WxC-β-SiC Nanocomposite Catalysts Used in Aqueous Phase Hydrogenation of Furfural
Abstract
:1. Introduction
2. Results and Discussion
2.1 Catalytical Transformation of Waste Biomass to THFA
3. Materials and Methods
3.1. Preparation of the Catalysts
3.2. Hydrogenation of Furfural
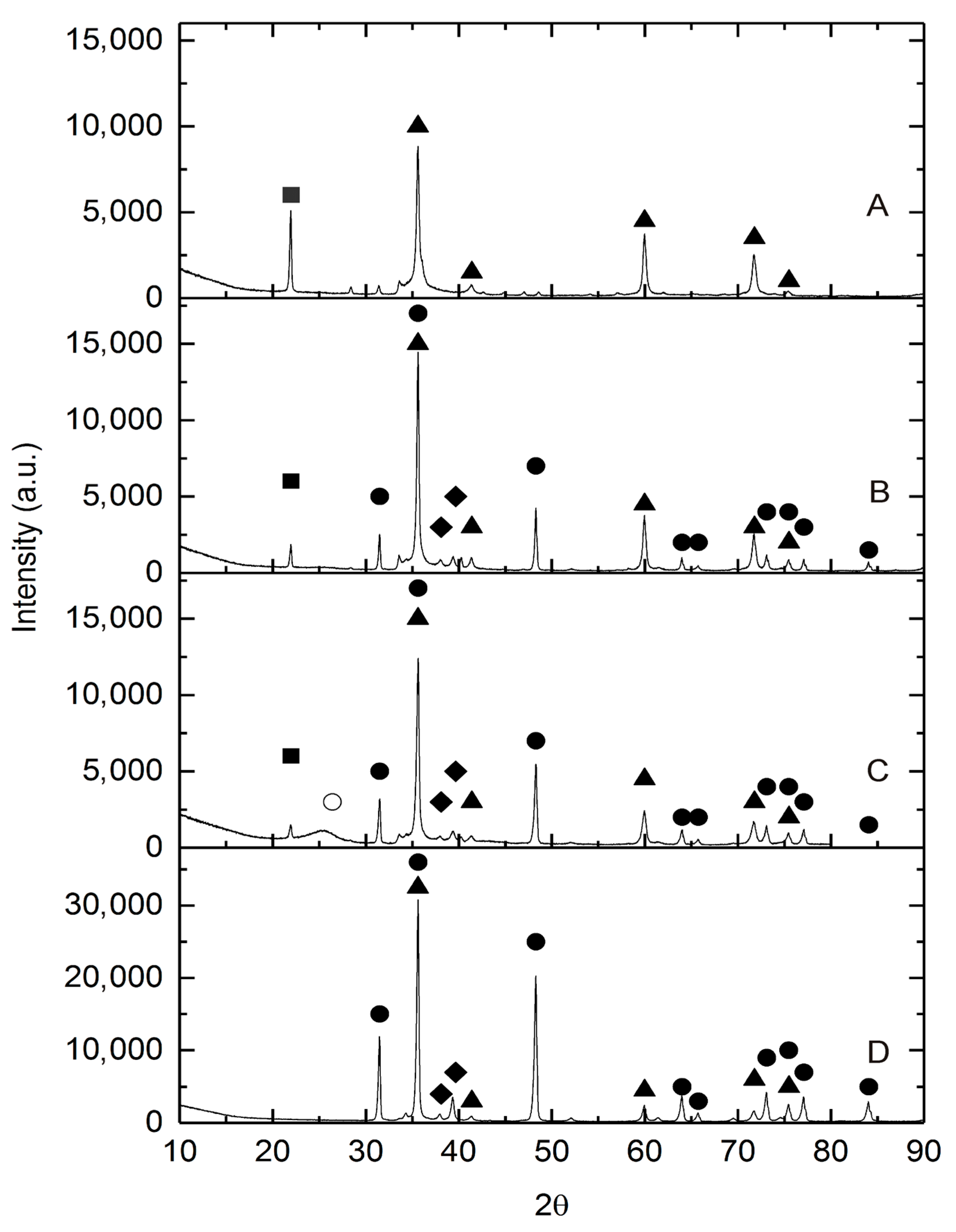
3.3. Analysis by X-ray Diffraction (XRD)
3.4. Low-Temperature N2 Adsorption/Desorption Measurements
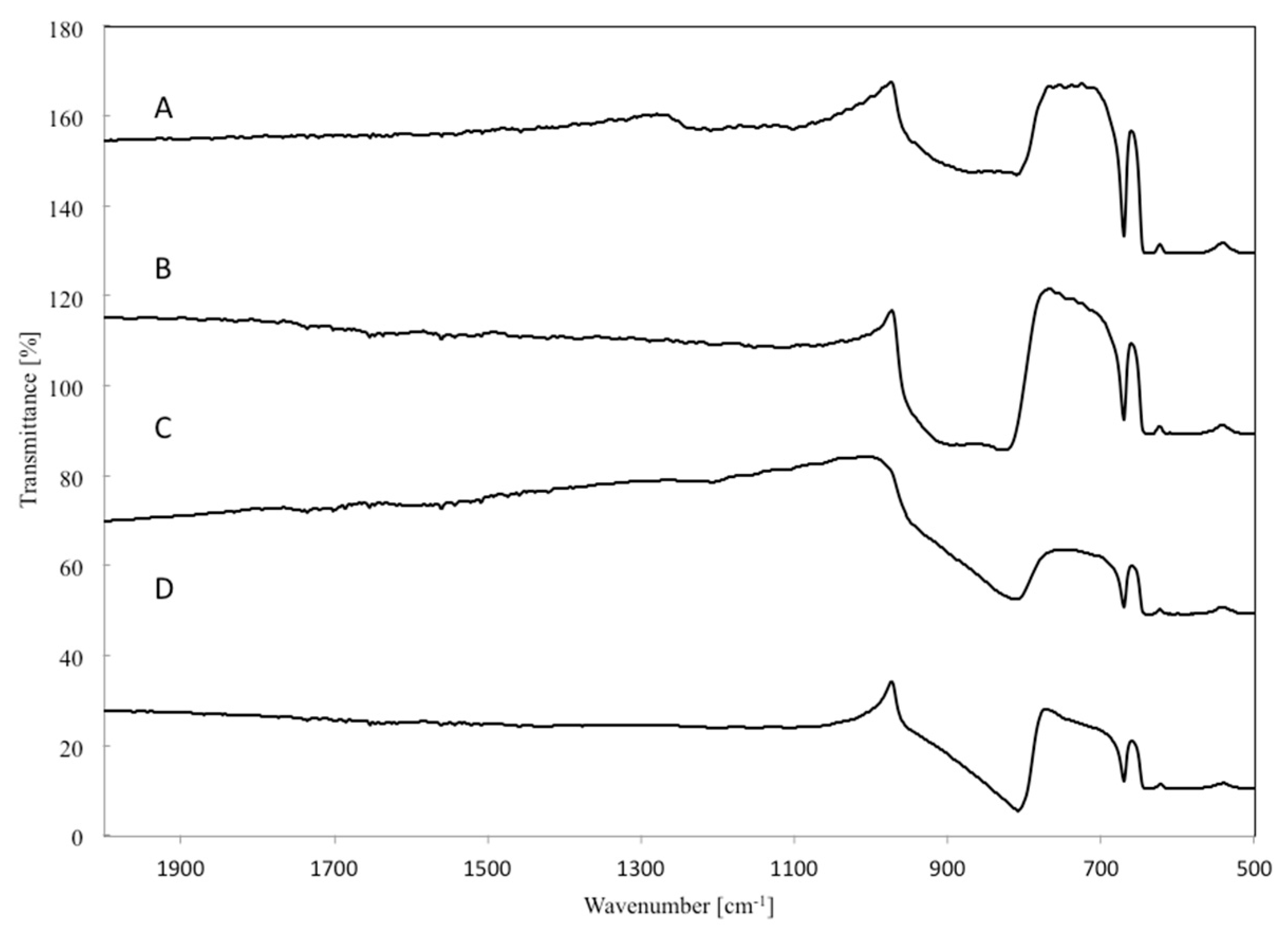
3.5. Fourier Transform Infrared (FTIR) Measurements
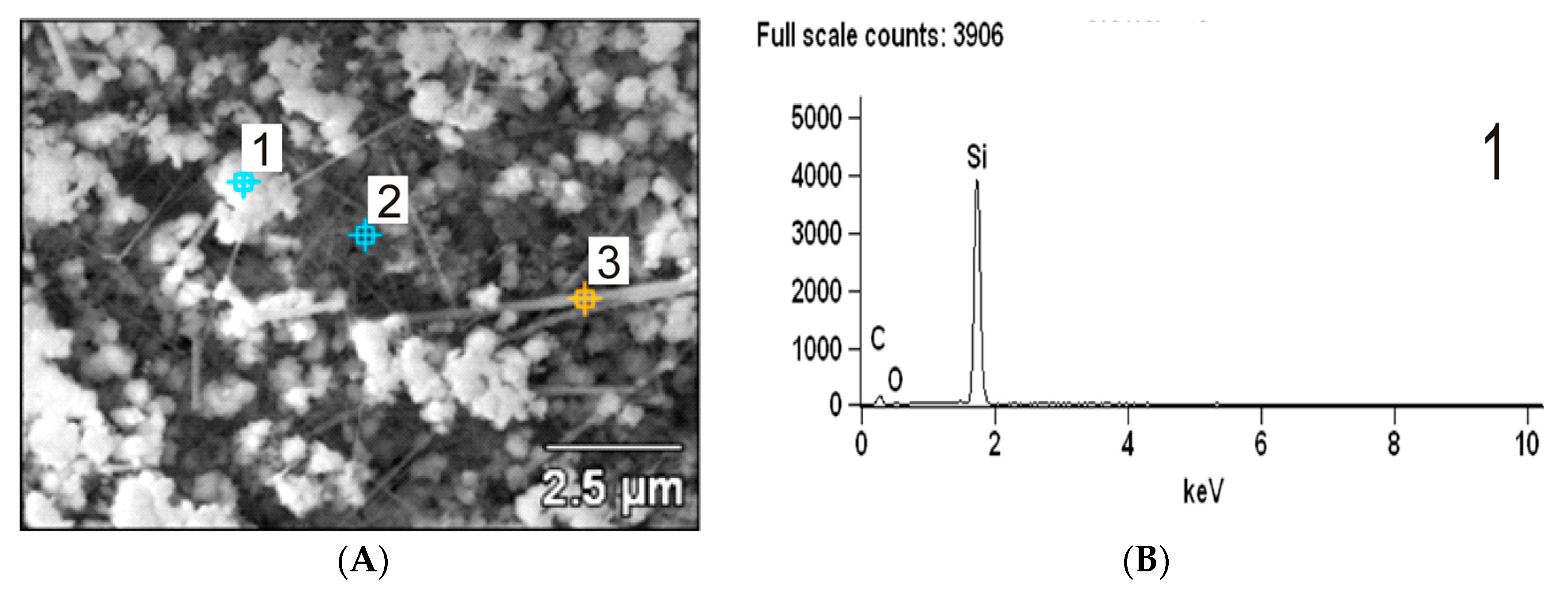
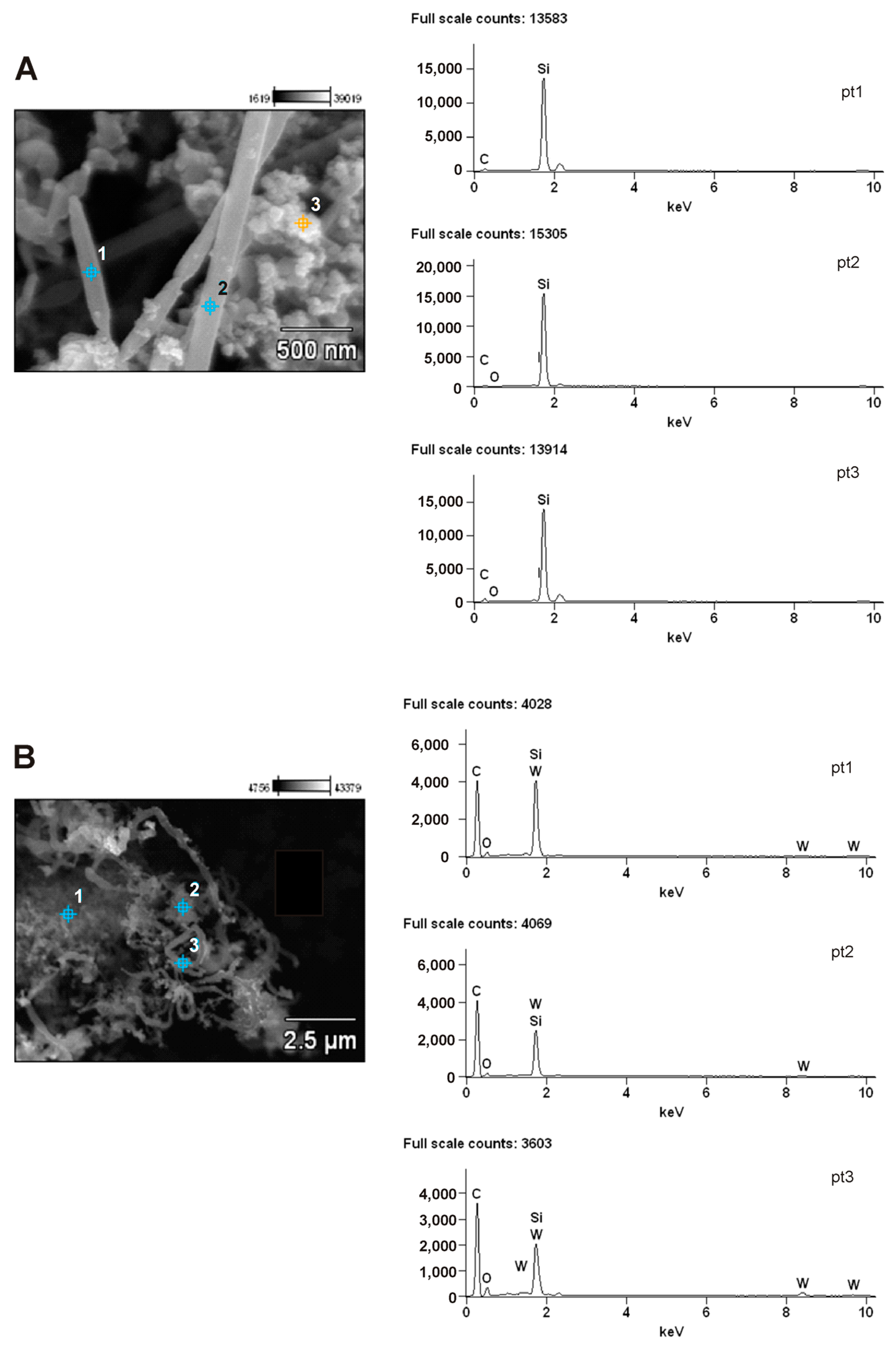
3.6. Scanning Electron Microscopy (SEM) Analysis of β-SiC and WxC-β-SiC
3.7. Transmission Electron Microscopy (TEM) Analysis
4. Conclusions
- (a)
- In the case of pure β-SiC catalysts, the activity of the systems depends on the presence of both β-SiC and SiO2 crystalline phases. This means that incomplete carbothermal reduction of SiO2 leads to the development of the system surface and improves its activity. However, the reduction of furfural is not selective and the process produces similar yields of FA and THFA.
- (b)
- While the addition of tungsten does not influence furfural conversion by β-SiC systems, it does affect the type of products produced in the reaction mixture. As the amount of tungsten in the β-SiC system was increased up to 10% by weight, the THFA yields also rose. The best selectivity to THFA was achieved with nanocomposite (10)WxC-β-SiC catalyst. The improved selectivity of (10)WxC-β-SiC catalyst to THFA is probably attributable to the sufficient dispersion of WC phase in this nanocomposite, in combination with its textural properties. The addition of larger amounts of W into the catalyst did not lead to further improvement of selectivity for THFA.
- (c)
- The results obtained by SEM, TEM and FTIR techniques reveal that the addition of tungsten into the β-SiC system facilitates the formation of whiskers and reduction of SiO2 by C in the carbothermal reduction process.
- (d)
- The nanocomposite (10)WxC-β-SiC catalyst was used successfully for the reduction of an aqueous solution of furfural obtained by acid hydrolysis of flax and oat straws. Despite the presence of trace amounts of organic impurities in the substratum obtained from the biomass, the (10)WxC-β-SiC catalyst was characterized by a very high activity and selectively for THFA. After optimization of the process conditions, this two-step catalytic process (I—acidic hydrolysis of biomass; II—catalytic reduction of Furfural to THFA) has potential to be used for the commercial production of green solvent from waste biomass.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yan, K.; Wu, G.; Lafleur, T.; Jarvis, C. Production, properties and catalytic hydrogenation of furfural to fuel additives and value-added chemicals. Renew. Sustain. Energy Rev. 2014, 38, 663–676. [Google Scholar] [CrossRef]
- Khan, F.A.; Vallat, A.; Süss-Fink, G. Highly selective low-temperature hydrogenation of furfuryl alcohol to tetrahydrofurfuryl alcohol catalysed by hectorite-supported ruthenium nanoparticles. Catal. Commun. 2011, 12, 1428–1431. [Google Scholar] [CrossRef]
- Yan, K.; Liao, J.; Wu, X.; Xie, X. A noble-metal free Cu-catalyst derived from hydrotalcite for highly efficient hydrogenation of biomass-derived furfural and levulinic acid. RSC Adv. 2013, 3, 3853. [Google Scholar] [CrossRef]
- Flaim, T.D.; Lamb, J.; Brewer, T. Microlithographic Substrate Cleaning and Compositions Therefor. U.S. Patent 5,362,608, 8 November 1994. [Google Scholar]
- Świętosławski, J.; Łabanowski, G. Gel for Pests Control and Against Tree Diseases by Means of Microinjection. PL Patent 196,977 B1, 29 February 2008. [Google Scholar]
- Vom Stein, T.; Klankermayer, J.; Leitner, W. Tailor-Made Fuels and Chemicals from Biomass in Catalysis for the Conversion of Biomass and its Derivatives. In Max Planck Research Library for the History and Development of Knowledge; Behrens, M., Datye, A.K., Eds.; Neopubli GmbH: Berlin, Germany, 2013. [Google Scholar]
- Wilson, H.B. Process for the Hydration of Furfural. U.S. Patent 2,487,054, 8 November 1949. [Google Scholar]
- Priickner, H. Preparation of Tetrahydrofuran. U.S. Patent 2,071,704, 23 February 1937. [Google Scholar]
- Lesiak, M.; Binczarski, M.; Karski, S.; Maniukiewicz, W.; Rogowski, J.; Szubiakiewicz, E.; Berlowska, J.; Dziugan, P.; Witonska, I. Hydrogenation of furfural over Pd–Cu/Al2O3 catalysts. The role ofinteraction between palladium and copper on determining catalytic properties. J. Mol. Catal. A Chem. 2014, 395, 337–348. [Google Scholar] [CrossRef]
- Merat, N.; Godowa, C.; Gaset, A. High selective production of tetrahydrofurfuryl alcohol: Catalytic hydrogenation of furfural and furfuryl alcohol. J. Chem. Technol. Biotechnol. 2007, 48, 145–159. [Google Scholar] [CrossRef]
- Chen, B.; Li, F.; Huang, Z.; Yuan, G. Tuning catalytic selectivity of liquid-phase hydrogenation of furfural via synergistic effects of supported bimetallic catalysts. Appl. Catal. A 2015, 500, 23–29. [Google Scholar] [CrossRef]
- Zhao, H.; Liu, C. Study of the catalysts for hydrogenation of furfuryl alcohol to tetrahydofurfuryl alcohol. Jingxi Huagong 2001, 18, 332, reprinted in Chem. Abstr. 2002, 136, 153064a. [Google Scholar]
- Liu, C.G.; Shen, R.H.; Xing, J.X.; Liu, Y.Q.; Zhao, R.Y.; Lu, C.B.; Liu, K. A Preparation Method of Tetrahydrofurfuryl Alcohol Catalyst and Tetrahydrofurfuryl Alcohol. CN Patent 1,280,980, 24 January 2001. [Google Scholar]
- Zhai, P.M.; Liu, C.G.; Shen, R.H.; Zhao, H.J.; Lu, C.B.; Liu, K. Preparation of supported nickel catalysts with high activity and selectivity and its use for hydrogenation of furfuryl alcohol. Shiyou Daxue Xuebao Ziran Kexueban 2000, 24, 25, reprinted in Chem. Abstr. 2001, 135, 21178k. [Google Scholar]
- Turtabaev, S.K. Catalytic hydrogenation of Furfurol. Bayandamalary Kaz. Resp. Ulttyk Gylym Akad. 2004, 4, 54, reprinted in Chem. Abstr. 2005, 143, 250040s. [Google Scholar]
- Zhao, H.J.; Xing, J.X.; Liu, C.G. Preparation of tetrahydrofurfuryl alcohol using hydrogenation of furfuryl alcohol catalysed by Raney nickel catalyst. Shiyou Daxue Xuebao Ziran Kexueban 2003, 27, 91, reprinted in Chem. Abstr. 2004, 141, 106330c. [Google Scholar]
- Liu, C.G.; Zhao, H.J.; Zhao, R.Y.; Ma, A.; Liu, Y.Q.; Xing, J.X.; Yin, C.L.; Shen, R.H. Method and Catalyst for Hydrogenation of Furfuryl Alcohol to Tetrahydrofurfuryl Alcohol. CN Patent 1,341,483, 27 March 2002. [Google Scholar]
- Merat, N.; Godawa, C.; Gasand, A. Hydrogenation selective de l’alcool furfurylique en alcool tetrahydrofurfurylique. J. Mol. Catal. 1990, 57, 397–415. [Google Scholar] [CrossRef]
- Tike, M.A.; Mahajani, V.V. Kinetics of liquid-phase hydrogenation of furfuryl alcohol to tetrahydrofurfuryl alcohol over a Ru/TiO2 catalyst. Ind. Eng. Chem. Res. 2007, 46, 3275–3282. [Google Scholar] [CrossRef]
- Seemuth, P.D. Hydrogenation Process using Cationic Ruthenium-Containing Zeolite. U.S. Patent 4,459,419, 10 July 1984. [Google Scholar]
- Sokol’skii, D.V.; Erzhanova, M.S.; Zibrova, N.A.; Tanatarova, S.B. Hydrogenation of furfuryl alcohol on mixtures of platinum and palladium black with aluminum oxide. Khim. Tekhnol. Silik. 1974, 275. reprinted in Chem. Abstr. 1975, 83, 43118a. [Google Scholar]
- Levy, R.B.; Boudart, M. Platinum-like behavior of tungsten carbide in surface catalysis. Science 1973, 181, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Rao, R.; Dandekar, A.; Baker, R.T.K.; Vannice, M.A. Properties of copper chromite catalysts in hydrogenation reactions. J. Catal. 1997, 171, 406–419. [Google Scholar] [CrossRef]
- Seo, G.; Chon, H. Hydrogenation of furfural over copper-containing catalysts. J. Catal. 1981, 67, 424–429. [Google Scholar] [CrossRef]
- Wojcik, B.H. Catalytic hydrogenation of furan compounds. J. Ind. Eng. Chem. 1948, 40, 210–216. [Google Scholar] [CrossRef]
- Bankman, M.; Ohmer, J.; Tacke, T. Formed Copper Catalyst for the Selective Hydrogenation of Furfural to Furfuryl Alcohol. U.S. Patent 5,591,873, 7 January 1997. [Google Scholar]
- Lee, S.P.; Chen, Y.W. Selective hydrogenation of furfural on Ni−P, Ni−B, and Ni−P−B ultrafine materials. Ind. Eng. Chem. Res. 1999, 38, 2548–2556. [Google Scholar] [CrossRef]
- Luo, H.; Li, H.; Zahuang, L. Furfural hydrogenation to furfuryl alcohol over a novel Ni–Co–B amorphous alloy catalyst. Chem. Lett. 2001, 5, 404–405. [Google Scholar] [CrossRef]
- Nowicki, J.; Maciejewski, Z. Uwodornienie furfuralu na katalizatorze Cu-Zn pod ciśnieniem atmosferycznym. Przem. Chem. 1997, 76, 53–54. [Google Scholar]
- Sitthisa, S.; Sooknoi, T.; Ma, Y.; Balbuena, P.B.; Resasco, D.E. Kinetics and mechanism of hydrogenation of furfural on Cu/SiO2 catalysts. J. Catal. 2011, 277, 1–13. [Google Scholar] [CrossRef]
- Sitthisa, S.; Resasco, D.E. Hydrodeoxygenation of furfural over supported metal catalysts: A comparative study of Cu, Pd and Ni. Catal. Lett. 2011, 141, 784–791. [Google Scholar] [CrossRef]
- Nagaraja, B.M.; Siva Kumar, V.; Shasikala, V.; Padmasri, A.H.; Sreedhar, B.; David Raju, B.; Rama Rao, K.S. A highly efficient Cu/MgO catalyst for vapour phase hydrogenation of furfural to furfuryl alcohol. Catal. Commun. 2003, 4, 287–293. [Google Scholar] [CrossRef]
- Rao, R.S.; Baker, R.T.K.; Vannice, M.A. Furfural hydrogenation over carbon-supported copper. Catal. Lett. 1999, 60, 51–57. [Google Scholar] [CrossRef]
- Burk, M.J.; Harper, T.; Gregory, P.; Lee, J.R.; Kalberg, C. Efficient rhodium-catalyzed hydrogenation of aldehydes and ketones. Tetrahedron Lett. 1994, 35, 4963–4966. [Google Scholar] [CrossRef]
- Karakhanov, E.A.; Neimerovets, E.B.; Dedov, A.G. Hydrogenation of Furfural on the Polymer-Containing Metallocomplex Catalysts; Vestnik Moskovskogo Universiteta Seriya 2 Khimiya: Moscow, Russia, 1985. [Google Scholar]
- Hao, X.Y.; Zahou, W.; Wang, J.W.; Zhang, Y.Q.; Liuo, S.X. A novel catalyst for the selective hydrogenation of furfural to furfuryl alcohol. Chem. Lett. 2005, 34, 1000–1001. [Google Scholar] [CrossRef]
- Reddy, B.M.; Reddy, G.K.; Rao, K.N.; Khan, A.; Garesh, I. Silica supported transition metal-based bimetallic catalysts for vapour phase selective hydrogenation of furfuraldehyde. J. Mol. Catal. A 2007, 265, 276–282. [Google Scholar] [CrossRef]
- Wu, J.; Shem, Y.M.; Liu, C.H.; Wang, H.B.; Geng, C.; Zhang, Z.X. Vapor phase hydrogenation of furfural to furfuryl alcohol over environmentally friendly Cu-Ca/SiO2 catalyst. Catal. Commun. 2005, 6, 633–637. [Google Scholar] [CrossRef]
- Binder, H.; Kohling, A.; Kuhn, W.; Lindner, W.; Sandstende, G. Tungsten carbide electrodes for fuel cells with acid electrolyte. Nature 1969, 224, 1299–1300. [Google Scholar] [CrossRef]
- Colton, R.J.; Huang, J.J.; Rabalais, J.W. Electronic structure of tungsten carbide and its catalytic behavior. Chem. Phys. Lett. 1975, 34, 337–339. [Google Scholar] [CrossRef]
- Rodrigues, J.A.J.; Cruz, G.M.; Bugli, G.; Boudart, M.; Djéga-Mariadassou, G. Nitride and carbide of molybdenum and tungsten as substitutes of iridium for the catalysts used for space communication. Catal. Lett. 1997, 45, 1–3. [Google Scholar] [CrossRef]
- Rogowski, J.; Kubiak, A.; Andrzejczuk, M. Carbothermal reduction of SiO2 promoted with tungsten and morphology of WC-W2C-β-SiC nanostructured composite material. Appl. Surf. Sci. 2015, 359, 177–187. [Google Scholar] [CrossRef]
- Terracciano, A.C.; Oliveira, S.D.; Siddhanti, D.; Blair, R.; Vasu, S.S.; Orlovskaya, N. Pd enhanced WC catalyst to promote heterogeneous methane combustion. Appl. Therm. Eng. 2017, 114, 663–672. [Google Scholar] [CrossRef]
- Wang, F.; He, P.; Li, Y.; Shifa, T.A.; Deng, Y.; Liu, K.; Wang, Q.; Wang, F.; Wen, Y.; Wang, Z.; et al. Interface engineered WxC@WS2 nanostructure for enhanced hydrogen evolution catalysis. Adv. Funct. Mater. 2017, 27, 1605802. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, F.; Jia, Y.; Yang, N.; Zhang, X. One-step ethanolysis of lignin into small-molecular aromatic hydrocarbons over nano-SiC catalyst. Bioresour. Technol. 2017, 226, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Qin, X.; Zhu, D.; Meng, Y.; Yang, L.; Sun, L.; Ming, Y. FeCl2-assisted synthesis and photoluminescence of β-SiC nanowires. Mater. Sci. Semicond. Process. 2015, 29, 155–160. [Google Scholar] [CrossRef]
- Liang, C.H.; Meng, G.W.; Zhang, L.D.; Wu, Y.C.; Cui, Z. Large-scale synthesis of β-SiC nanowires by using mesoporous silica embedded with Fe nanoparticles. Chem. Phys. Lett. 2000, 329, 323–328. [Google Scholar] [CrossRef]
- Attolini, G.; Rossi, F.; Negri, M.; Dhanabalan, S.C.; Bosi, M.; Boschi, F.; Lagonegro, P.; Lupo, P.; Salviati, G. Growth of SiC NWs by vapor phase technique using Fe as catalyst. Mater. Lett. 2014, 124, 169–172. [Google Scholar] [CrossRef]
- Chen, K.; Fang, M.; Huang, Z.; Huang, J.; Liu, Y. Catalytic synthesis and growth mechanism of SiC@SiO2 nanowires and their photoluminescence properties. Cryst. Eng. Commun. 2013, 15, 9032. [Google Scholar] [CrossRef]
- Wu, Z.S.; Deng, S.Z.; Xu, N.S.; Chen, J.; Zhou, J.; Chen, J. Needle-shaped silicon carbide nanowires: Synthesis and field electron emission properties. Appl. Phys. Lett. 2002, 80, 3829. [Google Scholar] [CrossRef]
- Yu, W.; Zheng, Y.; Yang, E.; Qiu, J.; Lan, R. Synthesis of β-SiC/SiO2 core-shell nanowires with the assistance of cerium oxide. J. Rare Earth 2010, 28, 365–368. [Google Scholar] [CrossRef]
- Meng, A.; Li, Z.; Zhang, J.; Gao, L.; Li, H. Synthesis and Raman scattering of β-SiC/SiO2 core–shell nanowires. J. Cryst. Growth 2007, 308, 263–268. [Google Scholar] [CrossRef]
- Li, Z.; Gao, W.; Meng, A.; Geng, Z.; Wan, L. Effects of Fe and Ni on the yield and morphology of the 1D SiC nanostructures prepared by chemical vapor reaction. J. Cryst. Growth 2008, 310, 4401–4406. [Google Scholar] [CrossRef]
- Wu, R.; Yang, G.; Gao, M.; Li, B.; Chen, J.; Zhai, R.; Pan, Y. Growth of SiC nanowires from NiSi solution. Cryst. Growth Des. 2009, 9, 100–104. [Google Scholar] [CrossRef]
- Senthil, K.; Yong, K. Enhanced field emission from density-controlled SiC nanowires. Mater. Chem. Phys. 2008, 112, 88–93. [Google Scholar] [CrossRef]
- Wu, R.; Chen, J.; Yang, G.; Wu, L.; Zhou, S.; Wang, J.; Pan, Y. Self-assembled one-dimensional hierarchical SiC nanostructures: Microstructure, growth mechanism, and optical properties. J. Cryst. Growth 2008, 310, 3573–3578. [Google Scholar] [CrossRef]
- Neumann, M.; Nöske, R.; Taubert, A.; Tiersch, B.; Strauch, P. Highly structured, biomorphous β-SiC with high specific surface area from Equisetaceae. J. Mater. Chem. 2012, 22, 9046–9051. [Google Scholar] [CrossRef]
- Qian, J.-M.; Jin, Z.-H. Preparation and characterization of porous, biomorphic SiC ceramic with hybrid pore structure. J. Eur. Ceram. Soc. 2006, 26, 1311–1316. [Google Scholar] [CrossRef]
- Merle-Mbjean, T.; Abdelmounim, E.; Quintard, P. Oxide layer on silicon carbide powder: A FT-IR investigation. J. Mol. Struct. 1995, 349, 105–108. [Google Scholar] [CrossRef]
- Ryu, Z.; Zheng, J.; Wang, M.; Zhang, B. Preparation and characterization of silicon carbide fibers from activated carbon fibers. Carbon 2002, 40, 715–720. [Google Scholar] [CrossRef]
- Preiss, H.; Berger, L.M.; Braun, M. Formation of black glasses and silicon carbide from binary carbonaceous/silica hydrogels. Carbon 1995, 33, 1739–1746. [Google Scholar] [CrossRef]
- Raman, V.; Bahl, O.P.; Dhawan, U. Synthesis of silicon carbide through the sol-gel process from different precursors. J. Mater. Sci. 1995, 30, 2686–2693. [Google Scholar] [CrossRef]
- Lin, L. Synthesis and optical property of large-scale centimetres-long silicon carbide nanowires by catalyst-free CVD route under superatmospheric pressure conditions. Nanoscale 2011, 3, 1582–1591. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Li, J.C.; Zhou, H.; Wang, H.; Wang, B.; Yan, H. Parameters determining crystallinity in β-SiC thin films prepared by catalytic chemical vapor deposition. J. Cryst. Growth 2004, 260, 176–180. [Google Scholar] [CrossRef]
- Dhage, S.; Lee, H.C.; Hassan, M.S.; Akhtar, M.S.; Kim, C.Y.; Sohn, J.M.; Kim, K.J.; Shin, H.S.; Yang, O.B. Formation of SiC nanowhiskers by carbothermic reduction of silica with activated carbon. Mater. Lett. 2009, 63, 174–176. [Google Scholar] [CrossRef]
- Ye, J.; Zhang, S.; Lee, W.E. Novel low temperature synthesis and characterisation of hollow silicon carbide spheresMicropor. Microporous Mesoporous Mater. 2012, 152, 25–30. [Google Scholar] [CrossRef]
Sample Availability: Samples of WxC-β-SiC nanocomposite catalysts are available from the authors. |




| Catalyst | Furfural Conversion (%) | Yield (%) | ||
|---|---|---|---|---|
| FA | THFA | Other Products * | ||
| β-SiC | 100 | 54.8 | 43.2 | 0 |
| (5)WxC-β-SiC | 100 | 49.7 | 48.9 | 1.3 |
| (10)WxC-β-SiC | 100 | 8.4 | 90.5 | 1.1 |
| (20)WxC-β-SiC | 67.6 | 42.2 | 24.6 | 0.3 |
| (10)WxC-β-SiC(A) | 100 | 53.2 | 46.8 | 0.4 |
| (10)WxC-β-SiC(B) | 69.4 | 58.1 | 11.3 | 0.1 |
| (10)WxC-β-SiC(C) | 35.1 | 29.6 | 5.9 | 0.2 |
| β-SiC(A) | 71.9 | 53.9 | 18.0 | 0.5 |
| β-SiC(B) | 73.4 | 60.7 | 12.7 | 0.3 |
| Catalyst | Temp. of Carbothermal Reduction (°C) | Time of Carbothermal Reduction (h) | Crystalline Phases | Size of SiC Crystallites (nm) | Size of WC Crystallites (nm) |
|---|---|---|---|---|---|
| β-SiC, | 1550 | 1.5 | β-SiC, SiO2 | 36 | - |
| (5)WxC-β-SiC | 1550 | 1.5 | β-SiC,SiC, WC, W2C, SiO2 | 37 | 50 |
| (10)WxC-β-SiC | 1550 | 1.5 | β-SiC,SiC, WC, W2C, SiO2 | 23 | 32 |
| (20)WxC-β-SiC | 1550 | 1.5 | β-SiC,SiC, WC, W2C, SiO2 | 18 | 47 |
| (10)WxC-β-SiC(A) | 1550 | 3.0 | β-SiC,SiC, WC, W2C | 35 | 37 |
| (10)WxC-β-SiC(B) | 1550 | 4.5 | β-SiC,SiC, WC, W2C | 37 | 42 |
| (10)WxC-β-SiC(C) | 1550 | 1.5 | β-SiC,SiC, WC, W2C | 25 | 23 |
| β-SiC(A) | 1550 | 3.0 | β-SiC | 36 | - |
| β-SiC(B) | 1550 | 1.5 | β-SiC | 20 | - |
| Catalyst | W wt % in the Starting Mixture | Surface Area (m2/g) | Total Pore Volume (cm3/g) | Average Pore Radius (nm) |
|---|---|---|---|---|
| β-SiC | 0 | 9.6 | 0.034 | 8.59 |
| (5)WxC-β-SiC | 5 | 45.4 | 0.145 | 6.17 |
| (10)WxC-β-SiC | 10 | 57.4 | 0.184 | 6.10 |
| (20)WxC-β-SiC | 20 | 10.0 | 0.035 | 6.67 |
| (10)WxC-β-SiC(A) | 10 | 29.2 | - | - |
| (10)WxC-β-SiC(B) | 10 | 24.9 | - | - |
| (10)WxC-β-SiC(C) | 10 | 31.0 | - | - |
| β-SiC(A) | 0 | 9.6 | - | - |
| β-SiC(B) | 0 | 11.2 | - | - |
| Origin of Furfural | Furfural Concentration (mol·L−1) | Conversion of Furfural (%) | Yield (%) | ||
|---|---|---|---|---|---|
| FA | THFA | Other Products * | |||
| COMMERCIAL, POCh Gliwice S.A. | 0.10 | 100 | 8.4 | 90.5 | 1.1 |
| Acidic hydrolysis of oat straw | 0.11 | 100 | 8.6 | 89.9 | 1.5 |
| Acidic hydrolysis of flax straw | 0.08 | 100 | 7.1 | 92.3 | 0.6 |
| Acidic hydrolysis of sugar beet pulp | 0.06 | 100 | 9.1 | 89.8 | 1.1 |
| Acidic hydrolysis of sugar beet leaves | 0.02 | 100 | 10.5 | 86.2 | 3.3 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rogowski, J.; Andrzejczuk, M.; Berlowska, J.; Binczarski, M.; Kregiel, D.; Kubiak, A.; Modelska, M.; Szubiakiewicz, E.; Stanishevsky, A.; Tomaszewska, J.; et al. WxC-β-SiC Nanocomposite Catalysts Used in Aqueous Phase Hydrogenation of Furfural. Molecules 2017, 22, 2033. https://doi.org/10.3390/molecules22112033
Rogowski J, Andrzejczuk M, Berlowska J, Binczarski M, Kregiel D, Kubiak A, Modelska M, Szubiakiewicz E, Stanishevsky A, Tomaszewska J, et al. WxC-β-SiC Nanocomposite Catalysts Used in Aqueous Phase Hydrogenation of Furfural. Molecules. 2017; 22(11):2033. https://doi.org/10.3390/molecules22112033
Chicago/Turabian StyleRogowski, Jacek, Mariusz Andrzejczuk, Joanna Berlowska, Michal Binczarski, Dorota Kregiel, Andrzej Kubiak, Magdalena Modelska, Elzbieta Szubiakiewicz, Andrei Stanishevsky, Jolanta Tomaszewska, and et al. 2017. "WxC-β-SiC Nanocomposite Catalysts Used in Aqueous Phase Hydrogenation of Furfural" Molecules 22, no. 11: 2033. https://doi.org/10.3390/molecules22112033
APA StyleRogowski, J., Andrzejczuk, M., Berlowska, J., Binczarski, M., Kregiel, D., Kubiak, A., Modelska, M., Szubiakiewicz, E., Stanishevsky, A., Tomaszewska, J., & Witonska, I. A. (2017). WxC-β-SiC Nanocomposite Catalysts Used in Aqueous Phase Hydrogenation of Furfural. Molecules, 22(11), 2033. https://doi.org/10.3390/molecules22112033








