Transformation of a Thermostable G-Quadruplex Structure into DNA Duplex Driven by Reverse Gyrase
Abstract
:1. Introduction
2. Results and Discussion
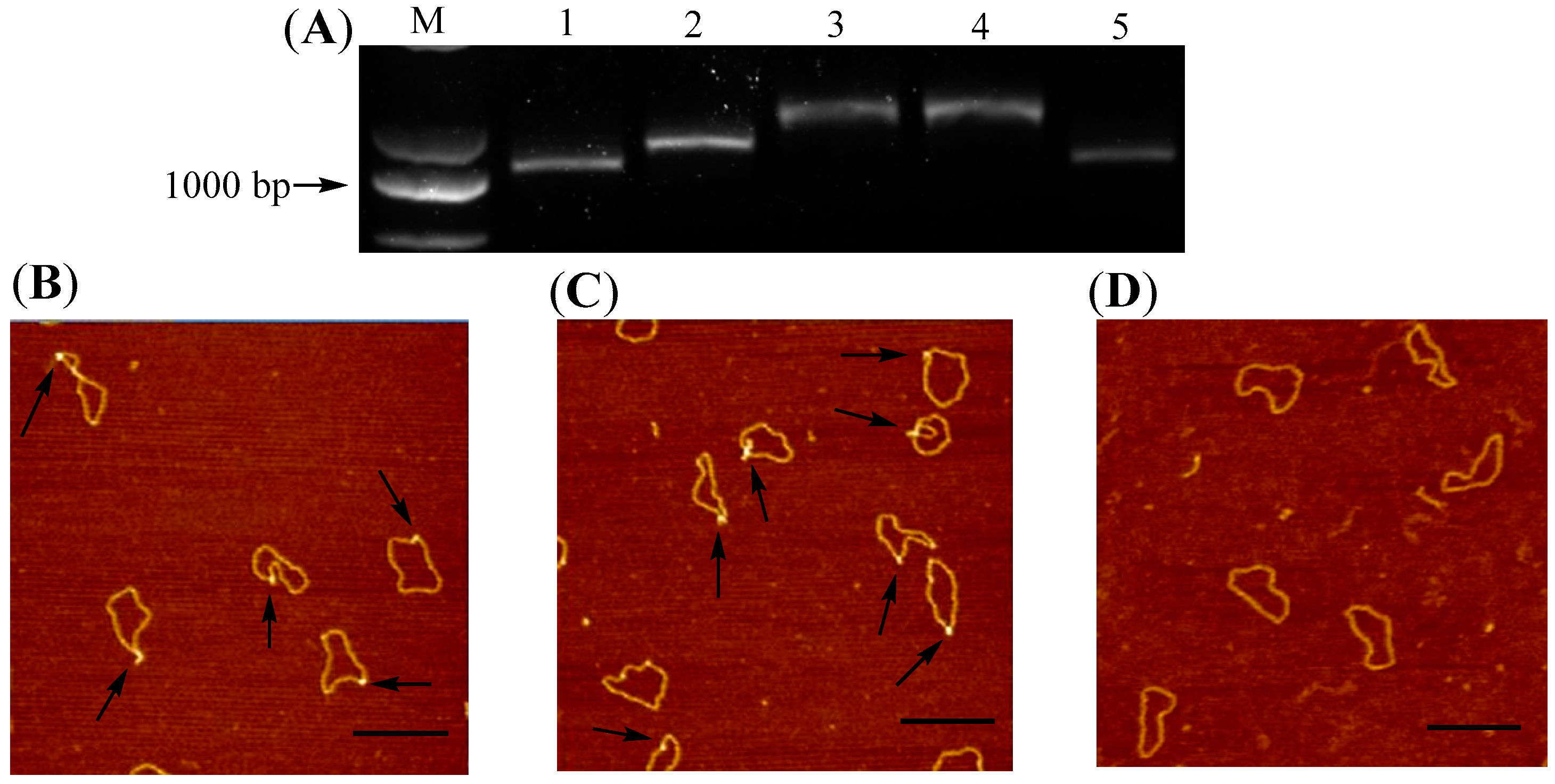
2.1. Synthesis and Structural Confirmation of G-Quadruplex-Containing DNA 1
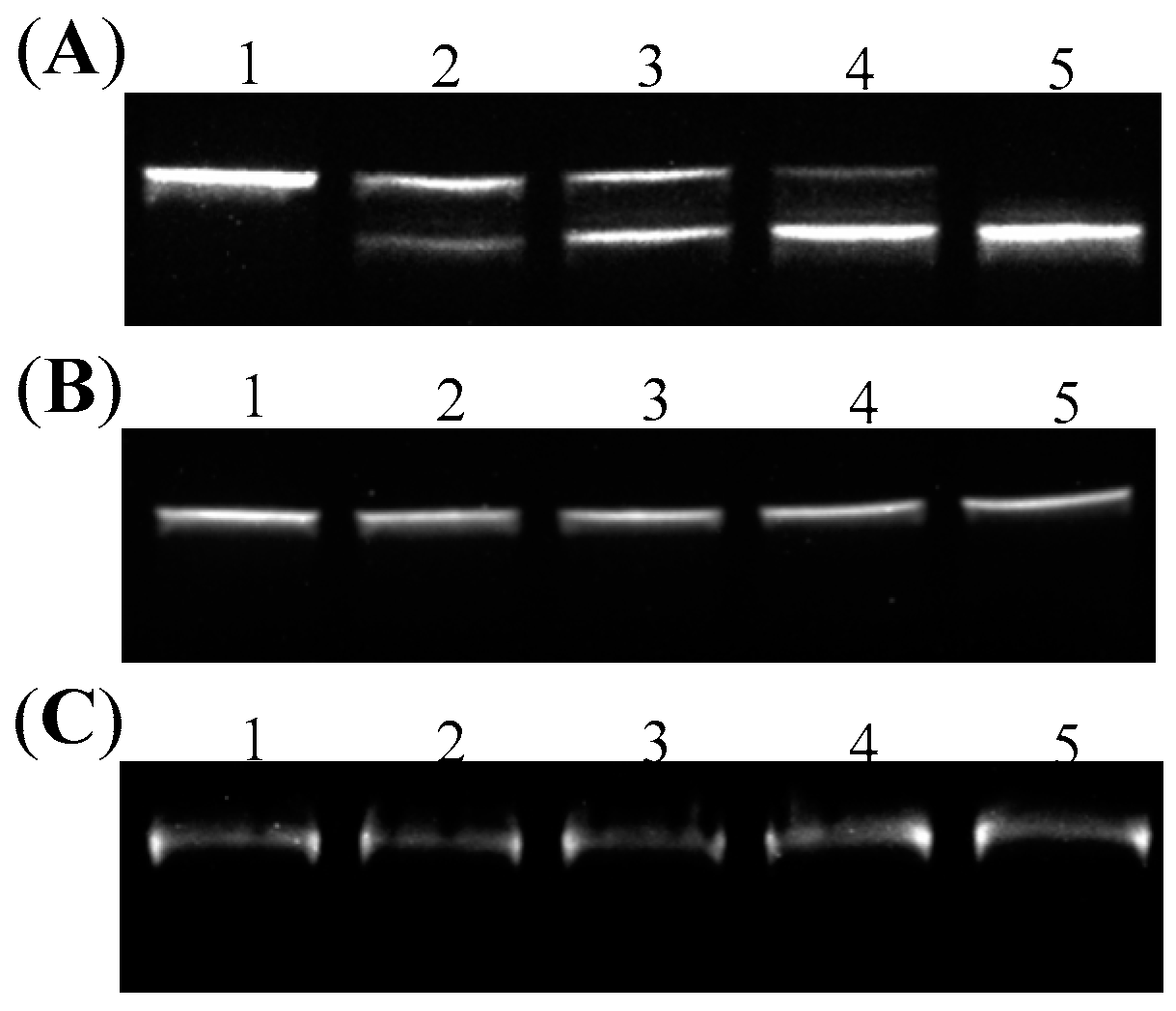
2.2. Introduction of Positive Supercoils and Dissociation of G-Quadruplex Driven by Reverse Gyrase
2.3. Comparison of Structural Parameters in AFM Images
2.4. Endonuclease Assays on DNA Topoisomers with or without G-Quadruplex
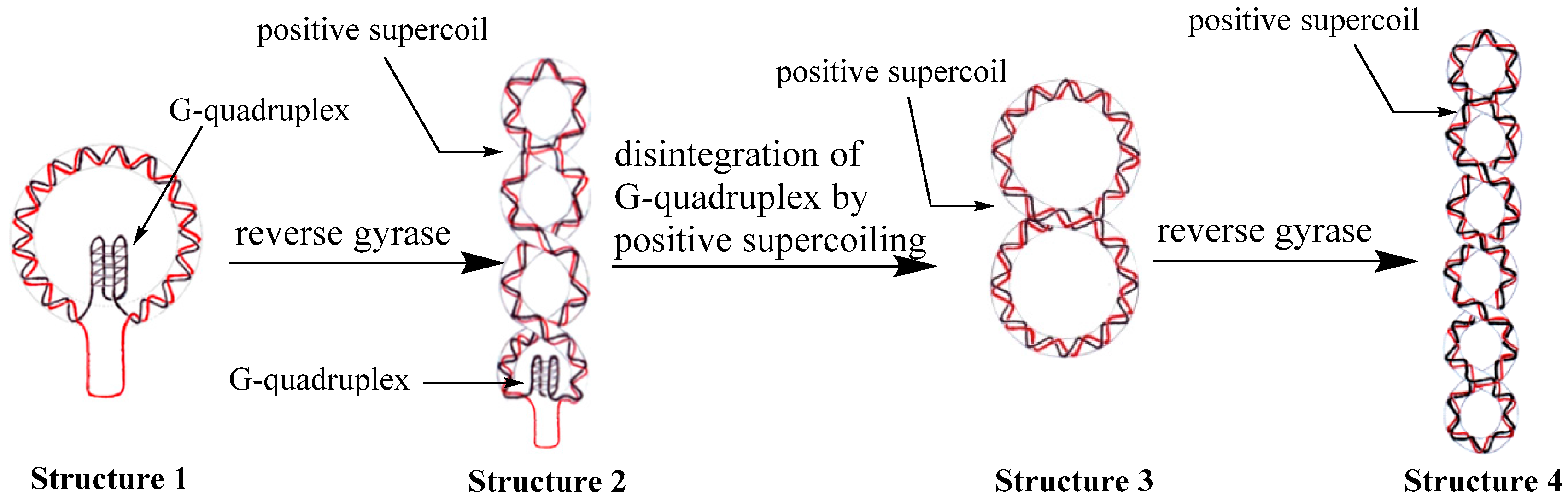
2.5. A Purposed Mechanism for Disintegration of G-Quadruplex Structures Driven by Reverse Gyrase
3. Materials and Methods
3.1. Reagents and General Information
3.2. Reactions of Nicking Endonuclease with Mini-Plasmid DNA
3.3. G-Quadruplex Formation from Duplex under Molecular Crowded Condition
3.4. Ligation Reaction for Sealing the Nicking Site
3.5. Reactions of Reverse Gyrase with Mini-Plasmid DNA
3.6. Reactions of Topo I with Supercoiledmini-Plasmid
3.7. Reactions of T7 Endonuclease I with Supercoiled-Plasmid
3.8. Gel Electrophoresis
3.9. Experimental Procedures for DNA Sample Preparations and AFM Examination
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kikuchi, A.; Asai, K. Reverse gyrase—A topoisomerase which introduces positive superhelical turns into DNA. Nature 1984, 309, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Nakasu, S.; Kikuchi, A. Reverse gyrase; ATP-dependent type I topoisomerase from Sulfolobus. EMBO J. 1985, 4, 2705–2710. [Google Scholar] [PubMed]
- Perugino, G.; Valenti, A.; D’Amaro, A.; Rossi, M.; Ciaramella, M. Reverse gyrase and genome stability in hyperthermophilic organisms. Biochem. Soc. Trans. 2009, 37, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Lulchev, P.; Klostermeier, D. Reverse gyrase—Recent advances and current mechanistic understanding of positive DNA supercoiling. Nucleic Acid Res. 2014, 42, 8200–8213. [Google Scholar] [CrossRef] [PubMed]
- Nadal, M. Reverse gyrase: An insight into the role of DNA-topoisomerases. Biochimie 2007, 89, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Vettone, A.; Perugino, G.; Rossi, M.; Valenti, A.; Ciaramella, M. Genome stability: Recent insights in the topoisomerase reverse gyrase and thermophilic DNA alkyltransferase. Extremophiles 2014, 18, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Heine, M.; Chandra, S.B. The linkage between reverse gyrase and hyperthermophiles: A review of their invariable association. J. Microbiol. 2009, 47, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Confalonieri, F.; Elie, C.; Nadal, M.; de La Tour, C.; Forterre, P.; Duguet, M. Reverse gyrase: A helicase-like domain and a type I topoisomerase in the same polypeptide. Proc. Natl. Acad. Sci. USA 1993, 90, 4753–4757. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Bacolla, A.; Wang, G.; Vasquez, K.M. Non-B DNA structure-induced genetic instability and evolution. Cell. Mol. Life Sci. 2010, 67, 43–62. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Majima, T. Conformational changes of non-B DNA. Chem. Soc. Rev. 2011, 40, 5893–5909. [Google Scholar] [CrossRef] [PubMed]
- Bacolla, A.; Wells, R.D. Non-B DNA conformations as determinants of mutagenesis and human disease. Mol. Carcinog. 2009, 48, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Boyer, A.S.; Grgurevic, S.; Cazaux, C.; Hoffmann, J.S. The human specialized DNA polymerases and non-B DNA: Vital relationships to preserve genome integrity. J. Mol. Biol. 2013, 425, 4767–4781. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Gertz, E.M.; Wojtowicz, D.; Zhabinskaya, D.; Levens, D.; Benham, C.J.; Schaffer, A.A.; Przytycka, T.M. Potential non-B DNA regions in the human genome are associated with higher rates of nucleotide mutation and expression variation. Nucleic Acid Res. 2014, 42, 12367–12379. [Google Scholar] [CrossRef] [PubMed]
- Bacolla, A.; Tainer, J.A.; Vasquez, K.M.; Cooper, D.N. Translocation and deletion breakpoints in cancer genomes are associated with potential non-B DNA-forming sequences. Nucleic Acid Res. 2016, 44, 5673–5688. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lv, B.; Zhang, H.; Lee, J.Y.; Li, T. Positive supercoiling affiliated with nucleosome formation repairs non-B DNA structures. Chem. Commun. 2014, 50, 10641–10644. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lv, B.; Zhang, H.; Lee, J.Y.; Li, T. Disintegration of cruciform and G-quadruplex structures during the course of helicase-dependent amplification (HDA). Bioorg. Med. Chem. Lett. 2015, 25, 1709–1714. [Google Scholar] [CrossRef] [PubMed]
- D’Amaro, A.; Rossi, M.; Ciaramella, M. Reverse gyrase: An unusual DNA manipulator of hyperthermophilic organisms. Ital. J. Biochem. 2007, 56, 103–109. [Google Scholar] [PubMed]
- Krah, R.; Kozyavkin, S.A.; Slesarev, A.I.; Gellert, M. A two-subunit type I DNA topoisomerase (reverse gyrase) from an extreme hyperthermophile. Proc. Natl. Acad. Sci. USA 1996, 93, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Lipps, H.J.; Rhodes, D. G-quadruplex structures: In vivo evidence and function. Trends Cell Biol. 2009, 19, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Simonsson, T. G-quadruplex DNA structures—Variations on a theme. Biol. Chem. 2001, 382, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Bochman, M.L.; Paeschke, K.; Zakian, V.A. DNA secondary structures: Stability and function of G-quadruplex structures. Nat. Rev. Genet. 2012, 13, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Hurley, L.H. Structure of the biologically relevant G-quadruplex in the c-MYC promoter. Nucl. Nucleosides, Nucleotides Nucleic Acids 2006, 25, 951–968. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P.; Chattopadhyay, S.; Chatterjee, S. G-Quadruplex surveillance in BCL-2 gene: A promising therapeutic intervention in cancer treatment. Drug Discov. Today 2017, 22, 1165–1186. [Google Scholar] [CrossRef] [PubMed]
- Huppert, J.L. Four-stranded nucleic acids: Structure, function and targeting of G-quadruplexes. Chem. Soc. Rev. 2008, 37, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.N.; Chaires, J.B.; Gray, R.D.; Trent, J.O. Stability and kinetics of G-quadruplex structures. Nucleic Acid Res. 2008, 36, 5482–5515. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wei, C.; Jia, G.; Wang, X.; Feng, Z.; Li, C. Human telomeric G-quadruplex formed from duplex under near physiological conditions: Spectroscopic evidence and kinetics. Biochimie 2009, 91, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Miyoshi, D.; Matsumura, S.; Nakano, S.; Sugimoto, N. Duplex dissociation of telomere DNAs induced by molecular crowding. J. Am. Chem. Soc. 2004, 126, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Kan, Z.Y.; Lin, Y.; Wang, F.; Zhuang, X.Y.; Zhao, Y.; Pang, D.W.; Hao, Y.H.; Tan, Z. G-quadruplex formation in human telomeric (TTAGGG) 4 sequence with complementary strand in close vicinity under molecularly crowded condition. Nucleic Acid Res. 2007, 35, 3646–3653. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, N.; Allan, J. Supercoiling in DNA and chromatin. Curr. Opin. Genet. Dev. 2014, 25, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Corless, S.; Gilbert, N. Effects of DNA supercoiling on chromatin architecture. Biophys. Rev. 2016, 8, 51–64. [Google Scholar] [CrossRef] [PubMed]
- Lv, B.; Li, D.; Zhang, H.; Lee, J.Y.; Li, T. DNA gyrase-driven generation of a G-quadruplex from plasmid DNA. Chem. Commun. 2013, 49, 8317–8319. [Google Scholar] [CrossRef] [PubMed]
- Chaires, J.B. Human telomeric G-quadruplex: Thermodynamic and kinetic studies of telomeric quadruplex stability. FEBS J. 2010, 277, 1098–1106. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.L.; Mergny, J.L.; Alberti, P. Stability of telomeric G-quadruplexes. Nucleic Acid Res. 2011, 39, 3282–3294. [Google Scholar] [CrossRef] [PubMed]
- Bates, A.D.; Maxwell, A. DNA Topology, 2nd ed.; Oxford University Press: Oxford, UK, 2005; p. 198. [Google Scholar]
- Cozzarelli, N.R.; Wang, J.C. DNA Topology and Its Biological Effects; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1990; p. 480. [Google Scholar]
- Pyne, A.L.; Hoogenboom, B.W. Imaging DNA structure by atomic force microscopy. Methods Mol. Biol. 2016, 1431, 47–60. [Google Scholar] [PubMed]
- Lyubchenko, Y.L. DNA structure and dynamics: An atomic force microscopy study. Cell Biochem. Biophys. 2004, 41, 75–98. [Google Scholar] [CrossRef]
- Li, D.; Lv, B.; Wang, Q.; Liu, Y.; Zhuge, Q. Direct observation of positive supercoils introduced by reverse gyrase through atomic force microscopy. Bioorg. Med. Chem. Lett. 2017, 27, 4086–4090. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Zhang, Z.; Cao, K.; Huang, X. Visualization of alkali-denatured supercoiled plasmid DNA by atomic force microscopy. Biochem. Biophys. Res. Commun. 2008, 374, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Neaves, K.J.; Huppert, J.L.; Henderson, R.M.; Edwardson, J.M. Direct visualization of G-quadruplexes in DNA using atomic force microscopy. Nucleic Acid Res. 2009, 37, 6269–6275. [Google Scholar] [CrossRef] [PubMed]
- Sinden, R.R. DNA Structure and Function; Academic Press: San Diego, CA, USA, 1994; p. 398. [Google Scholar]
- Ogawa, T.; Yogo, K.; Furuike, S.; Sutoh, K.; Kikuchi, A.; Kinosita, K.J. Direct observation of DNA overwinding by reverse gyrase. Proc. Natl. Acad. Sci. USA 2015, 112, 7495–7500. [Google Scholar] [CrossRef] [PubMed]
- Lyubchenko, Y.L.; Shlyakhtenko, L.S. AFM for analysis of structure and dynamics of DNA and protein-DNA complexes. Methods 2009, 47, 206–213. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |

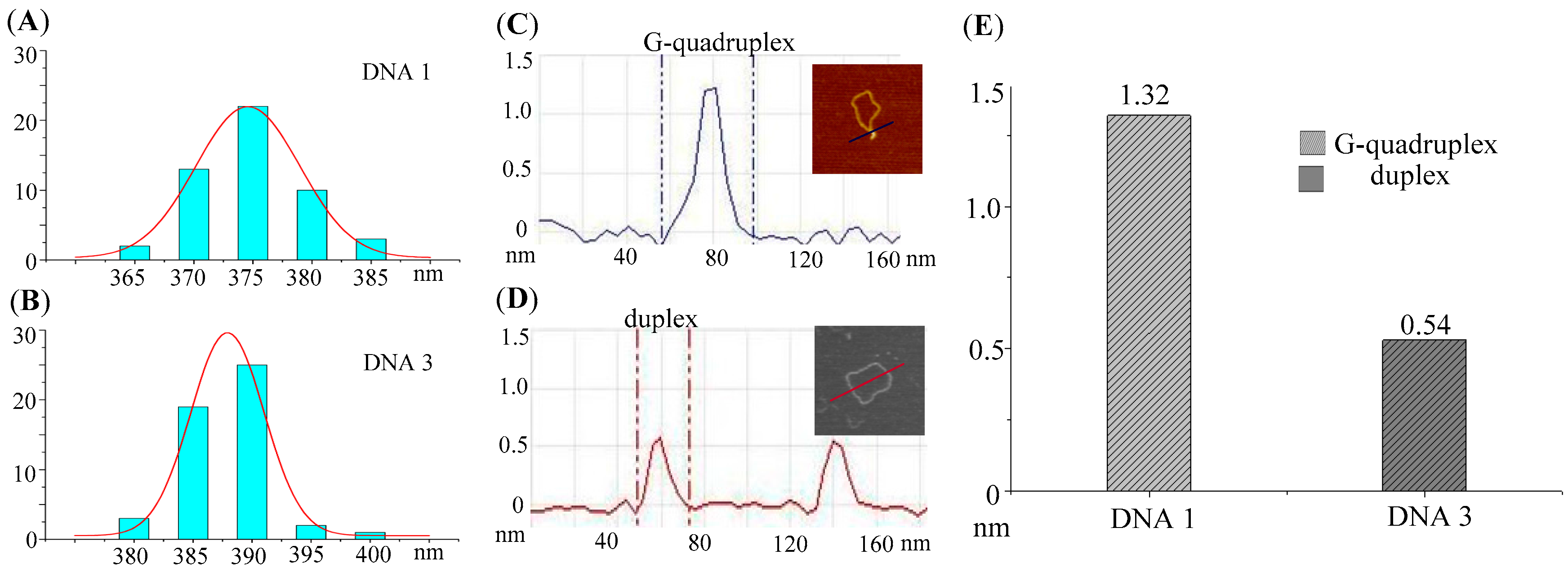
| Mini-Plasmid | Plasmids with G-Quadruplex (%) | Contour Lengtha | Height of Duplex | Height of G-Quadruplex | N |
|---|---|---|---|---|---|
| DNA 1 | 94 | 375.38 ± 3.3 nm | 0.54 ± 0.02 nm | 1.32 ± 0.04 nm | 50 |
| DNA 3 | 4 | 387.71 ± 3.0 nm | 0.54 ± 0.02 nm | N.A. | 50 |
| DNA C1 | 0 | 388.23 ± 2.1 nm | 0.53 ± 0.02 nm | N.A. | 50 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, D.; Wang, Q.; Liu, Y.; Liu, K.; Zhuge, Q.; Lv, B. Transformation of a Thermostable G-Quadruplex Structure into DNA Duplex Driven by Reverse Gyrase. Molecules 2017, 22, 2021. https://doi.org/10.3390/molecules22112021
Li D, Wang Q, Liu Y, Liu K, Zhuge Q, Lv B. Transformation of a Thermostable G-Quadruplex Structure into DNA Duplex Driven by Reverse Gyrase. Molecules. 2017; 22(11):2021. https://doi.org/10.3390/molecules22112021
Chicago/Turabian StyleLi, Dawei, Qiang Wang, Yun Liu, Kun Liu, Qiang Zhuge, and Bei Lv. 2017. "Transformation of a Thermostable G-Quadruplex Structure into DNA Duplex Driven by Reverse Gyrase" Molecules 22, no. 11: 2021. https://doi.org/10.3390/molecules22112021
APA StyleLi, D., Wang, Q., Liu, Y., Liu, K., Zhuge, Q., & Lv, B. (2017). Transformation of a Thermostable G-Quadruplex Structure into DNA Duplex Driven by Reverse Gyrase. Molecules, 22(11), 2021. https://doi.org/10.3390/molecules22112021





