Preparative Separation of Phenolic Compounds from Chimonanthus praecox Flowers by High-Speed Counter-Current Chromatography Using a Stepwise Elution Mode
Abstract
:1. Introduction
2. Results and Discussion
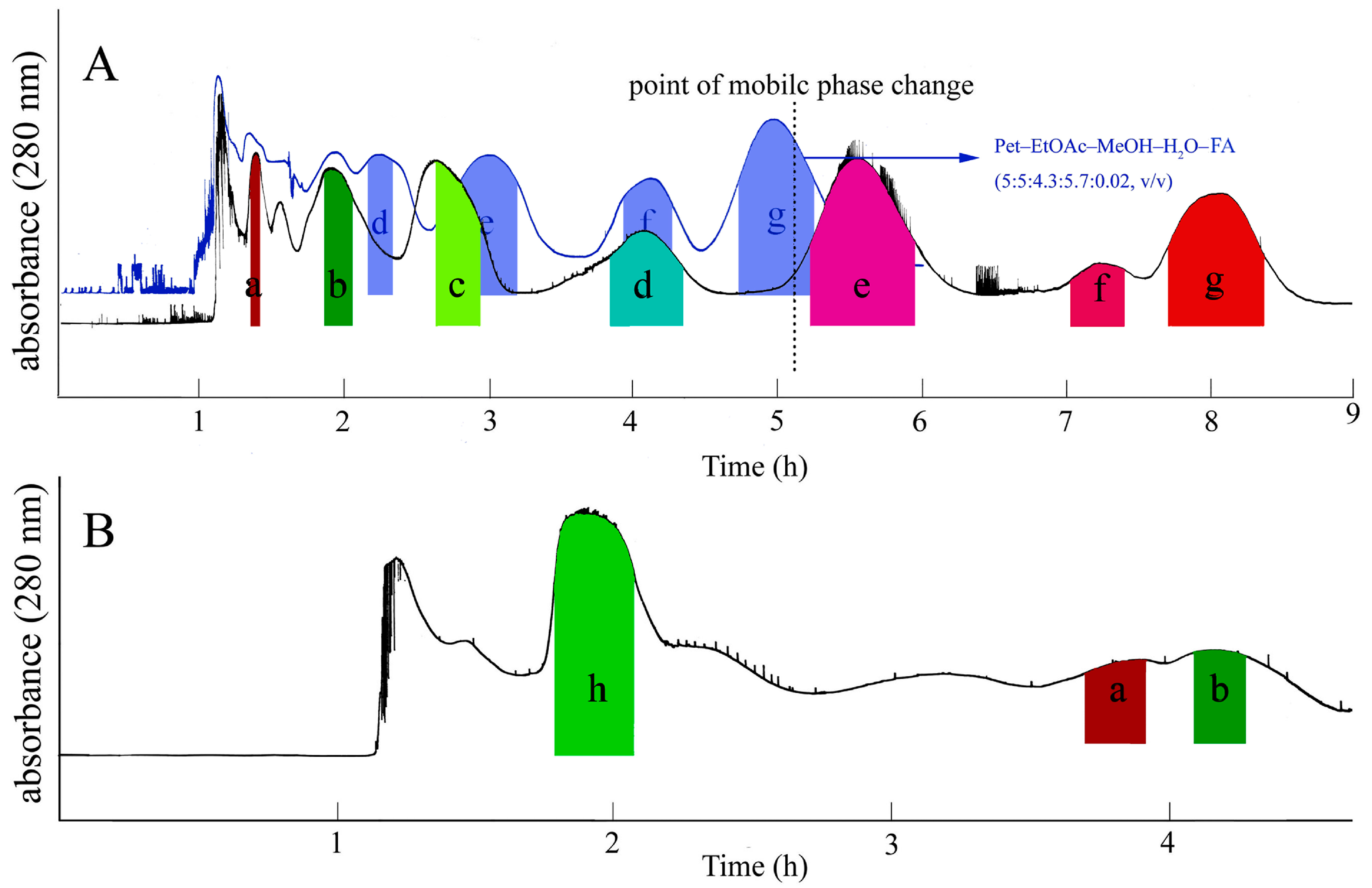
2.1. Selection of Two-Phase Solvent System
2.2. HSCCC Separation
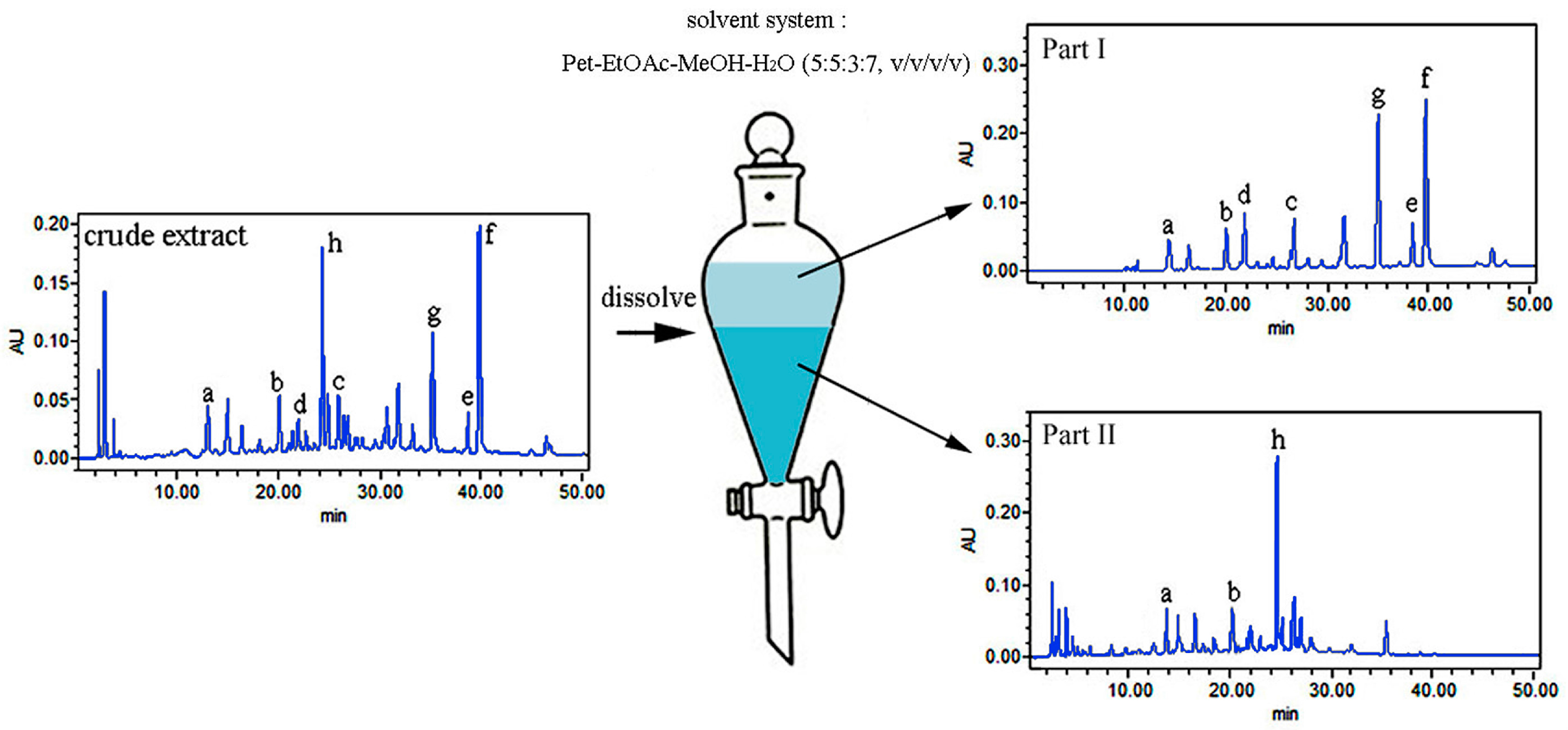
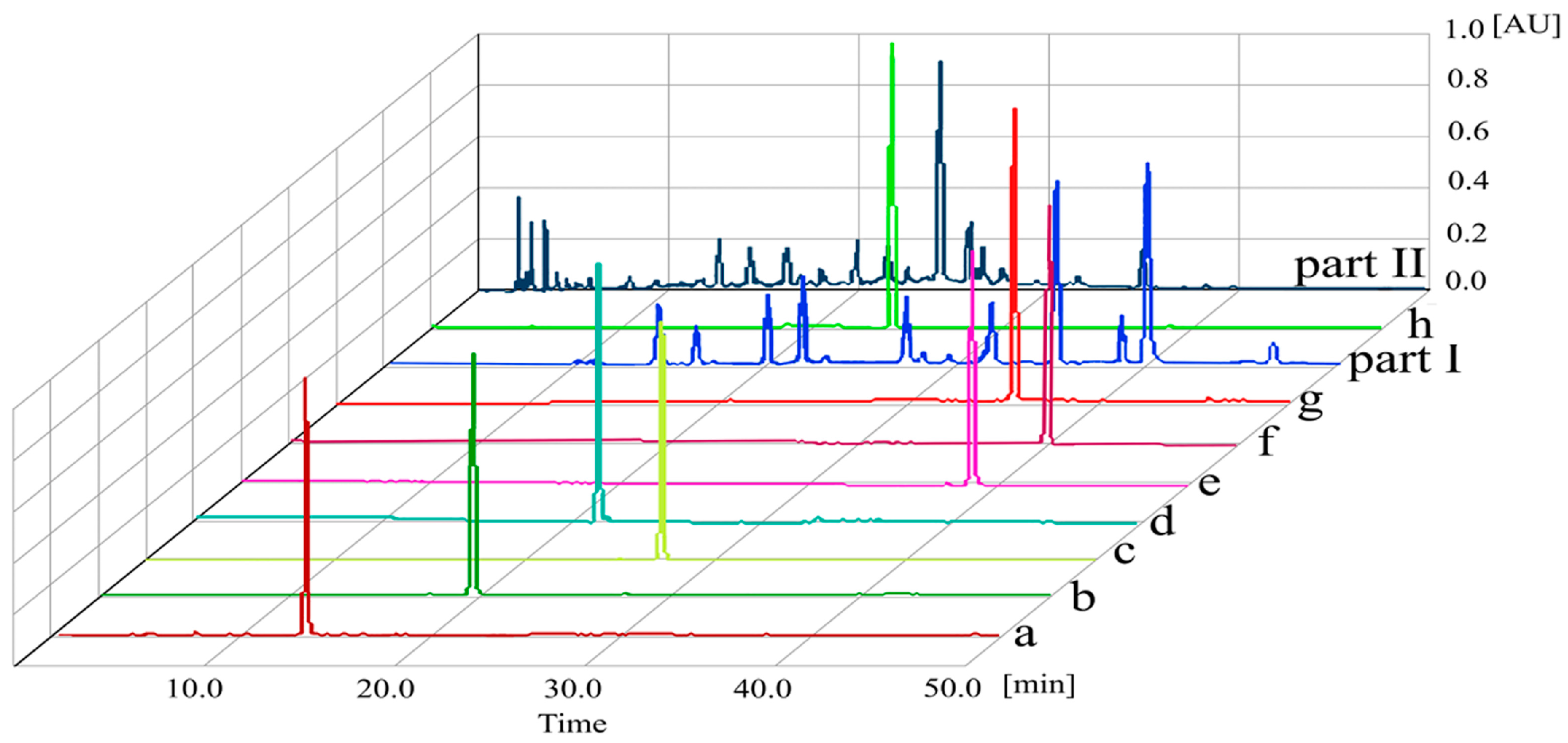
2.3. HPLC Analysis of Crude Extract and HSCCC Peaks
3. Experiment Section
3.1. Apparatus
3.2. Reagents and Materials
3.3. Preparation of Sample
3.4. Determination of Partition Coefficient (KD Value)
3.5. Separation Procedure
3.6. HPLC Analysis
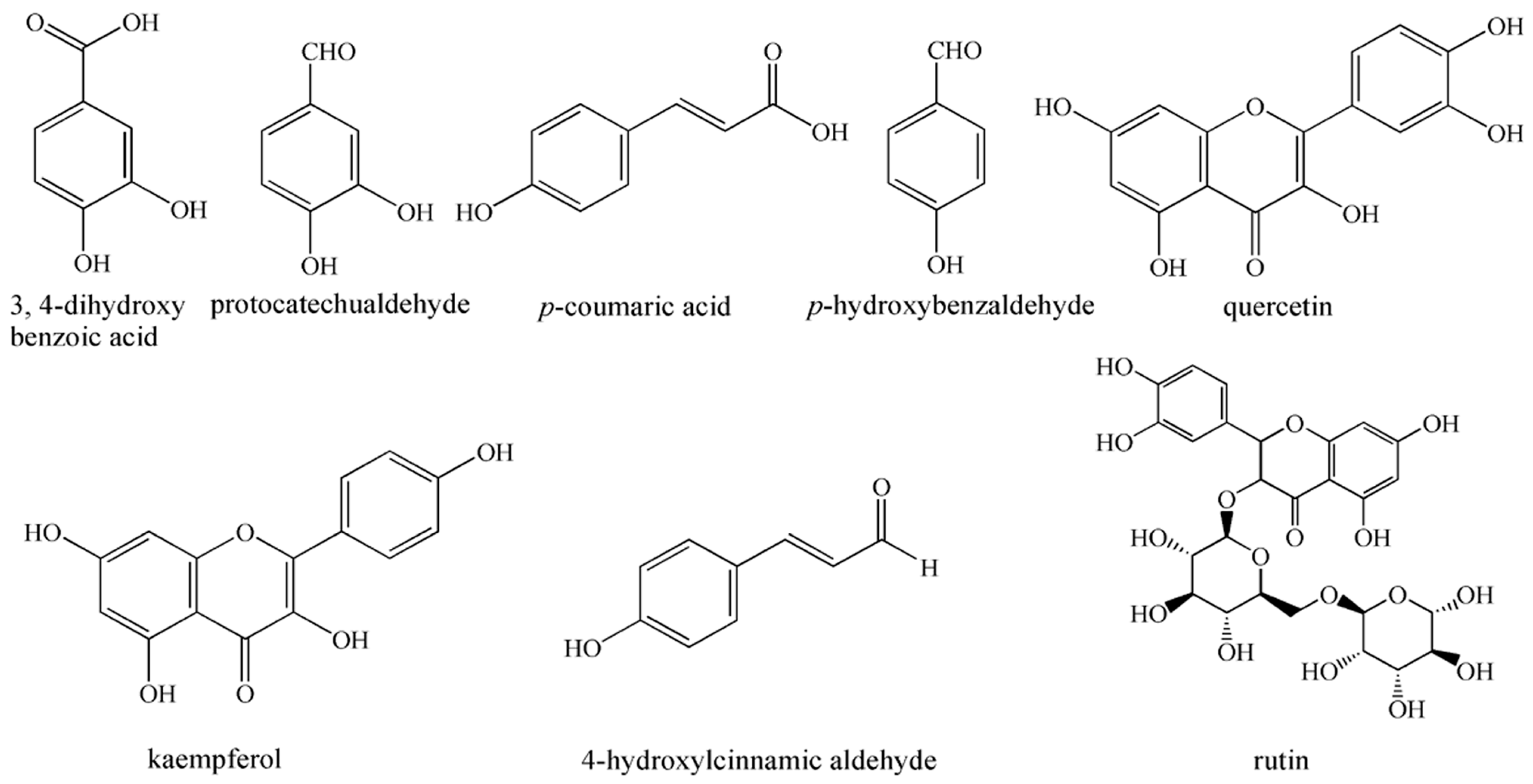
3.7. Identification of the Isolated Compounds
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Xiang, L.; Zhao, K.G.; Chen, L.Q. Molecular cloning and expression of Chimonanthus praecox farnesyl pyrophosphate synthase gene and its possible involvement in the biosynthesis of floral volatile sesquiterpenoids. Plant. Physiol. Biochem. 2010, 48, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.G.; Zhou, M.Q.; Chen, L.Q. Genetic diversity and discrimination of Chimonanthus praecox (L.) Link germplasm using ISSR and RAPD markers. Hort. Sci. 2007, 42, 1144–1148. [Google Scholar]
- Wang, L.Y.; Zhang, Z.B.; Zhou, Z.R.; Zhu, D. Advances of Studies on Chemical Composition and Pharmacological Activity of Chimonanthus Lindl. Lishizhen Med. Mater. Med. Res. 2012, 23, 3103–3106. [Google Scholar]
- Chen, D.W.; Chen, L.Q. The first intraspecific genetic linkage maps of Wintersweet Chimonanthus praecox (L.) Link based on AFLP and ISSR markers. Sci. Hortic. 2010, 1, 88–94. [Google Scholar] [CrossRef]
- Lv, J.F.; Zhang, L.L.; Chu, X.Z.; Zhou, J.F. Chemical composition, antioxidant and antimicrobial activity of the extracts of the flowers of the Chinese plant Chimonanthus praecox. Nat. Prod. Res. 2012, 14, 1363–1367. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Nakanishi, Y.; Ninomiya, K.; Matsuda, H.; Nakashima, S.; Miyashita, H.; Yoshikawa, M.; Hayakawa, T. Dimeric pyrrolidinoindoline-type alkaloids with melanogenesis inhibitory activity in flower buds of Chimonanthus praecox. J. Nat. Med. 2014, 3, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.Y.; Jiang, P. Development and clinical application of Chimonanthus praecox flower cough syrup. Acta Acad. Med. Suzhou 2000, 20, 122–123. [Google Scholar]
- Li, L.; Jiang, Z.H.; Chu, J. Study of Chimonanthus praecox flower in normal decimal immune system effects. Tianjin Pharm. 2000, 12, 29–31. [Google Scholar]
- Wang, X.; Li, F.W.; Geng, Y.L.; Sun, Q.L.; Shi, X.G. Antioxidant Effects of Chimonanthus praecox Flower Extract. Food Sci. 2005, 26, 518–520. [Google Scholar]
- Jiang, T.; Yuan, J.P.; Cheng, C.G.; Li, S.E.; Wang, X.; Chen, L.Z. Analysis of the Essential Oil from Chimonanthus Praecox. Chin. J. Spectrosc. Lab. 2005, 22, 1329–1332. [Google Scholar]
- Liu, D.F.; Sui, S.Z.; Ma, J.; Li, Z.N.; Guo, Y.L.; Luo, D.P.; Yang, J.F.; Li, M.Y. Transcriptomic Analysis of Flower Development in Wintersweet (Chimonanthus praecox). PLoS ONE 2014, 1, e86976. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.G.; Cao, H.; Lee, M.R.; Shen, D.L. Analysis of volatile compounds emitted from Chimonanthus praecox (L.) Link in different florescence and QSRR study of GC retention indices. Chromatographia 2009, 70, 1153–1162. [Google Scholar] [CrossRef]
- Shi, G.F.; An, L.J.; Jiang, B.; Guan, S.; Bao, Y.M. Alpinia protocatechuic acid protects against oxidative damage in vitro and reduces oxidative stress in vivo. Neuosci. Lett. 2006, 403, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Ueyama, Y.; Hashimoto, S.; Nii, H.; Furukawa, K. The volatile constituents of the flower concrete of Chimonanthus praecox Link. from China. Flavour Frag. J. 1990, 5, 85–88. [Google Scholar] [CrossRef]
- Kamaraj, S.; Vinodhkumar, R.; Anandakumar, P.; Jagan, S.; Ramakrishnan, G.; Devaki, T. The effects of quercetin on antioxidant status and tumor markers in the lung and serum of mice treated with benzo(a)pyrene. Biol. Pharm. Bull. 2007, 30, 2268–2273. [Google Scholar] [CrossRef] [PubMed]
- Loke, W.M.; Proudfoot, J.M.; Stewart, S.; McKinley, A.J.; Needs, P.W.; Kroon, P.A.; Hodgson, J.M.; Croft, K.D. Metabolic transformation has a profound effect on anti-inflammatory activity of flavonoids such as quercetin: Lack of association between antioxidant and lipoxygenase inhibitory activity. Biochem. Pharmacol. 2008, 7, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhang, Y.; Ding, X.R.; Chen, S.H.; Yang, J.; Wang, X.J.; Jia, G.L.; Chen, H.S.; Bo, X.C.; Wang, S.Q. Protocatechuic aldehyde inhibits hepatitis B virus replication both in vitro and in vivo. Antivir. Res. 2007, 74, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Zeng, D.M.; He, Q. Studies on chemical constituents of Chimonanthus praecox flower. Asia Pac. Tradit. Med. 2013, 9, 31–32. [Google Scholar]
- Skalicka-Wozniak, K.; Garrard, I. A comprehensive classification of solvent systems used for natural product purifications in countercurrent and centrifugal partition chromatography. Nat. Prod. Rep. 2015, 32, 1556–1561. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y. Golden rules and pitfalls in selecting optimum conditions for high-speed counter-current chromatography. J. Chromatogr. A 2005, 1065, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zheng, Z.J.; Guo, X.F.; Yuan, J.P.; Zheng, C.C. Preparative separation of gingerols from Zingiber officinale by high-speed counter-current chromatography using stepwise elution. Food Chem. 2011, 125, 1476–1480. [Google Scholar] [CrossRef]
- Sutherland, I.A.; Fisher, D. Role of counter-current chromatography in the modernisation of Chinese herbal medicines. J. Chromatogr. A 2009, 1216, 740–753. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Gao, H.F.; Zhao, J.L.; Wang, Q.; Zhou, L.G.; Han, J.G.; Yu, Z.; Yang, F.Y. Preparative Separation of Phenolic Compounds from Halimodendron halodendron by High-Speed Counter-Current Chromatography. Molecules 2010, 15, 5998–6007. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, Y.; Zhang, Y.; Liu, L.; Xu, Y.X.; Xu, Y.H.; Liu, T.H. Preparative isolation and purification of five flavonoid glycosides and one benzophenone galloyl glycoside from Psidium guajava by high-speed counter-current chromatography (HSCCC). Molecules 2013, 18, 15648–15661. [Google Scholar] [CrossRef] [PubMed]
- Chu, X.; Sun, A.L.; Liu, R.M. Preparative isolation and purification of five compounds from the Chinese medicinal herb Polygonum cuspidatum Sieb. et Zucc by high-speed counter-current chromatography. J. Chromatogr. A 2005, 1097, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.H.; Shu, X.K.; Jing, F.; Wang, X.; Lin, C.H.; Luo, A.Q. Preparative Separation of Alkaloids from Picrasma quassioides (D. Don) Benn. by Conventional and pH-Zone-Refining Countercurrent Chromatography. Molecules 2014, 19, 8752–8761. [Google Scholar] [CrossRef] [PubMed]
- Nechepurenko, I.V.; Polovinka, M.P.; Komarova, N.I.; Korchagina, D.V.; Salakhutdinov, N.F.; Nechepurenko, S.B. Low-molecular-weight phenolic compounds from Hedysarum theinum roots. Chem. Nat. Compd. 2008, 44, 31–34. [Google Scholar] [CrossRef]
- Kang, H.S.; Chol, J.H.; Cho, W.K.; Park, J.C.; Choi, J.S. A sphingolipid and tyrosinase inhibitors from the frulting body of Phellinus linteus. Arch. Pharm. Res. 2004, 27, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Youn, K.; Jun, M. Inhibitory effects of key compounds isolated from Corni fructus on BACE1 activity. Phytother. Res. 2012, 26, 1714–1718. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.J.; Liu, Q.; Xie, Y.X.; Zeng, H.L.; Zhang, L.; Jiang, X.Y.; Chen, X.Q. Separation of five flavonoids from tartary buckwheat (Fagopyrum tataricum (L.) Gaertn) grains via off-line two dimensional high-speed counter-current chromatography. Food Chem. 2015, 186, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Abdulla, R.; Yang, Y.; Aisa, H.A. Flavonoids from Gossypium hirsutum flowers. Chem. Nat. Compd. 2008, 44, 370–371. [Google Scholar] [CrossRef]
- Zhu, X.L.; Kou, Y.H.; Huang, X.F.; Kong, L.Y. Studies on the chemical constituents of Alpinia Calanga Willd. Mod. Chin. Med. 2008, 10, 13–15. [Google Scholar]
- Sample Availability: Samples of the phenolic compounds are available from the authors.
| Sample | Solvent System (v/v) | Partition Coefficient (KD) b | |||||||
|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | ||
| Crude extract a | Pet–EtOAc–MeOH–H2O 5:5:6:4 | 0.11 | 0.37 | 2.53 | 1.97 | 1.03 | 8.62 | >10 | <0.1 |
| Pet–EtOAc–MeOH–H2O 5:5:5:5 | 0.21 | 0.58 | 3.01 | 2.79 | 1.38 | >10 | >10 | <0.1 | |
| Pet–EtOAc–MeOH–H2O 5:5:3:7 | 0.79 | 1.32 | 5.36 | 5.21 | 3.01 | >10 | >10 | <0.1 | |
| Part I | Pet–EtOAc–MeOH–H2O–FA 5:5:4:6:0.02 | 0.17 | 0.25 | 0.43 | 0.99 | 1.26 | 1.77 | 2.37 | – |
| Pet–EtOAc–MeOH–H2O–FA 5:5:3:7:0.02 | 0.47 | 0.59 | 0.98 | 1.42 | 1.76 | 2.56 | 3.62 | – | |
| Pet–EtOAc–MeOH–H2O–FA 5:5:4.2:5.8:0.02 | 0.23 | 0.37 | 0.28 | 0.94 | 0.97 | 1.35 | 1.83 | – | |
| Pet–EtOAc–MeOH–H2O–FA 5:5:4.3:5.7:0.02 | 0.16 | 0.18 | 0.25 | 0.64 | 0.92 | 1.12 | 1.51 | – | |
| Part II | EtOAc–EtOH–FA–H2O 4:1:0.1:5 | – | – | – | – | – | – | – | 0.26 |
| EtOAc–n-BuOH–FA–H2O 4:1:0.1:5 | – | – | – | – | – | – | – | 0.47 | |
| EtOAc–n-BuOH–FA–H2O 4:1:0.5:5 | – | – | – | – | – | – | – | 0.92 | |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Zhang, Y.; Liu, Q.; Sun, C.; Li, J.; Yang, P.; Wang, X. Preparative Separation of Phenolic Compounds from Chimonanthus praecox Flowers by High-Speed Counter-Current Chromatography Using a Stepwise Elution Mode. Molecules 2016, 21, 1016. https://doi.org/10.3390/molecules21081016
Li H, Zhang Y, Liu Q, Sun C, Li J, Yang P, Wang X. Preparative Separation of Phenolic Compounds from Chimonanthus praecox Flowers by High-Speed Counter-Current Chromatography Using a Stepwise Elution Mode. Molecules. 2016; 21(8):1016. https://doi.org/10.3390/molecules21081016
Chicago/Turabian StyleLi, Huaizhi, Yongqing Zhang, Qian Liu, Changlei Sun, Jia Li, Peng Yang, and Xiao Wang. 2016. "Preparative Separation of Phenolic Compounds from Chimonanthus praecox Flowers by High-Speed Counter-Current Chromatography Using a Stepwise Elution Mode" Molecules 21, no. 8: 1016. https://doi.org/10.3390/molecules21081016
APA StyleLi, H., Zhang, Y., Liu, Q., Sun, C., Li, J., Yang, P., & Wang, X. (2016). Preparative Separation of Phenolic Compounds from Chimonanthus praecox Flowers by High-Speed Counter-Current Chromatography Using a Stepwise Elution Mode. Molecules, 21(8), 1016. https://doi.org/10.3390/molecules21081016






