Near-Infrared Emitting PbS Quantum Dots for in Vivo Fluorescence Imaging of the Thrombotic State in Septic Mouse Brain
Abstract
:1. Introduction
2. Results and Discussion
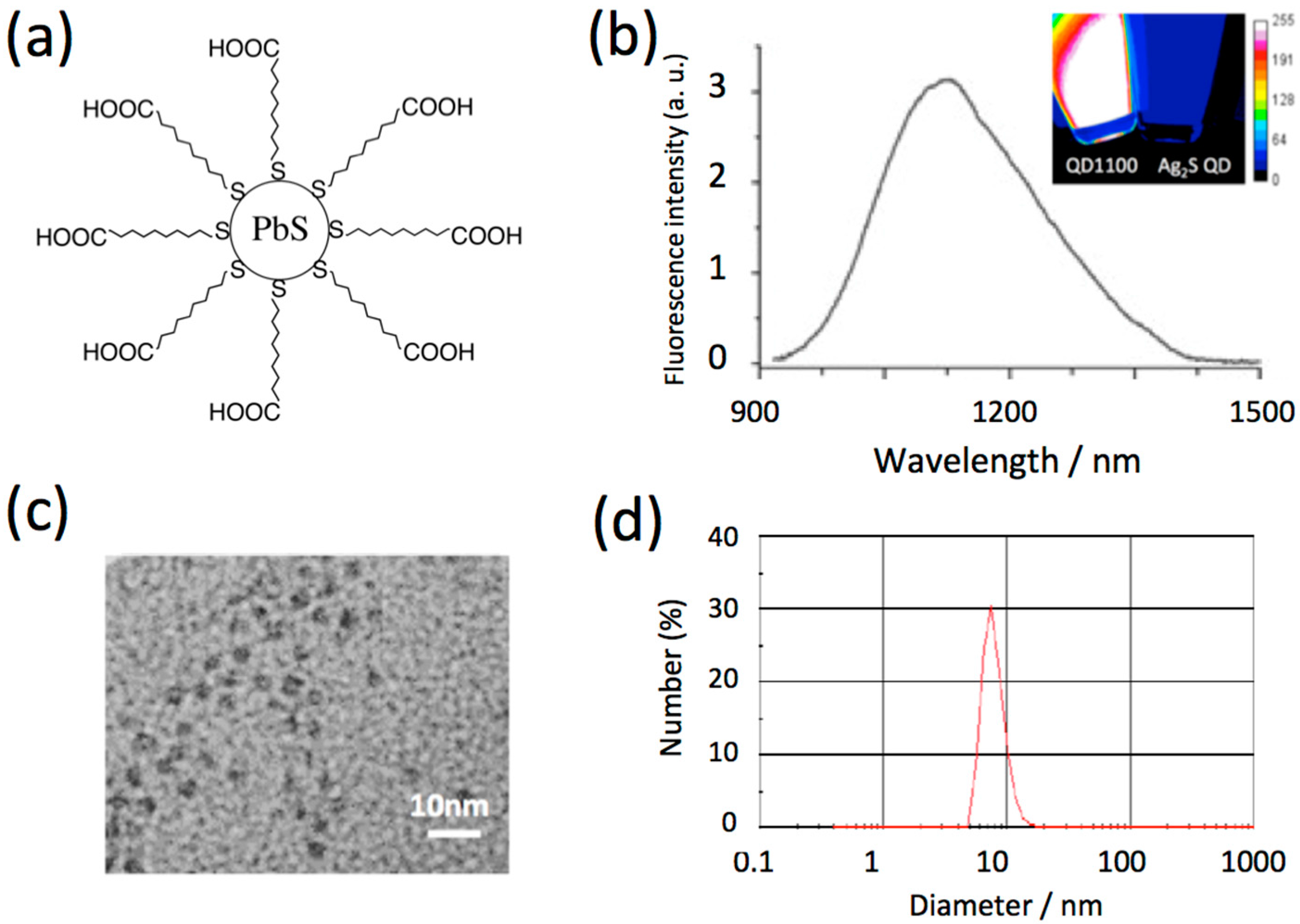
2.1. Characteristics of NIR Fluorescent PbS QDs
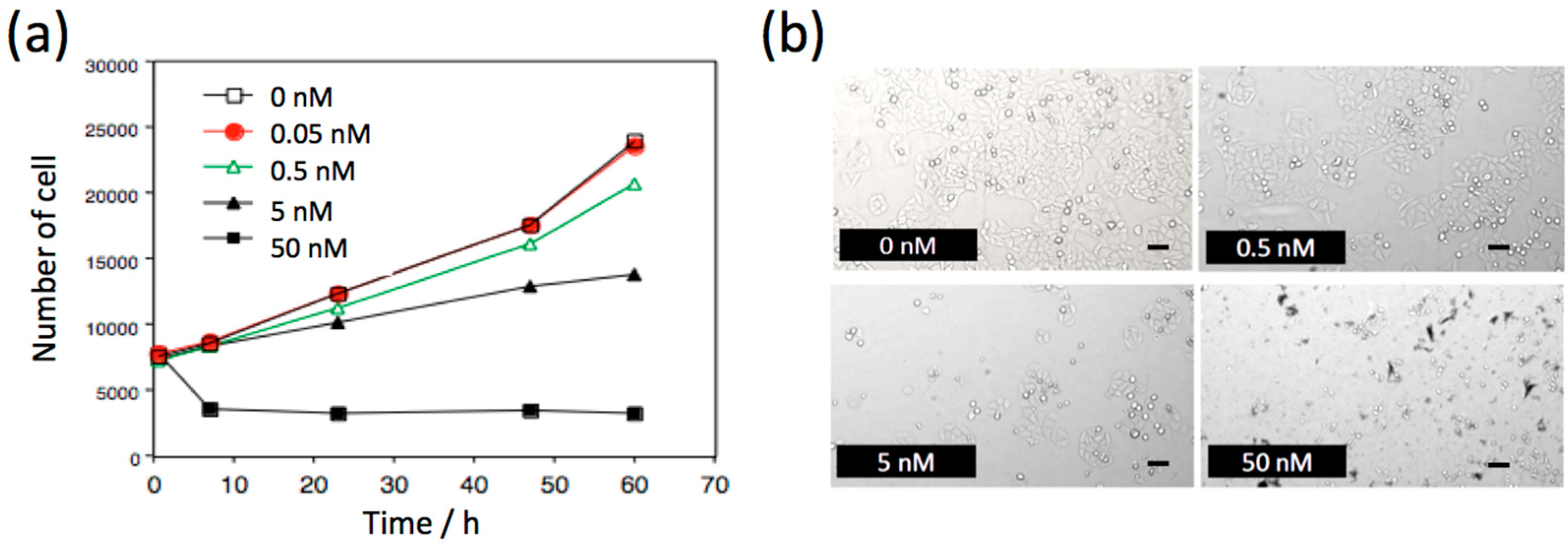
2.2. Biocompatibility of NIR Fluorescent PbS QDs
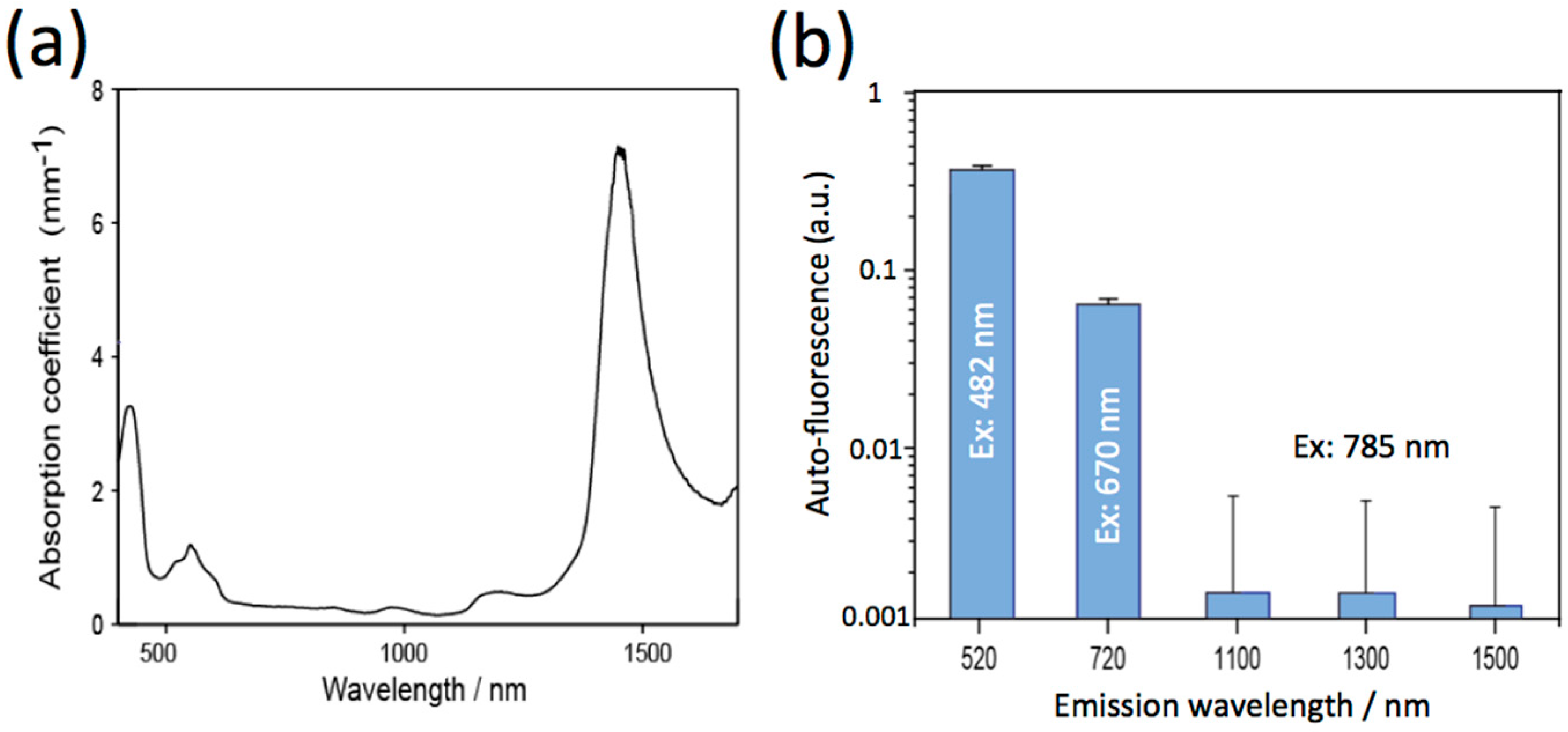
2.3. Optical Properties of Mouse Brain in the Visible and NIR Region
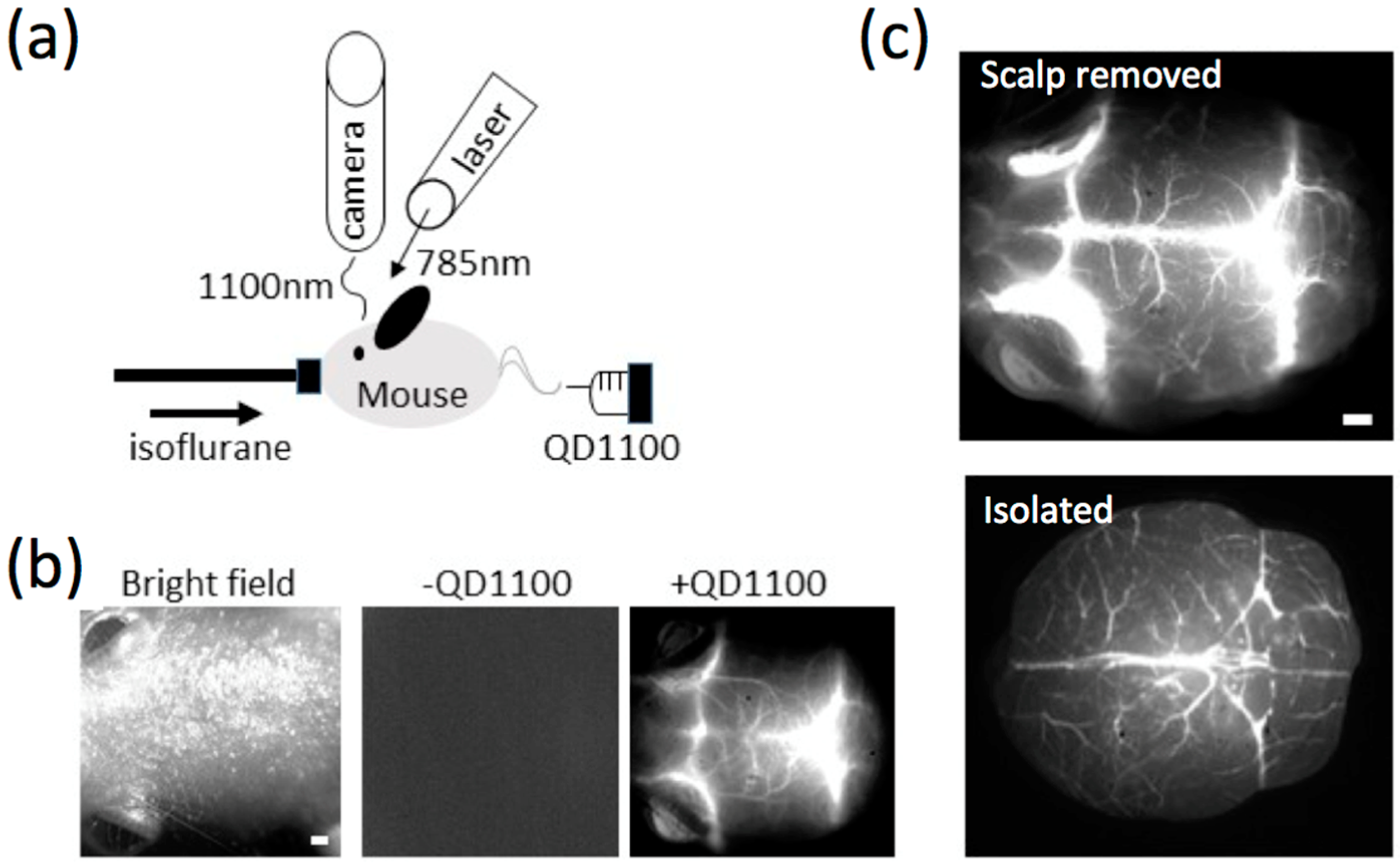
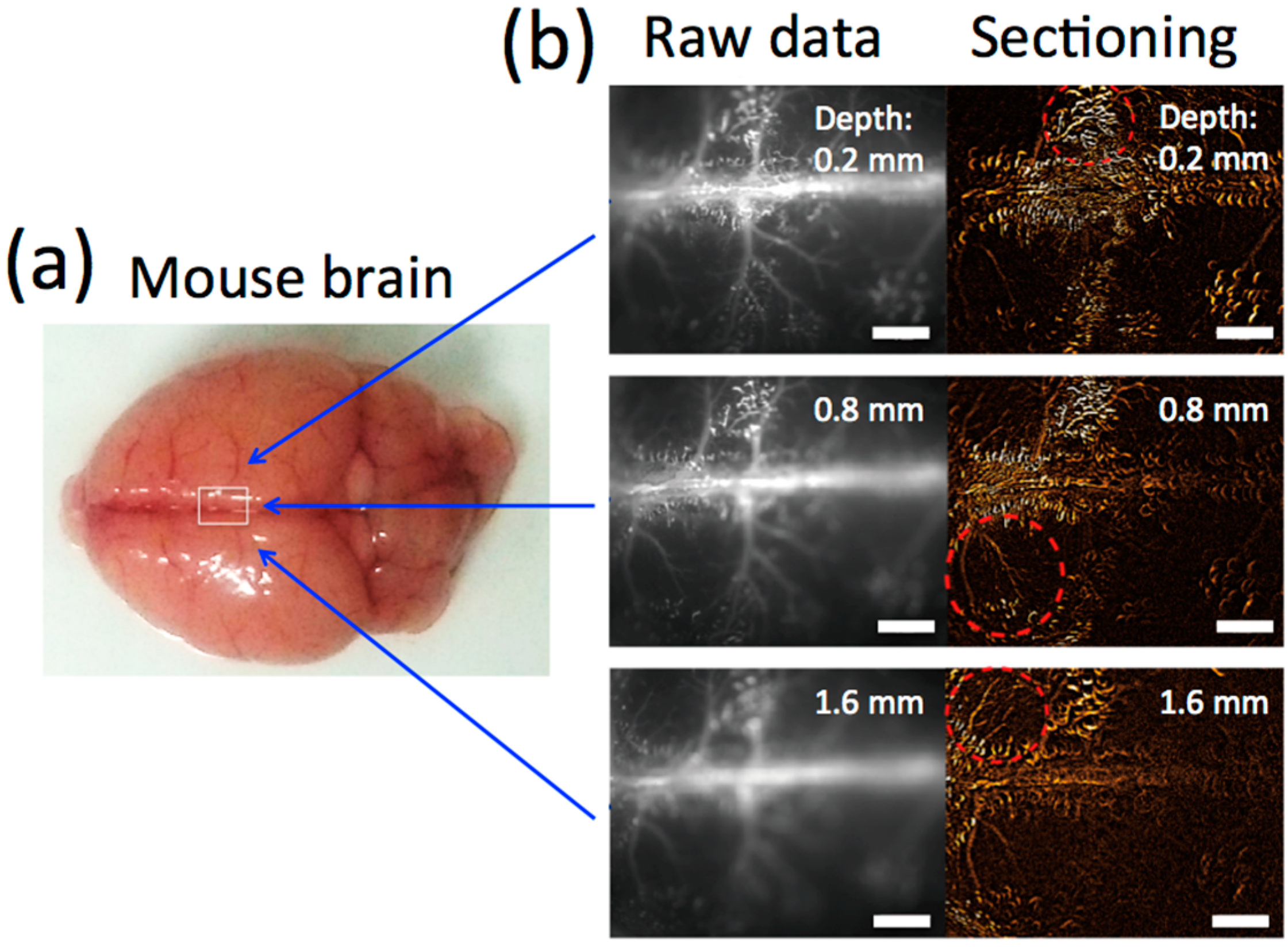
2.4. NIR Fluorescence Imaging of Cerebral Blood Vessels in Mice
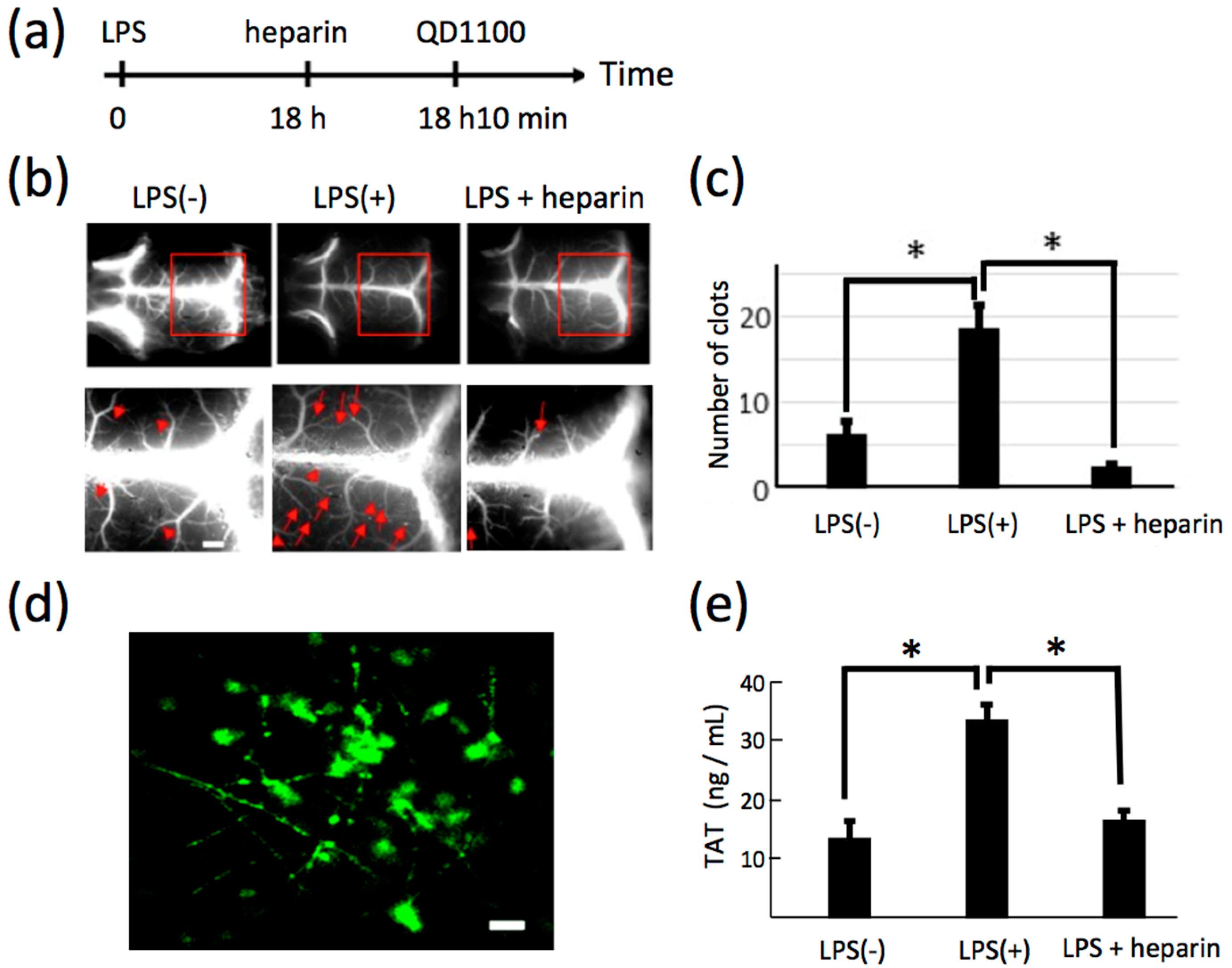
2.5. NIR Fluorescence Imaging of Lipopolysaccharide-Induced Thrombosis in Cerebral Blood Vessels in Living Mice
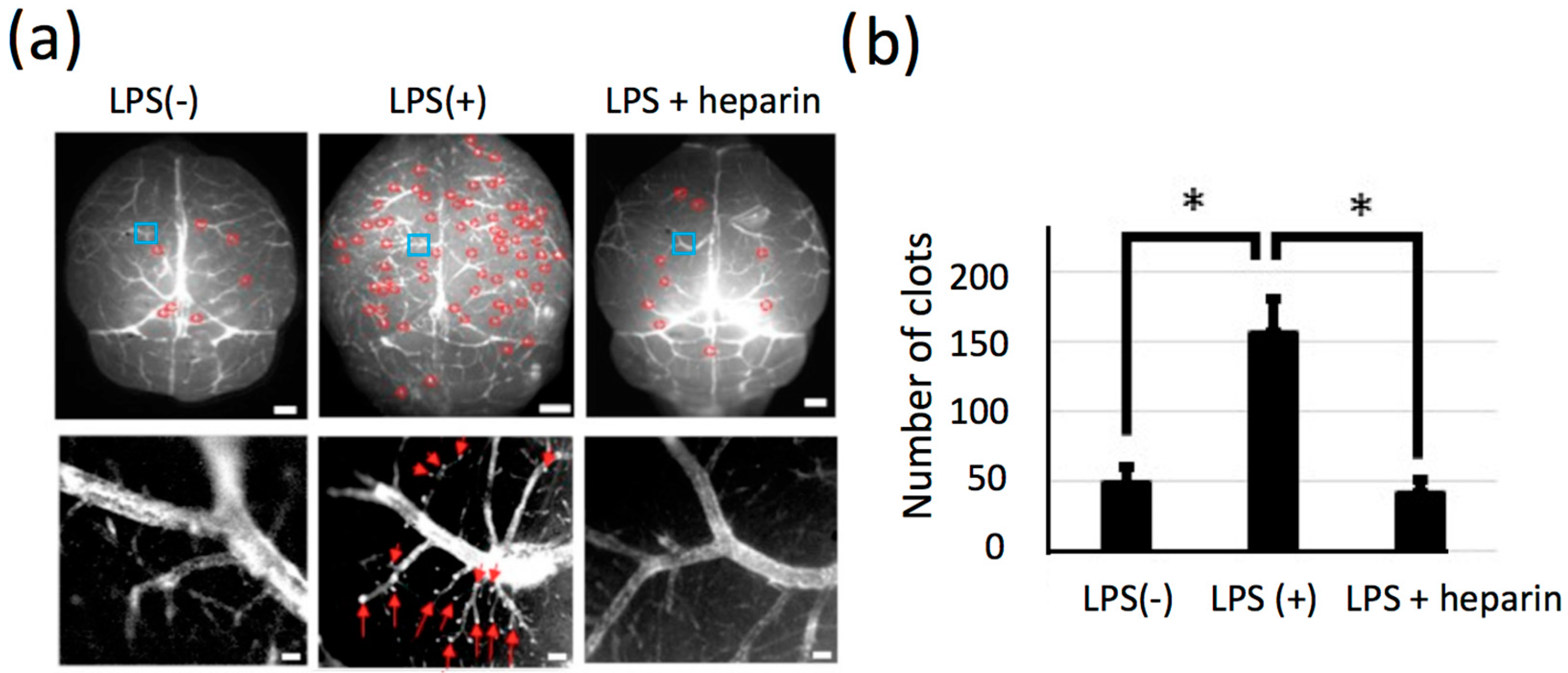
2.6. Significant Change in the Number of LPS-Induced Thrombi in Isolated Brains
3. Experimental Section
3.1. Chemicals
3.2. Synthesis and Characterization of NIR Fluorescent PbS QDs (QD1100)
3.3. Biocompatibility of QD1100
3.4. Mice
3.5. Optical Properties of Brain Tissue
3.6. Microscopy System for Cerebral Vessel Imaging
3.7. Immunofluorescence Staining of Brain Tissues
3.8. Enzyme-Linked Immunosorbent Assays (ELISA)
3.9. Statistical Analyses
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wngner, K.D.; Hiderbrandt, N. Quantum dots: Bright and versatle in vitro and in vivo fluorescence imaging biosensors. Chem. Soc. Rev. 2015, 44, 4792–4834. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, D.K.; Tanaka, S.; Inouye, Y.; Yoshizawa, K.; Watanabe, T.M.; Jin, T. Synthesis and characterization of Anti-HER2 antibody conjugated CdSe/CdZnS quantum dots for fluorescence imaging of breast cancer cells. Sensors (Basel) 2009, 9, 9332–9364. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Tiwari, D.K.; Tanaka, S.; Inouye, Y.; Yoshizawa, K.; Watanabe, T.M. Antibody-protein A conjugated quantum dots for multiplexed imaging of surface receptors in living cells. Mol. Biosyst. 2010, 6, 2325–2331. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Nakane, Y.; Mori, Y.; Hasegawa, M.; Yoshioka, Y.; Watanabe, T.M.; Gonda, K.; Ohuchi, N.; Jin, T. Multilayered, core/shell nanoprobes based on magnetic ferric oxide particles and quantum dots for multimodality imaging of breast cancer tumors. Biomaterials 2012, 33, 8486–8494. [Google Scholar] [CrossRef] [PubMed]
- Komatsuzaki, A.; Ohyanagi, T.; Tsukasaki, Y.; Miyanaga, Y.; Ueda, M.; Jin, T. Compact halo-ligand-conjugated quantum dots for multicolored single-molecule imaging of overcrowding GPCR proteins on cell membranes. Small 2014, 11, 1396–1401. [Google Scholar] [CrossRef] [PubMed]
- Tsukasaki, Y.; Morimatsu, M.; Nishimura, G.; Sakata, T.; Yasuda, H.; Komatsuzaki, A.; Watanabe, T.M.; Jin, T. Synthesis and optical properties of emission-tunable PbS/CdS core-shell quantum dots for in vivo fluorescence imaging in the second near-infrared window. RSC Adv. 2014, 4, 41164–41171. [Google Scholar] [CrossRef]
- Nakane, Y.; Tsukasaki, Y.; Sakata, T.; Yasuda, H.; Jin, T. Aqueous synthesis of glutathione-coated PbS quantum dots with tunable emission for non-invasive fluorescence imaging in the second near-infrared biological window (1000–1400 nm). Chem. Commun. 2013, 49, 7584–7586. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Tsukasaki, Y.; Komatsuzaki, A.; Sakata, T.; Yasuda, H.; Jin, T. Recombinant protein (EGFP-Protein G)-coated PbS quantum dots for in vitro and in vivo dual fluorescence (visible and second-NIR) imaging of breast tumors. Nanoscale 2015, 7, 5115–5119. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Imamura, Y. Applications of highly bright Pbs quantum dots to non-invasive near-infrared fluorescence imaging in the second optical window. ECS J. Solid State Sci. Technol. 2016, 5, R3138–R3145. [Google Scholar] [CrossRef]
- Welsher, K.; Liu, Z.; Sherlock, S.P.; Robinson, J.T.; Chen, Z.; Daranciang, D.; Dai, H. A route to brightly fluorescent carbon nanotubes for near-infrared imaging in mice. Nat. Nano 2009, 4, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Welsher, K.; Sherlock, S.P.; Dai, H. Deep-tissue anatomical imaging of mice using carbon nanotube fluorophores in the second near-infrared window. Proc. Natl. Acad. Sci. USA 2011, 108, 8943–8948. [Google Scholar] [CrossRef] [PubMed]
- Hong, G.; Lee, J.C.; Robinson, J.T.; Raaz, U.; Xie, L.; Huang, N.F.; Cooke, J.P.; Dai, H. Multifunctional in vivo vascular imaging using near-infrared II fluorescence. Nat. Med. 2012, 18, 1841–1846. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.T.; Hong, G.; Liang, Y.; Zhang, B.; Yaghi, O.K.; Dai, H. In vivo fluorescence imaging in the second near-infrared window with long circulating carbon nanotubes capable of ultrahigh tumor uptake. J. Am. Chem. Soc. 2014, 134, 10664–10669. [Google Scholar] [CrossRef] [PubMed]
- Hong, G.; Diano, S.; Chang, J.; Antaris, A.L.; Chen, C.; Zhang, B.; Zhao, S.; Atochin, D.N.; Huang, P.L.; Anderson, K.L.; et al. Through-skull fluorescence imaging of the brain in a new near-infrared window. Nat. Photonics 2014, 8, 723–730. [Google Scholar] [CrossRef]
- Naczynski, D.J.; Tan, M.C.; Zevon, M.; Wall, B.; Kohl, J.; Kulesa, A.; Chen, S.; Roth, C.M.; Riman, R.E.; Moghe, P.V. Rare-earth-doped biological composites as in vivo shortwave infrared reporters. Nat. Commun. 2013, 4, 2199. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Sordillo, L.A.; Rodoríguez-Contrera, A.; Alfano, R. Transmission in near-infrared optical windows for deep brain imaging. J. Biophotonics 2016, 9, 38–43. [Google Scholar] [CrossRef]
- Imamura, Y.; Wang, H.; Matsumoto, N.; Ogura, H.; Shimazu, T.; Jin, T.; Seiyama, A. Neurochemistry in the Pathophysiology of Septic Encephalopathy. In Neuroscience—Dealing with Frontiers; In-Tech Open Access Publisher: Rijeka, Croatias, 2012. [Google Scholar]
- Reis, C.; Wang, Y.; Akyol, O.; Ho, W.M.; Applegate, I.R.; Stier, G.; Martin, R.; Zhang, J.H. What’s new in traumatic brain injury: Update on tracking, monitoring and treatment. Int. J. Mol. Sci. 2015, 16, 11903–11965. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.A.; Ivanova, M.I.; Sawaya, M.R.; Cascio, D.; Reyes, F.; Shi, D.; Sangwan, S.; Guenther, E.L.; Johnson, L.M.; Zhang, M.; et al. Structure of the toxic core of α-synuclein from invisible crystals. Nature 2015, 525, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Bennewitz, M.F.; Saltzman, W.M. Nanotechnology for the delivery of drugs to the brain for epilepsy. Neurotherapeutics 2009, 6, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Shcharbina, N.; Shcharbin, D.; Bryszewska, M. Nanomaterials in stroke treatment: Perspectives. Stroke 2013, 44, 2351–2355. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Raoof, M.; Chen, Y.; Sumi, Y.; Sursal, T.; Junger, W.; Brohi, K.; Itagaki, K.; Hauser, C.J. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 2010, 464, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Gofton, T.E.; Young, G.B. Sepsis-associated encephalopathy. Nat. Rev. Neurol. 2012, 8, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Imamura, Y.; Hori, S.; Matsumoto, N.; Yamakawa, K.; Nakagawa, J.; Shimazaki, J.; Wang, H.; Itou, J.; Murakami, Y.; Mitani, S.; et al. Septic Encephalopathy; Nova Science Publishers: New York, NY, USA, 2013; Volume 8, p. 13. [Google Scholar]
- Stubbs, D.J.; Yamamoto, A.K.; Menon, D.K. Imaging in sepsis-associated encephalopathy[mdash]insights and opportunities. Nat. Rev. Neurol. 2013, 9, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Ziaja, M. Septic encephalopathy. Curr. Neurol. Neurosci. Rep. 2013, 13, 383. [Google Scholar] [CrossRef] [PubMed]
- Ten Cate, H.; Schoenmakers, S.H.; Franco, R.; Timmerman, J.J.; Groot, A.P.; Spek, C.A.; Reitsma, P.H. Microvascular coagulopathy and disseminated intravascular coagulation. Crit. Care Med. 2001, 29, S95–S97. [Google Scholar] [CrossRef] [PubMed]
- Levi, M.; Keller, T.T.; van Gorp, E.; ten Cate, H. Infection and inflammation and the coagulation system. Cardiovasc. Res. 2003, 60, 26–39. [Google Scholar] [CrossRef]
- Yamakawa, K.; Ogura, H.; Fujimi, S.; Morikawa, M.; Ogawa, Y.; Mohri, T.; Nakamori, Y.; Inoue, Y.; Kuwagata, Y.; Tanaka, H.; et al. Recombinant human soluble thrombomodulin in sepsis-induced disseminated intravascular coagulation: A multicenter propensity score analysis. Intensive Care Med. 2013, 39, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Semeraro, N.; Ammollo, C.T.; Semeraro, F.; Colucci, M. Sepsis-associated disseminated intravascular coagulation and thromboembolic disease. Mediterr. J. Hematol. Infect. Dis. 2010, 2, e2010024. [Google Scholar] [CrossRef] [PubMed]
- Semeraro, N.; Ammollo, C.T.; Semeraro, F.; Colucci, M. Sepsis, thrombosis and organ dysfunction. Thromb. Res. 2012, 129, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Abildgaard, U. Heparin/low molecular weight heparin and tissue factor pathway inhibitor. Haemostasis 1993, 23, 103–106. [Google Scholar] [CrossRef] [PubMed]
- Lupu, C.; Poulsen, E.; Roquefeuil, S.; Westmuckett, A.D.; Kakkar, V.V.; Lupu, F. Cellular effects of heparin on the production and release of tissue factor pathway inhibitor in human endothelial cells in culture. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 2251–2262. [Google Scholar] [CrossRef] [PubMed]
- Slofstra, S.H.; van’t Veer, C.; Buurman, W.A.; Reitsma, P.H.; ten Cate, H.; Spek, C.A. Low molecular weight heparin attenuates multiple organ failure in a murine model of disseminated intravascular coagulation. Crit. Care Med. 2005, 33, 1365–1370. [Google Scholar] [CrossRef] [PubMed]
- Ding, R.; Zhao, D.; Guo, R.; Zhang, Z.; Ma, X. Treatment with unfractionated heparin attenuates coagulation and inflammation in endotoxemic mice. Thromb. Res. 2011, 128, e160–e165. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Tong, J.; Tan, Y.; Zhu, S.; Yang, J.; Ji, M. Low molecular weight heparin prevents lipopolysaccharide induced-hippocampus-dependent cognitive impairments in mice. Int. J. Clin. Exp. Pathol. 2015, 8, 8881–8891. [Google Scholar] [PubMed]
- Wada, H.; Wakita, Y.; Nakase, T.; Shimura, M.; Hiyoyama, K.; Nagaya, S.; Deguchi, H.; Mori, Y.; Kaneko, T.; Deguchi, K.; et al. Increased plasma-soluble fibrin monomer levels in patients with disseminated intravascular coagulation. Am. J. Hematol. 1996, 51, 255–260. [Google Scholar] [CrossRef]
- Enjeti, A.K.; Ariyarajah, A.; D’Crus, A.; Seldon, M.; Lincz, L.F. Correlative analysis of nanoparticle tracking, flow cytometric and functional measurements for circulating microvesicles in normal subjects. Thromb. Res. 2016, 145, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Averett, R.D. Experimental mechanics of magnetic microparticle-induced strain on fibrin clots. J. Biomed. Mater. Res. A 2014, 102, 4365–4370. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Imamura, Y.; Matsumoto, N.; Yoshikawa, N.; Nakagawa, J.; Yamakawa, K.; Yamada, T.; Murakami, Y.; Mitani, S.; Jin, T.; et al. Matrix metalloproteinase-9 triggers the gap junction impairment and somatosensory neuronal dysfunction in septic encephalopathy. Biochem. Pharmacol. 2013, 2. [Google Scholar] [CrossRef]
- Saitoh, Y.; Terada, N.; Ohno, N.; Hamano, A.; Okumura, N.; Jin, T.; Saiki, I.; Ohno, S. Imaging of thrombosis and microcirculation in mouse lungs of initial melanoma metastasis with in vivo cryotechnique. Microvasc. Res. 2014, 91, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Imamura, Y.; Wang, H.; Matsumoto, N.; Muroya, T.; Shimazaki, J.; Ogura, H.; Shimazu, T. Interleukin-1beta causes long-term potentiation deficiency in a mouse model of septic encephalopathy. Neuroscience 2011, 187, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Imamura, Y.; Yoshikawa, N.; Murkami, Y.; Mitani, S.; Matsumoto, N.; Matsumoto, H.; Yamada, T.; Yamakawa, K.; Nakagawa, J.; Ogura, H.; et al. S Effect of histone acetylation on N-methyl-d-aspartate 2B receptor subunits and interleukin-1 receptors in association with nociception-related somatosensory cortex dysfunction in a mouse model of sepsis. Shock 2016, 45, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Not available.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imamura, Y.; Yamada, S.; Tsuboi, S.; Nakane, Y.; Tsukasaki, Y.; Komatsuzaki, A.; Jin, T. Near-Infrared Emitting PbS Quantum Dots for in Vivo Fluorescence Imaging of the Thrombotic State in Septic Mouse Brain. Molecules 2016, 21, 1080. https://doi.org/10.3390/molecules21081080
Imamura Y, Yamada S, Tsuboi S, Nakane Y, Tsukasaki Y, Komatsuzaki A, Jin T. Near-Infrared Emitting PbS Quantum Dots for in Vivo Fluorescence Imaging of the Thrombotic State in Septic Mouse Brain. Molecules. 2016; 21(8):1080. https://doi.org/10.3390/molecules21081080
Chicago/Turabian StyleImamura, Yukio, Sayumi Yamada, Setsuko Tsuboi, Yuko Nakane, Yoshikazu Tsukasaki, Akihito Komatsuzaki, and Takashi Jin. 2016. "Near-Infrared Emitting PbS Quantum Dots for in Vivo Fluorescence Imaging of the Thrombotic State in Septic Mouse Brain" Molecules 21, no. 8: 1080. https://doi.org/10.3390/molecules21081080
APA StyleImamura, Y., Yamada, S., Tsuboi, S., Nakane, Y., Tsukasaki, Y., Komatsuzaki, A., & Jin, T. (2016). Near-Infrared Emitting PbS Quantum Dots for in Vivo Fluorescence Imaging of the Thrombotic State in Septic Mouse Brain. Molecules, 21(8), 1080. https://doi.org/10.3390/molecules21081080






