1. Introduction
Oral infections, generally caused by bacteria and viruses, are very common diseases in the oral and maxillofacial regions including dental caries, periodontitis, gingivitis, oral mucosal infections, peri-implantitis, etc., which may be very mild or lead to serious problems [
1]. For example, peri-implantitis, a type of destructive inflammatory process surrounding dental implants that can give rise to progressive loss of alveolar bone around the implant, is considered one of the main causes of dental implant failure [
1,
2,
3]. Peri-implantitis is associated with the formation of a bacterial biofilm and compromised immunity at the implant/tissue interface. Microorganisms such as
Fusobacterium nucleatum (
F.n.) accumulate in the subgingival crevice around dental implants and play a significant role in accelerating the co-aggregation of pathogens, promoting the formation of biofilm, and thus inducing the development of peri-implantitis [
2,
3,
4]. Therefore, antibiotic treatments and the maintenance of antibacterial microenvironments are of vital importance for oral health.
The antibiotic treatments with conventional antibiotic drugs such as ampicillin, vancomycin, and gentamycine have become more and more complicated by the emergence of multi-drug resistant bacteria. Antimicrobial peptides (AMPs), since their initial discovery in the 1980s, have been demonstrated as a promising alternative to conventional antibiotics [
5]. AMPs represent a class of short cationic amphiphilic peptides presented ubiquitously in natural organisms as the innate immune system with a broad spectrum of activity against bacteria, viruses, fungi, and even transformed or cancerous cells [
5,
6,
7,
8]. Not like most of antibiotics that act on specific proteins, AMPs aim at bacterial membranes and subsequently disintegrate bacterial cell membranes to overcome the problem of antibiotic resistance and the microbial gene mutation. According to these advantages, AMPs have therefore been widely studied as alternatives to traditional antibiotics in recent years.
KSL (KKVVFKVKFK-CONH
2) is a novel antimicrobial decapeptide that is developed through synthetic combinatorial library technology showing a wide range of activity against microbial pathogens [
9,
10]. Previous studies have showed that KSL was stable in artificial saliva and acidic buffer, and could effectively inhibit the growth of oral biofilm and prevent the development of oral bacterial pathogens [
11,
12,
13]. Also, KSL-W (KKVVFWVKFK-CONH
2), the analogue of KSL, was proved to display improved stability in saliva and simulated gastric conditions with preserved antimicrobial activity [
11]. At the same time, KSL-W exhibits no cytotoxicity on host cells. Therefore, KSL-W is also widely accepted for effectively killing bacteria and controlling biofilm formation in oral applications [
14].
The site-specific delivery and controlled release of therapeutic drugs, bioactive proteins, and peptides have been attracting more and more attention in the field of biomaterials and drug delivery. Encapsulation in the form of nanoparticles or microspheres is considered an effective drug delivery method for proteins/peptides to protect their bioactivities, enhance the therapeutic efficiency, and reduce drug side-effects as well. In this study, prolonged release of KSL-W to the targeted diseased area is thought to be imperative to maintain long-term antimicrobial effect for satisfying infection control. Poly(lactide-
co-glycolide) (PLGA) microspheres have been extensively applied as drug delivery system because of their good biocompatibility and adjustable biodegradability [
15,
16]. However, the locally acidic micro-milieu associated with PLGA hydrolysis may result in protein aggregation and denaturation, and thus confine its applications [
17,
18]. Therefore, chitosan (CS), a positively charged linear polysaccharide, has been used with PLGA to neutralize the acidic degradation product of PLGA [
19,
20,
21,
22]. Chitosan is a biodegradable natural polymer with low toxicity, ideal biocompatibility, and excellent hemostatic and antimicrobial properties, which makes it suitable for many kinds of biomedical applications. Wang et al. successfully developed PLGA/CS composite microspheres through the emulsion method for the long-term delivery of oligopeptides derived from BMP-2 [
21,
22]. In our study, similar PLGA/CS microspheres were fabricated and studied for the sustained release of KSL-W.
In previous studies, PLGA microspheres were generally produced by phase separation, emulsification/solvent-extraction, or emulsification/solvent-evaporation [
23,
24]. However, from a technical standpoint, it is difficult to achieve desirable microspheres with uniform size distribution, high drug encapsulation, and good loading efficiency because smaller molecules can hardly enter PLGA microspheres sufficiently via conventional preparation methods. The electrospraying technique is a useful technique to prepare microspheres, embedding bioactive proteins through ejecting a polymer solution under high voltage power and ventilation conditions [
25,
26]. In addition, it is very easy to use electrospraying to completely control the drug encapsulation and size distribution of microspheres [
26].
In this paper, sphere-in-sphere PLGA/CS composite microspheres were fabricated by electrospraying and combined crosslinking-emulsion methods for increased encapsulation efficiency and prolonged antimicrobial peptide release.
3. Discussion
In this study, the PLGA/CS composite microspheres exhibited an excellent release profile for the sustained release of KSL-W in a controllable manner. The release profile of drug-loaded microspheres is affected by the properties of the MSs such as the drug/polymer mass ratio, the physicochemical properties of the drug and polymer, and the particle size. The PLGA microspheres fabricated by the emulsion electrospraying process had smooth surfaces and quite uniform particle diameters of around 7 μm, which made them able to be encapsulated in CS shells more evenly in a sphere-in-sphere structural pattern, probably favoring better drug release behavior. Moreover, the electrosprayed PLGA microspheres could reach much higher encapsulation efficiency of over 90% than the microspheres fabricated by the W/O/W emulsion-solvent evaporation method. Within the initial 10 days, the PLGA/CS MSs underwent a quick release period with about 30% KSL-W released. This phenomenon was mainly due to the easier and faster release of KSL-W distributed near microsphere surfaces in a diffusion-controlled mode [
26]. After that, the PLGA/CS MSs released about 60% KSL-W in the following 70 days in both diffusion- and degradation-controlled fashions. As a result, the initially quick release of the KSL-W contributed to resist acute inflammation, and the sustained release for up to 80 days from the PLGA/CS composite microspheres benefitted the maintenance of an antibacterial microenvironment. Moreover, all the formulations of PLGA/CS MSs showed similar drug release trends, which indicated that the concentration of released KSL-W could be easily adjusted by the amount initially added in PLGA and CS solutions. The more KSL-W added, the larger the released concentration.
Recently, AMPs have attracted increasing interest as an alternative to common antibiotics in biomedical applications against infections, mainly because of their broad-spectrum antibacterial activity and little bacterial resistance. Most AMPs are characterized by a positive net charge and hydrophobic amino acids that make them able to insert into and disintegrate negatively-charged bacterial cell membranes. However, as is well-known, most mammalian cells are also negatively charged; therefore, the long-acting antibacterial effect and the biosafety of AMPs on mammalian cells are also thought to be key issues for clinical applications. Some previous studies indicated that cationic AMPs present a certain level of cytotoxic and hemolytic activity to human cells and erythrocytes, respectively. Other studies showed that AMPs have an essential property to interact preferentially with bacteria compared to mammalian cells, due to the specific biophysical and biochemical properties of bacterial membranes. Therefore, the AMPs concentration in the local milieu should be accurately controlled to provoke both effective antibacterial activity and minimal cytotoxicity to ensure the health of mammalian cells.
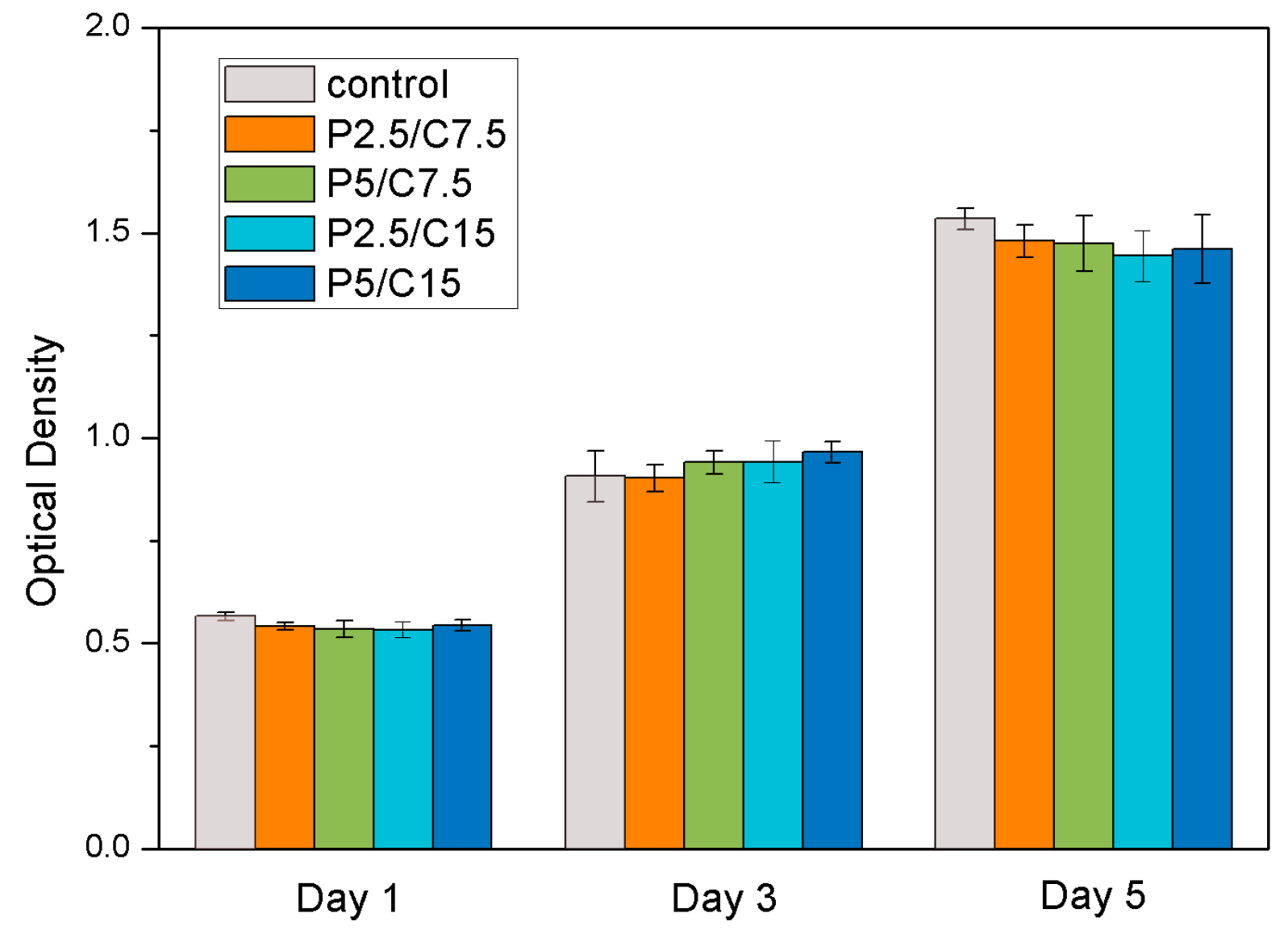
In our studies, PLGA/CS composite microspheres were fabricated for the controlled release of KSL-W. The mouse preosteoblast cell line, MC3T3-E1, was then used to evaluate the cytocompatibility of the released AMPs, indicating that the initially high concentration of AMPs released from all of the formulations of microspheres exhibited no cytotoxicity. At the same time, the KSL-W-loaded PLGA/CS MSs showed long-term antibacterial activity for up to 80 days.
F.n., evaluated here, is an early colonizer in the subgingival crevice around dental implants and plays a significant role in accelerating the co-aggregation of pathogens, promoting the formation of biofilm, and thus inducing the development of peri-implantitis. The minimum inhibitory concentration against
F.n. reported in the previous study is about 0.0156 mg/mL [
27]. Yet, the minimum concentration of KSL-W solution after 80 days of incubation in our study was about 0.0190 mg/mL, which was still higher than the minimum inhibitory concentration reported previously, showing effective antibiotic activity. A series of fabrication processes from encapsulation, and storage, to release did not destroy the antibacterial activity of KSL-W. Therefore, our results indicate that the sustained release system of AMPs based on PLGA/CS MSs have potential applications for the treatment of peri-implantitis and other oral diseases.
4. Materials and Methods
4.1. Chemicals
Antimicrobial peptide KSL-W (KKVVFWVKFK-CONH2, Mw: 1307) was custom-synthesized by Qiangyao Bio-Technology Co., Ltd. (purity > 90%, Shanghai, China). Poly (lactic-co-glycolic acid) (PLGA, 75:25, Mw: 5.0 × 104) was purchased from the Medical Equipment Research Institute (Jinan, China). Chitosan (Mw: 1 × 105–3 × 105) was purchased from the Bailingwei Science and Technology Co., Ltd. (Beijing, China). All other chemicals used were of analytical grade and obtained from Chemical Reagent Co., Ltd. (Beijing, China).
4.2. Preparation of PLGA Microspheres
KSL-W-loaded PLGA microspheres were prepared through emulsion electrospraying. The PLGA was dissolved in trichloromethane (CHCl3) at a concentration of 60 mg/mL. Different amounts of KSL-W (2.5 mg or 5.0 mg) were dissolved in 50 μL of deionized water and then added to 1 mL of the PLGA solution to form a W/O emulsion by sonication on ice with an ultrasonic crasher (Scientz-IID, Ningbo Science Biotechnology Co., Ltd., Ningbo, China) at a power of 300 W for 20 s. The emulsion solutions were then electrosprayed under an ambient atmosphere. The emulsion solutions were placed in a 1 mL syringe and passed through a blunt stainless-steel nozzle (inner diameter = 0.34 mm) at a constant flow rate of 1 mL/h using a micro-infusion pump. The applied voltage to the nozzle tip was 5.23 kV, and the distance between the nozzle tip to the aluminum collection plate was 20 cm. The organic solvent was removed during the electrospraying process by evaporation, leaving solid microspheres on the collection plate. The PLGA microspheres were then freeze-dried for 24 h to remove the residual solvent. Two formulations of KSL-W-loaded PLGA microspheres with different amounts of added peptide were abbreviated as P2.5 and P5.0, respectively.
4.3. Preparation of PLGA/CS Composite Microspheres
KSL-W-loaded PLGA/CS composite microspheres were prepared with the crosslinking emulsification method. First of all, the 2% aqueous acetic acid solution was prepared and used as the solvent to dissolve chitosan at a concentration of 3% (
w/
v). Then, 30 mg of different formulations of PLGA microspheres were added to 10 mL of 3% CS solution, respectively, whilst stirred magnetically at room temperature, during which different amounts of KSL-W solutions (7.5 mg, 15.0 mg, 7.5 mg, and 15.0 mg) were also added to the PLGA/CS mixture. After that, the PLGA/CS/KSL-W mixture was poured into a mixture of 60 mL liquid paraffin and 1.8 mL Span 80 to form the W/O emulsion, which was then stirred magnetically for 30 min at room temperature. We then dropped 5 mL of 5% (
w/
v) aqueous sodium tripolyphosphate (TPP) solution slowly into the emulsion while stirring mildly for an additional 3 h. Afterwards, the resulting emulsion was settled overnight, forming solid microspheres. The final PLGA/CS/KSL-W microspheres were washed three times with excess amounts of petroleum ether and isopropyl alcohol, and then freeze-dried for 24 h to remove the residual solvent [
19]. According to different amounts of peptides added within the PLGA and CS microspheres, the sequential formulations of products were abbreviated as P2.5/C7.5, P5/C7.5, P2.5/C15, and P5/C15.
4.4. Morphology Characterization of KSL-W-Loaded Microspheres
Scanning electron microscopy (JSM-7001F, JEOL Ltd., Tokyo, Japan) was used to characterize the surface morphology of microspheres. KSL-W-loaded PLGA microspheres and PLGA/CS microspheres were dispersed in water and ethyl alcohol, respectively, and then dropped on the surface of a metallic sample stand with a bi-adhesive carbon tape. After being dried under an ambient atmosphere, the samples were sputter-coated with gold and examined at an accelerating voltage of 15 kV.
In order to trace the distribution of KSL-W in the microspheres, FITC-conjugated KSL-W peptides were encapsulated within the microspheres and then examined using a Laser Scanning Confocal Microscope (Zeiss LSM710, excitation wavelength of 488 nm, Carl Zeiss Ltd, Oberkochen, Germany).
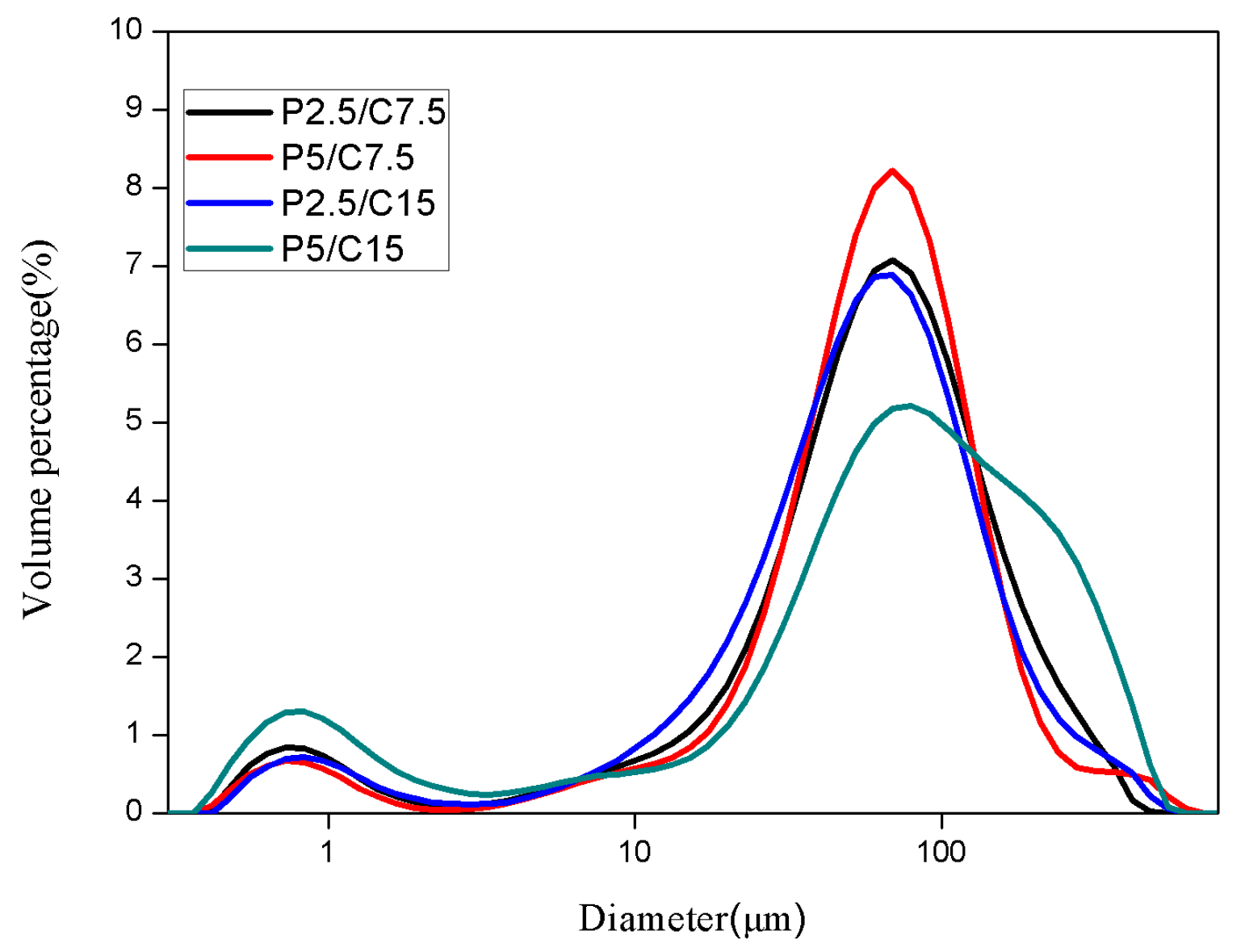
The size distribution of PLGA microspheres and PLGA/CS microspheres were determined using a laser particle size analyzer (Mastersizer 2000, Malvern Instruments Ltd., Worcestershire, UK). The PLGA/CS microspheres were dispersed in water.
4.5. Characterization of KSL-W Encapsulation Behavior
The KSL-W encapsulation behavior in the microspheres was characterized as the encapsulation efficiency (EE) and drug-loading rate (DLR), which were defined using the following equations:
For determining the amount of KSL-W loaded in PLGA microspheres, 10 mg of KSL-W-loaded PLGA microspheres were dissolved by adding 1 mL of acetonitrile and 3 mL of 0.01 M hydrochloric acid solution with shaking for 1 h in a wrist-action shaker. The extracted KSL-W in the supernatant was obtained after centrifugation. In order to calculate the total amount of KSL-W loaded in PLGA/CS microspheres, KSL-W was extracted from the microspheres through the following steps. First, 10 mg of PLGA/CS microspheres was dispersed in 2 mL of 2% aqueous acetic acid solution to dissolve the external chitosan thoroughly. After centrifugation, the supernatant (S1) was collected and the precipitate was dried and weighed. The precipitate was further dissolved by adding 0.7 mL acetonitrile and 1.3 mL 0.01 M of hydrochloric acid solution while stirring. After centrifugation, the supernatant (S2) was obtained. S1 and S2 were detected by a UV-VIS spectrophotometer to calculate the total mass of KSL-W within PLGA/CS MSs, which was marked as M1 for the mass of KSL-W in S1 and M2 for that in S2. The mass of KSL-W was calculated according to the extracted KSL-W solution concentration that was determined by UV-VIS spectrophotometer at 280 nm (UV759S, Shanghai Precision & Scientific Instrument Co., Ltd., Shanghai, China). The standard curve was plotted using the standard KSL-W solutions with given concentrations.
4.6. In Vitro Drug Release Profile
The in vitro release profile of KSL-W from PLGA/CS microspheres suspended in PBS was evaluated for up to 80 days. Briefly, 100 mg of KSL-W-loaded PLGA/CS microspheres were added into a 5 mL centrifuge tube containing 1 mL PBS (pH = 7.4), which was placed in a shaking bath (Model THZ-C, Taicang Laboratorial Equipment Factory in China) at 60 rpm and 37 °C. The microsphere suspensions were centrifuged at 5000 rpm for 10 min to retrieve the supernatants and replaced with 1 mL of fresh PBS at different intervals (4, 7, 10, 14, 21, 30, 35, 42, 50, 56, 64, 72, and 80 days). The aforementioned UV-VIS spectrophotometer (UV759S) method was used to determine the KSL-W concentrations in the released supernatant. Each formulation was examined in triplicate for preliminary statistical analysis.
4.7. The Stability Assessment of the KSL-W
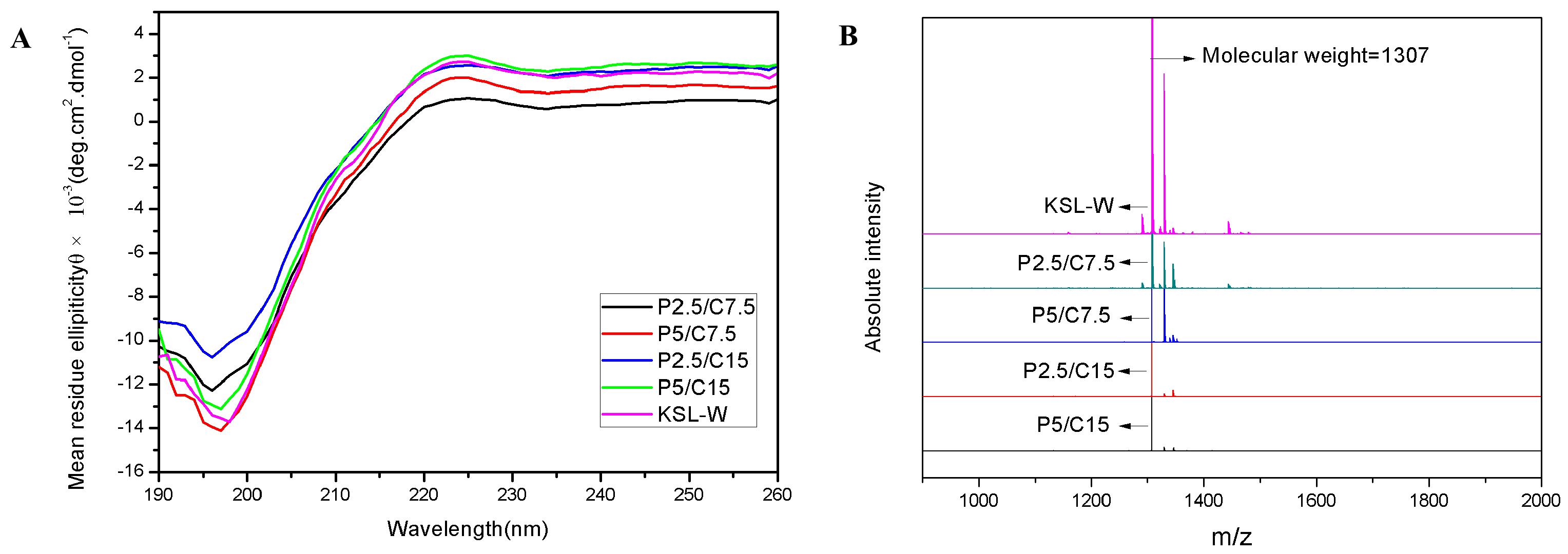
The structural stability of peptide KSL-W after encapsulation and release was assessed by the secondary structure and molecular weight.
The secondary structure of the KSL-W released from composite microspheres after 30 days of incubation was monitored by Far-UV circular dichroism (CD, J-715-150L, JASCO, Tokyo, Japan). Released KSL-W solution was analyzed at room temperature in a quartz cuvette with a path length of 0.1 cm and a scanning speed of 100 nm/min in a wavelength range of 190–260 nm.
Matrix-assisted laser desorption/ionization time-of-flight Mass Spectrometry (MALDI-TOF-MS, Autoflext, Bruker Daltonic Inc., Bremen, Germany) was used to monitor the molecular weights of the KSL-W released in the supernatant. Briefly, 2 μL of 4-hydroxy-cinnamic acids and 2 μL of released supernatant were mixed uniformly. Then, the resultant mixture was dried at room temperature for 1 μL. A nitrogen gas laser further dissociated the samples under the positive ion reflector mode. Lastly, we collected and analyzed the peak signals (m/z) of the samples.
4.8. The Biocompatibility of Peptide KSL-W
The biocompatibility of PLGA/CS composite microspheres loaded with different masses of KSL-W was assessed using CCK-8. MC3T3-E1 cells as representative cells were cultured in a dish containing α-MEM supplemented with 10% (v/v) fetal bovine serum (FBS, Life Technologies, Inc., Rockville, MD, USA), 100 IU/mL penicillin, and 100 IU/mL streptomycin. In order to obtain the drug containing extract, 10 mg each of the composite microspheres was incubated with α-MEM supplemented for a 10-day period at 37 °C. Then, 1 × 104 MC3T3-E1 cells per well were cultured in a humidified incubator with 5% CO2 atmosphere at 37 °C. After 24 h, cells were incubated with the drug extract. Fresh drug extract was replaced every two days and the CCK-8 assay was carried out after one, three, and five days. At each time point, the medium was removed, and fresh medium with CCK-8 (media:CCK-8 = 10:1) was added in each well for 3 h of incubation before measurement with a microplate reader (Bio-Rad, Model680, Bio-Rad Laboratories, Inc., Hercules, CA, USA) at a wavelength of 450 nm. Four repeated measurements for each group were carried out for statistical analysis.
4.9. The Antibacterial Assay
F.n. was used in this study to evaluate the antibacterial activity of the KSL-W-loaded PLGA microspheres. F.n. (ATCC 10953) was cultured in a liquid Bacto-tryptone-yeast extract-ascorbic acid-glucose 5 mg/mL NaCl solution, and incubated at 37 °C under anaerobic conditions (80% N2, 10% CO2, and 10% H2) for 48 h. Then, the bacterial cells were harvested by centrifugation at 10,000 rpm for 10 min, washed with sterile phosphate-buffered saline (PBS), and diluted to 1 × 108 CFU/mL by using a spectrophotometer-based standard curve calculation. The inhibition zone assay was performed using the agar diffusion method (Oxford cup method). Briefly, to determine the inhibition zone, bacterial cells were uniformly distributed on the surface of the blood agar plate (Bacto-tryptone-yeast extract-ascorbic acid-agar, with fresh defibrous sheep blood for more nutrition), and then the Oxford cups with a diameter of 6 mm were placed onto the agar plates. After incubating at 37 °C for half an hour, 0.25 mL of released liquid isolated from P2.5/C7.5 at different time intervals of 10 days, 30 days, 50 days, or 80 days was added into the Oxford cup. PBS served as a negative control. The inhibition zones forming around the cylinders were measured after three days of incubation.
4.10. Statistical Analysis
All numerical data are reported as the means ± standard deviation. The statistical analysis was performed with one-way analysis of variance and Student’s t-test. The data were considered statistically significant when p < 0.05. All of the data were analyzed using SPSS 13.0 software for Windows, Student Version (IBM Corporation, Armonk, NY, USA).