Grape Polyphenols’ Effects in Human Cardiovascular Diseases and Diabetes
Abstract
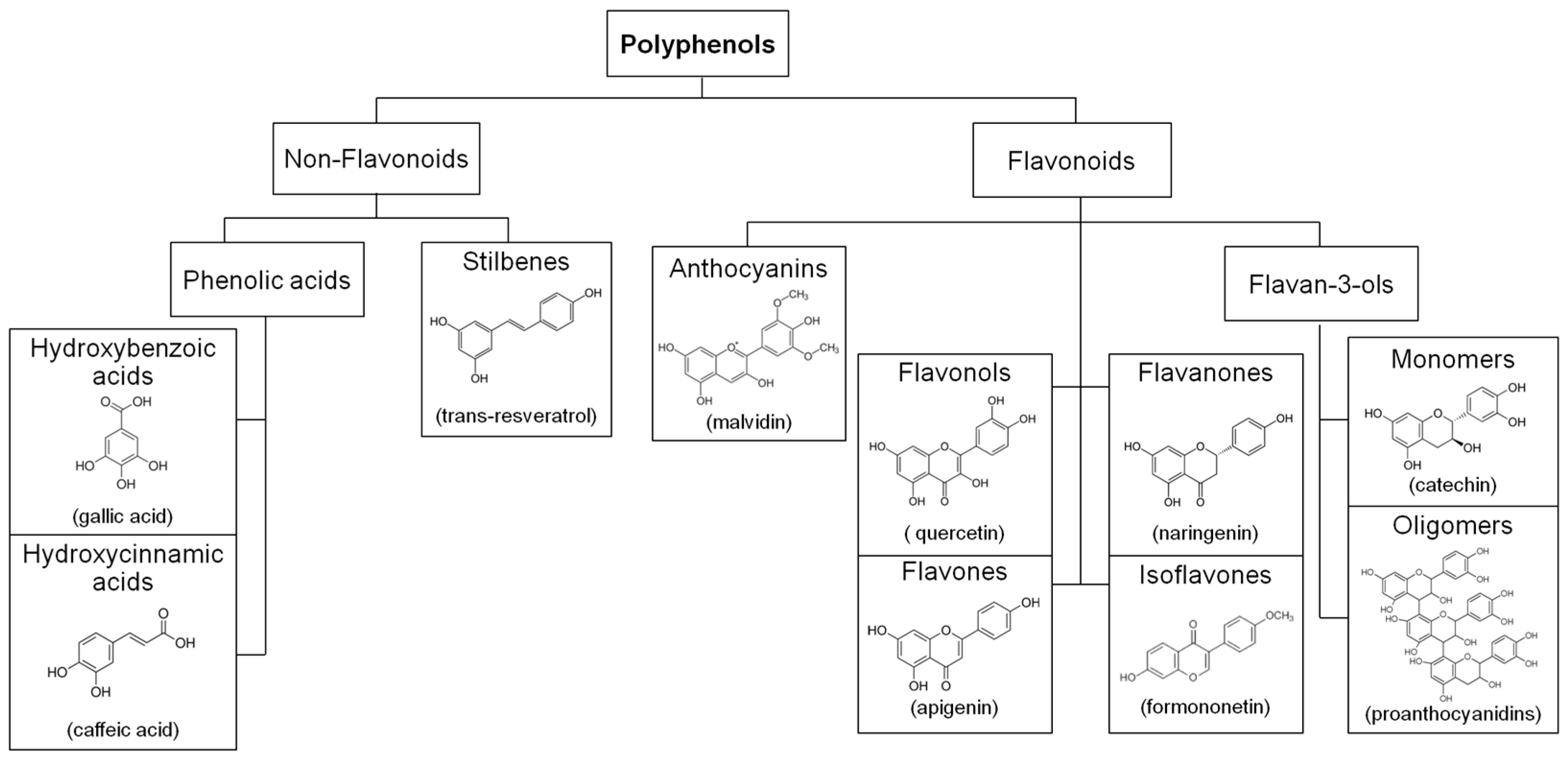
:1. Introduction
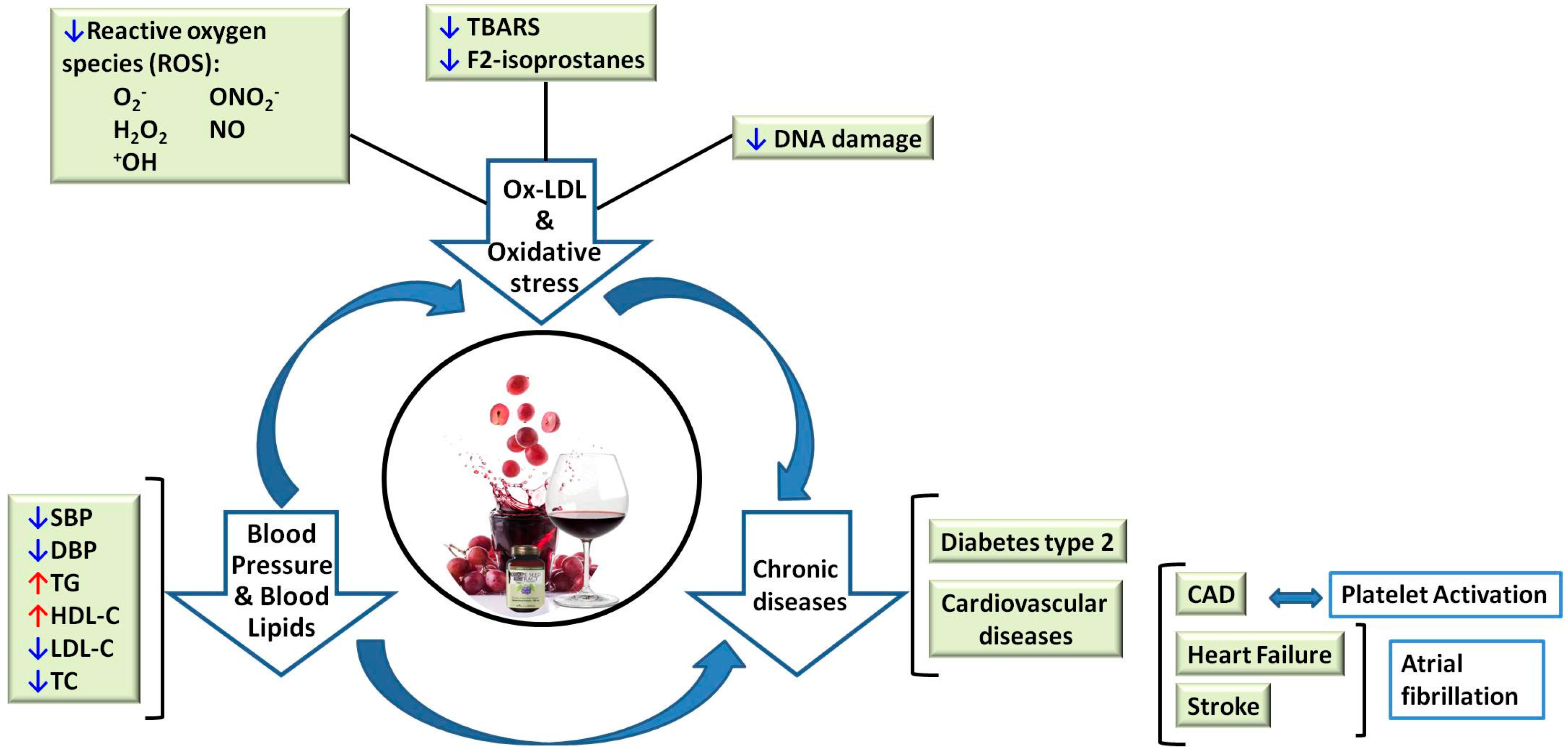
2. Cardiovascular Disease
2.1. Blood Pressure (BP)
2.2. Blood Lipids
2.3. LDL Oxidation and Oxidative Stress
2.4. Platelet Activation
2.5. Atrial Fibrillation
3. Diabetes Mellitus
4. Conclusions
Conflicts of Interest
References
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [PubMed]
- Lindberg, M.L.; Amsterdam, E.A. Alcohol, wine, and cardiovascular health. Clin. Cardiol. 2008, 31, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Friedman, L.A.; Kimball, A.W. Coronary heart disease mortality and alcohol consumption in framingham. Am. J. Epidemiol. 1986, 124, 481–489. [Google Scholar] [PubMed]
- Muntwyler, J.; Hennekens, C.H.; Buring, J.E.; Gaziano, J.M. Mortality and light to moderate alcohol consumption after myocardial infarction. Lancet 1998, 352, 1882–1885. [Google Scholar] [CrossRef]
- Gronbaek, M.; Deis, A.; Sorensen, T.I.; Becker, U.; Schnohr, P.; Jensen, G. Mortality associated with moderate intakes of wine, beer, or spirits. BMJ 1995, 310, 1165–1169. [Google Scholar] [CrossRef] [PubMed]
- Renaud, S.; de Lorgeril, M. Wine, alcohol, platelets, and the french paradox for coronary heart disease. Lancet 1992, 339, 1523–1526. [Google Scholar] [CrossRef]
- Kuhnau, J. The flavonoids. A class of semi-essential food components: Their role in human nutrition. World Rev. Nutr. Diet 1976, 24, 117–191. [Google Scholar] [PubMed]
- Hertog, M.G.; Hollman, P.C.; Katan, M.B.; Kromhout, D. Intake of potentially anticarcinogenic flavonoids and their determinants in adults in The Netherlands. Nutr. Cancer 1993, 20, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Rexrode, K.M.; Hu, F.; Albert, C.M.; Chae, C.U.; Rimm, E.B.; Stampfer, M.J.; Manson, J.E. Dietary intakes of flavonols and flavones and coronary heart disease in US women. Am. J. Epidemiol. 2007, 165, 1305–1313. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jime ́nez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention. National Diabetes Fact Sheet: National Estimates and General Information on Diabetes and Prediabetes in the United States, 2011; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention: Atlanta, GA, USA, 2011.
- Wang, X.; Ouyang, Y.Y.; Liu, J.; Zhao, G. Flavonoid intake and risk of CVD: A systematic review and meta-analysis of prospective cohort studies. Br. J. Nutr. 2014, 111, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hartley, L.; Flowers, N.; Holmes, J.; Clarke, A.; Stranges, S.; Hooper, L.; Rees, K. Green and black tea for the primary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2013, 6, CD009934. [Google Scholar]
- Wightman, J.D.; Heuberger, R.A. Effect of grape and other berries on cardiovascular health. J. Sci. Food Agric. 2015, 95, 1584–1597. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xia, N.; Förstermann, U. Cardiovascular effects and molecular targets of resveratrol. Nitric Oxide 2012, 26, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R.; Cushman, W.C.; Green, L.A.; Izzo, J.L., Jr.; Jones, D.W.; Materson, B.J.; Oparil, S.; Wright, J.T.; et al. Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003, 42, 1206–1252. [Google Scholar] [CrossRef] [PubMed]
- Papadogiannis, D.E.; Protogerou, A.D. Blood pressure variability: A confounder and a cardiovascular risk factor. Hypertens. Res. 2010, 3, 162–163. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Status Report on Noncommunicable Diseases 2014; WHO Press: Geneva, Switzerland, 2014. [Google Scholar]
- Lindsley, C.W. 2013 Trends and Statistics for Prescription Medications in the United States: CNS Highest Ranked and Record Number of Prescriptions Dispensed. ACS Chem. Neurosci. 2015, 6, 356–357. [Google Scholar] [CrossRef] [PubMed]
- Eckel, R.H.; Jakicic, J.M.; Ard, J.D.; de Jesus, J.M.; Houston Miller, N.; Hubbard, V.S.; Lee, I.M.; Lichtenstein, A.H.; Loria, C.M.; Millen, B.E.; et al. 2013 AHA/ACC Guideline on lifestyle management to reduce cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129, S76–S99. [Google Scholar] [CrossRef] [PubMed]
- Wengreen, H.; Munger, R.G.; Cutler, A.; Quach, A.; Bowles, A.; Corcoran, C.; Tschanz, J.T.; Norton, M.C.; Welsh-Bohmer, K.A. Prospective study of Dietary Approaches to Stop Hypertension- and Mediterranean-style dietary patterns and age-related cognitive change: The Cache County Study on Memory, Health and Aging. Am. J. Clin. Nutr. 2013, 98, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Lewington, S.; Clarke, R.; Qizilbash, N.; Peto, R.; Collins, R. Prospective Studies Collaboration. Age-specific relevance of usual blood pressure to vascular mortality: A meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002, 360, 1903–1913. [Google Scholar] [PubMed]
- Park, Y.K.; Kim, J.-S.; Kang, M.-H. Concord grape juice supplementation reduces blood pressure in Korean hypertensive men: Double-blind, placebo controlled intervention trial. Biofactors 2004, 22, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Dohadwala, M.M.; Hamburg, N.M.; Holbrook, M.; Kim, B.H.; Duess, M.-A.; Levit, A.; Titas, M.; Chung, W.B.; Vincent, F.B.; Caiano, T.L.; et al. Effect of grape juice on ambulatory blood pressure in pre-hypertension and Stage 1 hypertension. Am. J. Clin. Nutr. 2010, 92, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Ras, R.T.; Zock, P.L.; Zebregs, Y.E.; Johnston, N.R.; Webb, D.J.; Draijer, R. Effect of polyphenol-rich grape seed extract on ambulatory blood pressure in subjects with pre- and stage I hypertension. Br. J. Nutr. 2013, 110, 2234–2241. [Google Scholar] [CrossRef] [PubMed]
- Sivaprakasapillai, B.; Edirisinghe, I.; Randolph, J.; Steinberg, F.; Kappagoda, T. Effect of grape seed extract on blood pressure in subjects with the metabolic syndrome. Metabolism 2009, 58, 1743–1746. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M. Effect of grape seed extract and quercetin on cardiovascular and endothelial parameters in high-risk subjects. J. Biomed. Biotechnol. 2004, 5, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Sano, A.; Uchida, R.; Saito, M.; Shioya, N.; Komori, Y.; Tho, Y.; Hashizume, N. Beneficial effects of grape seed extract on malondialdehyde-modified LDL. J. Nutr. Sci. Vitaminol. 2007, 53, 174–182. [Google Scholar] [CrossRef] [PubMed]
- Van Mierlo, L.A.J.; Zock, P.L.; van der Knaap, H.C.M.; Draijer, R. Grape polyphenols do not affect vascular function in healthy men. J. Nutr. 2010, 140, 1769–1773. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.C.; Hodgson, J.M.; Croft, K.D.; Burke, V.; Beilin, L.J.; Puddey, I.B. The combination of vitamin C and grape-seed polyphenols increases blood pressure: A randomized, double-blind, placebo-controlled trial. J. Hypertens. 2005, 23, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Barona, J.; Aristizabal, J.C.; Blesso, C.N.; Volek, J.S.; Fernandez, M.L. Grape polyphenols reduce blood pressure and increase flow-mediated vasodilation in men with metabolic syndrome. J. Nutr. 2012, 142, 1626–1632. [Google Scholar] [CrossRef] [PubMed]
- Erlund, I.; Koli, R.; Alfthan, G.; Marniemi, J.; Puukka, P.; Mustonen, P.; Mattila, P.; Jula, A. Favorable effects of berry consumption on platelet function, blood pressure, and HDL cholesterol. Am. J. Clin. Nutr. 2008, 87, 323–331. [Google Scholar] [PubMed]
- Feringa, H.H.; Laskey, D.A.; Dickson, J.E.; Coleman, C.I. The effect of grape seed extract on cardiovascular risk markers: A meta-analysis of randomized controlled trials. J. Am. Diet. Assoc. 2011, 111, 1173–1181. [Google Scholar] [CrossRef] [PubMed]
- Draijer, R.; de Graaf, Y.; Slettenaar, M.; de Groot, E.; Wright, C.I. Consumption of a polyphenol-rich grape-wine extract lowers ambulatory blood pressure in mildly hypertensive subjects. Nutrients 2015, 7, 3138–3153. [Google Scholar] [CrossRef] [PubMed]
- Vaisman, N.; Niv, E. Daily consumption of red grape cell powder in a dietary dose improves cardiovascular parameters: A double blind, placebo-controlled, randomized study. Int. J. Food Sci. Nutr. 2015, 66, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Biesinger, S.; Michaels, H.A.; Quadros, A.S.; Qian, Y.; Rabovsky, A.B.; Badgerand, R.S.; Jalili, T. A combination of isolated phytochemicals and botanical extractlowers diastolic blood pressure in a randomized controlled trial of hypertensivsubjects. Eur. J. Clin. Nutr. 2016, 70, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Delles, C.; Dymott, J.A.; Neisius, U.; Rocchiccioli, J.P.; Bryce, G.J.; Moreno, M.U.; Carty, D.M.; Berg, G.A.; Hamilton, C.A.; Dominiczak, A.F. Reduced LDL-cholesterol levels in patients with coronary artery disease are paralleled by improved endothelial function: An observational study in patients from 2003 and 2007. Atherosclerosis 2010, 211, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.G.; Stukovsky, K.H.; Zhao, X.Q. Simultaneous low-density lipoprotein-C lowering and high-density lipoprotein-C elevation for optimum cardiovascular disease prevention with various drug classes, and their combinations: A meta-analysis of 23 randomized lipid trials. Curr. Opin. Lipidol. 2006, 17, 631–636. [Google Scholar] [CrossRef] [PubMed]
- NACB LMPGCommittee Members; Myers, G.L.; Christenson, R.H.M.; Cushman, M.; Ballantyne, C.M.; Cooper, G.R.; Pteiffer, C.M.; Grundy, S.M.; Labarthe, D.R.; Levy, D.; et al. National Academy of Clinical Biochemistry Laboratory Medicine Practice Guidelines: Emerging biomarkers for primary prevention of cardiovascular disease. Clin. Chem. 2009, 55, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Zern, T.L.; Fernandez, M.L. Cardioprotective effects of dietary polyphenols. J. Nutr. 2005, 135, 2291–2294. [Google Scholar] [PubMed]
- Castilla, P.; Echarri, R.; Davalos, A.; Cerrato, F.; Ortega, H.; Teruel, J.L.; Fernández-Lucas, M.; Gómez-Coronado, D.; Ortuño, J.; Lasunción, M.A. Concentrated red grape juice exerts antioxidant, hypolipidemic, and antiinflammatory effects in both hemodialysis patients and healthy subjects. Am. J. Clin. Nutr. 2006, 84, 252–262. [Google Scholar] [PubMed]
- Castilla, P.; Davalos, A.; Teruel, J.L.; Cerrato, F.; Fernandez-Lucas, M.; Merino, J.L.; Sánchez-Martín, C.C.; Ortuño, J.; Lasunción, M.A. Comparative effects of dietary supplementation with red grape juice and vitamin E on production of superoxide by circulating neutrophil NADPH oxidase in hemodialysis patients. Am. J. Clin. Nutr. 2008, 87, 1053–1061. [Google Scholar] [PubMed]
- Khadem-Ansari, M.H.; Rasmi, Y.; Ramezani, F. Effects of red grape juice consumption on high density lipoprotein-cholesterol, apolipoprotein AI, apolipoprotein B and homocysteine in healthy human volunteers. Open Biochem. J. 2010, 4, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Albers, A.R.; Varghese, S.; Vitseva, O.; Vita, J.A.; Freedman, J.E. The antiinflammatory effects of purple grape juice consumption in subjects with stable coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 2004, 24, e179–e180. [Google Scholar] [CrossRef] [PubMed]
- Stein, J.H.; Keevil, J.G.; Wiebe, D.A.; Aeschlimann, S.; Folts, J.D. Purple grape juice improves endothelial function and reduces the susceptibility of LDL cholesterol to oxidation in patients with coronary artery disease. Circulation 1999, 100, 1050–1055. [Google Scholar] [CrossRef] [PubMed]
- O’Byrne, D.J.; Devaraj, S.; Grundy, S.M.; Jialal, I. Comparison of the antioxidant effects of Concord grape juice flavonoids and α-tocopherol on markers of oxidative stress in healthy adults. Am. J. Clin. Nutr. 2002, 76, 1367–1374. [Google Scholar] [PubMed]
- Evans, M.; Wilson, D.; Guthrie, N. A randomized, double-blind, placebo-controlled, pilot study to evaluate the effect of whole grape extract on antioxidant status and lipid profile. J. Funct. Foods 2014, 7, 680–691. [Google Scholar] [CrossRef]
- Razavi, S.M.; Gholamin, S.; Eskandari, A.; Mohsenian, N.; Ghorbanihaghjo, A.; Delazar, A.; Rashtchizadeh, N.; Keshtkar-Jahromi, M.; Argani, H. Red grape seed extract improves lipid profiles and decreases oxidized low-density lipoprotein in patients with mild hyperlipidemia. J. Med. Food 2013, 16, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Vinson, J.A.; Proch, J.; Bose, P. MegaNatural® gold grapeseed extract: In vitro antioxidant and in vivo human supplementation studies. J. Med. Food 2001, 4, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Van der Made, S.M.; Plat, J.; Mensink, R.P. Resveratrol does not influence metabolic risk markers related to cardiovascular health in overweight and slightly obese subjects: A randomized, placebo-controlled crossover trial. PLoS ONE 2015, 10, e0118393. [Google Scholar] [CrossRef] [PubMed]
- Magyar, K.; Halmosi, R.; Palfi, A.; Feher, G.; Czopf, L.; Fulop, A.; Battyany, I.; Sumegi, B.; Toth, K.; Szabados, E. Cardioprotection by resveratrol: A human clinical trial in patients with stable coronary artery disease. Clin. Hemorheol. Microcirc. 2012, 50, 179–187. [Google Scholar] [PubMed]
- Teissedre, P.L.; Frankel, E.N.; Waterhouse, A.L.; Peleg, H.; German, J.B. Inhibition of in vitro human LDL oxidation by phenolic antioxidants from grapes and wines. J. Sci. Food Agric. 1996, 70, 55–61. [Google Scholar] [CrossRef]
- Teissedre, P.L.; Waterhouse, A.L.; Frankel, E.N. Principal phytochemicals in French syrah and grenache Rhône wines and their antioxidant activity in inhibiting oxidation of human low density lipoproteins. J. Int. Sci. Vigne Vin 1995, 29, 205–212. [Google Scholar] [CrossRef]
- Frankel, E.N.; Waterhouse, A.L.; Teissedre, P.L. Principal phenolic phytochemicals in selected California wines and their antioxidant activity in inhibiting oxidation of human low-density lipoproteins. J. Agric. Food Chem. 1995, 43, 890–894. [Google Scholar] [CrossRef]
- Miyagi, Y.; Miwa, K.; Inoue, H. Inhibition of human low-density lipoprotein oxidation by flavonoids in red wine and grape juice. Am. J. Cardiol. 1997, 80, 1627–1631. [Google Scholar] [CrossRef]
- Frankel, E.N.; Bosanek, C.A.; Meyer, A.S.; Silliman, K.; Kirk, L.L. Commercial grape juices inhibit the in vitro oxidation of human low-density lipoproteins. J. Agric. Food Chem. 1998, 46, 834–838. [Google Scholar] [CrossRef]
- Fuhrman, B.; Volkova, N.; Suraski, A.; Aviram, M. White wine with red wine-like properties: Increased extraction of grape skin polyphenols improves the antioxidant capacity of the derived white wine. J. Agric. Food Chem. 2001, 49, 3164–3168. [Google Scholar] [CrossRef] [PubMed]
- Toaldo, I.M.; Cruz, F.A.; de Alves, T.L.; de Gois, J.S.; Borges, D.L.; Cunha, H.P.; da Silva, E.L.; Bordignon-Luiz, M.T. Bioactive potential of Vitis labrusca L. grape juices from the Southern Region of Brazil: Phenolic and elemental composition and effect on lipid peroxidation in healthy subjects. Food Chem. 2015, 173, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Vigna, G.B.; Costantini, F.; Aldini, G.; Carini, M.; Catapano, A.; Schena, F.; Tangerini, A.; Zanca, R.; Bombardelli, E.; Morazzoni, P.; et al. Effect of a standardized grape seed extract on low-density lipoprotein susceptibility to oxidation in heavy smokers. Metabolism 2003, 52, 1250–1257. [Google Scholar] [CrossRef]
- Tomé-Carneiro, J.; Conzálvez, M.; Larrosa, M.; García-Almagro, F.J.; Avilés-Plaza, F.; Parra, S.; Yáñez-Gascón, M.J.; Ruiz-Ros, J.A.; García-Conesa, M.T.; Tomás-Barberán, F.A.; et al. Consumption of a grape extract supplement containing resveratrol decreases oxidized LDL and ApoB in patients undergoing primary prevention of cardiovascular disease: A triple-blind, 6-month follow-up, placebo-controlled, randomized trial. Mol. Nutr. Food Res. 2012, 56, 810–821. [Google Scholar] [CrossRef] [PubMed]
- Cathcart, M.K. Regulation of superoxide anion production by NADPH oxidase in monocytes/macrophages: Contributions to atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.E.; Parker, C., III; Li, L.; Perlman, J.A.; Frei, B.; Ivanov, V.; Deak, L.R.; Iafrati, M.D.; Folts, J.D. Select flavonoids and whole juice from purple grapes inhibit platelet function and enhance nitric oxide release. Circulation 2001, 103, 2792–2798. [Google Scholar] [CrossRef] [PubMed]
- Morrow, J.D. Quantification of Isoprostanes as Indices of Oxidant Stress and the Risk of Atherosclerosis in Humans. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Caccetta, R.A.; Burke, V.; Mori, T.A.; Beilen, L.J.; Puddey, I.B.; Croft, K.D. Red wine polyphenols, in the absence of alcohol, reduce lipid peroxidative stress in smoking subjects. Free Radic. Biol. Med. 2001, 30, 636–642. [Google Scholar] [CrossRef]
- Zern, T.L.; Wood, R.J.; Greene, C.; West, K.L.; Liu, Y.; Aggarwal, D.; Shachter, N.S.; Fernandez, M.L. Grape polyphenols exert a cardioprotective effect in pre- and postmenopausal women by lowering plasma lipids and reducing oxidative stress. J. Nutr. 2005, 135, 1911–1917. [Google Scholar] [PubMed]
- Hokayem, M.; Blond, E.; Vidal, H.; Lambert, K.; Meugnier, E.; Feillet-Coudray, C.; Coudray, C.; Pesenti, S.; Luyton, C.; Lambert-Porcheron, S.; et al. Grape polyphenols prevent induced oxidative stress and insulin resistance in first-degree relatives of type 2 diabetic patients. Diabetes Care 2013, 36, 1454–1461. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.K.; Lee, S.H.; Park, E.; Kim, J.S.; Kang, M.H. Changes in antioxidant status, blood pressure, and lymphocyte DNA damage from grape juice supplementation. Ann. N. Y. Acad. Sci. 2009, 1171, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Corredor, Z.; Rodríguez-Ribera, L.; Coll, E.; Montañés, R.; Diaz, J.M.; Ballarin, J.; Marcos, R.; Pastor, S. Unfermented grape juice reduce genomic damage on patienundergoing hemodialysis. Food Chem. Toxicol. 2016, 92, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Pace-Asciak, C.R.; Rounova, O.; Hahn, S.E.; Diamandis, E.P.; Goldberg, D.M. Wines and grape juices as modulators of platelet aggregation in healthy human subjects. Clin. Chim. Acta 1996, 246, 163–182. [Google Scholar] [CrossRef]
- Watkins, T.R.; Bierenbaum, M.L. Grape juice attenuates cardiovascular risk factors in the hyperlipemic subject. Pharm. Biol. 1998, 36, 75–80. [Google Scholar] [CrossRef]
- Keevil, J.G.; Osman, H.E.; Reed, J.D.; Folts, J.D. Grape Juice, But Not Orange Juice or Grapefruit Juice, Inhibits Human Platelet Aggregation. J. Nutr. 2000, 130, 53–56. [Google Scholar] [PubMed]
- Benjamin, E.J.; Levy, D.; Vaziri, S.M.; D’Agostino, R.B.; Belanger, A.J.; Wolf, P.A. Independent risk factors for atrial fibrillation in a populationbased cohort. The Framingham Heart Study. JAMA 1994, 271, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Cannon, C.P. An overview of stroke and the impact of atrial fibrillation. Am. J. Manag. Care 2010, 16, S273–S277. [Google Scholar] [PubMed]
- Lip, G.Y. Stroke in atrial fibrillation: Epidemiology and thromboprophylaxis. J. Thromb. Haemost. 2011, 9 (Suppl. 1), 344–351. [Google Scholar] [CrossRef] [PubMed]
- Nattel, S.; Harada, M. Atrial remodeling and atrial fibrillation: Recent advances and translational perspectives. J. Am. Coll. Cardiol. 2014, 63, 2335–2345. [Google Scholar] [CrossRef] [PubMed]
- Maisel, W.H.; Stevenson, L.W. Atrial fibrillation in heart failure: Epidemiology, pathophysiology, and rationale for therapy. Am. J. Cardiol. 2003, 91 (Suppl. 1), 2D–8D. [Google Scholar] [CrossRef]
- Larned, J.M.; Raja Laskar, S. Atrial fibrillation and heart failure. Congesrt. Heart Fail. 2009, 15, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Braunwald, E. Shattuck lecture—Cardiovascular medicine at the turn of the millennium: Triumphs, concerns, and opportunities. N. Engl. J. Med. 1997, 337, 1360–1369. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Wolf, P.A.; D’Agostino, R.B.; Silbershatz, H.; Kannel, W.B.; Levy, D. Impact of atrial fibrillation on the risk of death: The Framingham Heart Study. Circulation 1998, 98, 946–952. [Google Scholar] [CrossRef] [PubMed]
- Nattel, S.; Maguy, A.; LeBouter, S.; Yeh, Y.H. Arrhythmogenic ion-channel remodeling in the heart: Heart failure, myocardial infarction, and atrial fibrillation. Physiol. Rev. 2007, 87, 425–456. [Google Scholar] [CrossRef] [PubMed]
- Nerheim, P.; Birger-Botkin, S.; Piracha, L.; Olshansky, B. Heart failure and sudden death in patients with tachycardia-induced cardiomyopathy and recurrent tachycardia. Circulation 2004, 110, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Van Wagoner, D.R. Electrophysiological remodeling in human atrial fibrillation. Pacing Clin. Electrophysiol. 2003, 26, 1572–1575. [Google Scholar] [CrossRef] [PubMed]
- Ravens, U.; Poulet, C.; Wettwer, E.; Knaut, M. Atrial selectivity of antiarrhythmic drugs. J. Physiol. 2013, 591, 4087–4097. [Google Scholar] [CrossRef] [PubMed]
- Ehrlich, J.R.; Nattel, S. Novel approaches for pharmacological management of atrial fibrillation. Drugs 2009, 69, 757–774. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Sun, H.; Levesque, P. Antiarrhythmic drug therapy for atrial fibrillation: Focus on atrial selectivity and safety. Cardiovasc. Hematol. Agents Med. Chem. 2009, 7, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Negi, S.; Sovari, A.A.; Dudley, S.C., Jr. Atrial fibrillation: The emerging role of inflammation and oxidative stress. Cardiovasc. Hematol. Disord. Drug Targets 2010, 10, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Lin, J.L.; Lin, C.S.; Tsai, M.C.; Su, M.J.; Lai, L.P.; Huang, S.K. Activation of the calcineurin-nuclear factor of activated T-cell signal transduction pathway in atrial fibrillation. Chest 2004, 126, 1926–1932. [Google Scholar] [CrossRef]
- Doshi, D.; Morrow, J.P. Potential application of late sodium current blockade in the treatment of heart failure and atrial fibrillation. Rev. Cardiovasc. Med. 2009, 10 (Suppl. 1), S46–S52. [Google Scholar] [PubMed]
- Baczkó, I.; Light, P.E. Resveratrol and derivatives for the treatment of atrial fibrillation. Ann. N. Y. Acad. Sci. 2015, 1348, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- International Diabetes Federation. IDF Diabetes Atlas, 6th ed.; International Diabetes Federation: Brussels, Belgium, 2013; pp. 11–17. [Google Scholar]
- Roglic, G. WHO Global report on diabetes: A summary. Int. J. Non-Commun. Dis. 2016, 1, 3–8. [Google Scholar] [CrossRef]
- Thomas, T.; Pfeiffer, A.F.H. Foods for the prevention of diabetes: How do they work? Diabetes Metab. Res. Rev. 2012, 28, 25–49. [Google Scholar] [CrossRef] [PubMed]
- Stefek, M. Natural flavonoids as potential multifunctional agents in prevention of diabetic cataract. Interdiscip. Toxicol. 2011, 4, 6977. [Google Scholar] [CrossRef] [PubMed]
- Banihani, S.; Swedan, S.; Alguraan, Z. Pomegranate and type 2 diabetes. Nutr. Res. 2013, 33, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Dembinska-Kiec, A.; Mykkanen, O.; Kiec-Wilk, B.; Mykkanen, H. Antioxidant phytochemicals against type 2 diabetes. Br. J. Nutr. 2008, 99, ES109–ES117. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.B.; Högger, P. Influence of diabetes on the pharmacokinetic behavior of natural polyphenols. Curr. Drug Metab. 2014, 15, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.B.; Högger, P. Dietary Polyphenols and Type 2 Diabetes: Current Insights and Future Perspectives. Curr. Med. Chem. 2015, 22, 23–38. [Google Scholar] [CrossRef] [PubMed]
- Umeno, A.; Horie, M.; Murotomi, K.; Nakajima, Y.; Yoshida, Y. Antioxidative and Antidiabetic Effects of Natural Polyphenols and Isoflavones. Molecules 2016, 21, 708–723. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Kangath, A.; Burton-Freeman, B.; Jackson, L.S.; Edirisinghe, I. Polyphenol-rich fruits attenuate impaired endothelial function induced by glucose and free fatty acids in vitro in human endothelial cells. Int. J. Food Sci. Nutr. Diet. 2014, 3, 99–108. [Google Scholar]
- Aguirre, L.; Arias, N.; Macarulla, M.T.; Gracia, A.; Portillo, M.P. Beneficial effects of quercetin on obesity and diabetes. Open Nutraceut. J. 2011, 4, 189–198. [Google Scholar]
- Cao, H.; Xie, Y.X.; Chen, X.Q. Type 2 diabetes diminishes the benefits of dietary antioxidants: Evidence from the different free radical scavenging potential. Food Chem. 2015, 186, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Al-Awwadi, N.; Azay, J.; Poucheret, P.; Cassanas, G.; Krosniak, M.; Auger, C.; Gasc, F.; Rouanet, J.M.; Cros, G.; Teissedre, P.L. Antidiabetic activity of red wine polyphenolic extract, ethanol, or both in streptozotocin-treated rats. J. Agric. Food Chem. 2004, 52, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Al-Awwadi, N.A.; Bornet, A.; Azay, J.; Araiz, C.; Delbosc, S.; Cristol, J.P.; Linck, N.; Cros, G.; Teissedre, P.L. Red wine polyphenols alone or in association with ethanol prevent hypertension, cardiac hypertrophy, and production of reactive oxygen species in the insulin-resistant fructose-fed rat. J. Agric. Food Chem. 2004, 52, 5593–5607. [Google Scholar] [CrossRef] [PubMed]
- Pinent, M.; Blay, M.; Bladé, M.C.; Salvado, M.J.; Arola, L.; Ardévol, A. Grape seed-derived procyanidins have an antihyperglycemic effect in streptozotocin-induced diabetic rats and insulinomimetic activity in insulin-sensitive cell lines. Endocrinology 2004, 145, 4985–4990. [Google Scholar] [CrossRef] [PubMed]
- El-Alfy, A.T.; Ahmed, A.A.E.; Fatani, A.J. Protective effect of red grape seeds proanthocyanidins against induction of diabetes by alloxan in rats. Pharmacol. Res. 2005, 52, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Liu, Y.; Ren, G.; Yin, L.; Liang, X.; Geng, T.; Dang, H.; An, R. Resveratrol limits diabetes-associated cognitive decline in rats by preventing oxidative stress and inflammation and modulating hippocampal structural synaptic plasticity. Brain Res. 2016, 1650, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Polyphenols and Glycemic Control. Nutrientes 2016, 8, 17–44. [Google Scholar] [CrossRef] [PubMed]
- Zunino, S.J. Type 2 Diabetes and Glycemic Response to Grapes or Grape Products. J. Nutr. 2009, 139, 1794S–1800S. [Google Scholar] [CrossRef] [PubMed]
- Guilford, J.M.; Pezzuto, J.M. Wine and health: A review. Am. J. Enol. Vitic. 2011, 62, 471–486. [Google Scholar] [CrossRef]
- Wedick, N.M.; Pan, A.; Cassidy, A.; Rimm, E.B.; Sampson, L.; Rosner, B.; Willett, W.; Hu, F.B.; Sun, Q.; van Dam, R.M. Dietary flavonoid intakes and risk of type 2 diabetes in US men and women. Am. J. Clin. Nutr. 2012, 95, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.J.; Zhan, J.; Liu, X.L.; Wang, Y.; Ji, I.; He, Q.Q. Dietary flavonoids intake and risk of type 2 diabetes: A meta-analysis of prospective cohort studies. Clin. Nutr. 2014, 33, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, R.M.; Naidoo, N.; Landberg, R. Dietary flavonoids and the development of type 2 diabetes and cardiovascular diseases: Review of recent findings. Curr. Opin. Lipidol. 2013, 24, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Anhê, F.F.; Desjardins, Y.; Pilon, G.; Dudonné, S.; Genovese, M.I.; Lajolo, F.M.; Marette, A. Polyphenols and type 2 diabetes: A prospective review. PharmaNutrition 2013, 1, 105–114. [Google Scholar] [CrossRef]
- Cardona, F.; Andres-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuno, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Possemiers, S.; van de Wiele, T.; Guiot, Y.; Everard, A.; Rottier, O.; Geurts, L.; Naslain, D.; Neyrinck, A.; Lambert, D.M. Changes in gut microbiota control inflammation in obese mice through a mechanism involving GLP-2-driven improvement of gut permeability. Gut 2009, 58, 1091–1103. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Neyrinck, A.M.; Fava, F.; Knauf, C.; Burcelin, R.G.; Tuohy, K.M.; Gibson, G.; Delzenne, N.M. Selective increases of bifidobacteria in gut microflora improve high-fat-diet-induced diabetes in mice through a mechanism associated with endotoxaemia. Diabetologia 2007, 50, 2374–2383. [Google Scholar] [CrossRef] [PubMed]
- Yamakoshi, J.; Tokutake, S.; Kikuchi, M.; Kubota, Y.; Konishi, H.; Mitsuoka, T. Effect of proanthocyanidin-rich extract from grape seeds on human fecal flora and fecal odor. Microb. Ecol. Health Dis. 2001, 13, 25–31. [Google Scholar] [CrossRef]
- Queipo-Ortuno, M.I.; Boto-Ordonez, M.; Murri, M.; Gomez-Zumaquero, J.M.; Clemente-Postigo, M.; Estruch, R.; Cardona Diaz, F.; Andres-Lacueva, C.; Tinahones, F.J. Influence of red wine polyphenols and ethanol on the gut microbiota ecology and biochemical biomarkers. Am. J. Clin. Nutr. 2012, 95, 1323–1334. [Google Scholar] [CrossRef] [PubMed]
- Kar, P.; Laight, D.; Rooprai, H.K.; Shaw, K.M.; Cummings, M. Effects of grape seed extract in type 2 diabetic subjects at high cardiovascular risk: A double blind randomized placebo controlled trial examining metabolic markers, vascular tone, inflammation, oxidative stress and insulin sensitivity. Diabet. Med. 2009, 26, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Urquiaga, I.; D’Acuna, S.; Perez, D.; Dicenta, S.; Echeverria, G.; Rigotti, A.; Leighton, F. Wine grape pomace flour improves blood pressure, fasting glucose and protein damage in humans: A randomized controlled trial. Biol. Res. 2015, 48, 49. [Google Scholar] [CrossRef] [PubMed]
- Banini, A.E.; Boyd, L.C.; Allen, J.C.; Allen, H.G.; Sauls, D.L. Muscadine grape products intake, diet and blood constituents of non-diabetic and type 2 diabetic subjects. Nutrition 2006, 22, 1137–1145. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Ros, E.; Valderas-Martinez, P.; Casas, R.; Arranz, S.; Guillen, M.; Lamuela-Raventos, R.M.; Llorach, R.; Andres-Lacueva, C.; et al. Effects of red wine polyphenols and alcohol on glucose metabolism and the lipid profile: A randomized clinical trial. Clin. Nutr. 2013, 32, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, J.K.; Thomas, S.; Nanjan, M.J. Resveratrol supplementation improves glycemic control in type 2 diabetes mellitus. Nutr. Res. 2012, 32, 537–541. [Google Scholar] [CrossRef] [PubMed]
| Study Ref | Subject Description | Trial Type | n | Product | Intake (per Day) | Duration | Condition at Testing | Significant Results |
|---|---|---|---|---|---|---|---|---|
| Ras et al. 2013 [26] | Healthy | Parallel R PC | 70 | Grape seed extract | 300 mg | 8 wk | BP (days 5, 7, 61 and 63) Urine 24 h (days 7 and 63) Blood samples (0 and 2 h after breakfast) | ↓ SBP (p = 0.01) ↓ DBP (p = 0.01) |
| Sivaprakasapillai et al. 2009 [27] | Metabolic syndrome | Crossover R PC | 27 (11 H, 16 F) | Grape seed extract | 150 mg 300 mg | 4 wk | Fasted | ↓ SBP (p = 0.05) ↓ DBP (p = 0.05) No change in LDL and HDL choresterol ↓ Ox-LDL levels |
| Clifton 2004 [28] | Above average vascular risk 58 yr | Crossover R PC DB | 36 (24 M, 12 F) | Grape seed extract | 2 g | 4 wk | Fasted | ↓ SBP (p = 0.05) ↑ FMD (p < 0.05) |
| Sano et al. 2007 [29] | Healthy ≥30 yr and <70 yr | Crossover R PC DB | 61 (29 M, 32 F) | Grape seed extract | 200 mg 400 mg | 12 wk | Fasted at 0, 6 and 12 wk | No significant change in BP, LDL-C, HPL-C ↓ LDL and ox-LDL (12%–14%, p < 0.05) |
| van Mierlo et al. 2010 [30] | Healthy 31.4 ± 9.0 yr | Crossover R PC DB | 35 (35 M, 0 F) | Grape seed extract | 800 mg | 3 periods of 2 weeks | Post-prandial | No change in BP or FMD |
| Ward et al. 2005 [31] | Hypertensive 61.3 ± 6.3 yr | Parallel PC DB | 69 | Grape seed extract | 1000 mg | 6 wk | Fasted | No change in BP or FMD No change in ox-LDL No change in F2-isoprostanes |
| Vaisman et al. 2015 [36] | Pre- and mild-hypertension ≥35 yr and <70 yr | Parallel R PC DB | 50 (35 M, 15 F) | Red grape cell powder | 200 mg 400 mg | 12 wk | Fasted | ↓ DBP (p = 0.032) in 200 mg group |
| Biesinger et al. 2016 [37] | Metabolic syndrome | Crossover R PC DB | 18 | Combination of extract from seed and skin | 330 mg | 28 days | Fasted | ↓ DBP (p = 0.024) No change in SBP |
| Study Ref | Subject Description | Trial Type | n | Product | Intake (per Day) | Duration | Condition at Testing | Significant Results |
|---|---|---|---|---|---|---|---|---|
| Castilla et al. 2006 [42] | Healthy and hemodialysis patients | Crossover R PC | 38 (19 H, 19 F) | Red grape juice | 100 mL | 14 days | Fasted at time 0, 7 and 14 | ↓ TC, ↓ LDL-C, ↓ apo B-100 (p < 0.001); ↑ HPL-C and ↑ apo A-1 (p < 0.001) ↓ ox-LDL (35%) |
| Castilla et al. 2008 [43] | Hemodialysis patients | Crossover R PC | 32 | Red frape juice | 100 mL | 14 days | Fasted at time 0, 7 and 14 | ↓ TC, ↓ LDL-C, ↓ apo B-100 (p < 0.001); ↑ HPL-C and ↑ apo A-1 (p < 0.01) ↓ ox-LDL (65%) and NADPH (p < 0.01) |
| Khadem-Ansari et al. 2010 [44] | Healthy and non-smokers | Single arm intervention | 26 (26 M, 0 F) | Red grape juice | 300 mL | 1 month | Fasted 12 h before study entry and at the end of study | ↑ HLD-C (p < 0.0001) and ↑ apo B (p < 0.002) |
| Albers et al. 2004 [45] | CAD patients | Crossover R PC DB | 40 | Purple grape juice | 7 mL/kg | 14 days | Fasted | ↑ TG (p < 0.05) and no change in TC, HDL-C or LDL-C ↓ platelet (p = 0.02) |
| Stein et al. 1999 [45] | CAD 62.5 ± 12.7 yr | Single arm intervention | 15 (12 M, 3 F) | Concord grape juice | 7.7 ± 1.2 mL/kg | 14 days | Fasted (day 0); day 14, 4 mL/kg treatment consumed prior to test; time not given | ↑ TG (p < 0.001) and no change in TC, HDL-C or LDL-C ↑ LDL (35%) |
| O’Byrne et al. 2002 [47] | Healthy | Parallel R | 36 | Concord grape juice | 10 mL/kg | 2 wk | Fasted | ↑ TG (p < 0.05) and no change in TC, HDL-C or LDL-C ↑ LDL (10%) and ox-LDL (9%) No change in F2-isoprostanes |
| Evans et al. 2014 [48] | Pre-hypertensive/pre-diabetic | Crossover R PC DB | 24 | Grape extract | 350 mg | 6 wk | Fasted | ↑ HDL-C (p = 0.001), ↓ TC (p = 0.037) and no change in LDL-C or TG. |
| Razavi et al. 2013 [49] | Mild hyperlipidemic | Crossover R PC DB | 52 | Grape seed extract | 200 mg | 8 wk | Fasted | ↓ TC (p = 0.015), ↓ LDL-C (p = 0.014) ↓ ox-LDL (P = 0.008)) |
| Vinson et al. 2001 [50] | Healthy and hypercholesterolemic | Parallel R | 17 | Grape seed extract | 600 mg | 3 wk | Fasted | ↓ TC and ↓ LDL-C, p < 0.01; ↓ HDL-C, p < 0.05 |
| Study Ref | Subject Description | Trial Type | n | Product | Intake (per Day) | Duration | Condition at Testing | Significant Results |
|---|---|---|---|---|---|---|---|---|
| Toaldo et al. 2015 [57] | Healthy | Crossover R C | 24 (5 M, 19 F) | Grape juice | 400 mL | 2 wk | Fasted | ↓ TBARS (p < 0.05) |
| Vigna et al. 2003 [58] | Heavy smokers | Crossover R PC DB | 24 (24 M, 0 F) | Grape procyanidin extract | 75 mg | 4 wk | Fasted | No change in TC, TG, HDL-C and LDL-C ↓ TBARS (p < 0.01) and ↑ ox-LDL (p < 0.05) |
| Freedman et al. 2001 [60] | Healthy 30.6 ± 1.8 yr | Single arm intervention | 20 (12 M, 8 F) | Purple grape juice | 7 mL/kg | 14 days | Fasted | ↑ Platelet-NO (P<0.007) and ↓ Platelet-O2- (p < 0.05) |
| Caccetta et al. 2000 [62] | Smokers | Crossover R | 18 (18 M, 0 F) | Red wine, white wine and dealcoholized red wine | 375 mL | 2 wk | Fasted before and after each bevarage | ↓ F2-isoprostanes (p < 0.05) with dealcoholized red wine |
| Zern et al. 2005 [63] | Pre- and post-menopausal women | Crossover R PC | 44 (24 F pre-, 20 F post-) | Grape powder | 36 g | 4 wk | Fasted blood and non fasted urine | ↓ TG (p < 0.01); ↓ LDL-C, apo-b and apo-e (p < 0.05, ↓ F2-isoprastones (p < 0.05). No change in ox-LDL |
| Hokayem et al. 2013 [64] | Healthy overweight/obese T2D | Crossover R PC DB | 38 (18 M, 20 F) | Grape polyphenols | 2 g | 8 wk | Fasted | ↓ F2-isopreastones and TBARS (p < 0.05) ↓ hepatic insulin (p < 0.05) and glucose infusion rate (p < 0.05) |
| Park et al. 2009 [65] | Hypertensive men | Crossover R PC DB | 40 | Grape juice | 5.5 mL/kg | 8 wk | Fasted | ↓ DNA damage (p < 0.01) and ↓ DBP and SBP (p < 0.05) |
| Corredor et al. 2016 [66] | Chronic kidney disease | Crossover R C | 39 (21 M, 15 F) | Unfermented grape juice | 100 mL | 6 months | Blood samples before HD | ↓ ox-DNA damage and LDL and C |
| Study Ref | Subject Description | Trial Type | n | Product | Intake (per Day) | Duration | Condition at Testing | Significant Results |
|---|---|---|---|---|---|---|---|---|
| Yamakoshi et al. 2001 [116] | Healthy and elderly inpatients | Crossover R | 33 | Grape seed extract | 0.5 g | 2 wk | Freshly voided fecal by direct defecation | ↑ Bifidobacterium |
| Queipo-Ortuno et al. 2012 [117] | Healthy | Crossover R C | 10 (10 M, 0 F) | Red wine and dealcoholized red wine | 272 mL | 20 days | Fecal samples before and after treatment. Fasted blood and 24-h urine | ↑ Bifidobacterium |
| Kar et al. 2009 [118] | T2D subjects 61.8 ± 6.36 yr | Crossover R PC DB | 32 (16 M, 16 F) | Grape seed extract | 600 mg | 4 wk | Fasted | ↓ Fructosamine (p = 0.0004) ↓ TC (p = 0.05) TG and HDL-C stables |
| Urquiaga et al. 2015 [119] | Metabolic syndrome | Crossover R C | 38 | Red wine grape pomace | 20 g | 16 wk | Fasted | ↓ Postprandial insulin (p < 0.05) ↓ Protein damage |
| Banini et al. 2006 [120] | T2D subjects | Crossover R C | 29 | Grape juice, wine and dealcoholized wine | 150 mL | 28 days | Fasted | ↓ blood glucose, insulin and glycated hemoglobin |
| Chiva-Blanch et al. 2013 [121] | High CVD risk 60 ± 8 yr | Crossover R C | 67 (67 M, 0 F) | Red wine and dealcoholized red wine | 272 mL | 4 wk | Fasted and 24-h urine | ↓ insulin value ↓ LDL-C and ↑HLD-C ↓ Apo-B |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasines-Perea, Z.; Teissedre, P.-L. Grape Polyphenols’ Effects in Human Cardiovascular Diseases and Diabetes. Molecules 2017, 22, 68. https://doi.org/10.3390/molecules22010068
Rasines-Perea Z, Teissedre P-L. Grape Polyphenols’ Effects in Human Cardiovascular Diseases and Diabetes. Molecules. 2017; 22(1):68. https://doi.org/10.3390/molecules22010068
Chicago/Turabian StyleRasines-Perea, Zuriñe, and Pierre-Louis Teissedre. 2017. "Grape Polyphenols’ Effects in Human Cardiovascular Diseases and Diabetes" Molecules 22, no. 1: 68. https://doi.org/10.3390/molecules22010068
APA StyleRasines-Perea, Z., & Teissedre, P.-L. (2017). Grape Polyphenols’ Effects in Human Cardiovascular Diseases and Diabetes. Molecules, 22(1), 68. https://doi.org/10.3390/molecules22010068





