Abstract
Capsaicin, the pungent ingredient of the hot chili pepper, is known to act on the transient receptor potential cation channel vanilloid subfamily member 1 (TRPV1). TRPV1 is involved in somatic and visceral peripheral inflammation, in the modulation of nociceptive inputs to spinal cord and brain stem centers, as well as the integration of diverse painful stimuli. In this review, we first describe the chemical and pharmacological properties of capsaicin and its derivatives in relation to their analgesic properties. We then consider the biochemical and functional characteristics of TRPV1, focusing on its distribution and biological effects within the somatosensory and viscerosensory nociceptive systems. Finally, we discuss the use of capsaicin as an agonist of TRPV1 to model acute inflammation in slices and other ex vivo preparations.
1. Introduction and General Concepts
1.1. Chemical Features of Capsaicin
Capsaicin (trans-8-methyl-N-vanillyl-6-nonenamide—C18H27NO3) is a naturally occurring substance derived from the plants of the genus Capsicum, family Solanaceae. Capsaicin is a vanilloid as it contains a vanillyl group in its formula. Vanilloids belong to a class of organic chemicals referred to as protoalkaloids, i.e., alkaloids where nitrogen is located in the side chain. Alkaloids are a wide and rather heterogeneous group of compounds that are generally made of carbon, hydrogen and nitrogen. In addition to these, alkaloids often contain other elements among which is oxygen, as is the case for capsaicin. The first isolation of the vanilloid from paprika and cayenne dates back to 1876 and was reported by Thresh [1]. The first study on capsaicin structure dates back to 1920 [2].
The commercial production of capsaicin from natural sources primarily involves its isolation from Capsicum spp. Capsaicin can also be produced by the reaction of vanillylamine with 7-methyloct-5-ene-1-carboxylic acid chloride. The vanilloid appears in the form of a highly volatile, pungent, hydrophobic, colorless and odorless white crystalline powder. Once absorbed by the body, capsaicin is likely metabolized by dehydrogenation, giving rise to specific macrocyclic, -diene and -imide metabolites.
1.2. Natural Sources of Capsaicin
In the world, there are five known domesticated varieties of Capsicum spp.: C. annuum, C. frutescens, C. chinense, C. baccatum and C. pubescens. C. annuum (also known as red chili, paprika, gendot, curly chili) and C. frutescens (rawit) are very similar, to the point that some authors do not consider the two as different species [3]. The fruit of all these plants has a hot taste that derives from its content in capsaicinoid compounds: a group of amide acids from vanilinamine and fatty acid chain branched at C9 and C11. Capsaicinoids in Capsicum spp. for the most contain capsaicin, and, in lesser quantities, dihydrocapsaicin, homocapsaicin and homodihydrocapsaicin. Analysis of the levels of capsaicin in various Capsicum fruits showed that green paprika, yellow paprika and red paprika contained no capsaicin, while chili tanjung, red chili, red gendot, green gendot, green curly, japlak rawit, red curly, red rawit and green rawit (cayenne) contained 0.38; 0.83; 0.87; 0.88; 1.05; 1.09; 1.14; 1.85 and 2.11% capsaicin (w/w), respectively [4].
1.3. Cloning, General Distribution, Functional Properties and Biological Effects of the Capsaicin Receptor in Mammals
1.3.1. Cloning and General Distribution of TRPV1
The capsaicin receptor, named transient receptor potential vanilloid 1 receptor (TRPV1), was cloned in 1997 from rat dorsal root ganglia (DRGs) using a functional screening strategy for isolating candidate complementary DNA (cDNA) clones [5]. This newly cloned cDNA was initially named VR1, for vanilloid receptor subtype 1. Later, VR1 was identified to be a member of the transient receptor potential (TRP) family of cation channels and the nomenclature TRPV1 was adopted to denote this association. To date, TRPV1 has been cloned from human, guinea pig, rabbit, mouse and porcine tissues. Its distribution was mainly investigated in tissues and organs from human, rat and mouse, but also several other mammals among which are the other aforementioned species [6]. By reverse transcription-polymerase chain reaction (RT-PCR), TRPV1 was localized to human DRGs, brain, kidney, pancreas, testis, uterus, spleen, stomach, small intestine, lung and liver [7]. In rats, with an array of techniques including in situ hybridization, northern blotting, RT-PCR and immunocytochemistry (ICC), the receptor was localized in numerous areas of the central nervous system (CNS) including the cerebral cortex, striatum, hippocampus, central amygdala, thalamus, hypothalamus, cerebellum, locus cerulean, cochlear nuclei, spinal nucleus of the trigeminal nerve (SNTN), inferior olive and spinal cord [8]. In the peripheral nervous system (PNS), TRPV1 was detected in rat trigeminal ganglion (TG) and DRGs. Other rat organs expressing the receptor were the kidney, pancreas, placenta and urinary bladder [5,7,9]. In mice, TRPV1 was localized to similar districts of the CNS and PNS than in rats [10,11,12,13,14]; and a subset of smooth muscle cells in small arteries [15]. In the above localizations and species, TRPV1 was not only detected in intramural nerve fibers and plexuses, providing the visceral innervation to the organs and tissues listed above, but also in the mucosal epithelial cells. The latter are not the only non-neural cells expressing the receptor, as some cells of the immune system, e.g., the T-cells and the mast cells; the keratinocytes of the epidermis; the cells of inner root sheet and the infundibulum of hair follicles; differentiated sebocytes; the cells of sweat gland ducts and the secretory portion of eccrine sweat glands; and the vascular endothelium also express TRPV1 [16].
1.3.2. Functional Properties and Biological Effects of TRPV1
Initial studies on isolated cells demonstrated that capsaicin and other natural substances, as well as some physical activators and protons, activated TRPV1. Functionally, capsaicin, resinferatoxin (RTX) and heat activated Human Embryonic Kidney 293 (HEK 293) cells transfected with human or rat TRPV1 vector [5,7]. Mouse DRG neurons were activated by the same substances in patch-clamp whole- or single-cell recordings [17]. Capsaicin and acidic pH in Xenopus laevis oocytes injected with the human TRPV1 cDNA [7] effectively opened the receptor channel in two-electrode voltage clamp experiments. In addition, intracellular Ca2+ imaging provided further evidence that the receptor was activated by capsaicin, anandamide, olvanil, RTX and pH in HEK 293 cells transfected with rat [18], mouse [19] or human [20] TRPV1 cDNA. In neurons, cation (Ca2+) influx through TRPV1 causes membrane depolarization, leading to the activation of voltage-gated sodium channels and the generation of an action potential. It was very recently reported that the capsaicin-evoked action potentially follows a physical interaction between TRPV1 and anoctamin 1, a calcium-activated chloride channel, resulting from the entry of Ca2+ through the TRPV1 pore and that such interaction is relevant for the enhancement of nociception [21].
Broadly speaking and in the more complex in vivo or ex vivo context, TRPV1 has been linked to thermo-sensation (heat), autonomic thermoregulation, nociception, food intake regulation and multiple functions in the gastrointestinal (GI) tract [22]. Specifically in CNS, the receptor has also been involved in growth cone guidance, long-term depression, endocannabinoid signaling and osmosensing, the latter by a particular TRPV1 variant [16].
Notably, TRPV1 is also up regulated in several human pathological conditions including vulvodynia [23], GI inflammation, Crohn’s disease and ulcerative colitis [24,25].
In the light of the aforementioned observations, it is not surprising that capsaicin has been clinically used and proven to be of some benefit in obesity, cardiovascular and GI pathologies, various types of tumors, neurogenic bladder and certain dermatologic conditions, although many of these pharmacological effects appeared to be TRPV1-independent [22].
1.3.3. Other Molecular Targets of Capsaicin
Some of the TRPV1-independent pharmacological effects have led a debate on the possible existence of other receptor targets of capsaicin. There is contrasting evidence regarding the possibility that capsaicin may be an inhibitor of signal transducer and activator of transcription 3 (STAT3), although the lowest active dose (50 μM) necessary to inhibit STAT3 is significantly higher than the concentration (1–5 μM) usually required to stimulate TRPV1 [26].
It seems also possible that—at least under certain conditions—neurokinin 1 (NK1) and neurokinin 2 receptor-mediated mechanisms are involved in the induction of phosphorylated extracellular signal-regulated kinase (pERK) after stimulation of primary sensory neurons (PSNs) with capsaicin, an effect not solely directly linked to the binding of the vanilloid to TRPV1 channels [27]. However, as PSNs release substance P in response to capsaicin challenge, such a possibility still needs to be confirmed in full.
Very recently, additional potential targets of capsaicin were predicted by reverse docking and confirmed via chemical-protein interactome and molecular docking. By this approach capsaicin was identified as an inhibitor of carbonic anhydrase 2 [28].
1.4. Nociception and Pain
Nociception is the encoding of a noxious stimulus i.e., an actual or potential tissue damaging event [4] and its transduction into electric signals. Noxious stimuli are detected by nerve endings found throughout the body and originating from the PSNs, which represent the first element of a polyneuronal chain leading to the perception of pain. A class of PSNs, the nociceptors, respond to nociceptive stimuli but under certain conditions can be activated also by innocuous forms of the same stimulus e.g., in the case of heat and cold nociceptors. Nociceptors are polymodal receptors as they respond to stimuli of a heterogeneous nature: mechanical (e.g., high pressure), thermal (too high or too low temperatures) and chemical.
Once activated by an adequate stimulus, nociceptors in skin, muscles, joints or viscera generate a nerve signal (action potential) that is ultimately transferred to the somatosensory cortex, the parieto-insular cortex and to the anterior cingulate cortex where the sensation of pain is perceived. Nociceptive stimuli conveyed from somatic and visceral organs to cortical centers follow different routes. In particular, the trigeminal and spinal nerves solely provide somatic sensory fibers to the skin and the organs of locomotion, whereas most viscera have two distinct types of sensory nerves originating from the sympathetic (via the white communicating branches of the thoracic and lumbar nerves) and parasympathetic (via certain cranial nerves) divisions of the autonomic nervous system.
1.5. Basic Organization of Somatic and Visceral Pain Pathways
1.5.1. Somatic Pain Pathways
Somatic pain pathways collect stimuli from the skin, muscles, joints, ligaments and bones. With the exception of the head and the proximal regions of the neck which are innervated by the peripheral projections of the trigeminal PSNs, nociceptive stimuli from all other parts of the body are encoded by the PSNs of the DRGs. The main somatic pain pathways are the trigeminothalamic and the spinothalamic pathways. They are critical for the sensory discriminative aspects of pain perception. Both consist of a polysynaptic chain of three main neurons, often referred to as first, second and third order somatic sensory neurons. First order sensory neurons (the PSNs) are located in the TG, the proximal ganglia of the glossopharyngeal and vagus nerves, or the DRGs. Second order sensory neurons lie in the spinal cord dorsal horn, or the SNTN. These projection neurons give rise to axons that cross the midline and travel ascending all along the spinal cord and brainstem to reach the thalamus. Finally, third order sensory neurons reside in the ventral postero-lateral nucleus of the thalamus and eventually send their axons to the cerebral cortex (Figure 1). Modulation of nociceptive signals can occur all along this polyneuronal chain, primarily at the level of the substantia gelatinosa of the spinal cord dorsal horn or the SNTN and by several descending pathways that exert inhibitory or facilitatory effects onto the trigeminothalamic or the spinothalamic neurons.
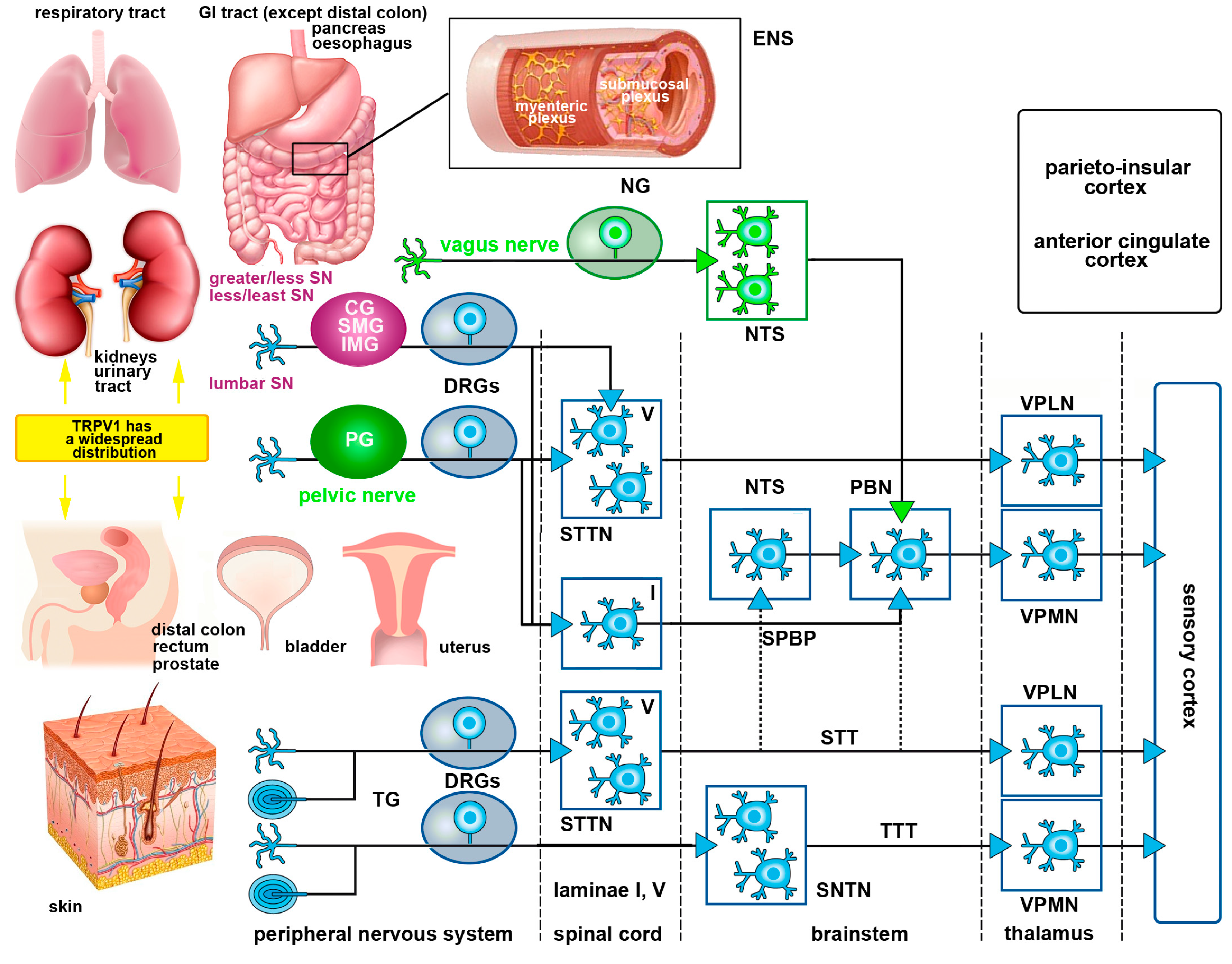
Figure 1.
Illustration depicting TRPV1 receptor distribution in body organs and the main visceral and somatic pain pathways. TRPV1 is found in several body organs and when its expression increases, it contributes to the development of visceral and somatic pain. Afferent fibers innervating the viscera project to the CNS following the course of autonomic sympathetic and parasympathetic nerves. Afferent sympathetic fibers originating from the thoracolumbar DRGs follow the hypogastric, lumbar and splanchnic nerves (SN) after traversing the sympathetic prevertebral (celiac ganglion—CG, superior mesenteric ganglion- SMG and inferior mesenteric ganglia—IMG) and paravertebral ganglia (in magenta). Ascending projections from lamina I neurons in the spinal cord travel along the spinoparabrachial pathway (SPBP) to the parabrachial nucleus (PBN), whereas projections from deep dorsal horn neurons (STTN) travel along the spinothalamic tract (STT) to thalamic nuclei (VPMN and VPLN). The parasympathetic sensory innervation (in green) follows the vagus and pelvic nerve that terminate in the brainstem and lumbosacral cord, respectively. The gastrointestinal tract also has its own autonomic intrinsic nervous system—the enteric nervous system (ENS) — constituted by the submucosal (Meissner’s) and myenteric (Auerbach’s) plexuses. Enteric plexuses play a key role for communication with the autonomic extrinsic nervous system in several GI functions. Nociceptive somatic inputs from all the parts of the body, except the head, are transmitted to spinothalamic projection neurons in the dorsal horn of the spinal cord. These neurons in turn, reach the neurons in the ventro-postero lateral nucleus of the thalamus (VPLN) through the STT. Nociceptive somatic inputs (for simplicity, only the skin is depicted but these inputs also derive from the muscles, tendons, bones and joints) from the head are relayed to the spinal nucleus of the trigeminal nerve (SNTN) and then, along the trigeminothalamic fibers to the ventro-postero medial nucleus of the thalamus (VPMN). Finally, nociceptive input is transferred to the sensory cortex where it is perceived as pain. Affective, emotional and autonomic aspects of pain are processed in other cortical areas (black).
Other ascending pathways are also important for the general dimension of pain and pain control, as they convey stimuli related to motivational and cognitive aspects (spinoreticular tract and spinoparabrachial tract), motor responses and affectivity (spinomesencephalic tract and spinoparabrachial tract) and neuroendocrine/autonomic responses (spinohypothalamic tract). Notably, the spinoparabrachial pathway is an important route of convergence of somatic and visceral nociceptive stimuli (see Section 1.5.2). For a general description of all these pathways, see [29].
1.5.2. Visceral Pain Pathways
Visceral pain arises from the internal organs including the heart and vessels, airway structures, GI tract and urinary and reproductive organs [30,31]. However, visceral pain is not evoked from all viscera and it is not always linked to a factual visceral injury [32,33]. The diffuse nature and difficulty in locating visceral pain is due to a relatively low density of visceral sensory innervation and extensive divergence of visceral input within the CNS. While in somatic pain, nociceptive C-fibers make synaptic contacts with second-order projection neurons in the upper laminae of the dorsal horn spinal cord and the substantia gelatinosa (lamina II) interneurons [34], input from the viscera has a more diffuse and less topographic distribution and specifically, reaches lamina I and the deep dorsal horn [31,35]. In patients, pain from different visceral organs can have differing areas of presentation, e.g., bladder to perineal area or heart to left arm and neck. Therefore, visceral pain is often localized to distant structures and thus, known as referred pain. Development of symptoms may entail referred pain to somatic structures within the same metameric field as the affected viscera [33]. In addition, secondary hyperalgesia of superficial or deep body wall tissues might occur due to viscerosomatic convergence [32,33,36,37]. Finally, visceral pain is often associated with marked motor and autonomic reflexes such as profuse sweating, nausea, vomiting, GI disturbances and changes in body temperature, blood pressure and heart rate [32,33,36,38,39].
Primary afferent fibers innervating the viscera project to the CNS through autonomic sympathetic and parasympathetic nerves. The sympathetic innervation follows the hypogastric, lumbar and splanchnic nerves which traverse both prevertebral and paravertebral ganglia, until they reach the thoracolumbar region of the spinal cord. The parasympathetic innervation is mediated by vagal and pelvic afferents that terminate in the brainstem and lumbosacral cord, respectively [40,41].
Visceral pain is primarily signaled by spinal afferents, while vagal afferents signal non-painful sensations such as hunger, satiety, fullness and nausea [39]. Although, vagal afferents may not signal pain directly, several studies have documented that stimulation of the vagus nerve attenuates somatic and visceral pain [39]. Vagal afferents originate from PSNs in nodose ganglion that project to the nucleus tractus solitarius (NTS) in the brainstem. Spinally-converging visceral afferents instead, synapse onto the second order neurons within the dorsal horn, which in turn project to higher centers through the dorsal column pathway, the parabrachial pathway and the spinothalamic tract (STT) [31]. Superficial dorsal horn projections, mostly form the spinoparabrachial pathway [42], are associated with autonomic and affective responses to painful stimuli [43]. Along with NTS projections from vagal afferents, spinoparabrachial projections are transmitted to limbic and cognitive higher relay centers including those parts of the brain involved in affectivity, such as the amygdala, hypothalamus and periaqueductal grey (PAG) [31,43]. Visceral spinothalamic projections travel contralaterally from the deep dorsal horn to the main thalamic projections located in the ventroposterior medial and lateral nuclei of the thalamus. The medial thalamic nuclei eventually project to the areas of the prefrontal cortex that are correlated with visceral pain [44,45,46] (Figure 1).
Furthermore, the GI tract besides its extrinsic innervation as mentioned above, has its own intrinsic nervous system, the enteric nervous system (ENS), composed by the submucosal (Meissner’s) and myenteric (Auerbach’s) plexuses [47]. Myenteric plexus lies between the longitudinal and circular muscle layers, whereas the submucosal plexus is found in the submucosa and relays sensory and motor responses to the myenteric plexus, prevertebral ganglia and spinal cord [47,48]. The ENS works autonomously but the digestive function requires communication with the parasympathetic and sympathetic innervations (Figure 1). The ENS contains sensory peptides, primary afferent neurons, interneurons and motor neurons and together with the extrinsic system, controls motor activity, secretion, absorption, blood flow and interactions with other organs such as the pancreas or gallbladder. This way, the gut provides sensory information to the CNS and the CNS can affect GI function [47,48].
1.6. Capsaicin as an Analgesic Medication
There is widespread use of capsaicin and other capsaicinoids in traditional medicine, not only in pain therapy but also in body temperature regulation, anti-obesity treatments, anticancer therapy and as antioxidant and antimicrobial agents [49]. In several South-American, African and Asian countries, the leaves and fruits of Capsicum spp. have long been in use in the treatment of painful menses, toothache and muscle pain [50]. After the discovery of TRPV1, capsaicin and numerous other natural and synthetic receptor agonists have attracted the attention of the academic and pharma community for their potential in the relief of chronic pain [22]. In national pharmacopoeias, topical capsaicin medications are registered for the treatment of painful states derived from neuralgia, diabetic neuropathy, osteoarthritis and rheumatoid arthritis. Capsaicin-based preparations are also in use to treat pain due to pruritus, psoriasis, mastectomy and bladder disorders.
2. The Capsaicin Receptor in Nociceptive Pathways
2.1. Structure and Physiology of the Capsaicin Receptor
2.1.1. Structure and Splice Variants of TRPV1
The painful sensations induced by capsaicin are consequent to its binding TRPV1. As mentioned in Section 1.3.1, TRPV1 is part of the TRP multigene superfamily that encodes a wide number of integral membrane ion channel proteins [16]. Ligand binding and activation profiles of these receptors are closely related but unique to each TRPV subfamily member. The formation of multimeric species [51] and/or heteromultimers among members of the family may increase their functional diversity. For example, TRPV1 and TRPV2 are co-expressed in the IV-VI layer neurons of the adult rat cerebral cortex, with occurrence of multimeric receptor complexes upon receptor activation in vitro [52].
Alternative splicing also occurs in the TRP gene family, expanding the number of functionally distinct TRP proteins and potentially providing tissue-specific regulation. Several TRPV1 splice variants have been reported in mice, rats and humans [53,54,55]. While the biological roles of these variants are unclear, the reported deletions lead to nonfunctional channels (mTRPV1b, VR.5'sv and TRPV1VAR) or channels with distinct properties, when examined in recombinant or native expression systems [56]. TRPV1α and TRPV1β are two cDNA variants of TRPV1 as a result of alternate splicing. TRPV1β is a dominant-negative regulator of TRPV1 responses, since it is not functional by itself but inhibits TRPV1α function during co-expression. The recently identified splice variant of the TRPV1 molecule, TRPV1b, produces a negative-dominant effect on the responsiveness of the TRPV1 channel which is increased by peripheral inflammatory processes. TRPV1b has been cloned from mouse DRG neurons, human and rat cDNA libraries respectively. TRPV1b is not sensitive to capsaicin and protons but responsive to heat. Furthermore, TRPV1b but not TRPV1, is expressed on sensory neurons which respond to heat but are not activated by capsaicin. Moreover, when co-expressed with TRPV1, TRPV1b reduces the responses to TRPV1 channel activators in a ratio-dependent manner [57].
2.1.2. Biochemistry and Physiology of TRPV1
TRPV1 is a non-selective cation channel that, similarly to all TRPVs, prefers calcium. The receptor is activated by capsaicin and noxious temperatures with a threshold in vitro of approximately 43 °C [5].
The threshold of activation in vitro suggests that TRPV1 is inactive at normal body temperature. However such a threshold is dynamically regulated and significantly lowered during inflammation [22]. TRPV1 is also activated, among others, by moderate heat, protons and anandamide, an endogenous ligand of both vanilloid and cannabinoid receptors [5,58]. The full list of natural substances to date reported to be capable of activating/sensitizing the receptor, aside from capsaicin, is reported in Table 1. To these, numerous other synthetic compounds should be added [59].

Table 1.
Natural activators/agonists of TRPV1 receptor. Some of these molecules not only act as receptor agonists but also as channel blockers. Abbreviations: DkTx = Double-knot toxin; HPETE = hydroperoxyeicosatetraenoic acid; NADA = arachidonoyl-dopamine; NGF = nerve growth factor; RTX = resinferatoxin; VaTx = vanillotoxins.
Like other TRP channels, TRPV1 is a putative six-transmembrane-spanning protein with a pore region localized between transmembrane segments 5 and 6 [5,60]. Numerous studies have led to the identification of the receptor regions and key amino acids involved in specific functions (multimerization, capsaicin action, proton action, heat activation, desensitization, permeability and phosphorylation and modulation by lipids and ATP) [61].
Activity of TRPV1 is positively regulated inside the cell after phosphorylation [5], as its N-terminus has several phosphorylation sites for protein kinases, among which is the calcium and calmodulin-dependent protein kinase II (CaMK II kinase). Some activators listed in Table 1, such as prostaglandins, bradykinin and prokineticin, modulate the activity of the receptor indirectly by activating several different protein kinases inside the cell [61]. Cleavage of phosphatidylinositol 4,5-bisphosphate (PIP2) by phospholipase C (PLC) is another intracellular activation pathway [62,63,64]. Besides PIP2, other negative regulators are intracellular calcium and calmodulin [61]. Instead, phosphatases desensitize TRPV1 after its dephosphorylation [65].
Depending on functional conditions, once activated TRPV1 may become sensitized or enter a refractory state. Studies on these phenomena were mainly carried out in vitro and their relevance in vivo still needs confirmation for the development of new medications. On cultured human respiratory epithelial cells, sensitization of TRPV1 receptors by antagonists’ pretreatment was shown to be a consequence of the translocation of existing receptors from the endoplasmic reticulum to the cell surface [66]. As an additional possibility, once activated by capsaicin, TRPV1 enters a long refractory state [67]. Refractoriness is a consequence of conformational changes in the receptor protein that depend on extracellular calcium and ultimately, close the channel pore [68].
It should be also mentioned that there seems to exist a marked difference in pharmacological responses between TRPV1 and native capsaicin receptors for the presence of regulatory proteins associated with TRPV1 in vivo, among which is Fas-associated factor 1 (FAF1). Notably, FAF1 was demonstrated to constitutively modulate the sensitivity of TRPV1 to various noxious stimuli in sensory neurons by forming an integral component of the vanilloid receptor complex [69].
2.2. Localization and Activation of TRPV1 in Pain Pathways
2.2.1. PSNs and Non-Neural Cells
Activation of TRPV1 during Neurogenic Inflammation of Skin and Mucosae
The sensitivity of TRPV1 to numerous physical and chemical activators and its widespread distribution indicate that the receptor may be crucial to the onset of inflammation. Activation of TRPV1 on the terminal endings of the sensory fibers derived from somatic and visceral PSNs and some non-neural cells (see Section 1.3.1) produces a calcium and sodium influx that ultimately results in the release of a cocktail of neuropeptides. Among these, the tachykinins substance P and neurokinin A (NKA) and the calcitonin-gene-related-peptide (CGRP) are of particular relevance. Epithelial, endothelial and smooth muscle cells, as well as resident immune cells, respond to these neuropeptides and give rise to a complex series of events collectively referred to as neurogenic inflammation. The main features of neurogenic inflammation, i.e., redness, swelling and pain, are a consequence of the vasodilation, plasma extravasation and hyperalgesia that follow binding of NKA, substance P and CGRP to their cognate receptors on the endothelial and smooth muscle cells [70]. In response to the release of these neuropeptides, mast cells, epithelial cells and immune cells release pro-inflammatory cytokines (e.g., IL1b, IL6, IL8, and TNFα) further contributing to the maintenance of the inflammatory state. The central role played by TRPV1 in the initiation and modulation of neurogenic inflammation suggests that the same factors which influence its functional expression or numbers, could also influence the organism’s response to inflammatory xenobiotics [71].
Other Effects of TRPV1 Activation in PSNs
At first, intradermal capsaicin injection into the plantar skin of the hind paw in mice contributes to the development of thermal and mechanical hyperalgesia [5]. Capsaicin also evokes a mechanical allodynia that could be reversed by both G protein and protein kinase inhibitors [90]. At the cell level, the activation of protein kinase A and C evoked by capsaicin results in the phosphorylation of the NMDA receptor subunit NR1 at Ser890/897 and Ser896 respectively, which could be blocked with specific inhibitors [91]. A role for protein kinase B (PKB)/Akt in DRG neurons was also demonstrated using the intradermal capsaicin model [92].
As a consequence of these cellular changes, not only the activation of TRPV1 releases NKA, substance P and CGRP from the peripheral terminals of PSNs contributing to the onset of pain and inflammation but it also blocks the axoplasmic transport of these and other neuropeptides [93], thereby depleting terminals of their neuropeptide content. This block of nociception by the exhaustion of its main mediators at periphery was initially believed to be primarily responsible for pain relief after local capsaicin treatment. However it was more recently understood that the analgesic effect of capsaicin is mainly a consequence of a loss of function of the somatic nociceptive fibers [94]. In fact, the capsaicin-induced defunctionalization of these fibers not only derives from the depletion of neuropeptides but also from the loss of membrane potential, the block of neurotrophic factor axonal trafficking, and a reversible retraction of cutaneous terminals [94]. Nerve endings collapse because of the calcium overload induced by activation of TRPV1, with loss of mitochondrial function, metabolic inhibition and disruption of the integrity of the terminal membrane [94]. These observations are fully in line with previous studies on TRPV1-bearing cells in vitro that, after challenge with nontoxic doses of capsaicinoids, RTX or olvanil, downregulated their TRPV1 expression and ultimately underwent cell death [95,96]. In keeping with these in vitro studies, RTX causes the loss of unmyelinated C fibers and a significant damage to myelinated Aδ fibers in adult rats. The agonist also depletes TRPV1 expressing neurons in DRGs with reduced thermal pain perception [97]. Therefore, the defunctionalization that follows the activation of TRPV1 by capsaicin and its agonists/activators causes long-term functional and structural alterations that are not limited at the peripheral terminals of the PSNs.
Finally, it needs to be mentioned that repeated topical application of capsaicin also leads to degeneration of the cutaneous autonomic nerve fibers [98]. As a result, there is a reduction of pain by a still not clearly understood additional mechanism that, likely involves a TRPV1-mediated calcium influx and glutamate release from these fibers [99].
2.2.2. Somatic Pathways
First-to-Second Order Neuron Synapses and Modulation in Substantia Gelatinosa
The painful effects of the administration of capsaicin to the skin occur very soon after an intradermal injection of the vanilloid, but the intensity of pain decreases progressively within twenty minutes. At the site of injection, the desensitization or defunctionalization of TRPV1-expressing primary afferents (see Section 2.1.1) results in a region of hypoalgesia. However, near the injection site, primary mechanical and heat hyperalgesia rapidly develop and last for about one day. It is widely agreed that primary mechanical and heat hyperalgesia are a consequence of the sensitization of PSNs [100]. Secondary mechanical hyperalgesia and allodynia instead appear in a progressively larger skin area after about fifteen minutes, to then slowly decrease in a few hours (allodynia) or in about one day (hyperalgesia) [101]. PSNs innervating the skin in a region of secondary mechanical allodynia and hyperalgesia are not sensitized by the capsaicin injection and thus, have a normal level of excitability. Therefore, an enhanced responsiveness (central sensitization) of the second order nociceptive STT or trigeminothalamic tract neurons is responsible for secondary mechanical allodynia and hyperalgesia [100].
The burning pain or sometimes itch, produced by the activation of peripheral TRPV1s [102] follows the discharge in polymodal C and mechano-heat Aδ cutaneous nociceptors. As mentioned in Section 2.2.1, these PSNs release a cocktail of neuropeptides at periphery. However, in the excited state, the central terminals of the PSNs release the same cocktail of neuropeptides and the fast amino acid transmitter glutamate in the spinal cord dorsal horn or the SNTN. These neurotransmitters act onto two main targets: the second order sensory STT projection neurons and the interneurons of the dorsal horn substantia gelatinosa [103]. Analogous effects are elicited onto the second order sensory trigeminothalamic tract neurons and the interneurons of the SNTN substantia gelatinosa. Notably, it was also the GABAergic interneurons to be affected by a topical injection of capsaicin into the skin, as demonstrated in vivo by the increased expression of proto-oncogene c-Fos in GABA-immunoreactive (IR) neurons following a challenge with the vanilloid [104].
Second-to-Third Order Neuron Synapses and Other Brain Areas Involved in Pain Control
In comparison to the enormous mass of data on first-to-second order neuron synapses, few studies have addressed the distribution and physiology of TRPV1 in supraspinal centers and more specifically, in STT synapses. Among the several areas of the brain that are directly or indirectly involved in the modulation of ascending nociceptive stimuli, TRPV1 has been localized in the somatosensory cortex, anterior cingulate cortex, insula, rostroventral medulla, PAG, amygdala and NTS [52,105,106]. Functionally, the role of the receptor is not totally clear, as capsaicin microinjected in the ventrolateral PAG had both hyperalgesic [107] and antinociceptive effects in rats [108,109,110]. The mechanisms of TRPV1 activation, function and sensitization of hypothetical supraspinal synapses in health and pathological pain conditions are discussed in [111].
Activation of TRPV1 during Pain Perception
Several lines of evidence converge to demonstrate that TRPV1 has indeed a role in bona fide pain perception. In a mouse model of neuropathic pain, a TRPV1-dependent glutamate-mediated cross talk among the prelimbic and infralimbic cortex neurons and glia participates to the generation of pain. In addition, single-unit extracellular recordings in vivo, following electrical stimulation of the basolateral amygdala or application of pressure on the hind paw, showed increased excitatory pyramidal neuron activity in these cortical areas, which also contained higher levels of the endocannabinoid 2-arachidonoylglycerol [112]. In another study, N-arachidonoyl-serotonin, which is a hybrid TRPV1 antagonist and fatty acid amide hydrolase inhibitor, normalized the imbalance between excitatory and inhibitory responses in the medial prefrontal cortex neurons, resulting in pain inhibition [113].
2.2.3. Visceral Pathways
Visceral pain represents a major clinical problem, yet far less is known about its mechanisms compared to somatic pain. Since viscera are not normally exposed to noxious heat or capsaicin, the presence of TRPV1 renders the visceral afferents sensitive to the mediators of inflammation, thus functionally serving as nociceptors. As it will be further discussed, TRPV1 expression is found widespread in visceral innervation of all body organs and, when upregulated, TRPV1 correlates significantly with the degree of visceral pain. The importance of TRPV1 in visceral innervation is also supported by the painful effects of capsaicin application in several animal models and reports of human studies.
TRPV1 Expression and Function in the Urinary Tract—Upper Urinary Tract
TRPV1 immunoreactivity is present in nerve terminals that course both the mucosa and muscular layer of the renal pelvis [114], with the exception of the rat kidney parenchyma [114]. TRPV1 may have a protective role in situations of exaggerated renal function and structural injury by attenuating the progression of renal fibrosis, possibly through down-regulation of the TGF-β/Smad2/3 signaling pathway [115]. In addition, TRPV1 attenuates renal inflammatory responses in mice subjected to DOCA-salt hypertension [116]. In the ureters, TRPV1 is detected in nerve terminals that course both the mucosa and muscular layer [114], where CGRP released from capsaicin-sensitive fibers acts as inhibitory transmitter, contributing to ureteral motility [117,118]. However, capsaicin also induces an inflammatory response in the ureters [45].
TRPV1 Expression and Function in the Urinary Tract—Lower Urinary Tract
In the lower urinary tract, TRPV1 expression is well documented not only in a large subpopulation of nerve fibers but also in non-neuronal cells [114]. In the rat bladder mucosa, most IR fibers are in close proximity to the basal cells of the transitional epithelium, while in the vesical muscular layer TRPV1-IR fibers impinge on the surface of the smooth muscle cells. Functional TRPV1 channels in urothelial cells, including the basal, intermediate and large superficial umbrella cells, have been confirmed in rodents by either RT-PCR or immunolabeling [9,119]. In the human bladder, TRPV1-IR was detected in nerve fibers coursing in the suburothelial connective tissue and in muscular layer [120,121,122]. Charrua et al. confirmed the presence of TRPV1 channels in human urothelial cells by measuring TRPV1 mRNA levels in tissue extracts [123]. TRPV1-IR has also been reported in the interstitial cells of the human bladder [124] which form a suburothelial network that may contribute to a fast spread of smooth muscle contractions [125]; in the smooth muscle cells; endothelium of capillaries and arteries (but not of veins); and mast cells [120].
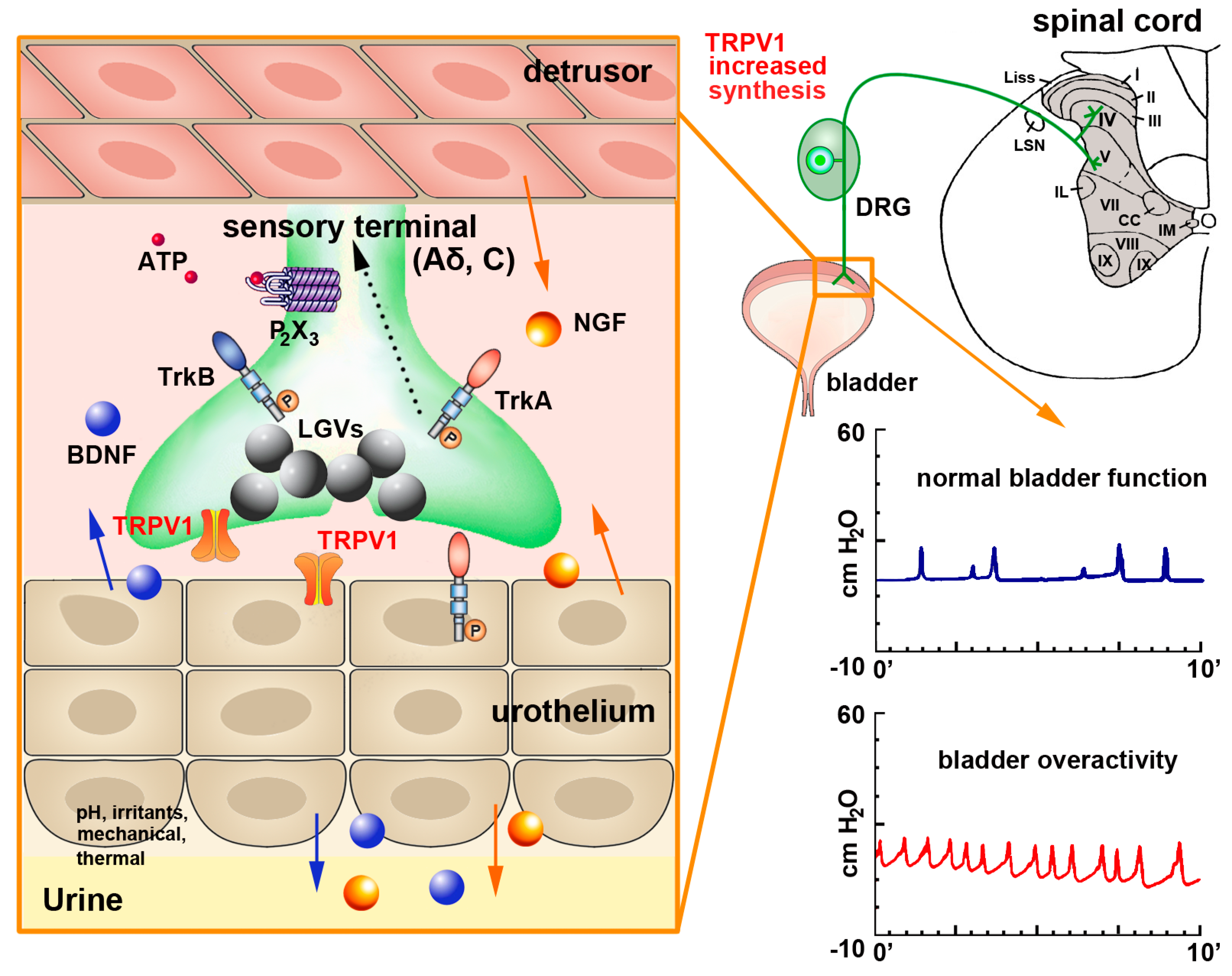
Given its extensive distribution, TRPV1 is important in regulating normal lower urinary tract function, and studies involving the desensitization of bladder sensory fibers by capsaicin and RTX have contributed to elucidate its mechanisms of action [126,127,128,129]. During bladder inflammation or spinal cord injury, TRPV1, substance P and CGRP expression is elevated [130] and this allows for the development of bladder overactivity and pain [126,129,131,132,133] (Figure 2). Pretreatment of rats with SDZ 249-665, a vanilloid compound reproducing capsaicin desensitization, attenuates inflammatory bladder hyperreflexia, referred hyperalgesia [134] and behavioral pain responses to intraperitoneal acetic acid in rats [135]. In mice with lipopolysaccharide (LPS)-induced bladder inflammation, an increase of the pain-evoked fos gene expression in sacral spinal cord neurons of wild type (WT) but not TRPV1 knock-out (KO) mice is observed, an effect that was accompanied by an increase of bladder reflex contractions [126,132,136]. Specifically, TRPV1 KO mice display greater short-term voluntary urination and abnormal urodynamic responses, with an increase in the frequency of non-voiding contractions, increased bladder capacity and inefficient voiding [136].
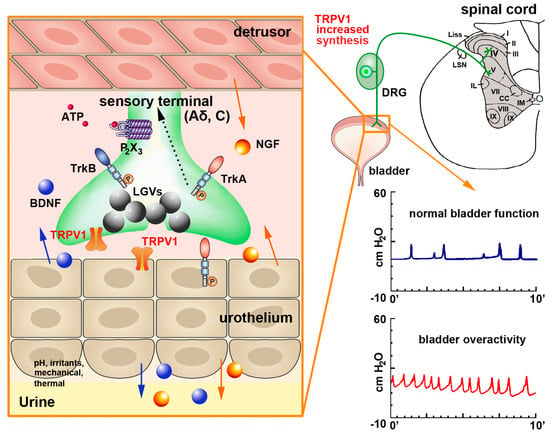
Figure 2.
Schematics of TRPV1 localization and function in the urinary bladder and its contribution to bladder dysfunction. In the urinary bladder, TRPV1 can be found in sensory afferents and in the urothelium. Upon mechanical injury or inflammation TRPV1 levels are increased together with those of substance P and CGRP (stored in large granular vesicles—LGVs—large gray spheres) Nerve growth factor (NGF—orange spheres) is also released by the detrusor smooth muscle and the urothelium (orange arrows). NGF activates tropomyosin-related kinase A (TrkA) receptors expressed on afferent terminals, contributing to sensitization of neuronal TRPV1. The TrkA-NGF complex is internalized and retrogradely transported (dashed line) to neurons in lumbosacral dorsal root ganglia (DRGs), where de novo transcription of TRPV1 and additional sensory ion channels (including purinergic P2X3 receptor for ATP—small red spheres) is initiated. These newly synthesized ion channels are anterogradely transported back to afferent terminals to contribute to peripheral hypersensitivity. The urothelium also potentially produces brain-derived nerve factor (BDNF—blue spheres), which binds to tropomyosin-related kinase B (TrkB) receptors, further contributing to sensitization. This also participates to the development of bladder over activity, as shown by the increase number of bladder contractions in the graph at bottom right of the figure.
However, in the rat the density of TRPV1-IR fibers coursing in the mucosa or in the muscular layer was shown to be rapidly and markedly reduced after application of capsaicin or RTX [114,137] or even after TRPV1 antagonists [57,126,132,138], which significantly enabled an improvement of bladder function after inflammation or spinal cord injury. Similar findings were reported after intra vesical RTX application in patients with neurogenic detrusor over activity [121,139].
TRPV1 is also localized in nerve terminals within the mucosa and muscular layer of the urethra [114,122]. In keeping with this observation, application of capsaicin produced an inhibitory effect on the nerve-mediated contractions of the rat proximal urethra, promoting its relaxation and thus, facilitating urine voiding [140]. However, capsaicin also induces an urethral inflammatory response [141].
TRPV1 Expression and Function in the Digestive Apparatus
TRPV1 in the digestive apparatus has been well documented in the afferents originating from PSNs in the DRGs, TG and nodose ganglion [142,143]. In general terms, TRPV1-IR fibers play an important role in modulating several physiological gut functions including motility, secretion, circulation and nociception [144,145]. However, TRPV1 is also emerging as a potential mechanosensor that mediates pain in various GI diseases [136,146,147].
Esophagus
Retrograde tracing and multiple labeling immunofluorescence studies have revealed that TRPV1-IR neurons projecting to the rat esophagus are located in the DRGs and the nodose ganglion [148]. TRPV1-IR nerve fibers deriving from these neurons are found in both the submucosal and myenteric plexuses of the esophagus. These fibers also express CGRP/substance P and neuronal nitric oxide synthase (nNOS) in the myenteric plexus of mice [149], and can be activated by capsaicin [150]. After exposure of the rat esophagus to acids, TRPV1 increases in DRGs [148,151]. In rats with chronic esophagitis, muscle afferent fibers exhibit a significantly greater response to capsaicin compared to non-inflamed naive animals [152]. This sensitized response is possibly due to an increase in the expression of TRPV1 channels in vagal afferent fibers after esophagitis. Activation of cervical and thoracic DRG neurons by intra-esophageal acid in vivo is lost in TRPV1 KO mice, suggesting that TRPV1 channels are required for acid-induced esophageal pain [153]. In keeping with this observation, TRPV1 KO mice develop a significantly less severe esophagitis after acid exposure [154]. Moreover, capsaicin causes enhanced relaxation of smooth muscle contraction in the esophagitis model, which supports the fact that acid reflux increases the expression of TRPV1 in primary afferent nerves [149]. In TRPV1 deficient mice, reflux promoting surgery induces significantly less mucosal injury and inflammation than in WT mice and pretreatment with the TRPV1 antagonist capsazepine in WT mice significantly inhibits esophagitis [152]. In cat spinal afferents, capsaicin-evoked stimulation of epithelial TRPV1 induces hypersensitivity and esophagitis due to the release of substance P and CGRP from esophageal sensory nerve endings [155].
Expression of TRPV1 is up-regulated also in human patients with esophagitis, gastro-esophageal reflux disease and non-erosive reflux disease [156,157,158]. The increase in the expression of TRPV1 correlates to intra-esophageal up regulation of nerve growth factor (NGF) and glial-cell-derived neurotrophic factor (GDNF) [157], which are important mediators involved in neuroplastic events and, therefore, contribute to enhancement of sensitization of the esophageal nerve fiber network.
Stomach
In rodents, most stomach-innervating neurons in nodose ganglia and DRGs have been shown to be IR for TRPV1 (80% and 71%, respectively) [142,151]. Furthermore, numerous TRPV1-IR nerve fibers were detected in the myenteric plexus, at the level of the fundus and the antrum. These fibers are in close contact with the myenteric ganglionic neurons. TRPV1-IR fibers were also observed within the circular and longitudinal muscle layers and at the level of the lamina propria of the mucosa. In these locations, TRPV1 has been found to co-localize with CGRP and substance P [159,160]. In humans, TRPV1 labeling was found in intra-cytoplasmic granules of the glandular parietal cells, in the gastric cells, in intramural ganglionic neurons and fibers, the latter being numerous in the submucosa and mucosa and, often, ending close to the parietal cells [161]. In addition, expression of TRPV1 protein and mRNA in a rat gastric mucosal epithelial cell line as well as in the mucosa of the intact rat stomach by Western blotting and RT–PCR, respectively, suggest that TRPV1 plays a protective role in these cells [162].
However, acute exposure of the rat gastric mucosa to a noxious HCl concentration has been shown to raise TRPV1 protein but not mRNA in DRG neurons innervating the stomach [151]. Capsaicin pre-treatment of rats prevents the behavioral pain reaction to gastric acid challenge [163]. In addition, central injection of capsaicin and anandamide stimulates gastric acid secretion in rats, via TRPV1 coupled with non-NMDA and GABAA receptor systems [164,165].
In humans, the intragastric administration of capsaicin increases the sensitivity to proximal gastric distension [166], and ingestion of capsaicin capsules induces gastric sensations of pressure, heartburn and heat [167].
Small Intestine
Intense labeling of TRPV1-IR nerve fibers is found surrounding the myenteric neurons within the enteric ganglia of jejunum and ileum. Fibers can be found also in interganglionic tracts and they diverge within myenteric ganglia to wrap around cell bodies of the myenteric neurons [143]. Occasionally, TRPV1-IR nerve fibers can also be located within the circular muscle layer, surrounding the enteric neurons, and the blood vessels of the submucosal plexus. Retrograde labeling and immunofluorescence studies in DRGs revealed high degrees of co-localization between TRPV1 and CGRP, substance P or nNOS, in neurons supplying the mouse jejunum [168]. TRPV1-IR nerve fibers in the rat jejunum derive from extrinsic neurons, and activation of TRPV1 produces a relaxation response that is in part, due to the release of CGRP [169].
In general, capsaicin stimulates, most likely via TRPV1, the extrinsic afferents of the gut [150] and its administration into the lumen of the alimentary canal evokes pain in mice [170]. Capsaicin is thought to evoke intestinal pain by stimulation of jejunal chemoreceptors, presumably expressing TRPV1 [171]. Afferent jejunal nerve fibers can be activated by capsaicin, but this effect is obviously lost in TRPV1 KO mice [147]. Patients with uninvestigated dyspepsia have been found hypersensitive to intrajejunal capsaicin infusion [172]. In addition, administration of capsaicin into the ileum of patients with an ileal stoma has been reported to cause mechanical hypersensitivity [173].
Large Intestine
About three quarters of the colonic splanchnic afferents express TRPV1 and show capsaicin responsiveness [174]. Notably, the TRPV1-IR area in the rectum is the largest in the isolated mouse lower GI tract. Numerous TRPV1-IR nerve fibers were found in the mucosa, submucosal layer (around blood vessels), in the myenteric plexus and in the circular and longitudinal muscle layer; these fibers were found to contain CGRP, substance P and nNOS [175]. In the distal colon, TRPV1 fibers were half of that in the rectum, and in the transverse and proximal colon their density was even lower [175,176,177].
During pathological conditions the levels of TRPV1 increase in colonic tissues of rodents and humans, in parallel with a hypersensitivity of the large intestine [146,178,179,180] that could be inhibited by antagonist administration [181,182]. Rats pretreated with capsaicin do not present the inflammation-induced hypersensitivity that follows experimental colonic distension [183,184]. In a rat model of dextran sulfate sodium (DSS) colitis, neonatal animals chemically deprived of TRPV1-expressing fibers by treatment with capsaicin, as well as those given a TRPV1 antagonist (JNJ 10185734) were both protected from the damaging effects of DSS [179]. In keeping with these observations, Jones et al. found that the zymosan-induced sensitization of colonic afferents was absent in TRPV1 KO mice [146]. A mouse post-inflammatory chronic hypersensitivity model using intracolonic trinitrobenzene sulphonic acid (TNBS), which induces colitis, also showed TRPV1 as an important mediator of mechanical and chemical visceral hyperalgesia [182,185]. Furthermore, several studies have reported elevated expression of TRPV1-IR nerve fibers in large intestine biopsies from patients with irritable bowel syndrome and painful Crohn’s disease [25,180,186], Hirschprung disease [187], rectal hypersensitivity and fecal urgency [180]. Also, the density of TRPV1-positive nerve fibers in the rectosigmoid colon correlates with pain severity in patients with irritable bowel syndrome [186].
Pancreas
Through retrograde labeling of pancreatic nerves and immunostaining, it has been possible to localize the pancreatic afferents expressing TRPV1 in the mouse T9-T12 DRGs (75% of neurons) and the nodose ganglion (35% of the neurons) [188]. Within the glandular parenchyma, TRPV1-IR nerve fibers originating from these neurons are in close proximity to pancreatic acini [189]. Also in pancreatic nerve fibers, TRPV1, CGRP and substance P were found to be co-expressed. Capsaicin injection into the pancreatic duct promotes substance P and CGRP release in the dorsal horn neurons upon stimulation of pancreatic sensory nerves [189,190,191]. In turn, substance P has been shown to stimulate plasma extravasation from pancreatic post capillary venules [192]. In keeping with this observation, blocking substance P with specific NK1 receptor antagonists, or genetic deletion of the NK1 receptor, reduces pancreatic edema and neutrophil infiltration [192,193].
Capsaicinized animals incur in a much less severe pancreatic inflammation compared to non-capsaicinized animals, as the capsaicin-sensitive neurons expressing TRPV1 were destroyed [194]. However, administration of capsazepine significantly reduces inflammation and pancreatic injury in a model of caerulein-induced acute pancreatitis [195]. Injection of capsaicin into the pancreatic duct of rats has also been shown to induce expression of fos in the neurons of the spinal dorsal horn, contributing to pancreatic pain [196].
TRPV1 Expression and Function in the Respiratory Tract
Fine axons expressing TRPV1 are diffusely distributed in the respiratory tract, specifically within the epithelium of the respiratory organs, including the trachea, bronchi, bronchioles and pulmonary alveoli. In these locations, TRPV1-IR fibers are also seen below the epithelium, surrounding the smooth muscles and blood vessels and around the alveolar wall [197]. Furthermore, most TRPV1-IR axons within the intrapulmonary airways co-express substance P and CGRP within and beneath the epithelium, around blood vessels, within airway smooth muscle and alveoli. However, only a small proportion of the nerves in the tracheal epithelium and only half the number of TRPV1 axons are immunopositive for substance P and CGRP [198].
During inflammation, lipoxygenase products, such as 15-HPETE, 15-HETE and leukotriene B4 (LTB4) released from the airways epithelial cells can directly activate TRPV1, thus contributing to neuronal hypersensitivity [199]. Increased production of NGF during asthma can potentiate inflammation through TRPV1 sensitization. In addition to these inflammatory mediators, acidic pH associated with inflammation can contribute further to TRPV1-mediated airway hypersensitivity during asthma [200].
TRPV1 Expression and Function in the Genital Tract
In the male genital tract, rat TRPV1 mRNA is detected in the testicles, prostate and penis [201]. Similarly, transcripts for human TRPV1 were isolated from the testicles, seminiferous tubules, corpus cavernosum, glans and its overlying skin, scrotal skin and prostate [201]. In addition, TRPV1 could be found in Sertoli cells, where it regulated the acidity of extracellular microenvironment, which is crucial to maintain male fertility [202]. TRPV1 KO mice present testicular hyperthermia, which results in massive germ cell depletion from the seminiferous tubules [203]. In the human prostate, TRPV1 has also been described in primary afferents that course the urethral mucosa, verumontanum, ejaculatory ducts and periurethral prostatic acini, both by immunocytochemistry and western blotting [204]. The rich TRPV1 sensory innervation found in the human prostate plays an important role in the development of chronic prostatitis (Chronic Prostatitis/Chronic Pelvic Pain Syndrome—CP/CPPS). Burning pain sensation is the main description of pain in patients with CP/CPPS, either upon urination or ejaculation and is directly related to activation of TRPV1-IR fibers [204,205]. In addition, CP/CPPS patients have increased heat sensitivity in the perineal area [206]. TRPV1 immunoreactivity is also found in the epithelial cells of the prostate [207], where application of capsaicin and RTX causes a calcium inflow, revertible by capsazepine [208]. Notably, although low levels of TRPV1 mRNA are detected in the prostate, high levels of the protein occur in both the epithelium and smooth muscle cells of the gland [209].
In the female genital tract, TRPV1 is expressed in the vagina [210] and myometrium of the uterus. Rat uterine cervical afferents in the hypogastric nerve express TRPV1 [211] and estrogens amplify the responses to painful stimuli of the uterine cervix due to an increase of TRPV1 expression in PSNs innervating the uterus [212]. On the other hand, the activation of nerve fibers in the uterine horn is reversed by intrauterine pretreatment with capsazepine [212,213].
In a study investigating TRPV1 innervation of the human uterus during pregnancy and labor, TRPV1-IR fibers were observed scattered throughout the stroma and around blood vessels and appeared more frequently in the subepithelium of the cervix uterus. An almost complete disappearance of TRPV1-IR fibers was observed in the pregnant uterus, but the cervical innervation remained high throughout pregnancy and labor and was likely responsible for pain during cervical ripening [214]. In addition, TRPV1-expressing fibers significantly increased in the vulvar epidermis and superficial dermis [23].
3. Experimental Modeling Nociception Using Capsaicin in Vitro and ex Vivo
Under normal conditions in vivo, capsaicin and capsaicinoids only have access to cutaneous and mucosal TRPV1-expressing sensory fibers of the digestive tract, the airways and the conjunctiva. However, given the widespread expression of the receptor in tissues and organs, the molecule has been used as an agonist of TRPV1 in numerous in vitro and ex vivo studies. Most studies on isolated cells have been fundamental to understand the biology and function of TRPV1 as summarized in Section 1.3.2 and Section 2.1. Other studies in vitro led to a better understanding of the cellular pathways that are activated following receptor activation by the vanilloid.
It is well established that assessment of functional activity of TRPV1 can be evaluated by quantification of ATP release by capsaicin stimulation. In urothelial cells, this process was shown to be potentiated by NGF treatment and dependent on TrkA activation via phosphatidylinositol-3-kinase (PI3K) and protein kinase C signaling [215]. The relation between TRPV1 channels and NGF-induced pain is well established in somatic and visceral pain, and studies in heterologous expression systems contributed to demonstrate the biochemical mechanism through which bradykinin and NGF produce hypersensitivity and might explain how the activation of PLC regulates the activity of other members of the TRP channel family in PSNs [81].
Ex vivo systems are less commonly in use but they are very promising as a bridge, linking intracellular pathways studies and functional anatomy. In general, these preparations include at least bundles of primary fibers and a target organ, such as the intestine or the skin. For example, using an ex vivo colon-pelvic nerve-L6 DRG-and spinal cord preparation, mechanically sensitive colonic afferents were shown to segregate into high firing and low-firing frequency fibers based on firing frequency and distension response thresholds. Notably, nearly all low frequency afferents expressed TRPV1 and could be sensitized by luminal application of capsaicin, in contrast to the high-frequency ones [178].
Using acute jejunal preparations, capsaicin induced typical excitatory responses on sensory afferents of WT mice, which lasted briefly and were followed by desensitization with higher concentration of capsaicin. In contrast, capsaicin had neither excitatory nor desensitizing effects on jejunal nerves of TRPV1 KO mice [147].
Finally, capsaicin-evoked action potentials on isolated skin-nerve preparations were significantly decreased in cannabinoid 1 receptor KO mice [216].
Spinal cord slices (Figure 3) are another type of ex vivo preparation that proved to be useful for a better understanding of the central modulatory effects of TRPV1. Most diffuse are acute spinal cord slices, at times with attached dorsal roots and/or DRGs. However, the spinal cord was also organotypically cultivated thereby leading to deafferentation and disappearance of the primary sensory input to the spinal cord dorsal horn [217]. Initial studies on acute spinal cord slices were carried out with an electrophysiological approach and were focused onto lamina II, given its pivotal role in the modulation of nociception. Experiments aiming to establish whether the central and peripheral effects of capsaicin onto the internalization of the preferred substance P receptor NK1 had different mechanisms led to the demonstration that capsaicin produced neurokinin release by a direct action, i.e., a TRPV1-mediated influx of Ca2+, on primary afferents terminals. The vanilloid also increased the firing of action potentials, and the first effect but not the second largely bypassed NMDA and GABAB modulatory mechanisms [218]. These observations were consistent with the previous demonstration that capsaicin induced a strong rise of [Ca2+]i in rat spinal cord slices during the course of laser scanning confocal microscope imaging experiments [219]. Converging to the demonstration of a cross-talk between central TRPV1 and peptide release, N-(4-tertiarybutylphenyl)-4-(3-cholorphyridin-2-yl)tetrahydropryazine-1(2H)-carbox-amide, a selective TRPV1 antagonist, inhibited the capsaicin-induced release of CGRP and substance P in rat spinal cord slices [220]. Subsequent electrophysiological observations showed that primary afferent stimulation with capsaicin differentially potentiated excitatory and inhibitory inputs to spinal lamina II outer and inner neurons [221]. Moreover, in another study, administration of capsaicin to lamina II neurons in slices from mice pups resulted in an increase of spontaneous inhibitory postsynaptic currents. GABAergic inhibitory interneurons in laminae I, III and IV were excited because of the release of substance P, which, in turn, reduced the activity of lamina II neurons [14]. Experiments on lamina I neurons also demonstrated that purinergic P2X receptor-expressing fibers were capsaicin-sensitive nociceptive afferents [222].
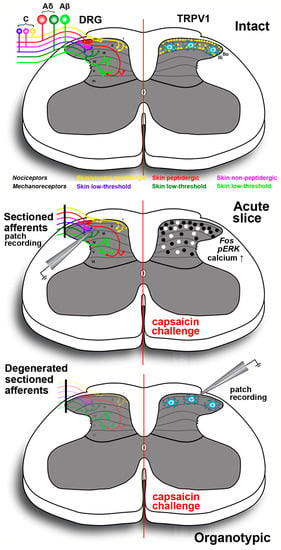
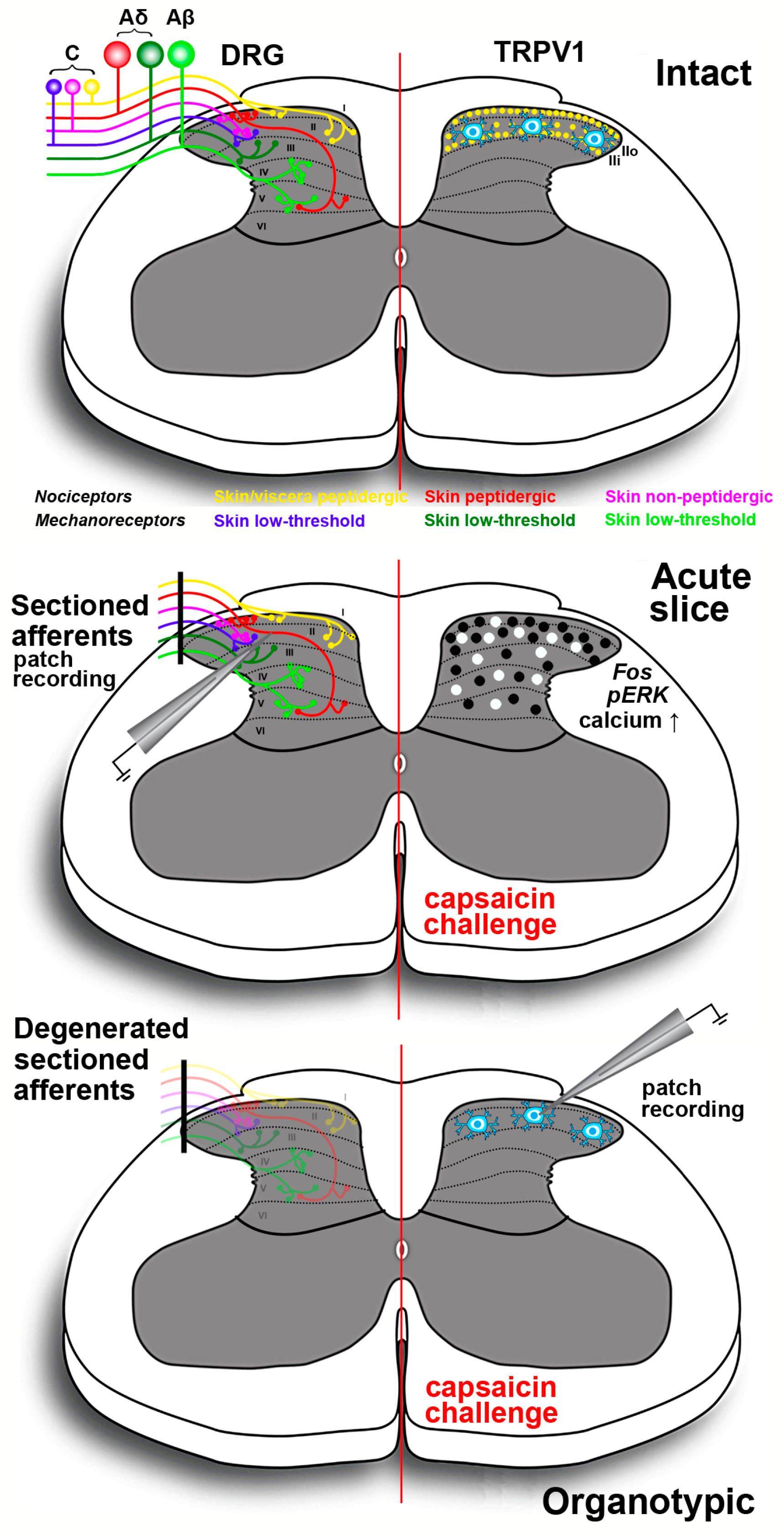
Figure 3.
Schematic drawing of the spinal cord slices preparations exploiting the agonist action of capsaicin onto TRPV1 expressing neurons and fibers in the dorsal horn. Top panel shows at left the laminar distribution of primary afferent terminals originating from different classes of nociceptors in DRG neurons; at right the distribution of TRPV1 in peptidergic terminals (yellow dots) originating from skin and viscera in laminae I-II. Receptors are less densely localized in the outer part of lamina II (IIo) as compared to lamina I and the inner part of lamina II (IIi). Lamina II also contains TRPV1-IR interneurons that are spared in organotypically cultured slices (bottom panel). For simplicity, proprioceptive muscle afferents are not represented. In acute slices (middle panel) primary afferents are severed at dorsal roots but their terminals remain functional for several hours allowing performing electrophysiological recording, calcium imaging and proto-oncogene c-Fos/pERK immunocytochemistry. The black dots indicate the distribution of proto-oncogene c-Fos/pERK-IR neurons after capsaicin challenge. White dots exemplify the location of the cells responding to the vanilloid with increased intracellular calcium concentration in real-time confocal imaging. In organotypic cultures (bottom panel) sectioned primary afferents degenerate and thus, only the TRPV1-IR lamina II neurons are spared. This type of preparation is useful to isolate the effects of TRPV1 activation on these neurons, in the absence of primary afferent input.
Other investigators used a different approach exploiting the use of capsaicin as a tool to stimulate peptidergic primary afferents in acute slices. This approach was based on the immunocytochemical localization of pERK or expression of the proto-oncogene c-Fos. ERK phosphorylation is known to occur in central sensitization and capsaicin stimulation of C fibers induced eight to tenfold increase of pERK in superficial dorsal horn neurons [223]. The effect was similar to that obtained after direct electrical stimulation of these fibers or intraplantar injection of capsaicin in an intact animal. More recently, a study has compared the pERK and proto-oncogene c-Fos responses of mouse spinal cord slices subjected to octreotide administration. Octreotide is a synthetic antinociceptive analog of somatostatin, one of the neuropeptides involved in the negative modulation of pain signals in the dorsal horn. In acute slices, octreotide reduced the response to capsaicin as measured by expression of proto-oncogene c-Fos and pERK, and it was concluded that the use of Fos and pERK immunoreactivity in vitro was a valuable tool in investigating the activation of spinal nociceptive pathways and to test potentially antinociceptive molecules [224].
Other ex vivo approaches were based on the use of real-time confocal imaging of cell-permeant calcium indicators or voltage-sensitive dyes. The strong calcium response induced by capsaicin was used in acute rat or mouse slices to mimic the central effects of inflammation and to study the ying and yang of BDNF and GDNF in the modulation of the excitatory and inhibitory input to lamina II interneurons [225,226]. Finally, the neurotoxic effects of neonatal capsaicin were exploited to prepare slices from C fibers’ depleted rats and to study the effects of GABA and excitatory amino-acid receptors antagonists on the primary afferent excitatory input to the spinal dorsal horn, by imaging slices from young pups after anterograde labeling with a voltage-sensitive dye from the dorsal root attached to the spinal cord slice [227].
4. Therapeutic Use of Capsaicin
Capsaicin has been used in several clinical settings as a topical medication to treat pain derived from different conditions. The USA regulatory authorities have approved capsaicin as an 8% dermal patch for treating local pain. These patches contain 640 mcg/cm2 synthetic capsaicin with a total dose of 179 mg in one patch [228]. Lower doses showed no clinical benefits [229,230,231] or only short-term efficacy [232,233]. In general, it seems that the vanilloid is effective in the treatment of neuropathic but not inflammatory pain. For example, capsaicin was not effective against inflammatory osteoarthritic pain [234], and a high-dose capsaicin patch was of no benefit in targeting persistent pain after inguinal herniorrhaphy [235]. However, instillation of 15 mg capsaicin (Anesiva 4975) immediately prior to wound closure displayed some efficacy in controlling pain after total knee arthroplasty [236], and the vanilloid reduced abdominal pain of patients affected by irritable bowel syndrome after oral administration [237]. Notably, zucapsaicin, a synthetic cis isomer of natural capsaicin [238] was shown to be therapeutically effective in relieving pain deriving from knee osteoarthritis. The mechanism of action and clinical indications of zucapsaicin are similar to that capsaicin, but zucapsaicin is better tolerated. Therefore, zucapsaicin could become a valuable drug for treating certain forms of inflammatory pain such as osteoarthritic or intestinal pain and headaches.
An 8% capsaicin patch was reported to be safe and effective in controlling neuropathic pain resulting from several conditions, with clear improvement in pain attacks, sleep duration and quality of life [239]. Local pain and erythema were the commonest adverse effects in about 10% of the patients. Capsaicin patch preparations (NGX-4010 or Qutenza®) were also of benefit in post-herpetic neuralgia and painful HIV-associated distal sensory polyneuropathy, alone [239,240,241,242,243,244,245,246,247,248] or in association with 4% lidocaine topical preparations [249,250]. Notably, the outcomes of a recent interdisciplinary expert workshop lead to the conclusion that response rates in patients with or without lidocaine pretreatment were comparable [251]. According to Derry et al. 2013 [252], high-concentration topical capsaicin is similar to other therapies for chronic pain. However, the high cost of single and repeated applications suggest that such a therapy should be preferentially used when other available therapies have failed. Moreover, the same authors concluded that this type of treatment should not be used repeatedly without substantial documented pain relief as, even when efficacy is established, there are unknown risks of repeated application for long periods, especially on epidermal innervation. In addition, it should be mentioned that a revision of the Special Interest Group on Neuropathic Pain (NeuPSIG) recommendations for the pharmacotherapy of neuropathic pain, based on the results of a systematic review and meta-analysis of existing clinical data, only led to weak recommendation for the use and proposal as second line therapy of capsaicin high-concentration patches [253].
Injectable capsaicin preparations are also in the course of clinical evaluation. Adlea (ALGRX-4975) is an injectable highly purified form of capsaicin formulated for long lasting pain relief [254]. It is currently under investigation for treatment of intermetatarsal neuromas [255], lateral epicondylitis [256] and end stage osteoarthritis [257]. However, a recent study has tested the effects of intradermal capsaicin in healthy volunteers and led to the conclusion that an injection of capsaicin at different depths in the skin had effects on heart rate and blood pressure but not on pain [258]. Authors worried that their results might have implications for the pharmacology and analgesic predictive value of the model of intradermal capsaicin.
Palvanil (N-palmitoyl-vanillamide) is a non-pungent capsaicin- like compound found in low amounts in Capsicum plants [259]. Palvanil has slower kinetics of TRPV1 activation and is a stronger desensitizer of TRPV1 than capsaicin [259]. When administered systemically at analgesic doses in mice, it produced significantly fewer aversive effects on body temperature and bronchoconstriction as compared to capsaicin [260]. These observations have a potential for subsequent translational studies.
Acknowledgments
The original work described in this paper was funded by local grants from the University of Turin.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| 12-HPETE | 12-hydroperoxyeicosatetraenoic acid |
| ATP | Adenosine triphosphate |
| CaMK II kinase | Calmodulin- dependent protein kinase II |
| CGRP | Calcitonin Gene-Related Peptide |
| CP/CPPS | Chronic Prostatitis/Chronic Pelvic Pain Syndrome |
| DNA | Deoxyribonucleic acid |
| DRG | Dorsal Root Ganglion |
| ENS | Enteric Nervous System |
| GABA | γ-Aminobutyric acid |
| GI tract | Gastrointestinal tract |
| HEK 293 | Human Embryonic Kidney 293 |
| IR | Immunoreactive |
| 15-HPETE | 15-hydroperoxyeicosatetraenoic acid |
| BDNF | Brain Derived Neurotrophic Factor |
| cDNA | complementary DNA |
| CNS | Central Nervous System |
| DkTx | Double-knot toxin |
| DOCA | Deoxycorticosterone acetate |
| DSS | Dextran sulfate sodium |
| FAF1 | Fas-Associated Factor 1 |
| GDNF | Glial cell-Derived Neurotrophic Factor |
| HCl | Acid chloride |
| ICC | Immunocytochemistry |
| KO | Knock-out |
| LB4 | Leukotriene B4 |
| LPS | Lipopolysaccharide |
| NADA | Arachidonoyl-dopamine |
| NGF | Nerve Growth Factor |
| NKA | Neurokinin A |
| NMDA receptor | N-methyl-D-aspartate receptor |
| NOS | Nitric Oxide Synthase |
| NTS | Nucleus Tractus Solitarius |
| PAG | Periaqueductal grey |
| Palvanil | N-palmitoyl-vanillamide |
| pERK | Phosphorylated Extracellular signal-Regulated Kinase |
| PI3K | Phosphatidylinositol-3-Kinase |
| PIP2 | Phosphatidylinositol 4,5-bisphosphate |
| PKB/Akt | Protein Kinase B/Akt |
| PKC | Protein Kinase C |
| PNS | Peripheral Nervous System |
| PSNs | Primary Sensory Neurons |
| RT-PCR | Reverse Transcription-Polymerase Chain Reaction |
| RTX | Resinferatoxin |
| SNTN | Spinal Nucleus of the Trigeminal Nerve |
| STAT3 | Signal Transducer and Activator of Transcription 3 |
| STT | Spinothalamic Tract |
| TG | Trigeminal Ganglion |
| TNBS | Trinitrobenzene sulphonic acid |
| TrkA | Tropomyosin kinase A receptor |
| TRP | Transient Receptor Potential |
| TRPV1 | Transient Receptor Potential cation channel Vanilloid subfamily member 1 |
| VaTx | Vanillotoxins |
| VR1 | Vanilloid Receptor subtype 1 |
| WT | Wild Type |
References
- Thresh, J.C. Isolation of capsacin. Pharm. J. Tr. 1876, 6, 941–947. [Google Scholar]
- Nelson, E.K. The constitution of capsaicin, the pungent principle of capsicum. II. J. Am. Chem. Soc. 1920, 42, 597–599. [Google Scholar]
- De, A.K. (Ed.) Capsicum: The Genus Capsicum; CRC Press: London, UK, 2003.
- Musfiroh, I.; Mutakin, M.; Angelina, T.; Muchtaridi, M. Capsaicin level of various capsicum fruits. Int. J. Pharm. Pharm. Sci. 2013, 5, 248–251. [Google Scholar]
- Caterina, M.J.; Schumacher, M.A.; Tominaga, M.; Rosen, T.A.; Levine, J.D.; Julius, D. The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature 1997, 389, 816–824. [Google Scholar] [PubMed]
- Krause, J.E.; Chenard, B.L.; Cortright, D.N. Transient receptor potential ion channels as targets for the discovery of pain therapeutics. Curr. Opin. Investig. Drugs 2005, 6, 48–57. [Google Scholar] [PubMed]
- Hayes, P.; Meadows, H.J.; Gunthorpe, M.J.; Harries, M.H.; Duckworth, D.M.; Cairns, W.; Harrison, D.C.; Clarke, C.E.; Ellington, K.; Prinjha, R.K.; et al. Cloning and functional expression of a human orthologue of rat vanilloid receptor-1. Pain 2000, 88, 205–215. [Google Scholar] [CrossRef]
- Mezey, E.; Toth, Z.E.; Cortright, D.N.; Arzubi, M.K.; Krause, J.E.; Elde, R.; Guo, A.; Blumberg, P.M.; Szallasi, A. Distribution of mrna for vanilloid receptor subtype 1 (vr1), and vr1-like immunoreactivity, in the central nervous system of the rat and human. Proc. Natl. Acad. Sci. USA 2000, 97, 3655–3660. [Google Scholar] [CrossRef] [PubMed]
- Birder, L.A.; Kanai, A.J.; de Groat, W.C.; Kiss, S.; Nealen, M.L.; Burke, N.E.; Dineley, K.E.; Watkins, S.; Reynolds, I.J.; Caterina, M.J. Vanilloid receptor expression suggests a sensory role for urinary bladder epithelial cells. Proc. Natl. Acad. Sci. USA 2001, 98, 13396–13401. [Google Scholar] [CrossRef] [PubMed]
- Cavanaugh, D.J.; Chesler, A.T.; Braz, J.M.; Shah, N.M.; Julius, D.; Basbaum, A.I. Restriction of transient receptor potential vanilloid-1 to the peptidergic subset of primary afferent neurons follows its developmental downregulation in nonpeptidergic neurons. J. Neurosci. 2011, 31, 10119–10127. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Hoon, M.A. Ablation of trpv1 neurons reveals their selective role in thermal pain sensation. Mol. Cell. Neurosci. 2010, 43, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Tisel, S.M.; Orestes, P.; Bhangoo, S.K.; Hoon, M.A. Trpv1-lineage neurons are required for thermal sensation. EMBO J. 2011, 30, 582–593. [Google Scholar] [CrossRef] [PubMed]
- Cristino, L.; de Petrocellis, L.; Pryce, G.; Baker, D.; Guglielmotti, V.; di Marzo, V. Immunohistochemical localization of cannabinoid type 1 and vanilloid transient receptor potential vanilloid type 1 receptors in the mouse brain. Neuroscience 2006, 139, 1405–1415. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, F.; Salio, C.; Vergnano, A.M.; Merighi, A. Vanilloid receptor-1 (trpv1)-dependent activation of inhibitory neurotransmission in spinal substantia gelatinosa neurons of mouse. Pain 2007, 129, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Cavanaugh, D.J.; Chesler, A.T.; Jackson, A.C.; Sigal, Y.M.; Yamanaka, H.; Grant, R.; O'Donnell, D.; Nicoll, R.A.; Shah, N.M.; Julius, D.; et al. Trpv1 reporter mice reveal highly restricted brain distribution and functional expression in arteriolar smooth muscle cells. J. Neurosci. 2011, 31, 5067–5077. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome Biol 2011, 12, 218. [Google Scholar] [CrossRef] [PubMed]
- Caterina, M.J.; Leffler, A.; Malmberg, A.B.; Martin, W.J.; Trafton, J.; Petersen-Zeitz, K.R.; Koltzenburg, M.; Basbaum, A.I.; Julius, D. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 2000, 288, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Smart, D.; Gunthorpe, M.J.; Jerman, J.C.; Nasir, S.; Gray, J.; Muir, A.I.; Chambers, J.K.; Randall, A.D.; Davis, J.B. The endogenous lipid anandamide is a full agonist at the human vanilloid receptor (hvr1). Br. J. Pharmacol. 2000, 129, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Correll, C.C.; Phelps, P.T.; Anthes, J.C.; Umland, S.; Greenfeder, S. Cloning and pharmacological characterization of mouse trpv1. Neurosci Lett 2004, 370, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Smart, D.; Jerman, J.C.; Gunthorpe, M.J.; Brough, S.J.; Ranson, J.; Cairns, W.; Hayes, P.D.; Randall, A.D.; Davis, J.B. Characterisation using flipr of human vanilloid vr1 receptor pharmacology. Eur. J. Pharmacol. 2001, 417, 51–58. [Google Scholar] [CrossRef]
- Takayama, Y.; Uta, D.; Furue, H.; Tominaga, M. Pain-enhancing mechanism through interaction between trpv1 and anoctamin 1 in sensory neurons. Proc. Natl. Acad. Sci. USA 2015, 112, 5213–5218. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Vij, A.S.; Sharma, M. Mechanisms and clinical uses of capsaicin. Eur. J. Pharmacol. 2013, 720, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Tympanidis, P.; Casula, M.A.; Yiangou, Y.; Terenghi, G.; Dowd, P.; Anand, P. Increased vanilloid receptor vr1 innervation in vulvodynia. Eur. J. Pain 2004, 8, 129–133. [Google Scholar] [CrossRef]
- Geppetti, P.; Trevisani, M. Activation and sensitisation of the vanilloid receptor: Role in gastrointestinal inflammation and function. Br. J. Pharmacol. 2004, 141, 1313–1320. [Google Scholar] [CrossRef] [PubMed]
- Yiangou, Y.; Facer, P.; Dyer, N.H.; Chan, C.L.; Knowles, C.; Williams, N.S.; Anand, P. Vanilloid receptor 1 immunoreactivity in inflamed human bowel. Lancet 2001, 357, 1338–1339. [Google Scholar] [CrossRef]
- Bhutani, M.; Pathak, A.K.; Nair, A.S.; Kunnumakkara, A.B.; Guha, S.; Sethi, G.; Aggarwal, B.B. Capsaicin is a novel blocker of constitutive and interleukin-6-inducible stat3 activation. Clin. Cancer Res. 2007, 13, 3024–3032. [Google Scholar] [CrossRef] [PubMed]
- Donnerer, J.; Liebmann, I. The nk1 receptor antagonist sr140333 inhibits capsaicin-induced erk phosphorylation in sensory neurons. Pharmacology 2006, 77, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.Y.; Ling, Q.Z.; Chen, S.J. Identification of a potential target of capsaicin by computational target fishing. Evid. Based Complement. Alternat. Med. 2015, 2015, 983951. [Google Scholar] [CrossRef] [PubMed]
- Merighi, A.; Salio, C.; Ghirri, A.; Lossi, L.; Ferrini, F.; Betelli, C.; Bardoni, R. Bdnf as a pain modulator. Prog. Neurobiol. 2008, 85, 297–317. [Google Scholar] [CrossRef] [PubMed]
- Bonica, J.J. Evolution and current status of pain programs. J. Pain Symptom Manage. 1990, 5, 368–374. [Google Scholar] [CrossRef]
- Hunt, S.P.; Mantyh, P.W. The molecular dynamics of pain control. Nat. Rev. Neurosci. 2001, 2, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Cervero, F.; Laird, J.M. Visceral pain. Lancet 1999, 353, 2145–2148. [Google Scholar] [CrossRef]
- McMahon, S.B.; Dmitrieva, N.; Koltzenburg, M. Visceral pain. Br. J. Anaesth. 1995, 75, 132–144. [Google Scholar] [CrossRef] [PubMed]
- Abrahams, V.C.; Swett, J.E. The pattern of spinal and medullary projections from a cutaneous nerve and a muscle nerve of the forelimb of the cat: A study using the transganglionic transport of hrp. J. Comp. Neurol. 1986, 246, 70–84. [Google Scholar] [CrossRef] [PubMed]
- Morgan, C.; Nadelhaft, I.; deGroat, W.C. The distribution within the spinal cord of visceral primary afferent axons carried by the lumbar colonic nerve of the cat. Brain Res. 1986, 398, 11–17. [Google Scholar] [CrossRef]
- Giamberardino, M.A. Recent and forgotten aspects of visceral pain. Eur. J. Pain 1999, 3, 77–92. [Google Scholar] [CrossRef] [PubMed]
- Sikandar, S.; Dickenson, A.H. Visceral pain: The ins and outs, the ups and downs. Curr. Opin. Support. Palliat. Care 2012, 6, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Blackshaw, L.A. Visceral pain readouts in experimental medicine. Neurogastroenterol. Motil. 2012, 24, 891–894. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, J.N. Visceral pain: The neurophysiological mechanism. Handb. Exp. Pharmacol. 2009, 31–74. [Google Scholar]
- Gebhart, G.F.J.J. Bonica lecture--2000: Physiology, pathophysiology, and pharmacology of visceral pain. Reg. Anesth. Pain Med. 2000, 25, 632–638. [Google Scholar] [CrossRef] [PubMed]
- Gebhart, G.F. Pathobiology of visceral pain: Molecular mechanisms and therapeutic implications iv. Visceral afferent contributions to the pathobiology of visceral pain. Am. J. Physiol. Gastrointest. Liver Physiol. 2000, 278, G834–G838. [Google Scholar] [PubMed]
- Todd, A.J. Anatomy of primary afferents and projection neurones in the rat spinal dorsal horn with particular emphasis on substance p and the neurokinin 1 receptor. Exp. Physiol. 2002, 87, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Bester, H.; Menendez, L.; Besson, J.M.; Bernard, J.F. Spino (trigemino) parabrachiohypothalamic pathway: Electrophysiological evidence for an involvement in pain processes. J. Neurophysiol. 1995, 73, 568–585. [Google Scholar] [PubMed]
- Bushnell, M.C.; Duncan, G.H. Sensory and affective aspects of pain perception: Is medial thalamus restricted to emotional issues? Exp. Brain Res. 1989, 78, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Groenewegen, H.J. Organization of the afferent connections of the mediodorsal thalamic nucleus in the rat, related to the mediodorsal-prefrontal topography. Neuroscience 1988, 24, 379–431. [Google Scholar] [CrossRef]
- Mertz, H.; Morgan, V.; Tanner, G.; Pickens, D.; Price, R.; Shyr, Y.; Kessler, R. Regional cerebral activation in irritable bowel syndrome and control subjects with painful and nonpainful rectal distention. Gastroenterology 2000, 118, 842–848. [Google Scholar] [CrossRef]
- Furness, J.B. Types of neurons in the enteric nervous system. J. Auton Nerv. Syst. 2000, 81, 87–96. [Google Scholar] [CrossRef]
- Furness, J.B. Novel gut afferents: Intrinsic afferent neurons and intestinofugal neurons. Auton Neurosci. 2006, 125, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Meghvansi, M.K.; Siddiqui, S.; Khan, M.H.; Gupta, V.K.; Vairale, M.G.; Gogoi, H.K.; Singh, L. Naga chilli: A potential source of capsaicinoids with broad-spectrum ethnopharmacological applications. J. Ethnopharmacol. 2010, 132, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bhagowati, R.R.; Changkija, S. Genetic variability and traditional practices in naga king chili of nagaland. Asian Agri-Hist. 2009, 13, 171–180. [Google Scholar]
- Vetter, I.; Cheng, W.; Peiris, M.; Wyse, B.D.; Roberts-Thomson, S.J.; Zheng, J.; Monteith, G.R.; Cabot, P.J. Rapid, opioid-sensitive mechanisms involved in transient receptor potential vanilloid 1 sensitization. J. Biol. Chem. 2008, 283, 19540–19550. [Google Scholar] [CrossRef] [PubMed]
- Liapi, A.; Wood, J.N. Extensive co-localization and heteromultimer formation of the vanilloid receptor-like protein trpv2 and the capsaicin receptor trpv1 in the adult rat cerebral cortex. Eur. J. Neurosci. 2005, 22, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Hu, H.Z.; Colton, C.K.; Wood, J.D.; Zhu, M.X. An alternative splicing product of the murine trpv1 gene dominant negatively modulates the activity of trpv1 channels. J. Biol. Chem. 2004, 279, 37423–37430. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.; Zavala, K.; Fahimi, A.; Lee, J.; Xue, Q.; Eilers, H.; Schumacher, M.A. Transcription factors sp1 and sp4 regulate trpv1 gene expression in rat sensory neurons. Mol. Pain 2011, 7, 44. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.A.; Eilers, H. Trpv1 splice variants: Structure and function. Front. Biosci. (Landmark Ed.) 2010, 15, 872–882. [Google Scholar] [CrossRef] [PubMed]
- Lyall, V.; Heck, G.L.; Vinnikova, A.K.; Ghosh, S.; Phan, T.H.; Alam, R.I.; Russell, O.F.; Malik, S.A.; Bigbee, J.W.; DeSimone, J.A. The mammalian amiloride-insensitive non-specific salt taste receptor is a vanilloid receptor-1 variant. J. Physiol. 2004, 558, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Charrua, A.; Cruz, C.D.; Narayanan, S.; Gharat, L.; Gullapalli, S.; Cruz, F.; Avelino, A. Grc-6211, a new oral specific trpv1 antagonist, decreases bladder overactivity and noxious bladder input in cystitis animal models. J. Urol. 2009, 181, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Caterina, M.J.; Malmberg, A.B.; Rosen, T.A.; Gilbert, H.; Skinner, K.; Raumann, B.E.; Basbaum, A.I.; Julius, D. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron 1998, 21, 531–543. [Google Scholar] [CrossRef]
- Alexander, S.P.; Catterall, W.A.; Kelly, E.; Marrion, N.; Peters, J.A.; Benson, H.E.; Faccenda, E.; Pawson, A.J.; Sharman, J.L.; Southan, C.; et al. The concise guide to pharmacology 2015/16: Voltage-gated ion channels. Br. J. Pharmacol. 2015, 172, 5904–5941. [Google Scholar] [CrossRef] [PubMed]
- Planells-Cases, R.; Garcia-Sanz, N.; Morenilla-Palao, C.; Ferrer-Montiel, A. Functional aspects and mechanisms of trpv1 involvement in neurogenic inflammation that leads to thermal hyperalgesia. Pflugers Arch. 2005, 451, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Szallasi, A.; Cortright, D.N.; Blum, C.A.; Eid, S.R. The vanilloid receptor trpv1: 10 years from channel cloning to antagonist proof-of-concept. Nat. Rev. Drug Discov. 2007, 6, 357–372. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, D.P.; Nau, C. Regulation of ca2+-dependent desensitization in the vanilloid receptor trpv1 by calcineurin and camp-dependent protein kinase. J. Biol. Chem. 2005, 280, 13424–13432. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, D.P.; Nau, C. Desensitization of capsaicin-activated currents in the vanilloid receptor trpv1 is decreased by the cyclic amp-dependent protein kinase pathway. J. Biol. Chem. 2003, 278, 50080–50090. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, T.; Simon, S.A. Trpv1 receptors and signal transduction. In Trp ion Channel Function in Sensory Transduction and Cellular Signaling Cascades; Liedtke, W.B., Heller, S., Eds.; Taylor & Francis Group: Boca Raton, FL, USA, 2007. [Google Scholar]
- Ma, W.; Quirion, R. Inflammatory mediators modulating the transient receptor potential vanilloid 1 receptor: Therapeutic targets to treat inflammatory and neuropathic pain. Expert Opin. Ther. Targets 2007, 11, 307–320. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yu, H.M.; Zhou, X.D.; Kolosov, V.P.; Perelman, J.M. Study on trpv1-mediated mechanism for the hypersecretion of mucus in respiratory inflammation. Mol. Immunol. 2013, 53, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Szallasi, A.; Blumberg, P.M. Complex regulation of trpv1 by vanilloids. In Trp Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades; Liedtke, W.B., Heller, S., Eds.; Taylor & Francis Group: Boca Raton, FL, USA, 2007. [Google Scholar]
- Planells-Cases, R.; Valente, P.; Ferrer-Montiel, A.; Qin, F.; Szallasi, A. Complex regulation of trpv1 and related thermo-trps: Implications for therapeutic intervention. Adv. Exp. Med. Biol. 2011, 704, 491–515. [Google Scholar] [PubMed]
- Kim, S.; Kang, C.; Shin, C.Y.; Hwang, S.W.; Yang, Y.D.; Shim, W.S.; Park, M.Y.; Kim, E.; Kim, M.; Kim, B.M.; et al. Trpv1 recapitulates native capsaicin receptor in sensory neurons in association with fas-associated factor 1. J. Neurosci. 2006, 26, 2403–2412. [Google Scholar] [CrossRef] [PubMed]
- Richardson, J.D.; Vasko, M.R. Cellular mechanisms of neurogenic inflammation. J. Pharmacol. Exp. Ther. 2002, 302, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Veronesi, B.; Oortgiesen, M. The trpv1 receptor: Target of toxicants and therapeutics. Toxicol. Sci. 2006, 89, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Jordt, S.E.; Tominaga, M.; Julius, D. Acid potentiation of the capsaicin receptor determined by a key extracellular site. Proc. Natl. Acad. Sci. USA 2000, 97, 8134–8139. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Wada, M.; Masu, M. Potentiation of capsaicin receptor activity by metabotropic atp receptors as a possible mechanism for atp-evoked pain and hyperalgesia. Proc. Natl. Acad. Sci. USA 2001, 98, 6951–6956. [Google Scholar] [CrossRef] [PubMed]
- Ahern, G.P.; Wang, X.; Miyares, R.L. Polyamines are potent ligands for the capsaicin receptor trpv1. J. Biol. Chem. 2006, 281, 8991–8995. [Google Scholar] [CrossRef] [PubMed]
- Hwang, S.W.; Cho, H.; Kwak, J.; Lee, S.Y.; Kang, C.J.; Jung, J.; Cho, S.; Min, K.H.; Suh, Y.G.; Kim, D.; et al. Direct activation of capsaicin receptors by products of lipoxygenases: Endogenous capsaicin-like substances. Proc. Natl. Acad. Sci. USA 2000, 97, 6155–6160. [Google Scholar] [CrossRef] [PubMed]
- McHugh, D.; McMaster, R.S.; Pertwee, R.G.; Roy, S.; Mahadevan, A.; Razdan, R.K.; Ross, R.A. Novel compounds that interact with both leukotriene b4 receptors and vanilloid trpv1 receptors. J. Pharmacol. Exp. Ther. 2006, 316, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.M.; Bisogno, T.; Trevisani, M.; Al-Hayani, A.; De Petrocellis, L.; Fezza, F.; Tognetto, M.; Petros, T.J.; Krey, J.F.; Chu, C.J.; et al. An endogenous capsaicin-like substance with high potency at recombinant and native vanilloid vr1 receptors. Proc. Natl. Acad. Sci. USA 2002, 99, 8400–8405. [Google Scholar] [CrossRef] [PubMed]
- Zygmunt, P.M.; Petersson, J.; Andersson, D.A.; Chuang, H.; Sorgard, M.; Di Marzo, V.; Julius, D.; Hogestatt, E.D. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature 1999, 400, 452–457. [Google Scholar] [PubMed]
- Moriyama, T.; Higashi, T.; Togashi, K.; Iida, T.; Segi, E.; Sugimoto, Y.; Tominaga, T.; Narumiya, S.; Tominaga, M. Sensitization of trpv1 by ep1 and ip reveals peripheral nociceptive mechanism of prostaglandins. Mol. Pain 2005, 1, 3. [Google Scholar] [CrossRef] [PubMed]
- Cesare, P.; Dekker, L.V.; Sardini, A.; Parker, P.J.; McNaughton, P.A. Specific involvement of pkc-epsilon in sensitization of the neuronal response to painful heat. Neuron 1999, 23, 617–624. [Google Scholar] [CrossRef]
- Chuang, H.H.; Prescott, E.D.; Kong, H.; Shields, S.; Jordt, S.E.; Basbaum, A.I.; Chao, M.V.; Julius, D. Bradykinin and nerve growth factor release the capsaicin receptor from ptdins(4,5)p2-mediated inhibition. Nature 2001, 411, 957–962. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Cho, H.; Hwang, S.W.; Jung, J.; Shin, C.Y.; Lee, S.Y.; Kim, S.H.; Lee, M.G.; Choi, Y.H.; Kim, J.; et al. Bradykinin-12-lipoxygenase-vr1 signaling pathway for inflammatory hyperalgesia. Proc. Natl. Acad. Sci. USA 2002, 99, 10150–10155. [Google Scholar] [CrossRef] [PubMed]
- Negri, L.; Lattanzi, R.; Giannini, E.; Colucci, M.; Margheriti, F.; Melchiorri, P.; Vellani, V.; Tian, H.; de Felice, M.; Porreca, F. Impaired nociception and inflammatory pain sensation in mice lacking the prokineticin receptor pkr1: Focus on interaction between pkr1 and the capsaicin receptor trpv1 in pain behavior. J. Neurosci. 2006, 26, 6716–6727. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, L.S.; Ahern, G.P. Induction of vanilloid receptor channel activity by protein kinase c. Nature 2000, 408, 985–990. [Google Scholar] [CrossRef] [PubMed]
- McNamara, F.N.; Randall, A.; Gunthorpe, M.J. Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (trpv1). Br. J. Pharmacol. 2005, 144, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Blair, N.T.; Clapham, D.E. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in a vanilloid-independent mechanism. J. Neurosci. 2005, 25, 8924–8937. [Google Scholar] [CrossRef] [PubMed]
- Cuypers, E.; Yanagihara, A.; Karlsson, E.; Tytgat, J. Jellyfish and other cnidarian envenomations cause pain by affecting trpv1 channels. FEBS Lett. 2006, 580, 5728–5732. [Google Scholar] [CrossRef] [PubMed]
- Siemens, J.; Zhou, S.; Piskorowski, R.; Nikai, T.; Lumpkin, E.A.; Basbaum, A.I.; King, D.; Julius, D. Spider toxins activate the capsaicin receptor to produce inflammatory pain. Nature 2006, 444, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Bohlen, C.J.; Priel, A.; Zhou, S.; King, D.; Siemens, J.; Julius, D. A bivalent tarantula toxin activates the capsaicin receptor, trpv1, by targeting the outer pore domain. Cell 2010, 141, 834–845. [Google Scholar] [CrossRef] [PubMed]
- Sluka, K.A.; Willis, W.D. The effects of g-protein and protein kinase inhibitors on the behavioral responses of rats to intradermal injection of capsaicin. Pain 1997, 71, 165–178. [Google Scholar] [CrossRef]
- Zou, X.; Lin, Q.; Willis, W.D. Role of protein kinase a in phosphorylation of nmda receptor 1 subunits in dorsal horn and spinothalamic tract neurons after intradermal injection of capsaicin in rats. Neuroscience 2002, 115, 775–786. [Google Scholar] [CrossRef]
- Sun, R.; Yan, J.; Willis, W.D. Activation of protein kinase b/akt in the periphery contributes to pain behavior induced by capsaicin in rats. Neuroscience 2007, 144, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Gamse, R.; Petsche, U.; Lembeck, F.; Jancso, G. Capsaicin applied to peripheral nerve inhibits axoplasmic transport of substance p and somatostatin. Brain Res. 1982, 239, 447–462. [Google Scholar] [CrossRef]
- Anand, P.; Bley, K. Topical capsaicin for pain management: Therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br. J. Anaesth. 2011, 107, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Johansen, M.E.; Reilly, C.A.; Yost, G.S. Trpv1 antagonists elevate cell surface populations of receptor protein and exacerbate trpv1-mediated toxicities in human lung epithelial cells. Toxicol. Sci. 2006, 89, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Thomas, K.C.; Sabnis, A.S.; Johansen, M.E.; Lanza, D.L.; Moos, P.J.; Yost, G.S.; Reilly, C.A. Transient receptor potential vanilloid 1 agonists cause endoplasmic reticulum stress and cell death in human lung cells. J. Pharmacol. Exp. Ther. 2007, 321, 830–838. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.L.; Khan, G.M.; Alloway, K.D.; Chen, S.R. Resiniferatoxin induces paradoxical changes in thermal and mechanical sensitivities in rats: Mechanism of action. J. Neurosci. 2003, 23, 2911–2919. [Google Scholar] [PubMed]
- Nolano, M.; Simone, D.A.; Wendelschafer-Crabb, G.; Johnson, T.; Hazen, E.; Kennedy, W.R. Topical capsaicin in humans: Parallel loss of epidermal nerve fibers and pain sensation. Pain 1999, 81, 135–145. [Google Scholar] [CrossRef]
- Sikand, P.; Premkumar, L.S. Potentiation of glutamatergic synaptic transmission by protein kinase c-mediated sensitization of trpv1 at the first sensory synapse. J. Physiol. 2007, 581, 631–647. [Google Scholar] [CrossRef] [PubMed]
- Treede, R.D.; Meyer, R.A.; Raja, S.N.; Campbell, J.N. Peripheral and central mechanisms of cutaneous hyperalgesia. Prog. Neurobiol. 1992, 38, 397–421. [Google Scholar] [CrossRef]
- Ringkamp, M.; Meyer, R.A. Physiology of nociceptors. Pain 2008, 97–114. [Google Scholar]
- Simone, D.A.; Ochoa, J. Early and late effects of prolonged topical capsaicin on cutaneous sensibility and neurogenic vasodilatation in humans. Pain 1991, 47, 285–294. [Google Scholar] [CrossRef]
- Willis, W.D.; Coggeshall, R.E. (Eds.) Sensory Mechanisms of the Spinal Cord. Primary Afferent Neurons and the Spinal Dorsal Horn; Plenum Press: Berlin, Germany, 2004.
- Zou, X.; Lin, Q.; Willis, W.D. Nmda or non-nmda receptor antagonists attenuate increased fos expression in spinal dorsal horn gabaergic neurons after intradermal injection of capsaicin in rats. Neuroscience 2001, 106, 171–182. [Google Scholar] [CrossRef]
- Millan, M.J. Descending control of pain. Prog. Neurobiol. 2002, 66, 355–474. [Google Scholar] [CrossRef]
- Doyle, M.W.; Bailey, T.W.; Jin, Y.H.; Andresen, M.C. Vanilloid receptors presynaptically modulate cranial visceral afferent synaptic transmission in nucleus tractus solitarius. J. Neurosci. 2002, 22, 8222–8229. [Google Scholar] [PubMed]
- McGaraughty, S.; Chu, K.L.; Bitner, R.S.; Martino, B.; El Kouhen, R.; Han, P.; Nikkel, A.L.; Burgard, E.C.; Faltynek, C.R.; Jarvis, M.F. Capsaicin infused into the pag affects rat tail flick responses to noxious heat and alters neuronal firing in the rvm. J. Neurophysiol. 2003, 90, 2702–2710. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, E.; de Novellis, V.; Marabese, I.; Cuomo, D.; Rossi, F.; Berrino, L.; Rossi, F.; Maione, S. Interaction between vanilloid and glutamate receptors in the central modulation of nociception. Eur. J. Pharmacol. 2002, 439, 69–75. [Google Scholar] [CrossRef]
- Starowicz, K.; Cristino, L.; Di Marzo, V. Trpv1 receptors in the central nervous system: Potential for previously unforeseen therapeutic applications. Curr. Pharm. Des. 2008, 14, 42–54. [Google Scholar] [PubMed]
- Starowicz, K.; Maione, S.; Cristino, L.; Palazzo, E.; Marabese, I.; Rossi, F.; de Novellis, V.; Di Marzo, V. Tonic endovanilloid facilitation of glutamate release in brainstem descending antinociceptive pathways. J. Neurosci. 2007, 27, 13739–13749. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, E.; Luongo, L.; de Novellis, V.; Rossi, F.; Marabese, I.; Maione, S. Transient receptor potential vanilloid type 1 and pain development. Curr. Opin. Pharmacol. 2012, 12, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Giordano, C.; Cristino, L.; Luongo, L.; Siniscalco, D.; Petrosino, S.; Piscitelli, F.; Marabese, I.; Gatta, L.; Rossi, F.; Imperatore, R.; et al. Trpv1-dependent and -independent alterations in the limbic cortex of neuropathic mice: Impact on glial caspases and pain perception. Cereb. Cortex 2012, 22, 2495–2518. [Google Scholar] [CrossRef] [PubMed]
- de Novellis, V.; Vita, D.; Gatta, L.; Luongo, L.; Bellini, G.; De Chiaro, M.; Marabese, I.; Siniscalco, D.; Boccella, S.; Piscitelli, F.; et al. The blockade of the transient receptor potential vanilloid type 1 and fatty acid amide hydrolase decreases symptoms and central sequelae in the medial prefrontal cortex of neuropathic rats. Mol. Pain 2011, 7, 7. [Google Scholar] [CrossRef] [PubMed]
- Avelino, A.; Cruz, C.; Nagy, I.; Cruz, F. Vanilloid receptor 1 expression in the rat urinary tract. Neuroscience 2002, 109, 787–798. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, D.H. Protective effect of trpv1 against renal fibrosis via inhibition of tgf-beta/smad signaling in doca-salt hypertension. Mol. Med. 2011, 17, 1204–1212. [Google Scholar] [PubMed]
- Wang, Y.; Wang, D.H. Aggravated renal inflammatory responses in trpv1 gene knockout mice subjected to doca-salt hypertension. Am. J. Physiol. Ren. Physiol. 2009, 297, F1550–F1559. [Google Scholar] [CrossRef] [PubMed]
- Conte, B.; Maggi, C.A.; Giachetti, A.; Parlani, M.; Lopez, G.; Manzini, S. Intraurethral capsaicin produces reflex activation of the striated urethral sphincter in urethane-anesthetized male rats. J. Urol. 1993, 150, 1271–1277. [Google Scholar] [PubMed]
- Maggi, C.A.; Giuliani, S.; Santicioli, P.; Abelli, L.; Meli, A. Visceromotor responses to calcitonin gene-related peptide (cgrp) in the rat lower urinary tract: Evidence for a transmitter role in the capsaicin-sensitive nerves of the ureter. Eur. J. Pharmacol. 1987, 143, 73–82. [Google Scholar] [CrossRef]
- Birder, L.A. Involvement of the urinary bladder urothelium in signaling in the lower urinary tract. Proc. West. Pharmacol. Soc. 2001, 44, 85–86. [Google Scholar] [PubMed]
- Lazzeri, M.; Vannucchi, M.G.; Zardo, C.; Spinelli, M.; Beneforti, P.; Turini, D.; Faussone-Pellegrini, M.S. Immunohistochemical evidence of vanilloid receptor 1 in normal human urinary bladder. Eur. Urol. 2004, 46, 792–798. [Google Scholar] [CrossRef] [PubMed]
- Apostolidis, A.; Brady, C.M.; Yiangou, Y.; Davis, J.; Fowler, C.J.; Anand, P. Capsaicin receptor trpv1 in urothelium of neurogenic human bladders and effect of intravesical resiniferatoxin. Urology 2005, 65, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Avelino, A.; Cruz, F. Trpv1 (vanilloid receptor) in the urinary tract: Expression, function and clinical applications. Naunyn Schmiedebergs Arch. Pharmacol. 2006, 373, 287–299. [Google Scholar] [CrossRef] [PubMed]
- Charrua, A.; Reguenga, C.; Cordeiro, J.M.; Correiade-Sa, P.; Paule, C.; Nagy, I.; Cruz, F.; Avelino, A. Functional transient receptor potential vanilloid 1 is expressed in human urothelial cells. J. Urol. 2009, 182, 2944–2950. [Google Scholar] [CrossRef] [PubMed]
- Ost, D.; Roskams, T.; van Der Aa, F.; de Ridder, D. Topography of the vanilloid receptor in the human bladder: More than just the nerve fibers. J. Urol. 2002, 168, 293–297. [Google Scholar] [CrossRef]
- Sui, G.P.; Wu, C.; Fry, C.H. Electrical characteristics of suburothelial cells isolated from the human bladder. J. Urol. 2004, 171, 938–943. [Google Scholar] [CrossRef] [PubMed]
- Dinis, P.; Charrua, A.; Avelino, A.; Yaqoob, M.; Bevan, S.; Nagy, I.; Cruz, F. Anandamide-evoked activation of vanilloid receptor 1 contributes to the development of bladder hyperreflexia and nociceptive transmission to spinal dorsal horn neurons in cystitis. J. Neurosci. 2004, 24, 11253–11263. [Google Scholar] [CrossRef] [PubMed]
- Cruz, F.; Guimaraes, M.; Silva, C.; Reis, M. Suppression of bladder hyperreflexia by intravesical resiniferatoxin. Lancet 1997, 350, 640–641. [Google Scholar] [CrossRef]
- Cruz, F.; Guimaraes, M.; Silva, C.; Rio, M.E.; Coimbra, A.; Reis, M. Desensitization of bladder sensory fibers by intravesical capsaicin has long lasting clinical and urodynamic effects in patients with hyperactive or hypersensitive bladder dysfunction. J. Urol. 1997, 157, 585–589. [Google Scholar] [CrossRef]
- Cruz, C.D.; Charrua, A.; Vieira, E.; Valente, J.; Avelino, A.; Cruz, F. Intrathecal delivery of resiniferatoxin (rtx) reduces detrusor overactivity and spinal expression of trpv1 in spinal cord injured animals. Exp. Neurol. 2008, 214, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Avelino, A.; Cruz, F. Peptide immunoreactivity and ultrastructure of rat urinary bladder nerve fibers after topical desensitization by capsaicin or resiniferatoxin. Auton Neurosci. 2000, 86, 37–46. [Google Scholar] [CrossRef]
- Avelino, A.; Charrua, A.; Frias, B.; Cruz, C.; Boudes, M.; de Ridder, D.; Cruz, F. Transient receptor potential channels in bladder function. Acta Physiol. (Oxf.) 2013, 207, 110–122. [Google Scholar] [CrossRef] [PubMed]
- Charrua, A.; Cruz, C.D.; Cruz, F.; Avelino, A. Transient receptor potential vanilloid subfamily 1 is essential for the generation of noxious bladder input and bladder overactivity in cystitis. J. Urol. 2007, 177, 1537–1541. [Google Scholar] [CrossRef] [PubMed]
- Mukerji, G.; Yiangou, Y.; Agarwal, S.K.; Anand, P. Transient receptor potential vanilloid receptor subtype 1 in painful bladder syndrome and its correlation with pain. J. Urol. 2006, 176, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Jaggar, S.I.; Scott, H.C.; James, I.F.; Rice, A.S. The capsaicin analogue sdz249–665 attenuates the hyper-reflexia and referred hyperalgesia associated with inflammation of the rat urinary bladder. Pain 2001, 89, 229–235. [Google Scholar] [CrossRef]
- Urban, L.; Campbell, E.A.; Panesar, M.; Patel, S.; Chaudhry, N.; Kane, S.; Buchheit, K.; Sandells, B.; James, I.F. In vivo pharmacology of sdz 249–665, a novel, non-pungent capsaicin analogue. Pain 2000, 89, 65–74. [Google Scholar] [CrossRef]
- Birder, L.A.; Nakamura, Y.; Kiss, S.; Nealen, M.L.; Barrick, S.; Kanai, A.J.; Wang, E.; Ruiz, G.; de Groat, W.C.; Apodaca, G.; et al. Altered urinary bladder function in mice lacking the vanilloid receptor trpv1. Nat. Neurosci. 2002, 5, 856–860. [Google Scholar] [CrossRef] [PubMed]
- Avelino, A.; Cruz, F.; Coimbra, A. Intravesical resiniferatoxin desensitizes rat bladder sensory fibres without causing intense noxious excitation. A c-fos study. Eur. J. Pharmacol. 1999, 378, 17–22. [Google Scholar] [CrossRef]
- Vizzard, M.A. Alterations in spinal cord fos protein expression induced by bladder stimulation following cystitis. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 278, R1027–R1039. [Google Scholar] [PubMed]
- Brady, C.M.; Apostolidis, A.N.; Harper, M.; Yiangou, Y.; Beckett, A.; Jacques, T.S.; Freeman, A.; Scaravilli, F.; Fowler, C.J.; Anand, P. Parallel changes in bladder suburothelial vanilloid receptor trpv1 and pan-neuronal marker pgp9.5 immunoreactivity in patients with neurogenic detrusor overactivity after intravesical resiniferatoxin treatment. BJU Int. 2004, 93, 770–776. [Google Scholar] [CrossRef] [PubMed]
- Andersson, P.O.; Malmgren, A.; Uvelius, B. Functional responses of different muscle types of the female rat urethra in vitro. Acta Physiol. Scand. 1990, 140, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Maggi, C.A.; Santicioli, P.; Abelli, L.; Parlani, M.; Capasso, M.; Conte, B.; Giuliani, S.; Meli, A. Regional differences in the effects of capsaicin and tachykinins on motor activity and vascular permeability of the rat lower urinary tract. Naunyn Schmiedebergs Arch. Pharmacol. 1987, 335, 636–645. [Google Scholar] [CrossRef] [PubMed]
- Patterson, L.M.; Zheng, H.; Ward, S.M.; Berthoud, H.R. Vanilloid receptor (vr1) expression in vagal afferent neurons innervating the gastrointestinal tract. Cell Tissue Res. 2003, 311, 277–287. [Google Scholar] [PubMed]
- Ward, S.M.; Bayguinov, J.; Won, K.J.; Grundy, D.; Berthoud, H.R. Distribution of the vanilloid receptor (vr1) in the gastrointestinal tract. J. Comp. Neurol. 2003, 465, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Holzer, P. Trpv1 and the gut: From a tasty receptor for a painful vanilloid to a key player in hyperalgesia. Eur. J. Pharmacol. 2004, 500, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Bartho, L.; Benko, R.; Patacchini, R.; Petho, G.; Holzer-Petsche, U.; Holzer, P.; Lazar, Z.; Undi, S.; Illenyi, L.; Antal, A.; et al. Effects of capsaicin on visceral smooth muscle: A valuable tool for sensory neurotransmitter identification. Eur. J. Pharmacol. 2004, 500, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.C., 3rd; Xu, L.; Gebhart, G.F. The mechanosensitivity of mouse colon afferent fibers and their sensitization by inflammatory mediators require transient receptor potential vanilloid 1 and acid-sensing ion channel 3. J. Neurosci. 2005, 25, 10981–10989. [Google Scholar] [CrossRef] [PubMed]
- Rong, W.; Hillsley, K.; Davis, J.B.; Hicks, G.; Winchester, W.J.; Grundy, D. Jejunal afferent nerve sensitivity in wild-type and trpv1 knockout mice. J. Physiol. 2004, 560, 867–881. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, B.; Medda, B.K.; Lazarova, Z.; Bansal, N.; Shaker, R.; Sengupta, J.N. Effect of reflux-induced inflammation on transient receptor potential vanilloid one (trpv1) expression in primary sensory neurons innervating the oesophagus of rats. Neurogastroenterol. Motil. 2007, 19, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Hosoya, T.; Ishikawa, E.; Tashima, K.; Amagase, K.; Kato, S.; Murayama, T.; Horie, S. Distribution of transient receptor potential cation channel subfamily v member 1-expressing nerve fibers in mouse esophagus. Histochem. Cell Biol. 2014, 142, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Blackshaw, L.A.; Page, A.J.; Partosoedarso, E.R. Acute effects of capsaicin on gastrointestinal vagal afferents. Neuroscience 2000, 96, 407–416. [Google Scholar] [CrossRef]
- Schicho, R.; Florian, W.; Liebmann, I.; Holzer, P.; Lippe, I.T. Increased expression of trpv1 receptor in dorsal root ganglia by acid insult of the rat gastric mucosa. Eur. J. Neurosci. 2004, 19, 1811–1818. [Google Scholar] [CrossRef] [PubMed]
- Peles, S.; Medda, B.K.; Zhang, Z.; Banerjee, B.; Lehmann, A.; Shaker, R.; Sengupta, J.N. Differential effects of transient receptor vanilloid one (trpv1) antagonists in acid-induced excitation of esophageal vagal afferent fibers of rats. Neuroscience 2009, 161, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Blackshaw, L.A. Transient receptor potential cation channels in visceral sensory pathways. Br. J. Pharmacol. 2014, 171, 2528–2536. [Google Scholar] [CrossRef] [PubMed]
- Fujino, K.; de la Fuente, S.G.; Takami, Y.; Takahashi, T.; Mantyh, C.R. Attenuation of acid induced oesophagitis in vr-1 deficient mice. Gut 2006, 55, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; de la Monte, S.; Ma, J.; Hong, J.; Tong, M.; Cao, W.; Behar, J.; Biancani, P.; Harnett, K.M. Hcl-activated neural and epithelial vanilloid receptors (trpv1) in cat esophageal mucosa. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 297, G135–G143. [Google Scholar] [CrossRef] [PubMed]
- Matthews, P.J.; Aziz, Q.; Facer, P.; Davis, J.B.; Thompson, D.G.; Anand, P. Increased capsaicin receptor trpv1 nerve fibres in the inflamed human oesophagus. Eur. J. Gastroenterol. Hepatol. 2004, 16, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Shieh, K.R.; Yi, C.H.; Liu, T.T.; Tseng, H.L.; Ho, H.C.; Hsieh, H.T.; Chen, C.L. Evidence for neurotrophic factors associating with trpv1 gene expression in the inflamed human esophagus. Neurogastroenterol. Motil. 2010, 22, 971–977, e252. [Google Scholar] [CrossRef] [PubMed]
- Bhat, Y.M.; Bielefeldt, K. Capsaicin receptor (trpv1) and non-erosive reflux disease. Eur. J. Gastroenterol. Hepatol. 2006, 18, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Sharrad, D.F.; Hibberd, T.J.; Kyloh, M.A.; Brookes, S.J.; Spencer, N.J. Quantitative immunohistochemical co-localization of trpv1 and cgrp in varicose axons of the murine oesophagus, stomach and colorectum. Neurosci. Lett. 2015, 599, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Horie, S.; Michael, G.J.; Priestley, J.V. Co-localization of trpv1-expressing nerve fibers with calcitonin-gene-related peptide and substance p in fundus of rat stomach. Inflammopharmacology 2005, 13, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Faussone-Pellegrini, M.S.; Taddei, A.; Bizzoco, E.; Lazzeri, M.; Vannucchi, M.G.; Bechi, P. Distribution of the vanilloid (capsaicin) receptor type 1 in the human stomach. Histochem. Cell Biol. 2005, 124, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Aihara, E.; Nakamura, A.; Xin, H.; Matsui, H.; Kohama, K.; Takeuchi, K. Expression of vanilloid receptors in rat gastric epithelial cells: Role in cellular protection. Biochem. Pharmacol. 2003, 66, 1115–1121. [Google Scholar] [CrossRef]
- Lamb, K.; Kang, Y.M.; Gebhart, G.F.; Bielefeldt, K. Gastric inflammation triggers hypersensitivity to acid in awake rats. Gastroenterology 2003, 125, 1410–1418. [Google Scholar] [CrossRef] [PubMed]
- Minowa, S.; Ishihara, S.; Tsuchiya, S.; Horie, S.; Murayama, T. Capsaicin- and anandamide-induced gastric acid secretion via vanilloid receptor type 1 (trpv1) in rat brain. Brain Res. 2005, 1039, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Minowa, S.; Tsuchiya, S.; Horie, S.; Watanabe, K.; Murayama, T. Stimulatory effect of centrally injected capsaicin, an agonist of vanilloid receptors, on gastric acid secretion in rats. Eur. J. Pharmacol. 2001, 428, 349–356. [Google Scholar] [CrossRef]
- Lee, K.J.; Vos, R.; Tack, J. Effects of capsaicin on the sensorimotor function of the proximal stomach in humans. Aliment. Pharmacol. Ther. 2004, 19, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Hammer, J.; Vogelsang, H. Characterization of sensations induced by capsaicin in the upper gastrointestinal tract. Neurogastroenterol. Motil. 2007, 19, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.L.; Bornstein, J.C.; Anderson, C.R. Distinct chemical classes of medium-sized transient receptor potential channel vanilloid 1-immunoreactive dorsal root ganglion neurons innervate the adult mouse jejunum and colon. Neuroscience 2008, 156, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, M.; Kuramoto, H.; Takaki, M. Combined determination with functional and morphological studies of origin of nerve fibers expressing transient receptor potential vanilloid 1 in the myenteric plexus of the rat jejunum. Auton Neurosci. 2004, 116, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Laird, J.M.; Martinez-Caro, L.; Garcia-Nicas, E.; Cervero, F. A new model of visceral pain and referred hyperalgesia in the mouse. Pain 2001, 92, 335–342. [Google Scholar] [CrossRef]
- Schmidt, B.; Hammer, J.; Holzer, P.; Hammer, H.F. Chemical nociception in the jejunum induced by capsaicin. Gut 2004, 53, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Hammer, J. Effect of repeated capsaicin ingestion on intestinal chemosensation and mechanosensation. Aliment. Pharmacol. Ther. 2006, 24, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Drewes, A.M.; Schipper, K.P.; Dimcevski, G.; Petersen, P.; Gregersen, H.; Funch-Jensen, P.; Arendt-Nielsen, L. Gut pain and hyperalgesia induced by capsaicin: A human experimental model. Pain 2003, 104, 333–341. [Google Scholar] [CrossRef]
- Brierley, S.M.; Jones, R.C., 3rd; Xu, L.; Gebhart, G.F.; Blackshaw, L.A. Activation of splanchnic and pelvic colonic afferents by bradykinin in mice. Neurogastroenterol. Motil. 2005, 17, 854–862. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Hosoya, T.; Tashima, K.; Namiki, T.; Murayama, T.; Horie, S. Distribution of transient receptor potential vanilloid 1 channel-expressing nerve fibers in mouse rectal and colonic enteric nervous system: Relationship to peptidergic and nitrergic neurons. Neuroscience 2011, 172, 518–534. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Kurosawa, E.; Terui, H.; Hosoya, T.; Tashima, K.; Murayama, T.; Priestley, J.V.; Horie, S. Localization of trpv1 and contractile effect of capsaicin in mouse large intestine: High abundance and sensitivity in rectum and distal colon. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 297, G348–G360. [Google Scholar] [CrossRef] [PubMed]
- Christianson, J.A.; Traub, R.J.; Davis, B.M. Differences in spinal distribution and neurochemical phenotype of colonic afferents in mouse and rat. J. Comp. Neurol. 2006, 494, 246–259. [Google Scholar] [CrossRef] [PubMed]
- Malin, S.A.; Christianson, J.A.; Bielefeldt, K.; Davis, B.M. Tprv1 expression defines functionally distinct pelvic colon afferents. J. Neurosci. 2009, 29, 743–752. [Google Scholar] [CrossRef] [PubMed]
- Kimball, E.S.; Wallace, N.H.; Schneider, C.R.; D'Andrea, M.R.; Hornby, P.J. Vanilloid receptor 1 antagonists attenuate disease severity in dextran sulphate sodium-induced colitis in mice. Neurogastroenterol. Motil. 2004, 16, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.L.; Facer, P.; Davis, J.B.; Smith, G.D.; Egerton, J.; Bountra, C.; Williams, N.S.; Anand, P. Sensory fibres expressing capsaicin receptor trpv1 in patients with rectal hypersensitivity and faecal urgency. Lancet 2003, 361, 385–391. [Google Scholar] [CrossRef]
- Kihara, N.; de la Fuente, S.G.; Fujino, K.; Takahashi, T.; Pappas, T.N.; Mantyh, C.R. Vanilloid receptor-1 containing primary sensory neurones mediate dextran sulphate sodium induced colitis in rats. Gut 2003, 52, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Miranda, A.; Nordstrom, E.; Mannem, A.; Smith, C.; Banerjee, B.; Sengupta, J.N. The role of transient receptor potential vanilloid 1 in mechanical and chemical visceral hyperalgesia following experimental colitis. Neuroscience 2007, 148, 1021–1032. [Google Scholar] [CrossRef] [PubMed]
- Plourde, V.; St-Pierre, S.; Quirion, R. Calcitonin gene-related peptide in viscerosensitive response to colorectal distension in rats. Am. J. Physiol. 1997, 273, G191–G196. [Google Scholar] [PubMed]
- Delafoy, L.; Raymond, F.; Doherty, A.M.; Eschalier, A.; Diop, L. Role of nerve growth factor in the trinitrobenzene sulfonic acid-induced colonic hypersensitivity. Pain 2003, 105, 489–497. [Google Scholar] [CrossRef]
- Hughes, P.A.; Brierley, S.M.; Martin, C.M.; Brookes, S.J.; Linden, D.R.; Blackshaw, L.A. Post-inflammatory colonic afferent sensitisation: Different subtypes, different pathways and different time courses. Gut 2009, 58, 1333–1341. [Google Scholar] [CrossRef] [PubMed]
- Akbar, A.; Yiangou, Y.; Facer, P.; Brydon, W.G.; Walters, J.R.; Anand, P.; Ghosh, S. Expression of the trpv1 receptor differs in quiescent inflammatory bowel disease with or without abdominal pain. Gut 2010, 59, 767–774. [Google Scholar] [CrossRef] [PubMed]
- Facer, P.; Knowles, C.H.; Tam, P.K.; Ford, A.P.; Dyer, N.; Baecker, P.A.; Anand, P. Novel capsaicin (vr1) and purinergic (p2x3) receptors in hirschsprung's intestine. J. Pediatr. Surg. 2001, 36, 1679–1684. [Google Scholar] [CrossRef] [PubMed]
- Fasanella, K.E.; Christianson, J.A.; Chanthaphavong, R.S.; Davis, B.M. Distribution and neurochemical identification of pancreatic afferents in the mouse. J. Comp. Neurol. 2008, 509, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Wick, E.C.; Hoge, S.G.; Grahn, S.W.; Kim, E.; Divino, L.A.; Grady, E.F.; Bunnett, N.W.; Kirkwood, K.S. Transient receptor potential vanilloid 1, calcitonin gene-related peptide, and substance p mediate nociception in acute pancreatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G959–G969. [Google Scholar] [CrossRef] [PubMed]
- Liddle, R.A. The role of transient receptor potential vanilloid 1 (trpv1) channels in pancreatitis. Biochim. Biophys. Acta 2007, 1772, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Wick, E.C.; Pikios, S.; Grady, E.F.; Kirkwood, K.S. Calcitonin gene-related peptide partially mediates nociception in acute experimental pancreatitis. Surgery 2006, 139, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Figini, M.; Emanueli, C.; Grady, E.F.; Kirkwood, K.; Payan, D.G.; Ansel, J.; Gerard, C.; Geppetti, P.; Bunnett, N. Substance p and bradykinin stimulate plasma extravasation in the mouse gastrointestinal tract and pancreas. Am. J. Physiol. 1997, 272, G785–G793. [Google Scholar] [PubMed]
- Bhatia, M.; Saluja, A.K.; Hofbauer, B.; Frossard, J.L.; Lee, H.S.; Castagliuolo, I.; Wang, C.C.; Gerard, N.; Pothoulakis, C.; Steer, M.L. Role of substance p and the neurokinin 1 receptor in acute pancreatitis and pancreatitis-associated lung injury. Proc. Natl. Acad. Sci. USA 1998, 95, 4760–4765. [Google Scholar] [CrossRef] [PubMed]
- Nathan, J.D.; Peng, R.Y.; Wang, Y.; McVey, D.C.; Vigna, S.R.; Liddle, R.A. Primary sensory neurons: A common final pathway for inflammation in experimental pancreatitis in rats. Am. J. Physiol. Gastrointest. Liver Physiol. 2002, 283, G938–G946. [Google Scholar] [CrossRef] [PubMed]
- Nathan, J.D.; Patel, A.A.; McVey, D.C.; Thomas, J.E.; Prpic, V.; Vigna, S.R.; Liddle, R.A. Capsaicin vanilloid receptor-1 mediates substance p release in experimental pancreatitis. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 281, G1322–G1328. [Google Scholar] [PubMed]
- Hoogerwerf, W.A.; Zou, L.; Shenoy, M.; Sun, D.; Micci, M.A.; Lee-Hellmich, H.; Xiao, S.Y.; Winston, J.H.; Pasricha, P.J. The proteinase-activated receptor 2 is involved in nociception. J. Neurosci. 2001, 21, 9036–9042. [Google Scholar] [PubMed]
- Watanabe, N.; Horie, S.; Michael, G.J.; Spina, D.; Page, C.P.; Priestley, J.V. Immunohistochemical localization of vanilloid receptor subtype 1 (trpv1) in the guinea pig respiratory system. Pulm. Pharmacol. Ther. 2005, 18, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Horie, S.; Michael, G.J.; Keir, S.; Spina, D.; Page, C.P.; Priestley, J.V. Immunohistochemical co-localization of transient receptor potential vanilloid (trpv)1 and sensory neuropeptides in the guinea-pig respiratory system. Neuroscience 2006, 141, 1533–1543. [Google Scholar] [CrossRef] [PubMed]
- Pingle, S.C.; Matta, J.A.; Ahern, G.P. Capsaicin receptor: Trpv1 a promiscuous trp channel. In Transient Receptor Potential (TRP) Channels; Springer: Berlin, Germany, 2007; pp. 155–171. [Google Scholar]
- Jia, Y.; McLeod, R.L.; Hey, J.A. Trpv1 receptor: A target for the treatment of pain, cough, airway disease and urinary incontinence. Drug News Perspect. 2005, 18, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Stein, R.J.; Santos, S.; Nagatomi, J.; Hayashi, Y.; Minnery, B.S.; Xavier, M.; Patel, A.S.; Nelson, J.B.; Futrell, W.J.; Yoshimura, N.; et al. Cool (trpm8) and hot (trpv1) receptors in the bladder and male genital tract. J. Urol. 2004, 172, 1175–1178. [Google Scholar] [CrossRef] [PubMed]
- Auzanneau, C.; Norez, C.; Antigny, F.; Thoreau, V.; Jougla, C.; Cantereau, A.; Becq, F.; Vandebrouck, C. Transient receptor potential vanilloid 1 (trpv1) channels in cultured rat sertoli cells regulate an acid sensing chloride channel. Biochem. Pharmacol. 2008, 75, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Mizrak, S.C.; Gadella, B.M.; Erdost, H.; Ozer, A.; van Pelt, A.M.; van Dissel-Emiliani, F.M. Spermatogonial stem cell sensitivity to capsaicin: An in vitro study. Reprod. Biol. Endocrinol. 2008, 6, 52. [Google Scholar] [CrossRef] [PubMed]
- Dinis, P.; Charrua, A.; Avelino, A.; Nagy, I.; Quintas, J.; Ribau, U.; Cruz, F. The distribution of sensory fibers immunoreactive for the trpv1 (capsaicin) receptor in the human prostate. Eur. Urol. 2005, 48, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Litwin, M.S.; McNaughton-Collins, M.; Fowler, F.J., Jr.; Nickel, J.C.; Calhoun, E.A.; Pontari, M.A.; Alexander, R.B.; Farrar, J.T.; O'Leary, M.P. The national institutes of health chronic prostatitis symptom index: Development and validation of a new outcome measure. Chronic prostatitis collaborative research network. J. Urol. 1999, 162, 369–375. [Google Scholar] [CrossRef]
- Turini, D.; Beneforti, P.; Spinelli, M.; Malagutti, S.; Lazzeri, M. Heat/burning sensation induced by topical application of capsaicin on perineal cutaneous area: New approach in diagnosis and treatment of chronic prostatitis/chronic pelvic pain syndrome? Urology 2006, 67, 910–913. [Google Scholar] [CrossRef] [PubMed]
- Czifra, G.; Varga, A.; Nyeste, K.; Marincsak, R.; Toth, B.I.; Kovacs, I.; Kovacs, L.; Biro, T. Increased expressions of cannabinoid receptor-1 and transient receptor potential vanilloid-1 in human prostate carcinoma. J. Cancer Res. Clin. Oncol. 2009, 135, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.G.; Sanchez, A.M.; Collado, B.; Malagarie-Cazenave, S.; Olea, N.; Carmena, M.J.; Prieto, J.C.; Diaz-Laviada, I.I. Expression of the transient receptor potential vanilloid 1 (trpv1) in lncap and pc-3 prostate cancer cells and in human prostate tissue. Eur. J. Pharmacol. 2005, 515, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.P.; Pu, X.Y.; Wang, X.H. Distribution profiles of transient receptor potential melastatin-related and vanilloid-related channels in prostatic tissue in rat. Asian J. Androl. 2007, 9, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Pierce, A.N.; Zhang, Z.; Fuentes, I.M.; Wang, R.; Ryals, J.M.; Christianson, J.A. Neonatal vaginal irritation results in long-term visceral and somatic hypersensitivity and increased hypothalamic-pituitary-adrenal axis output in female mice. Pain 2015, 156, 2021–2031. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.; Conklin, D.; Clyne, B.B.; Stanislaus, J.D.; Eisenach, J.C. Uterine cervical afferents in thoracolumbar dorsal root ganglia express transient receptor potential vanilloid type 1 channel and calcitonin gene-related peptide, but not p2x3 receptor and somatostatin. Anesthesiology 2006, 104, 651–657. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Liu, B.; Du, D.; Eisenach, J.C.; Tong, C. Estrogen amplifies pain responses to uterine cervical distension in rats by altering transient receptor potential-1 function. Anesth. Analg. 2007, 104, 1246–1250, tables of contents. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.Y.; Chang, H.M.; Lee, S.D.; Huang, P.C.; Chen, G.D.; Lai, C.H.; Lai, C.Y.; Chiu, C.H.; Tung, K.C.; Lin, T.B. Trpv1 mediates the uterine capsaicin-induced nmda nr2b-dependent cross-organ reflex sensitization in anesthetized rats. Am. J. Physiol. Renal Physiol. 2008, 295, F1324–F1335. [Google Scholar] [CrossRef] [PubMed]
- Tingaker, B.K.; Ekman-Ordeberg, G.; Facer, P.; Irestedt, L.; Anand, P. Influence of pregnancy and labor on the occurrence of nerve fibers expressing the capsaicin receptor trpv1 in human corpus and cervix uteri. Reprod Biol. Endocrinol. 2008, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Coelho, A.; Wolf-Johnston, A.S.; Shinde, S.; Cruz, C.D.; Cruz, F.; Avelino, A.; Birder, L.A. Urinary bladder inflammation induces changes in urothelial nerve growth factor and trpv1 channels. Br. J. Pharmacol. 2015, 172, 1691–1699. [Google Scholar] [CrossRef] [PubMed]
- Fioravanti, B.; De Felice, M.; Stucky, C.L.; Medler, K.A.; Luo, M.C.; Gardell, L.R.; Ibrahim, M.; Malan, T.P., Jr.; Yamamura, H.I.; Ossipov, M.H.; et al. Constitutive activity at the cannabinoid cb1 receptor is required for behavioral response to noxious chemical stimulation of trpv1: Antinociceptive actions of cb1 inverse agonists. J. Neurosci. 2008, 28, 11593–11602. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, F.; Salio, C.; Lossi, L.; Gambino, G.; Merighi, A. Modulation of inhibitory neurotransmission by the vanilloid receptor type 1 (trpv1) in organotypically cultured mouse substantia gelatinosa neurons. Pain 2010, 150, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Lao, L.J.; Song, B.; Marvizon, J.C. Neurokinin release produced by capsaicin acting on the central terminals and axons of primary afferents: Relationship with n-methyl-d-aspartate and gaba(b) receptors. Neuroscience 2003, 121, 667–680. [Google Scholar] [CrossRef]
- Merighi, A. Costorage and coexistence of neuropeptides in the mammalian cns. Prog. Neurobiol. 2002, 66, 161–190. [Google Scholar] [CrossRef]
- Kanai, Y.; Nakazato, E.; Fujiuchi, A.; Hara, T.; Imai, A. Involvement of an increased spinal trpv1 sensitization through its up-regulation in mechanical allodynia of cci rats. Neuropharmacology 2005, 49, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.Z.; Pan, H.L. Primary afferent stimulation differentially potentiates excitatory and inhibitory inputs to spinal lamina ii outer and inner neurons. J. Neurophysiol. 2004, 91, 2413–2421. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Gu, J.G. A p2x receptor-mediated nociceptive afferent pathway to lamina i of the spinal cord. Mol. Pain 2005, 1, 4. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, Y.; Kohno, T.; Zhuang, Z.Y.; Brenner, G.J.; Wang, H.; Van Der Meer, C.; Befort, K.; Woolf, C.J.; Ji, R.R. Ionotropic and metabotropic receptors, protein kinase a, protein kinase c, and src contribute to c-fiber-induced erk activation and camp response element-binding protein phosphorylation in dorsal horn neurons, leading to central sensitization. J. Neurosci. 2004, 24, 8310–8321. [Google Scholar] [CrossRef] [PubMed]
- Ferrini, F.; Russo, A.; Salio, C. Fos and perk immunoreactivity in spinal cord slices: Comparative analysis of in vitro models for testing putative antinociceptive molecules. Ann. Anat. 2014, 196, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Salio, C.; Ferrini, F.; Muthuraju, S.; Merighi, A. Presynaptic modulation of spinal nociceptive transmission by glial cell line-derived neurotrophic factor (gdnf). J. Neurosci. 2014, 34, 13819–13833. [Google Scholar] [CrossRef] [PubMed]
- Merighi, A.; Bardoni, R.; Salio, C.; Lossi, L.; Ferrini, F.; Prandini, M.; Zonta, M.; Gustincich, S.; Carmignoto, G. Presynaptic functional trkb receptors mediate the release of excitatory neurotransmitters from primary afferent terminals in lamina ii (substantia gelatinosa) of postnatal rat spinal cord. Dev. Neurobiol. 2008, 68, 457–475. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, H.; Kiritoshi, T.; Murase, K. Effect of excitatory and inhibitory agents and a glial inhibitor on optically-recorded primary-afferent excitation. Mol. Pain 2008, 4, 39. [Google Scholar] [CrossRef] [PubMed]
- Qutenza. Official website of qutenza (trademarked) patch. Avaliable online: http://www.qutenza.com/_docs/qutenza_full_PI_.pdf (accessed on 17 June 2016).
- Kulkantrakorn, K.; Lorsuwansiri, C.; Meesawatsom, P. 0.025% capsaicin gel for the treatment of painful diabetic neuropathy: A randomized, double-blind, crossover, placebo-controlled trial. Pain Pract. 2013, 13, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Derry, S.; Moore, R.A. Topical capsaicin (low concentration) for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2012, 9, CD010111. [Google Scholar] [PubMed]
- Teixeira, M.J.; Menezes, L.M.; Silva, V.; Galhardoni, R.; Sasson, J.; Okada, M.; Duarte, K.P.; Yeng, L.T.; Andrade, D.C. Liposomal topical capsaicin in post-herpetic neuralgia: A safety pilot study. Arq. Neuropsiquiatr. 2015, 73, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Casanueva, B.; Rodero, B.; Quintial, C.; Llorca, J.; Gonzalez-Gay, M.A. Short-term efficacy of topical capsaicin therapy in severely affected fibromyalgia patients. Rheumatol. Int. 2013, 33, 2665–2670. [Google Scholar] [CrossRef] [PubMed]
- Griebeler, M.L.; Morey-Vargas, O.L.; Brito, J.P.; Tsapas, A.; Wang, Z.; Carranza Leon, B.G.; Phung, O.J.; Montori, V.M.; Murad, M.H. Pharmacologic interventions for painful diabetic neuropathy: An umbrella systematic review and comparative effectiveness network meta-analysis. Ann. Intern. Med. 2014, 161, 639–649. [Google Scholar] [CrossRef] [PubMed]
- Altman, R.D.; Barthel, H.R. Topical therapies for osteoarthritis. Drugs 2011, 71, 1259–1279. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, J.M.; Ringsted, T.K.; Petersen, M.; Sommer, C.; Uceyler, N.; Werner, M.U. A capsaicin (8%) patch in the treatment of severe persistent inguinal postherniorrhaphy pain: A randomized, double-blind, placebo-controlled trial. PLoS ONE 2014, 9, e109144. [Google Scholar] [CrossRef] [PubMed]
- Hartrick, C.T.; Pestano, C.; Carlson, N.; Hartrick, S. Capsaicin instillation for postoperative pain following total knee arthroplasty: A preliminary report of a randomized, double-blind, parallel-group, placebo-controlled, multicentre trial. Clin. Drug Investig. 2011, 31, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, M.; Porta, S. Effect of red pepper on symptoms of irritable bowel syndrome: Preliminary study. Dig. Dis. Sci. 2011, 56, 3288–3295. [Google Scholar] [CrossRef] [PubMed]
- Salat, K.; Jakubowska, A.; Kulig, K. Zucapsaicin for the treatment of neuropathic pain. Expert Opin. Investig. Drugs 2014, 23, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Maihofner, C.; Heskamp, M.L. Prospective, non-interventional study on the tolerability and analgesic effectiveness over 12 weeks after a single application of capsaicin 8% cutaneous patch in 1044 patients with peripheral neuropathic pain: First results of the quepp study. Curr. Med. Res. Opin. 2013, 29, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.; Simpson, D.M.; Moyle, G.; Brew, B.J.; Schifitto, G.; Larbalestier, N.; Orkin, C.; Fisher, M.; Vanhove, G.F.; Tobias, J.K. Ngx-4010, a capsaicin 8% patch, for the treatment of painful hiv-associated distal sensory polyneuropathy: Integrated analysis of two phase iii, randomized, controlled trials. AIDS Res. Ther. 2013, 10, 5. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.M.; Brown, S.; Tobias, J.K.; Vanhove, G.F.; Group, N.-C.S. Ngx-4010, a capsaicin 8% dermal patch, for the treatment of painful hiv-associated distal sensory polyneuropathy: Results of a 52-week open-label study. Clin. J. Pain 2014, 30, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Clifford, D.B.; Simpson, D.M.; Brown, S.; Moyle, G.; Brew, B.J.; Conway, B.; Tobias, J.K.; Vanhove, G.F.; Group, N.-C.S. A randomized, double-blind, controlled study of ngx-4010, a capsaicin 8% dermal patch, for the treatment of painful hiv-associated distal sensory polyneuropathy. J. Acquir. Immune Defic. Syndr. 2012, 59, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Irving, G.; Backonja, M.; Rauck, R.; Webster, L.R.; Tobias, J.K.; Vanhove, G.F. Ngx-4010, a capsaicin 8% dermal patch, administered alone or in combination with systemic neuropathic pain medications, reduces pain in patients with postherpetic neuralgia. Clin. J. Pain 2012, 28, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Maihofner, C.G.; Heskamp, M.L. Treatment of peripheral neuropathic pain by topical capsaicin: Impact of pre-existing pain in the quepp-study. Eur. J. Pain 2014, 18, 671–679. [Google Scholar] [CrossRef] [PubMed]
- Martini, C.H.; Yassen, A.; Krebs-Brown, A.; Passier, P.; Stoker, M.; Olofsen, E.; Dahan, A. A novel approach to identify responder subgroups and predictors of response to low- and high-dose capsaicin patches in postherpetic neuralgia. Eur. J. Pain 2013, 17, 1491–1501. [Google Scholar] [CrossRef] [PubMed]
- Mou, J.; Paillard, F.; Turnbull, B.; Trudeau, J.; Stoker, M.; Katz, N.P. Qutenza (capsaicin) 8% patch onset and duration of response and effects of multiple treatments in neuropathic pain patients. Clin. J. Pain 2014, 30, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Mou, J.; Paillard, F.; Turnbull, B.; Trudeau, J.; Stoker, M.; Katz, N.P. Efficacy of qutenza(r) (capsaicin) 8% patch for neuropathic pain: A meta-analysis of the qutenza clinical trials database. Pain 2013, 154, 1632–1639. [Google Scholar] [CrossRef] [PubMed]
- Hoper, J.; Helfert, S.; Heskamp, M.L.; Maihofner, C.G.; Baron, R. High concentration capsaicin for treatment of peripheral neuropathic pain: Effect on somatosensory symptoms and identification of treatment responders. Curr. Med. Res. Opin. 2014, 30, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Webster, L.R.; Peppin, J.F.; Murphy, F.T.; Lu, B.; Tobias, J.K.; Vanhove, G.F. Efficacy, safety, and tolerability of ngx-4010, capsaicin 8% patch, in an open-label study of patients with peripheral neuropathic pain. Diabetes Res. Clin. Pract. 2011, 93, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Jensen, T.S.; Hoye, K.; Fricova, J.; Vanelderen, P.; Ernault, E.; Siciliano, T.; Marques, S. Tolerability of the capsaicin 8% patch following pretreatment with lidocaine or tramadol in patients with peripheral neuropathic pain: A multicentre, randomized, assessor-blinded study. Eur. J. Pain 2014, 18, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- Treede, R.D.; Wagner, T.; Kern, K.U.; Husstedt, I.W.; Arendt, G.; Birklein, F.; Cegla, T.; Freynhagen, R.; Gockel, H.H.; Heskamp, M.L.; et al. Mechanism- and experience-based strategies to optimize treatment response to the capsaicin 8% cutaneous patch in patients with localized neuropathic pain. Curr. Med. Res. Opin. 2013, 29, 527–538. [Google Scholar] [CrossRef] [PubMed]
- Derry, S.; Sven-Rice, A.; Cole, P.; Tan, T.; Moore, R.A. Topical capsaicin (high concentration) for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2013, 2, CD007393. [Google Scholar] [PubMed]
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; McNicol, E.; Baron, R.; Dworkin, R.H.; Gilron, I.; Haanpaa, M.; Hansson, P.; Jensen, T.S.; et al. Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. Lancet Neurol. 2015, 14, 162–173. [Google Scholar] [CrossRef]
- Remadevi, R.; Szallisi, A. Adlea (algrx-4975), an injectable capsaicin (trpv1 receptor agonist) formulation for longlasting pain relief. IDrugs 2008, 11, 120–132. [Google Scholar] [PubMed]
- Diamond, E.; Richards, P.; Miller, T. Algrx 4975 reduces pain of intermetatarsal neuroma: Preliminary results from a randomized, double-blind, placebo-controlled, phase ii multicenter clinical trial. J. Pain 2006, 7, S41. [Google Scholar] [CrossRef]
- Richards, P.; Vasko, G.; Stasko, I.; Lacko, M.; Hewson, G. Algrx 4975 reduces pain of acute lateral epicondylitis: Preliminary results from a randomized, double-blind, placebo-controlled, phase ii multicenter clinical trial. J. Pain 2006, 7, S3. [Google Scholar] [CrossRef]
- Cantillon, M.; Vause, E.; Sykes, D.; Russel, R.; Moon, A.; Hughes, S. Preliminary safety, tolerability and efficacy of algrx 4975 in osteoarthristis (oa) of the knee. J. Pain 2005, 6, S39. [Google Scholar]
- Silberberg, A.; Moeller-Bertram, T.; Wallace, M.S. A randomized, double-blind, crossover study to evaluate the depth response relationship of intradermal capsaicin-induced pain and hyperalgesia in healthy adult volunteers. Pain Med. 2015, 16, 745–752. [Google Scholar] [CrossRef] [PubMed]
- De Petrocellis, L.; Guida, F.; Moriello, A.S.; De Chiaro, M.; Piscitelli, F.; de Novellis, V.; Maione, S.; Di Marzo, V. N-palmitoyl-vanillamide (palvanil) is a non-pungent analogue of capsaicin with stronger desensitizing capability against the trpv1 receptor and anti-hyperalgesic activity. Pharmacol. Res. 2011, 63, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Luongo, L.; Costa, B.; D'Agostino, B.; Guida, F.; Comelli, F.; Gatta, L.; Matteis, M.; Sullo, N.; De Petrocellis, L.; de Novellis, V.; et al. Palvanil, a non-pungent capsaicin analogue, inhibits inflammatory and neuropathic pain with little effects on bronchopulmonary function and body temperature. Pharmacol. Res. 2012, 66, 243–250. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).