Rapid Determination of Major Compounds in the Ethanol Extract of Geopropolis from Malaysian Stingless Bees, Heterotrigona itama, by UHPLC-Q-TOF/MS and NMR
Abstract
1. Introduction
2. Results and Discussion
2.1. Identification of Compounds from EEGP by UHPLC-Q-TOF/MS
2.1.1. Identification of Compounds in EEGP Based on Known Authentic Standards
2.1.2. Identification of Compounds in EEGP using METLIN and MSC Software
2.2. Identification of Unknown Compounds using Preparative HPLC (PHPLC) and NMR
3. Materials and Methods
3.1. Chemicals
3.2. UHPLC-Q-TOF/MS Analysis of EEGP
3.2.1. Sample Preparation
3.2.2. UHPLC System and Mass Spectrometry
3.3. PHPLC Purification and NMR Analysis for Identification of Unknown Compounds in EEGP
3.3.1. PHPLC
3.3.2. NMR
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dutra, R.P.; Nogueira, A.M.C.; de Oliveira Marques, R.R.; Costa, M.C.P.; Ribeiro, M.N.S. Avaliação farmacognóstica de geoprópolis de Melipona fasciculata Smith da Baixada maranhense, Brasil. Rev. Bras. Farmacogn. 2008, 18, 557–562. [Google Scholar] [CrossRef]
- Dutra, R.P.; Abreu, B.V.; Cunha, M.S.; Batista, M.C.; Torres, L.M.; Nascimento, F.R.; Ribeiro, M.N.; Guerra, R.N. Phenolic acids, hydrolyzable tannins, and antioxidant activity of geopropolis from the stingless bee Melipona fasciculata Smith. J. Agric. Food Chem. 2014, 62, 2549–2557. [Google Scholar] [CrossRef] [PubMed]
- Quezada-Euán, J.J.G.; de Jesús May-Itzá, W.; González-Acereto, J.A. Meliponiculture in Mexico: Problems and perspective for development. Bee World 2001, 82, 160–167. [Google Scholar] [CrossRef]
- Araujo, M.J.; Bosco, S.M.; Sforcin, J.M. Pythium insidiosum: Inhibitory effects of propolis and geopropolis on hyphal growth. Braz. J. Microbiol. 2016, 47, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Liberio, S.A.; Pereira, A.L.; Dutra, R.P.; Reis, A.S.; Araujo, M.J.; Mattar, N.S.; Silva, L.A.; Ribeiro, M.N.; Nascimento, F.R.; Guerra, R.N.; et al. Antimicrobial activity against oral pathogens and immunomodulatory effects and toxicity of geopropolis produced by the stingless bee Melipona fasciculata Smith. BMC Complement. Altern. Med. 2011, 11. [Google Scholar] [CrossRef] [PubMed]
- Duailibe, S.A.; Gonçalves, A.G.; Ahid, F.J. Effect of a propolis extract on streptococcus mutans counts in vivo. J. Appl. Oral Sci. 2007, 15, 420–423. [Google Scholar] [CrossRef] [PubMed]
- Da Cunha, M.G.; Franchin, M.; de Carvalho Galvão, L.C.; de Ruiz, A.L.; de Carvalho, J.E.; Ikegaki, M.; de Alencar, S.M.; Koo, H.; Rosalen, P.L. Antimicrobial and antiproliferative activities of stingless bee Melipona scutellaris geopropolis. BMC Complement. Altern. Med. 2013, 13, 23. [Google Scholar] [CrossRef] [PubMed]
- Cinegaglia, N.C.; Bersano, P.R.; Araujo, M.J.; Bufalo, M.C.; Sforcin, J.M. Anticancer effects of geopropolis produced by stingless bees on canine osteosarcoma cells in vitro. Evid. Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef]
- Araujo, M.; Bufalo, M.; Conti, B.; Fernandes, A.R.Y., Jr.; Trusheva, B.; Bankova, V.; Sforcin, J. The chemical composition and pharmacological activities of geopropolis produced by Melipona fasciculata Smith in northeast Brazil. J. Mol. Pathophysiol. 2015, 4, 12–20. [Google Scholar] [CrossRef]
- Franchin, M.; de Cunha, M.G.; Denny, C.; Napimoga, M.H.; Cunha, T.M.; Bueno-Silva, B.; de Alencar, M.S.; Ikegaki, M.; Luiz Rosalen, P. Bioactive Fraction of Geopropolis from Melipona. scutellaris Decreases Neutrophils Migration in the Inflammatory Process: Involvement of Nitric Oxide Pathway. Evid. Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.; Zakaria, A.J.; Ismail, Z.; Mohd, K.S. Antibacterial and Phenolic Content of Propolis Produced by Two Malaysian Stingless Bees, Heterotrigona itama and Geniotrigona thoracica. Int. J. Pharm. Phytochem. Res. 2016, 8, 156–161. [Google Scholar]
- Ibrahim, N.; Mohd Niza, N.F.S.; Mohd Rodi, M.M.; Zakaria, A.J.; Ismail, Z.; Mohd, K.S. Chemical and biological analyses of Malaysian stingless bee propolis extracts. Malays. J. Anal. Sci. 2016, 20, 413–422. [Google Scholar] [CrossRef]
- Wang, S.; Chen, P.; Xu, Y.; Li, X.; Fan, X. Characterization of the chemical constituents in Da-Huang-Gan-Cao-Tang by liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry and liquid chromatography coupled with ion trap mass spectrometry. J. Sep. Sci. 2014, 37, 1748–1761. [Google Scholar] [CrossRef] [PubMed]
- Nina, N.; Quispe, C.; Jimenez-Aspee, F.; Theoduloz, C.; Feresin, G.E.; Lima, B.; Leiva, E.; Schmeda-Hirschmann, G. Antibacterial Activity, Antioxidant Effect and Chemical Composition of Propolis from the Region del Maule, Central Chile. Molecules 2015, 20, 18144–18167. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.W.; Zhu, S.; Yoshimatsu, K.; Komatsu, K. MS/MS similarity networking accelerated target profiling of triterpene saponins in Eleutherococcus senticosus leaves. Food Chem. 2017, 227, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Qing, Z.X.; Zhao, H.; Tang, Q.; Mo, C.M.; Huang, P.; Cheng, P.; Yang, P.; Yang, X.Y.; Liu, X.B.; Zheng, Y.J.; et al. Systematic identification of flavonols, flavonol glycosides, triterpene and siraitic acid glycosides from Siraitia grosvenorii using high-performance liquid chromatography/quadrupole-time-of-flight mass spectrometry combined with a screening strategy. J. Pharm. Biomed. Anal. 2017, 138, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Velikova, M.; Bankova, V.; Marcucci, M.C.; Tsvetkova, I.; Kujumgiev, A. Chemical Composition and Biological Activity of Propolis from Brazilian Meliponinae. Z. Naturforsch. C 2000, 55, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.S. Comparison of propolis from Apis mellifera and Tetragonisca angustula. Apidologie 2003, 34, 291–298. [Google Scholar] [CrossRef]
- Massaro, F.C.; Brooks, P.R.; Wallace, H.M.; Russell, F.D. Cerumen of Australian stingless bees (Tetragonula carbonaria): Gas chromatography-mass spectrometry fingerprints and potential anti-inflammatory properties. Naturwissenschaften 2011, 98, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Araújo, K.S.; dos Santos Júnior, J.F.; Sato, M.O.; Finco, F.D.B.A.; Soares, I.M.; dos Santos Barbosa, R.; da Costa Alvim, T.; Ascêncio, S.D.; Mariano, S.M.B. Physicochemical properties and antioxidant capacity of propolis of stingless bees (Meliponinae) and Apisfrom two regions of Tocantins, Brazil. Acta Amaz. 2016, 46, 61–68. [Google Scholar] [CrossRef]
- De Souza, S.A.; Camara, C.A.; da Silva, E.M.S.; Silva, T.M. Composition and Antioxidant Activity of Geopropolis Collected by Melipona subnitida (Jandaira) Bees. Evid. Based Complement. Altern. Med. 2013. [Google Scholar] [CrossRef]
- Da Silva, E.C.C.; Muniz, M.P.; de Cássia Saraiva Nunomura, R.; Nunomura, S.M.; Zilse, G.A.C. Phenolic constituents and antioxidant activity of geopropolis from two species of amazonian stingless bees. Quim. Nova 2013, 36, 628–633. [Google Scholar] [CrossRef]
- Leong, K.H.; Looi, C.Y.; Loong, X.M.; Cheah, F.K.; Supratman, U.; Litaudon, M.; Mustafa, M.R.; Awang, K. Cycloart-24-ene-26-ol-3-one, a New Cycloartane Isolated from Leaves of Aglaia exima Triggers Tumour Necrosis Factor-Receptor 1-Mediated Caspase-Dependent Apoptosis in Colon Cancer Cell Line. PLoS ONE 2016, 11, e0152652. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Xu, J.; Zhang, J.; Liu, W.Y.; Xie, N.; Chen, L.; Feng, F.; Qu, W. Novel tirucallane triterpenoids from the stem bark of Toona sinensis. Fitoterapia 2016, 112, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Velikova, M.; Bankova, V.; Tsvetkova, I.; Kujumgiev, A.; Marcucci, M.C. Antibacterial ent-kaurene from Brazilian propolis of native stingless bees. Fitoterapia 2000, 71, 693–696. [Google Scholar] [CrossRef]
- Çelemli, Ö.G. Chemical Properties of Propolis Collected by Stingless Bees; Springer: New York, NY, USA, 2013; pp. 525–537. [Google Scholar]
- Torres-González, A.; López-Rivera, P.; Duarte-Lisci, G.; López-Ramírez, Á.; Correa-Benítez, A.; Rivero-Cruz, J.F. Analysis of Volatile Components from Melipona beecheii Geopropolis from Southeast Mexico by Headspace Solid-Phase Microextraction. Nat. Prod. Res. 2016, 30, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Sanpa, S.; Popova, M.; Bankova, V.; Tunkasiri, T.; Eitssayeam, S.; Chantawannakul, P. Antibacterial Compounds from Propolis of Tetragonula laeviceps and Tetrigona melanoleuca (Hymenoptera: Apidae) from Thailand. PLoS ONE 2015, 10, e0126886. [Google Scholar] [CrossRef] [PubMed]
- Pino, J.A.; Marbot, R.; Delgado, A.; Zumárraga, C.; Sauri, E. Volatile Constituents of Propolis from Honey Bees and Stingless Bees from Yucatán. J. Essent. Oil Res. 2006, 18, 53–56. [Google Scholar] [CrossRef]
- Qiu, X.; Zhang, J.; Huang, Z.; Zhu, D.; Xu, W. Profiling of phenolic constituents in Polygonum multiflorum Thunb by combination of ultra-high-pressure liquid chromatography with linear ion trap-Orbitrap mass spectrometry. J. Chromatogr. A 2013, 1292, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.X.; Ge, A.H.; Yu, X.A.; Jiao, X.C.; Li, J.; He, J.; Tian, J.; Liu, W.; Azietaku, J.T.; Zhang, B.L.; et al. Simultaneous determination of four phenolic acids and seven alkaloids in rat plasma after oral administration of traditional Chinese medicinal preparation Jinqi Jiangtang Tablet by LC-ESI-MS/MS. J. Pharm. Biomed. Anal. 2016, 117, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, D.; Zheng, S.; Wu, L.; Huang, L.; Chen, S. Ultra-performance liquid chromatography-quadrupole/time-of-flight mass spectrometry with multivariate statistical analysis for exploring potential chemical markers to distinguish between raw and processed Rheum palmatum. BMC Complement. Altern. Med. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Thiruvengadam, M.; Praveen, N.; Kim, E.H.; Kim, S.H.; Chung, I.M. Production of anthraquinones, phenolic compounds and biological activities from hairy root cultures of Polygonum multiflorum Thunb. Protoplasma 2014, 251, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Jerković, I.; Tuberoso, C.I.G.; Kuś, P.M.; Marijanović, Z.; Kranjac, M. Screening of Coffea spp. honey by different methodologies: Theobromine and caffeine as chemical markers. RSC Adv. 2014, 4, 60557–60562. [Google Scholar] [CrossRef]
- Xu, M.L.; Zheng, M.S.; Lee, Y.K.; Moon, D.C.; Lee, C.S.; Woo, M.H.; Jeong, B.S.; Lee, E.S.; Jahng, Y.; Chang, H.W.; et al. A new stilbene glucoside from the roots ofPolygonum multiflorum Thunb. Arch. Pharm. Res. 2006, 29, 946–951. [Google Scholar] [CrossRef] [PubMed]
- Jirovetz, L.; Buchbauer, G.; Stoilova, I.; Stoyanova, A.; Krastanov, A.; Schmidt, E. Chemical Composition and Antioxidant Properties of Clove Leaf Essential Oil. J. Agric. Food Chem. 2006, 54, 6303–6307. [Google Scholar] [CrossRef] [PubMed]
- Dhalwal, K.; Shinde, V.M.; Mahadik, K.R. Efficient and Sensitive Method for Quantitative Determination and Validation of Umbelliferone, Carvone and Myristicin in Anethum graveolens and Carum carvi Seed. Chromatographia 2007, 67, 163–167. [Google Scholar] [CrossRef]
- Gemiarto, A.T.; Ninyio, N.N.; Lee, S.W.; Logis, J.; Fatima, A.; Chan, E.W.; Lim, C.S. Isoprenyl caffeate, a major compound in manuka propolis, is a quorum-sensing inhibitor in Chromobacterium violaceum. Antonie Van Leeuwenhoek 2015, 108, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Han, Y.; Yao, J.; Li, X.; Li, Y.; Xu, P.; Xue, M. Structural elucidation of the metabolites of lapachol in rats by liquid chromatography-tandem mass spectrometry. J. Chromatogr. B 2014, 944, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.A.; Su, B.N.; Keller, W.J.; Mehta, R.G.; Kinghorn, A.D. Antioxidant Xanthones from the Pericarp of Garcinia mangostana (Mangosteen). J. Agric. Food Chem. 2006, 54, 2077–2082. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Zhao, H.; Yuan, X.; Zou, S.; Wang, Q.; Ma, C.; Ren, Z.; Wang, Y. In vitro Protective Effect of Ganoderol A Isolated from Ganadermalucidum Against Ultraviolet A Radiation and its Anti-inflammatory Properties. Trop. J. Pharm. Res. 2015, 14, 415–421. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2014, 31, 160–258. [Google Scholar] [CrossRef] [PubMed]
- Gu, Q.; Yi, X.; Zhang, Z.; Yan, H.; Shi, J.; Zhang, H. A facile method for simultaneous analysis of phytosterols, erythrodiol, uvaol, tocopherols and lutein in olive oils by LC-MS. Anal. Methods 2016, 8, 1373–1380. [Google Scholar] [CrossRef]
- Parvez, M.; Gul, W.; Yousaf, M.; Choudhary, M.I.; Khan, M.R. Taraxerone. Acta Crystallographica 1999, 55, 213–215. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |
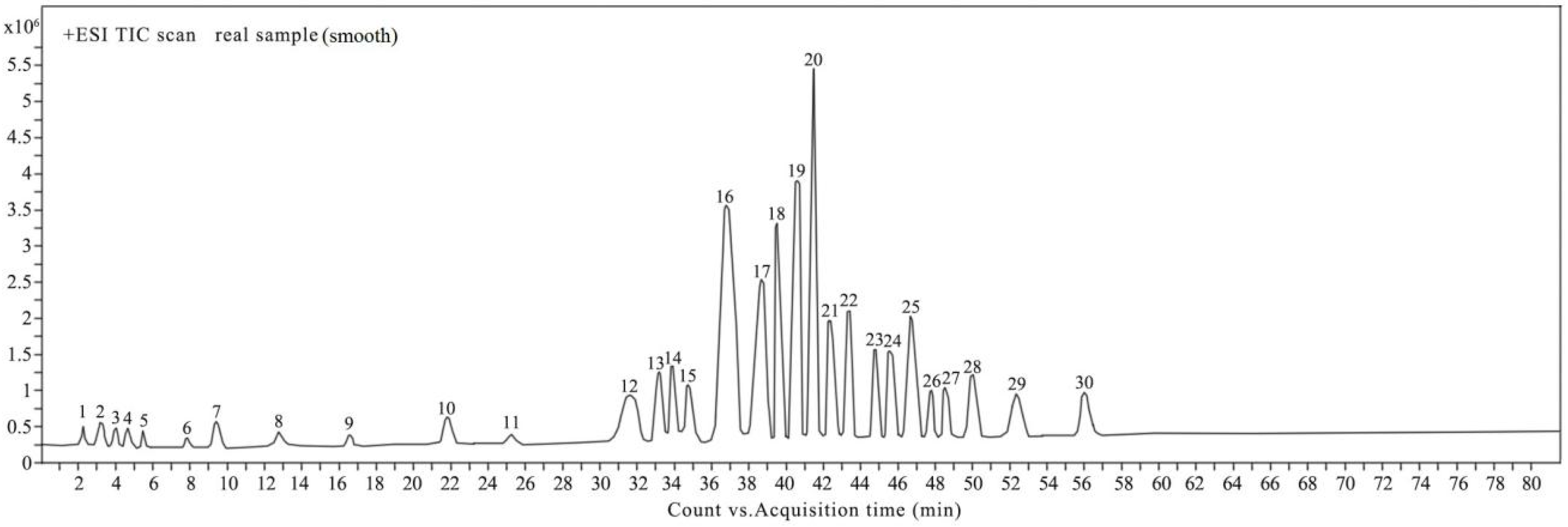




| Peak | Rt | m/z (−) | Error (ppm) | m/z (+) | Error (ppm) | Formula | MS/MS (m/z) (−) | MS/MS (m/z) (+) | Identification | Confirmation |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2.2 | 169.0144 | 1.18 | 171.0283 | −2.92 | C7H6O5 | 125.0234, 107.0128 | / | Gallic acid [17,30] | MSC and authentic standard |
| 2 | 3.1 | 179.0342 | −4.47 | 181.0489 | −3.31 | C9H8O4 | 135.0399,109.0301 | / | Caffeic acid [18] | MSC and authentic standard |
| 3 | 3.9 | 353.0876 | −0.56 | 355.1019 | −1.41 | C16H18O9 | 191.0129, 179.0488, 173.0004, 161.0535, 154.9881 | / | Caffeoylquinic acid [31] | MSC |
| 4 | 4.6 | 331.1031 | −1.21 | 333.1170 | −3.00 | C14H20O9 | 211.0224, 169.0133, 125.0232, 107.0127 | / | Gallic acid-hexose [32] | MSC |
| 5 | 5.5 | 167.0362 | 1.20 | 169.0491 | −2.37 | C8H8O4 | 153.0206, 108.0213 | / | Vanillic acid [33] | MSC |
| 6 | 7.9 | 167.0354 | 2.39 | / | C8H8O4 | 153.0211, 108.0214 | / | Isomer of vanillic acid [33] | MSC | |
| 7 | 9.3 | 195.0877 | 0 | C8H10N4O2 | / | 150.0888, 138.0668, 135.0477, 110. 0713 | Caffeine [34] | MSC | ||
| 8 | 12.8 | 151.0402 | 0.66 | 153.0543 | −1.96 | C8H8O3 | 137.0233, 123.0463, 107.0120 | / | Vanillin [33] | MSC |
| 9 | 16.5 | 197.0452 | −1.52 | 199.0611 | 5.02 | C9H10O5 | 153.0481, 124.0161, 107.0477, 106.0062 | / | Syringic acid [33] | MSC and authentic standard |
| 10 | 21.8 | 153.0192 | −0.65 | 155.0344 | 3.23 | C7H6O4 | 123.0416, 109.0285 | / | Protocatechuic acid [33] | MSC |
| 11 | 25.2 | 125.0244 | 0 | 127.0391 | 0.79 | C6H6O3 | 107.0130 | / | Pyrogallol [33] | MSC |
| 12 | 31.8 | 121.0293 | −1.65 | 123.0440 | −0.81 | C7H6O2 | 105.0349 | / | Benzoic acid | authentic standard |
| 13 | 33.1 | 583.1118 | 4.29 | / | C28H24O14 | 431.0997, 313.0578, 269.0467,169.0143, 125.0242 | / | Vitexin-O-gallate [35] | MSC | |
| 14 | 34.0 | 583.1120 | 4.63 | C28H24O14 | 431.0970, 313.0577, 269.0461, 169.0142, 125.0234 | / | Isomer of vitexin- O-gallate [35] | MSC | ||
| 15 | 34.9 | 205.0863 | −3.41 | 207.1014 | −0.97 | C12H14O3 | / | 189.0526, 150.0297, 149.0232, 122.0335 | Acetyleugenol [36] | MSC |
| 16 | 36.8 | 271.0613 | 0.37 | 273.0754 | −1.10 | C15H12O5 | 229.0477, 211.0348, 187.0375, 151.0012 | / | Pinobanksin | MSC and authentic standard |
| 17 | 38.4 | 161.0243 | −0.62 | 163.0396 | 3.68 | C9H6O3 | / | 135.0447, 133.0286, 107.0512, 105.0347 | Umbelliferone [37] | MSC |
| 18 | 39.7 | 285.0417 | 4.21 | 287.0557 | 2.44 | C15H10O6 | 255.0341, 239.0376, 227.0384, 211.0421, 199.0574, 124.0143, 107.0135 | / | Kaempferol [33] | MSC and authentic standard |
| 19 | 40.6 | 247.0972 | −1.62 | 249.1123 | 0.80 | C14H16O4 | 217.0884, 172.0888, 144.0939 | / | Prenyl caffeate [38] | MSC |
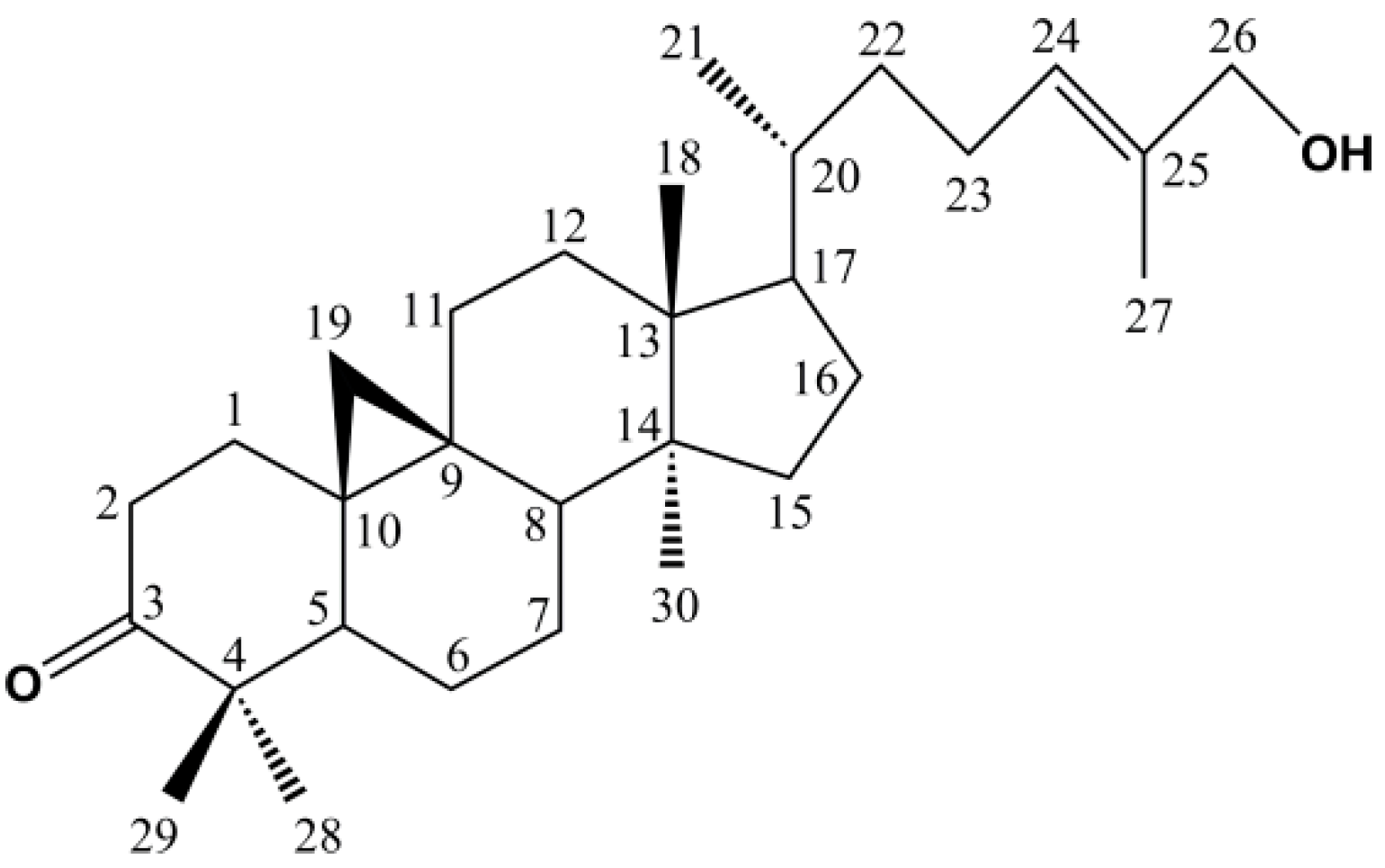
| 20 | 41.3 | 439.3583 | 0.23 | 441.3725 | −0.45 | C30H48O2 | / | / | 24(E)-cycloart-24-ene-26-ol-3-one [23] | NMR |
| 21 | 42.4 | 241.0874 | 1.66 | 243.1017 | 0.41 | C15H14O3 | / | 225.0560, 183.0806, 149.0597, 133.0665 | Lapachol [39] | MSC |
| 22 | 43.2 | 441.3736 | −0.45 | 443.3882 | −0.45 | C30H50O2 | / | / | 20-hydroxy-24-dammaren-3-one [24] | NMR |
| 23 | 44.9 | 407.1362 | 3.44 | 409.1488 | −1.22 | C20H24O9 | 245.0832, 230.0594, 202.0612, 187.0403, 173.0605, 137.0224 | / | Torachrysone-O-hexose [30] | MSC |
| 24 | 45.7 | 449.1462 | 2.00 | 451.1506 | −0.66 | C22H26O10 | 407.1322, 245.0482, 230.0065, 202.0516 | / | Torachrysone-O-(acetyl)-hexose [30] | MSC |
| 25 | 46.7 | 559.1467 | 1.79 | 561.1554 | −1.60 | C27H28O13 | 313.0575, 287.0938, 245.0827, 230.0596, 215.0367, 169.0143, 125.0232 | / | Torachrysone-O-(galloyl)-hexose [30] | MSC |
| 26 | 47.8 | 409.1656 | −0.24 | 411.1801 | −0.24 | C24H26O6 | / | 393.1688, 355.1167, 341.1009, 299.0541, 195.1013 | Mangostin [40] | MSC |
| 27 | 48.7 | 437.3424 | −0.23 | 439.3570 | −0.23 | C30H46O2 | / | 421.3449, 249.1849, 235.1698, 167.1074 | Ganoderol A [41] | MSC |
| 28 | 50.0 | 427.3582 | 0 | 429.3712 | −3.93 | C29H48O2 | / | 411.3611, 193.1608 | Saringosterol [42] | MSC |
| 29 | 52.3 | 411.3629 | −0.73 | 413.3757 | −5.08 | C29H48O | / | 395.3651, 135.1171 | Stigmasterol [43] | MSC |
| 30 | 56.0 | 423.3630 | −0.47 | 425.3777 | −0.24 | C30H48O | / | 407.3648, 271.2086, 135.1169, 109.1019 | Taraxerone [44] | MSC |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, L.; Yu, M.; Sun, M.; Xue, X.; Wang, T.; Cao, W.; Sun, L. Rapid Determination of Major Compounds in the Ethanol Extract of Geopropolis from Malaysian Stingless Bees, Heterotrigona itama, by UHPLC-Q-TOF/MS and NMR. Molecules 2017, 22, 1935. https://doi.org/10.3390/molecules22111935
Zhao L, Yu M, Sun M, Xue X, Wang T, Cao W, Sun L. Rapid Determination of Major Compounds in the Ethanol Extract of Geopropolis from Malaysian Stingless Bees, Heterotrigona itama, by UHPLC-Q-TOF/MS and NMR. Molecules. 2017; 22(11):1935. https://doi.org/10.3390/molecules22111935
Chicago/Turabian StyleZhao, Lingling, Mengjiao Yu, Minghui Sun, Xiaofeng Xue, Tongtong Wang, Wei Cao, and Liping Sun. 2017. "Rapid Determination of Major Compounds in the Ethanol Extract of Geopropolis from Malaysian Stingless Bees, Heterotrigona itama, by UHPLC-Q-TOF/MS and NMR" Molecules 22, no. 11: 1935. https://doi.org/10.3390/molecules22111935
APA StyleZhao, L., Yu, M., Sun, M., Xue, X., Wang, T., Cao, W., & Sun, L. (2017). Rapid Determination of Major Compounds in the Ethanol Extract of Geopropolis from Malaysian Stingless Bees, Heterotrigona itama, by UHPLC-Q-TOF/MS and NMR. Molecules, 22(11), 1935. https://doi.org/10.3390/molecules22111935





