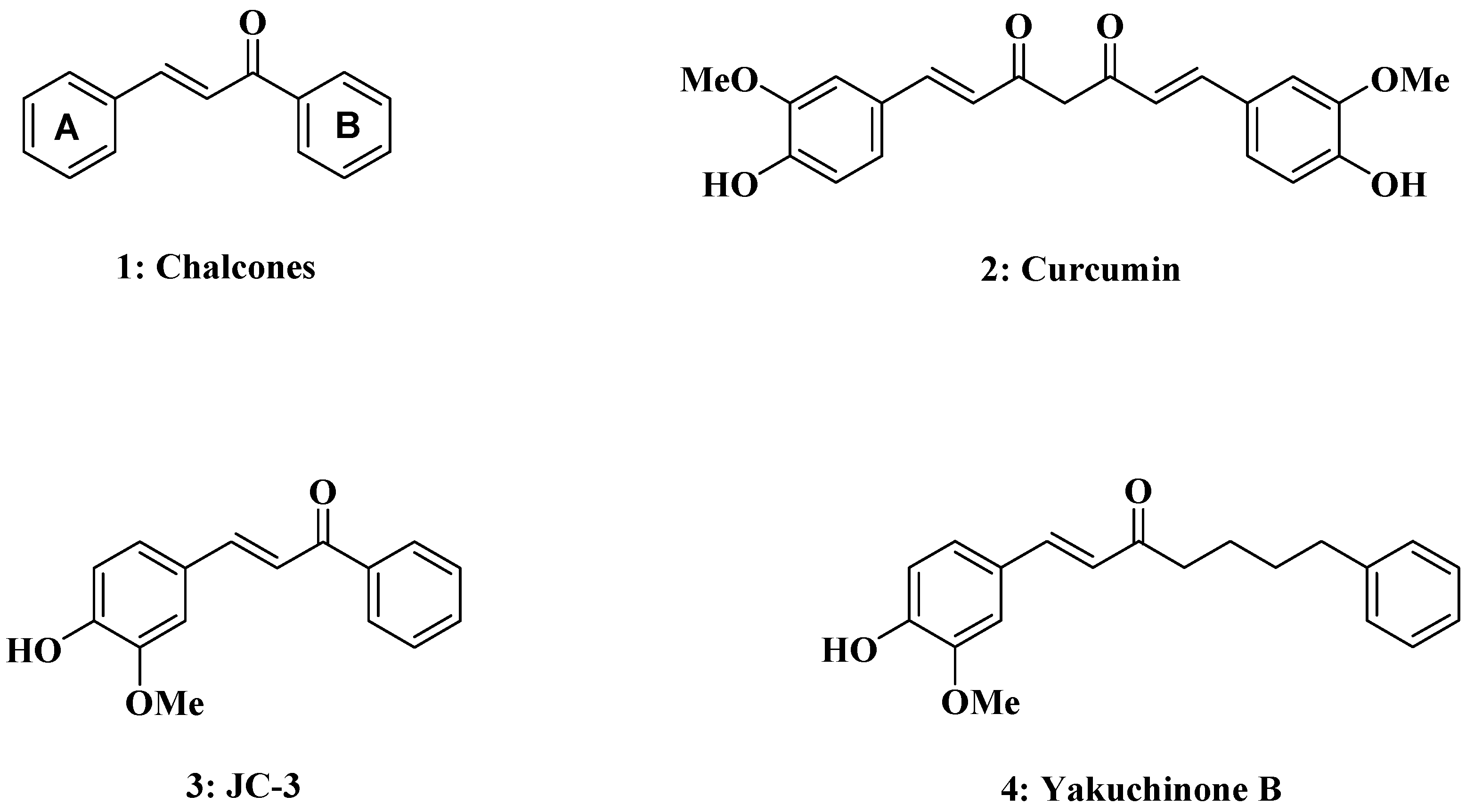
Practical Synthesis of Chalcone Derivatives and Their Biological Activities
Abstract
:1. Introduction
2. Results and Discussion
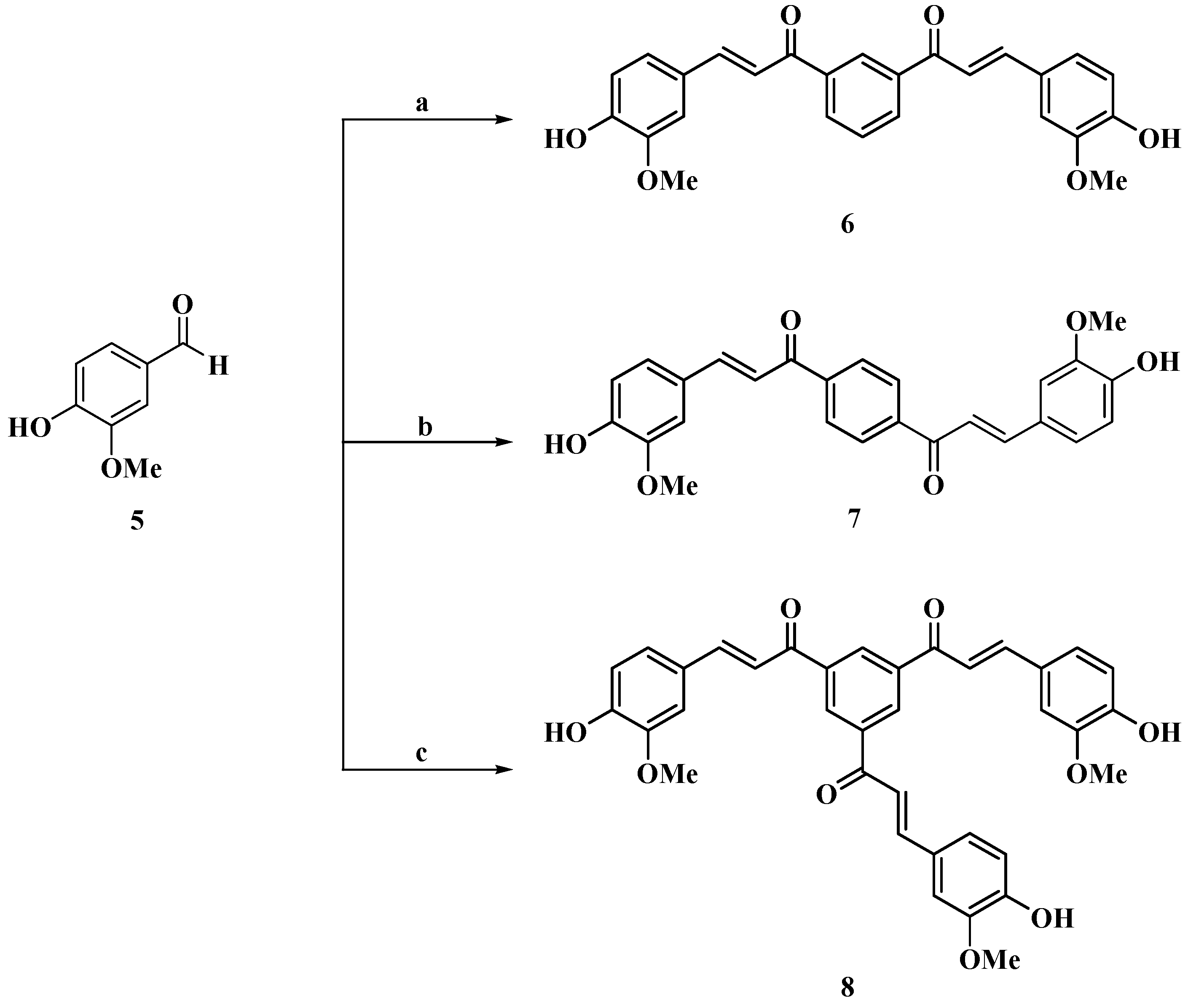
2.1. Chemistry
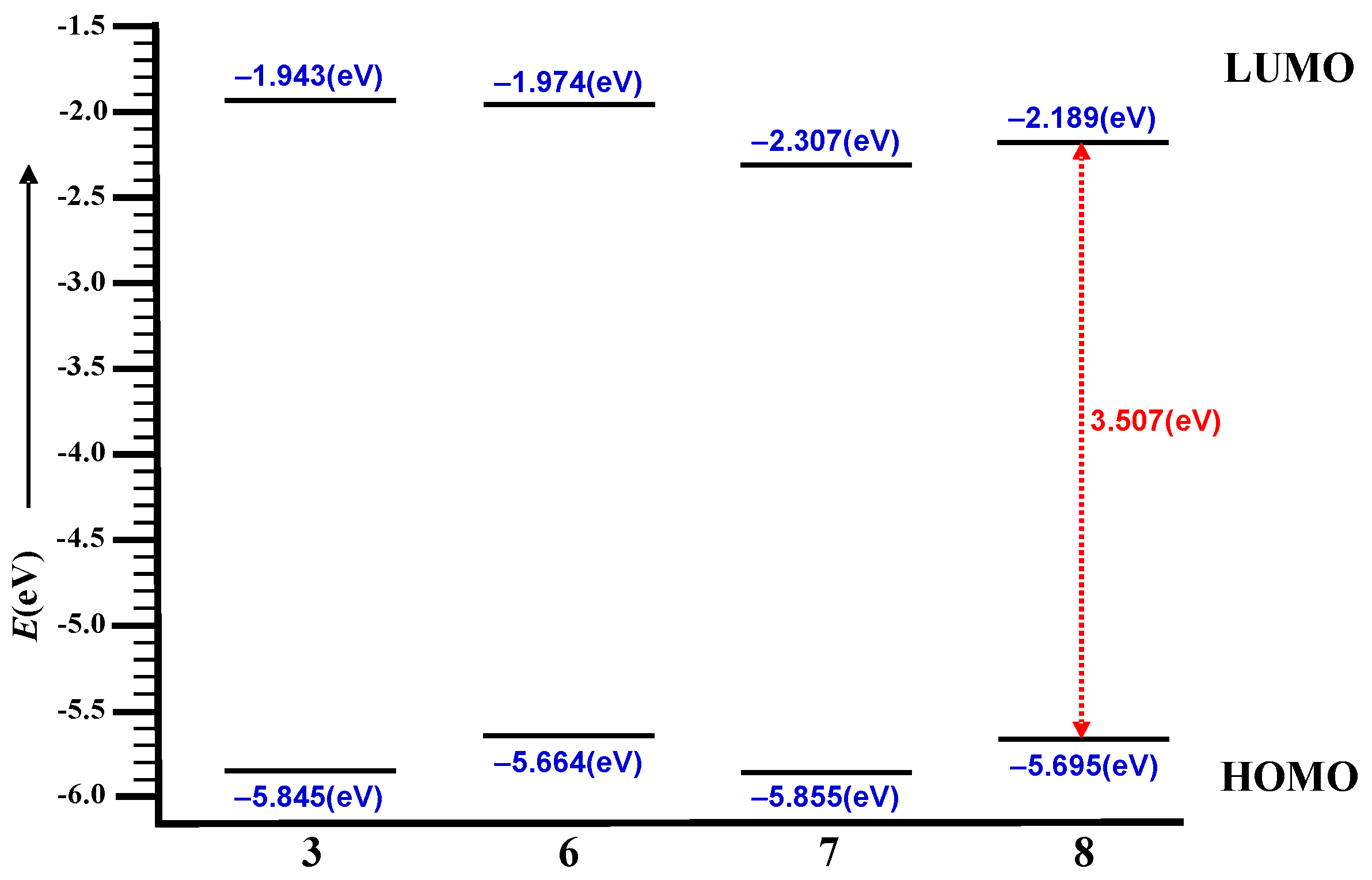
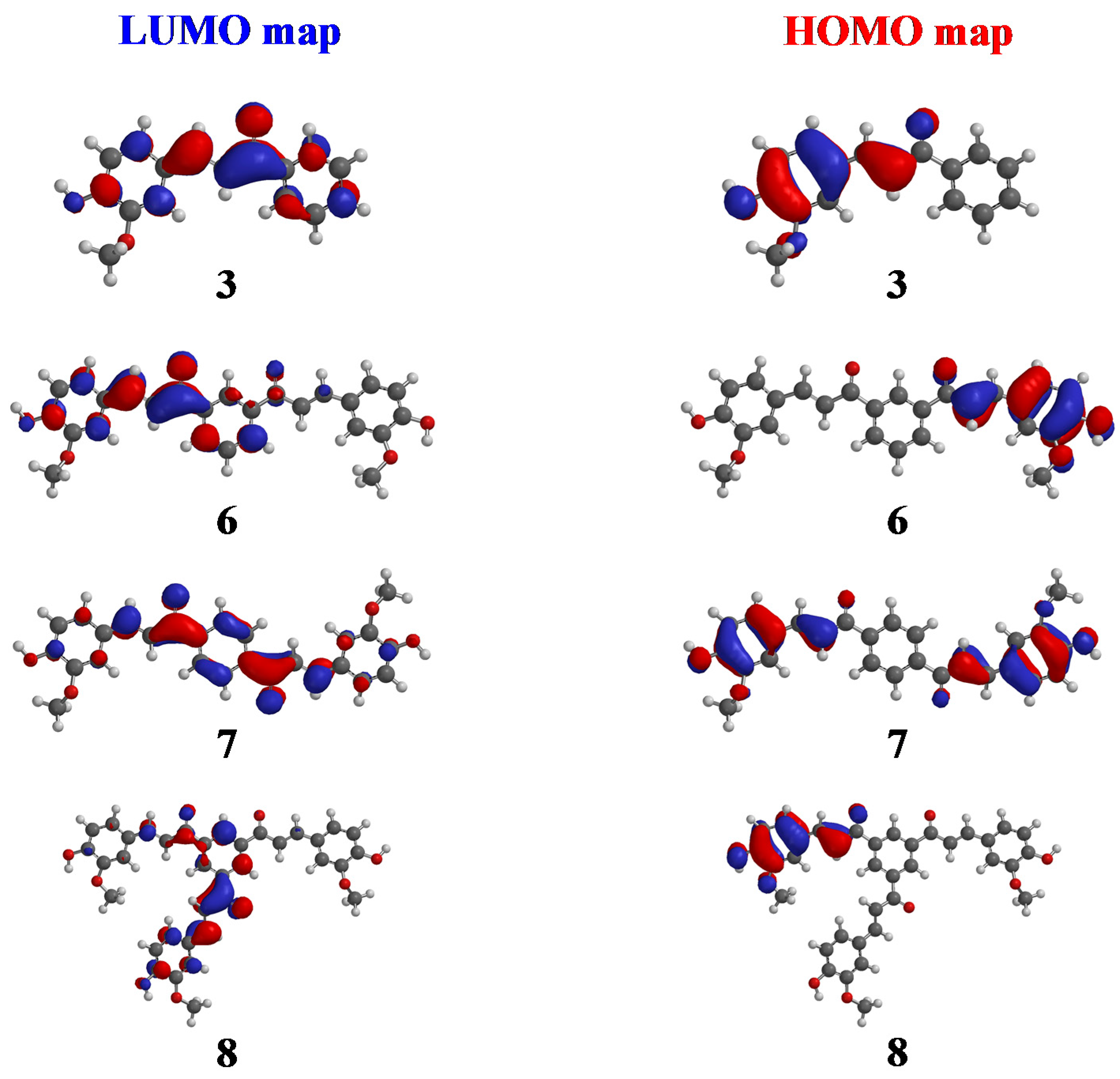
2.2. Molecular Modeling
2.3. Biological Evaluation
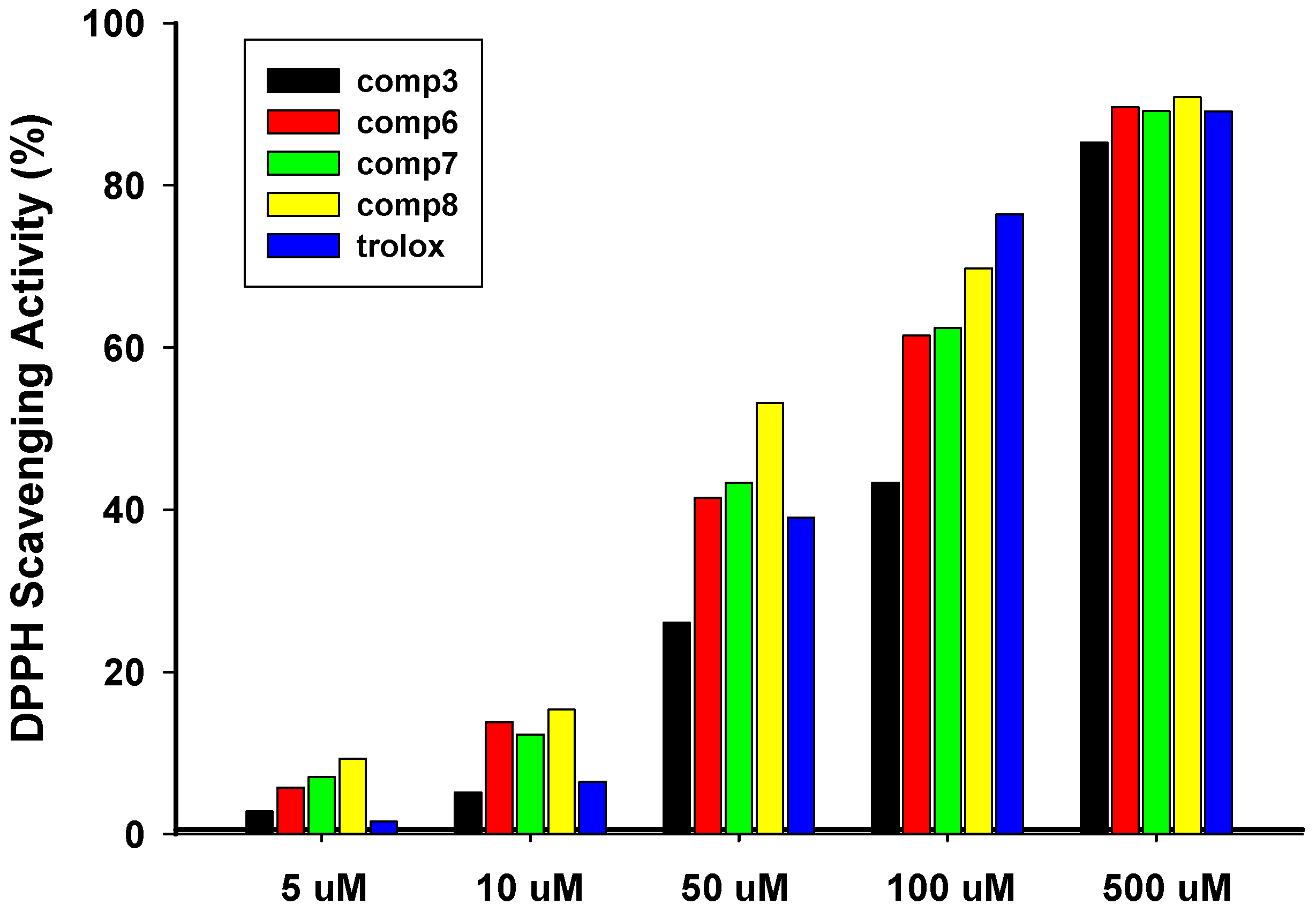
2.3.1. Radical Scavenging Activity
2.3.2. Inhibition of NO Generation
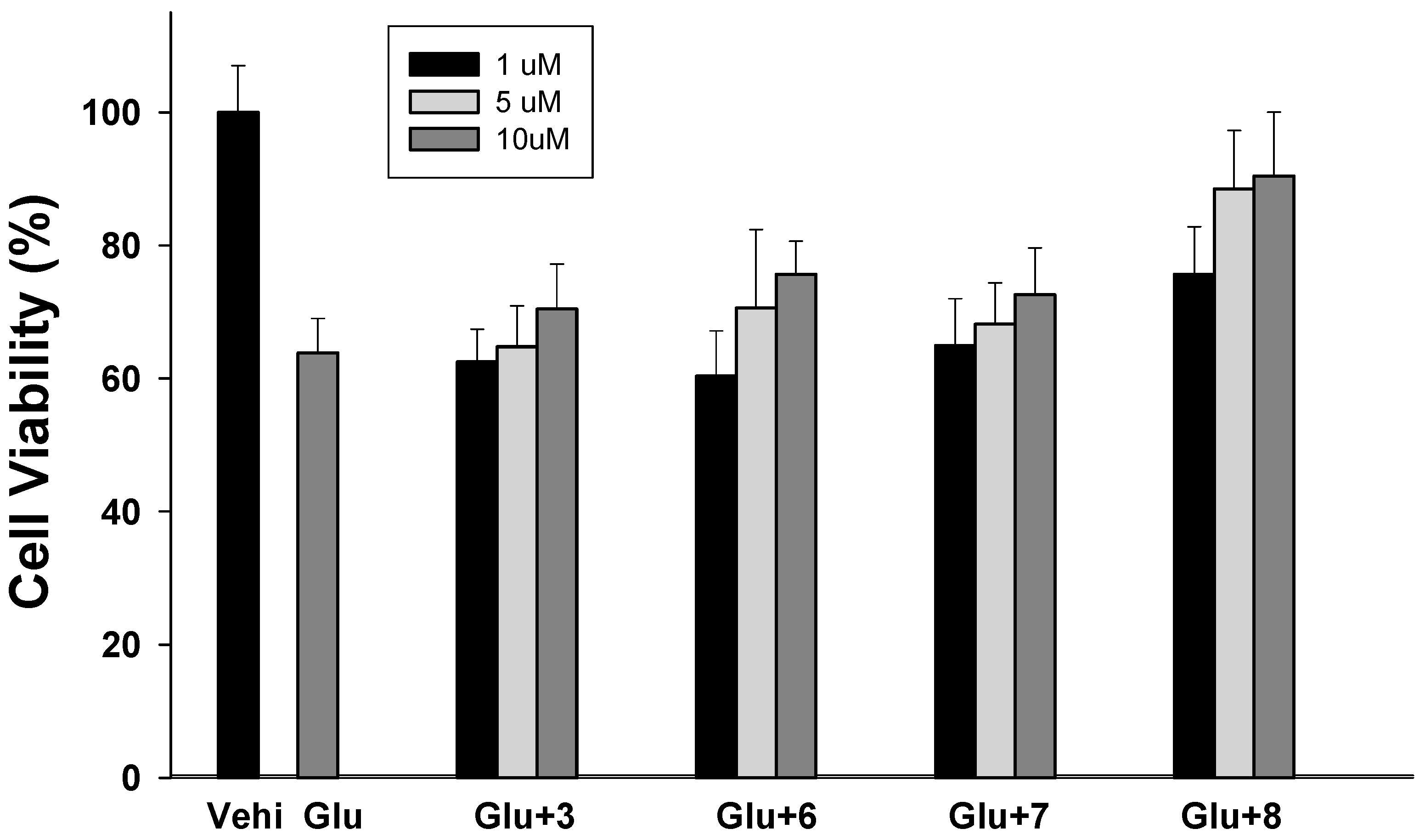
2.3.3. Neuroprotective Activity: Inhibition of Glutamate-Induced Neurotoxicity
3. Materials and Methods
3.1. Synthesis
3.2. Molecular Modeling
3.3. Biology-Measurement of Cell Viability
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Viana, G.S.B.; Bandeira, M.A.M.; Matos, F.J.A. Analgesic and antiinflammatory effects of chalcones isolated from Myracrodruon urundeuva Allemão. Phytomedicine 2003, 10, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Rojas, J.; Payá, M.; Domínguez, J.N.; Ferrándiz, M.L. ttCH, a selective inhibitor of inducible nitric oxide synthase expression with antiarthritic properties. Eur. J. Pharmcol. 2003, 465, 183–189. [Google Scholar] [CrossRef]
- Nowakowska, Z. A review of anti-infective and anti-inflammatory chalcones. Eur. J. Med. Chem. 2007, 42, 125–137. [Google Scholar] [CrossRef] [PubMed]
- Al Rahim, M.; Nakajima, A.; Misawa, N.; Shindo, K.; Adachi, K.; Shizuri, Y.; Ohizumi, Y.; Yamakuni, T. A novel diol-derivative of chalcone produced by bioconversion, 3-(2,3-dihydroxyphenyl)-1-phenylpropan-1-one, activates PKA/MEK/ERK signaling and antagonizes Aβ-inhibition of the cascade in cultured rat CNS neurons. Eur. J. Pharmacol. 2008, 600, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, S.F.; Larsen, M.; Boesen, T.; Schonning, K.; Kromann, H. Cationic chalcone antibiotics. Design, synthesis, and mechanism of action. J. Med. Chem. 2005, 48, 2667–2677. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.A.; Shaharyar, M.; De Clercq, E. Synthesis of 5-(4-hydroxy-3-methylphenyl)-5-(substituted phenyl)-4,5-dihydro-1H-1-pyrazolyl-4-pyridylmethanone derivatives with anti-viral activity. J. Enzyme Inhib. Med. Chem. 2007, 22, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Onyilagha, J.C.; Malhotra, B.; Elder, M.; French, C.J.; Towers, G.H.N. Comparative studies of inhibitory activities of chalcones on tomato ringspot virus (ToRSV). Can. J. Plant Pathol. 1997, 19, 133–137. [Google Scholar] [CrossRef]
- Konieczny, M.T.; Konieczny, W.; Sabisz, M.; Skladanowski, A.; Wakiec, R.; Augustynowicz-Kopec, E.; Zwolska, Z. Synthesis of isomeric, oxathiolone fused chalcones, and comparison of their activity toward various microorganisms and human cancer cells line. Chem. Pharm. Bull. 2007, 55, 817–820. [Google Scholar] [CrossRef] [PubMed]
- Gschwendt, M.; Kittstein, W.; Furstenberger, G.; Marks, F. The mouse ear edema: A quantitatively evaluable assay for tumor promoting compounds and for inhibitors of tumor promotion. Cancer Lett. 1984, 25, 177–185. [Google Scholar] [CrossRef]
- Bhale, P.S.; Chavan, H.V.; Dongare, S.B.; Shringare, S.N.; Mule, Y.B.; Nagane, S.S.; Bandgar, B.P. Synthesis of extended conjugated indolyl chalcones as potent anti-breast cancer, anti-inflammatory and antioxidant agents. Bioorg. Med. Chem. Lett. 2017, 27, 1502–1507. [Google Scholar] [CrossRef] [PubMed]
- Sokmen, M.; Khan, M.A. The antioxidant activity of some curcuminoids and chalcones. Inflammopharmacology 2016, 24, 81–86. [Google Scholar]
- Hofmann, E.; Webster, J.; Do, T.; Kline, R.; Snider, L.; Hauser, R.; Higginbottom, G.; Campbell, A.; Ma, L.; Paula, S. Hydroxylated chalcones with dual properties: Xanthine oxidase inhibitors and radical scavengers. Bioorg. Med. Chem. 2016, 24, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.L.; Stevens, J.F.; Ivanov, V.; McCall, M.; Frei, B.; Deinzer, M.L.; Buhler, D.R. Antioxidant and prooxidant actions of prenylated and nonprenylated chalcones and flavanones in vitro. J. Agric. Food. Chem. 2000, 48, 3876–3884. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, C.; Kawasaki, N.; Miyataka, H.; Jayachandran, E.; Kim, I.H.; Kirk, K.L.; Taguchi, T.; Takeuchi, Y.; Hori, H.; Satoh, T. Synthesis and biological activities of fluorinated chalcone derivatives. Bioorg. Med. Chem. 2002, 10, 699–706. [Google Scholar] [CrossRef]
- Park, P.H.; Kim, H.S.; Hur, J.; Jin, X.Y.; Jin, Y.L.; Sohn, D.H. YL-I-108, a synthetic chalcone derivative, inhibits lipopolysaccharide-stimulated nitric oxide production in RAW 264.7 murine macrophages: Involvement of heme oxygenase-1 induction and blockade of activator protein-1. Arch. Pharm. Res. 2009, 32, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Ansari, F.L.; Umbreen, S.; Hussain, L.; Makhmoor, T.; Nawaz, S.A.; Lodhi, M.A.; Khan, S.N.; Shaheen, F.; Choudhary, M.I.; Atta-ur-Rahman. Syntheses and biological activities of chalcone and 1,5-benzothiazepine derivatives: Promising new free-radical scavengers, and esterase, urease, and α-glucosidase inhibitors. Chem. Biodivers. 2005, 2, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Burnham, W.S.; Sidwell, R.W.; Tolman, R.L.; Stout, M.G. Synthesis and antiviral activity of 4′-hydroxy-5,6,7,8-tetramethoxyflavone. J. Med. Chem. 1972, 15, 1075–1076. [Google Scholar] [CrossRef] [PubMed]
- Boeck, P.; Falcao, C.A.B.; Leal, P.C.; Yunes, R.A.; Cechinel, V.; Torres-Santos, E.C.; Rossi-Bergmann, B. Synthesis of chalcone analogues with increased antileishmanial activity. Bioorg. Med. Chem. 2006, 14, 1538–1545. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, J.N.; Leon, C.; Rodrigues, J.; de Dominguez, N.G.; Gut, J.; Rosenthal, P.J. Synthesis and evaluation of new antimalarial phenylurenyl chalcone derivatives. J. Med. Chem. 2005, 48, 3654–3658. [Google Scholar] [CrossRef] [PubMed]
- Quintin, J.; Desrivot, J.; Thoret, S.; Le Menez, P.; Cresteil, T.; Lewin, G. Synthesis and biological evaluation of a series of tangeretin-derived chalcones. Bioorg. Med. Chem. Lett. 2009, 19, 167–169. [Google Scholar] [CrossRef] [PubMed]
- Prakash, O.; Kumar, A.; Sadana, A.; Prakash, R.; Singh, S.P.; Claramunt, R.M.; Sanz, D.; Alkorta, I.; Elguero, J. Study of the reaction of chalcone analogs of dehydroacetic acid and o-aminothiophenol: Synthesis and structure of 1,5-benzothiazepines and 1,4-benzothiazines. Tetrahedron 2005, 61, 6642–6651. [Google Scholar] [CrossRef]
- Weber, W.M.; Hunsaker, L.A.; Abcouwer, S.F.; Deck, L.M.; Vander Jagt, D.L. Anti-oxidant activities of curcumin and related enones. Bioorg. Med. Chem. 2005, 13, 3811–3820. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Jung, J.C.; Kim, D.H.; Ryu, J.H.; Lee, Y.; Jung, M.; Oh, S. The neuroprotective effects of benzylideneacetophenone derivatives on excitotoxicity and inflammation via phosphorylated janus tyrosine kinase 2/phosphorylated signal transducer and activator of transcription 3 and mitogen-activated protein K pathways. J. Pharmacol. Exp. Ther. 2009, 328, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Yun, J.; Jung, J.C.; Oh, S.; Jung, Y.S. Anti-tumor activity of benzylideneacetophenone derivatives via proteasomal inhibition in prostate cancer cells. Pharmazie 2016, 71, 274–279. [Google Scholar] [PubMed]
- Lee, H.J.; Kim, J.S.; Yoon, J.W.; Kim, H.-D.; Ryu, J.-H. Suppression of inducible nitric oxide synthase expression by yakuchinones and their analogues. Chem. Pharm. Bull. 2006, 54, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Winter, E.; Devantier, N.P.; Chiaradia-Delatorre, L.D.; Gauthier, C.; Yunes, R.A.; Nunes, R.J.; Creczynski-Pasa, T.B.; Di Pietro, A. Symmetric bis-chalcones as a new type of breast cancer resistance protein inhibitors with a mechanism different from that of chromones. J. Med. Chem. 2014, 57, 2930–2941. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Jung, J.C.; Oh, S. Synthesis of 1,3-diphenyl-2-propen-1-one derivatives and evaluation of their biological activities. Bioorg. Med. Chem. 2007, 15, 4098–4105. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.-C.; Jang, S.; Lee, Y.; Min, D.; Lim, E.; Jung, H.; Oh, M.; Oh, S.; Jung, M. Efficient synthesis and neuroprotective effect of substituted 1,3-diphenyl-2-propen-1-ones. J. Med. Chem. 2008, 51, 4054–4058. [Google Scholar] [CrossRef] [PubMed]
- Eddarir, S.; Cotelle, N.; Bakkour, Y.; Rolando, C. An efficient synthesis of chalcones based on the Suzuki reaction. Tetrahedron Lett. 2003, 44, 5359–5363. [Google Scholar] [CrossRef]
- Lawinski, M.; Sledzinski, Z.; Kubasik-Juraniec, J.; Spodnik, J.H.; Wozniak, M.; Boguslawski, W. Does resveratrol prevent free radical-induced acute pancreatitis? Pancreas 2005, 31, 43–47. [Google Scholar] [PubMed]
- Kimura, Y.; Okuda, H.; Arichi, S. Effects of stilbenes on arachidonate metabolism in leukocytes. Biochim. Biophys. Acta Lipids Lipid Metab. 1985, 834, 275–278. [Google Scholar]
- Theodosis-Nobelos, P.; Athanasekou, C.; Rekka, E.A. Dual antioxidant structures with potent anti-inflammatory, hypolipidemic and cytoprotective properties. Bioorg. Med. Chem. Lett. 2017, 27, 4800–4804. [Google Scholar] [CrossRef] [PubMed]
- Dewar, M.J.S.; Zoebisch, E.G.; Healy, E.F.; Stewart, J.J.P. AM1: A new general purpose quantum mechanical molecular model. J. Am. Chem. Soc. 1985, 107, 3902–3909. [Google Scholar] [CrossRef]
- Kohn, W.; Becks, A.D.; Parr, R.G. Density functional theory of electronic structure. J. Phys. Chem. 1996, 100, 12974–12980. [Google Scholar] [CrossRef]
- Spartan’06; Wavefunction Inc.: Irvine, CA, USA, 2006.
Sample Availability: Samples of the compounds 6,7,8 are available from the authors. |

| Compounds | Energy | E. HOMO a | E. LUMO b | ΔE c |
|---|---|---|---|---|
| (au) | (eV) | (eV) | (eV) | |
| 3 | −843.796 | −5.845 | −1.943 | 3.901 |
| 6 | −1455.342 | −5.664 | −1.974 | 3.690 |
| 7 | −1455.334 | −5.855 | −2.307 | 3.548 |
| 8 | −2066.890 | −5.695 | −2.189 | 3.507 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, J.-C.; Lee, Y.; Min, D.; Jung, M.; Oh, S. Practical Synthesis of Chalcone Derivatives and Their Biological Activities. Molecules 2017, 22, 1872. https://doi.org/10.3390/molecules22111872
Jung J-C, Lee Y, Min D, Jung M, Oh S. Practical Synthesis of Chalcone Derivatives and Their Biological Activities. Molecules. 2017; 22(11):1872. https://doi.org/10.3390/molecules22111872
Chicago/Turabian StyleJung, Jae-Chul, Yongnam Lee, Dongguk Min, Mankil Jung, and Seikwan Oh. 2017. "Practical Synthesis of Chalcone Derivatives and Their Biological Activities" Molecules 22, no. 11: 1872. https://doi.org/10.3390/molecules22111872
APA StyleJung, J.-C., Lee, Y., Min, D., Jung, M., & Oh, S. (2017). Practical Synthesis of Chalcone Derivatives and Their Biological Activities. Molecules, 22(11), 1872. https://doi.org/10.3390/molecules22111872




