Photocatalytic Water Splitting—The Untamed Dream: A Review of Recent Advances
Abstract
:1. Introduction
1.1. Hydrogen and Related Concerns
1.2. Hydrogen Evolution by Solar Energy
1.3. Photocatalytic Water Splitting
2. Photocatalytic Reactions
2.1. Types of Reaction
2.1.1. Photochemical Reactions
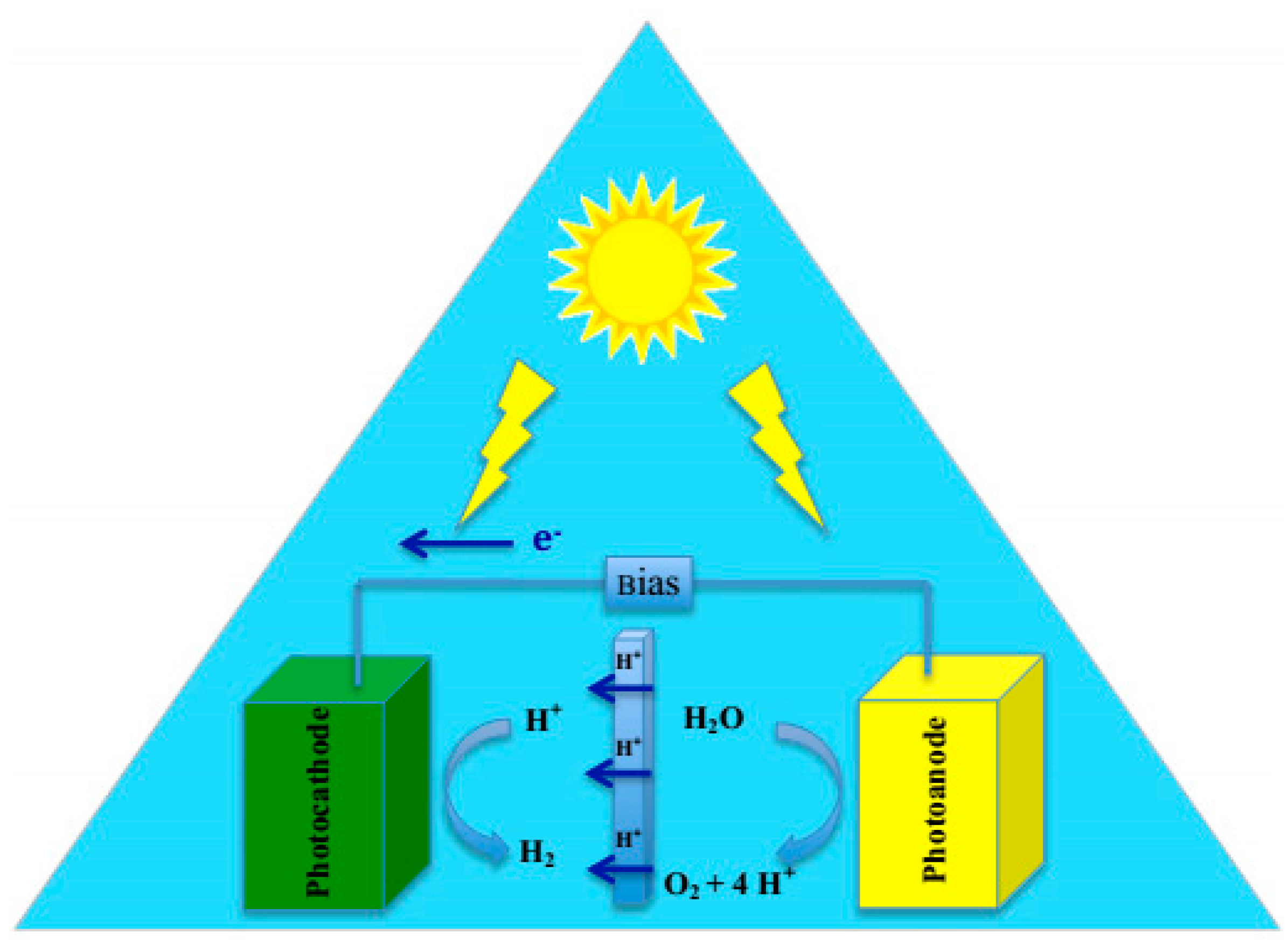
2.1.2. Photo-Electrochemical Reactions
2.2. Reaction Setup
3. Photocatalytic Condition
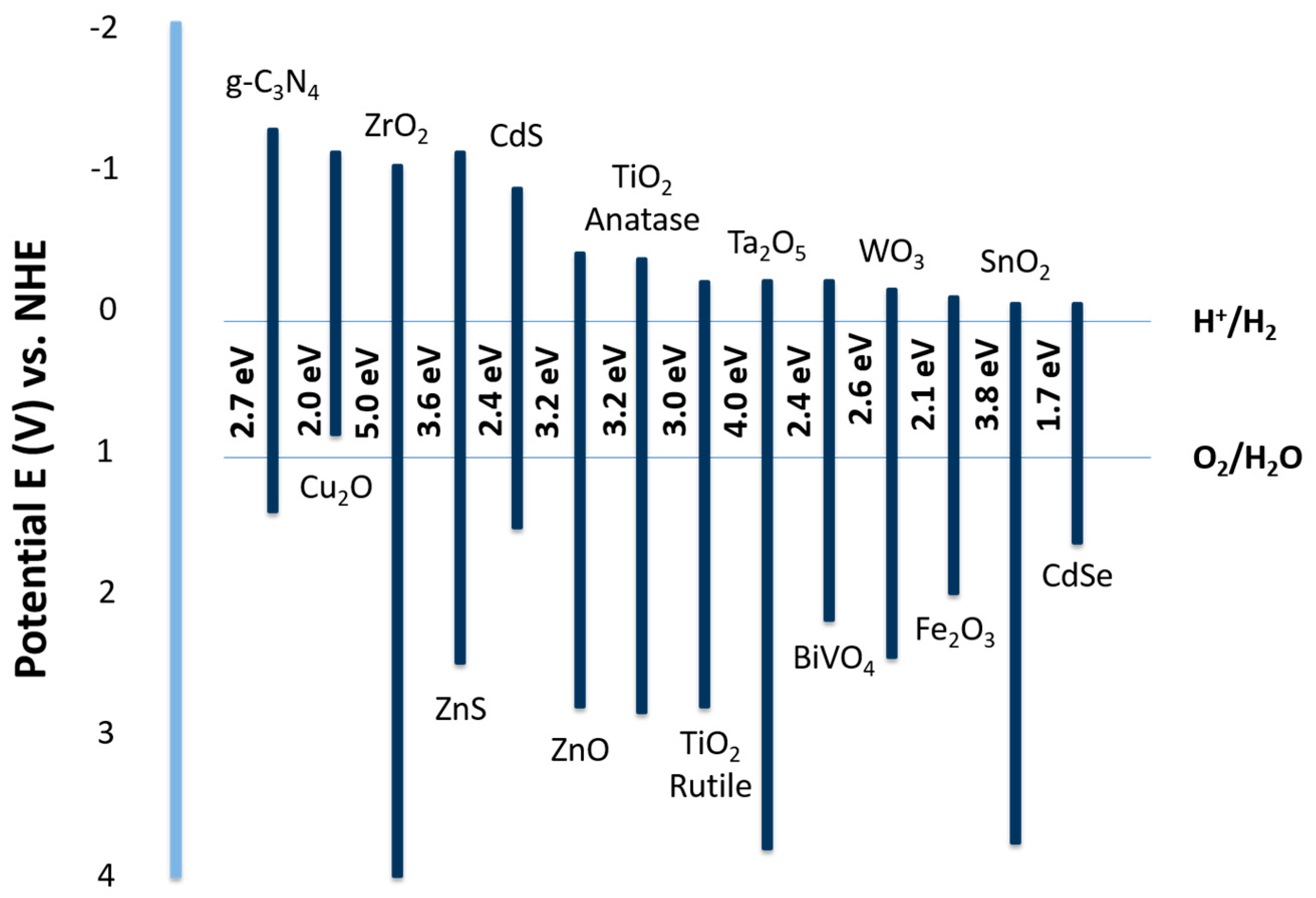
Surface and Band Structure
4. Photocatalyst Materials
4.1. Design and Description
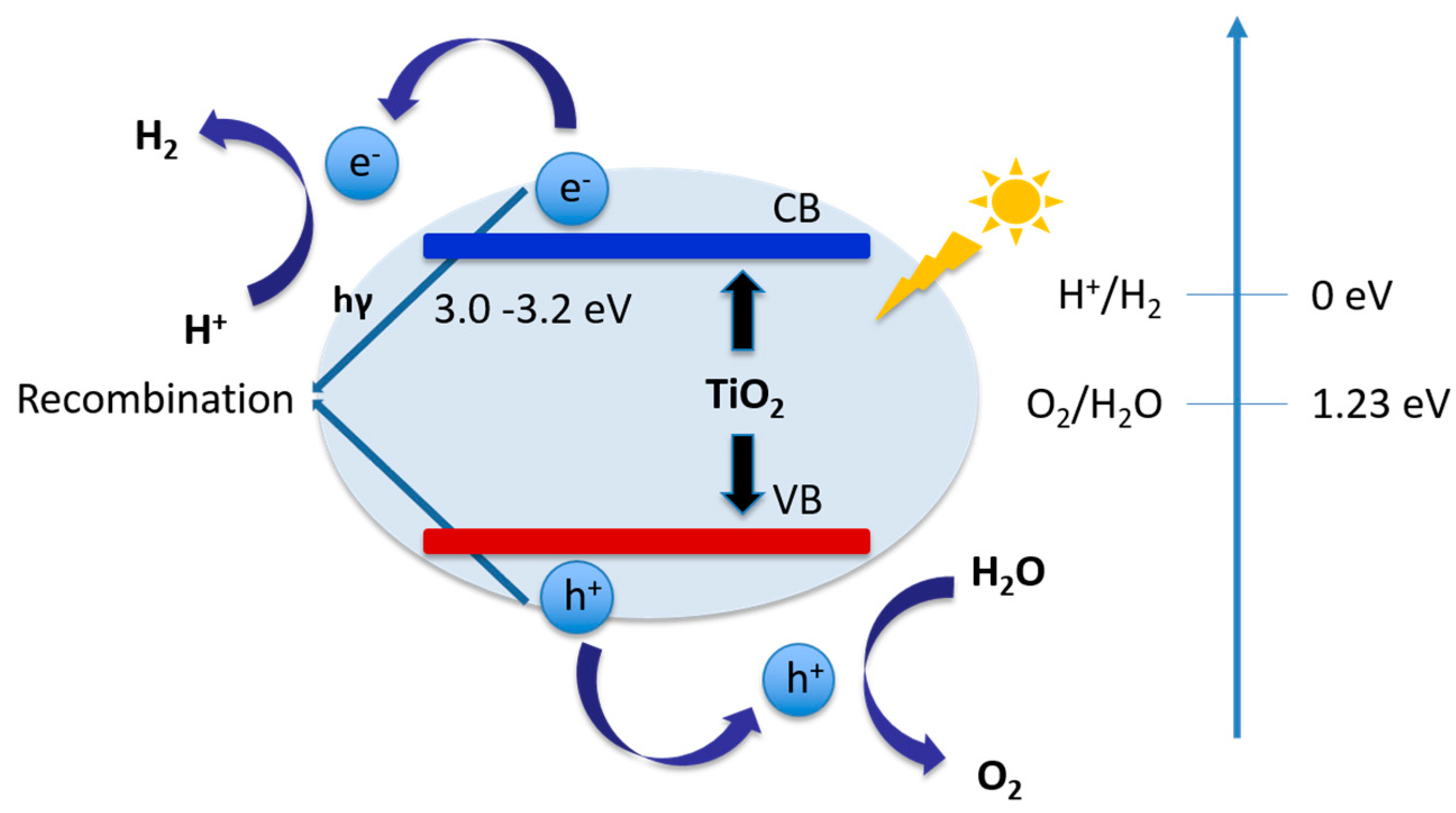
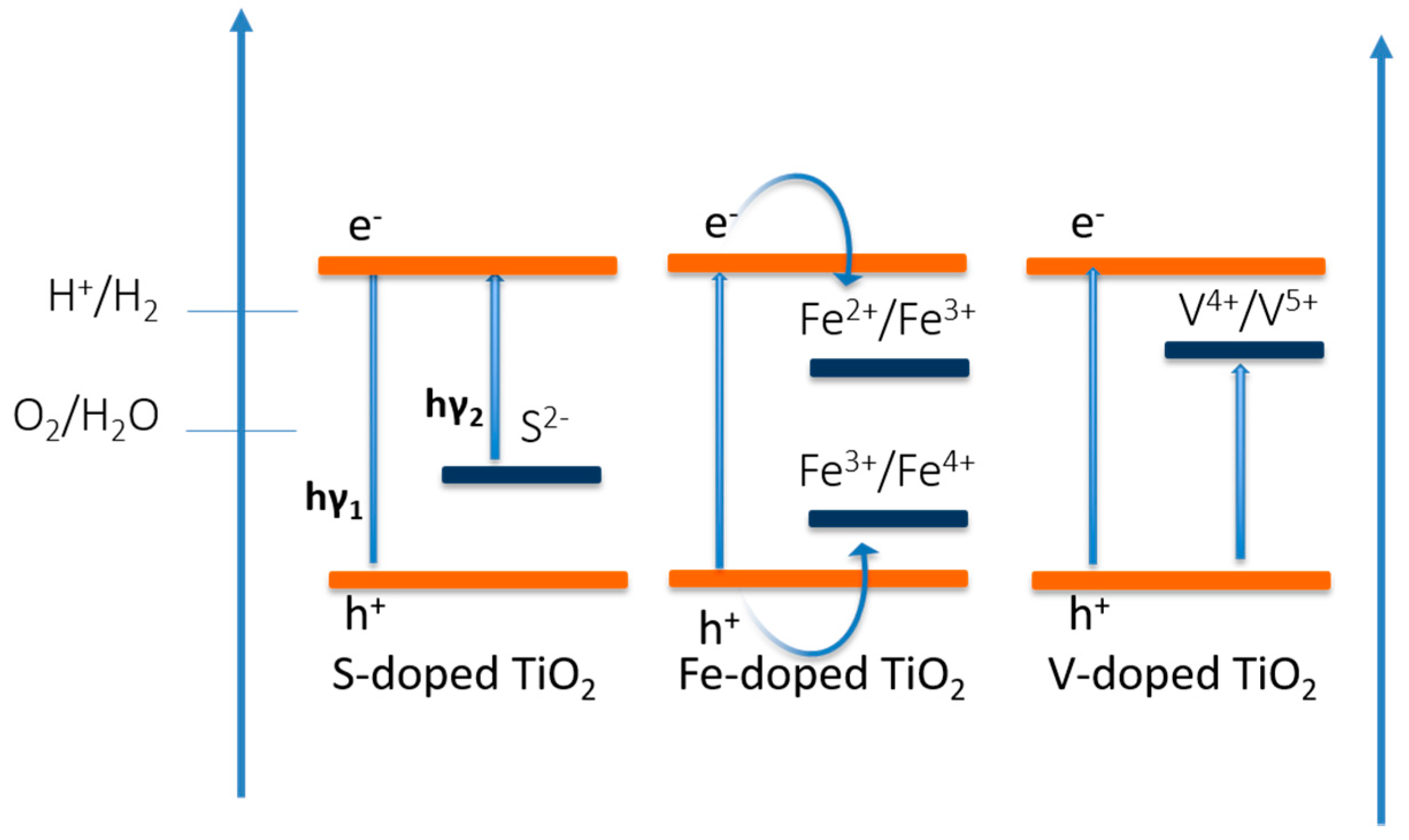
4.2. TiO2
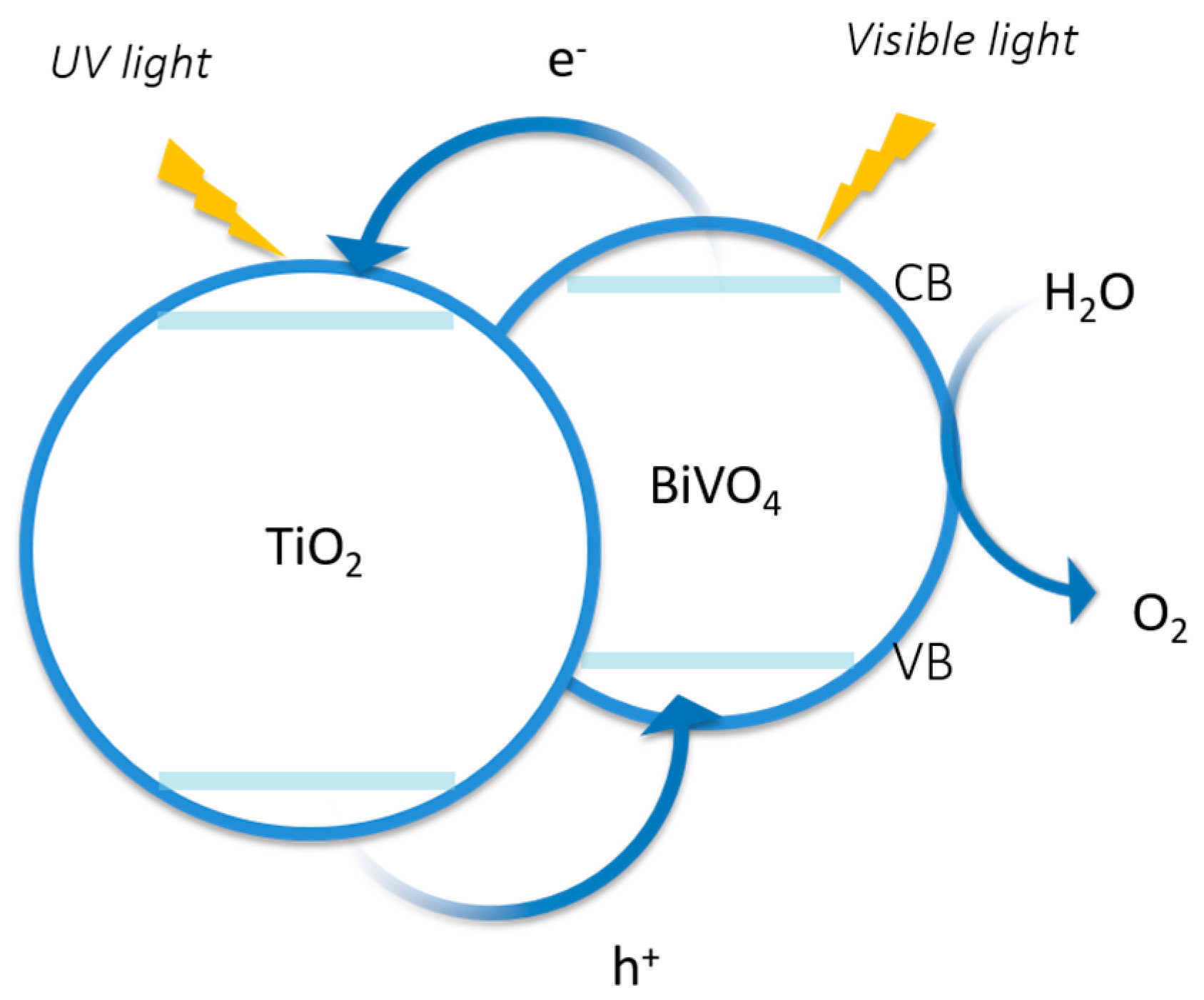
4.3. Metal Oxides
4.4. Metal Sulfides
4.5. Nitrides
5. Theoretical Modeling of Photocatalytic Water Splitting
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Pao, H.-T.; Tsai, C.-M. CO2 emissions, energy consumption and economic growth in BRIC countries. Energy Policy 2010, 38, 7850–7860. [Google Scholar] [CrossRef]
- Davis, S.J.; Caldeira, K. Consumption-based accounting of CO2 emissions. Proc. Natl. Acad. Sci. USA 2010, 107, 5687–5692. [Google Scholar] [CrossRef] [PubMed]
- Dodman, D. Blaming cities for climate change? An analysis of urban greenhouse gas emissions inventories. Environ. Urban. 2009, 21, 185–201. [Google Scholar] [CrossRef]
- Moniz, S.; Shevlin, S.A.; Martin, D.; Guo, Z.; Tang, J. Visible-Light Driven Heterojunction Photocatalysts for Water Splitting—A Critical Review. Energy Environ. Sci. 2015, 8, 731–759. [Google Scholar] [CrossRef]
- Byrne, J.; Hughes, K.; Rickerson, W.; Kurdgelashvili, L. American policy conflict in the greenhouse: Divergent trends in federal, regional, state, and local green energy and climate change policy. Energy Policy 2007, 35, 4555–4573. [Google Scholar] [CrossRef]
- Solomon, S.; Plattner, G.-K.G.; Knutti, R.; Friedlingstein, P. Irreversible climate change due to carbon dioxide emissions. Proc. Natl. Acad. Sci. USA 2009, 106, 1704–1709. [Google Scholar] [CrossRef] [PubMed]
- Di Primio, R.; Horsfield, B.; Guzman-Vega, M. Determining the temperature of petroleum formation from the kinetic properties of petroleum asphaltenes. Nature 2000, 406, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Barbier, E. Geothermal energy technology and current status: An overview. Renew. Sustain. Energy Rev. 2002, 6, 3–65. [Google Scholar] [CrossRef]
- Dincer, I.; Zamfirescu, C.; Dinçer, İ.; Zamfirescu, C.; Dincer, I.; Zamfirescu, C. Sustainable Energy Systems and Applications; Springer Science & Business Media: New York, NY, USA, 2011; Volume 6. [Google Scholar]
- Parida, B.; Iniyan, S.; Goic, R. A review of solar photovoltaic technologies. Renew. Sustain. Energy Rev. 2011, 15, 1625–1636. [Google Scholar] [CrossRef]
- Xie, W.T.; Dai, Y.J.; Wang, R.Z.; Sumathy, K. Concentrated solar energy applications using Fresnel lenses: A review. Renew. Sustain. Energy Rev. 2011, 15, 2588–2606. [Google Scholar] [CrossRef]
- Zamfirescu, C.; Dincer, I.; Naterer, G.F.; Banica, R. Quantum efficiency modeling and system scaling-up analysis of water splitting with Cd1-xZnxS solid-solution photocatalyst. Chem. Eng. Sci. 2013, 97, 235–255. [Google Scholar] [CrossRef]
- Turner, J. A Sustainable hydrogen production. Science 2004, 305, 972–974. [Google Scholar] [CrossRef] [PubMed]
- Melián, E.P.; Díaz, O.G.; Méndez, A.O.; López, C.R.; Suárez, M.N.; Rodríguez, J.M.D.; Navío, J.A.; Hevia, D.F.; Peña, J.P. Efficient and affordable hydrogen production by water photo-splitting using TiO2-based photocatalysts. Int. J. Hydrog. Energy 2013, 38, 2144–2155. [Google Scholar] [CrossRef]
- Zhu, J.; Zäch, M. Nanostructured materials for photocatalytic hydrogen production. Curr. Opin. Colloid Interface Sci. 2009, 14, 260–269. [Google Scholar] [CrossRef]
- Midilli, A.; Ay, M.; Dincer, I.; Rosen, M.A. On hydrogen and hydrogen energy strategies I : Current status and needs. Renew. Sustain. Energy Rev. 2005, 9, 255–271. [Google Scholar] [CrossRef]
- Chiarello, G.L.; Aguirre, M.H.; Selli, E. Hydrogen production by photocatalytic steam reforming of methanol on noble metal-modified TiO2. J. Catal. 2010, 273, 182–190. [Google Scholar] [CrossRef]
- Hou, K.; Hughes, R. The kinetics of methane steam reforming over a Ni/α-Al2O catalyst. Chem. Eng. J. 2001, 82, 311–328. [Google Scholar] [CrossRef]
- Czernik, S.; Evans, R.; French, R. Hydrogen from biomass-production by steam reforming of biomass pyrolysis oil. Catal. Today 2007, 129, 265–268. [Google Scholar] [CrossRef]
- Ni, M.; Leung, D.Y.C.; Leung, M.K.H.; Sumathy, K. An overview of hydrogen production from biomass. Fuel Process. Technol. 2006, 87, 461–472. [Google Scholar] [CrossRef]
- Xie, Q.; Wang, Y.; Pan, B.; Wang, H.; Su, W.; Wang, X. A novel photocatalyst LaOF: Facile fabrication and photocatalytic hydrogen production. Catal. Commun. 2012, 27, 21–25. [Google Scholar] [CrossRef]
- Luo, J.; Im, J.-H.; Mayer, M.T.; Schreier, M.; Nazeeruddin, M.K.; Park, N.-G.; Tilley, S.D.; Fan, H.J.; Gratzel, M. Water photolysis at 12.3% efficiency via perovskite photovoltaics and Earth-abundant catalysts. Science 2014, 345, 1593–1596. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.L.; Lee, M.S. Enhanced TiO2 photocatalysis by Cu in hydrogen production from aqueous methanol solution. Int. J. Hydrog. Energy 2004, 29, 1601–1605. [Google Scholar] [CrossRef]
- Liao, C.-H.; Huang, C.-W.; Wu, J.C.S. Hydrogen Production from Semiconductor-based Photocatalysis via Water Splitting. Catalysts 2012, 2, 490–516. [Google Scholar] [CrossRef]
- Steinfeld, A. Solar hydrogen production via a two-step water-splitting thermochemical cycle based on Zn/ZnO redox reactions. Int. J. Hydrog. Energy 2002, 27, 611–619. [Google Scholar] [CrossRef]
- Akkerman, I.; Janssen, M.; Rocha, J.; Wijffels, R.H. Photobiological hydrogen production: Photochemical efficiency and bioreactor design. Int. J. Hydrog. Energy 2002, 27, 1195–1208. [Google Scholar] [CrossRef]
- Das, D.; Veziroglu, T.N. Advances in biological hydrogen production processes. Int. J. Hydrog. Energy 2008, 33, 6046–6057. [Google Scholar] [CrossRef]
- Guan, Y.; Deng, M.; Yu, X.; Zhang, W. Two-stage photo-biological production of hydrogen by marine green alga Platymonas subcordiformis. Biochem. Eng. J. 2004, 19, 69–73. [Google Scholar] [CrossRef]
- Lewis, N.S.; Nocera, D.G. Powering the planet: Chemical challenges in solar energy utilization. Proc. Natl. Acad. Sci. USA 2006, 103, 15729–15735. [Google Scholar]
- Kudo, A. Photocatalysis and solar hydrogen production. Pure Appl. Chem. 2007, 79, 1917–1927. [Google Scholar] [CrossRef]
- Department of Energy. Fuel Cell Technologies Office Multi-Year Research, Development, and Demonstration Plan. Available online: http://energy.gov/eere/fuelcells/downloads/fuel-cell-technologies-office-multi-year-research-development-and-22 (accessed on 9 May 2016).
- Kudo, A.; Miseki, Y. Heterogeneous photocatalyst materials for water splitting. Chem. Soc. Rev. 2009, 38, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Linsebigler, A.L.; Yates, J.T., Jr.; Lu, G. Photocatalysis on TiO2 Surfaces: Principles, Mechanisms, and Selected Results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Zou, Z.; Ye, J.; Sayama, K.; Arakawa, H. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst. Nature 2001, 414, 625–627. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Halmann, M.; Grätzel, M. Energy Resources through Photochemistry and Catalysis; Academic Press: New York, NY, USA, 1983. [Google Scholar]
- Serpone, N.; Pelizzetti, E. Photocatalysis: Fundamentals and Applications; Wiley: New York, NY, USA, 1989. [Google Scholar]
- Pleskov, Y.V.; Gurevich, Y.Y. Semiconductor Photoelectrochemistry; Consultants Bureau: New York, NY, USA, 1986. [Google Scholar]
- Yoneyama, H.; Sakamoto, H.; Tamura, H. A Photo-Electrochemical Cell With Production of Hydrogen and Oxygen By a Cell Reaction. Electrochim. Acta 1975, 20, 341–345. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.H.; Leung, D.Y.C.; Sumathy, K. A review and recent developments in photocatalytic water-splitting using TiO2 for hydrogen production. Renew. Sustain. Energy Rev. 2007, 11, 401–425. [Google Scholar] [CrossRef]
- Sivula, K.; van de Krol, R. Semiconducting materials for photoelectrochemical energy conversion. Nat. Rev. Mater. 2016, 70, 15010:1–15010:7. [Google Scholar]
- Zhou, W.; Li, W.; Wang, J.-Q.; Qu, Y.; Yang, Y.; Xie, Y.; Zhang, K.; Wang, L.; Fu, H.; Zhao, D. Ordered Mesoporous Black TiO2 as Highly Efficient Hydrogen Evolution Photocatalyst. J. Am. Chem. Soc. 2014, 136, 9280–9283. [Google Scholar] [CrossRef] [PubMed]
- Kay, A.; Cesar, I.; Gratzel, M. New Benchmark for Water Photooxidation by Nanostructured α-Fe2O3 Films. J. Am. Chem. Soc. 2006, 128, 15714–15721. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Magesh, G.; Youn, D.H.; Jang, J.-W.; Kubota, J.; Domen, K.; Lee, J.S. Single-crystalline, wormlike hematite photoanodes for efficient solar water splitting. Sci. Rep. 2013, 3, 2681:1–2681:8. [Google Scholar] [CrossRef] [PubMed]
- Henrich, V.E.; Cox, P.A. The Surface Science of Metal Oxides; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Walsh, A.; Yan, Y.; Huda, M.N.; Al-Jassim, M.M.; Wei, S.-H. Band Edge Electronic Structure of BiVO4: Elucidating the Role of the Bi s and V d Orbitals. Chem. Mater. 2009, 21, 547–551. [Google Scholar] [CrossRef]
- Cooper, J.K.; Gul, S.; Toma, F.M.; Chen, L.; Glans, P.-A.; Guo, J.; Ager, J.W.; Yano, J.; Sharp, I.D. Electronic Structure of Monoclinic BiVO4. Chem. Mater. 2014, 26, 5365–5373. [Google Scholar] [CrossRef]
- Lu, W.; Yuan, D.; Sculley, J.; Zhao, D.; Krishna, R.; Zhou, H.-C. Sulfonate-grafted porous polymer networks for preferential CO2 adsorption at low pressure. J. Am. Chem. Soc. 2011, 133, 18126–18129. [Google Scholar] [CrossRef] [PubMed]
- Kudo, A.; Ueda, K.; Kato, H.; Mikami, I. Photocatalytic O2 evolution under visible light irradiation on BiVO4 in aqueous AgNO3 solution. Catal. Lett. 1998, 53, 229–230. [Google Scholar] [CrossRef]
- Sayama, K.; Nomura, A.; Arai, T.; Sugita, T.; Abe, R.; Yanagida, M.; Oi, T.; Iwasaki, Y.; Abe, Y.; Sugihara, H. Photoelectrochemical decomposition of water into H2 and O2 on porous BiVO4 thin-film electrodes under visible light and significant effect of Ag ion treatment. J. Phys. Chem. B 2006, 110, 11352–11360. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Tsubota, T.; Mooij, L.P.A.; van de Krol, R. Highly Improved Quantum Efficiencies for Thin Film BiVO4 Photoanodes. J. Phys. Chem. C 2011, 115, 17594–17598. [Google Scholar] [CrossRef]
- Ye, H.; Lee, J.; Jang, J.S.; Bard, A.J. Rapid Screening of BiVO4-Based Photocatalysts by Scanning Electrochemical Microscopy (SECM) and Studies of Their Photoelectrochemical Properties. J. Phys. Chem. C 2010, 114, 13322–13328. [Google Scholar] [CrossRef]
- Zhong, D.K.; Choi, S.; Gamelin, D.R. Near-Complete Suppression of Surface Recombination in Solar Photoelectrolysis by “Co-Pi” Catalyst-Modified W:BiVO4. J. Am. Chem. Soc. 2011, 133, 18370–18377. [Google Scholar] [CrossRef] [PubMed]
- Yourey, J.E.; Bartlett, B.M. Electrochemical deposition and photoelectrochemistry of CuWO4, a promising photoanode for water oxidation. J. Mater. Chem. 2011, 21, 7651–7660. [Google Scholar] [CrossRef]
- McDonald, K.J.; Choi, K.-S. Synthesis and Photoelectrochemical Properties of Fe2O3/ZnFe2O4 Composite Photoanodes for Use in Solar Water Oxidation. Chem. Mater. 2011, 23, 4863–4869. [Google Scholar] [CrossRef]
- Ida, S.; Yamada, K.; Matsunaga, T.; Hagiwara, H.; Matsumoto, Y.; Ishihara, T. Preparation of p-Type CaFe2O4 Photocathodes for Producing Hydrogen from Water. J. Am. Chem. Soc. 2010, 132, 17343–17345. [Google Scholar] [CrossRef] [PubMed]
- Patil, R.; Kelkar, S.; Naphade, R.; Ogale, S. Low temperature grown CuBi2O4 with flower morphology and its composite with CuO nanosheets for photoelectrochemical water splitting. J. Mater. Chem. A 2014, 2, 3661–3668. [Google Scholar] [CrossRef]
- Joshi, U.A.; Maggard, P.A. CuNb3O8: A p-Type Semiconducting Metal Oxide Photoelectrode. J. Phys. Chem. Lett. 2012, 3, 1577–1581. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhang, J.; Gong, J. Tantalum-based semiconductors for solar water splitting. Chem. Soc. Rev. 2014, 43, 4395–4422. [Google Scholar] [CrossRef] [PubMed]
- Higashi, M.; Domen, K.; Abe, R. Highly Stable Water Splitting on Oxynitride TaON Photoanode System under Visible Light Irradiation. J. Am. Chem. Soc. 2012, 134, 6968–6971. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Minegishi, T.; Zhang, L.; Zhong, M.; Gunawan, M.; Nakabayashi, M.; Ma, G.; Hisatomi, T.; Katayama, M.; Ikeda, S.; et al. Enhancement of Solar Hydrogen Evolution from Water by Surface Modification with CdS and TiO2 on Porous CuInS2 Photocathodes Prepared by an Electrodeposition-Sulfurization Method. Angew. Chem. Int. Ed. 2014, 53, 11808–11812. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Minegishi, T.; Nakabayashi, M.; Suzuki, Y.; Seki, K.; Shibata, N.; Kubota, J.; Domen, K. Durable hydrogen evolution from water driven by sunlight using (Ag,Cu)GaSe2 photocathodes modified with CdS and CuGa3Se5. Chem. Sci. 2015, 6, 894–901. [Google Scholar] [CrossRef]
- Moriya, M.; Minegishi, T.; Kumagai, H.; Katayama, M.; Kubota, J.; Domen, K. Stable Hydrogen Evolution from CdS-Modified CuGaSe2 Photoelectrode under Visible-Light Irradiation. J. Am. Chem. Soc. 2013, 135, 3733–3735. [Google Scholar] [CrossRef] [PubMed]
- Seger, B.; Laursen, A.B.; Vesborg, P.C.K.; Pedersen, T.; Hansen, O.; Dahl, S.; Chorkendorff, I. Hydrogen Production Using a Molybdenum Sulfide Catalyst on a Titanium-Protected n+p-Silicon Photocathode. Angew. Chem. Int. Ed. 2012, 51, 9128–9131. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Sun, J.; Tang, J.; Yang, P. Zn-Doped p-Type Gallium Phosphide Nanowire Photocathodes from a Surfactant-Free Solution Synthesis. Nano Lett. 2012, 12, 5407–5411. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, M.; Sun, R.-Q.; Wang, X. A Facile Band Alignment of Polymeric Carbon Nitride Semiconductors to Construct Isotype Heterojunctions. Angew. Chem. 2012, 124, 10292–10296. [Google Scholar] [CrossRef]
- Boettcher, S.W.; Warren, E.L.; Putnam, M.C.; Santori, E.A.; Turner-Evans, D.; Kelzenberg, M.D.; Walter, M.G.; McKone, J.R.; Brunschwig, B.S.; et al. Photoelectrochemical hydrogen evolution using Si microwire arrays. J. Am. Chem. Soc. 2011, 133, 1216–1219. [Google Scholar] [CrossRef] [PubMed]
- McKone, J.R.; Warren, E.L.; Bierman, M.J.; Boettcher, S.W.; Brunschwig, B.S.; Lewis, N.S.; Gray, H.B. Evaluation of Pt, Ni, and Ni-Mo electrocatalysts for hydrogen evolution on crystalline Si electrodes. Energy Environ. Sci. 2011, 4, 3573–3583. [Google Scholar] [CrossRef]
- Kainthla, R.C. Significant Efficiency Increase in Self-Driven Photoelectrochemical Cell for Water Photoelectrolysis. J. Electrochem. Soc. 1987, 134, 841–845. [Google Scholar] [CrossRef]
- Kempa, T.J.; Tian, B.; Kim, D.R.; Jinsong, H.; Xiaolin, Z.; Lieber, C.M. Single and tandem axial p-i-n nanowire photovoltaic devices. Nano Lett. 2008, 8, 3456–3460. [Google Scholar] [CrossRef] [PubMed]
- Tanigawa, S.; Irie, H. Visible-light-sensitive two-step overall water-splitting based on band structure control of titanium dioxide. Appl. Catal. B Environ. 2016, 180, 1–5. [Google Scholar] [CrossRef]
- Kandiel, T.A.; Takanabe, K. Solvent-induced deposition of Cu–Ga–In–S nanocrystals onto a titanium dioxide surface for visible-light-driven photocatalytic hydrogen production. Appl. Catal. B Environ. 2016, 184, 264–269. [Google Scholar] [CrossRef]
- Zhu, Z.; Chen, J.Y.; Su, K.Y.; Wu, R.J. Efficient hydrogen production by water-splitting over Pt-deposited C-HS-TiO2 hollow spheres under visible light. J. Taiwan Inst. Chem. Eng. 2016, 60, 222–228. [Google Scholar] [CrossRef]
- Li, L.; Yan, J.; Wang, T.; Zhao, Z.-J.; Zhang, J.; Gong, J.; Guan, N. Sub-10 nm rutile titanium dioxide nanoparticles for efficient visible-light-driven photocatalytic hydrogen production. Nat. Commun. 2015, 6, 5881. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Hisatomi, T.; Ma, S.S.K.; Li, Y.; Domen, K. Core/shell structured La- and Rh-Codoped SrTiO3 as a hydrogen evolution photocatalyst in Z-scheme overall water splitting under visible light irradiation. Chem. Mater. 2014, 26, 4144–4150. [Google Scholar] [CrossRef]
- Chen, Y.; Zhao, S.; Wang, X.; Peng, Q.; Lin, R.; Wang, Y.; Shen, R.; Cao, X.; Zhang, L.; Zhou, G.; et al. Synergetic Integration of Cu1.94S-ZnxCd1-xS Heteronanorods for Enhanced Visible-light-driven Photocatalytic Hydrogen Production. J. Am. Chem. Soc. 2016, 138, 4286–4289. [Google Scholar] [PubMed]
- Dontsova, D.; Fettkenhauer, C.; Papaefthimiou, V.; Schmidt, J.; Antonietti, M. 1,2,4-Triazole-Based Approach to Noble-Metal-Free Visible-Light Driven Water Splitting over Carbon Nitrides. Chem. Mater. 2015, 28, 772–778. [Google Scholar] [CrossRef]
- Fujito, H.; Kunioku, H.; Kato, D.; Suzuki, H.; Higashi, M.; Kageyama, H.; Abe, R. Layered Perovskite Oxychloride Bi4NbO8Cl: A Stable Visible Light Responsive Photocatalyst for Water Splitting. J. Am. Chem. Soc. 2016, 138, 2082–2085. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.; Sun, Z.; Jia, H.; Lu, D.; Du, P. A cocatalyst-free CdS nanorod/ZnS nanoparticle composite for high-performance visible-light-driven hydrogen production from water. J. Mater. Chem. A Mater. Energy Sustain. 2016, 4, 675–683. [Google Scholar] [CrossRef]
- Yue, X.; Yi, S.; Wang, R.; Zhang, Z.; Qiu, S. Cadmium Sulfide and Nickel Synergetic Co-catalysts Supported on Graphitic Carbon Nitride for Visible-Light-Driven Photocatalytic Hydrogen Evolution. Sci. Rep. 2016, 6, 22268:1–22268:9. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.; Cheng, F.; Lang, D. Hierarchical Layered WS2/Graphene-Modified CdS Nanorods for Efficient Photocatalytic Hydrogen Evolution. ChemSusChem 2016, 9, 996–1002. [Google Scholar] [CrossRef] [PubMed]
- Agegnehu, A.K.; Pan, C.-J.; Tsai, M.-C.; Rick, J.; Su, W.-N.; Lee, J.-F.; Hwang, B.-J. Visible light responsive noble metal-free nanocomposite of V-doped TiO2 nanorod with highly reduced graphene oxide for enhanced solar H2 production. Int. J. Hydrog. Energy 2016, 41, 6752–6762. [Google Scholar] [CrossRef]
- Zhang, G.; Lan, Z.-A.; Lin, L.; Lin, S.; Wang, X. Overall water splitting by Pt/g-C3N4 photocatalysts without using sacrificial agents. Chem. Sci. 2016, 7, 3062–3066. [Google Scholar] [CrossRef]
- Zhang, J.; Jin, X.; Morales-Guzman, P.I.; Yu, X.; Liu, H.; Zhang, H.; Razzari, L.; Claverie, J.P. Engineering the Absorption and Field Enhancement Properties of Au-TiO2 Nanohybrids via Whispering Gallery Mode Resonances for Photocatalytic Water Splitting. ACS Nano 2016, 10, 4496–4503. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Hisatomi, T.; Jia, Q.; Tokudome, H.; Zhong, M.; Wang, C.; Pan, Z.; Takata, T.; Nakabayashi, M.; Shibata, N.; et al. Scalable water splitting on particulate photocatalyst sheets with a solar-to-hydrogen energy conversion efficiency exceeding 1. Nat. Mater. 2016, 15, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Gujral, S.S.; Simonov, A.N.; Higashi, M.; Fang, X.-Y.; Abe, R.; Spiccia, L. Highly Dispersed Cobalt Oxide on TaON as Efficient Photoanodes for Long-Term Solar Water Splitting. ACS Catal. 2016, 6, 3404–3417. [Google Scholar] [CrossRef]
- Yuan, Y.-J.; Chen, D.-Q.; Huang, Y.-W.; Yu, Z.-T.; Zhong, J.-S.; Chen, T.-T.; Tu, W.-G.; Guan, Z.-J.; Cao, D.-P.; et al. MoS2 Nanosheet-Modified CuInS2 Photocatalyst for Visible-Light-Driven Hydrogen Production from Water. ChemSusChem 2016, 9, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.L.; Wang, C.H.; Zhao, B.; Dong, J.; Lu, F.; Wang, W.H.; Wang, W.C.; Wu, G.J.; Cui, J.Z.; Cheng, P. A Semi-Conductive Copper-Organic Framework with Two Types of Photocatalytic Activity. Angew. Chem.-Int. Ed. 2016, 55, 4938–4942. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.S. Light work with water. Nature 2001, 414, 589–590. [Google Scholar] [CrossRef] [PubMed]
- Currao, A. Photoelectrochemical water splitting. Chimia (Aarau) 2007, 61, 815–819. [Google Scholar] [CrossRef]
- Ikeda, S.; Itani, T.; Nango, K.; Matsumura, M. Overall water splitting on tungsten-based photocatalysts with defect pyrochlore structure. Catal. Lett. 2004, 98, 229–233. [Google Scholar] [CrossRef]
- Reece, S.Y.; Hamel, J.A.; Sung, K.; Jarvi, T.D.; Esswein, A.J.; Pijpers, J.J.H.; Nocera, D.G. Wireless Solar Water Splitting Using Silicon-Based Semiconductors and Earth-Abundant Catalysts. Science 2011, 334, 645–648. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Li, T.; Wen, B.; Tang, J.; Hu, Z.; Liu, L.-M.; Qu, J.; Huang, C.P.; Liu, H. New Insights into Defect-Mediated Heterostructures for Photoelectrochemical Water Splitting. Adv. Energy Mater. 2016, 6. [Google Scholar] [CrossRef]
- Rajeshwar, K.; Thomas, A.; Janaky, C.; Janáky, C. Photocatalytic Activity of Inorganic Semiconductor Surfaces: Myths, Hype, and Reality. J. Phys. Chem. Lett. 2015, 6, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yu, X. Hydrothermal Synthesis and Photocatalytic Activity of Zinc Oxide Hollow Spheres. Environ. Sci. Technol. 2008, 42, 4902–4907. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Li, L.; Yi, T.; Zhang, W.; Zhang, X.; Wang, L. Microwave assisted synthesis of sheet-like Cu/BiVO4 and its activities of various photocatalytic conditions. J. Solid State Chem. 2015, 229, 141–149. [Google Scholar] [CrossRef]
- Luo, Z.; Poyraz, A.S.; Kuo, C.-H.; Miao, R.; Meng, Y.; Chen, S.-Y.; Jiang, T.; Wenos, C.; Suib, S.L. Crystalline Mixed Phase (Anatase/Rutile) Mesoporous Titanium Dioxides for Visible Light Photocatalytic Activity. Chem. Mater. 2015, 27, 6–17. [Google Scholar] [CrossRef]
- Nithya, V.D.; Hanitha, B.; Surendran, S.; Kalpana, D.; Kalai Selvan, R. Effect of pH on the sonochemical synthesis of BiPO4 nanostructures and its electrochemical properties for pseudocapacitors. Ultrason. Sonochem. 2015, 22, 300–310. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, H.; Kamarudin, S.K.; Minggu, L.J.; Kassim, M. Hydrogen from photo-catalytic water splitting process: A review. Renew. Sustain. Energy Rev. 2015, 43, 599–610. [Google Scholar] [CrossRef]
- DeMeo, D.; Sonkusale, S.; MacNaughton, S.; Vandervelde, T. Nanowires-Implementations and Applications; InTech Europe: Rijeka, Croatia, 2011. [Google Scholar]
- Dubale, A.A.; Pan, C.-J.; Tamirat, A.G.; Chen, H.-M.; Su, W.-N.; Chen, C.-H.; Rick, J.; Ayele, D.W.; Aragaw, B.A.; Lee, J.-F.; et al. Heterostructured Cu2O/CuO decorated with nickel as a highly efficient photocathode for photoelectrochemical water reduction. J. Mater. Chem. A 2015, 3, 12482–12499. [Google Scholar] [CrossRef]
- Hou, Y.; Zuo, F.; Dagg, A.; Feng, P. A Three-Dimensional Branched Cobalt-Doped α-Fe2O3 Nanorod/MgFe2O4 Heterojunction Array as a Flexible Photoanode for Efficient Photoelectrochemical Water Oxidation. Angew. Chem. 2013, 125, 1286–1290. [Google Scholar] [CrossRef]
- Li, Z.; Qu, Y.; He, G.; Humayun, M.; Chen, S.; Jing, L. Enhanced visible-light activities for PEC water reduction of CuO nanoplates by coupling with anatase TiO2 and mechanism. Appl. Surf. Sci. 2015, 351, 681–685. [Google Scholar] [CrossRef]
- Samaele, N.; Amornpitoksuk, P.; Suwanboon, S. Effect of pH on the morphology and optical properties of modified ZnO particles by SDS via a precipitation method. Powder Technol. 2010, 203, 243–247. [Google Scholar] [CrossRef]
- Tan, G.; Zhang, L.; Ren, H.; Wei, S.; Huang, J.; Xia, A. Effects of pH on the hierarchical structures and photocatalytic performance of BiVO4 powders prepared via the microwave hydrothermal method. ACS Appl. Mater. Interfaces 2013, 5, 5186–5193. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Yang, C.; Li, Q.; Cao, W.; Li, T. The pH-controlled morphology transition of BiVO4 photocatalysts from microparticles to hollow microspheres. Mater. Lett. 2015, 145, 52–55. [Google Scholar] [CrossRef]
- Obregón, S.; Caballero, A.; Colón, G. Hydrothermal synthesis of BiVO4: Structural and morphological influence on the photocatalytic activity. Appl. Catal. B Environ. 2012, 117–118, 59–66. [Google Scholar]
- Sun, S.; Wang, W.; Zhou, L.; Xu, H. Efficient Methylene Blue Removal over Hydrothermally Synthesized Starlike BiVO4. Ind. Eng. Chem. Res. 2009, 48, 1735–1739. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, T.; Zhao, X.; Zhu, Y. Controllable synthesis of Bi2MoO6 and effect of morphology and variation in local structure on photocatalytic activities. Appl. Catal. B Environ. 2010, 98, 138–146. [Google Scholar] [CrossRef]
- Ge, M.; Liu, L.; Chen, W.; Zhou, Z. Sunlight-driven degradation of Rhodamine B by peanut-shaped porous BiVO4 nanostructures in the H2O2-containing system. CrystEngComm 2012, 14, 1038–1044. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, X.-B.B.; Wang, L.-W.W.; Wang, W.-K.K.; Long, L.L.; Li, W.-W.W.; Yu, H.Q. Synthesis of a highly efficient BiOCl single-crystal nanodisk photocatalyst with exposing {001} facets. ACS Appl. Mater. Interfaces 2014, 6, 7766–7772. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, J.; Meng, D.; Xing, Y.; Wang, C. Enhanced photocatalytic activity of hierarchically structured BiVO4 oriented along {040} facets with different morphologies. Mater. Lett. 2015, 147. [Google Scholar] [CrossRef]
- Guijarro, N.; Prévot, M.S.; Sivula, K. Surface modification of semiconductor photoelectrodes. Phys. Chem. Chem. Phys. 2015, 17, 15655–15674. [Google Scholar] [CrossRef] [PubMed]
- Meshram, S.P.; Adhyapak, P.V.; Mulik, U.P.; Amalnerkar, D.P. Facile synthesis of CuO nanomorphs and their morphology dependent sunlight driven photocatalytic properties. Chem. Eng. J. 2012, 204–206, 158–168. [Google Scholar] [CrossRef]
- Lu, Y.; Shang, H.; Shi, F.; Chao, C.; Zhang, X.; Zhang, B. Preparation and efficient visible light-induced photocatalytic activity of m-BiVO4 with different morphologies. J. Phys. Chem. Solids 2015, 85, 44–50. [Google Scholar] [CrossRef]
- Wu, Q.; Han, R.; Chen, P.; Qi, X.; Yao, W. Novel synthesis and photocatalytic performance of BiVO4 with tunable morphologies and macroscopic structures. Mater. Sci. Semicond. Process. 2015, 38, 271–277. [Google Scholar] [CrossRef]
- Liu, W.; Zhao, G.; An, M.; Chang, L. Solvothermal synthesis of nanostructured BiVO4 with highly exposed (010) facets and enhanced sunlight-driven photocatalytic properties. Appl. Surf. Sci. 2015, 357, 1053–1063. [Google Scholar] [CrossRef]
- Meng, X.; Zhang, L.; Dai, H.; Zhao, Z.; Zhang, R.; Liu, Y. Surfactant-assisted hydrothermal fabrication and visible-light-driven photocatalytic degradation of methylene blue over multiple morphological BiVO4 single-crystallites. Mater. Chem. Phys. 2011, 125, 59–65. [Google Scholar] [CrossRef]
- Hu, Y.; Li, D.; Sun, F.; Wang, H.; Weng, Y.; Xiong, W.; Shao, Y. One-pot template-free synthesis of heterophase BiVO4 microspheres with enhanced photocatalytic activity. RSC Adv. 2015, 5, 54882–54889. [Google Scholar] [CrossRef]
- Liu, W.; Yu, Y.; Cao, L.; Su, G.; Liu, X.; Zhang, L.; Wang, Y. Synthesis of monoclinic structured BiVO4 spindly microtubes in deep eutectic solvent and their application for dye degradation. J. Hazard. Mater. 2010, 181, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Yang, X.; Ma, F.; Li, K.; Xu, L.; Yuan, X.; Guo, Y. Additive-free controllable fabrication of bismuth vanadates and their photocatalytic activity toward dye degradation. Appl. Surf. Sci. 2010, 256, 2215–2222. [Google Scholar] [CrossRef]
- Zhao, Y.; Xie, Y.; Zhu, X.; Yan, S.; Wang, S. Surfactant-free synthesis of hyperbranched monoclinic bismuth vanadate and its applications in photocatalysis, gas sensing, and lithium-ion batteries. Chem. Eur. J. 2008, 14, 1601–1606. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.; Wang, D.; Wang, L.; Li, H.; Wang, P.; Jiang, T.; Xie, T. Hydrothermal synthesis and photoelectric properties of BiVO4 with different morphologies: An efficient visible-light photocatalyst. Appl. Surf. Sci. 2011, 257, 7758–7762. [Google Scholar] [CrossRef]
- Jiang, H.; Dai, H.; Meng, X.; Ji, K.; Zhang, L.; Deng, J. Porous olive-like BiVO4: Alcoho-hydrothermal preparation and excellent visible-light-driven photocatalytic performance for the degradation of phenol. Appl. Catal. B Environ. 2011, 105, 326–334. [Google Scholar] [CrossRef]
- Wang, Q.; Jiang, H.; Ding, S.; Noh, H.M.; Moon, B.K.; Choi, B.C.; Shi, J.; Jeong, J.H. Butterfly-like BiVO4: Synthesis and Visible Light Photocatalytic Activity. Synth. React. Inorg. Met. Nano-Metal Chem. 2015, 46, 483–488. [Google Scholar] [CrossRef]
- Gu, S.; Li, W.; Wang, F.; Wang, S.; Zhou, H.; Li, H. Synthesis of buckhorn-like BiVO4 with a shell of CeOx nanodots: Effect of heterojunction structure on the enhancement of photocatalytic activity. Appl. Catal. B Environ. 2015, 170–171, 186–194. [Google Scholar] [CrossRef]
- Guo, Z.; Li, P.; Che, H.; Wang, G.; Wu, C.; Zhang, X.; Mu, J. One-dimensional spindle-like BiVO4/TiO2 nanofibers heterojunction nanocomposites with enhanced visible light photocatalytic activity. Ceram. Int. 2016, 42, 4517–4525. [Google Scholar] [CrossRef]
- Ju, P.; Wang, Y.; Sun, Y.; Zhang, D. Controllable one-pot synthesis of a nest-like Bi2WO6/BiVO4 composite with enhanced photocatalytic antifouling performance under visible light irradiation. Dalt. Trans. 2016, 45, 4588–4602. [Google Scholar] [CrossRef] [PubMed]
- Bao, N.; Yin, Z.; Zhang, Q.; He, S.; Hu, X.; Miao, X. Synthesis of flower-like monoclinic BiVO4/surface rough TiO2 ceramic fiber with heterostructures and its photocatalytic property. Ceram. Int. 2016, 42, 1791–1800. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, F.; Wang, M.; Gao, X.; Luo, Y.; Xue, J.; Zhang, Y.; Ding, J.; Sun, S.; Bao, J.; et al. A shuriken-shaped m-BiVO4/{001}-TiO2 heterojunction: Synthesis, structure and enhanced visible light photocatalytic activity. Appl. Catal. A Gen. 2015, in press. [Google Scholar] [CrossRef]
- Bao, J. Photoelectrochemical water splitting: A new use for bandgap engineering. Nat. Nanotechnol. 2015, 10, 19–20. [Google Scholar] [CrossRef] [PubMed]
- Marschall, R. Semiconductor Composites: Strategies for Enhancing Charge Carrier Separation to Improve Photocatalytic Activity. Adv. Funct. Mater. 2014, 24, 2421–2440. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Q.; Xueying, Z.; Wang, F.; Safdar, M.; He, J. Visible light driven type II heterostructures and their enhanced photocatalysis properties: A review. Nanoscale 2013, 5, 8326–8339. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hoffmann, M.W.G.; Shen, H.; Fabrega, C.; Prades, J.D.; Andreu, T.; Hernandez-Ramirez, F.; Mathur, S. Enhanced photoelectrochemical activity of an excitonic staircase in CdS@TiO2 and CdS@anatase@rutile TiO2 heterostructures. J. Mater. Chem. 2012, 22, 20472–20476. [Google Scholar] [CrossRef]
- Li, J.; Cushing, S.K.; Zheng, P.; Senty, T.; Meng, F.; Bristow, A.D.; Manivannan, A.; Wu, N. Solar Hydrogen Generation by a CdS-Au-TiO2 Sandwich Nanorod Array Enhanced with Au Nanoparticle as Electron Relay and Plasmonic Photosensitizer. J. Am. Chem. Soc. 2014, 136, 8438–8449. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, C.; Xu, J.; Liu, X.; Zou, R.; Ouyang, L.; Xu, X.; Chen, X.; Xing, H. Fabrication of ZnO/CdS/ Cu2ZnSnS4 p-n heterostructure nanorod arrays via a solution-based route. CrystEngComm 2013, 15, 1139–1145. [Google Scholar] [CrossRef]
- Wang, X.; Yin, L.; Liu, G.; Wang, L.; Saito, R.; Lu, G.Q.M.; Cheng, H.-M. Polar interface-induced improvement in high photocatalytic hydrogen evolution over ZnO-CdS heterostructures. Energy Environ. Sci. 2011, 4, 3976–3979. [Google Scholar] [CrossRef]
- Pihosh, Y.; Turkevych, I.; Mawatari, K.; Uemura, J.; Kazoe, Y.; Kosar, S.; Makita, K.; Sugaya, T.; Matsui, T.; Fujita, D.; et al. Photocatalytic generation of hydrogen by core-shell WO3/BiVO4 nanorods with ultimate water splitting efficiency. Sci. Rep. 2015, 5, 11141:1–11141:10. [Google Scholar] [CrossRef] [PubMed]
- Ji, L.; McDaniel, M.D.; Wang, S.; Posadas, A.B.; Li, X.; Huang, H.; Lee, J.C.; Demkov, A.A.; Bard, A.J.; Ekerdt, J.G.; et al. A silicon-based photocathode for water reduction with an epitaxial SrTiO3 protection layer and a nanostructured catalyst. Nat. Nanotechnol. 2015, 10, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Liao, L.; Zhang, Q.; Su, Z.; Zhao, Z.; Wang, Y.; Li, Y.; Lu, X.; Wei, D.; Feng, G.; Yu, Q.; et al. Efficient solar water-splitting using a nanocrystalline CoO photocatalyst. Nat. Nanotechnol. 2014, 9, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Paz, D.S.; Foletto, E.L.; Bertuol, D.A.; Jahn, S.L.; Collazzo, G.C.; da Silva, S.S.; Chiavone-Filho, O.; do Nascimento, C.A.O. CuO/ZnO coupled oxide films obtained by the electrodeposition technique and their photocatalytic activity in phenol degradation under solar irradiation. Water Sci. Technol. 2013, 68, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- Ong, W.-J.; Tan, L.-L.; Ng, Y.H.; Yong, S.-T.; Chai, S.-P. Graphitic Carbon Nitride (g-C3N4)-Based Photocatalysts for Artificial Photosynthesis and Environmental Remediation: Are We a Step Closer To Achieving Sustainability? Chem. Rev. 2016, 116, 7159–7329. [Google Scholar] [CrossRef] [PubMed]
- Khakpash, N.; Simchi, A.; Jafari, T. Adsorption and solar light activity of transition-metal doped TiO2 nanoparticles as semiconductor photocatalyst. J. Mater. Sci. Mater. Electron. 2012, 23, 659–667. [Google Scholar] [CrossRef]
- Miyauchi, M.; Irie, H.; Liu, M.; Qiu, X.; Yu, H.; Sunada, K.; Hashimoto, K. Visible-light-sensitive Photocatalysts. Nanocluster-grafted Titanium Dioxide for Indoor Environmental Remediation. J. Phys. Chem. Lett. 2016, 7, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Pan, L.; Song, J.J.; Mi, W.; Zou, J.J.; Wang, L.; Zhang, X. Titanium-defected undoped anatase TiO2 with p-type conductivity, room-temperature ferromagnetism, and remarkable photocatalytic performance. J. Am. Chem. Soc. 2015, 137, 2975–2983. [Google Scholar] [CrossRef] [PubMed]
- Fei, T.; Rongshu, Z.; Kelin, S.; Feng, O.; Gang, C. Synergistic Photocatalytic Degradation of Phenol Using Precious Metal Supported Titanium Dioxide with Hydrogen Peroxide. Environ. Eng. Sci. 2016, 33, 185–192. [Google Scholar]
- Li, B.; Zhao, J.; Liu, J.; Shen, X.; Mo, S.; Tong, H. Bio-templated synthesis of hierarchically ordered macro-mesoporous anatase titanium dioxide flakes with high photocatalytic activity. RSC Adv. 2015, 5, 15572–15578. [Google Scholar] [CrossRef]
- Ismail, A.A.; Bahnemann, D.W. Photochemical splitting of water for hydrogen production by photocatalysis: A review. Sol. Energy Mater. Sol. Cells 2014, 128, 85–101. [Google Scholar] [CrossRef]
- Miao, R.; Luo, Z.; Zhong, W.; Chen, S.-Y.; Jiang, T.; Dutta, B.; Nasr, Y.; Zhang, Y.; Suib, S.L. Mesoporous TiO2 modified with carbon quantum dots as a high-performance visible light photocatalyst. Appl. Catal. B Environ. 2016, 189, 26–38. [Google Scholar] [CrossRef]
- Chen, X.; Mao, S.S. Titanium dioxide nanomaterials: Synthesis, properties, modifications and applications. Chem. Rev. 2007, 107, 2891–2959. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Cowan, A.J.; Durrant, J.R.; Klug, D.R. Mechanism of O2 Production from Water Splitting: Nature of Charge Carriers in Nitrogen Doped Nanocrystalline TiO2 Films and Factors Limiting O2 Production. J. Phys. Chem. C 2011, 115, 3143–3150. [Google Scholar] [CrossRef]
- Piskunov, S.; Lisovski, O.; Begens, J.; Bocharov, D.; Zhukovskii, Y.F.; Wessel, M.; Spohr, E. C-, N-, S-, and Fe-Doped TiO2 and SrTiO3 Nanotubes for Visible-Light-Driven Photocatalytic Water Splitting: Prediction from First Principles. J. Phys. Chem. C 2015, 119, 18686–18696. [Google Scholar] [CrossRef]
- Luo, H.; Takata, T.; Lee, Y.; Zhao, J.; Domen, K. Yushan Photocatalytic Activity Enhancing for Titanium Dioxide by Co-doping with Bromine and Chlorine. Chem. Mater. 2004, 16, 846–849. [Google Scholar] [CrossRef]
- Wang, H.; Dong, S.; Chang, Y.; Faria, J.L. Enhancing the photocatalytic properties of TiO2 by coupling with carbon nanotubes and supporting gold. J. Hazard. Mater. 2012, 235–236, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Gao, B.; Chen, G.Z.; Mokaya, R.; Sotiropoulos, S.; Li Puma, G. Carbon nanotube/titanium dioxide (CNT/TiO2) core-shell nanocomposites with tailored shell thickness, CNT content and photocatalytic/ photoelectrocatalytic properties. Appl. Catal. B Environ. 2011, 110, 50–57. [Google Scholar] [CrossRef]
- Silva, C.G.; Faria, J.L. Photocatalytic oxidation of benzene derivatives in aqueous suspensions: Synergic effect induced by the introduction of carbon nanotubes in a TiO2 matrix. Appl. Catal. B Environ. 2010, 101, 81–89. [Google Scholar] [CrossRef]
- Xu, Y.-J.; Zhuang, Y.; Fu, X. New Insight for Enhanced Photocatalytic Activity of TiO2 by Doping Carbon Nanotubes: A Case Study on Degradation of Benzene and Methyl Orange. J. Phys. Chem. C 2010, 114, 2669–2676. [Google Scholar] [CrossRef]
- Babu, V.J.; Vempati, S.; Uyar, T.; Ramakrishna, S. Review of one-dimensional and two-dimensional nanostructured materials for hydrogen generation. Phys. Chem. Chem. Phys. 2015, 17, 2960–2986. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shen, S.; Guo, L.; Mao, S.S. Semiconductor-based photocatalytic hydrogen generation. Chem. Rev. 2010, 110, 6503–6570. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K. Z-scheme water splitting using two different semiconductor photocatalysts. ACS Catal. 2013, 3, 1486–1503. [Google Scholar] [CrossRef]
- Resasco, J.; Zhang, H.; Kornienko, N.; Becknell, N.; Lee, H.; Guo, J.; Briseno, A.L.; Yang, P. TiO2 /BiVO4 Nanowire Heterostructure Photoanodes Based on Type II Band Alignment. ACS Cent. Sci. 2016, 2, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-T.; Chan, A.; Al-Azri, Z.H.N.; Dosado, A.G.; Nadeem, M.A.; Sun-Waterhouse, D.; Idriss, H.; Waterhouse, G.I.N. Effect of TiO2 polymorph and alcohol sacrificial agent on the activity of Au/TiO2 photocatalysts for H2 production in alcohol-water mixtures. J. Catal. 2015, 329, 499–513. [Google Scholar] [CrossRef]
- Reza Gholipour, M.; Dinh, C.-T.; Béland, F.; Do, T.-O. Nanocomposite heterojunctions as sunlight-driven photocatalysts for hydrogen production from water splitting. Nanoscale 2015, 7, 8187–8208. [Google Scholar] [CrossRef] [PubMed]
- Majeed, I.; Nadeem, M.A.A.; Al-Oufi, M.; Nadeem, M.A.A.; Waterhouse, G.I.N.; Badshah, A.; Metson, J.B.; Idriss, H. On the role of metal particle size and surface coverage for photo-catalytic hydrogen production: A case study of the Au/CdS system. Appl. Catal. B Environ. 2016, 182, 266–276. [Google Scholar] [CrossRef]
- Sreethawong, T.; Yoshikawa, S. Comparative investigation on photocatalytic hydrogen evolution over Cu-, Pd-, and Au-loaded mesoporous TiO2 photocatalysts. Catal. Commun. 2005, 6, 661–668. [Google Scholar] [CrossRef]
- Tian, Y.; Tatsuma, T. Mechanisms and applications of plasmon-induced charge separation at TiO2 films loaded with gold nanoparticles. J. Am. Chem. Soc. 2005, 127, 7632–7637. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Liu, D.; Mubeen, S.; Chuong, T.T.; Moskovits, M.; Stucky, G.D. Anisotropic Growth of TiO2 onto Gold Nanorods for Plasmon-Enhanced Hydrogen Production from Water Reduction. J. Am. Chem. Soc. 2016, 138, 1114–1117. [Google Scholar] [PubMed]
- Dutta, S.K.; Mehetor, S.K.; Pradhan, N. Metal semiconductor heterostructures for photocatalytic conversion of light energy. J. Phys. Chem. Lett. 2015, 6, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wu, Z.; Wang, J.; Elzatahry, A.A.; Zhao, D. A Perspective on Mesoporous TiO2 Materials. Chem. Mater. 2014, 26, 287–298. [Google Scholar] [CrossRef]
- Zheng, X.; Kuang, Q.; Yan, K.; Qiu, Y.; Qiu, J.; Yang, S. Mesoporous TiO2 Single Crystals: Facile Shape-, Size-, and Phase-Controlled Growth and Efficient Photocatalytic Performance. ACS Appl. Mater. Interfaces 2013, 5, 11249–11257. [Google Scholar] [CrossRef] [PubMed]
- Jitputti, J.; Suzuki, Y.; Yoshikawa, S. Synthesis of TiO2 nanowires and their photocatalytic activity for hydrogen evolution. Catal. Commun. 2008, 9, 1265–1271. [Google Scholar] [CrossRef]
- Regonini, D.; Teloeken, A.C.; Alves, A.K.; Berutti, F.A.; Gajda-Schrantz, K.; Bergmann, C.P.; Graule, T.; Clemens, F. Electrospun TiO2 Fiber Composite Photoelectrodes for Water Splitting. ACS Appl. Mater. Interfaces 2013, 5, 11747–11755. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.; Huang, B.; Wang, Z.; Ma, X.; Zhang, R.; Zhang, X.; Qin, X.; Dai, Y.; Whangbo, M.-H. Ag6Si2O7: A Silicate Photocatalyst for the Visible Region. Chem. Mater. 2014, 26, 3873–3875. [Google Scholar] [CrossRef]
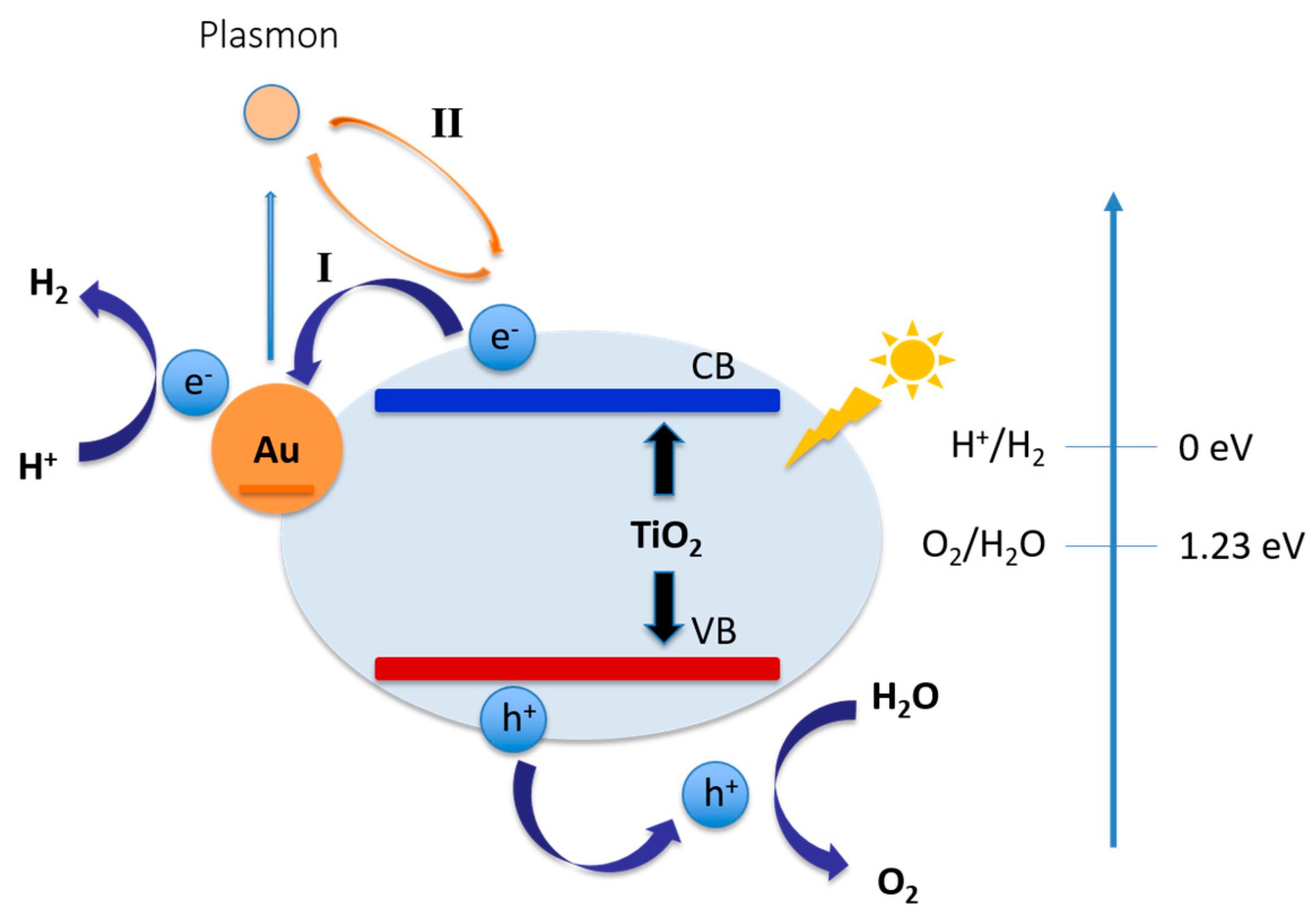
- Cheng, H.; Fuku, K.; Kuwahara, Y.; Mori, K.; Yamashita, H. Harnessing single-active plasmonic nanostructures for enhanced photocatalysis under visible light. J. Mater. Chem. A 2015, 3, 5244–5258. [Google Scholar] [CrossRef]
- Bian, Z.; Tachikawa, T.; Zhang, P.; Fujitsuka, M.; Majima, T. Au/TiO2 Superstructure-Based Plasmonic Photocatalysts Exhibiting Efficient Charge Separation and Unprecedented Activity. J. Am. Chem. Soc. 2013, 136, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Clavero, C. Plasmon-induced hot-electron generation at nanoparticle/metal-oxide interfaces for photovoltaic and photocatalytic devices. Nat. Photonics 2014, 8, 95–103. [Google Scholar] [CrossRef]
- Mubeen, S.; Lee, J.; Liu, D.; Stucky, G.D.; Moskovits, M. Panchromatic Photoproduction of H2 with Surface Plasmons. Nano Lett. 2015, 15, 2132–2136. [Google Scholar] [CrossRef] [PubMed]
- Moskovits, M. The case for plasmon-derived hot carrier devices. Nat. Nanotechnol. 2015, 10, 6–8. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Tang, J.; Chen, H.M.; Liu, B.; Yang, P. A Fully Integrated Nanosystem of Semiconductor Nanowires for Direct Solar Water Splitting. Nano Lett. 2013, 13, 2989–2992. [Google Scholar] [CrossRef] [PubMed]
- Mubeen, S.; Lee, J.; Singh, N.; Krämer, S.; Stucky, G.D.; Moskovits, M. An autonomous photosynthetic device in which all charge carriers derive from surface plasmons. Nat. Nanotechnol. 2013, 8, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Cherqui, C.; Bigelow, N.W.; Duscher, G.; Straney, P.J.; Millstone, J.E.; Masiello, D.J.; Camden, J.P. Spatially Mapping Energy Transfer from Single Plasmonic Particles to Semiconductor Substrates via STEM/EELS. Nano Lett. 2015, 15, 3465–3471. [Google Scholar] [CrossRef] [PubMed]
- Long, R.; Rao, Z.; Mao, K.; Li, Y.; Zhang, C.; Liu, Q.; Wang, C.; Li, Z.-Y.; Wu, X.; Xiong, Y. Efficient Coupling of Solar Energy to Catalytic Hydrogenation by Using Well-Designed Palladium Nanostructures. Angew. Chem. 2015, 127, 2455–2460. [Google Scholar] [CrossRef]
- Wang, X.; Zhu, M.; Fu, W.; Huang, C.; Gu, Q.; Zeng, T.H.; Dai, Y.; Sun, Y. Au nano dumbbells catalyzed the cutting of graphene oxide sheets upon plasmon-enhanced reduction. RSC Adv. 2016, 6, 46218–46225. [Google Scholar] [CrossRef]
- Wang, X.; Long, R.; Liu, D.; Yang, D.; Wang, C.; Xiong, Y. Enhanced full-spectrum water splitting by confining plasmonic Au nanoparticles in N-doped TiO2 bowl nanoarrays. Nano Energy 2016, 24, 87–93. [Google Scholar] [CrossRef]
- Lou, Z.; Fujitsuka, M.; Majima, T. Pt–Au Triangular Nanoprisms with Strong Dipole Plasmon Resonance for Hydrogen Generation Studied by Single-Particle Spectroscopy. ACS Nano 2016, 10, 6299–6305. [Google Scholar] [CrossRef] [PubMed]
- Vigderman, L.; Khanal, B. P.; Zubarev, E. R. Functional Gold Nanorods: Synthesis, Self-Assembly, and Sensing Applications. Adv. Mater. 2012, 24, 4811–4841. [Google Scholar] [CrossRef] [PubMed]
- Skrabalak, S.E.; Au, L.; Li, X.; Xia, Y. Facile synthesis of Ag nanocubes and Au nanocages. Nat. Protoc. 2007, 2, 2182–2190. [Google Scholar] [CrossRef] [PubMed]
- Manjavacas, A.; García de Abajo, F.J. Tunable plasmons in atomically thin gold nanodisks. Nat. Commun. 2014, 5, 3548:1–3548:7. [Google Scholar] [CrossRef] [PubMed]
- Millstone, J.E.; Métraux, G.S.; Mirkin, C.A. Controlling the Edge Length of Gold Nanoprisms via a Seed-Mediated Approach. Adv. Funct. Mater. 2006, 16, 1209–1214. [Google Scholar] [CrossRef]
- Zheng, Z.; Tachikawa, T.; Majima, T. Plasmon-Enhanced Formic Acid Dehydrogenation Using Anisotropic Pd-Au Nanorods Studied at the Single-Particle Level. J. Am. Chem. Soc. 2015, 137, 948–957. [Google Scholar] [CrossRef] [PubMed]
- Madelung, O. Semiconductors: Data Handbook; Springer Berlin Heidelberg: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Schmitz, G.; Gassmann, P.; Franchy, R. A combined scanning tunneling microscopy and electron energy loss spectroscopy study on the formation of thin, well-ordered β-Ga2O3 films on CoGa(001). J. Appl. Phys. 1998, 83, 2533–2538. [Google Scholar] [CrossRef]
- Perevalov, T.V.; Shaposhnikov, A.V.; Gritsenko, V.A.; Wong, H.; Han, J.H.; Kim, C.W. Electronic structure of α-Al2O3: Ab initio simulations and comparison with experiment. JETP Lett. 2007, 85, 165–168. [Google Scholar] [CrossRef]
- Schwertmann, U.; Cornell, R. The Iron Oxides. Structure, Properties, Reactions Occurrences and Uses; VCH: Weinheim, Germany, 1996. [Google Scholar]
- Kim, K.J.; Park, Y.R. Optical investigation of charge-transfer transitions in spinel Co3O4. Solid State Commun. 2003, 127, 25–28. [Google Scholar] [CrossRef]
- Walsh, A.; Watson, G.W. The origin of the stereochemically active Pb(II) lone pair: DFT calculations on PbO and PbS. J. Solid State Chem. 2005, 178, 1422–1428. [Google Scholar] [CrossRef]
- Walsh, A.; Watson, G.W. Electronic structures of rocksalt, litharge, and herzenbergite SnO by density functional theory. Phys. Rev. B 2004, 70, 235114:1–235114:7. [Google Scholar] [CrossRef]
- Geurts, J.; Rau, S.; Richter, W.; Schmitte, F.J. SnO films and their oxidation to SnO2: Raman scattering, IR reflectivity and X-ray diffraction studies. Thin Solid Films 1984, 121, 217–225. [Google Scholar] [CrossRef]
- Dolocan, V. Some electrical properties of Bi2O3 thin films. Phys. Status Solidi 1978, 45, K155–K157. [Google Scholar] [CrossRef]
- Kong, L.; Chen, H.; Hua, W.; Zhang, S.; Chen, J. Mesoporous bismuth titanate with visible-light photocatalytic activity. Chem. Commun. 2008, 40, 4977–4979. [Google Scholar] [CrossRef] [PubMed]
- Hosogi, Y.; Shimodaira, Y.; Kato, H.; Kobayashi, H.; Kudo, A. Role of Sn2+ in the Band Structure of SnM2O6 and Sn2M2O7 (M = Nb and Ta) and Their Photocatalytic Properties. Chem. Mater. 2008, 20, 1299–1307. [Google Scholar] [CrossRef]
- Kisch, H. Visible Light Induced Photoelectrochemical Properties of n-BiVO4 and n-BiVO4/p-Co3O4. J. Phys. Chem. C 2008, 112, 548–554. [Google Scholar]
- Sayama, K.; Nomura, A.; Zou, Z.; Abe, R.; Abe, Y.; Arakawa, H. Photoelectrochemical decomposition of water on nanocrystalline BiVO4 film electrodes under visible light. Chem. Commun. 2003, 23, 2908–2909. [Google Scholar] [CrossRef]
- Mishra, M.; Chun, D.-M. α-Fe2O3 as a photocatalytic material: A review. Appl. Catal. A Gen. 2015, 498, 126–141. [Google Scholar] [CrossRef]
- Haghighat, S.; Dawlaty, J.M. Continuous representation of the proton and electron kinetic parameters in the pH-potential space for water oxidation on hematite. J. Phys. Chem. C 2015, 119, 6619–6625. [Google Scholar] [CrossRef]
- Morales-Guio, C.G.; Mayer, M.T.; Yella, A.; Tilley, S.D.; Grätzel, M.; Hu, X. An Optically Transparent Iron Nickel Oxide Catalyst for Solar Water Splitting. J. Am. Chem. Soc. 2015, 137, 9927–9936. [Google Scholar] [CrossRef] [PubMed]
- Amer, A.W.; El-Sayed, M.A.; Allam, N.K. Tuning the Photoactivity of Zirconia Nanotubes-Based Photoanodes via Ultra-Thin Layers of ZrN: An Effective Approach Towards Visible Light-Water Splitting. J. Phys. Chem. C 2016, 120, 7025–7032. [Google Scholar] [CrossRef]
- Sivula, K.; Le Formal, F.; Gratzel, M. WO3-Fe2O3 photoanodes for water splitting: A host scaffold, guest absorber approach. Chem. Mater. 2009, 21, 2862–2867. [Google Scholar] [CrossRef]
- Zhan, X.; Wang, Z.; Wang, F.; Cheng, Z.; Xu, K.; Wang, Q.; Safdar, M.; He, J. Efficient CoO nanowire array photocatalysts for H2 generation. Appl. Phys. Lett. 2014, 105, 153903:1–153903:5. [Google Scholar] [CrossRef]
- Jasieniak, J.; Califano, M.; Watkins, S.E. Size-Dependent Valence and Conduction Band-Edge Energies of Semiconductor Nanocrystals. ACS Nano 2011, 5, 5888–5902. [Google Scholar] [CrossRef] [PubMed]
- Asai, R.; Nemoto, H.; Jia, Q.; Saito, K.; Iwase, A.; Kudo, A. A visible light responsive rhodium and antimony-codoped SrTiO3 powdered photocatalyst loaded with an IrO2 cocatalyst for solar water splitting. Chem. Commun. 2014, 50, 2543–2546. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, K.; Jia, Q.; Kudo, A.; Onishi, H. Time-Resolved Infrared Absorption Study of SrTiO3 Photocatalysts Codoped with Rhodium and Antimony. J. Phys. Chem. C 2013, 117, 19101–19106. [Google Scholar] [CrossRef]
- Takata, T.; Domen, K. Defect engineering of photocatalysts by doping of aliovalent metal cations for efficient water splitting. J. Phys. Chem. C 2009, 113, 19386–19388. [Google Scholar] [CrossRef]
- Kang, H.W.; Lim, S.N.; Song, D.; Park, S. Bin Organic-inorganic composite of g-C3N4–SrTiO3:Rh photocatalyst for improved H2 evolution under visible light irradiation. Int. J. Hydrog. Energy 2012, 37, 11602–11610. [Google Scholar] [CrossRef]
- Lü, X.; Ding, S.; Lin, T.; Mou, X.; Hong, Z.; Huang, F. Ta2O5 nanowires: A novel synthetic method and their solar energy utilization. Dalton Trans. 2012, 41, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Tao, C.; Xu, L.; Guan, J. Well-dispersed mesoporous Ta2O5 submicrospheres: Enhanced photocatalytic activity by tuning heating rate at calcination. Chem. Eng. J. 2013, 229, 371–377. [Google Scholar] [CrossRef]
- Mao, L.; Zhu, S.; Ma, J.; Shi, D.; Chen, Y.; Chen, Z.; Yin, C.; Li, Y.; Zhang, D. Superior H2 production by hydrophilic ultrafine Ta2O5 engineered covalently on graphene. Nanotechnology 2014, 25, 215401:1–215401:9. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Lin, T.; Cui, H.; Zhao, W.; Zhang, H.; Huang, F. Gray Ta2O5 Nanowires with Greatly Enhanced Photocatalytic Performance. ACS Appl. Mater. Interfaces 2016, 8, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Sun, J.Z.; Hong, J.; Li, H.Y.; Chen, H.Z.; Wang, M. Carbon Nanotube/CdS Core-Shell Nanowires Prepared by a Simple Room-Temperature Chemical Reduction Method. Adv. Mater. 2004, 16, 84–87. [Google Scholar] [CrossRef]
- Huang, Z.-F.; Song, J.; Li, K.; Tahir, M.; Wang, Y.-T.; Pan, L.; Wang, L.; Zhang, X.; Zou, J.-J. Hollow Cobalt-Based Bimetallic Sulfide Polyhedra for Efficient All-pH-Value Electrochemical and Photocatalytic Hydrogen Evolution. J. Am. Chem. Soc. 2016, 138, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Ran, J.; Zhang, J.; Yu, J.; Jaroniec, M.; Qiao, S.Z. Earth-abundant cocatalysts for semiconductor-based photocatalytic water splitting. Chem. Soc. Rev. 2014, 43, 7787–7812. [Google Scholar] [CrossRef] [PubMed]
- Reiss, P.; Protière, M.; Li, L. Core/shell semiconductor nanocrystals. Small 2009, 5, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Yu, J.; Jaroniec, M.; Gong, J.R. Noble Metal-Free Reduced Graphene Oxide-ZnxCd1–xS Nanocomposite with Enhanced Solar Photocatalytic H2-Production Performance. Nano Lett. 2012, 12, 4584–4589. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Meng, H.; Zhou, P.; Zheng, Y.; Wang, J.; Yu, J.; Gong, J. Zn1–xCdxS solid solutions with controlled bandgap and enhanced visible-light photocatalytic H2-production activity. ACS Catal. 2013, 3, 882–889. [Google Scholar] [CrossRef]
- Pan, Y.X.; Zhuang, H.Q.; Hong, J.D.; Fang, Z.; Liu, H.; Liu, B.; Huang, Y.Z.; Xu, R. Cadmium Sulfide Quantum Dots Supported on Gallium and Indium Oxide for Visible-Light-Driven Hydrogen Evolution from Water. ChemSusChem 2014, 7, 2537–2544. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, G.; Lu, G.Q.; Cheng, H.M. Stable photocatalytic hydrogen evolution from water over ZnO-CdS core-shell nanorods. Int. J. Hydrog. Energy 2010, 35, 8199–8205. [Google Scholar] [CrossRef]
- Hou, J.; Wang, Z.; Kan, W.; Jiao, S.; Zhu, H.; Kumar, R.V. Efficient visible-light-driven photocatalytic hydrogen production using CdS@TaON core-shell composites coupled with graphene oxide nanosheets. J. Mater. Chem. 2012, 22, 7291–7299. [Google Scholar] [CrossRef]
- Bai, S.; Shen, X. Graphene–inorganic nanocomposites. RSC Adv. 2012, 2, 64–98. [Google Scholar] [CrossRef]
- Cheng, Z.; Wang, Z.; Shifa, T.A.; Wang, F.; Zhan, X.; Xu, K.; Liu, Q.; He, J. Au plasmonics in a WS2-Au-CuInS2 photocatalyst for significantly enhanced hydrogen generation. Appl. Phys. Lett. 2015, 107, 223902:1–223902:5. [Google Scholar] [CrossRef]
- Kato, H.; Asakura, K.; Kudo, A. Highly efficient water splitting into H2 and O2 over lanthanum-doped NaTaO3 photocatalysts with high crystallinity and surface nanostructure. J. Am. Chem. Soc. 2003, 125, 3082–3089. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Takata, T.; Hara, M.; Saito, N.; Inoue, Y.; Kobayashi, H.; Domen, K. GaN:ZnO solid solution as a photocatalyst for visible-light-driven overall water splitting. J. Am. Chem. Soc. 2005, 127, 8286–8287. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Teramura, K.; Domen, K. Effect of post-calcination on photocatalytic activity of (Ga1-xZnx)(N1-xOx) solid solution for overall water splitting under visible light. J. Catal. 2008, 254, 198–204. [Google Scholar] [CrossRef]
- Domen, K. Photocatalytic Water Splitting Using Oxynitride and Nitride Semiconductor Powders for Production of Solar Hydrogen. Interphase 2013, 22, 57–62. [Google Scholar]
- Zhen, C.; Wang, L.; Liu, G.; Lu, G.Q.M.; Cheng, H.-M. Template-free synthesis of Ta3N5 nanorod arrays for efficient photoelectrochemical water splitting. Chem. Commun. 2013, 49, 3019–3021. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.; Takata, T.; Nakabayashi, M.; Hisatomi, T.; Shibata, N.; Minegishi, T.; Domen, K. Mg-Zr Cosubstituted Ta3N5 Photoanode for Lower-Onset-Potential Solar-Driven Photoelectrochemical Water Splitting. J. Am. Chem. Soc. 2015, 137, 12780–12783. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Maeda, K.; Thomas, A.; Takanabe, K.; Xin, G.; Carlsson, J.M.; Domen, K.; Antonietti, M. A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat. Mater. 2009, 8, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Zhang, M.; Ye, X.; Qiu, X.; Lin, S.; Wang, X. Iodine modified carbon nitride semiconductors as visible light photocatalysts for hydrogen evolution. Adv. Mater. 2014, 26, 805–809. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Chen, X.; Antonietti, M.; Wang, X. Synthesis of transition metal-modified carbon nitride polymers for selective hydrocarbon oxidation. ChemSusChem 2011, 4, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, Y.; Wang, X. Two-dimensional covalent carbon nitride nanosheets: Synthesis, functionalization, and applications. Energy Environ. Sci. 2015, 3092, 3092–3108. [Google Scholar] [CrossRef]
- Martin, D.J.; Reardon, P.J.T.; Moniz, S.J.A.; Tang, J. Visible Light-Driven Pure Water Splitting by a Nature-Inspired Organic Semiconductor-Based System. J. Am. Chem. Soc. 2014, 136, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.-P.; Wang, L.-C.; Luo, J.; Nie, Y.-C.; Xing, Q.-J.; Luo, X.-B.; Du, H.-M.; Luo, S.-L.; Suib, S.L. Synthesis and efficient visible light photocatalytic H2 evolution of a metal-free g-C3N4/graphene quantum dots hybrid photocatalyst. Appl. Catal. B Environ. 2016, 193, 103–109. [Google Scholar] [CrossRef]
- Etacheri, V.; Di Valentin, C.; Schneider, J.; Bahnemann, D.; Pillai, S.C. Visible-Light Activation of TiO2 Photocatalysts: Advances in Theory and Experiments. J. Photochem. Photobiol. C Photochem. Rev. 2015, 25, 1–29. [Google Scholar] [CrossRef]
- Nazeeruddin, M.K.; De Angelis, F.; Fantacci, S.; Selloni, A.; Viscardi, G.; Liska, P.; Ito, S.; Takeru, B.; Grätzel, M. Combined experimental and DFT-TDDFT computational study of photoelectrochemical cell ruthenium sensitizers. J. Am. Chem. Soc. 2005, 127, 16835–16847. [Google Scholar] [CrossRef] [PubMed]
- Nayak, P.K.; Periasamy, N. Calculation of electron affinity, ionization potential, transport gap, optical band gap and exciton binding energy of organic solids using “solvation” model and DFT. Org. Electron. 2009, 10, 1396–1400. [Google Scholar] [CrossRef]
- Chai, S.; Wen, S.-H.; Huang, J.-D.; Han, K.-L. Density functional theory study on electron and hole transport properties of organic pentacene derivatives with electron-withdrawing substituent. J. Comput. Chem. 2011, 32, 3218–3225. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, F.; Fantacci, S.; Selloni, A. Alignment of the dye’s molecular levels with the TiO2 band edges in dye-sensitized solar cells: A DFT-TDDFT study. Nanotechnology 2008, 19, 424002–424008. [Google Scholar] [CrossRef] [PubMed]
- Wendt, S.; Seitsonen, A.; Kim, Y.; Knapp, M.; Idriss, H.; Over, H. Complex redox chemistry on the RuO2 (100) surface: Experiment and theory. Surf. Sci. 2002, 505, 137–152. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, H.; Tomov, I.V.; Wolfsberg, M.; Ding, X.; Rentzepis, P.M. Transient structures and kinetics of the ferrioxalate redox reaction studied by time-resolved EXAFS, optical spectroscopy, and DFT. J. Phys. Chem. A 2007, 111, 9326–9335. [Google Scholar] [CrossRef] [PubMed]
- Kohn, W.; Sham, L.J. Self-Consistent Equations Including Exchange and Correlation Effects. Phys. Rev. 1965, 140, A1133–A1138. [Google Scholar] [CrossRef]
- Hohenberg, P. Inhomogeneous Electron Gas. Phys. Rev. 1964, 136, B864–B871. [Google Scholar] [CrossRef]
- Burke, K. Perspective on density functional theory. J. Chem. Phys. 2012, 136, 150901. [Google Scholar] [CrossRef] [PubMed]
- Lejaeghere, K.; Bihlmayer, G.; Bjorkman, T.; Blaha, P.; Blugel, S.; Blum, V.; Caliste, D.; Castelli, I.E.; Clark, S.J.; Dal Corso, A.; et al. Reproducibility in density functional theory calculations of solids. Science 2016, 351, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Waroquiers, D.; Lherbier, A.; Miglio, A.; Stankovski, M.; Poncé, S.; Oliveira, M.J.T.; Giantomassi, M.; Rignanese, G.-M.; Gonze, X. Band widths and gaps from the Tran-Blaha functional: Comparison with many-body perturbation theory. Phys. Rev. B 2013, 87, 075121:1–075121:15. [Google Scholar] [CrossRef]
- Hedin, L.; Lundqvist, S. Effects of Electron-Electron and Electron-Phonon Interactions on the One-Electron States of Solids. Solid State Phys. 1970, 23. [Google Scholar] [CrossRef]
- Aulbur, W.G.; Jönsson, L.; Wilkins, J.W. Quasiparticle Calculations in Solids. Solid State Phys. 1999, 54. [Google Scholar] [CrossRef]
- Becke, A.D.; Johnson, E.R. A simple effective potential for exchange. J. Chem. Phys. 2006, 124, 221101:1–221101:4. [Google Scholar] [CrossRef] [PubMed]
- Tran, F.; Blaha, P. Accurate band gaps of semiconductors and insulators with a semilocal exchange-correlation potential. Phys. Rev. Lett. 2009, 102, 226401:1–226401:4. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Walther, C.F.J.; Kuc, A.; Heine, T. Density Functional Theory and Beyond for Band-Gap Screening: Performance for Transition-Metal Oxides and Dichalcogenides. J. Chem. Theory Comput. 2013, 9, 2950–2958. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Wang, D.; Zhou, X.; Li, C. A theoretical study on the mechanism of photocatalytic oxygen evolution on BiVO4 in aqueous solution. Chemistry 2013, 19, 1320–1326. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zhang, F.; Wang, D.; Yang, J.; Li, M.; Zhu, J.; Zhou, X.; Han, H.; Li, C. Spatial separation of photogenerated electrons and holes among {010} and {110} crystal facets of BiVO4. Nat. Commun. 2013, 4, 1432–1438. [Google Scholar] [CrossRef] [PubMed]
- Xiang, C.; Haber, J.; Marcin, M.; Mitrovic, S.; Jin, J.; Gregoire, J.M. Mapping Quantum Yield for (Fe-Zn-Sn-Ti)Ox Photoabsorbers Using a High Throughput Photoelectrochemical Screening System. ACS Comb. Sci. 2014, 16, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Butler, K.T.; Kumagai, Y.; Oba, F.; Walsh, A. Screening procedure for structurally and electronically matched contact layers for high-performance solar cells: Hybrid perovskites. J. Mater. Chem. C 2016, 4, 1149–1158. [Google Scholar] [CrossRef]
- Wu, Y.; Lazic, P.; Hautier, G.; Persson, K.; Ceder, G. First principles high throughput screening of oxynitrides for water-splitting photocatalysts. Energy Environ. Sci. 2013, 6, 157–168. [Google Scholar] [CrossRef]
- Miró, P.; Audiffred, M.; Heine, T. An atlas of two-dimensional materials. Chem. Soc. Rev. 2014, 43, 6537–6554. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Cheng, H.; Takeda, O.; Zhu, H. Unique 3D heterojunction photoanode design to harness charge transfer for efficient and stable photoelectrochemical water splitting. Energy Environ. Sci. 2015, 8, 1348–1357. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, Y.; Liu, K.; Guo, L.; Yuan, Q.; Dong, B. Multichannel-Improved Charge-Carrier Dynamics in Well-Designed Hetero-nanostructural Plasmonic Photocatalysts toward Highly Efficient Solar-to-Fuels Conversion. Adv. Mater. 2015, 27, 5906–5914. [Google Scholar] [CrossRef] [PubMed]
- Serpone, N.; Emeline, A.V. Semiconductor photocatalysis—Past, present, and future outlook. J. Phys. Chem. Lett. 2012, 3, 673–677. [Google Scholar] [CrossRef] [PubMed]

| Photocatalysts | Band Gap (eV) | Illumination | Hydrogen Production | Ref. |
|---|---|---|---|---|
| Pt, Cr, Ta Doped TiO2 | N/A | Visible light (>420 nm) | 11.7 μmol·h−1·g−1 | [71] |
| Cu-Ga-In-S/TiO2 | N/A | 300 W Xe arc lamp (385–740 nm) | 50.6 μmol·h−1 | [72] |
| 1 wt.%Pt/C-HS-TiO2 | 2.94 | Visible light | 5713.6 μmol·h−1·g−1 | [73] |
| Platinized sub-10 nm rutile TiO2 (1 wt.% Pt) | 2.7–2.9 | Xe lamp (PLS-SXE, 300–2500 nm) with (UVREF: 320–400 nm, ca. 83 mW·cm−2; UVCUT400: 400–780 nm, ca. 80 mW·cm−2) | 932 μmol·h−1·g−1 visible light 1954 μmol·h−1·g−1 simulated solar light | [74] |
| Rh- and La-codoped SrTiO3 | N/A | 300 W Xe lamp fitted with a cutoff filter (λ > 420 nm) | 84 μmol·h−1 | [75] |
| Cu1.94S-ZnxCd1−xS (0 ≤ x ≤ 1) | 2.57−3.88 | visible-light irradiation (λ > 420 nm) | 7735 μmol·h−1·g−1 | [76] |
| MoS2/Co2O3/poly(heptazine imide) | N/A | visible light irradiation | 0.67 μmol·h−1 | [77] |
| Bi4NbO8Cl | 2.4 | visible light | 6.25 μmol·h−1 | [78] |
| CdS nanorod/ ZnS nanoparticle | N/A | visible light irradiation (>420 nm) | 239,000 μmol·h−1·g−1 | [79] |
| Ni/CdS/g-C3N4 | N/A | 300 W Xe lamp (≥420 nm) | 1258.7 μmol·h−1·g−1 | [80] |
| CdS/WS/graphene | N/A | visible light irradiation (>420 nm) | 1842 μmol·h−1·g−1 | [81] |
| V-doped TiO2/RGO | N/A | visible light irradiation | 160 μmol·h−1 | [82] |
| Pt/g-C3N4 Conjugated Polymers | 2.56 | visible light irradiation (>420 nm) | 1.2 μmol·h−1 | [83] |
| Au–TiO2 Nanohybrids | N/A | Vis-NIR irradiation (>420 nm ) | 647,000 μmol·h−1·g−1 | [84] |
| SrTiO3:La,Rh/Au/BiVO4:Mo | N/A | 300 W Xe lamp fitted with a cutoff filter (λ > 420 nm) | 90 μmol·h−1 | [85] |
| CoOx-B/TiO2-TaON | N/A | 150 W Xe Lamp arc (>400 nm) | 40 μmol·h−1 | [86] |
| MoS2/CuInS2 | N/A | 300 W Xe lamp fitted with a cutoff filter (λ > 420 nm) | 202 μmol·h−1·g−1 | [87] |
| Copper-Organic Framework; H2PtCl6 | 2.1 | UV-Visible irradiation | 30 μmol·h−1·g−1 | [88] |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jafari, T.; Moharreri, E.; Amin, A.S.; Miao, R.; Song, W.; Suib, S.L. Photocatalytic Water Splitting—The Untamed Dream: A Review of Recent Advances. Molecules 2016, 21, 900. https://doi.org/10.3390/molecules21070900
Jafari T, Moharreri E, Amin AS, Miao R, Song W, Suib SL. Photocatalytic Water Splitting—The Untamed Dream: A Review of Recent Advances. Molecules. 2016; 21(7):900. https://doi.org/10.3390/molecules21070900
Chicago/Turabian StyleJafari, Tahereh, Ehsan Moharreri, Alireza Shirazi Amin, Ran Miao, Wenqiao Song, and Steven L. Suib. 2016. "Photocatalytic Water Splitting—The Untamed Dream: A Review of Recent Advances" Molecules 21, no. 7: 900. https://doi.org/10.3390/molecules21070900
APA StyleJafari, T., Moharreri, E., Amin, A. S., Miao, R., Song, W., & Suib, S. L. (2016). Photocatalytic Water Splitting—The Untamed Dream: A Review of Recent Advances. Molecules, 21(7), 900. https://doi.org/10.3390/molecules21070900






