Predicting the Global Potential Distribution of Four Endangered Panax Species in Middle-and Low-Latitude Regions of China by the Geographic Information System for Global Medicinal Plants (GMPGIS)
Abstract
1. Introduction
2. Results
2.1. Ecological Factors
2.2. Potential Distribution
2.2.1. Global Potential Distribution
- (1)
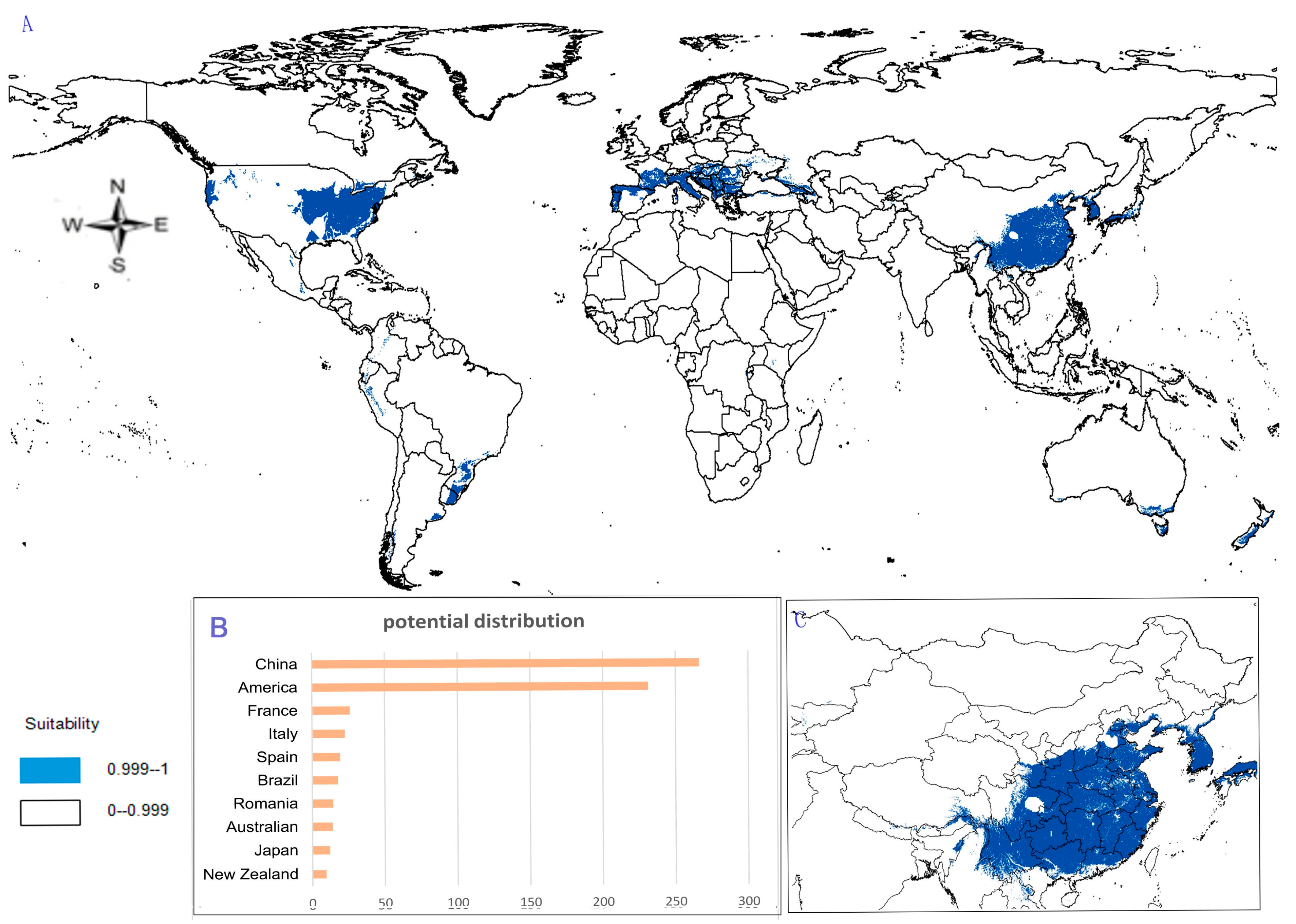
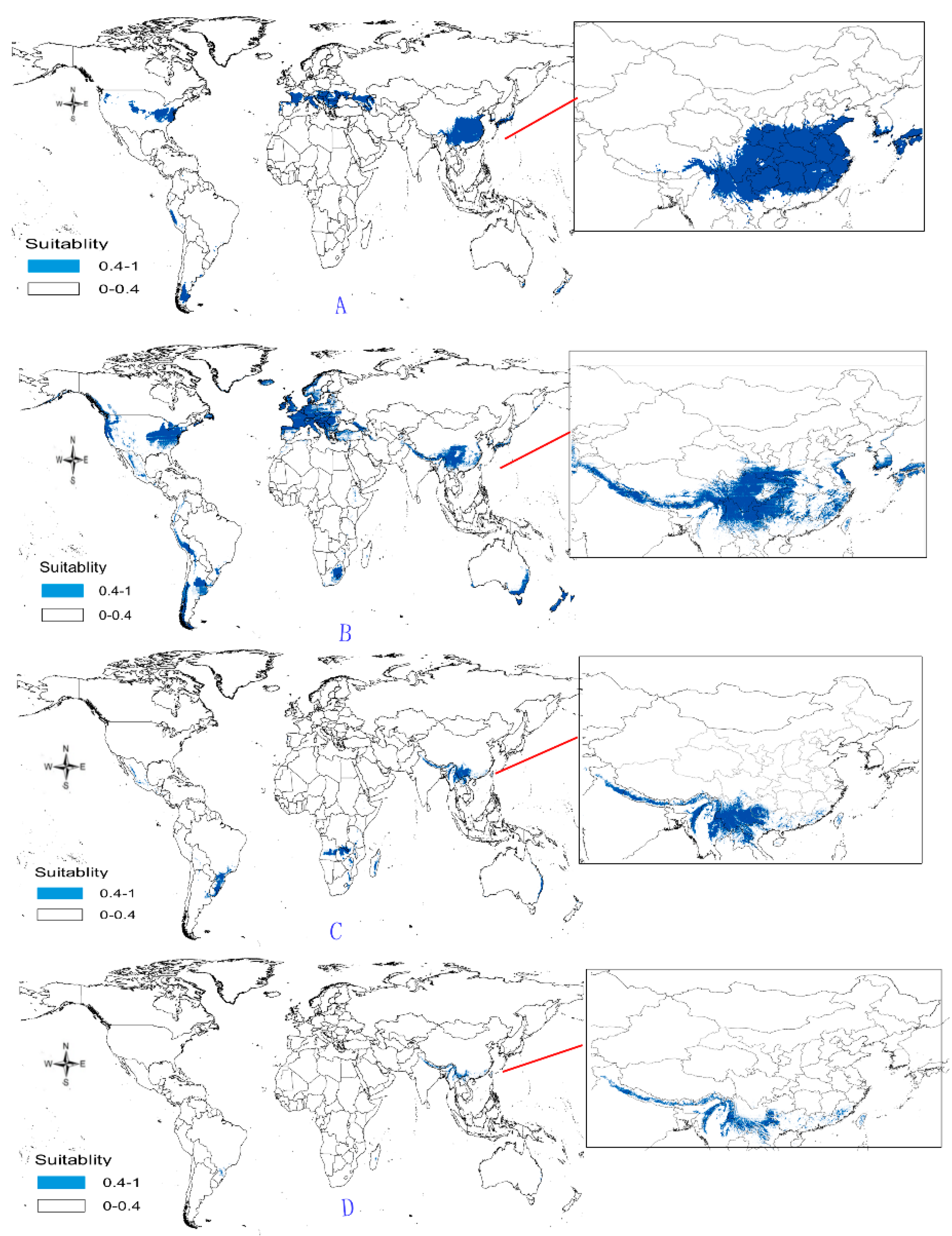
- Potential distribution of P. japonicus: The global potential distribution of P. japonicus is obtained by GMPGIS based on the ecological factors and soil, the total area is 118.29 × 105 km2. P. japonicus is distributed mainly in North and South America, Asia, Europe, Oceania, and other regions (Figure 3A). The leading distribution areas are Southeast Asia and North America, which include China, Japan, South Korea, North Korea, the United States, and Canada. The top three distribution areas are China (2662.98 × 103 km2), United States (2312.34 × 103 km2), and France (260.81 × 103 km2) as shown in Figure 3B.
- (2)
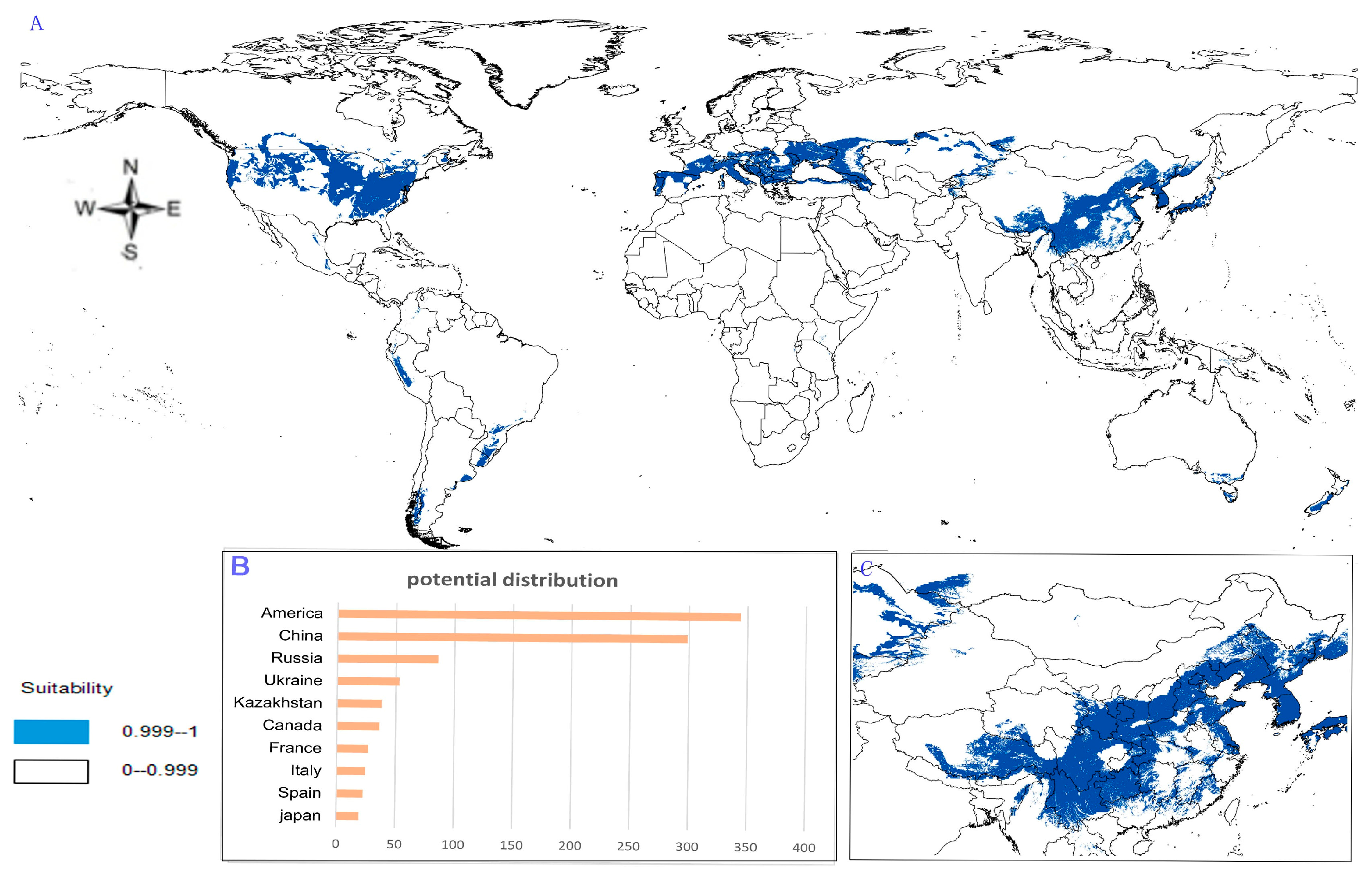
- Potential distribution of P. japonicus var. major: The potential distribution regions for this plant are found in North America, Asia, and Europe (Figure 4A). The total global potential distribution area is 77.5 × 105 km2, and the top three distribution areas are the United States (3438.73 × 103 km2), China (2986.11 × 103 km2), and Russia (861.09 × 103 km2) (Figure 4B).
- (3)
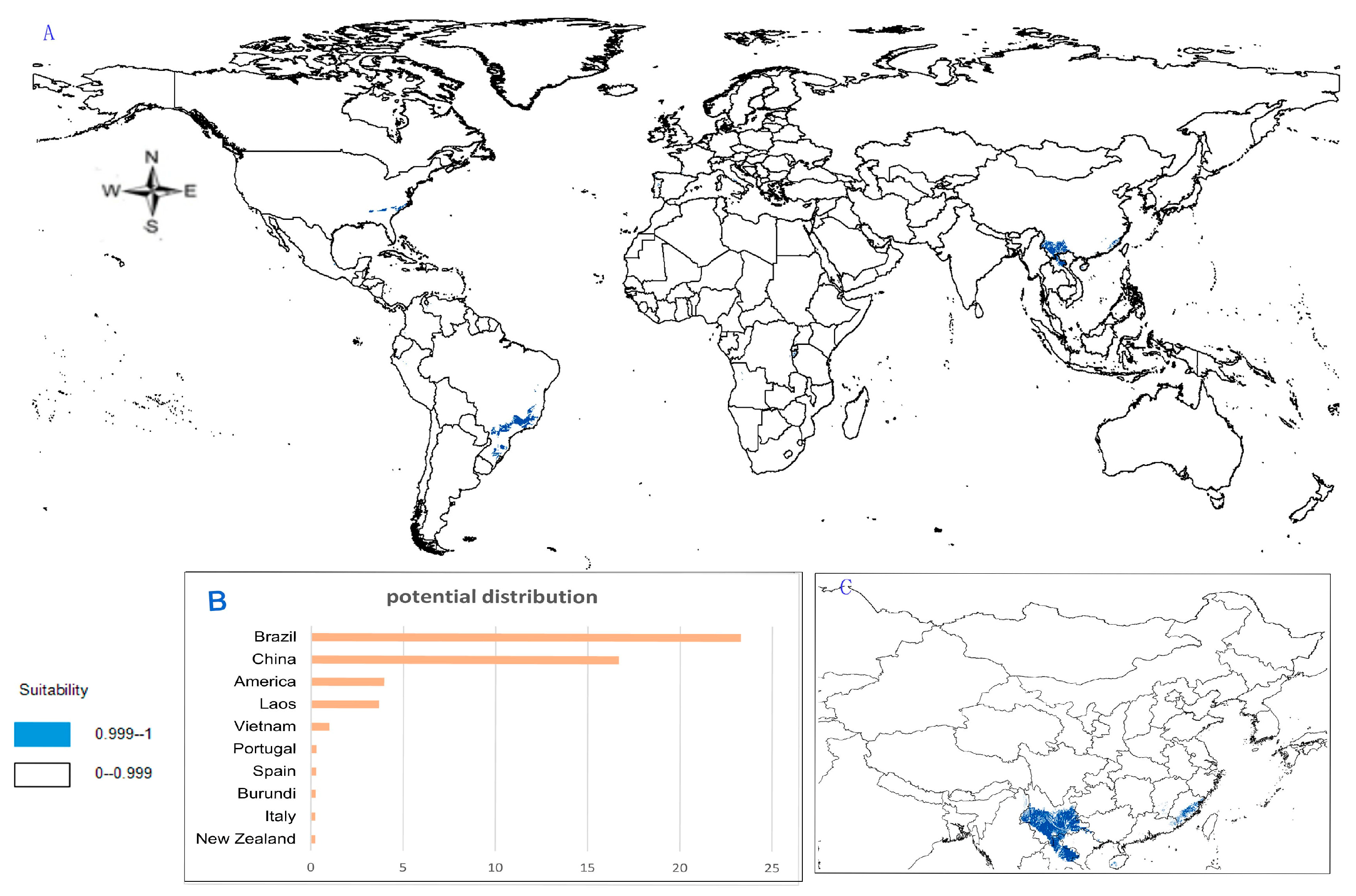
- Potential distribution of P. zingiberensis: The global potential suitable distribution of P. zingiberensis is obtained by GMPGIS (Figure 5A). The map shows the limited distribution of this species, which is distributed in a small part of Asia and South America. The total global potential suitable distribution area is 5.09 × 105 km2 ,the top three distribution areas are Brazil (232.79 × 103 km2), China (166.71 × 103 km2), and the United States (39.58 × 103 km2) shown in Figure 5B. Thus, the introduction and cultivation of P. zingiberensis should be prioritized in these countries and regions.
- (4)
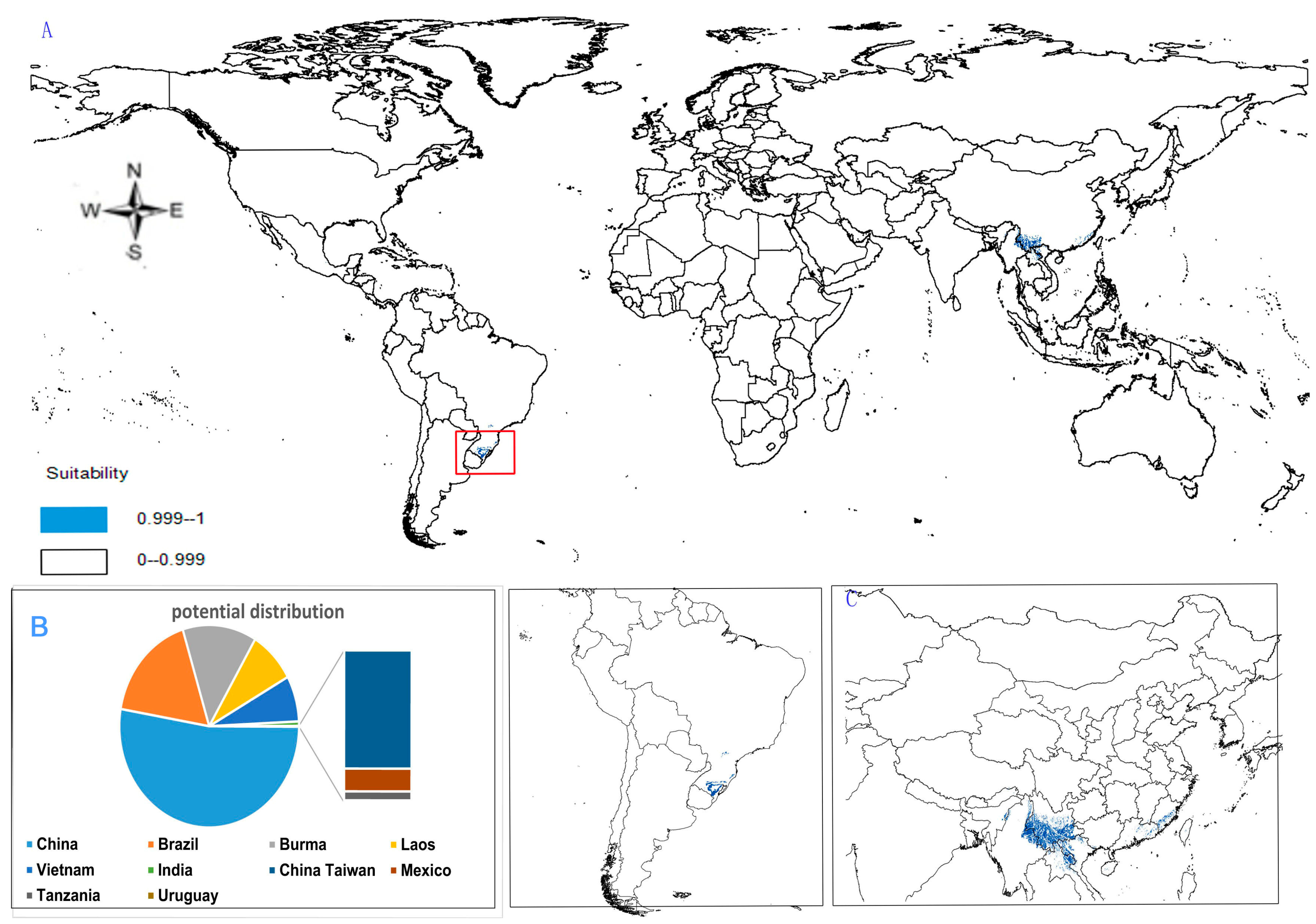
- Potential distribution of P. stipuleanatus: The global potential suitable distribution of this plant is limited to several countries in Asia and South America (Figure 6A), the total area is 2.05 × 105 km2. The top three distribution areas are China (108.03 × 103 km2), Brazil (35.92 × 103 km2), and Burma (27.33 × 103 km2) (Figure 6B).
2.2.2. Chinese Potential Distribution
3. Discussion
3.1. Impact of Environmental Variables on Medicinal Plants
3.2. Distribution of Suitable Habitats
3.3. Features of GMPGIS
- (1)
- GMPGIS adopts the multi-index comprehensive evaluation for the quantitative and spatial analyses of the four medicinal plants.
- (2)
- The results intuitively show the range of ecological factors and the best potential ecological areas of the plants.
- (3)
- GMPGIS also explores the area with similar climates and soils of sampling points for medicinal plants.
- (4)
- This system houses over 240 medicinal plants global sampling points.
- (5)
- The model has high accuracy for medicinal plants.
4. Methods
4.1. GMPGIS Database
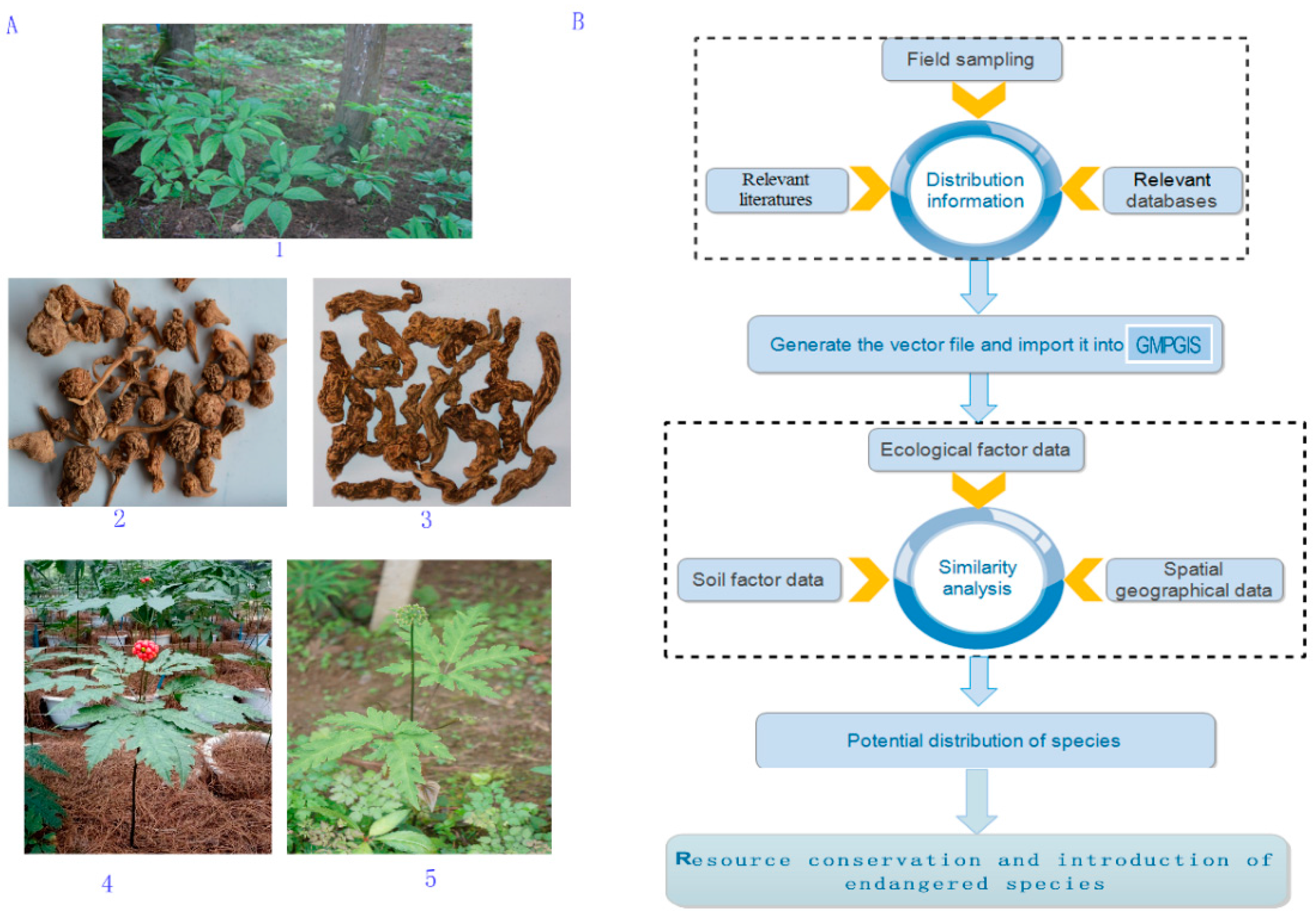
4.2. Data Collection
4.3. Principle and Algorithm of GMPGIS
- (1)
- Step 1: Linear normalization is performed on the original data. Suppose that minA and maxA were the minimum and maximum values of a layer A. Linear normalization maps a value Vi of A to Vi in the range [newminA, newmaxA] by computing the following:
- (2)
- In our study, an improved k-means was adopted to evaluate the ecological suitability models. A range-based partitioning technique uses the critical size of a cluster Di to represent that cluster. Conceptually, the critical of a cluster is its marginal value di. The range can be defined in various ways, such as by the polyhedron assigned to the cluster. The difference between an object p ∊ Di and di, the representative of the cluster, is measured by dist (p, di), where dist (x, y) is the Euclidean distance between two points, x and y. The quality of cluster Di can be measured by the within-cluster variation, which is the sum of the squared error between all points in Di and the range di, defined as follows:wherewhere E is the sum of the squared error for all points in the data set, p is the point in space representing a given object, and di is the range of cluster Di (both p and di were multidimensional). newminA is the minimum value after standardizing the layer; newmaxA is the maximum value after standardizing the layer.
- (3)
- According to the results of the distance calculation [Mind, Maxd], the grid was classified, and the most similar ecological area was discovered.
- (4)
- The suitable soil layer and climatic factors in the Euclidean distance layer were intersected.
4.4. Analysis by MaxEnt
5. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yang, X.; Wang, R.; Zhang, S.; Zhu, W.; Tang, J.; Liu, J.; Chen, P.; Zhang, D.; Ye, W.; Zheng, Y. Polysaccharides from Panax japonicus C.A. Meyer and their antioxidant activities. Carbohydr. Polym. 2014, 101, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Rai, A.; Yamazaki, M.; Takahashi, H.; Nakamura, M.; Kojoma, M.; Suzuki, H.; Saito, K. RNA-seq Transcriptome Analysis of Panax japonicus, and Its Comparison with Other Panax Species to Identify Potential Genes Involved in the Saponins Biosynthesis. Front. Plant Sci. 2016, 7, e0144. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, R.; Zeng, W.; Zhu, W.; Zhang, X.; Wu, C.; Song, J.; Zheng, Y.; Chen, P. Resource investigation of traditional medicinal plant Panax japonicus (T. Nees) C.A. Mey and its varieties in China. J. Ethnopharmacol. 2015, 166, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.X.; Li, M.R.; Li, Y.L.; Jiang, P.; Zhang, C.; Pan, Y.Z.; Liu, B.; Xiao, H.X.; Li, L.F. The impacts of polyploidy, geographic and ecological isolations on the diversification of Panax (Araliaceae). BMC Plant Biol. 2015, 15, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, Y.; Wang, Y.; Abozeid, A.; Zu, Y.G.; Zhang, X.N.; Tang, Z.H. GC-MS Metabolomic Analysis to Reveal the Metabolites and Biological Pathways Involved in the Developmental Stages and Tissue Response of Panax ginseng. Molecules 2017, 22, 496. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, C.Z.; Zhu, X.Y.; Wan, J.Y.; Zhang, J.; Li, W.; Ruan, C.C.; Yuan, C.S. Dynamic Changes in Neutral and Acidic Ginsenosides with Different Cultivation Ages and Harvest Seasons: Identification of Chemical Characteristics for Panax ginseng Quality Control. Molecules 2017, 22, 734. [Google Scholar] [CrossRef] [PubMed]
- Xia, P.; Bai, Z.; Liang, T.; Yang, D.; Liang, Z.; Yan, X.; Liu, Y. High-performance liquid chromatography based chemical fingerprint analysis and chemometric approaches for the identification and distinction of three endangered Panax plants in Southeast Asia. J. Sep. Sci. 2016, 39, 3880–3888. [Google Scholar] [CrossRef] [PubMed]
- Pimm, S.L.; Jenkins, C.N.; Abell, R.; Brooks, T.M.; Gittleman, J.L.; Joppa, L.N.; Raven, P.H.; Roberts, C.M.; Sexton, J.O. The biodiversity of species and their rates of extinction, distribution, and protection. Science 2014, 344, 1246752. [Google Scholar] [CrossRef] [PubMed]
- Herrick, K.A.; Huettmann, F.; Lindgren, M.A. A global model of avian influenza prediction in wild birds: The importance of northern regions. Vet. Res. 2017, 44, 42. [Google Scholar] [CrossRef] [PubMed]
- Han, X.S.; Guo, Y.M.; Mi, C.R.; Huettmann, F. Machine Learning Model Analysis of Breeding Habitats for the Blacknecked Crane in Central AsianUplands under Anthropogenic Pressures. Sci. Rep. 2017, 7, 6144. [Google Scholar] [CrossRef]
- Mi, C.R.; Zu, Q.; He, L. Climate change would enlarge suitable planting areas of sugarcanes in China. Int. J. Plant Prod. 2017, 11, 151–166. [Google Scholar]
- Jiao, S.G.; Huettmann, F.; Guo, Y.; Li, X.; Ouyang, Y. Advanced long-term birdbanding and climate data mining in spring confirm passerine population declines forhe Northeast Chinese-Russian flyway. Glob. Planet. Chang. 2016, 144, 17–33. [Google Scholar] [CrossRef]
- Guo, Y.M.; Huettmann, F.; Guo, Y.; Li, X.; Ouyang, Y. Climate envelope predictions indicatean enlarged suitable wintering distribution for Great Bustards (Otis tarda dybowski) in China for the 21st century. Peerj 2016, 4, e1630. [Google Scholar] [CrossRef]
- Warren, D.L.; Seifert, S.N. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. A Publ. Ecol. Soc. Am. 2011, 21, 335–342. [Google Scholar] [CrossRef]
- Mi, C. Why choose Random Forest to predict rare species distribution with few samples in large undersampled areas? Three Asian crane species models provide supporting evidence. Peerj 2017, 5, e2849. [Google Scholar] [CrossRef] [PubMed]
- Kalkvik, H.M.; Parkinson, C.L. Investigating niche and lineage diversification in widely distributed taxa: Phylogeography and ecological niche modeling of the Peromyscus maniculatus species group. Ecography 2012, 35, 54–64. [Google Scholar] [CrossRef]
- Meng, X.X.; Huang, L.F.; Dong, L.L.; Li, X.-W.; Wei, F.G.; Chen, Z.J.; Wu, J.J.; Sun, C.Z.; Yu, Y.-Q.; Chen, S.L. Analysis of global ecology of Panax notoginseng in suitability and quality. Acta Pharm. Sin. 2016, 51, 1483–1493. [Google Scholar]
- Shen, L.; Wu, J.J.; Li, X.-W.; Xu, J.; Dong, L.L.; Sang, M.C.; Sun, C.Z.; Fujihara, N.; Chen, S.L. A study of global ecological adaptability and field selection practices of Panax ginseng. China J. Chin. Mater. Med. 2016, 41, 3314–3322. [Google Scholar]
- Sánchez-Fernández, D.; Lobo, J.M. Species distribution models that do not incorporate global data misrepresent potential distributions: A case study using Iberian diving beetles. Divers. Distrib. 2015, 17, 163–171. [Google Scholar] [CrossRef]
- Nag, C.; Chetan, N.; Karanth, K.P.; Praveen, K.K.; Gururaja, K.V.; Vasudeva, G.K. Delineating ecological boundaries of Hanuman langur species complex in peninsular India using MaxEnt modeling approach. PLoS ONE 2014, 9, e87804. [Google Scholar] [CrossRef]
- Qin, Z; Zhang, J.E.; Ditommaso, A.; Wang, R.L.; Wu, R.S. Predicting invasions of Wedelia trilobata (L.) Hitchc. With Maxent and GARP models. J. Plant Res. 2015, 128, 673–755. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.W.; Cross, D.E.; Crump, H.L.; Drost, C.J.; Thomas, C.J. Climate suitability for European ticks: Assessing species distribution models against null models and projection under AR5 climate. Parasites Vectors 2015, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Gormley, A.M.; Woodford, L. Using presence-only and presence–absence data to estimate the current and potential distributions of established invasive species. J. Appl. Ecol. 2011, 48, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.P. A framework for using niche models to estimate impacts of climate change on species distributions. Ann. N. Y. Acad. Sci. 2013, 1297, 8–28. [Google Scholar] [CrossRef] [PubMed]
- Guisan, A.; Tingley, R.; Baumgartner, J.B.; Naujokaitislewis, I.; Sutcliffe, P.R.; Tulloch, A.I.; Regan, T.J.; Brotons, L.; Mcdonaldmadden, E.; Mantykapringle, C. Predicting species distributions for conservation decisions. Ecol. Lett. 2013, 16, 1424–1435. [Google Scholar] [CrossRef] [PubMed]
- Folmer, E.O.; Olff, H.; Piersma, T. How well do food distributions predict spatial distributions of shorebirds with different degrees of self-organization? J. Anim. Ecol. 2010, 79, 747–756. [Google Scholar] [CrossRef] [PubMed]
- Moat, J.; Williams, J.; Baena, S.; Wilkinson, T.; Gole, T.W.; Challa, Z.K.; Demissew, S.; Davis, A.P. Resilience potential of the Ethiopian coffee sector under climate change. Nat. Plants 2017, 3, 17081. [Google Scholar] [CrossRef] [PubMed]
- Elith, J.; Phillips, S.J.; Hastie, T.; Dudík, M.; Chee, Y.E.; Yates, C.J. A statistical explanation of MaxEnt for ecologists. Divers. Distrib. 2011, 17, 43–57. [Google Scholar] [CrossRef]
- Wang, E.H.; Liu, S.L.; Song, Z. Study Progression about Medicinal Plant Cold-resistance of Physiological. Chin. Agric. Sci. Bull. 2014, 30, 79–84. [Google Scholar]
- Hou, Y.; Ma, Y.; Zou, L.S. The Effcts of ecological factors on the secondary metabolites in medicinal plants and their research methods. Lishizhen Med. Mater. Med. Res. 2015, 26, 187–190. [Google Scholar]
- Xie, C.X.; Suo, F.M.; Jia, G.L. Correlation between ecological factors and ginsenosides. Acta Ecol. Sin. 2011, 31, 7551–7553. [Google Scholar]
- Jia, G.-L.; Huang, L.-F.; Suo, F.-M. Correlation between ginsenoside contents in Panax ginseng roots and ecological factors, and ecological division of ginseng plantation in China. Chin. J. Plant Ecol. 2012, 36, 302–312. [Google Scholar]
- Han, Z.M.; Zhao, S.J.; Liu, C.J. Effects of shading on growth and quality of triennial Clematis manshurica Rupr. Acta Ecol. Sin. 2011, 31, 6005–6012. [Google Scholar]
- Zuang, W.J.; Yu, R.C.; Zhou, T.J. Spatial Distribution and Temporal Variation of Soil Moisture over China Part II: The Evaluations for Coupled Models’ Simulations. Chin. J. Atmos. Sci. 2008, 32, 1128–1146. [Google Scholar]
- Lu, S.P.; Sui, X.-X.; Sun, Q. Biological Functions of Secondary Metabolism of Medicinal Plants and Influences of Ecological Environment. Nat. Prod. Res. Dev. 2006, 18, 1027–1032. [Google Scholar]
- Ma, R.-J.; Jiang, Z.-G. Impact of global climate change on wildlife. Acta Ecol. Sin. 2005, 25, 3061–3066. [Google Scholar]
- Zou, H.Y.; Zhou, D.; Zhao, H. Determination of Chikusetsusaponin V and IV a in Panax Japonicus by RP-HPLC. J. Liaoning Univ. Tradit. Chin. Med. 2015, 17, 52–54. [Google Scholar]
- Wu, J.; Tang, H.; Huang, L.F. Research and analysis of globally ecological suitability for Taxus plants. Acta Pharm. Sin. 2017, 52, 1186–1195. [Google Scholar]
- Chen, S.L.; Xiang, L.; Li, L.; Lan, W.U.; Huang, L.F.; Dong, Z.; Peng, S. Global strategy and raw material production on artemisinin resources regeneration (in Chinese). Chin. Sci. Bull. 2017, 62, 1982–1996. [Google Scholar] [CrossRef]
- Hijmans, R.; Cameron, S.; Parra, J.L.; Jones, P.G.; Andy, J. Very high resolution interpolation climate surfaces for global lant areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
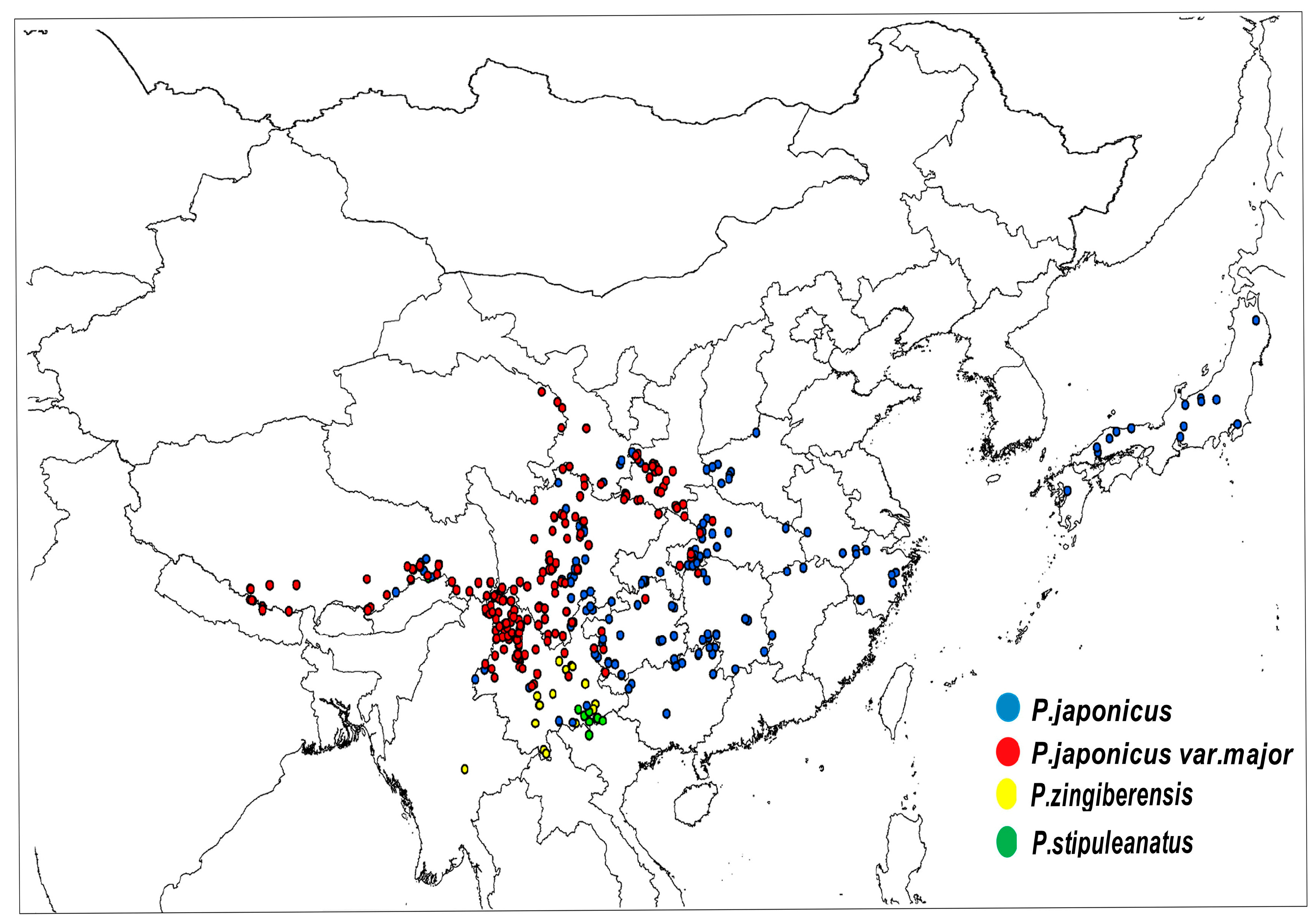
| Latin Name of Species | Sampling Distribution | Sampling Points |
|---|---|---|
| P. japonicus | Global: China, Japan, North Korea China: Yunnan, Guizhou, Sichuan, Hubei, etc. | 176 |
| P. japonicus var.major | China: Shaanxi, Sichuan, Yunan, Gansu, Guizhou, etc. | 221 |
| P. zingiberensis | Global: China, Burma China: Yunnan | 102 |
| P. stipuleanatus | Global: China, Burma China: Yunnan | 101 |
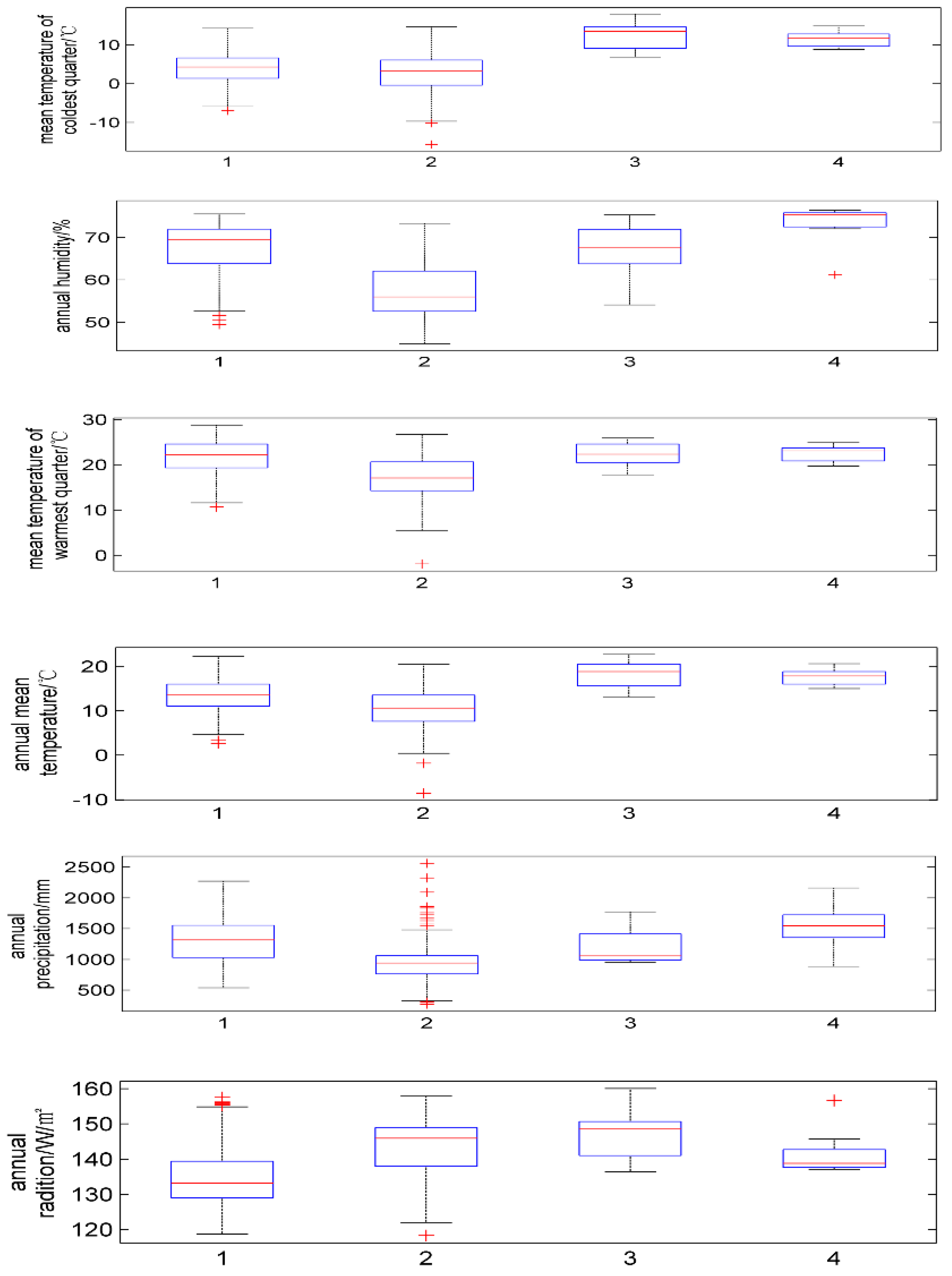
| Latin Name of Species | Annual Mean Temperature/°C | Mean Temperature of Coldest Quarter/°C | Mean Temperature of Warmest Quarter/°C | Annual Precipitation/mm | Annual Humidity/% | Annual Average Radiation/w·m−2 |
|---|---|---|---|---|---|---|
| P. japonicus | 2.6~22.3 | −7.0~14.3 | 10.7~28.8 | 539~2273 | 49.4~75.5 | 118.7~157.6 |
| soil types: Lixisols, Arenosols, Chernozems, Luvisols, Ferralsols, Acrisols, Andosols, etc. | ||||||
| P. japonicus var.major | −8.6~20.5 | −17.5~14.7 | −1.9~26.7 | 272~2562 | 44.9~73.3 | 118.3~157.9 |
| soil types: Lixisols, Chernozems, Greyzems, Leptosols, Arenosols, etc. | ||||||
| P. zingiberensis | 13~22.7 | 6.9~18.0 | 17.8~25.9 | 957~1772 | 54.1~75.3 | 136.3~160.1 |
| soil types: Acrisols, Arenosols, Arenosols, etc. | ||||||
| P. stipuleanatus | 15~20.6 | 8.8–14.9 | 19.7~24.9 | 888~2161 | 61.2~76.4 | 136.9~156.0 |
| soil types: Acrisols, Arenosols, etc. | ||||||
| Variable | Contribution (%) | |||
|---|---|---|---|---|
| P. japonicus | P. japonicus var. major | P. zingiberensis | P. stipuleanatus | |
| Cold | 55.5 | 44.7 | 45.6 | 43.0 |
| Pre | 36.0 | 5.7 | 37.8 | 42.3 |
| Tem | 4.5 | 7.1 | 4.9 | 2.8 |
| Warm | 3.0 | 4.5 | 9.4 | 9.7 |
| Rad | 0.6 | 37.7 | 2.0 | 0.1 |
| hum | 0.4 | 2.4 | 0.3 | 2.0 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, Z.; Wu, J.; Meng, X.; Li, J.; Huang, L. Predicting the Global Potential Distribution of Four Endangered Panax Species in Middle-and Low-Latitude Regions of China by the Geographic Information System for Global Medicinal Plants (GMPGIS). Molecules 2017, 22, 1630. https://doi.org/10.3390/molecules22101630
Du Z, Wu J, Meng X, Li J, Huang L. Predicting the Global Potential Distribution of Four Endangered Panax Species in Middle-and Low-Latitude Regions of China by the Geographic Information System for Global Medicinal Plants (GMPGIS). Molecules. 2017; 22(10):1630. https://doi.org/10.3390/molecules22101630
Chicago/Turabian StyleDu, Zhixia, Jie Wu, Xiangxiao Meng, Jinhua Li, and Linfang Huang. 2017. "Predicting the Global Potential Distribution of Four Endangered Panax Species in Middle-and Low-Latitude Regions of China by the Geographic Information System for Global Medicinal Plants (GMPGIS)" Molecules 22, no. 10: 1630. https://doi.org/10.3390/molecules22101630
APA StyleDu, Z., Wu, J., Meng, X., Li, J., & Huang, L. (2017). Predicting the Global Potential Distribution of Four Endangered Panax Species in Middle-and Low-Latitude Regions of China by the Geographic Information System for Global Medicinal Plants (GMPGIS). Molecules, 22(10), 1630. https://doi.org/10.3390/molecules22101630





