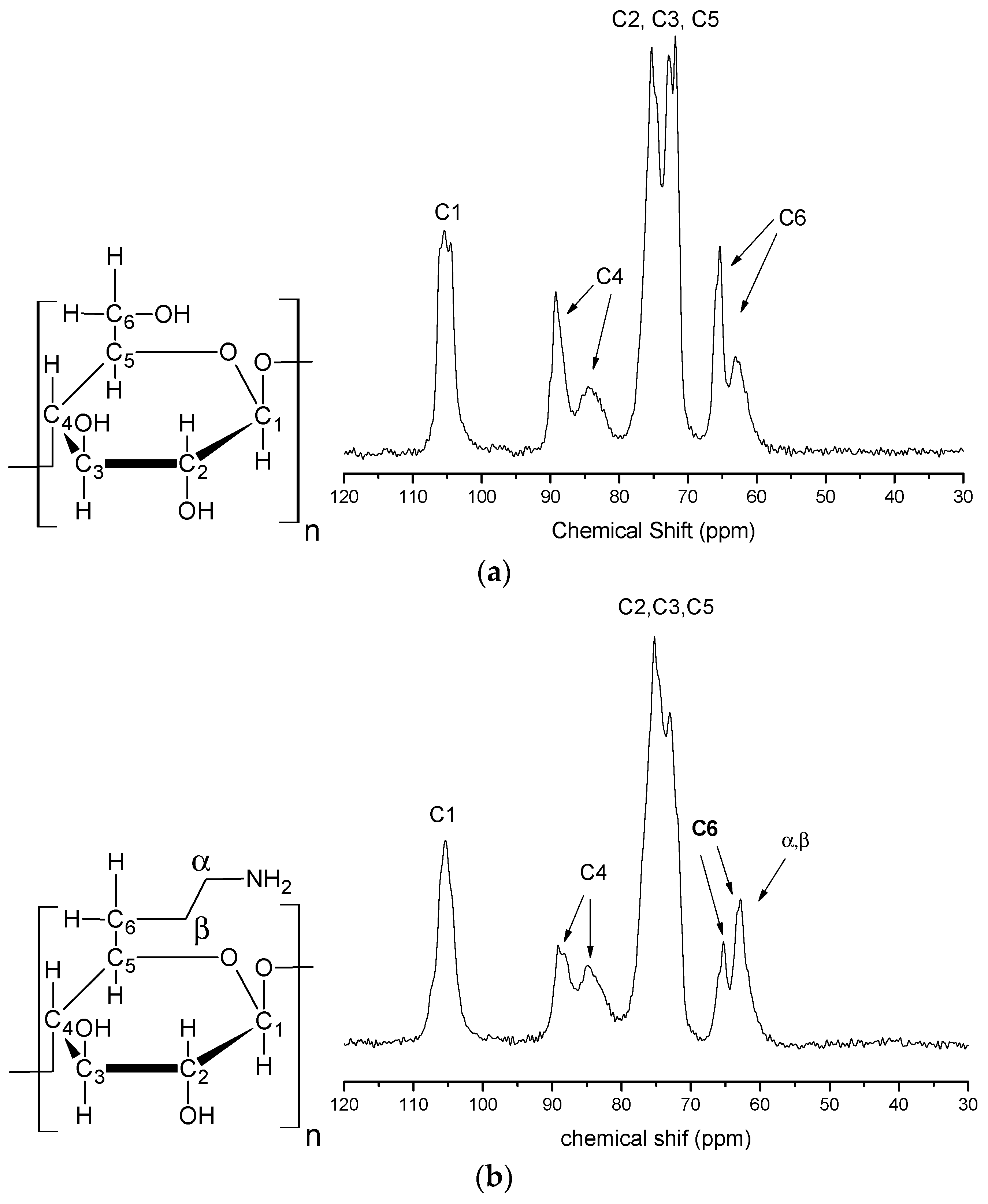
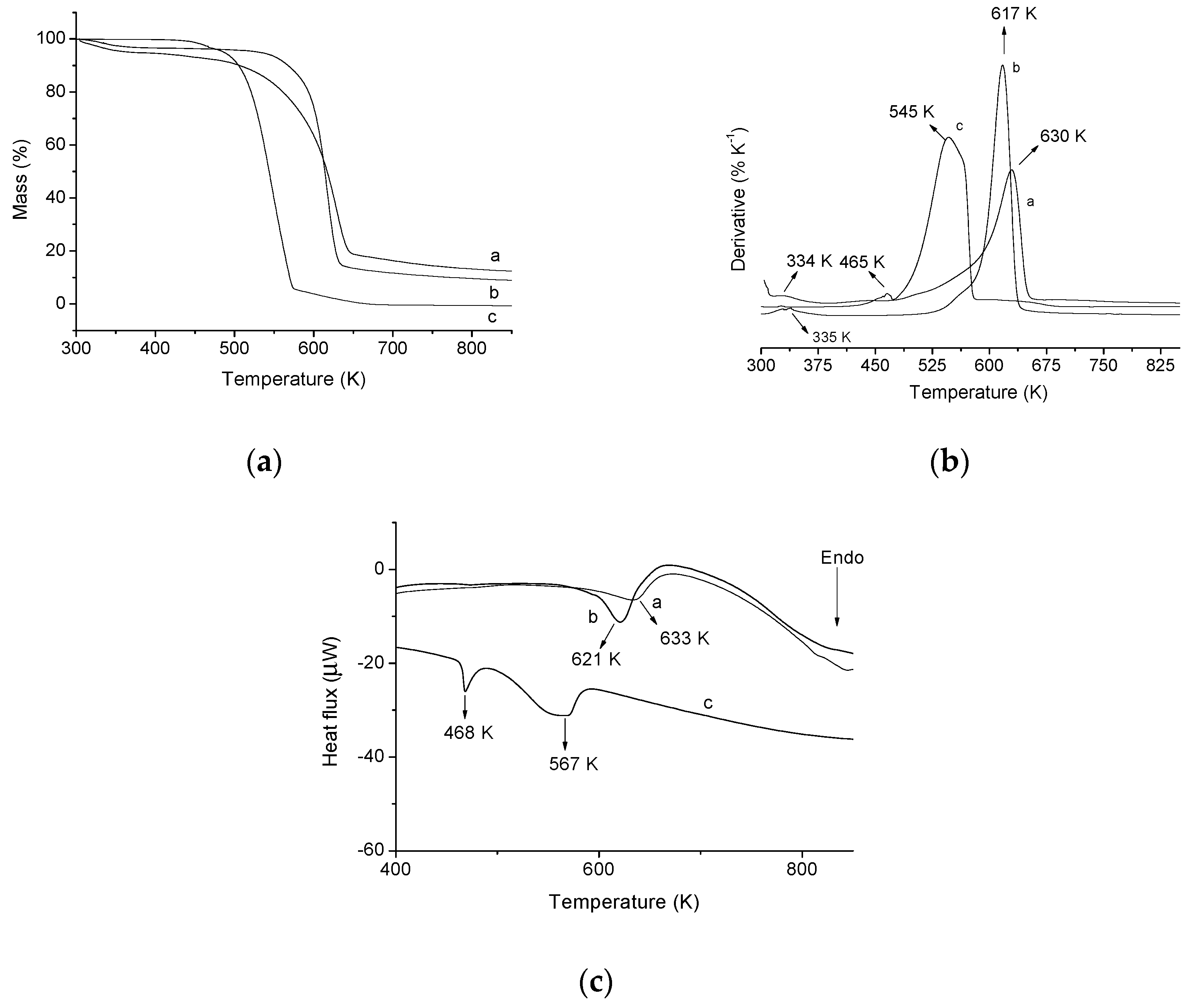
2.1. Product Characterization
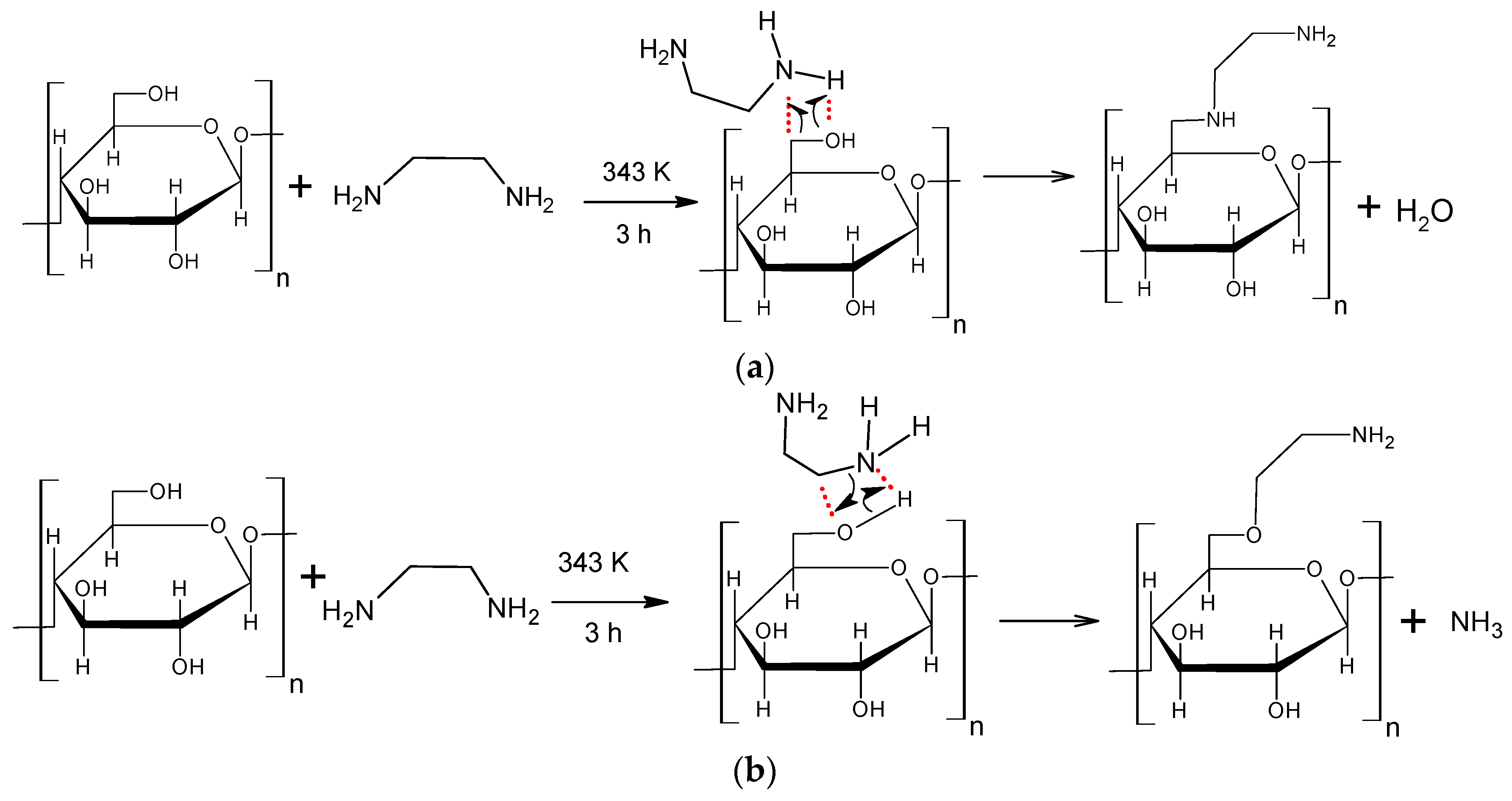
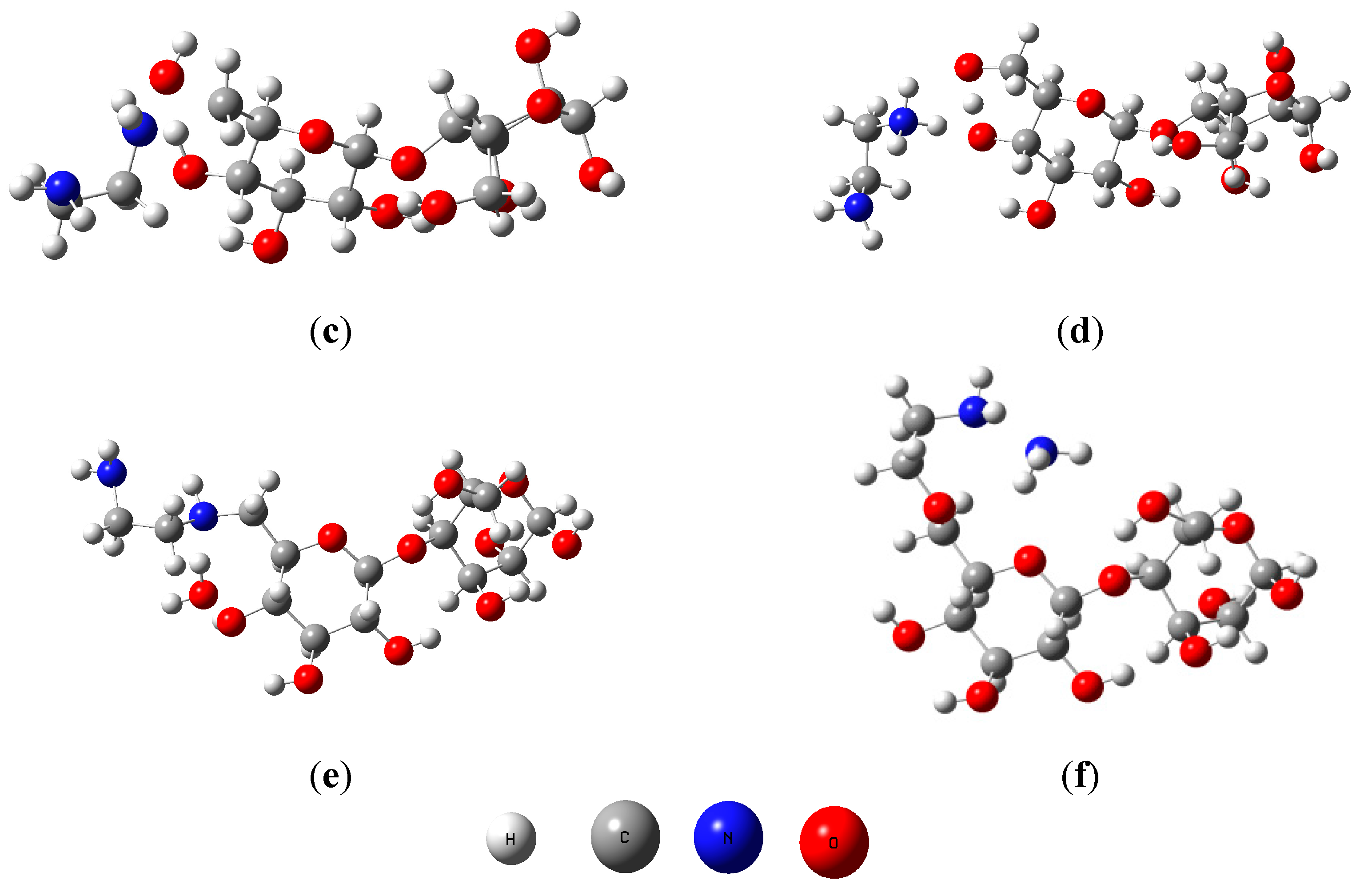
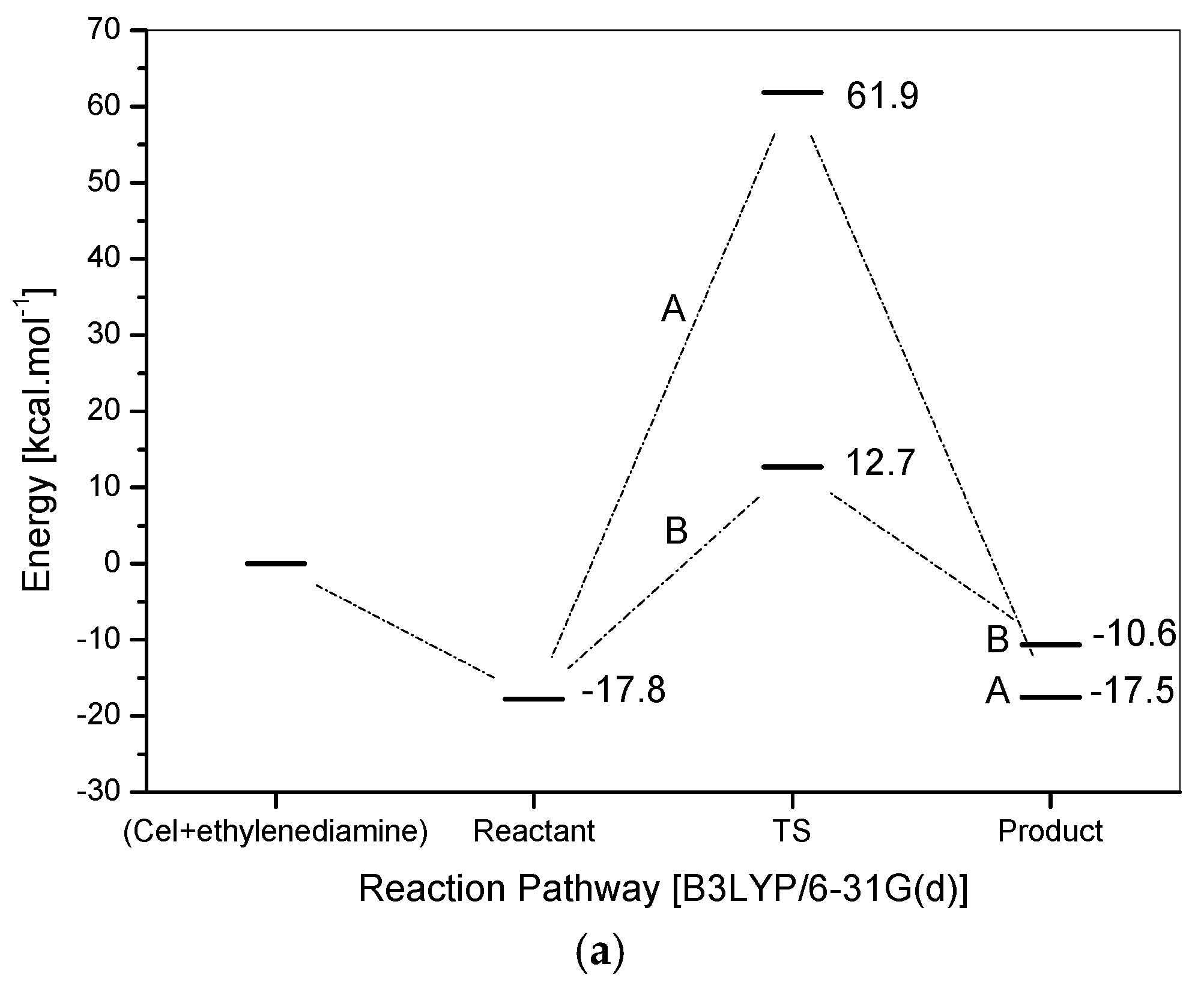
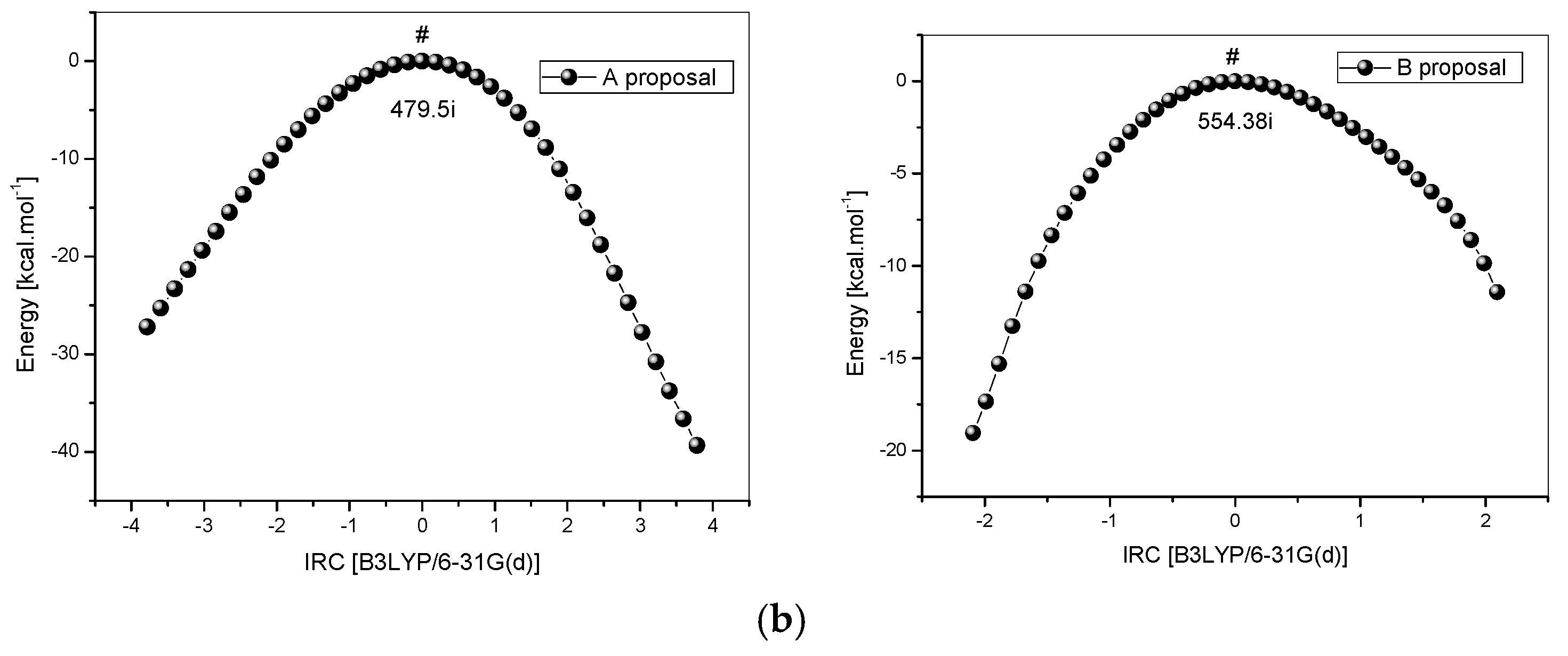
2.2. Reaction Mechanism and Theoretical Calculations
2.3. Adsorption Test
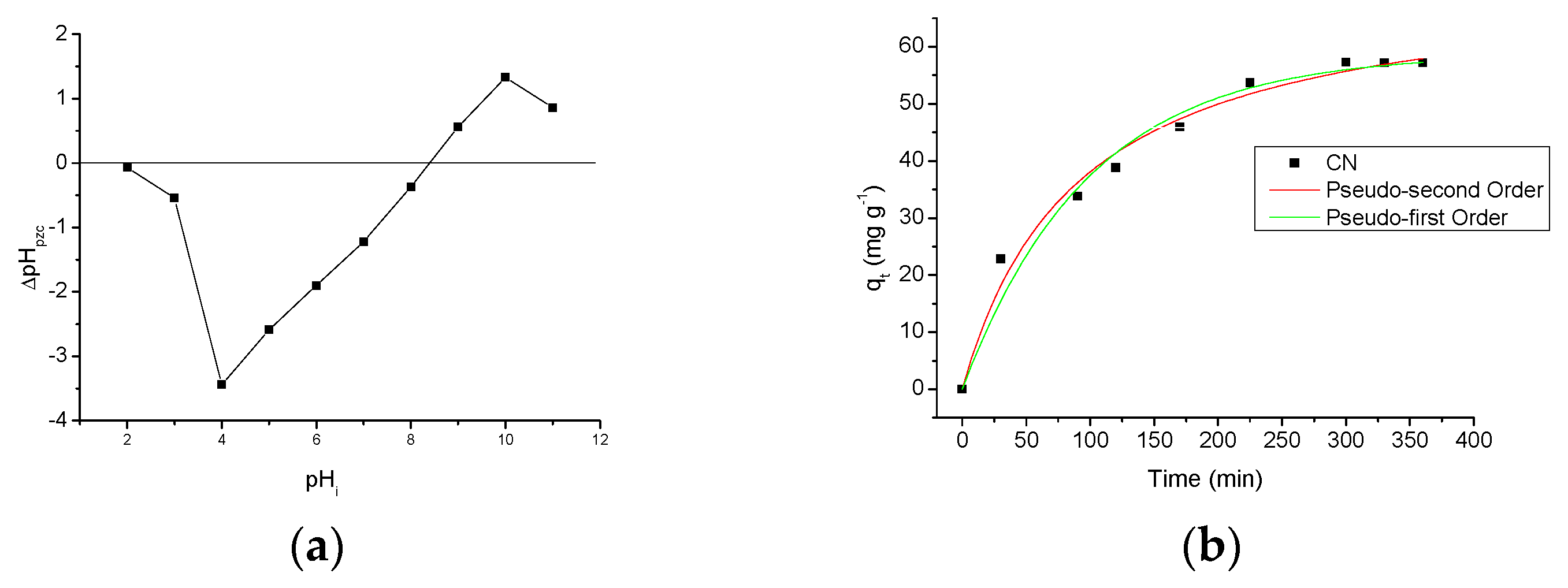
2.3.1. Point of Zero Charge (pHpzc)
2.3.2. Time Effect
2.3.3. Effect of pH
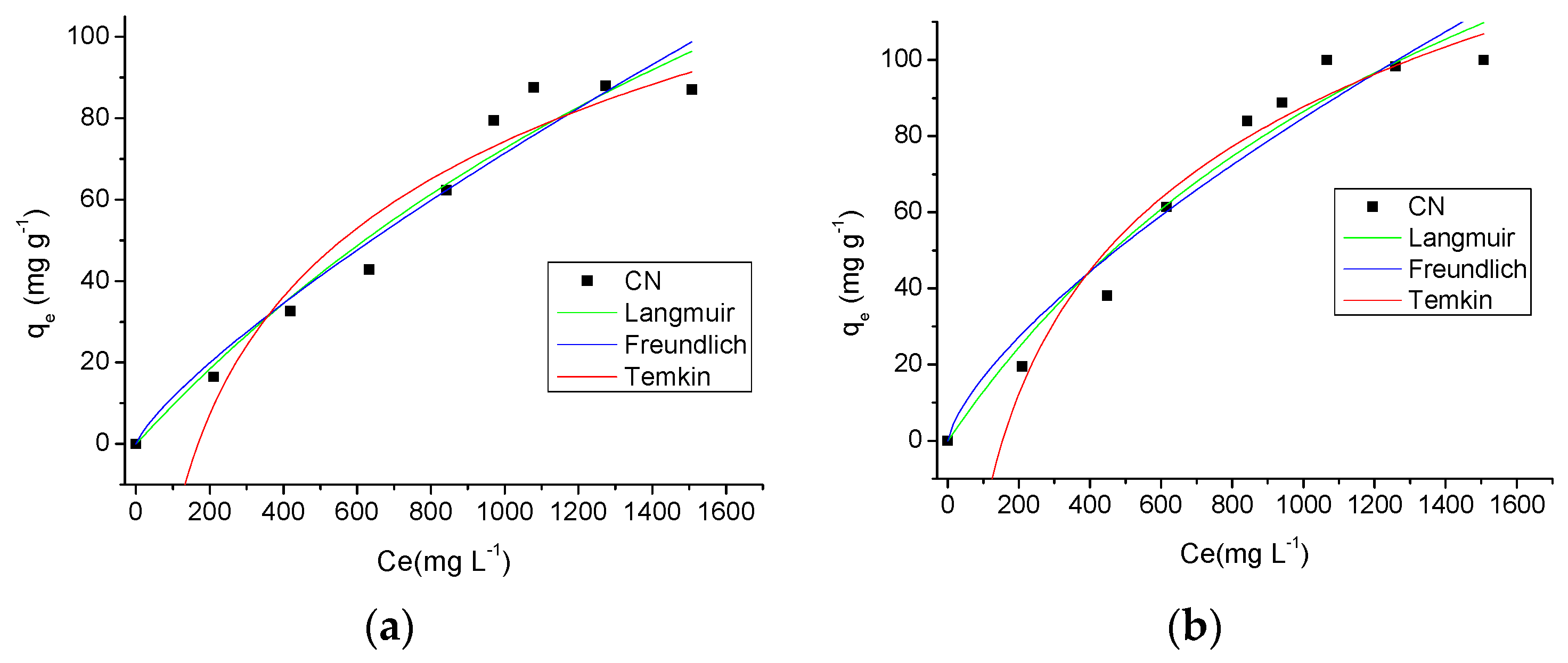
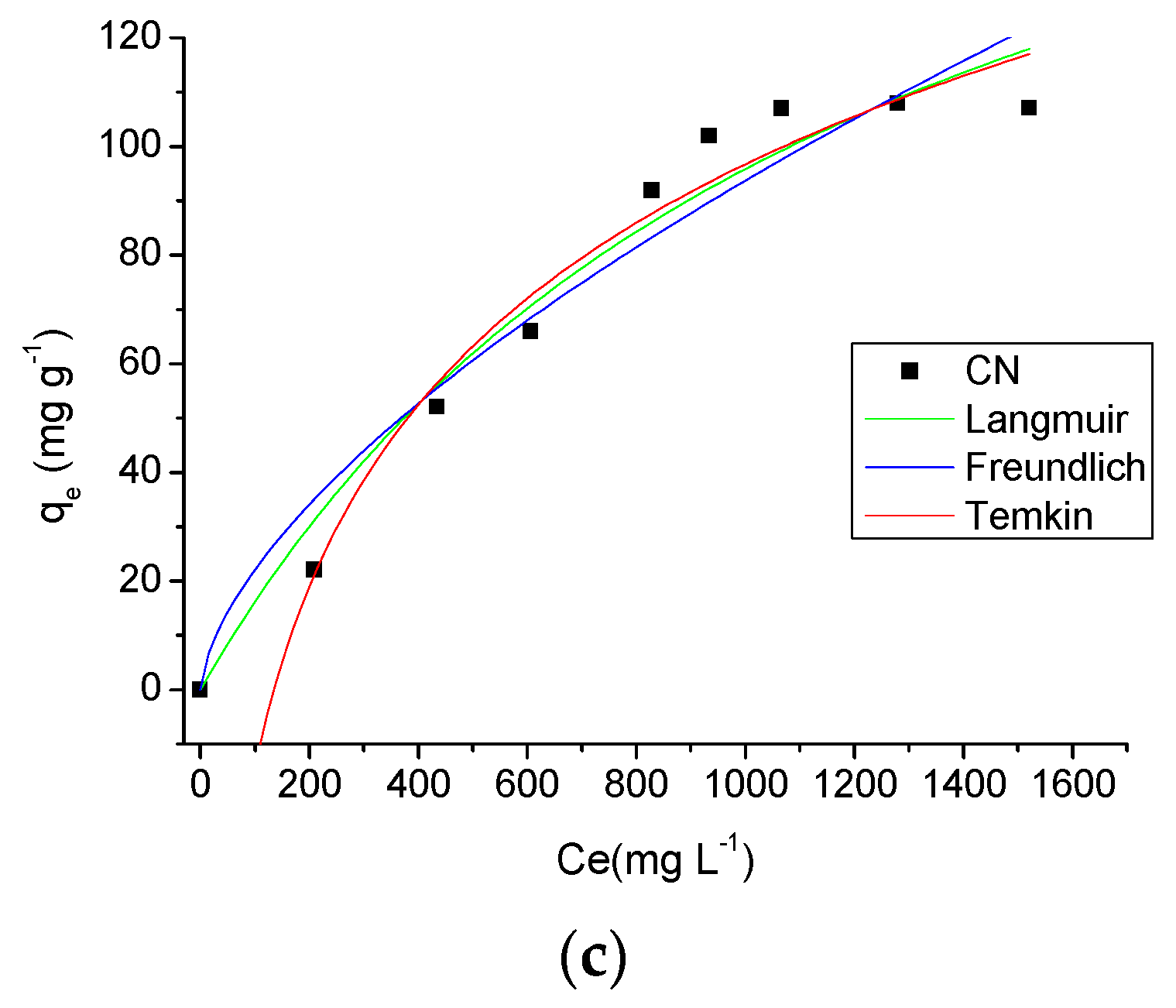
2.3.4. Effect of Concentration and Temperature
2.3.5. Ionic Strength
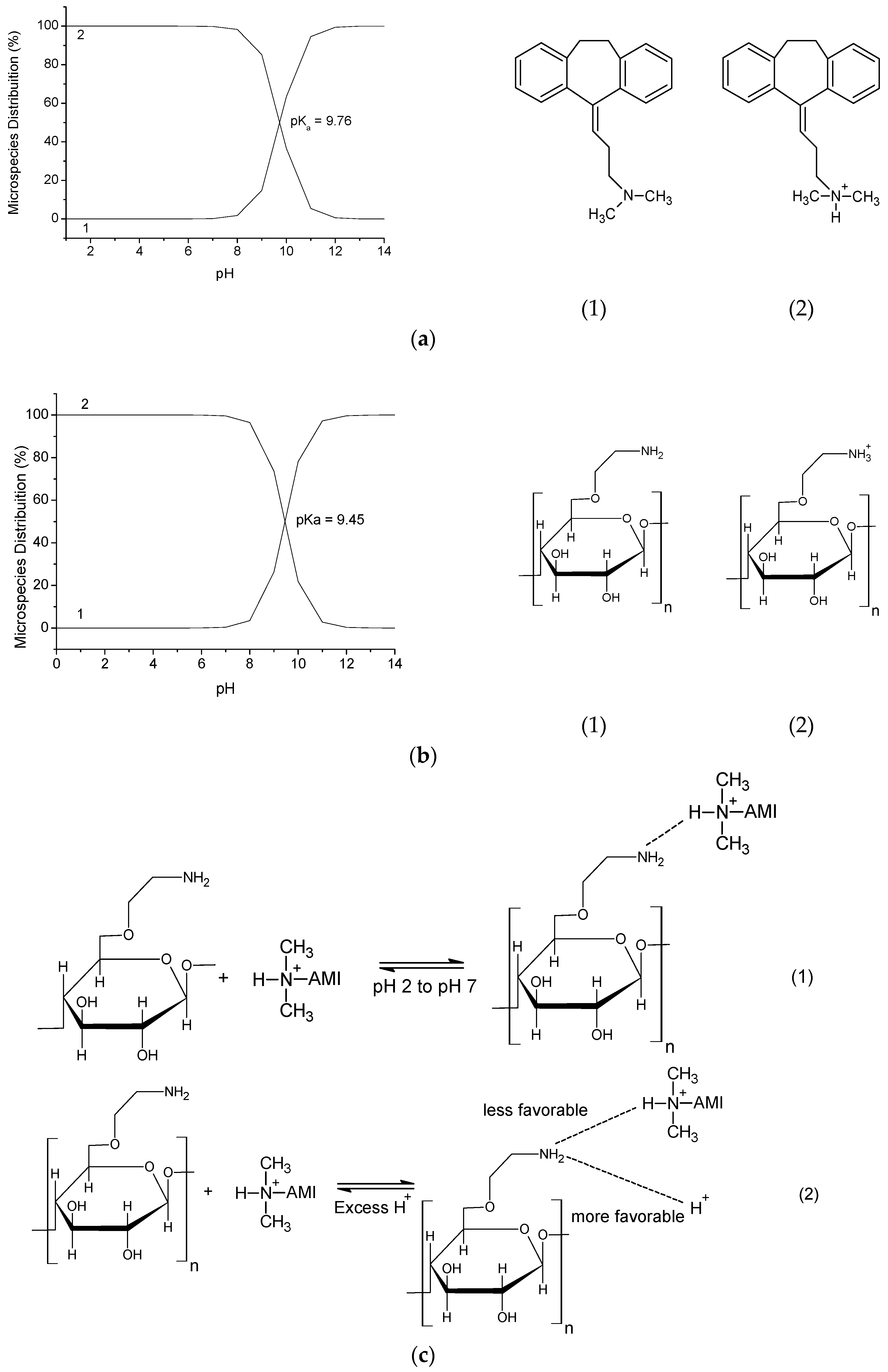
2.4. Drug/Polymer Interaction
3. Materials and Methods
3.1. Materials
3.2. Cellulose Chemical Modification with Ethylenediamine
3.3. Characterizations
3.4. Adsorption
3.4.1. Point of Zero Charge (pHpzc)
3.4.2. Influence of Time
3.4.3. Effect of pH
3.4.4. Study of Concentration and Temperature
3.4.5. Ionic Strength
3.5. Computational Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Nabais, J.M.V.; Ledesma, B.; Laginhas, C. Removal of amitriptyline from aqueous media using activated carbons. Adsorpt. Sci. Technol. 2012, 30, 255–263. [Google Scholar] [CrossRef]
- Chang, P.; Jiang, W.; Li, Z.; Kuo, C.; Jean, J.; Chen, W.; Lv, G. Mechanism of amitriptyline adsorption on Ca-montmotillonite (SAz-2). J. Hazard. Mater. 2014, 277, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, R.D.S.; Morais, A.I.S.; Osajima, J.A.; Nunes, L.C.C.; da Silva Filho, E.C. Development of new phosphated cellulose for application as an efficient biomaterial for the incorporation/release of amitriptyline. Int. J. Biol. Macromol. 2016, 86, 362–375. [Google Scholar] [CrossRef] [PubMed]
- Lv, G.; Stockwell, C.; Niles, J.; Minegar, S.; Li, Z.; Jiang, W. Uptake and retention of amitriptyline by Kaolinite. J. Colloid Interface Sci. 2013, 411, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Real, F.J.; Benitez, F.J.; Acero, J.L.; Roldan, G.; Casas, F. Elimination of the emerging contaminants amitriptyline hydrochloride, methyl salicylate, and 2-phenoxyethanol in ultrapure water and secondary effluents by photolytic and radicalary pathways. Ind. Eng. Chem. Res. 2012, 51, 16209–16215. [Google Scholar] [CrossRef]
- Wijekoon, K.C.; Hai, F.I.; Kang, J.; Price, W.E.; Guo, W.; Ngo, H.H.; Nghiem, L.D. The Fate of pharmaceuticals, steroid hormones, phytoestrogens, UV-filters and pesticides during MBR treatment. Bioresour. Technol. 2013, 144, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Wijekoon, K.C.; Hai, F.I.; Kang, J.; Price, W.E.; Cath, T.Y.; Nghiem, L.D. Rejection and fate of trace organic compounds (TrOCs) during membrane distillation. J. Membr. Sci. 2014, 453, 636–642. [Google Scholar] [CrossRef]
- Silva, L.S.; Lima, L.C.B.; Silva, F.C.; Matos, J.M.E.; Santos, M.R.M.C.; Santos Júnior, L.S.; Sousa, K.S.; da Silva Filho, E.C. Dye anionic sorption in aqueous solution onto a cellulose surface chemically modified with aminoethanethiol. Chem. Eng. J. 2013, 218, 89–98. [Google Scholar] [CrossRef]
- Alencar, J.M.; Oliveira, F.J.V.E.; Airoldi, C.; da Silva Filho, E.C. Organophilic nickel phyllosilicate for reactive blue dye removal. Chem. Eng. J. 2014, 15, 332–340. [Google Scholar] [CrossRef]
- Da Silva Filho, E.C.; de Melo, J.C.P.; Airoldi, C. Preparation of ethylenediamine—Anchored cellulose and determination of thermochemical data for the interaction between cations and basic centers at the solid/liquid interface. Carbohydr. Res. 2006, 341, 2842–2850. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Filho, E.C.; Júnior, L.S.S.; Santos, M.R.M.C.; Fonseca, M.G.; Sousa, K.S.; Santana, S.A.A.; Airoldi, C. Thermochemistry of interaction between cellulose modified with 2-aminomethylpyridine and divalent cations. J. Therm. Anal. Calorim. 2013, 114, 423–429. [Google Scholar] [CrossRef]
- Da Silva Filho, E.C.; da Silva, L.S.; Lima, L.C.B.; Santos Júnior, L.S.; Santos, M.R.M.C.; de Matos, J.M.E.; Airoldi, C. Thermodynamic data of 6-(4-aminobutylamino)-6-deoxycellulose sorbent for cátion removal from aqueous solutions. Sep. Sci. Technol. 2011, 46, 2566–2574. [Google Scholar] [CrossRef]
- Da Silva Filho, E.C.; Santos Júnior, L.S.; Silva, M.M.F.; Fonseca, M.G.; Santana, S.A.A.; Airoldi, C. Surface cellulose modification with 2-aminomethylpyridine for copper, cobalt, nickel and zinc removal from aqueous solutions. Mater. Res. 2013, 16, 79–87. [Google Scholar] [CrossRef]
- Torres, J.D.; Faria, E.A.; Prado, A.G.S. Thermodynamic studies of the interaction at the solid/liquid interface between metal ions and cellulose modified with ethylenediamine. J. Hazard. Mater. 2006, 129, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Li, W.; Xu, Q.; Sun, Q. Amino-functionalized nanocrystalline cellulose as an adsorbent for anionic dyes. Cellulose 2015, 22, 2443–2456. [Google Scholar] [CrossRef]
- Hu, D.; Wang, L. Adsorption of ciprofloxacin from aqueous solutions onto cationic and anionic flax noil cellulose. Desalin. Water Treat. 2016, 57, 1–14. [Google Scholar] [CrossRef]
- Hu, D.; Wang, L. Adsorption of amoxicillin onto quaternized cellulose from flax noil: Kinetic, equilibrium and thermodynamic study. J. Taiwan Inst. Chem. Eng. 2016, 64, 227–234. [Google Scholar] [CrossRef]
- Bezerra, R.D.S.; Morais, A.I.S.; Osajima, J.A.; Nunes, L.C.C.; Silva Filho, E.C. Cellulose phosphate applied in the removal of the drug acetaminophen from aqueous media. Mater. Sci. Forum 2016, 869, 745–749. [Google Scholar] [CrossRef]
- Calisto, V.; Ferreira, C.I.A.; Santos, S.M.; Gil, M.V.; Otero, M.; Esteves, V.I. Production of adsorbents by pyrolysis of paper mill sludge and application on the removal citalopram from water. Bioresour. Technol. 2014, 166, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Jaria, G.; Calisto, V.; Gil, M.V.; Otero, M.; Esteves, V.I. Removal of fluoxetine from water by adsorbent materials produced from paper mill sludge. J. Colloid Interface Sci. 2015, 448, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Filho, E.C.; Lima, L.C.B.; Sousa, K.S.; Fonseca, M.G. Calorimetry studies for interaction in solid/liquid interface between the modified cellulose and divalent cátion. J. Therm. Anal. Calorim. 2013, 114, 57–66. [Google Scholar] [CrossRef]
- Silva, L.S.; Lima, L.C.B.; Ferreira, F.J.L.; Silva, M.S.; Osajima, J.A.; Bezerra, R.D.S.; da Silva Filho, E.C. Sorption of the anionic reactive red RB dye in cellulose: Assessment of kinetic, thermodynamic, and equilibrium data. Open Chem. 2015, 13, 801–812. [Google Scholar] [CrossRef]
- Bezerra, R.D.S.; Silva, M.M.F.; Morais, A.I.S.; Santos, M.R.M.C.; Airoldi, C.; da Silva Filho, E.C. Natural cellulose for ranitidine drug removal from aqueous solutions. J. Environ. Chem. Eng. 2014, 2, 605–611. [Google Scholar] [CrossRef]
- Da Silva Filho, E.C.; Santana, S.A.A.; Melo, H.C.P.; Oliveira, F.J.V.E.; Airoldi, C. X-ray diffraction and thermogravimetry data of cellulose, chlorodeoxycellulose and aminodeoxycellulose. J. Therm. Anal. Calorim. 2010, 100, 315–321. [Google Scholar] [CrossRef]
- Segal, L.; Creely, J.J.; Martin, A.E.; Conrad, C.M. An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text. Res. J. 1959, 29, 786–794. [Google Scholar] [CrossRef]
- Da Silva Filho, E.C.; Lima, L.C.B.; Silva, F.C.; Sousa, K.S.; Fonseca, M.G.; Santana, S.A.A. Immobilization of ethylene sulfide in aminated cellulose for removal of the divalent cations. Carbohydr. Polym. 2013, 92, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Pavia, D.L.; Lampman, G.M.; Kriz, G.S.; Vyvyan, J.A. Introduction to Spectroscopy, 4th ed.; Brooks/Cole: Belmont, CA, USA, 2009. [Google Scholar]
- Isogai, A. NMR Analysis of cellulose dissolved in aqueous NaOH solutions. Cellulose 1997, 4, 99–107. [Google Scholar] [CrossRef]
- McMurry, J.E. Amines and Heterocycles. In Organic Chemistry, 7th ed.; Thomson Brooks/Cole: Belmont, CA, USA, 2008. [Google Scholar]
- Fathi, M.R.; Asfaram, A.; Farhangi, A. Removal of Direct Red 23 from aqueous solutions using corn stalks: Isotherms, kinetics and thermodynamic studies. Spectrochim. Acta Mol. Biomol. Spectrosc. 2015, 135, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Chowdhury, S.; das Saha, P. Adsorption of Crystal Violet from aqueous solution onto NaOH-modified rice husk. Carbohydr. Polym. 2011, 86, 1533–1541. [Google Scholar] [CrossRef]
- Chang, Y.K.; Chu, L.; Tsai, J.C.; Chiu, S.J. Kinetic study of immobilized lysozyme on the extrudate-shaped NaY zeolite. Process Biochem. 2006, 41, 1864–1874. [Google Scholar] [CrossRef]
- Sirvio, A.S.; Hasa, T.; Leiviska, T.; Liimatainen, H.; Hormi, O. Biophosphonate nanocellulose in the removal of vanadium(V) from water. Cellulose 2016, 23, 689–697. [Google Scholar] [CrossRef]
- Bezerra, R.D.S.; Silva, M.M.F.; Morais, A.I.S.; Osajima, J.A.; Santos, M.R.M.C.; Airoldi, C.; da Silva Filho, E.C. Phosphated cellulose as an efficient biomaterial for aqueous drug ranitidine removal. Materials 2014, 7, 7907–7924. [Google Scholar] [CrossRef] [PubMed]
- Oshima, T.; Taguchi, S.; Ohe, K.; Baba, Y. Phosphorylated bacterial cellulose for adsorption of proteins. Carbohydr. Polym. 2011, 83, 953–958. [Google Scholar] [CrossRef]
- Saleh, T.A.; Haladu, S.A.; Ali, S.A. A novel cross-linked pH-responsive tetrapolymer: Synthesis, characterization and sorption evaluation towards Cr(III). Chem. Eng. J. 2015, 269, 9–19. [Google Scholar] [CrossRef]
- Alshehri, S.M.; Naushad, M.; Ahamad, T.; Alothman, Z.A.; Aldalbahi, A. Synthesis, characterization of curcumin based ecofriendly antimicrobial bio-adsorbent for the removal of phenol from aqueous medium. Chem. Eng. J. 2014, 254, 181–189. [Google Scholar] [CrossRef]
- Yilmaz, M.S.; Ozdemir, O.D.; Piskin, S. Synthesis and characterization of MCM-41 with different methods and adsorption of Sr2+ on MCM-41. Res. Chem. Intermed. 2015, 41, 199–211. [Google Scholar] [CrossRef]
- Cheng, L.; Ye, L.; Sun, D.; Wu, T.; Li, Y. Removal of petroleum sulfonate from aqueous solution by hydroxide precipitates generated from leaching solution of white mud. Chem. Eng. J. 2015, 264, 672–680. [Google Scholar] [CrossRef]
- Wan, Z.; Chen, W.; Liu, C.; Liu, Y.; Dong, C. Preparation and characterization of γ-AlOOH@CS magnetic nanoparticle as a novel adsorbent for removing fluoride from drinking water. J. Colloid Interface Sci. 2015, 443, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.A.; Alrozi, R. Removal of malachite green dye from aqueous using rambutan peel-based activated carbon: Equilibrium, kinetic and thermodynamic studies. Chem. Eng. J. 2011, 171, 510–516. [Google Scholar] [CrossRef]
- Tsai, Y.L.; Chang, P.H.; Gao, Z.Y.; Xu, X.Y.; Chen, Y.H.; Wang, Z.H.; Chen, X.Y.; Yang, Z.Y.; Wang, T.H.; Jean, J.S.; et al. Amitriptyline removal using palygorskite clay. Chemosphere 2016, 155, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Boldvai, J.; Götz, M.; Posgay-Kovács, E. Thermoanalytische untersuchung zyklischer antidepressiva. J. Therm. Anal. 1983, 27, 57–67. [Google Scholar] [CrossRef]
- Chauvet, A.; Masse, J. Stude thermoanalytique de substances psychotherapeutiques. III. Antidepresseurs chlorhydrated d’imipramine, chlorhydrate de clomipramine, chlorhdrate de nortriptyline, chlorhydrate d’amitriptyline. Thermochim. Acta 1983, 65, 179–196. [Google Scholar] [CrossRef]
- Abu-Eittah, R.H.; Kamel, L.T. Kinetics and thermodynamic parameters of the thermal decomposition of imipramine hydrochloride and trimipramine maleate. Int. J. Chem. Kinet. 2003, 35, 166–179. [Google Scholar] [CrossRef]
- Lagergren, S. Zur theorie der sogenannten adsorption geloster stoffe, Kungliga Svenska Vetenskapsakademiens. Handlingar 1898, 24, 1–39. [Google Scholar]
- Ho, Y.S.; Mckay, G. Kinetic models for the sorption of dye from aqueous solution by wood. Process Saf. Environ. Prot. 1998, 76, 183–191. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Bhattacharyya, K.G.; Sharma, A. Adsorption of Pb(II) from aqueous solution by Azadirachta indica (Neem) leaf powder. J. Hazard. Mater. 2004, 113, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Freundlich, H.M.F. Uber die adsorption in losungen. J. Phys. Chem. 1906, 57, 385–470. [Google Scholar] [CrossRef]
- Temkin, M.J.; Pyzhev, V. Recent modifications to Langmuir isotherms. Acta Phys. 1940, 12, 217–222. [Google Scholar]
- Sun, X.; Chen, J.H.; Su, Z.; Huang, Y.; Dong, X. Highly effective removal of Cu(II) by a novel 3-aminopropyltriethoxysilane functionalized polyethyleneimine/sodium alginate porous membrane adsorbent. Chem. Eng. J. 2016, 290, 1–11. [Google Scholar] [CrossRef]
- Vieira, A.P.; Santana, S.A.A.; Bezerra, C.W.B.; Silva, H.A.S.; Chaves, J.A.P.; de Melo, J.C.P.; da Silva Filho, E.C.; Airoldi, C. Kinetics and thermodynamics of textile dye adsorption from aqueous solutions using babassu coconut mesocarp. J. Hazard. Mater. 2009, 166, 1272–1278. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian 09, Revision B.01; Gaussian Inc.: Wallingford, CT, USA, 2010. [Google Scholar]
- Burke, K. The ABC of DFT. 2007. Available online: http://dft.uci.edu/research.php (accessed on 7 May 2016).
- Morgon, N.H.; Custodio, R. Teoria do Funcional de Densidade. Quim. Nova 1995, 18, 44–55. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact Exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Kohn, W.; Sham, L.J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 1965, 140, 1133–1138. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. B 1988, 37, 785–789. [Google Scholar] [CrossRef]
- Hariharan, P.C.; Pople, J.A. The influence of polarization functions on molecular orbital hydrogenation energies. Theor. Chem. Acta 1973, 28, 213–222. [Google Scholar] [CrossRef]
- Peng, C.; Ayala, P.Y.; Schlegel, H.B. Using redundant internal coordinates to optimize equilibrium geometries and transition states. J. Comp. Chem. 1996, 17, 49–56. [Google Scholar] [CrossRef]
- Peng, C.; Schlegel, H.B. Combining synchronous transit and quasi-Newton methods to find transition states. Isr. J. Chem. 1993, 33, 449–454. [Google Scholar] [CrossRef]
- Hratchian, H.P.; Schlegel, H.B. Theory and Applications of Computational Chemistry: The First 40 Years; Dykstra, C.E., Frenking, G., Kim, K.S., Scuseria, G., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 195–249. [Google Scholar]
- Fukui, K. The Path of chemical reactions—The IRC approach. Acc. Chem. Res. 1981, 14, 363–368. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
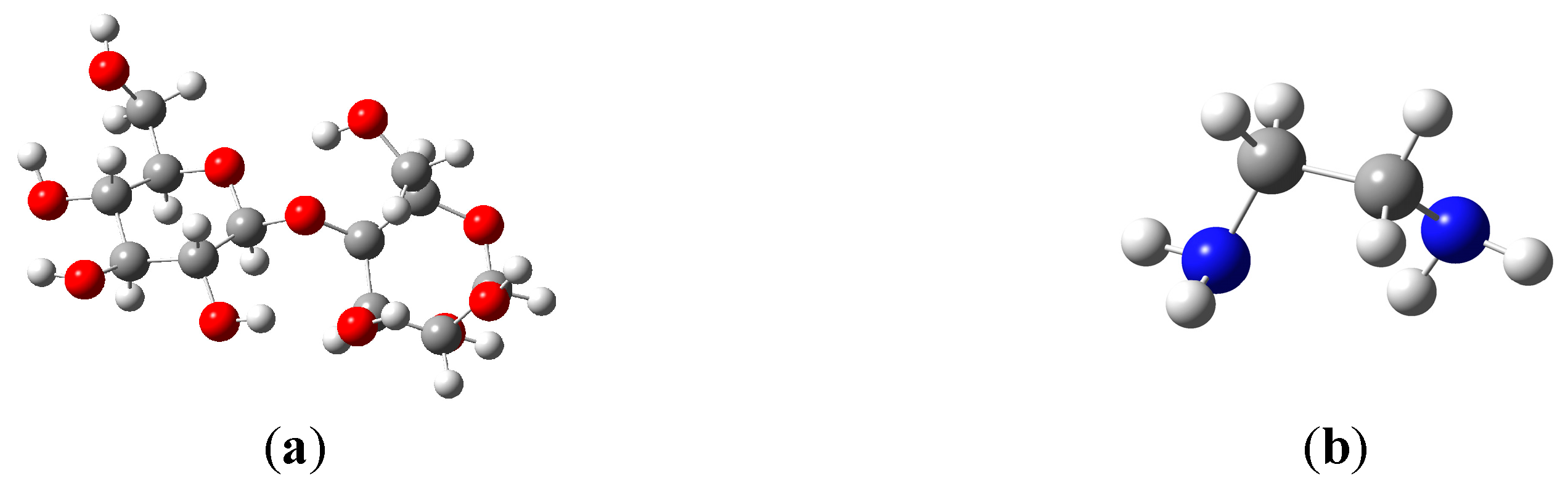
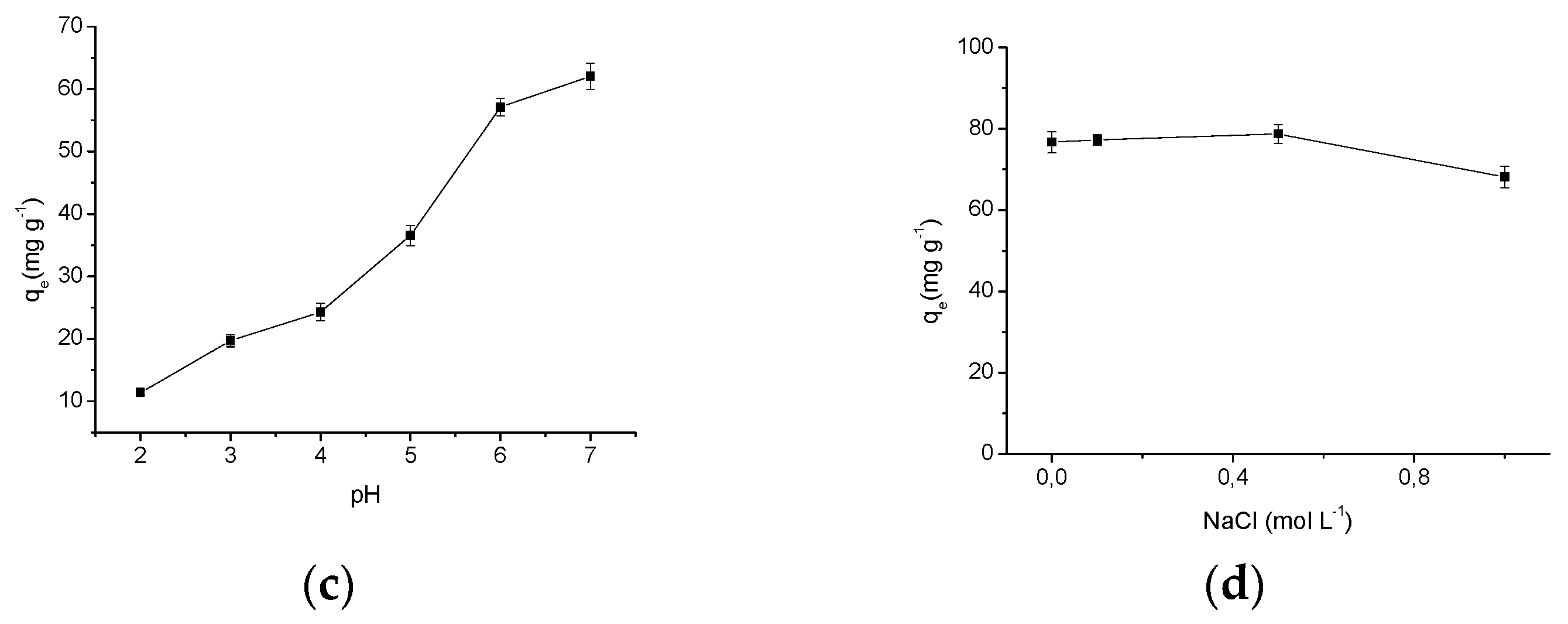
| Element | Pristine Microcrystalline Cellulose | CN Polymer | ||
|---|---|---|---|---|
| % | mmol·g−1 | % | mmol·g−1 | |
| C | 41.57 ± 0.15 | 34.64 ± 0.12 | 41.54 ± 0.02 | 34.62 ± 0.01 |
| N | - | - | 1.10 ± 0.01 | 0.78 ± 0.02 |
| Model | Parameters | CN Polymer |
|---|---|---|
| Pseudo-first Order | K1 | 0.0101 (min−1) |
| qe(cal) | 58.7742 (mg·g−1) | |
| R2 | 0.9732 | |
| Pseudo-second Order | K2 | 0.0002 (g·mg−1·min−1) |
| qe(cal) | 72.4653 (mg·g−1) | |
| R2 | 0.9837 |
| Model | Parameters | CN Polymer |
|---|---|---|
| T = 298 K | ||
| Langmuir | qmax | 273.2882 (mg·g−1) |
| KL | 0.0004 (L·mg−1) | |
| R2 | 0.9538 | |
| RL | 0.7180 | |
| Freundlich | nf | 1.2637 |
| Kf | 0.3019 (L·g−1) | |
| R2 | 0.9433 | |
| Temkin | nT | 13.7418 |
| KT | 0.0060 (L·g−1) | |
| R2 | 0.9456 | |
| T = 308 K | ||
| Langmuir | qmax | 234.5308 (mg·g−1) |
| KL | 0.0006 (L·mg−1) | |
| R2 | 0.9582 | |
| RL | 0.6143 | |
| Freundlich | nf | 1.4190 |
| Kf | 0.6517 (L·g−1) | |
| R2 | 0.9387 | |
| Temkin | nT | 18.1816 |
| KT | 0.0065 (L·g−1) | |
| R2 | 0.9652 | |
| T = 318 K | ||
| Langmuir | qmax | 311.7546 (mg·g−1) |
| KL | 0.0008 (L·mg−1) | |
| R2 | 0.5329 | |
| RL | 0.9627 | |
| Freundlich | nf | 1.5923 |
| Kf | 1.2238 (L·g−1) | |
| R2 | 0.9371 | |
| Temkin | nT | 17.6408 |
| KT | 0.0074 (L·g−1) | |
| R2 | 0.9734 |
| Polymer | ΔH° (kJ·mol−1) | ΔS° (J·mol−1·K−1) | ΔG° (kJ·mol−1) | ||
|---|---|---|---|---|---|
| 298 K | 308 K | 318 K | |||
| CN | 7.7094 | 4.4285 | 6.3897 | 6.3454 | 6.3011 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
D. S. Bezerra, R.; C. Leal, R.; S. da Silva, M.; I. S. Morais, A.; H. C. Marques, T.; A. Osajima, J.; B. Meneguin, A.; Da S. Barud, H.; C. da Silva Filho, E. Direct Modification of Microcrystalline Cellulose with Ethylenediamine for Use as Adsorbent for Removal Amitriptyline Drug from Environment. Molecules 2017, 22, 2039. https://doi.org/10.3390/molecules22112039
D. S. Bezerra R, C. Leal R, S. da Silva M, I. S. Morais A, H. C. Marques T, A. Osajima J, B. Meneguin A, Da S. Barud H, C. da Silva Filho E. Direct Modification of Microcrystalline Cellulose with Ethylenediamine for Use as Adsorbent for Removal Amitriptyline Drug from Environment. Molecules. 2017; 22(11):2039. https://doi.org/10.3390/molecules22112039
Chicago/Turabian StyleD. S. Bezerra, Roosevelt, Régis C. Leal, Mateus S. da Silva, Alan I. S. Morais, Thiago H. C. Marques, Josy A. Osajima, Andréia B. Meneguin, Hernane Da S. Barud, and Edson C. da Silva Filho. 2017. "Direct Modification of Microcrystalline Cellulose with Ethylenediamine for Use as Adsorbent for Removal Amitriptyline Drug from Environment" Molecules 22, no. 11: 2039. https://doi.org/10.3390/molecules22112039
APA StyleD. S. Bezerra, R., C. Leal, R., S. da Silva, M., I. S. Morais, A., H. C. Marques, T., A. Osajima, J., B. Meneguin, A., Da S. Barud, H., & C. da Silva Filho, E. (2017). Direct Modification of Microcrystalline Cellulose with Ethylenediamine for Use as Adsorbent for Removal Amitriptyline Drug from Environment. Molecules, 22(11), 2039. https://doi.org/10.3390/molecules22112039











