Synthesis, Single Crystal X-ray Analysis, and Antifungal Profiling of Certain New Oximino Ethers Bearing Imidazole Nuclei
Abstract
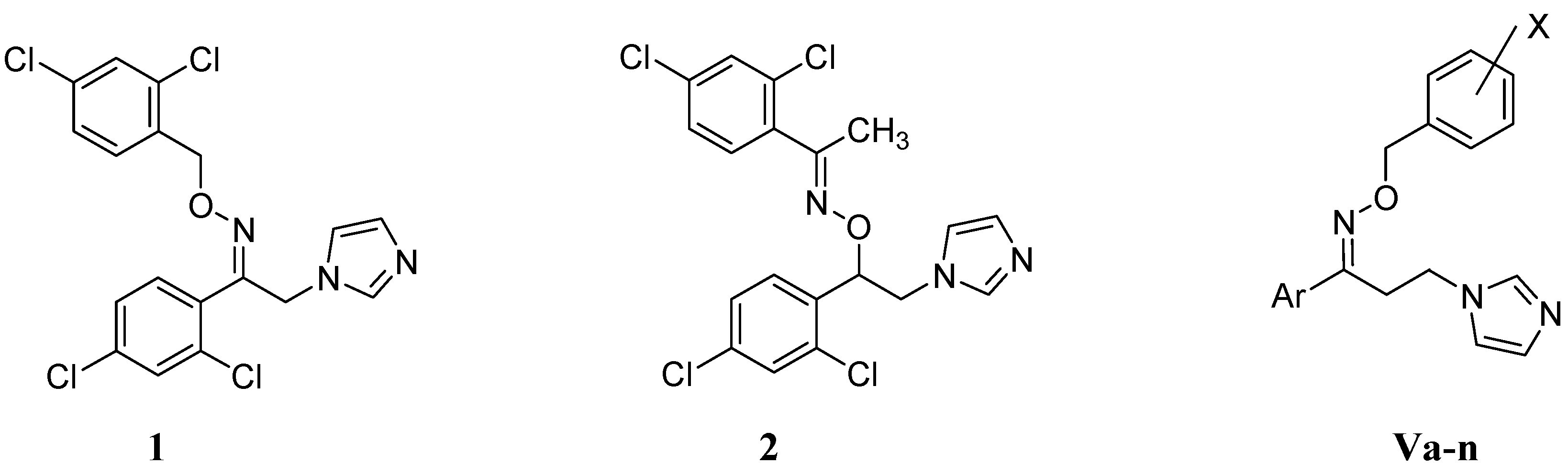
:1. Introduction
2. Results and Discussion
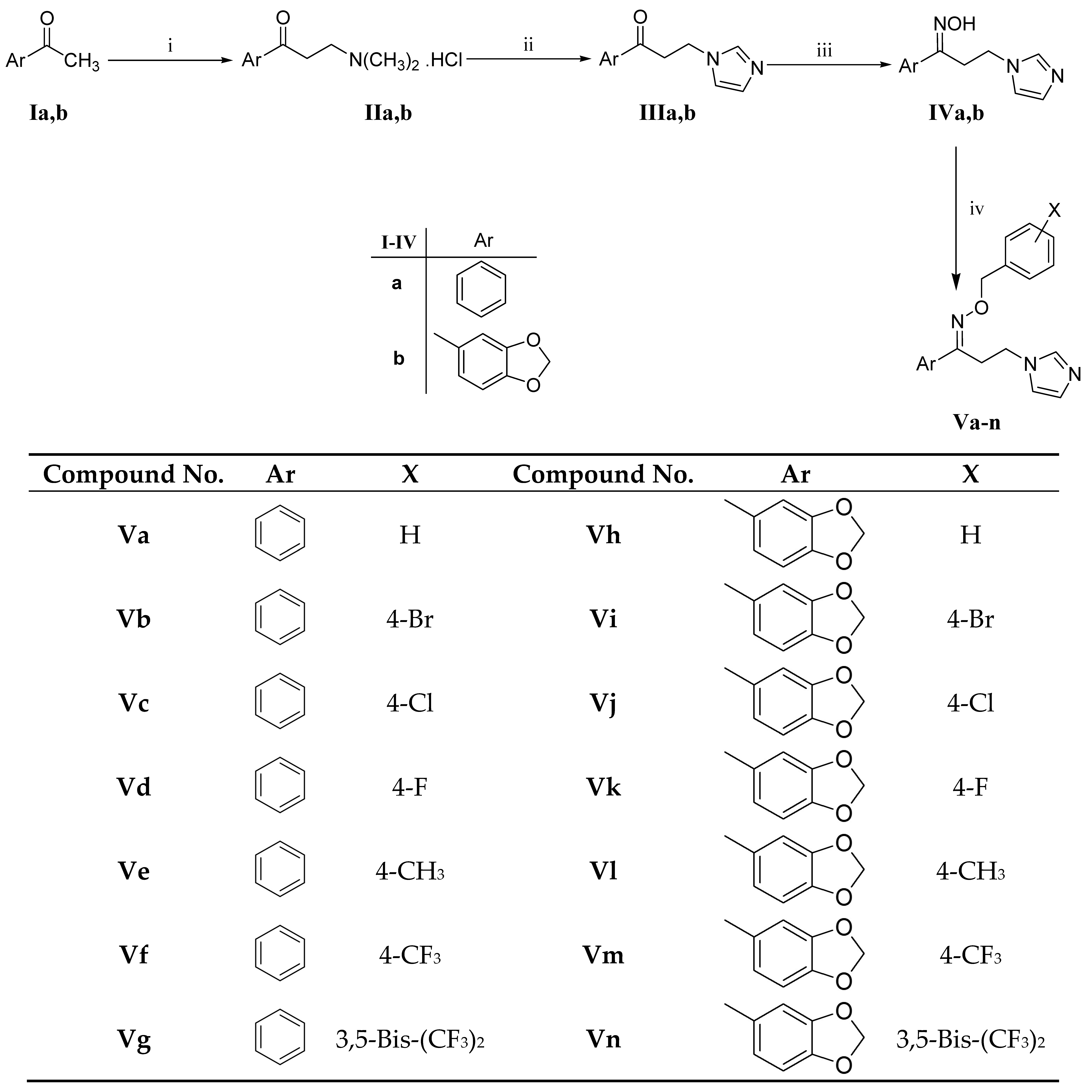
2.1. Chemistry
2.2. Crystal Structure of Compound Vi
2.3. Antifungal Evaluation
2.4. Structure–Activity Relationships
3. Experimental
3.1. General
3.2. Chemistry
3.2.1. Synthesis of (1E)-1-(2H-1,3-benzodioxol-5-yl)-N-hydroxy-3-(1H-imidazol-1-yl)propan-1-imine (IV)
3.2.2. Synthesis of the Oximino Ethers Va–n
3.3. Crystal Structure Determination
3.4. Antifungal Activity
3.4.1. Materials
3.4.2. Organisms
3.4.3. Preparation of Fungal Inocula
3.4.4. Preparation of the Tested Compound Solutions
3.4.5. Antifungal Susceptibility Studies
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Denning, D.W.; Hope, W.W. Therapy for fungal diseases: Opportunities and priorities. Trends Microbiol. 2010, 18, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Castelli, M.V.; Butassi, E.; Monteiro, M.C.; Svetaz, L.A.; Vicente, F.; Zacchino, S.A. Novel antifungal agents: A patent review (2011–present). Expert Opin. Ther. Pat. 2014, 24, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, S.Y.; Garzan, A.; Dennis, E.K.; Shrestha, S.K.; Garneau-Tsodikova, S. Repurposing antipsychotic drugs into antifungal agents: Synergistic combinations of azoles and bromperidol derivatives in the treatment of various fungal infections. Eur. J. Med. Chem. 2017, 139, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Moudgal, V.; Sobel, J. Antifungals to treat Candida albicans. Expert Opin. Pharmacother. 2010, 11, 2037–2048. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.; Lass-Flörl, C. Changing epidemiology of systemic fungal infections. Clin. Microbiol. Infect. 2008, 14, 5–24. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Wang, Y.; Wang, W.; Wang, S.; Xu, B.; Fan, G.; Dong, G.; Liu, Y.; Yao, J.; Miao, Z. Discovery of highly potent triazole antifungal derivatives by heterocycle-benzene bioisosteric replacement. Eur. J. Med. Chem. 2013, 64, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.; Wilson, D.; Drew, R.; Perfect, J. Azole antifungals: 35 years of invasive fungal infection management. Expert Rev. Anti-Infect. Ther. 2015, 13, 787–798. [Google Scholar] [CrossRef] [PubMed]
- Ghannoum, M.A.; Rice, L.B. Antifungal agents: Mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin. Microbiol. Rev. 1999, 12, 501–517. [Google Scholar] [PubMed]
- Lempers, V.J.; Martial, L.C.; Schreuder, M.F.; Blijlevens, N.M.; Burger, D.M.; Aarnoutse, R.E.; Brüggemann, R.J. Drug-interactions of azole antifungals with selected immunosuppressants in transplant patients: Strategies for optimal management in clinical practice. Curr. Opin. Pharmacol. 2015, 24, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Luca, L.D. Naturally occurring and synthetic imidazoles: Their chemistry and their biological activities. Curr. Med. Chem. 2006, 13, 1–23. [Google Scholar] [PubMed]
- Rossello, A.; Bertini, S.; Lapucci, A.; Macchia, M.; Martinelli, A.; Rapposelli, S.; Herreros, E.; Macchia, B. Synthesis, antifungal activity, and molecular modeling studies of new inverted oxime ethers of oxiconazole. J. Med. Chem. 2002, 45, 4903–4912. [Google Scholar] [CrossRef] [PubMed]
- Aboul-Enein, M.N.; El-Azzouny, A.A.; Attia, M.I.; Saleh, O.A.; Kansoh, A.L. Synthesis and anti-Candida potential of certain novel 1-[(3-substituted-3-phenyl)propyl]-1H-imidazoles. Arch. Pharm. 2011, 344, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Roman, G.; Mares, M.; Nastasa, V. A novel antifungal agent with broad spectrum: 1-(4-Biphenylyl)-3-(1H-imidazol-1-yl)-1-propanone. Arch. Pharm. 2013, 346, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Attia, M.I.; Zakaria, A.S.; Almutairi, M.S.; Ghoneim, S.W. In vitro anti-Candida activity of certain new 3-(1H-imidazol-1-yl)propan-1-one oxime esters. Molecules 2013, 18, 12208–12221. [Google Scholar] [CrossRef] [PubMed]
- Leite, A.C.L.; da Silva, K.P.; de Souza, I.A.; de Araújo, J.M.; Brondani, D.J. Synthesis, antitumour and antimicrobial activities of new peptidyl derivatives containing the 1,3-benzodioxole system. Eur. J. Med. Chem. 2004, 39, 1059–1065. [Google Scholar] [CrossRef] [PubMed]
- Himaja, M.; Vandana, K.; Ranjitha, A.; Ramana, M.; Karigar, A. Synthesis, docking studies and antioxidant activity of 1,3-benzodioxole-5-carboxyl amino acids and dipeptides. Int. Res. J. Pharm. 2011, 2, 57–61. [Google Scholar]
- Attia, M.I.; Kansoh, A.L.; El-Brollosy, N.R. Antimicrobial pyrimidinones II: Synthesis and antimicrobial evaluation of certain novel 5,6-disubstituted 2-(substituted amino) alkylthiopyrimidin-4(3H)-ones. Monatsh. Chem. 2014, 145, 1825–1837. [Google Scholar] [CrossRef]
- Allen, F.H.; Kennard, O.; Watson, D.G.; Brammer, L.; Orpen, A.G.; Taylor, R. Tables of bond lengths determined by X-ray and neutron diffraction. Part 1. Bond lengths in organic compounds. J. Chem. Soc. Perkin Trans. 1987, 12, S1–S19. [Google Scholar] [CrossRef]
- El Hage, S.; Lajoie, B.; Feuillolay, C.; Roques, C.; Baziard, G. Synthesis, antibacterial and antifungal activities of bifonazole derivatives. Arch. Pharm. 2011, 344, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Al-Wabli, R.I.; Al-Ghamdi, A.R.; Ghabbour, H.A.; Attia, M.I. Crystal structure of 1-(2H-1,3-benzodioxol-5-yl)-3-(1H-imidazol-1-yl)propan-1-one, C13H12N2O3. Z. Krist. New Cryst. Struct. 2017, 232, 437–439. [Google Scholar] [CrossRef]
- Al-Wabli, R.I.; Al-Ghamdi, A.R.; Ghabbour, H.A.; Al-Agamy, M.H.; Monicka, J.C.; Joe, I.H.; Attia, M.I. Synthesis, X-ray single crystal structure, molecular docking and DFT computations on N-[(1E)-1-(2H-1,3-benzodioxol-5-yl)-3-(1H-imidazol-1-yl)propyl idene]hydroxylamine: A new potential antifungal agent precursor. Molecules 2017, 22, 373. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A Found. Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G. SHELXTL-PC, Version 5.1; Siemens Analytical Instruments Inc.: Madison, WI, USA, 1997.
Sample Availability: Samples of the synthesized compounds are available from the corresponding authors. |
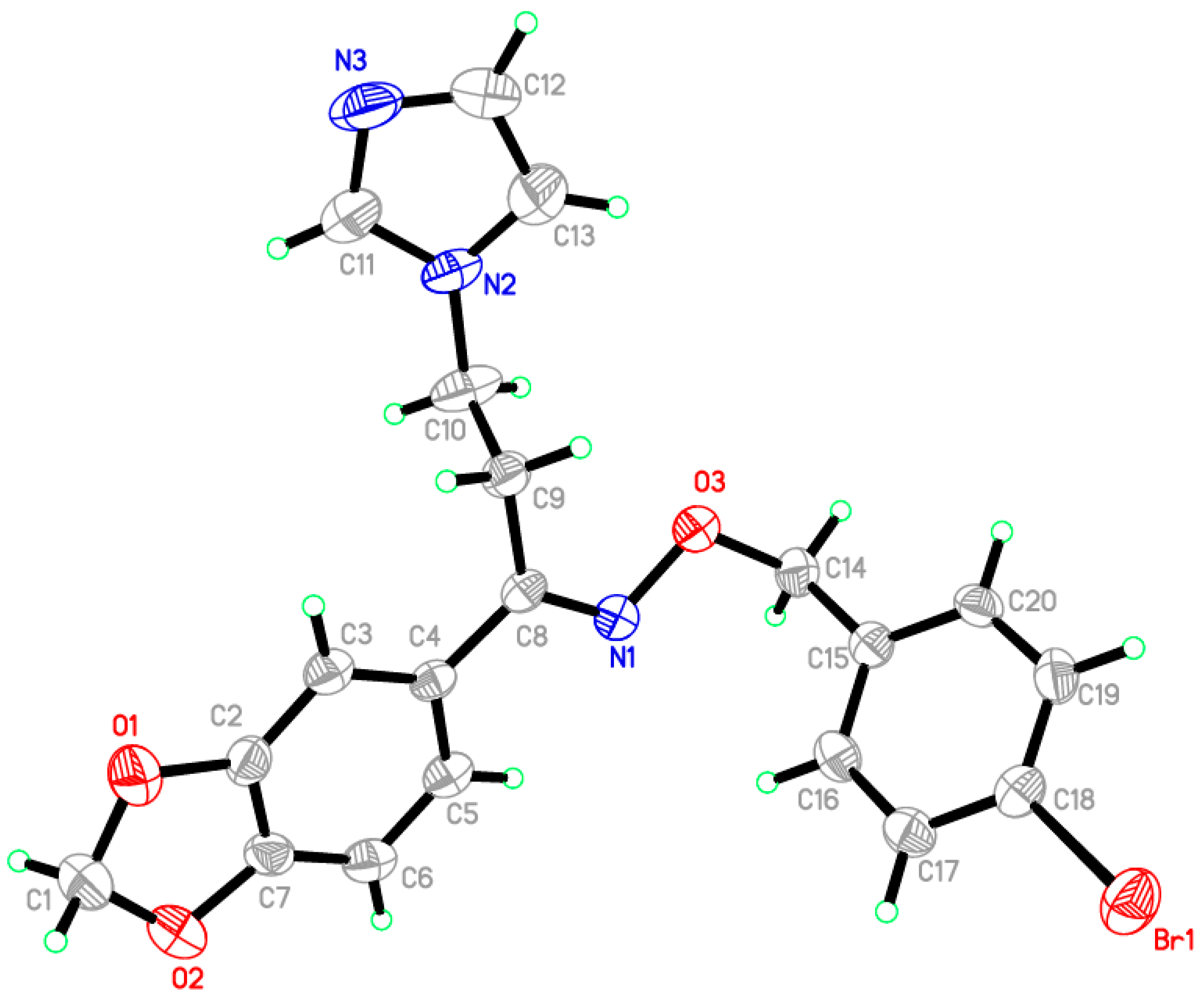

| Bond Lengths | |||
| Br1–C18 | 1.905(3) | N1–C8 | 1.290(4) |
| O1–C1 | 1.432(5) | N2–C10 | 1.462(5) |
| O1–C2 | 1.386(4) | N2–C11 | 1.337(5) |
| O2–C1 | 1.428(5) | N2–C13 | 1.347(6) |
| O2–C7 | 1.373(4) | N3–C11 | 1.313(6) |
| O3–N1 | 1.410(4) | N3–C12 | 1.343(6) |
| O3–C14 | 1.442(4) | ||
| Bond Angles | |||
| C1–O1–C2 | 105.9(3) | O2–C7–C2 | 110.5(3) |
| C1–O2–C7 | 105.9(3) | O2–C7–C6 | 128.4(3) |
| N1–O3–C14 | 107.6(2) | N1–C8–C4 | 114.9(3) |
| O3–N1–C8 | 111.8(3) | N1–C8–C9 | 123.1(3) |
| C10–N2–C11 | 127.2(3) | N2–C10–C9 | 112.3(3) |
| C10–N2–C13 | 126.8(3) | N2–C11–N3 | 112.3(4) |
| C11–N2–C13 | 106.0(3) | N3–C12–C13 | 110.2(4) |
| C11–N3–C12 | 104.8(4) | N2–C13–C12 | 106.7(4) |
| O1–C1–O2 | 108.0(3) | O3–C14–C15 | 113.3(3) |
| O1–C2–C3 | 127.9(3) | Br1–C18–C17 | 119.4(2) |
| O1–C2–C7 | 109.3(3) | Br1–C18–C19 | 119.3(3) |
| D–H···A | D–H | H···A | D···A | D–H···A |
|---|---|---|---|---|
| C3–H3A···N3 | 0.9300 | 2.5300 | 3.453(5) | 169.00 |
| Symmetry codes: (i) –x − 2, −y, −z + 1. | ||||
| Compound No. | Candida albicans | Candida tropicalis | Candida parapsilosis | Asperagillus niger | ||||
|---|---|---|---|---|---|---|---|---|
| DIZ ± SD (mm) | MIC (µmol/mL) | DIZ ± SD (mm) | MIC (µmol/mL) | DIZ ± SD (mm) | MIC (µmol/mL) | DIZ ± SD (mm) | MIC (µmol/mL) | |
| Va | 18 ± 1.00 | 0.209 | 16 ± 1.16 | 0.419 | 20 ± 0.68 | 0.105 | 21 ± 1.14 | 0.105 |
| Vb | 23 ± 0.63 | 0.083 | 18 ± 0.97 | 0.083 | 24 ± 0.45 | 0.042 | 22 ± 0.94 | 0.083 |
| Vc | 22 ± 0.52 | 0.094 | 22 ± 0.63 | 0.094 | 23 ± 0.61 | 0.047 | 19 ± 0.75 | 0.047 |
| Vd | 18 ± 1.12 | 0.198 | 15 ± 1.21 | 0.198 | 19 ± 0.85 | 0.049 | 12 ± 0.30 | 0.198 |
| Ve | 19 ± 1.10 | 0.050 | 17 ± 1.14 | 0.100 | 20 ± 1.00 | 0.050 | 12 ± 0.41 | 0.201 |
| Vf | 18 ± 0.95 | 0.686 | 18 ± 1.13 | 0.343 | 21 ± 0.43 | 0.043 | 22 ± 0.99 | 0.172 |
| Vg | 21 ± 1.13 | 0.073 | 20 ± 0.91 | 0.145 | 22 ± 0.58 | 0.036 | 13 ± 0.64 | 0.290 |
| Vh | 14 ± 0.50 | 0.183 | 10 ± 0.58 | 0.183 | 13 ± 0.60 | 0.003 | 20 ± 0.50 | 0.733 |
| Vi | 15 ± 1.20 | 0.149 | 15 ± 0.30 | 0.149 | 14 ± 0.50 | 0.002 | 13 ± 1.53 | 0.299 |
| Vj | 15 ± 0.30 | 0.167 | 14 ± 0.60 | 0.167 | 21 ± 1.00 | 0.010 | 16 ± 1.31 | 0.667 |
| Vk | 15 ± 0.58 | 0.174 | 12 ± 0.58 | 0.174 | 13 ± 0.58 | 0.044 | 23 ± 0.50 | 0.697 |
| Vl | 16 ± 1.00 | 0.176 | 13 ± 0.60 | 0.176 | 19 ± 1.00 | 0.006 | 12 ± 0.58 | 0.705 |
| Vm | 15 ± 0.58 | 0.153 | 13 ± 0.40 | 0.153 | 23 ± 0.60 | 0.019 | 14 ± 1.00 | 0.307 |
| Vn | 13 ± 0.40 | 0.527 | 15 ± 1.20 | 0.527 | 19 ± 0.58 | 0.002 | 11 ± 0.20 | >1.05 |
| Fluconazole | 18 ± 1.10 | 0.051 | 19 ± 1.00 | 0.045 | 19 ± 0.90 | 0.047 | ND | ND |
| Ketokonazole | ND | ND | ND | ND | ND | ND | 29 ± 0.60 | 0.02 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Wabli, R.I.; Al-Ghamdi, A.R.; Ghabbour, H.A.; Al-Agamy, M.H.; Attia, M.I. Synthesis, Single Crystal X-ray Analysis, and Antifungal Profiling of Certain New Oximino Ethers Bearing Imidazole Nuclei. Molecules 2017, 22, 1895. https://doi.org/10.3390/molecules22111895
Al-Wabli RI, Al-Ghamdi AR, Ghabbour HA, Al-Agamy MH, Attia MI. Synthesis, Single Crystal X-ray Analysis, and Antifungal Profiling of Certain New Oximino Ethers Bearing Imidazole Nuclei. Molecules. 2017; 22(11):1895. https://doi.org/10.3390/molecules22111895
Chicago/Turabian StyleAl-Wabli, Reem I., Alwah R. Al-Ghamdi, Hazem A. Ghabbour, Mohamed H. Al-Agamy, and Mohamed I. Attia. 2017. "Synthesis, Single Crystal X-ray Analysis, and Antifungal Profiling of Certain New Oximino Ethers Bearing Imidazole Nuclei" Molecules 22, no. 11: 1895. https://doi.org/10.3390/molecules22111895
APA StyleAl-Wabli, R. I., Al-Ghamdi, A. R., Ghabbour, H. A., Al-Agamy, M. H., & Attia, M. I. (2017). Synthesis, Single Crystal X-ray Analysis, and Antifungal Profiling of Certain New Oximino Ethers Bearing Imidazole Nuclei. Molecules, 22(11), 1895. https://doi.org/10.3390/molecules22111895





